The Intra- and Extra-Telomeric Role of TRF2 in the DNA Damage Response
Abstract
:1. Introduction
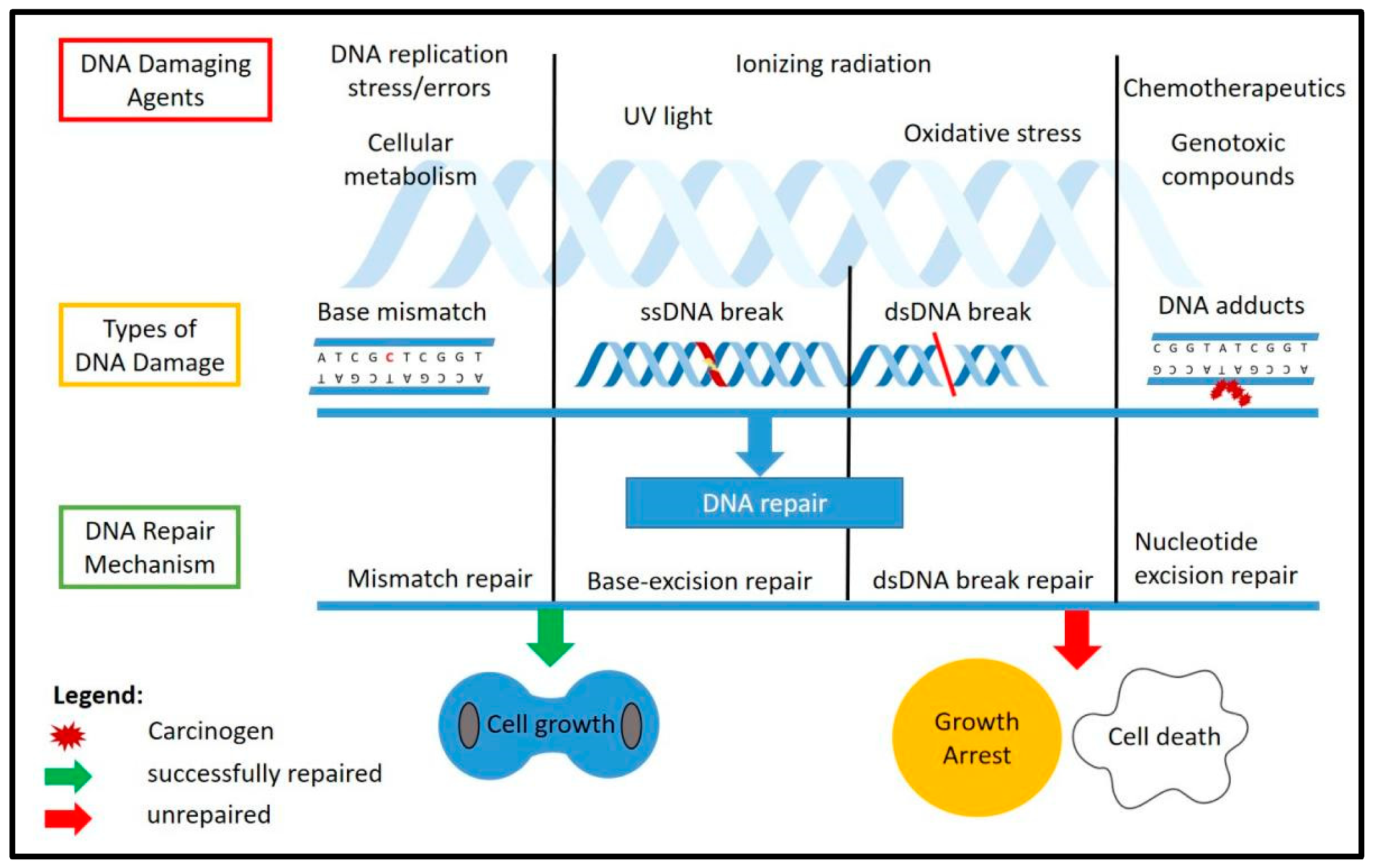
2. DNA Damage Response (DDR)
3. TRF2 Post-Translational Modification and Its Involvement with DDR
4. TRF2 Protects Telomeres by Blocking the Activation of DDR
5. TRF2 Inhibits the ATM Kinase
6. TRF2 Involved in Non-Homologous End Joining (NHEJ)
| Repair Mechanism | Gene | Known Function | Known Function | Location | Reference (s) |
|---|---|---|---|---|---|
| ATM Kinase | ATM | Initial sensing of double-strand breaks of DNA | TRF2 inhibits the activation (autophosphorylation) of ATM on S1981 at the telomeres | Telomeres and non-telomeric regions | [12,104,125] |
| γH2AX | Recruitment and activation of other DNA repair factors | TRF2 localized at the same site upon DSB induction | Non-telomeric regions | [104,126] | |
| Cell Cycle | pChk2 | Cell cycle regulation | Inhibited by TRF2 via ATM kinase inhibition | Telomeres and non-telomeric regions | [102,127,128] |
| MRE11 | Part of the Mre11-Rad50-Nbs1 (MRN) complex to facilitate DNA damage repair | Mediates the maintenance of t-loop and DNA repair at the telomeres with TRF2/RAP1 | Telomeres and non-telomeric regions | [129,130] | |
| Apollo (SNM1B) | Involved in the repair of DNA lesions | Localization at the telomeres is TRF2-dependent | Telomeres | [131,132] | |
| Direct interaction with endogenous TRF2 | [133] | ||||
| Rap1 | Regulation of telomere structure and integrity | Interacts with TRF2 to inhibit NHEJ | Telomeres | [3] | |
| Ku70/80 | Initiates and is required for c-NHEJ and suppresses alt-NHEJ | Interacts with TRF2 to inhibit c-NHEJ | Telomeres and non-telomeric regions | [119,134,135] | |
| DNA-PK | DNA double-strand break repair complex | Inhibited by TRF2 to prevent c-NHEJ | Telomeres and non-telomeric regions | [136] | |
| 53BP1 | Early response to DNA double-strand breaks | Localization at DSB induced by inhibition of TRF2 | Telomeres and non-telomeric regions | [53,120,137] | |
| BER | pol β | Base-excision repair | Physically bound to TRF2 at the NH2-terminal and COOH-terminal domains | Telomeres and non-telomeric regions | [138,139,140] |
| FEN1 | Base-excision repair | Physically bound to TRF2 at the NH2-terminal and COOH-terminal domains | Telomeres and non-telomeric regions | [139,140] | |
| APE1 | Base-excision repair | Indirectly stimulated by TRF2 | Telomeres | [136] | |
| Lig1 | Base-excision repair | Indirectly stimulated by TRF2 | Telomeres | [140] | |
| PARP1 | DNA nick sensor activated rapidly and transiently in response to DNA damage | Recruits TRF2 at DNA damage site | Non-telomeric regions | [92,97] | |
| Others | LAP1 | A lamina-associated protein that was recently found to be involved in DDR | Forms a complex with TRF2 upon DNA damage induction and binds at DNA damage site | Non-telomeric regions | [141] |
| RTEL1 | A helicase essential in DDR | Recruited by TRF2 at telomeres to promote t-loop unwinding | Telomeres and non-telomeric regions (not TRF2-dependent) | [142,143,144] |
7. TRF2 Interacts with Factors of Base Excision Repair (BER)
8. TRF2 Involvement in the DDR Outside of the Telomeres
9. Other Known Extra-Telomeric Functions of TRF2
10. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- De Lange, T. Shelterin: The protein complex that shapes and safeguards human telomeres. Genes Dev. 2005, 19, 2100–2110. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Yang, Y.; van Overbeek, M.; Donigian, J.R.; Baciu, P.; de Lange, T.; Lei, M. A Shared Docking Motif in TRF1 and TRF2 Used for Differential Recruitment of Telomeric Proteins. Science 2008, 319, 1092–1096. [Google Scholar] [CrossRef] [PubMed]
- Rai, R.; Chen, Y.; Lei, M.; Chang, S. TRF2-RAP1 is required to protect telomeres from engaging in homologous recombination-mediated deletions and fusions. Nat. Commun. 2016, 7, 10881. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Özlem Arat, N.; Griffith, J.D. Human Rap1 Interacts Directly with Telomeric DNA and Regulates TRF2 Localization at the Telomere. J. Biol. Chem. 2012, 287, 41583–41594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ye, J.Z.-S.; Donigian, J.R.; van Overbeek, M.; Loayza, D.; Luo, Y.; Krutchinsky, A.N.; Chait, B.T.; de Lange, T. TIN2 Binds TRF1 and TRF2 Simultaneously and Stabilizes the TRF2 Complex on Telomeres. J. Biol. Chem. 2004, 279, 47264–47271. [Google Scholar] [CrossRef] [Green Version]
- Yang, Q.; Zheng, Y.-L.; Harris, C.C. POT1 and TRF2 Cooperate to Maintain Telomeric Integrity. Mol. Cell. Biol. 2005, 25, 1070–1080. [Google Scholar] [CrossRef] [Green Version]
- Kendellen, M.F.; Barrientos, K.S.; Counter, C.M. POT1 Association with TRF2 Regulates Telomere Length. Mol. Cell. Biol. 2009, 29, 5611–5619. [Google Scholar] [CrossRef] [Green Version]
- Hu, C.; Rai, R.; Huang, C.; Broton, C.; Long, J.; Xu, Y.; Xue, J.; Lei, M.; Chang, S.; Chen, Y. Structural and functional analyses of the mammalian TIN2-TPP1-TRF2 telomeric complex. Cell Res. 2017, 27, 1485–1502. [Google Scholar] [CrossRef]
- Lim, C.J.; Zaug, A.J.; Kim, H.J.; Cech, T.R. Reconstitution of human shelterin complexes reveals unexpected stoichiometry and dual pathways to enhance telomerase processivity. Nat. Commun. 2017, 8, 1075. [Google Scholar] [CrossRef] [Green Version]
- Stansel, R.M.; de Lange, T.; Griffith, J.D. T-loop assembly in vitro involves binding of TRF2 near the 3’ telomeric overhang. EMBO J. 2001, 20, 5532–5540. [Google Scholar] [CrossRef] [Green Version]
- Timashev, L.A.; De Lange, T. Characterization of t-loop formation by TRF. Nucleus 2020, 11, 164–177. [Google Scholar] [CrossRef]
- Karlseder, J.; Hoke, K.; Mirzoeva, O.K.; Bakkenist, C.; Kastan, M.B.; Petrini, J.; De Lange, T. The Telomeric Protein TRF2 Binds the ATM Kinase and Can Inhibit the ATM-Dependent DNA Damage Response. PLoS Biol. 2004, 2, e240. [Google Scholar] [CrossRef]
- Wong, S.K.; Ima-Nirwana, S.; Chin, K.-Y. Can telomere length predict bone health? A review of current evidence. Bosn. J. Basic Med. Sci. 2020, 20, 423–429. [Google Scholar] [CrossRef]
- Muñoz, P.; Blanco, R.; Blasco, M.A. Role of the TRF2 Telomeric Protein in Cancer and Aging. Cell Cycle 2006, 5, 718–721. [Google Scholar] [CrossRef]
- Huda, N.; Abe, S.; Gu, L.; Mendonca, M.S.; Mohanty, S.; Gilley, D. Recruitment of TRF2 to laser-induced DNA damage sites. Free. Radic. Biol. Med. 2012, 53, 1192–1197. [Google Scholar] [CrossRef]
- Kong, X.; Cruz, G.M.S.; Trinh, S.L.; Zhu, X.-D.; Berns, M.W.; Yokomori, K. Biphasic recruitment of TRF2 to DNA damage sites promotes non-sister chromatid homologous recombination repair. J. Cell Sci. 2018, 131, 219311. [Google Scholar] [CrossRef] [Green Version]
- Rothkamm, K.; Krüger, I.; Thompson, L.H.; Löbrich, M. Pathways of DNA Double-Strand Break Repair during the Mammalian Cell Cycle. Mol. Cell. Biol. 2003, 23, 5706–5715. [Google Scholar] [CrossRef] [Green Version]
- Fagagna, F.d.A.d.; Reaper, P.M.; Clay-Farrace, L.; Fiegler, H.; Carr, P.; von Zglinicki, T.; Saretzki, G.; Carter, N.P.; Jackson, S.P. A DNA damage checkpoint response in telomere-initiated senescence. Nature 2003, 426, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, I.; Yoshida, Y.; Suda, M.; Minamino, T. DNA Damage Response and Metabolic Disease. Cell Metab. 2014, 20, 967–977. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turgeon, M.-O.; Perry, N.J.S.; Poulogiannis, G. DNA Damage, Repair, and Cancer Metabolism. Front. Oncol. 2018, 8, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sinha, R.P.; Häder, D.-P. UV-induced DNA damage and repair: A review. Photochem. Photobiol. Sci. 2002, 1, 225–236. [Google Scholar] [CrossRef]
- Rastogi, R.P.; Richa; Kumar, A.; Tyagi, M.B.; Sinha, R.P. Molecular Mechanisms of Ultraviolet Radiation-Induced DNA Damage and Repair. J. Nucleic Acids 2010, 2010, 1–32. [Google Scholar] [CrossRef] [Green Version]
- Sachs, R.K.; Chen, P.-L.; Hahnfeldt, P.J.; Hlatky, L.R. DNA damage caused by ionizing radiation. Math. Biosci. 1992, 112, 271–303. [Google Scholar] [CrossRef]
- Leadon, S.A. Repair of DNA damage produced by ionizing radiation: A minireview. Semin. Radiat. Oncol. 1996, 6, 295–305. [Google Scholar] [CrossRef]
- Awang, N.; Kismin, D.N.A.; Kamaludin, N.F.; Ghazali, A.R.b. Genotoxic Effects on buccal Cells of Workers Exposed to Fogging Sprays during Fogging Operation. Biomed. J. Sci. Tech. Res. 2017, 1, 1341–1345. [Google Scholar] [CrossRef] [Green Version]
- Sopian, N.; Jalaludin, J.; Abu Bakar, S.; Hamedon, T.; Latif, M. Exposure to Particulate PAHs on Potential Genotoxicity and Cancer Risk among School Children Living Near the Petrochemical Industry. Int. J. Environ. Res. Public Health 2021, 18, 2575. [Google Scholar] [CrossRef] [PubMed]
- Morrison, A.; Johnson, A.; Johnston, L.; Sugino, A. Pathway correcting DNA replication errors in Saccharomyces cerevisiae. EMBO J. 1993, 12, 1467–1473. [Google Scholar] [CrossRef]
- Brown, T.A. Mutation, Repair and Recombination. In Genomes, 2nd ed.; Wiley-Liss: Oxford, UK, 2002. [Google Scholar]
- Boyce Kylie, J.; Wang, Y.; Verma, S.; Shakya Viplendra, P.S.; Xue, C.; Idnurm, A.; Alspaugh, J.A. Mismatch Repair of DNA Replication Errors Contributes to Microevolution in the Pathogenic Fungus Cryptococcus neoformans. mBio 2017, 8, e00595-17. [Google Scholar] [CrossRef] [Green Version]
- Kunkel, T.A.; Erie, D.A. Eukaryotic Mismatch Repair in Relation to DNA Replication. Annu. Rev. Genet. 2015, 49, 291–313. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Yu, Y.; Hamrick, H.E.; Duerksen-Hughes, P.J. ATM, ATR and DNA-PK: Initiators of the cellular genotoxic stress responses. Carcinogenesis 2003, 24, 1571–1580. [Google Scholar] [CrossRef] [Green Version]
- Coates, P.J.; Lorimore, S.A.; Wright, E.G. Cell and tissue responses to genotoxic stress. J. Pathol. 2005, 205, 221–235. [Google Scholar] [CrossRef] [PubMed]
- Tsolou, A.; Nelson, G.; Trachana, V.; Chondrogianni, N.; Saretzki, G.; von Zglinicki, T.; Gonos, E.S. The 19S proteasome subunit Rpn7 stabilizes DNA damage foci upon genotoxic insult. IUBMB Life 2012, 64, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Caldecott, K.W. Single-strand break repair and genetic disease. Nat. Rev. Genet. 2008, 9, 619–631. [Google Scholar] [CrossRef] [PubMed]
- Anindya, R. Single-stranded DNA damage: Protecting the single-stranded DNA from chemical attack. DNA Repair 2020, 87, 102804. [Google Scholar] [CrossRef] [PubMed]
- Scully, R.; Panday, A.; Elango, R.; Willis, N.A. DNA double-strand break repair-pathway choice in somatic mammalian cells. Nat. Rev. Mol. Cell Biol. 2019, 20, 698–714. [Google Scholar] [CrossRef]
- Li, G.-M. Mechanisms and functions of DNA mismatch repair. Cell Res. 2008, 18, 85–98. [Google Scholar] [CrossRef] [Green Version]
- Krokan, H.E.; Bjørås, M. Base Excision Repair. Cold Spring Harbor Perspect. Biol. 2013, 5, a012583. [Google Scholar] [CrossRef] [PubMed]
- Spivak, G. Nucleotide excision repair in humans. DNA Repair 2015, 36, 13–18. [Google Scholar] [CrossRef] [Green Version]
- Wright, W.D.; Shah, S.S.; Heyer, W.-D. Homologous recombination and the repair of DNA double-strand breaks. J. Biol. Chem. 2018, 293, 10524–10535. [Google Scholar] [CrossRef] [Green Version]
- Chiruvella, K.K.; Liang, Z.; Wilson, T.E. Repair of Double-Strand Breaks by End Joining. Cold Spring Harb. Perspect. Biol. 2013, 5, a012757. [Google Scholar] [CrossRef]
- Rondeau, S.; Vacher, S.; De Koning, L.; Briaux, A.; Schnitzler, A.; Chemlali, W.; Callens, C.; Lidereau, R.; Bièche, I. ATM has a major role in the double-strand break repair pathway dysregulation in sporadic breast carcinomas and is an independent prognostic marker at both mRNA and protein levels. Br. J. Cancer 2015, 112, 1059–1066. [Google Scholar] [CrossRef] [PubMed]
- Niida, H.; Nakanishi, M. DNA damage checkpoints in mammals. Mutagenesis 2005, 21, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Nastasi, C.; Mannarino, L.; D’Incalci, M. DNA Damage Response and Immune Defense. Int. J. Mol. Sci. 2020, 21, 7504. [Google Scholar] [CrossRef] [PubMed]
- Andrs, M.; Korabecny, J.; Jun, D.; Hodny, Z.; Bartek, J.; Kuca, K. Phosphatidylinositol 3-Kinase (PI3K) and Phosphatidylinositol 3-Kinase-Related Kinase (PIKK) Inhibitors: Importance of the Morpholine Ring. J. Med. Chem. 2015, 58, 41–71. [Google Scholar] [CrossRef]
- Lovejoy, C.A.; Cortez, D. Common mechanisms of PIKK regulation. DNA Repair 2009, 8, 1004–1008. [Google Scholar] [CrossRef] [Green Version]
- Menolfi, D.; Zha, S. ATM, ATR and DNA-PKcs kinases—The lessons from the mouse models: Inhibition ≠ deletion. Cell Biosci. 2020, 10, 1–15. [Google Scholar] [CrossRef]
- Guo, F.; Li, J.; Du, W.; Zhang, S.; O’Connor, M.; Thomas, G.; Kozma, S.; Zingarelli, B.; Pang, Q.; Zheng, Y. mTOR regulates DNA damage response through NF-κB-mediated FANCD2 pathway in hematopoietic cells. Leukemia 2013, 27, 2040–2046. [Google Scholar] [CrossRef] [Green Version]
- Alao, J.-P.; Legon, L.; Rallis, C. Crosstalk between the mTOR and DNA Damage Response Pathways in Fission Yeast. Cells 2021, 10, 305. [Google Scholar] [CrossRef] [PubMed]
- Hiom, K. DNA Repair: How to PIKK a Partner. Curr. Biol. 2005, 15, R473–R475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klapper, W.; Qian, W.; Schulte, C.; Parwaresch, R. DNA damage transiently increases TRF2 mRNA expression and telomerase activity. Leukemia 2003, 17, 2007–2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saha, B.; Zitnik, G.; Johnson, S.; Nguyen, Q.; Risques, R.A.; Martin, G.M.; Oshima, J. DNA damage accumulation and TRF2 degradation in atypical Werner syndrome fibroblasts with LMNA mutations. Front. Genet. 2013, 4, 129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takai, H.; Smogorzewska, A.; de Lange, T. DNA Damage Foci at Dysfunctional Telomeres. Curr. Biol. 2003, 13, 1549–1556. [Google Scholar] [CrossRef] [Green Version]
- Dinami, R.; Porru, M.; Amoreo, C.A.; Sperduti, I.; Mottolese, M.; Buglioni, S.; Marinelli, D.; Maugeri-Saccà, M.; Sacconi, A.; Blandino, G.; et al. TRF2 and VEGF-A: An unknown relationship with prognostic impact on survival of colorectal cancer patients. J. Exp. Clin. Cancer Res. 2020, 39, 1–13. [Google Scholar] [CrossRef]
- Diehl, M.C.; Idowu, M.O.; Kimmelshue, K.N.; York, T.P.; Jackson-Cook, C.K.; Turner, K.C.; Holt, S.E.; Elmore, L.W. Elevated TRF2 in advanced breast cancers with short telomeres. Breast Cancer Res. Treat. 2010, 127, 623–630. [Google Scholar] [CrossRef]
- Nijjar, T.; Bassett, E.; Garbe, J.; Takenaka, Y.; Stampfer, M.R.; Gilley, D.; Yaswen, P. Accumulation and altered localization of telomere-associated protein TRF2 in immortally transformed and tumor-derived human breast cells. Oncogene 2005, 24, 3369–3376. [Google Scholar] [CrossRef]
- Knecht, H.; Sawan, B.; Lichtensztejn, D.; Lemieux, B.; Wellinger, R.J.; Mai, S. The 3D nuclear organization of telomeres marks the transition from Hodgkin to Reed–Sternberg cells. Leukemia 2008, 23, 565–573. [Google Scholar] [CrossRef] [Green Version]
- Lajoie, V.; Lemieux, B.; Sawan, B.; Lichtensztejn, D.; Lichtensztejn, Z.; Wellinger, R.; Mai, S.; Knecht, H. LMP1 mediates multinuclearity through downregulation of shelterin proteins and formation of telomeric aggregates. Blood 2015, 125, 2101–2110. [Google Scholar] [CrossRef] [Green Version]
- Knecht, H.; Johnson, N.A.; Haliotis, T.; Lichtensztejn, D.; Mai, S. Disruption of direct 3D telomere–TRF2 interaction through two molecularly disparate mechanisms is a hallmark of primary Hodgkin and Reed–Sternberg cells. Lab. Investig. 2017, 97, 772–781. [Google Scholar] [CrossRef]
- Muñoz, P.; Blanco, R.; Flores, J.M.; Blasco, M.A. XPF nuclease-dependent telomere loss and increased DNA damage in mice overexpressing TRF2 result in premature aging and cancer. Nat. Genet. 2005, 37, 1063–1071. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wu, X. Abnormal function of telomere protein TRF2 induces cell mutation and the effects of environmental tumor-promoting factors (Review). Oncol. Rep. 2021, 46, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Matsutani, N.; Yokozaki, H.; Tahara, E.; Tahara, H.; Kuniyasu, H.; Haruma, K.; Chayama, K.; Yasui, W.; Tahara, E. Expression of telomeric repeat binding factor 1 and 2 and TRF1-interacting nuclear protein 2 in human gastric carcinomas. Int. J. Oncol. 2001, 19, 507–512. [Google Scholar] [CrossRef]
- Bellon, M.; Datta, A.; Brown, M.; Pouliquen, J.-F.; Couppié, P.; Kazanji, M.; Nicot, C. Increased expression of telomere length regulating factors TRF1, TRF2 and TIN2 in patients with adult T-cell leukemia. Int. J. Cancer 2006, 119, 2090–2097. [Google Scholar] [CrossRef] [PubMed]
- Nera, B.; Huang, H.-S.; Lai, T.; Xu, L. Elevated levels of TRF2 induce telomeric ultrafine anaphase bridges and rapid telomere deletions. Nat. Commun. 2015, 6, 10132. [Google Scholar] [CrossRef] [PubMed]
- Muhammad Imran, S.A. The Role of TRF2 in Regulating Neural Progenitor Cells Proliferation and Surivival; Imperial College London: London, UK, 2019. [Google Scholar]
- Walker, J.R.; Zhu, X.-D. Post-translational modifications of TRF1 and TRF2 and their roles in telomere maintenance. Mech. Ageing Dev. 2012, 133, 421–434. [Google Scholar] [CrossRef] [PubMed]
- Hornbeck, P.V.; Zhang, B.; Murray, B.; Kornhauser, J.M.; Latham, V.; Skrzypek, E. PhosphoSitePlus, 2014: Mutations, PTMs and recalibrations. Nucleic Acids Res. 2015, 43, D512–D520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chi, Y.; Welcker, M.; Hizli, A.A.; Posakony, J.J.; Aebersold, R.; Clurman, B.E. Identification of CDK2 substrates in human cell lysates. Genome Biol. 2008, 9, R149. [Google Scholar] [CrossRef] [Green Version]
- Sarek, G.; Kotsantis, P.; Ruis, P.; Van Ly, D.; Margalef, P.; Borel, V.; Zheng, X.-F.; Flynn, H.; Snijders, B.; Chowdhury, D.; et al. CDK phosphorylation of TRF2 controls t-loop dynamics during the cell cycle. Nat. Cell Biol. 2019, 575, 523–527. [Google Scholar] [CrossRef]
- Picco, V.; Coste, I.; Giraud-Panis, M.-J.; Renno, T.; Gilson, E.; Pagès, G. ERK1/2/MAPK pathway-dependent regulation of the telomeric factor TRF. Oncotarget 2016, 7, 46615–46627. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.-T.; Lim, D.-S.; Canman, C.E.; Kastan, M.B. Substrate Specificities and Identification of Putative Substrates of ATM Kinase Family Members. J. Biol. Chem. 1999, 274, 37538–37543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanaka, H.; Mendonca, M.S.; Bradshaw, P.S.; Hoelz, D.J.; Malkas, L.H.; Meyn, M.S.; Gilley, D. DNA damage-induced phosphorylation of the human telomere-associated protein TRF. Proc. Natl. Acad. Sci. USA 2005, 102, 15539–15544. [Google Scholar] [CrossRef] [Green Version]
- Matsuoka, S.; Rotman, G.; Ogawa, A.; Shiloh, Y.; Tamai, K.; Elledge, S.J. Ataxia telangiectasia-mutated phosphorylates Chk2 in vivo and in vitro. Proc. Natl. Acad. Sci. USA 2000, 97, 10389–10394. [Google Scholar] [CrossRef] [Green Version]
- Buscemi, G.; Zannini, L.; Fontanella, E.; Lecis, D.; Lisanti, S.; Delia, D. The Shelterin Protein TRF2 Inhibits Chk2 Activity at Telomeres in the Absence of DNA Damage. Curr. Biol. 2009, 19, 874–879. [Google Scholar] [CrossRef] [Green Version]
- Zhou, J.; Deng, Z.; Norseen, J.; Lieberman, P.M. Regulation of Epstein-Barr Virus Origin of Plasmid Replication (OriP) by the S-Phase Checkpoint Kinase Chk. J. Virol. 2010, 84, 4979–4987. [Google Scholar] [CrossRef] [Green Version]
- Geiss-Friedlander, R.; Melchior, F. Concepts in sumoylation: A decade on. Nat. Rev. Mol. Cell Biol. 2007, 8, 947–956. [Google Scholar] [CrossRef]
- Yang, Y.; He, Y.; Wang, X.; Liang, Z.; He, G.; Zhang, P.; Zhu, H.; Xu, N.; Liang, S. Protein SUMOylation modification and its associations with disease. Open Biol. 2017, 7, 170167. [Google Scholar] [CrossRef]
- Her, J.; Jeong, Y.Y.; Chung, I.K. PIAS1-mediated sumoylation promotes STUbL-dependent proteasomal degradation of the human telomeric protein TRFFEBS. Letters 2015, 589, 3277–3286. [Google Scholar]
- Potts, P.R.; Yu, H. The SMC5/6 complex maintains telomere length in ALT cancer cells through SUMOylation of telomere-binding proteins. Nat. Struct. Mol. Biol. 2007, 14, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Yalçin, Z.; Selenz, C.; Jacobs, J.J.L. Ubiquitination and SUMOylation in Telomere Maintenance and Dysfunction. Front. Genet. 2017, 8, 67. [Google Scholar] [CrossRef] [Green Version]
- Peuscher, M.; Jacobs, J.J. Posttranslational control of telomere maintenance and the telomere damage response. Cell Cycle 2012, 11, 1524–1534. [Google Scholar] [CrossRef] [Green Version]
- Sun, H.; Leverson, J.D.; Hunter, T. Conserved function of RNF4 family proteins in eukaryotes: Targeting a ubiquitin ligase to SUMOylated proteins. EMBO J. 2007, 26, 4102–4112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, Y.-C.; Oram, M.; Bielinsky, A.-K. SUMO-Targeted Ubiquitin Ligases and Their Functions in Maintaining Genome Stability. Int. J. Mol. Sci. 2021, 22, 5391. [Google Scholar] [CrossRef]
- Groocock, L.M.; Nie, M.; Prudden, J.; Moiani, D.; Wang, T.; Cheltsov, A.; Rambo, R.P.; Arvai, A.S.; Hitomi, C.; Tainer, J.A.; et al. RNF 4 interacts with both SUMO and nucleosomes to promote the DNA damage response. EMBO Rep. 2014, 15, 601–608. [Google Scholar] [CrossRef] [Green Version]
- Fujita, K.; Horikawa, I.; Mondal, A.M.; Jenkins, L.M.M.; Appella, E.; Vojtesek, B.; Bourdon, J.-C.; Lane, D.; Harris, C.C. Positive feedback between p53 and TRF2 during telomere-damage signalling and cellular senescence. Nat. Cell Biol. 2010, 12, 1205–1212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Her, Y.R.; Chung, I.K. p300-mediated acetylation of TRF2 is required for maintaining functional telomeres. Nucleic Acids Res. 2013, 41, 2267–2283. [Google Scholar] [CrossRef] [Green Version]
- Dutto, I.; Scalera, C.; Prosperi, E. CREBBP and p300 lysine acetyl transferases in the DNA damage response. Cell. Mol. Life Sci. 2018, 75, 1325–1338. [Google Scholar] [CrossRef] [PubMed]
- Hassa, P.O.; Buerki, C.; Lombardi, C.; Imhof, R.; Hottiger, M.O. Transcriptional Coactivation of Nuclear Factor-κB-dependent Gene Expression by p300 Is Regulated by Poly(ADP)-ribose Polymerase-1*. J. Biol. Chem. 2003, 278, 45145–45153. [Google Scholar] [CrossRef] [Green Version]
- Chaudhuri, A.R.; Nussenzweig, A.R.C.A. The multifaceted roles of PARP1 in DNA repair and chromatin remodelling. Nat. Rev. Mol. Cell Biol. 2017, 18, 610–621. [Google Scholar] [CrossRef] [PubMed]
- Haince, J.-F.; Kozlov, S.; Dawson, V.; Dawson, T.M.; Hendzel, M.; Lavin, M.F.; Poirier, G.G. Ataxia Telangiectasia Mutated (ATM) Signaling Network Is Modulated by a Novel Poly(ADP-ribose)-dependent Pathway in the Early Response to DNA-damaging Agents. J. Biol. Chem. 2007, 282, 16441–16453. [Google Scholar] [CrossRef] [Green Version]
- Sousa, F.; Matuo, R.; Soares, D.G.; Escargueil, A.; Henriques, J.A.; Larsen, A.K.; Saffi, J. PARPs and the DNA damage response. Carcinogenesis 2012, 33, 1433–1440. [Google Scholar] [CrossRef] [Green Version]
- Gomez, M.; Wu, J.; Schreiber, V.; Dunlap, J.; Dantzer, F.; Wang, Y.; Liu, Y. PARP1 Is a TRF2-associated Poly(ADP-Ribose)Polymerase and Protects Eroded Telomeres. Mol. Biol. Cell 2006, 17, 1686–1696. [Google Scholar] [CrossRef] [Green Version]
- Mao, Z.; Seluanov, A.; Jiang, Y.; Gorbunova, V. TRF2 is required for repair of nontelomeric DNA double-strand breaks by homologous recombination. Proc. Natl. Acad. Sci. USA 2007, 104, 13068–13073. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zalzman, M.; Meltzer, W.A.; Portney, B.A.; Brown, R.A.; Gupta, A. The Role of Ubiquitination and SUMOylation in Telomere Biology. Curr. Issues Mol. Biol. 2020, 35, 85–98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huda, N.; Tanaka, H.; Mendonca, M.S.; Gilley, D. DNA Damage-Induced Phosphorylation of TRF2 Is Required for the Fast Pathway of DNA Double-Strand Break Repair. Mol. Cell. Biol. 2009, 29, 3597–3604. [Google Scholar] [CrossRef] [Green Version]
- Spengler, D. The Protein Kinase Aurora-C Phosphorylates TRF. Cell Cycle 2007, 6, 2579–2580. [Google Scholar] [CrossRef] [Green Version]
- Dantzer, F.; Giraud-Panis, M.-J.; Jaco, I.; Amé, J.-C.; Schultz, I.; Blasco, M.; Koering, C.-E.; Gilson, E.; Murcia, J.M.-D.; de Murcia, G.; et al. Functional Interaction between Poly(ADP-Ribose) Polymerase 2 (PARP-2) and TRF2: PARP Activity Negatively Regulates TRF. Mol. Cell. Biol. 2004, 24, 1595–1607. [Google Scholar] [CrossRef] [Green Version]
- Baker, A.M.; Fu, Q.; Hayward, W.; Victoria, S.; Pedroso, I.M.; Lindsay, S.M.; Fletcher, T.M. The Telomere Binding Protein TRF2 Induces Chromatin Compaction. PLoS ONE 2011, 6, e19124. [Google Scholar] [CrossRef]
- De Lange, T. How Telomeres Solve the End-Protection Problem. Science 2009, 326, 948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palm, W.; de Lange, T. How Shelterin Protects Mammalian Telomeres. Annu. Rev. Genet. 2008, 42, 301–334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Steensel, B.; Smogorzewska, A.; de Lange, T. TRF2 Protects Human Telomeres from End-to-End Fusions. Cell 1998, 92, 401–413. [Google Scholar] [CrossRef] [Green Version]
- Saint-Léger, A.; Koelblen, M.; Civitelli, L.; Bah, A.; Djerbi, N.; Giraud-Panis, M.-J.; Londono-Vallejo, A.; Ascenzioni, F.; Gilson, E. The basic N-terminal domain of TRF2 limits recombination endonuclease action at human telomeres. Cell Cycle 2014, 13, 2469–2474. [Google Scholar] [CrossRef] [Green Version]
- Arnoult, N.; Karlseder, J. Complex interactions between the DNA-damage response and mammalian telomeres. Nat. Struct. Mol. Biol. 2015, 22, 859–866. [Google Scholar] [CrossRef]
- Blasco, M.A. Telomeres and human disease: Ageing, cancer and beyond. Nat. Rev. Genet. 2005, 6, 611–622. [Google Scholar] [CrossRef]
- Celli, G.B.; de Lange, T. DNA processing is not required for ATM-mediated telomere damage response after TRF2 deletion. Nat. Cell Biol. 2005, 7, 712–718. [Google Scholar] [CrossRef]
- Denchi, E.L.; de Lange, T. Protection of telomeres through independent control of ATM and ATR by TRF2 and POT. Nature 2007, 448, 1068–1071. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, G.; Jurk, D.; Marques, F.M.; Correia-Melo, C.; Hardy, T.L.D.; Gackowska, A.; Anderson, R.; Taschuk, M.; Mann, J.; Passos, J.F. Telomeres are favoured targets of a persistent DNA damage response in ageing and stress-induced senescence. Nat. Commun. 2012, 3, 708. [Google Scholar] [CrossRef]
- Okamoto, K.; Bartocci, C.; Ouzounov, I.; Diedrich, J.K.; Iii, J.R.Y.; Denchi, E.L. A two-step mechanism for TRF2-mediated chromosome-end protection. Nat. Cell Biol. 2013, 494, 502–505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anderson, R.; Lagnado, A.; Maggiorani, D.; Walaszczyk, A.; Dookun, E.; Chapman, J.; Birch, J.; Salmonowicz, H.; Ogrodnik, M.; Jurk, D.; et al. Length-independent telomere damage drives post-mitotic cardiomyocyte senescence. EMBO J. 2019, 38, 100492. [Google Scholar] [CrossRef] [PubMed]
- Hunter, T. Protein kinases and phosphatases: The Yin and Yang of protein phosphorylation and signaling. Cell 1995, 80, 225–236. [Google Scholar] [CrossRef] [Green Version]
- Lavin, M.; Shiloh, Y. THE GENETIC DEFECT IN ATAXIA-TELANGIECTASIA. Annu. Rev. Immunol. 1997, 15, 177–202. [Google Scholar] [CrossRef]
- Uziel, T.; Lerenthal, Y.; Moyal, L.; Andegeko, Y.; Mittelman, L.; Shiloh, Y. Requirement of the MRN complex for ATM activation by DNA damage. EMBO J. 2003, 22, 5612–5621. [Google Scholar] [CrossRef] [Green Version]
- Bakkenist, C.J.; Kastan, M.B. DNA damage activates ATM through intermolecular autophosphorylation and dimer dissociation. Nat. Cell Biol. 2003, 421, 499–506. [Google Scholar] [CrossRef]
- Derheimer, F.A.; Kastan, M.B. Multiple roles of ATM in monitoring and maintaining DNA integrity. FEBS Lett. 2010, 584, 3675–3681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kozlov, S.V.; Graham, M.E.; Jakob, B.; Tobias, F.; Kijas, A.W.; Tanuji, M.; Chen, P.; Robinson, P.J.; Taucher-Scholz, G.; Suzuki, K.; et al. Autophosphorylation and ATM activation: Additional sites add to the complexity. J. Biol. Chem. 2011, 286, 9107–9119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumaran, M.; Fazry, S. The effect of MRN complex and ATM kinase inhibitors on Zebrafish embryonic development. AIP Conf. Proc. 2018, 1940, 020074. [Google Scholar]
- Doksani, Y.; Wu, J.Y.; De Lange, T.; Zhuang, X. Super-Resolution Fluorescence Imaging of Telomeres Reveals TRF2-Dependent T-loop Formation. Cell 2013, 155, 345–356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, H.H.Y.; Pannunzio, N.R.; Adachi, N.; Lieber, M.R. Non-homologous DNA end joining and alternative pathways to double-strand break repair. Nat. Rev. Mol. Cell Biol. 2017, 18, 495–506. [Google Scholar] [CrossRef] [PubMed]
- Ribes-Zamora, A.; Indiviglio, S.M.; Mihalek, I.; Williams, C.; Bertuch, A.A. TRF2 Interaction with Ku Heterotetramerization Interface Gives Insight into c-NHEJ Prevention at Human Telomeres. Cell Rep. 2013, 5, 194–206. [Google Scholar] [CrossRef] [Green Version]
- Dimitrova, N.; Chen, Y.-C.M.; Spector, D.L.; De Lange, T. 53BP1 promotes non-homologous end joining of telomeres by increasing chromatin mobility. Nat. Cell Biol. 2008, 456, 524–528. [Google Scholar] [CrossRef]
- Mao, A.S.; Mooney, D.J. Regenerative medicine: Current therapies and future directions. Proc. Natl. Acad. Sci. USA 2015, 112, 14452–14459. [Google Scholar] [CrossRef] [Green Version]
- Konishi, A.; de Lange, T. Cell cycle control of telomere protection and NHEJ revealed by a ts mutation in the DNA-binding domain of TRF. Genes Dev. 2008, 22, 1221–1230. [Google Scholar] [CrossRef] [Green Version]
- Smogorzewska, A.; Karlseder, J.; Holtgreve-Grez, H.; Jauch, A.; de Lange, T. DNA Ligase IV-Dependent NHEJ of Deprotected Mammalian Telomeres in G1 and G. Curr. Biol. 2002, 12, 1635–1644. [Google Scholar] [CrossRef] [Green Version]
- Roy, S.; Roy, S.; Kar, M.; Thakur, S.; Akhter, Y.; Kumar, A.; Delogu, F.; Padhi, S.; Saha, A.; Banerjee, B. p38 MAPK pathway and its interaction with TRF2 in cisplatin induced chemotherapeutic response in head and neck cancer. Oncogenesis 2018, 7, 53. [Google Scholar] [CrossRef]
- Muraki, K.; Nabetani, A.; Nishiyama, A.; Ishikawa, F. Essential roles of Xenopus TRF2 in telomere end protection and replication. Genes Cells 2011, 16, 728–739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bradshaw, P.S.; Stavropoulos, D.J.; Meyn, M.S. Human telomeric protein TRF2 associates with genomic double-strand breaks as an early response to DNA damage. Nat. Genet. 2005, 37, 193–197. [Google Scholar] [CrossRef]
- Oh, H.; Wang, S.; Prahash, A.; Sano, M.; Moravec, C.S.; Taffet, G.E.; Michael, L.H.; Youker, K.; Entman, M.L.; Schneider, M. Telomere attrition and Chk2 activation in human heart failure. Proc. Natl. Acad. Sci. USA 2003, 100, 5378–5383. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zannini, L.; Delia, D.; Buscemi, G. CHK2 kinase in the DNA damage response and beyond. J. Mol. Cell Biol. 2014, 6, 442–457. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, X.-D.; Küster, B.; Mann, M.; Petrini, J.H.J.; Lange, T.d. Cell-cycle-regulated association of RAD50/MRE11/NBS1 with TRF2 and human telomeres. Nat. Genet. 2000, 25, 347–352. [Google Scholar] [CrossRef]
- Lee, J.-H.; Paull, T.T. Direct Activation of the ATM Protein Kinase by the Mre11/Rad50/Nbs1 Complex. Science 2004, 304, 93. [Google Scholar] [CrossRef]
- Lenain, C.; Bauwens, S.; Amiard, S.; Brunori, M.; Giraud-Panis, M.-J.; Gilson, E. The Apollo 5′ Exonuclease Functions Together with TRF2 to Protect Telomeres from DNA Repair. Curr. Biol. 2006, 16, 1303–1310. [Google Scholar] [CrossRef]
- Ye, J.; Lenain, C.; Bauwens, S.; Rizzo, A.; Saint-Léger, A.; Poulet, A.; Benarroch, D.; Magdinier, F.; Morere, J.; Amiard, S.; et al. TRF2 and Apollo Cooperate with Topoisomerase 2α to Protect Human Telomeres from Replicative Damage. Cell 2010, 142, 230–242. [Google Scholar] [CrossRef]
- Van Overbeek, M.; de Lange, T. Apollo, an Artemis-Related Nuclease, Interacts with TRF2 and Protects Human Telomeres in S Phase. Curr. Biol. 2006, 16, 1295–1302. [Google Scholar] [CrossRef] [Green Version]
- Song, K.; Jung, D.; Jung, Y.; Lee, S.-G.; Lee, I. Interaction of human Ku70 with TRF. FEBS Lett. 2000, 481, 81–85. [Google Scholar] [CrossRef] [Green Version]
- Ribes-Zamora, A.; Mihalek, I.; Lichtarge, O.; Bertuch, A.A. Distinct faces of the Ku heterodimer mediate DNA repair and telomeric functions. Nat. Struct. Mol. Biol. 2007, 14, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Bombarde, O.; Boby, C.; Gomez, D.; Frit, P.; Giraud-Panis, M.-J.; Gilson, E.; Salles, B.; Calsou, P. TRF2/RAP1 and DNA–PK mediate a double protection against joining at telomeric ends. EMBO J. 2010, 29, 1573–1584. [Google Scholar] [CrossRef] [PubMed]
- Mirman, Z.; de Lange, T. 53BP1: A DSB escort. Genes Dev. 2020, 34, 7–23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fotiadou, P.; Henegariu, O.; Sweasy, J.B. DNA Polymerase β Interacts with TRF2 and Induces Telomere Dysfunction in a Murine Mammary Cell Line. Cancer Res. 2004, 64, 3830. [Google Scholar] [CrossRef] [Green Version]
- Muftuoglu, M.; Wong, H.K.; Imam, S.Z.; Wilson, D.M.; Bohr, V.A.; Opresko, P. Telomere Repeat Binding Factor 2 Interacts with Base Excision Repair Proteins and Stimulates DNA Synthesis by DNA Polymerase β. Cancer Res. 2006, 66, 113–124. [Google Scholar] [CrossRef] [Green Version]
- Miller, A.S.; Balakrishnan, L.; Buncher, N.A.; Opresko, P.L.; Bambara, R.A. Telomere proteins POT1, TRF1 and TRF2 augment long-patch base excision repair in vitro. Cell Cycle 2012, 11, 998–1007. [Google Scholar] [CrossRef] [Green Version]
- Pereira, C.D.; Martins, F.; Santos, M.; Müeller, T.; Silva, O.A.B.D.C.E.; Rebelo, S. Nuclear Accumulation of LAP1:TRF2 Complex During DNA Damage Response Uncovers a Novel Role for LAP. Cells 2020, 9, 1804. [Google Scholar] [CrossRef]
- Porreca, R.M.; Glousker, G.; Awad, A.; Fernandez, M.I.M.; Gibaud, A.; Naucke, C.; Cohen, S.B.; Bryan, T.M.; Tzfati, Y.; Draskovic, I.; et al. Human RTEL1 stabilizes long G-overhangs allowing telomerase-dependent over-extension. Nucleic Acids Res. 2018, 46, 4533–4545. [Google Scholar] [CrossRef] [Green Version]
- Awad, A.; Glousker, G.; Lamm, N.; Tawil, S.; Hourvitz, N.; Smoom, R.; Revy, P.; Tzfati, Y. Full length RTEL1 is required for the elongation of the single-stranded telomeric overhang by telomerase. Nucleic Acids Res. 2020, 48, 7239–7251. [Google Scholar] [CrossRef]
- Bauwens, S.; Lototska, L.; Koundrioukoff, S.; Debatisse, M.; Ye, J.; Gilson, E.; Mendez-Bermudez, A. The Telomeric Protein TRF2 Regulates Replication Origin Activity within Pericentromeric Heterochromatin. Life 2021, 11, 267. [Google Scholar] [CrossRef]
- Fouché, N.; Cesare, A.; Willcox, S.; Özgür, S.; Compton, S.A.; Griffith, J.D. The Basic Domain of TRF2 Directs Binding to DNA Junctions Irrespective of the Presence of TTAGGG Repeats. J. Biol. Chem. 2006, 281, 37486–37495. [Google Scholar] [CrossRef] [Green Version]
- Lieberman, P.M. Resistance from the flanks: A role for telomere repeat factor 2 in chemotherapeutic drug resistance. Cancer Biol. Ther. 2006, 5, 957–958. [Google Scholar] [CrossRef]
- Simonet, T.; Zaragosi, L.-E.; Philippe, C.; Lebrigand, K.; Schouteden, C.; Augereau, A.; Bauwens, S.; Ye, J.; Santagostino, M.; Giulotto, E.; et al. The human TTAGGG repeat factors 1 and 2 bind to a subset of interstitial telomeric sequences and satellite repeats. Cell Res. 2011, 21, 1028–1038. [Google Scholar] [CrossRef] [Green Version]
- Yang, D.; Xiong, Y.; Kim, H.; He, Q.; Li, Y.; Chen, R.; Songyang, Z. Human telomeric proteins occupy selective interstitial sites. Cell Res. 2011, 21, 1013–1027. [Google Scholar] [CrossRef] [Green Version]
- Mukherjee, A.K.; Sharma, S.; Bagri, S.; Kutum, R.; Kumar, P.; Hussain, A.; Singh, P.; Saha, D.; Kar, A.; Dash, D.; et al. Telomere repeat–binding factor 2 binds extensively to extra-telomeric G-quadruplexes and regulates the epigenetic status of several gene promoters. J. Biol. Chem. 2019, 294, 17709–17722. [Google Scholar] [CrossRef] [Green Version]
- Hanaoka, S.; Nagadoi, A.; Nishimura, Y. Comparison between TRF2 and TRF1 of their telomeric DNA-bound structures and DNA-binding activities. Protein Sci. 2009, 14, 119–130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, Y.; Lee, S.; Bang, S.; Kim, S.; Choi, K.; Lee, C.; Lee, S.-G.; Kim, C.J.; Song, K.; Lee, I. TRF2 is in neuroglial cytoplasm and induces neurite-like processes. FEBS Lett. 2003, 557, 129–132. [Google Scholar] [CrossRef] [Green Version]
- Purohit, G.; Mukherjee, A.K.; Sharma, S.; Chowdhury, S.; Mukherjee, A.K. Extratelomeric Binding of the Telomere Binding Protein TRF2 at the PCGF3 Promoter Is G-Quadruplex Motif-Dependent. Biochemistry 2018, 57, 2317–2324. [Google Scholar] [CrossRef] [PubMed]
- Bochman, M.; Paeschke, K.; Zakian, V.A. DNA secondary structures: Stability and function of G-quadruplex structures. Nat. Rev. Genet. 2012, 13, 770–780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moye, A.L.; Porter, K.C.; Cohen, S.B.; Phan, T.; Zyner, K.; Sasaki, N.; Lovrecz, G.; Beck, J.; Bryan, T.M. Telomeric G-quadruplexes are a substrate and site of localization for human telomerase. Nat. Commun. 2015, 6, 7643. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rhodes, D.; Lipps, H.J. G-quadruplexes and their regulatory roles in biology. Nucleic Acids Res. 2015, 43, 8627–8637. [Google Scholar] [CrossRef] [Green Version]
- Haensel-Hertsch, R.; Di Antonio, M.; Balasubramanian, S. DNA G-quadruplexes in the human genome: Detection, functions and therapeutic potential. Nat. Rev. Mol. Cell Biol. 2017, 18, 279–284. [Google Scholar] [CrossRef]
- Bryan, T.M. G-Quadruplexes at Telomeres: Friend or Foe? Molecules 2020, 25, 3686. [Google Scholar] [CrossRef]
- De Magis, A.; Manzo, S.G.; Russo, M.; Marinello, J.; Morigi, R.; Sordet, O.; Capranico, G. DNA damage and genome instability by G-quadruplex ligands are mediated by R loops in human cancer cells. Proc. Natl. Acad. Sci. USA 2019, 116, 816–825. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wood, A.M.; Danielsen, J.M.R.; Lucas, C.A.; Rice, E.L.; Scalzo, D.; Shimi, T.; Goldman, R.D.; Smith, E.D.; Le Beau, M.M.; Kosak, S.T. TRF2 and lamin A/C interact to facilitate the functional organization of chromosome ends. Nat. Commun. 2014, 5, 5467. [Google Scholar] [CrossRef] [Green Version]
- Gonzalez-Suarez, I.; Redwood, A.B.; Perkins, S.M.; Vermolen, B.; Lichtensztejin, D.; Grotsky, D.A.; Morgado-Palacin, L.; Gapud, E.J.; Sleckman, B.P.; Sullivan, T.; et al. Novel roles for A-type lamins in telomere biology and the DNA damage response pathway. EMBO J. 2009, 28, 2414–2427. [Google Scholar] [CrossRef]
- Bronshtein, I.; Kepten, E.; Kanter, I.; Berezin, S.; Lindner, M.; Redwood, A.B.; Mai, S.; Gonzalo, S.; Foisner, R.; Shav-Tal, Y.; et al. Loss of lamin A function increases chromatin dynamics in the nuclear interior. Nat. Commun. 2015, 6, 8044. [Google Scholar] [CrossRef] [Green Version]
- Wood, A.M.; Laster, K.; Rice, E.L.; Kosak, S.T. A beginning of the end: New insights into the functional organization of telomeres. Nucleus 2015, 6, 172–178. [Google Scholar] [CrossRef] [Green Version]
- Smith, E.D.; Garza-Gongora, A.G.; MacQuarrie, K.L.; Kosak, S.T. Interstitial telomeric loops and implications of the interaction between TRF2 and lamin A/C. Differentiation 2018, 102, 19–26. [Google Scholar] [CrossRef]
- Kim, H.; Lee, O.-H.; Xin, H.; Chen, L.-Y.; Qin, J.; Chae, H.K.; Lin, S.-Y.; Safari, A.; Liu, D.; Songyang, Z. TRF2 functions as a protein hub and regulates telomere maintenance by recognizing specific peptide motifs. Nat. Struct. Mol. Biol. 2009, 16, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Feuerhahn, S.; Chen, L.-Y.; Luke, B.; Porro, A. No DDRama at chromosome ends: TRF2 takes centre stage. Trends Biochem. Sci. 2015, 40, 275–285. [Google Scholar] [CrossRef]
- Hussain, T.; Saha, D.; Purohit, G.; Kar, A.; Mukherjee, A.K.; Sharma, S.; Sengupta, S.; Dhapola, P.; Maji, B.; Vedagopuram, S.; et al. Transcription regulation of CDKN1A (p21/CIP1/WAF1) by TRF2 is epigenetically controlled through the REST repressor complex. Sci. Rep. 2017, 7, 11541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ovando-Roche, P.; Yu, J.S.; Testori, S.; Ho, C.; Cui, W. TRF2-Mediated Stabilization of hREST4 Is Critical for the Differentiation and Maintenance of Neural Progenitors. Stem Cells 2014, 32, 2111–2122. [Google Scholar] [CrossRef] [Green Version]
- Zhang, P.; Furukawa, K.; Opresko, P.L.; Xu, X.; Bohr, V.A.; Mattson, M.P. TRF2 dysfunction elicits DNA damage responses associated with senescence in proliferating neural cells and differentiation of neurons. J. Neurochem. 2006, 97, 567–581. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Casaday-Potts, R.; Precht, P.; Jiang, H.; Liu, Y.; Pazin, M.; Mattson, M.P. Nontelomeric splice variant of telomere repeat-binding factor 2 maintains neuronal traits by sequestering repressor element 1-silencing transcription factor. Proc. Natl. Acad. Sci. USA 2011, 108, 16434–16439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grammatikakis, I.; Zhang, P.; Mattson, M.P.; Gorospe, M. The long and the short of TRF2 in neurogenesis. Cell Cycle 2016, 15, 3026–3032. [Google Scholar] [CrossRef] [Green Version]
- Robin, J.D.; Burbano, M.J.; Peng, H.; Croce, O.; Thomas, J.L.; Laberthonniere, C.; Renault, V.; Lototska, L.; Pousse, M.; Tessier, F.; et al. Mitochondrial function in skeletal myofibers is controlled by a TRF2-SIRT3 axis over lifetime. Aging Cell 2020, 19, e13097. [Google Scholar] [CrossRef]
- Cherfils-Vicini, J.; Iltis, C.; Cervera, L.; Pisano, S.; Croce, O.; Sadouni, N.; Győrffy, B.; Collet, R.; Renault, V.M.; Rey-Millet, M.; et al. Cancer cells induce immune escape via glycocalyx changes controlled by the telomeric protein TRF. EMBO J. 2019, 38, 100012. [Google Scholar] [CrossRef]
- Biroccio, A.; Cherfils-Vicini, J.; Augereau, A.; Pinte, S.; Bauwens, S.; Ye, J.; Simonet, T.; Horard, B.; Jamet, K.; Cervera, L.; et al. TRF2 inhibits a cell-extrinsic pathway through which natural killer cells eliminate cancer cells. Nat. Cell Biol. 2013, 15, 818–828. [Google Scholar] [CrossRef] [PubMed]
- Imran, S.; Yazid, M.; Idrus, R.; Maarof, M.; Nordin, A.; Razali, R.; Lokanathan, Y. Is There an Interconnection between Epithelial–Mesenchymal Transition (EMT) and Telomere Shortening in Aging? Int. J. Mol. Sci. 2021, 22, 3888. [Google Scholar] [CrossRef] [PubMed]

| Modification | Amino Acids | Modifying Enzymes | Function | Reference (s) |
|---|---|---|---|---|
| Phosphorylation | Ser20 | Chk2 | Phosphorylation of TRF2 by Chk2 decreases TRF2 binding to telomeric DNA. S20 phosphorylation is required for TRF2 interaction with G-rich RNA and recruitment of ORC to OriP. | [74,75] |
| Ser365 | CDK | Regulates the assembly and disassembly of t-loop during the cell-cycle or DNA replication, which protects the telomeres from replicative stress. | [69] | |
| Thr188 | ATM | Early recruitment at the DNA break site and may be involved in DNA damage response/repair. | [72,95] | |
| Thr358 | Aurora C | Possible function in cell replication and unwinding of the chromosome. | [96] | |
| SUMOylation | IK140TE, LK245SE, and RK333DE | MMS21 | Stabilization of TRF2 and recruitment at the telomeres. | [79,80] |
| - | PIAS1 | Interacts with RNF4 to regulate TRF2 level at the telomeres. | [78] | |
| Ubiquitylation | K173 K180 K184 | Siah1 | Siah1 is a p53-inducible E3 ligase with a C3H4-type RING finger domain. Knockdown of Siah1 stabilizes TRF2 and telomere length maintenance. | [85] |
| - | RNF4 | Interacts together with PIAS1 in modulating TRF2 role in telomere maintenance and protection through regulating the level of TRF2 at the telomeres. Acts downstream of the ATM-kinase pathway in DDR. | [78,84] | |
| Acetylation | K293 | P300 | Required for TRF2 stabilization and efficient binding at the telomeres. | [86] |
| Poly(ADP-ribosyl)ation or PARsylation | - | PARP1, PARP2 | Reduces the binding of TRF2 to telomeres that may contribute to dysfunctional telomeres. | [92,97] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imran, S.A.M.; Yazid, M.D.; Cui, W.; Lokanathan, Y. The Intra- and Extra-Telomeric Role of TRF2 in the DNA Damage Response. Int. J. Mol. Sci. 2021, 22, 9900. https://doi.org/10.3390/ijms22189900
Imran SAM, Yazid MD, Cui W, Lokanathan Y. The Intra- and Extra-Telomeric Role of TRF2 in the DNA Damage Response. International Journal of Molecular Sciences. 2021; 22(18):9900. https://doi.org/10.3390/ijms22189900
Chicago/Turabian StyleImran, Siti A. M., Muhammad Dain Yazid, Wei Cui, and Yogeswaran Lokanathan. 2021. "The Intra- and Extra-Telomeric Role of TRF2 in the DNA Damage Response" International Journal of Molecular Sciences 22, no. 18: 9900. https://doi.org/10.3390/ijms22189900






