The In Vitro Anti-Pseudomonal Activity of Cu2+, Strawberry Furanone, Gentamicin, and Lytic Phages Alone and in Combination: Pros and Cons
Abstract
:1. Introduction
2. Results
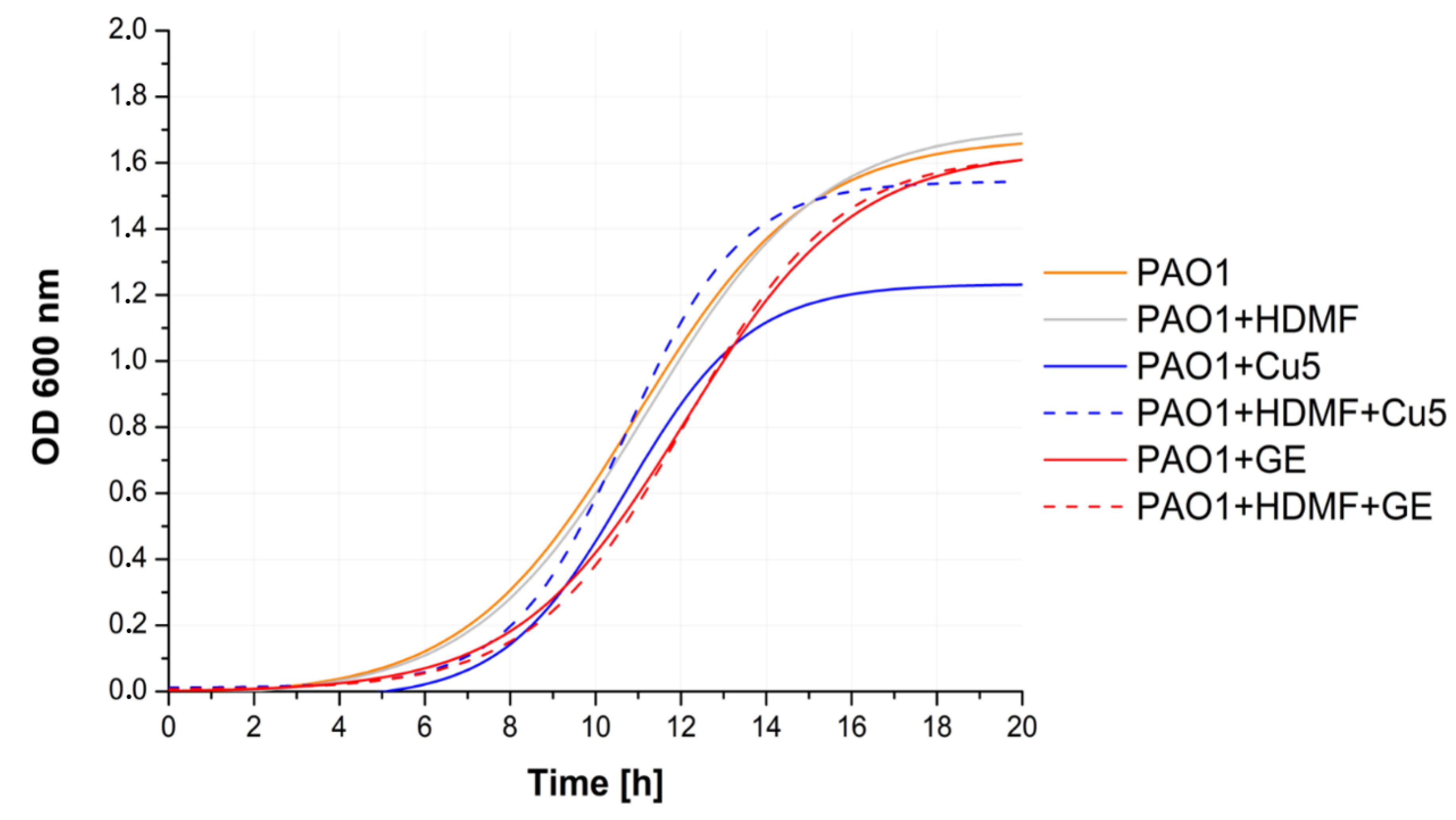
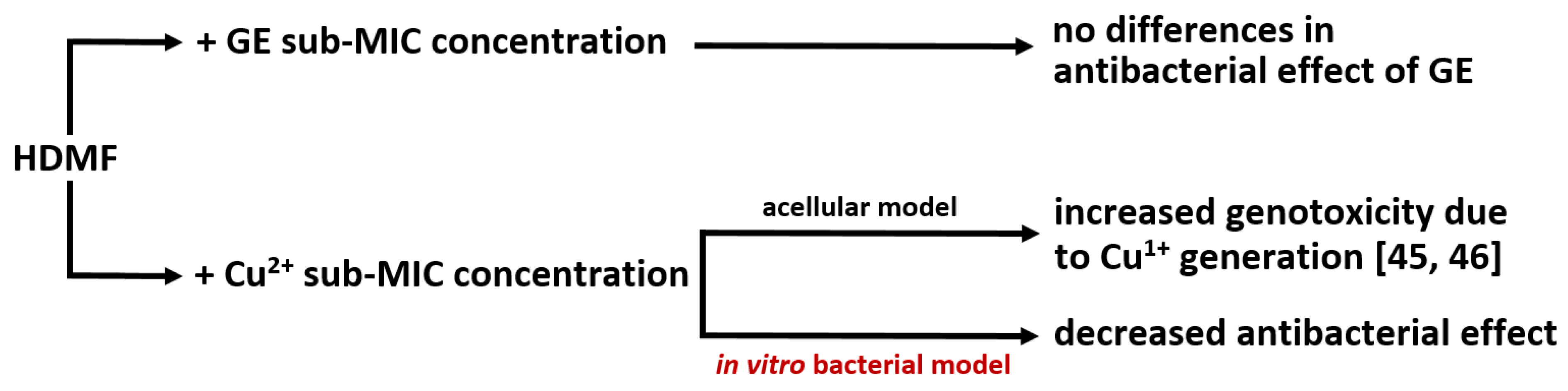
2.1. Cu2+ in Combination with GE Show Additive Antibacterial Effect, Whereas HDMF Neutralizes the Copper-Induced Virulence and Toxicity
2.2. Cu2+ and HDMF Do Not Enhance the Drug Resistance Emergence when Applied for 20 h
2.3. Cu2+ and GE Reduce the Effective Propagation of Lytic Phages Whilst HDMF Is Neutral
2.4. Cu2+ Reduce the Number of Infective Phage Particles Whilst HDMF and GE Are Neutral
2.5. Cu2+ Irreversibly Inactivate Phage Infectivity but Does Not Disturb the Virion Morphology
3. Discussion
4. Materials and Methods
4.1. Bacterial Strain and Phages
4.2. Antibacterial Activity Assays of Tested Chemicals
4.3. Anti-Virulent Activity of Tested Agents
- RVF—relative virulence factor production per cell unit;
- qFV—quantification of virulence factor.
4.4. Growth of P. aeruginosa PAO1 in the Presence of Tested Agents Alone or with Phages
4.5. Antibiotic Sensitivity Patterns
4.6. Phage Propagation in the Presence of Different Agents
4.7. Phage Particles Inactivation by Different Agents
4.8. Copper Ions Impact on Phage Particles Morphology
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HDMF | 4-hydroxy-2,5-dimethyl-3(2H)-furanone |
| GE | Gentamicin |
| Cu | Copper (II) nitrate hemi (pentahydrate) |
| CFU | Colony Forming Units |
| PFU | Plaque Forming Units |
| MIC | Minimum Inhibitory Concentration |
| MOI | Multiplicity of Infection |
References
- Rice, L.B. Federal funding for the study of antimicrobial resistance in nosocomial pathogens: No ESKAPE. J. Infect. Dis. 2008, 197, 1079–1081. [Google Scholar] [CrossRef]
- Mulani, M.S.; Kamble, E.E.; Kumkar, S.N.; Tawre, M.S.; Pardesi, K.R. Emerging strategies to combat ESKAPE pathogens in the era of antimicrobial resistance: A review. Front. Microbiol. 2019, 10, 539. [Google Scholar] [CrossRef] [PubMed]
- Guła, G.; Dorotkiewicz-Jach, A.; Korzekwa, K.; Valvano, M.A.; Drulis-Kawa, Z. Complex signaling networks controlling dynamic molecular changes in Pseudomonas aeruginosa biofilm. Curr. Med. Chem. 2019, 26, 1979–1993. [Google Scholar] [CrossRef]
- Azam, M.W.; Khan, A.U. Updates on the pathogenicity status of Pseudomonas aeruginosa. Drug Discov. Today 2019, 24, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Bulitta, J.B.; Ly, N.S.; Landersdorfer, C.B.; Wanigaratne, N.A.; Velkov, T.; Yadav, R.; Oliver, A.; Martin, L.; Shin, B.S.; Forrest, A.; et al. Two mechanisms of killing of Pseudomonas aeruginosa by tobramycin assessed at multiple inocula via mechanism-based modeling. Antimicrob. Agents Chemother. 2015, 59, 2315–2327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suwalsky, M.; Ungerer, B.; Quevedo, L.; Aguilar, F.; Sotomayor, C.P. Cu2+ ions interact with cell membranes. J. Inorg. Biochem. 1998, 70, 233–238. [Google Scholar] [CrossRef]
- Dorotkiewicz-Jach, A.; Augustyniak, D.; Olszak, T.; Drulis-Kawa, Z. Modern therapeutic approaches against Pseudomonas aeruginosa infections. Curr. Med. Chem. 2015, 22, 1642–1664. [Google Scholar] [CrossRef]
- Drulis-Kawa, Z.; Majkowska-Skrobek, G.; Maciejewska, B.; Delattre, A.-S.; Lavigne, R. Learning from bacteriophages-advantages and limitations of phage and phage-encoded protein applications. Curr. Protein Pept. Sci. 2013, 13, 699–722. [Google Scholar] [CrossRef] [Green Version]
- Maciejewska, B.; Olszak, T.; Drulis-Kawa, Z. Applications of bacteriophages versus phage enzymes to combat and cure bacterial infections: An ambitious and also a realistic application? Appl. Microbiol. Biotechnol. 2018, 102, 2563–2581. [Google Scholar] [CrossRef] [Green Version]
- Tagliaferri, T.L.; Jansen, M.; Horz, H.P. Fighting pathogenic bacteria on two fronts: Phages and antibiotics as combined strategy. Front. Cell. Infect. Microbiol. 2019, 9, 22. [Google Scholar] [CrossRef]
- Torres-Barceló, C.; Gurney, J.; Gougat-Barberá, C.; Vasse, M.; Hochberg, M.E. Transient negative effects of antibiotics on phages do not jeopardise the advantages of combination therapies. FEMS Microbiol. Ecol. 2018, 94, fiy107. [Google Scholar] [CrossRef]
- Chan, B.K.; Sistrom, M.; Wertz, J.E.; Kortright, K.E.; Narayan, D.; Turner, P.E. Phage selection restores antibiotic sensitivity in MDR Pseudomonas aeruginosa. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Hentzer, M.; Wu, H.; Andersen, J.B.; Riedel, K.; Rasmussen, T.B.; Bagge, N.; Kumar, N.; Schembri, M.A.; Song, Z.; Kristoffersen, P.; et al. Attenuation of Pseudomonas aeruginosa virulence by quorum sensing inhibitors. EMBO J. 2003, 22, 3803–3815. [Google Scholar] [CrossRef]
- Rezzoagli, C.; Archetti, M.; Mignot, I.; Baumgartner, M.; Kümmerli, R. Combining antibiotics with antivirulence compounds can have synergistic effects and reverse selection for antibiotic resistance in Pseudomonas aeruginosa. PLoS Biol. 2020, 18, e3000805. [Google Scholar] [CrossRef]
- Proctor, C.R.; McCarron, P.A.; Ternan, N.G. Furanone quorum-sensing inhibitors with potential as novel therapeutics against Pseudomonas aeruginosa. J. Med. Microbiol. 2020, 69, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.C.; Zhang, C.; Moon, S.; Oh, Y.S. Inhibitory effects of 4-hydroxy-2,5-dimethyl-3(2H)-furanone (HDMF) on acyl-homoserine lactone-mediated virulence factor production and biofilm formation in Pseudomonas aeruginosa PAO1. J. Microbiol. 2014, 52, 734–742. [Google Scholar] [CrossRef]
- Vincent, M.; Hartemann, P.; Engels-Deutsch, M. Antimicrobial applications of copper. Int. J. Hyg. Environ. Health 2016, 219, 585–591. [Google Scholar] [CrossRef]
- Vincent, M.; Duval, R.E.; Hartemann, P.; Engels-Deutsch, M. Contact killing and antimicrobial properties of copper. J. Appl. Microbiol. 2018, 124, 1032–1046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hobman, J.L.; Crossman, L.C. Bacterial antimicrobial metal ion resistance. J. Med. Microbiol. 2015, 64, 471–497. [Google Scholar] [CrossRef]
- Pietsch, F.; O’Neill, A.J.; Ivask, A.; Jenssen, H.; Inkinen, J.; Kahru, A.; Ahonen, M.; Schreiber, F. Selection of resistance by antimicrobial coatings in the healthcare setting. J. Hosp. Infect. 2020, 106, 115–125. [Google Scholar] [CrossRef]
- Rader, K.J.; Carbonaro, R.F.; van Hullebusch, E.D.; Baken, S.; Delbeke, K. The fate of copper added to surface water: Field, laboratory, and modeling studies. Environ. Toxicol. Chem. 2019, 38, 1386–1399. [Google Scholar] [CrossRef] [Green Version]
- Grass, G.; Rensing, C.; Solioz, M. Metallic copper as an antimicrobial surface. Appl. Environ. Microbiol. 2011, 77, 1541–1547. [Google Scholar] [CrossRef] [Green Version]
- Sagripanti, J.L.; Goering, P.L.; Lamanna, A. Interaction of copper with DNA and antagonism by other metals. Toxicol. Appl. Pharmacol. 1991, 110, 477–485. [Google Scholar] [CrossRef]
- Reina, G.; Peng, S.; Jacquemin, L.; Andrade, A.F.; Bianco, A. Hard nanomaterials in time of viral pandemics. ACS Nano 2020, 14, 9364–9388. [Google Scholar] [CrossRef] [PubMed]
- Moradali, M.F.; Ghods, S.; Rehm, B.H.A. Pseudomonas aeruginosa lifestyle: A paradigm for adaptation, survival, and persistence. Front. Cell. Infect. Microbiol. 2017, 7, 39. [Google Scholar] [CrossRef] [Green Version]
- Yoder, M.F.; Kisaalita, W.S. Iron specificity of a biosensor based on fluorescent pyoverdin immobilized in sol-gel glass. J. Biol. Eng. 2011, 5, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Poole, K. At the Nexus of Antibiotics and Metals: The impact of Cu and Zn on antibiotic activity and resistance. Trends Microbiol. 2017, 25, 820–832. [Google Scholar] [CrossRef] [PubMed]
- Teitzel, G.M.; Geddie, A.; De Long, S.K.; Kirisits, M.J.; Whiteley, M.; Parsek, M.R. Survival and growth in the presence of elevated copper: Transcriptional profiling of copper-stressed Pseudomonas aeruginosa. J. Bacteriol. 2006, 188, 7242–7256. [Google Scholar] [CrossRef] [Green Version]
- Thaden, J.T.; Lory, S.; Gardner, T.S. Quorum-sensing regulation of a copper toxicity system in Pseudomonas aeruginosa. J. Bacteriol. 2010, 192, 2557–2568. [Google Scholar] [CrossRef] [Green Version]
- Novoa-Aponte, L.; Ramírez, D.; Argüello, J.M. The interplay of the metallosensor CueR with two distinct CopZ chaperones defines copper homeostasis in Pseudomonas aeruginosa. J. Biol. Chem. 2019, 294, 4934–4945. [Google Scholar] [CrossRef] [Green Version]
- Wretlind, B.; Pavlovskis, O.R. Pseudomonas aeruginosa elastase and its role in pseudomonas infections. Rev. Infect. Dis. 1983, 5 (Suppl. 5), 998–1004. [Google Scholar] [CrossRef]
- Jayaseelan, S.; Ramaswamy, D.; Dharmaraj, S. Pyocyanin: Production, applications, challenges and new insights. World J. Microbiol. Biotechnol. 2014, 30, 1159–1168. [Google Scholar] [CrossRef]
- Yan, S.; Wu, G. Can biofilm be reversed through Quorum Sensing in Pseudomonas aeruginosa? Front. Microbiol. 2019, 10, 1582. [Google Scholar] [CrossRef] [Green Version]
- Minandri, F.; Imperi, F.; Frangipani, E.; Bonchi, C.; Visaggio, D.; Facchini, M.; Pasquali, P.; Bragonzi, A.; Visca, P. Role of iron uptake systems in Pseudomonas aeruginosa virulence and airway infection. Infect. Immun. 2016, 84, 2324–2335. [Google Scholar] [CrossRef] [Green Version]
- Banin, E.; Vasil, M.L.; Greenberg, E.P. Iron and Pseudomonas aeruginosa biofilm formation. Proc. Natl. Acad. Sci. USA 2005, 102, 11076–11081. [Google Scholar] [CrossRef] [Green Version]
- Poppe, J.; Reichelt, J.; Blankenfeldt, W. Pseudomonas aeruginosa pyoverdine maturation enzyme PvdP has a noncanonical domain architecture and affords insight into a new subclass of tyrosinases. J. Biol. Chem. 2018, 293, 14926–14936. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Visca, P.; Imperi, F.; Lamont, I.L. Pyoverdine siderophores: From biogenesis to biosignificance. Trends Microbiol. 2007, 15, 22–30. [Google Scholar] [CrossRef]
- Ringel, M.T.; Brüser, T. The biosynthesis of pyoverdines. Microb. Cell 2018, 5, 424–437. [Google Scholar] [CrossRef] [PubMed]
- Sung, W.S.; Jung, H.J.; Park, K.; Kim, H.S.; Lee, I.S.; Lee, D.G. 2,5-dimethyl-4-hydroxy-3(2H)-furanone (DMHF); antimicrobial compound with cell cycle arrest in nosocomial pathogens. Life Sci. 2007, 80, 586–591. [Google Scholar] [CrossRef]
- Harrison, J.J.; Ceri, H.; Stremick, C.A.; Turner, R.J. Biofilm susceptibility to metal toxicity. Environ. Microbiol. 2004, 6, 1220–1227. [Google Scholar] [CrossRef] [PubMed]
- Harrison, J.J.; Turner, R.J.; Ceri, H. Persister cells, the biofilm matrix and tolerance to metal cations in biofilm and planktonic Pseudomonas aeruginosa. Environ. Microbiol. 2005, 7, 981–994. [Google Scholar] [CrossRef]
- Murakami, K.; Haneda, M.; Makino, T.; Yoshino, M. Prooxidant action of furanone compounds: Implication of reactive oxygen species in the metal-dependent strand breaks and the formation of 8-hydroxy-2′-deoxyguanosine in DNA. Food Chem. Toxicol. 2007, 45, 1258–1262. [Google Scholar] [CrossRef]
- Yamashita, N.; Murata, M.; Inoue, S.; Hiraku, Y.; Yoshinaga, T.; Kawanishi, S. Superoxide formation and DNA damage induced by a fragrant furanone in the presence of copper(II). Mutat. Res.-Fundam. Mol. Mech. Mutagen. 1998, 397, 191–201. [Google Scholar] [CrossRef]
- Kozłowski, H.; Kowalik-Jankowska, T.; Jezowska-Bojczuk, M. Chemical and biological aspects of Cu2+ interactions with peptides and aminoglycosides. Coord. Chem. Rev. 2005, 249, 2323–2334. [Google Scholar] [CrossRef]
- Kadurugamuwa, J.L.; Beveridge, T.J. Natural release of virulence factors in membrane vesicles by Pseudomonas aeruginosa and the effect of aminoglycoside antibiotics on their release. J. Antimicrob. Chemother. 1997, 40, 615–621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dalecki, A.G.; Crawford, C.L.; Wolschendorf, F. Copper and Antibiotics: Discovery, modes of action, and opportunities for medicinal applications. In Advances in Microbial Physiology; Academic Press: Cambridge, MA, USA, 2017; Volume 70, pp. 193–260. [Google Scholar] [CrossRef]
- Warnes, S.L.; Caves, V.; Keevil, C.W. Mechanism of copper surface toxicity in Escherichia coli O157:H7 and Salmonella involves immediate membrane depolarization followed by slower rate of DNA destruction which differs from that observed for Gram-positive bacteria. Environ. Microbiol. 2012, 14, 1730–1743. [Google Scholar] [CrossRef] [PubMed]
- Lesniak, W.; Harris, W.R.; Yudenfreund Kravitz, J.; Schacht, J.; Pecoraro, V.L. Solution chemistry of copper(II)-gentamicin complexes: Relevance to metal-related aminoglycoside toxicity. Inorg. Chem. 2003, 42, 1420–1429. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Feng, Z.; Yu, Z.; Chen, Y.; Li, P.; Yang, Z.; Li, S.; Jin, S. Preparation of chitosan-Cu2+/NH3 physical hydrogel and its properties. Int. J. Biol. Macromol. 2019, 133, 67–75. [Google Scholar] [CrossRef]
- Ashfaq, M.; Verma, N.; Khan, S. Highly effective Cu/Zn-carbon micro/nanofiber-polymer nanocomposite-based wound dressing biomaterial against the P. aeruginosa multi- and extensively drug-resistant strains. Mater. Sci. Eng. C 2017, 77, 630–641. [Google Scholar] [CrossRef] [PubMed]
- Ahire, J.J.; Hattingh, M.; Neveling, D.P.; Dicks, L.M.T. Copper-containing anti-biofilm nanofiber scaffolds as a wound dressing material. PLoS ONE 2016, 11, e0152755. [Google Scholar] [CrossRef] [Green Version]
- Singh, N.; Paknikar, K.M.; Rajwade, J. Gene expression is influenced due to ‘nano’ and ‘ionic’ copper in pre-formed Pseudomonas aeruginosa biofilms. Environ. Res. 2019, 175, 367–375. [Google Scholar] [CrossRef]
- Torres-Barceló, C.; Hochberg, M.E. Evolutionary rationale for phages as complements of antibiotics. Trends Microbiol. 2016, 24, 249–256. [Google Scholar] [CrossRef]
- Torres-Barceló, C.; Arias-Sánchez, F.I.; Vasse, M.; Ramsayer, J.; Kaltz, O.; Hochberg, M.E. A window of opportunity to control the bacterial pathogen Pseudomonas aeruginosa combining antibiotics and phages. PLoS ONE 2014, 9, e106628. [Google Scholar] [CrossRef] [Green Version]
- Chaudhry, W.N.; Concepcion-Acevedo, J.; Park, T.; Andleeb, S.; Bull, J.J.; Levin, B.R. Synergy and order effects of antibiotics and phages in killing Pseudomonas aeruginosa biofilms. PLoS ONE 2017, 12, e0168615. [Google Scholar] [CrossRef]
- Uchiyama, J.; Shigehisa, R.; Nasukawa, T.; Mizukami, K.; Takemura-Uchiyama, I.; Ujihara, T.; Murakami, H.; Imanishi, I.; Nishifuji, K.; Sakaguchi, M.; et al. Piperacillin and ceftazidime produce the strongest synergistic phage–antibiotic effect in Pseudomonas aeruginosa. Arch. Virol. 2018, 163, 1941–1948. [Google Scholar] [CrossRef] [PubMed]
- Grote, J.; Krysciak, D.; Streit, W.R. Phenotypic heterogeneity, a phenomenon that may explain why quorum sensing does not always result in truly homogenous cell behavior. Appl. Environ. Microbiol. 2015, 81, 5280–5289. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Dennehy, J.J. Differential bacteriophage mortality on exposure to copper. Appl. Environ. Microbiol. 2011, 77, 6878–6883. [Google Scholar] [CrossRef] [Green Version]
- Giachino, A.; Waldron, K.J. Copper tolerance in bacteria requires the activation of multiple accessory pathways. Mol. Microbiol. 2020, 114, 377–390. [Google Scholar] [CrossRef]
- Quintana, J.; Novoa-Aponte, L.; Argüello, J.M. Copper homeostasis networks in the bacterium Pseudomonas aeruginosa. J. Biol. Chem. 2017, 292, 15691–15704. [Google Scholar] [CrossRef] [Green Version]
- Ladomersky, E.; Petris, M.J. Copper tolerance and virulence in bacteria. Metallomics 2015, 7, 957–964. [Google Scholar] [CrossRef] [Green Version]
- Danis-Wlodarczyk, K.; Olszak, T.; Arabski, M.; Wasik, S.; Majkowska-Skrobek, G.; Augustyniak, D.; Gula, G.; Briers, Y.; Jang, H.B.; Vandenheuvel, D.; et al. Characterization of the newly isolated lytic bacteriophages KTN6 and KT28 and their efficacy against Pseudomonas aeruginosa biofilm. PLoS ONE 2015, 10, e0127603. [Google Scholar] [CrossRef]
- Danis-Wlodarczyk, K.; Vandenheuvel, D.; Jang, H.B.; Briers, Y.; Olszak, T.; Arabski, M.; Wasik, S.; Drabik, M.; Higgins, G.; Tyrrell, J.; et al. A proposed integrated approach for the preclinical evaluation of phage therapy in Pseudomonas infections. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [Green Version]
- Ceyssens, P.J.; Brabban, A.; Rogge, L.; Lewis, M.S.; Pickard, D.; Goulding, D.; Dougan, G.; Noben, J.P.; Kropinski, A.; Kutter, E.; et al. Molecular and physiological analysis of three Pseudomonas aeruginosa phages belonging to the “N4-like viruses”. Virology 2010, 405, 26–30. [Google Scholar] [CrossRef] [Green Version]
- Brennan-Krohn, T.; Kirby, J.E. When one drug is not enough: Context, methodology, and future prospects in antibacterial synergy testing. Clin. Lab. Med. 2019, 39, 345–358. [Google Scholar] [CrossRef]
- Das, T.; Manefield, M. Pyocyanin promotes extracellular DNA release in Pseudomonas aeruginosa. PLoS ONE 2012, 7, e0046718. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoegy, F.; Mislin, G.L.A.; Schalk, I.J. Pyoverdine and pyochelin measurements. Methods Mol. Biol. 2014, 1149, 293–301. [Google Scholar] [CrossRef]
- Rust, L.; Messing, C.R.; Iglewski, B.H. Elastase assays. Methods Enzymol. 1994, 235, 554–562. [Google Scholar] [CrossRef] [PubMed]








| Antibacterial/Anti-Virulent Agents | ∆log PFU/mL (Initial 107 PFU/mL) | ||
|---|---|---|---|
| Phage KT28 | Phage KTN4 | Phage LUZ19 | |
| control | +2 | +1 | +3 |
| HDMF 10 µM | +2 | +1 | +3 |
| GE 1 µg/mL | - | - | +1 |
| Cu 5 mM | +2 | +1 | +3 |
| Cu 10 mM | −2 | −5 | −5 |
| Antibacterial/Anti-Virulent Agents | PFU/mL Reduction of the Initial 107 [∆log] | |||
|---|---|---|---|---|
| Phage KT28 | Phage KTN4 | Phage LUZ19 | ||
| Neutral agents | HDMF 10 µM | - | - | - |
| GE 1 µg/mL | - | - | - | |
| HDMF 10 µM + GE 1 µg/mL | - | - | - | |
| Reducing agents | Cu 5 mM | - | −2 | −2 |
| HDMF 10 µM + Cu 5 mM | - | −2 | −2 | |
| Cu 5 mM + GE 1 µg/mL | - | −2 | −2 | |
| Cu 10 mM | −2 | −5 | −5 | |
| HDMF 10 µM + Cu 10 mM | −2 | −5 | −5 | |
| Cu 10 mM + GE 1 µg/mL | −2 | −5 | −5 | |
| Phage | Taxonomy (Family, Genus) | Genome Size [bp] | GenBank Accession Number | Recognized Bacterial Receptor | References |
|---|---|---|---|---|---|
| KT28 * | Myoviridae Pbunavirus | 66,381 | KP340287 | LPS | [62] |
| KTN4 * | Myoviridae Phikzvirus | 279,593 | KU521356 | T4P | [63] |
| LUZ19 ** | Autographiviridae, Phikmvvirus | 43,548 | NC_010326 | T4P | [64] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dorotkiewicz-Jach, A.; Markwitz, P.; Drulis-Kawa, Z. The In Vitro Anti-Pseudomonal Activity of Cu2+, Strawberry Furanone, Gentamicin, and Lytic Phages Alone and in Combination: Pros and Cons. Int. J. Mol. Sci. 2021, 22, 9830. https://doi.org/10.3390/ijms22189830
Dorotkiewicz-Jach A, Markwitz P, Drulis-Kawa Z. The In Vitro Anti-Pseudomonal Activity of Cu2+, Strawberry Furanone, Gentamicin, and Lytic Phages Alone and in Combination: Pros and Cons. International Journal of Molecular Sciences. 2021; 22(18):9830. https://doi.org/10.3390/ijms22189830
Chicago/Turabian StyleDorotkiewicz-Jach, Agata, Pawel Markwitz, and Zuzanna Drulis-Kawa. 2021. "The In Vitro Anti-Pseudomonal Activity of Cu2+, Strawberry Furanone, Gentamicin, and Lytic Phages Alone and in Combination: Pros and Cons" International Journal of Molecular Sciences 22, no. 18: 9830. https://doi.org/10.3390/ijms22189830






