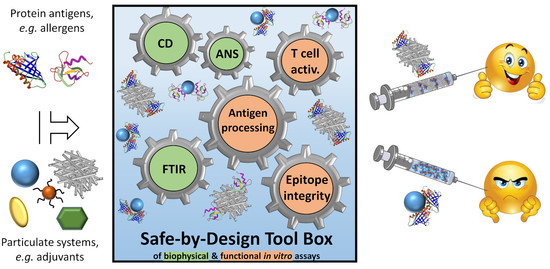
Structural Alterations of Antigens at the Material Interface: An Early Decision Toolbox Facilitating Safe-by-Design Nanovaccine Development
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of the Synthesized SiO2 NPs and Alhydrogel®
2.2. Efficient Conjugation of Allergens with SiO2 NPs and Alhydrogel®
2.3. Conjugation of Allergens Decreases Their Proteolytic Stability
2.4. Changes in the 3D Fold of the Allergen upon (Nano)Particle Conjugation
2.5. Structural Integrity of T Cell and IgE Epitopes
3. Materials and Methods
3.1. Patients and Sera
3.2. Synthesis and Physicochemical Characterization of SiO2 NPs
3.3. Determination of the Efficiency of Conjugation
3.4. Simulation of In Vitro Endolysosomal Degradation Using Microsomes
3.5. Evaluation of the Changes in the Protein Structure
3.6. Determination of the Structural and Functional Integrity of T Cell Epitope by T Cell Activation Assay
3.7. Determination of the Structural and Functional Integrity of IgE Epitopes by Mediator Release Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yun, C.-H.; Cho, C.-S. Nanoparticles to improve the efficacy of vaccines. Pharmaceutics 2020, 12, 418. [Google Scholar] [CrossRef]
- Ilyinskii, P.O.; Roy, C.J.; O’Neil, C.P.; Browning, E.A.; Pittet, L.A.; Altreuter, D.H.; Alexis, F.; Tonti, E.; Shi, J.; Basto, P.A. Adjuvant-carrying synthetic vaccine particles augment the immune response to encapsulated antigen and exhibit strong local immune activation without inducing systemic cytokine release. Vaccine 2014, 32, 2882–2895. [Google Scholar] [CrossRef] [Green Version]
- Getts, D.R.; Shea, L.D.; Miller, S.D.; King, N.J. Harnessing nanoparticles for immune modulation. Trends Immunol. 2015, 36, 419–427. [Google Scholar] [CrossRef] [Green Version]
- Thakur, N.; Thakur, S.; Chatterjee, S.; Das, J.; Sil, P.C. Nanoparticles as smart carriers for enhanced cancer immunotherapy. Front. Chem. 2020, 8, 1217. [Google Scholar] [CrossRef]
- Perciani, C.T.; Liu, L.Y.; Wood, L.; MacParland, S.A. Enhancing immunity with nanomedicine: Employing nanoparticles to harness the immune system. ACS Nano 2020, 15, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.; Duschl, A.; Himly, M. Nanotechnology-based vaccines for allergen-specific immunotherapy: Potentials and challenges of conventional and novel adjuvants under research. Vaccines 2020, 8, 237. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, P.; Bhatia, E.; Sharma, S.; Ahamad, N.; Banerjee, R. Advancements in prophylactic and therapeutic nanovaccines. Acta Biomater. 2020, 108, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Zhang, F.; Ni, Q.; Niu, G.; Chen, X. Efficient nanovaccine delivery in cancer immunotherapy. ACS Nano 2017, 11, 2387–2392. [Google Scholar] [CrossRef] [PubMed]
- Hua, S.; De Matos, M.B.; Metselaar, J.M.; Storm, G. Current trends and challenges in the clinical translation of nanoparticulate nanomedicines: Pathways for translational development and commercialization. Front. Pharmacol. 2018, 9, 790. [Google Scholar] [CrossRef]
- Tinkle, S.; McNeil, S.E.; Mühlebach, S.; Bawa, R.; Borchard, G.; Barenholz, Y.; Tamarkin, L.; Desai, N. Nanomedicines: Addressing the scientific and regulatory gap. Ann. N. Y. Acad. Sci. 2014, 1313, 35–56. [Google Scholar] [CrossRef] [PubMed]
- De Mattia, F.; Chapsal, J.-M.; Descamps, J.; Halder, M.; Jarrett, N.; Kross, I.; Mortiaux, F.; Ponsar, C.; Redhead, K.; McKelvie, J. The consistency approach for quality control of vaccines–A strategy to improve quality control and implement 3rs. Biologicals 2011, 39, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Akkermans, A.; Chapsal, J.-M.; Coccia, E.M.; Depraetere, H.; Dierick, J.-F.; Duangkhae, P.; Goel, S.; Halder, M.; Hendriksen, C.; Levis, R. Animal testing for vaccines. Implementing replacement, reduction and refinement: Challenges and priorities. Biologicals 2020, 68, 92–107. [Google Scholar] [CrossRef]
- Liu, Y.; Hardie, J.; Zhang, X.; Rotello, V.M. Effects of Engineered Nanoparticles on the Innate Immune System; Seminars in Immunology; Elsevier: Amsterdam, The Netherlands, 2017; pp. 25–32. [Google Scholar]
- Ciabattini, A.; Nardini, C.; Santoro, F.; Garagnani, P.; Franceschi, C.; Medaglini, D. Vaccination in the Elderly: The Challenge of Immune Changes with Aging; Seminars in Immunology; Elsevier: Amsterdam, The Netherlands, 2018; pp. 83–94. [Google Scholar]
- Kang, S.-J.; Jung, S.I. Age-related morbidity and mortality among patients with covid-19. Infect. Chemother. 2020, 52, 154. [Google Scholar] [CrossRef] [PubMed]
- Nikolich-Žugich, J. The twilight of immunity: Emerging concepts in aging of the immune system. Nat. Immunol. 2018, 19, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Haynes, L.; Eaton, S.M.; Burns, E.M.; Rincon, M.; Swain, S.L. Inflammatory cytokines overcome age-related defects in cd4 t cell responses in vivo. J. Immunol. 2004, 172, 5194–5199. [Google Scholar] [CrossRef]
- Hamad-Schifferli, K. Exploiting the novel properties of protein coronas: Emerging applications in nanomedicine. Nanomedicine 2015, 10, 1663–1674. [Google Scholar] [CrossRef]
- Schmutz, M.; Borges, O.; Jesus, S.; Borchard, G.; Perale, G.; Zinn, M.; Sips, Ä.A.; Soeteman-Hernandez, L.G.; Wick, P.; Som, C. A methodological safe-by-design approach for the development of nanomedicines. Front. Bioeng. Biotechnol. 2020, 8, 258. [Google Scholar] [CrossRef]
- HogenEsch, H.; O’Hagan, D.T.; Fox, C.B. Optimizing the utilization of aluminum adjuvants in vaccines: You might just get what you want. NPJ Vaccines 2018, 3, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Willhite, C.C.; Karyakina, N.A.; Yokel, R.A.; Yenugadhati, N.; Wisniewski, T.M.; Arnold, I.M.; Momoli, F.; Krewski, D. Systematic review of potential health risks posed by pharmaceutical, occupational and consumer exposures to metallic and nanoscale aluminum, aluminum oxides, aluminum hydroxide and its soluble salts. Crit. Rev. Toxicol. 2014, 44, 1–80. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Tovar, G.; Palestino, G.; Rosales-Mendoza, S. An overview on the role of silica-based materials in vaccine development. Expert Rev. Vaccines 2016, 15, 1449–1462. [Google Scholar] [CrossRef]
- Kitzmüller, C.; Wallner, M.; Deifl, S.; Mutschlechner, S.; Walterskirchen, C.; Zlabinger, G.; Ferreira, F.; Bohle, B. A hypoallergenic variant of the major birch pollen allergen shows distinct characteristics in antigen processing and t-cell activation. Allergy 2012, 67, 1375–1382. [Google Scholar] [CrossRef] [PubMed]
- Wallner, M.; Himly, M.; Neubauer, A.; Erler, A.; Hauser, M.; Asam, C.; Mutschlechner, S.; Ebner, C.; Briza, P.; Ferreira, F. The influence of recombinant production on the immunologic behavior of birch pollen isoallergens. PLoS ONE 2009, 4, e8457. [Google Scholar] [CrossRef] [PubMed]
- Oyewumi, M.O.; Kumar, A.; Cui, Z. Nano-microparticles as immune adjuvants: Correlating particle sizes and the resultant immune responses. Expert Rev. Vaccines 2010, 9, 1095–1107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finnie, K.S.; Bartlett, J.R.; Barbé, C.J.; Kong, L. Formation of silica nanoparticles in microemulsions. Langmuir 2007, 23, 3017–3024. [Google Scholar] [CrossRef]
- Harris, J.R.; Soliakov, A.; Lewis, R.J.; Depoix, F.; Watkinson, A.; Lakey, J.H. Alhydrogel® adjuvant, ultrasonic dispersion and protein binding: A tem and analytical study. Micron 2012, 43, 192–200. [Google Scholar] [CrossRef]
- Shardlow, E.; Mold, M.; Exley, C. From stock bottle to vaccine: Elucidating the particle size distributions of aluminum adjuvants using dynamic light scattering. Front. Chem. 2017, 4, 48. [Google Scholar] [CrossRef]
- Huang, M.; Wang, W. Factors affecting alum–protein interactions. Int. J. Pharm. 2014, 466, 139–146. [Google Scholar] [CrossRef]
- Lee, J.G.; Lannigan, K.; Shelton, W.A.; Meissner, J.; Bharti, B. Adsorption of myoglobin and corona formation on silica nanoparticles. Langmuir 2020, 36, 14157–14165. [Google Scholar] [CrossRef]
- Kubiak-Ossowska, K.; Tokarczyk, K.; Jachimska, B.; Mulheran, P.A. Bovine serum albumin adsorption at a silica surface explored by simulation and experiment. J. Phys. Chem. B 2017, 121, 3975–3986. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Himly, M.; Nony, E.; Chabre, H.; Van Overtvelt, L.; Neubauer, A.; Van Ree, R.; Buchheit, K.H.; Vieths, S.; Moingeon, P.; Ferreira, F. Standardization of allergen products: 1. Detailed characterization of gmp-produced recombinant bet v 1.0101 as biological reference preparation. Allergy 2009, 64, 1038–1045. [Google Scholar] [CrossRef]
- Kubiak-Ossowska, K.; Jachimska, B.; Mulheran, P.A. How negatively charged proteins adsorb to negatively charged surfaces: A molecular dynamics study of bsa adsorption on silica. J. Phys. Chem. B 2016, 120, 10463–10468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Henzler, K.; Haupt, B.R.; Lauterbach, K.; Wittemann, A.; Borisov, O.; Ballauff, M. Adsorption of β-lactoglobulin on spherical polyelectrolyte brushes: Direct proof of counterion release by isothermal titration calorimetry. J. Am. Chem. Soc. 2010, 132, 3159–3163. [Google Scholar] [CrossRef] [PubMed]
- Machado, Y.; Freier, R.; Scheiblhofer, S.; Thalhamer, T.; Mayr, M.; Briza, P.; Grutsch, S.; Ahammer, L.; Fuchs, J.E.; Wallnoefer, H.G. Fold stability during endolysosomal acidification is a key factor for allergenicity and immunogenicity of the major birch pollen allergen. J. Allergy Clin. Immunol. 2016, 137, 1525–1534. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scheiblhofer, S.; Laimer, J.; Machado, Y.; Weiss, R.; Thalhamer, J. Influence of protein fold stability on immunogenicity and its implications for vaccine design. Expert Rev. Vaccines 2017, 16, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Jürets, A.; Wallner, M.; Briza, P.; Ruzek, S.; Hainzl, S.; Pichler, U.; Kitzmüller, C.; Bohle, B.; Huber, C.G. Assessing protein immunogenicity with a dendritic cell line-derived endolysosomal degradome. PLoS ONE 2011, 6, e17278. [Google Scholar] [CrossRef] [Green Version]
- Jahn-Schmid, B.; Radakovics, A.; Lüttkopf, D.; Scheurer, S.; Vieths, S.; Ebner, C.; Bohle, B. Bet v 1142-156 is the dominant t-cell epitope of the major birch pollen allergen and important for cross-reactivity with bet v 1–related food allergens. J. Allergy Clin. Immunol. 2005, 116, 213–219. [Google Scholar] [CrossRef]
- Winter, P.; Stubenvoll, S.; Scheiblhofer, S.; Joubert, I.A.; Strasser, L.; Briganser, C.; Soh, W.T.; Hofer, F.; Kamenik, A.S.; Dietrich, V. In silico design of phl p 6 variants with altered folding stability significantly impacts antigen processing, immunogenicity and immune polarization. bioRxiv 2020, 11, 1824. [Google Scholar]
- Kamenik, A.S.; Hofer, F.; Handle, P.H.; Liedl, K.R. Dynamics rationalize proteolytic susceptibility of the major birch pollen allergen bet v 1. Front. Mol. Biosci. 2020, 7, 18. [Google Scholar] [CrossRef] [Green Version]
- Freier, R.; Dall, E.; Brandstetter, H. Protease recognition sites in bet v 1a are cryptic, explaining its slow processing relevant to its allergenicity. Sci. Rep. 2015, 5, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Kofler, S.; Asam, C.; Eckhard, U.; Wallner, M.; Ferreira, F.; Brandstetter, H. Crystallographically mapped ligand binding differs in high and low ige binding isoforms of birch pollen allergen bet v 1. J. Mol. Biol. 2012, 422, 109–123. [Google Scholar] [CrossRef] [Green Version]
- Kishore, D.; Kundu, S.; Kayastha, A.M. Thermal, chemical and ph induced denaturation of a multimeric β-galactosidase reveals multiple unfolding pathways. PLoS ONE 2012, 7, e50380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thalhamer, T.; Dobias, H.; Stepanoska, T.; Pröll, M.; Stutz, H.; Dissertori, O.; Lackner, P.; Ferreira, F.; Wallner, M.; Thalhamer, J. Designing hypoallergenic derivatives for allergy treatment by means of in silico mutation and screening. J. Allergy Clin. Immunol. 2010, 125, 926–934.e910. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, F.; Ebner, C.; Kramer, B.; Casari, G.; Briza, P.; Kungl, A.J.; Grimm, R.; Jahn-schmid, B.; Breiteneder, H.; Kraft, D. Modulation of ige reactivity of allergens by site-directed mutagenesis: Potential use of hypoallergenic variants for immunotherapy. FASEB J. 1998, 12, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Aglas, L.; Bethanis, A.; Chrusciel, P.; Stolz, F.; Gruen, M.; Jaakkola, U.-M.; Jongejan, L.; Yatkin, E.; Van Ree, R. In vivo induction of functional inhibitory igg antibodies by a hypoallergenic bet v 1 variant. Front. Immunol. 2020, 11, 2118. [Google Scholar] [CrossRef] [PubMed]
- Spangfort, M.D.; Mirza, O.; Ipsen, H.; Van Neerven, R.J.; Gajhede, M.; Larsen, J.N. Dominating ige-binding epitope of bet v 1, the major allergen of birch pollen, characterized by x-ray crystallography and site-directed mutagenesis. J. Immunol. 2003, 171, 3084–3090. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mirza, O.; Henriksen, A.; Ipsen, H.; Larsen, J.N.; Wissenbach, M.; Spangfort, M.D.; Gajhede, M. Dominant epitopes and allergic cross-reactivity: Complex formation between a fab fragment of a monoclonal murine igg antibody and the major allergen from birch pollen bet v 1. J. Immunol. 2000, 165, 331–338. [Google Scholar] [CrossRef] [Green Version]
- Malik, M.A.; Wani, M.Y.; Hashim, M.A. Microemulsion method: A novel route to synthesize organic and inorganic nanomaterials: 1st nano update. Arab. J. Chem. 2012, 5, 397–417. [Google Scholar] [CrossRef] [Green Version]
- Wallner, M.; Hauser, M.; Himly, M.; Zaborsky, N.; Mutschlechner, S.; Harrer, A.; Asam, C.; Pichler, U.; van Ree, R.; Briza, P. Reshaping the bet v 1 fold modulates th polarization. J. Allergy Clin. Immunol. 2011, 127, 1571–1578.e1579. [Google Scholar] [CrossRef] [Green Version]
- Soh, W.T.; Aglas, L.; Mueller, G.A.; Gilles, S.; Weiss, R.; Scheiblhofer, S.; Huber, S.; Scheidt, T.; Thompson, P.M.; Briza, P. Multiple roles of bet v 1 ligands in allergen stabilization and modulation of endosomal protease activity. Allergy 2019, 74, 2382–2393. [Google Scholar] [CrossRef] [Green Version]
- Kavan, D.; Man, P. Mstools—Web based application for visualization and presentation of hxms data. Int. J. Mass Spectrom. 2011, 302, 53–58. [Google Scholar] [CrossRef]
- Micsonai, A.; Wien, F.; Bulyáki, É.; Kun, J.; Moussong, É.; Lee, Y.-H.; Goto, Y.; Réfrégiers, M.; Kardos, J. Bestsel: A web server for accurate protein secondary structure prediction and fold recognition from the circular dichroism spectra. Nucleic Acids Res. 2018, 46, W315–W322. [Google Scholar] [CrossRef] [PubMed]
- Vogel, L.; Lüttkopf, D.; Hatahet, L.; Haustein, D.; Vieths, S. Development of a functional in vitro assay as a novel tool for the standardization of allergen extracts in the human system. Allergy 2005, 60, 1021–1028. [Google Scholar] [CrossRef] [PubMed]







| Sample | Technique | Number Mean Diameter (nm) | PDI | Size (Z Average) (nm) | Zeta Potential (mV) |
|---|---|---|---|---|---|
| SiO2 NPs | DLS | 100.3 ± 3.4 | 0.025 | 120.2 ± 1.2 | −38.9 ± 2.8 |
| SiO2 NPs | NTA | 102.4 ± 39.3 | - | - | - |
| Alhydrogel® | DLS | 585.9 ± 174.2 | 0.345 | 1082.0 ± 63.4 | +18.0 ± 1.5 |
| Sample | Zeta Potential (mV) |
|---|---|
| SiO2 NPs | −38.9 ± 2.8 |
| SiO2 NPs + Bet v 1 | −25.7 ± 5.8 |
| SiO2 NPs + BM4 | −19.2 ± 5.5 |
| Alhydrogel® | +18.0 ± 1.5 |
| Alhydrogel® + Bet v 1 | +31.2 ± 1.2 |
| Alhydrogel® + BM4 | +32.3 ± 1.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnson, L.; Aglas, L.; Soh, W.T.; Geppert, M.; Hofer, S.; Hofstätter, N.; Briza, P.; Ferreira, F.; Weiss, R.; Brandstetter, H.; et al. Structural Alterations of Antigens at the Material Interface: An Early Decision Toolbox Facilitating Safe-by-Design Nanovaccine Development. Int. J. Mol. Sci. 2021, 22, 10895. https://doi.org/10.3390/ijms221910895
Johnson L, Aglas L, Soh WT, Geppert M, Hofer S, Hofstätter N, Briza P, Ferreira F, Weiss R, Brandstetter H, et al. Structural Alterations of Antigens at the Material Interface: An Early Decision Toolbox Facilitating Safe-by-Design Nanovaccine Development. International Journal of Molecular Sciences. 2021; 22(19):10895. https://doi.org/10.3390/ijms221910895
Chicago/Turabian StyleJohnson, Litty, Lorenz Aglas, Wai Tuck Soh, Mark Geppert, Sabine Hofer, Norbert Hofstätter, Peter Briza, Fatima Ferreira, Richard Weiss, Hans Brandstetter, and et al. 2021. "Structural Alterations of Antigens at the Material Interface: An Early Decision Toolbox Facilitating Safe-by-Design Nanovaccine Development" International Journal of Molecular Sciences 22, no. 19: 10895. https://doi.org/10.3390/ijms221910895
APA StyleJohnson, L., Aglas, L., Soh, W. T., Geppert, M., Hofer, S., Hofstätter, N., Briza, P., Ferreira, F., Weiss, R., Brandstetter, H., Duschl, A., & Himly, M. (2021). Structural Alterations of Antigens at the Material Interface: An Early Decision Toolbox Facilitating Safe-by-Design Nanovaccine Development. International Journal of Molecular Sciences, 22(19), 10895. https://doi.org/10.3390/ijms221910895