Bioactive Phytochemicals from Mulberry: Potential Anti-Inflammatory Effects in Lipopolysaccharide-Stimulated RAW 264.7 Macrophages
Abstract
:1. Introduction
2. Results
2.1. Isolation and Identification of the Compounds
2.2. Effects of Compounds 1–22 on Nitric Oxide (NO) Production
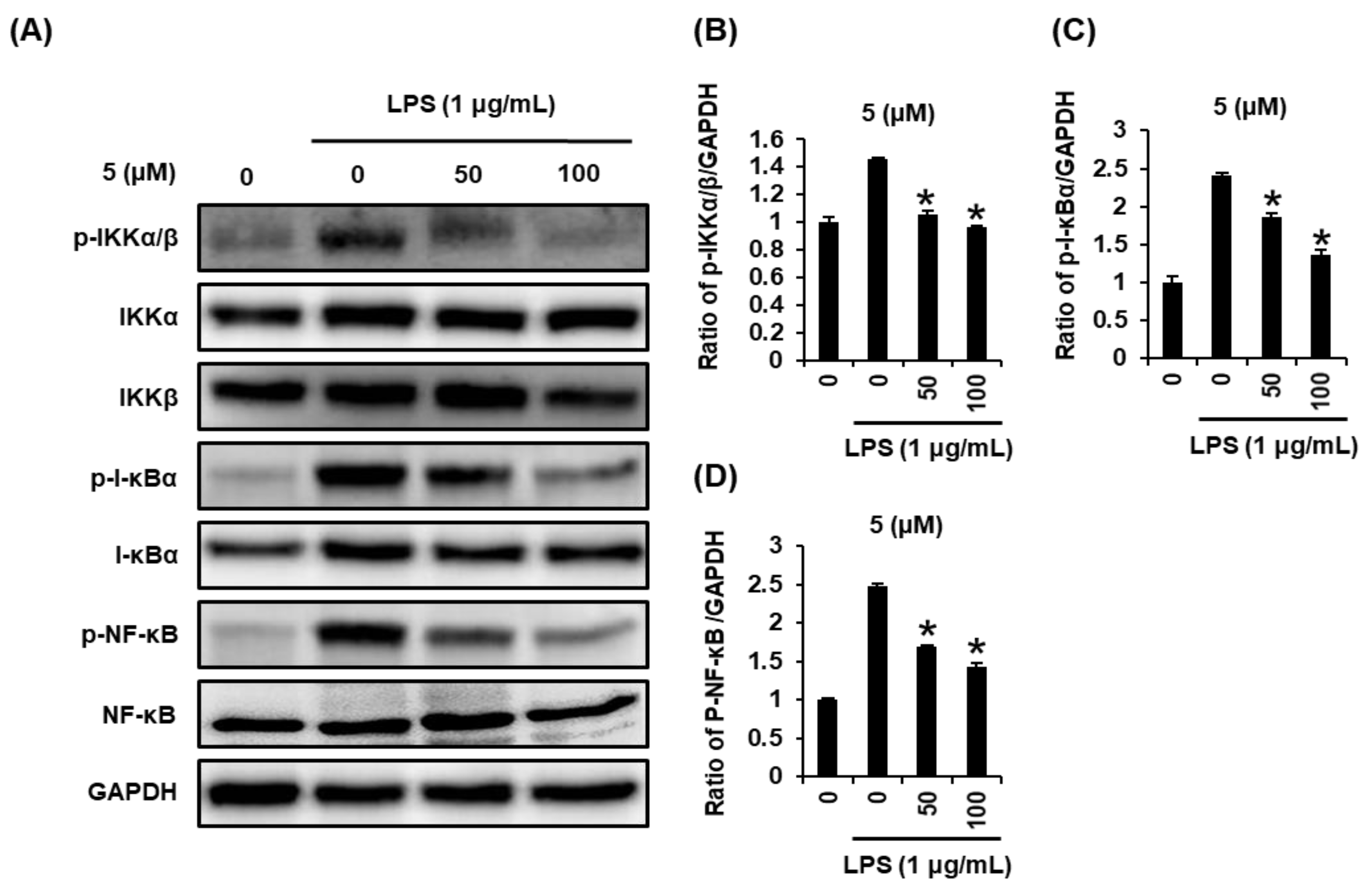
2.3. Effects of Compound 5 on the LPS-Induced Expression of IKKα/β, I-κBα, and NF-κB in RAW 264.7 Mouse Macrophages
2.4. Effects of Compound 5 on the LPS-Induced Expression of iNOS and COX-2 in RAW 264.7 Mouse Macrophages
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Plant Material
4.3. Extraction and Isolation
4.4. RAW 264.7 Cells Culture
4.5. Measurement of Viability of RAW 264.7 Cells
4.6. Measurement of NO Produced by RAW 264.7 Cells
4.7. Western Blot Analysis
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bhatarrai, G.; Choi, J.-W.; Seong, S.H.; Nam, T.-J.; Jung, H.A.; Choi, J.S. Anti-inflammatory, Anti-glycation, Anti-tyrosinase and CDK4 Inhibitory Activities of Alaternin (=7-Hydroxyemodin). Nat. Prod. Sci. 2021, 27, 28–35. [Google Scholar] [CrossRef]
- Rho, T.; Jeong, H.W.; Hong, Y.D.; Yoon, K.; Cho, J.Y.; Yoon, K.D. Identification of a novel triterpene saponin from Panax ginseng seeds, pseudoginsenoside RT8, and its antiinflammatory activity. J. Ginseng Res. 2020, 44, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-S.; Jin, H.-G.; Lee, H.; Lee, D.-S.; Woo, E.-R. Phytochemical Constituents of the Root Bark from Morus alba and Their Il-6 Inhibitory Activity. Nat. Prod. Sci. 2019, 25, 268–274. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Ma, Z.F.; Luo, X.; Li, X. Effects of mulberry fruit (Morus alba L.) consumption on health outcomes: A mini-review. Antioxidants 2018, 7, 69. [Google Scholar] [CrossRef] [Green Version]
- Butkhup, L.; Samappito, W.; Samappito, S. Phenolic composition and antioxidant activity of white mulberry (Morus alba L.) fruits. Int. J. Food Sci. Technol. 2013, 48, 934–940. [Google Scholar] [CrossRef]
- Gungor, N.; Sengul, M. Antioxidant activity, total phenolic content and selected physicochemical properties of white mulberry (Morus alba L.) fruits. Int. J. Food Prop. 2008, 11, 44–52. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.S.; Lim, S.H.; Lee, S.R.; Choi, C.-I.; Kim, K.H. Antioxidant and Anti-Inflammatory Effects of White Mulberry (Morus alba L.) Fruits on Lipopolysaccharide-Stimulated RAW 264.7 Macrophages. Molecules 2021, 26, 920. [Google Scholar] [CrossRef]
- Razavi, R.; Molaei, R.; Moradi, M.; Tajik, H.; Ezati, P.; Yordshahi, A.S. Biosynthesis of metallic nanoparticles using mulberry fruit (Morus alba L.) extract for the preparation of antimicrobial nanocellulose film. Appl. Nanosci. 2020, 10, 465–476. [Google Scholar] [CrossRef]
- Da Villa, G.; Ianiro, G.; Mangiola, F.; Del Toma, E.; Vitale, A.; Gasbarrini, A.; Gasbarrini, G. White mulberry supplementation as adjuvant treatment of obesity. J. Biol. Regul. Homeost. Agents 2014, 28, 141–145. [Google Scholar] [CrossRef]
- Mahboubi, M. Morus alba (mulberry), a natural potent compound in management of obesity. Pharmacol. Res. 2019, 146, 104341. [Google Scholar] [CrossRef]
- Lee, D.; Yu, J.S.; Lee, S.R.; Hwang, G.S.; Kang, K.S.; Park, J.G.; Kim, H.Y.; Kim, K.H.; Yamabe, N. Beneficial effects of bioactive compounds in mulberry fruits against cisplatin-induced nephrotoxicity. Int. J. Mol. Sci. 2018, 19, 1117. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.S.; Lee, D.; Lee, S.R.; Lee, J.W.; Choi, C.-I.; Jang, T.S.; Kang, K.S.; Kim, K.H. Chemical characterization of cytotoxic indole acetic acid derivative from mulberry fruit (Morus alba L.) against human cervical cancer. Bioorg. Chem. 2018, 76, 28–36. [Google Scholar] [CrossRef]
- Lee, S.R.; Park, J.Y.; Yu, J.S.; Lee, S.O.; Ryu, J.-Y.; Choi, S.-Z.; Kang, K.S.; Yamabe, N.; Kim, K.H. Odisolane, a novel oxolane derivative, and antiangiogenic constituents from the fruits of mulberry (Morus alba L.). J. Agric. Food Chem. 2016, 64, 3804–3809. [Google Scholar] [CrossRef]
- Song, J.H.; Lee, D.; Lee, S.R.; Yu, J.S.; Jang, T.S.; Nam, J.W.; Kim, K.H.; Kang, K.S. Identification of bioactive heterocyclic compounds from mulberry and their protective effect against streptozotocin-induced apoptosis in INS-1 cells. Mol. Med. Rep. 2018, 17, 5982–5987. [Google Scholar] [CrossRef] [Green Version]
- Kuang, H.X.; Yang, B.Y.; Xia, Y.G.; Feng, W.S. Chemical constituents from the flower of Datura metel L. Arch. Pharm. Res. 2008, 31, 1094–1097. [Google Scholar] [CrossRef]
- Savidge, R.A.; Forster, H. Coniferyl alcohol metabolism in conifers-II. Coniferyl alcohol and dihydroconiferyl alcohol biosynthesis. Phytochemistry 2001, 57, 1095–1103. [Google Scholar] [CrossRef]
- Li, L.Y.; Seeram, N.P. Further Investigation into Maple Syrup Yields 3 New Lignans, a New Phenylpropanoid, and 26 Other Phytochemicals. J. Agric. Food Chem. 2011, 59, 7708–7716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delauney, A.J.; Verma, D.P.S. Proline Biosynthesis and Osmoregulation in Plants. Plant J. 1993, 4, 215–223. [Google Scholar] [CrossRef]
- Adamczeski, M.; Reed, A.R.; Crews, P. New and Known Diketopiperazines from the Caribbean Sponge, Calyx cf. Podatypa. J. Nat. Prod. 1995, 58, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, M.I.; Naheed, N.; Abbaskhan, A.; Musharraf, S.G.; Siddiqui, H.; Atta, U.R. Phenolic and other constituents of fresh water fern Salvinia molesta. Phytochemistry 2008, 69, 1018–1023. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Kim, M.C.; Um, J.Y.; Hong, S.H. The beneficial effect of vanillic acid on ulcerative colitis. Molecules 2010, 15, 7208–7217. [Google Scholar] [CrossRef]
- Giovannini, C.; Straface, E.; Modesti, D.; Coni, E.; Cantafora, A.; De Vincenzi, M.; Malorni, W.; Masella, R. Tyrosol, the major olive oil biophenol, protects against oxidized-LDL-induced injury in Caco-2 cells. J. Nutr. 1999, 129, 1269–1277. [Google Scholar] [CrossRef] [Green Version]
- Le, H.T.; Ha Do, T.; Minh, C.T.; Kim, T.H.; Van Kiem, P.; Thuan, N.D.; Na, M. Constituents from the stem barks of Canarium bengalense with cytoprotective activity against hydrogen peroxide-induced hepatotoxicity. Arch. Pharm. Res. 2012, 35, 87–92. [Google Scholar] [CrossRef]
- Ul’chenko, N.; Gusakova, S.; Glushenkova, A. Oxygenated acids of the lipids of Onopordum acanthium seeds. Chem. Nat. Compd. 1993, 29, 447–450. [Google Scholar] [CrossRef]
- Chaurasia, N.; Wichtl, M. Sterols and steryl glycosides from Urtica dioica. J. Nat. Prod. 1987, 50, 881–885. [Google Scholar] [CrossRef]
- Zhang, X.; Julien-David, D.; Miesch, M.; Geoffroy, P.; Raul, F.; Roussi, S.; Aoudé-Werner, D.; Marchioni, E. Identification and quantitative analysis of β-sitosterol oxides in vegetable oils by capillary gas chromatography–mass spectrometry. Steroids 2005, 70, 896–906. [Google Scholar] [CrossRef]
- Rodriguez, J.; Nunez, L.; Peixinho, S.; Jimenez, C. Isolation and synthesis of the first natural 6-hydroximino 4-en-3-one- steroids from the sponges Cinachyrella spp. Tetrahedron Lett. 1997, 38, 1833–1836. [Google Scholar] [CrossRef]
- Zhao, C.C.; Shao, J.H.; Li, X.; Xu, J.; Zhang, P. Antimicrobial constituents from fruits of Ailanthus altissima SWINGLE. Arch. Pharm. Res. 2005, 28, 1147–1151. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Akihisa, T.; Yasukawa, K.; Takido, M.; Tamura, T. Structures of five hydroxylated sterols from the seeds of Trichosanthes kirilowii MAXIM. Chem. Pharm. Bull. 1995, 43, 1813–1817. [Google Scholar] [CrossRef] [Green Version]
- Pettit, G.R.; Numata, A.; Cragg, G.M.; Herald, D.L.; Takada, T.; Iwamoto, C.; Riesen, R.; Schmidt, J.M.; Doubek, D.L.; Goswami, A. Isolation and structures of schleicherastatins 1–7 and schleicheols 1 and 2 from the teak forest medicinal tree Schleichera oleosa. J. Nat. Prod. 2000, 63, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Lei, X.; Li, J.; Liu, B.; Zhang, N.; Liu, H. Separation and identification of four new compounds with antibacterial activity from Portulaca oleracea L. Molecules 2015, 20, 16375–16387. [Google Scholar] [CrossRef] [Green Version]
- Tsukamoto, S.; Tatsuno, M.; van Soest, R.W.; Yokosawa, H.; Ohta, T. New Polyhydroxy Sterols: Proteasome Inhibitors from a Marine Sponge Acanthodendrilla sp. J. Nat. Prod. 2003, 66, 1181–1185. [Google Scholar] [CrossRef]
- Zhang, Z.-Y.; Ma, N.; Tao, L.-J.; Gong, X.-Y.; Ye, W.-C.; Wang, L. Linear peptides containing D-leucine with neuroprotective activities from the Leech Whitmania pigra Whitman. J. Nat. Prod. 2019, 82, 2349–2353. [Google Scholar] [CrossRef]
- Lim, H.H.; Lee, S.O.; Kim, S.Y.; Yang, S.J.; Lim, Y. Anti-inflammatory and antiobesity effects of mulberry leaf and fruit extract on high fat diet-induced obesity. Exp. Biol. Med. 2013, 238, 1160–1169. [Google Scholar] [CrossRef]
- Liu, C.-J.; Lin, J.-Y. Anti-inflammatory and anti-apoptotic effects of strawberry and mulberry fruit polysaccharides on lipopolysaccharide-stimulated macrophages through modulating pro-/anti-inflammatory cytokines secretion and Bcl-2/Bak protein ratio. Food Chem. Toxicol. 2012, 50, 3032–3039. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Lee, M.-S.; Choi, A.-J.; Kim, C.-T.; Kim, Y. Anti-inflammatory effects of high hydrostatic pressure extract of mulberry (Morus alba) fruit on LPS-stimulated RAW264. 7 cells. Molecules 2019, 24, 1425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qian, Z.; Wu, Z.; Huang, L.; Qiu, H.; Wang, L.; Li, L.; Yao, L.; Kang, K.; Qu, J.; Wu, Y. Mulberry fruit prevents LPS-induced NF-κB/pERK/MAPK signals in macrophages and suppresses acute colitis and colorectal tumorigenesis in mice. Sci. Rep. 2015, 5, 17348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, H.; Ku, S.-K.; Choi, H.; Bae, J.-S. Three diketopiperazines from marine-derived bacteria inhibit LPS-induced endothelial inflammatory responses. Bioorg. Med. Chem. Lett. 2016, 26, 1873–1876. [Google Scholar] [CrossRef]
- Tweedie, D.; Luo, W.; Short, R.G.; Brossi, A.; Holloway, H.W.; Li, Y.; Yu, Q.-s.; Greig, N.H. A cellular model of inflammation for identifying TNF-α synthesis inhibitors. J. Neurosci. Methods 2009, 183, 182–187. [Google Scholar] [CrossRef] [Green Version]
- Pinsky, D.J.; Cai, B.; Yang, X.; Rodriguez, C.; Sciacca, R.R.; Cannon, P.J. The lethal effects of cytokine-induced nitric oxide on cardiac myocytes are blocked by nitric oxide synthase antagonism or transforming growth factor beta. J. Clin. Investig. 1995, 95, 677–685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhaskaran, N.; Shukla, S.; Srivastava, J.K.; Gupta, S. Chamomile: An anti-inflammatory agent inhibits inducible nitric oxide synthase expression by blocking RelA/p65 activity. Int. J. Mol. Med. 2010, 26, 935–940. [Google Scholar] [CrossRef] [Green Version]
- Israël, A. The IKK complex, a central regulator of NF-κB activation. Cold Spring Harb. Perspect. Biol. 2010, 2, a000158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oeckinghaus, A.; Ghosh, S. The NF-κB family of transcription factors and its regulation. Cold Spring Harb. Perspect. Biol. 2009, 1, a000034. [Google Scholar] [CrossRef] [PubMed]
- Cuzzocrea, S.; Salvemini, D. Molecular mechanisms involved in the reciprocal regulation of cyclooxygenase and nitric oxide synthase enzymes. Kidney Int. 2007, 71, 290–297. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.; Lee, D.; Ryoo, R.; Kim, J.-C.; Park, H.B.; Kang, K.S.; Kim, K.H. Calvatianone, a sterol possessing a 6/5/6/5-fused ring system with a contracted tetrahydrofuran b-ring, from the fruiting bodies of Calvatia nipponica. J. Nat. Prod. 2020, 83, 2737–2742. [Google Scholar] [CrossRef]
- Lee, S.R.; Kang, H.; Yoo, M.J.; Yu, J.S.; Lee, S.; Yi, S.A.; Beemelmanns, C.; Lee, J.; Kim, K.H. Anti-adipogenic pregnane steroid from a Hydractinia-associated fungus, Cladosporium sphaerospermum SW67. Nat. Prod. Sci. 2020, 26, 230–235. [Google Scholar] [CrossRef]
- Lee, S.; Ryoo, R.; Choi, J.H.; Kim, J.-H.; Kim, S.-H.; Kim, K.H. Trichothecene and tremulane sesquiterpenes from a hallucinogenic mushroom Gymnopilus junonius and their cytotoxicity. Arch. Pharmacal Res. 2020, 43, 214–223. [Google Scholar] [CrossRef]
- Jo, M.S.; Lee, S.; Yu, J.S.; Baek, S.C.; Cho, Y.-C.; Kim, K.H. Megastigmane derivatives from the cladodes of Opuntia humifusa and their nitric oxide inhibitory activities in macrophages. J. Nat. Prod. 2020, 83, 684–692. [Google Scholar] [CrossRef]
- Yu, J.S.; Li, C.; Kwon, M.; Oh, T.; Lee, T.H.; Kim, D.H.; Ahn, J.S.; Ko, S.-K.; Kim, C.S.; Cao, S. Herqueilenone A, a unique rearranged benzoquinone-chromanone from the hawaiian volcanic soil-associated fungal strain Penicillium herquei FT729. Bioorg. Chem. 2020, 105, 104397. [Google Scholar] [CrossRef]
- Ha, J.W.; Kim, J.; Kim, H.; Jang, W.; Kim, K.H. Mushrooms: An Important Source of Natural Bioactive Compounds. Nat. Prod. Sci. 2020, 26, 118–131. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, D.; Lee, S.R.; Kang, K.S.; Kim, K.H. Bioactive Phytochemicals from Mulberry: Potential Anti-Inflammatory Effects in Lipopolysaccharide-Stimulated RAW 264.7 Macrophages. Int. J. Mol. Sci. 2021, 22, 8120. https://doi.org/10.3390/ijms22158120
Lee D, Lee SR, Kang KS, Kim KH. Bioactive Phytochemicals from Mulberry: Potential Anti-Inflammatory Effects in Lipopolysaccharide-Stimulated RAW 264.7 Macrophages. International Journal of Molecular Sciences. 2021; 22(15):8120. https://doi.org/10.3390/ijms22158120
Chicago/Turabian StyleLee, Dahae, Seoung Rak Lee, Ki Sung Kang, and Ki Hyun Kim. 2021. "Bioactive Phytochemicals from Mulberry: Potential Anti-Inflammatory Effects in Lipopolysaccharide-Stimulated RAW 264.7 Macrophages" International Journal of Molecular Sciences 22, no. 15: 8120. https://doi.org/10.3390/ijms22158120









