The Association of Ovarian Teratoma and Anti-N-Methyl-D-Aspartate Receptor Encephalitis: An Updated Integrative Review
Abstract
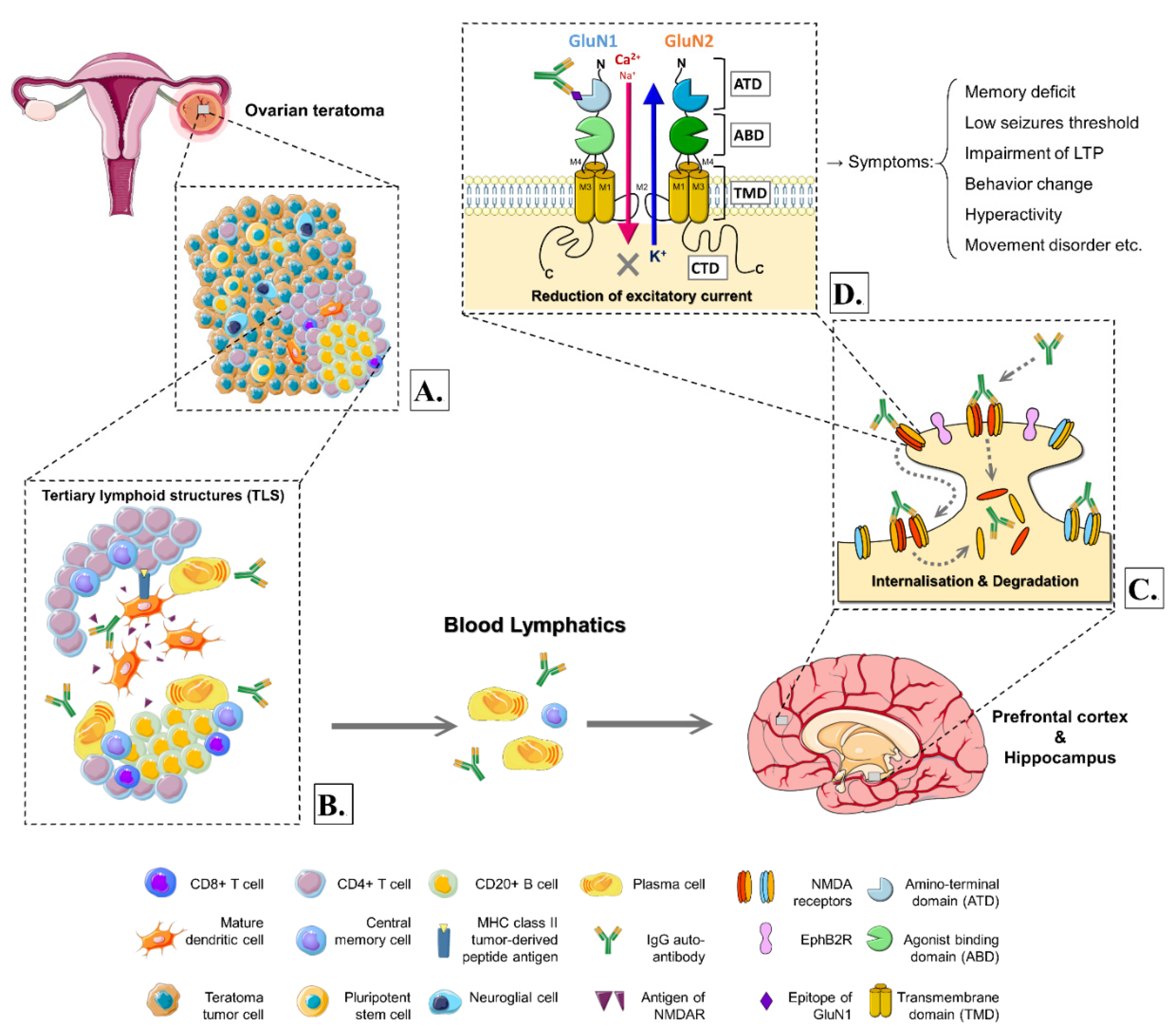
:1. Introduction
1.1. Ovarian Teratoma
1.2. Paraneoplastic Neurological Syndrome (PNS) and Anti-N-Methyl-D-Aspartate Receptor (Anti-NMDAR) Encephalitis
2. The Association of Anti-NMDAR Encephalitis and Ovarian Teratoma
3. The Hypothetical Mechanisms and Models of the Pathogenesis
3.1. The Triggers and Peculiar Cell Composition in the Ovarian Teratomas with Anti-NMDAR Encephalitis
3.2. The Microenvironment Involving in Ovarian Teratomas with Anti-NMDAR Encephalitis
3.3. The Association with Teratoma-Related Anti-NMDAR Encephalitis and Blood–Brain Barrier Integrity
3.4. The Molecular Basis for Structures and Physiological Features of NMDAR
3.5. The Role of NMDARs in Ovarian Teratomas with Anti-NMDAR Encephalitis
3.6. The Hypothetical Mechanism of the Pathogenesis and Evidence of Animal Models
4. Treatment and Detection for Patients with Ovarian Teratomas and Anti-NMDAR Encephalitis
5. Outcome and Associations of Ovarian Teratoma-Related Anti-NMDAR Encephalitis
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kraggerud, S.M.; Hoei-Hansen, C.E.; Alagaratnam, S.; Skotheim, R.I.; Abeler, V.M.; Meyts, E.R.-D.; Lothe, R.A. Molecular Characteristics of Malignant Ovarian Germ Cell Tumors and Comparison with Testicular Counterparts: Implications for Pathogenesis. Endocr. Rev. 2013, 34, 339–376. [Google Scholar] [CrossRef]
- Ovarian Germ cCell Tumors: Pathology, Epidemiology, Clinical Manifestations, and Diagnosis. Available online: https://www.uptodate.com/contents/ovarian-germ-cell-tumors-pathology-epidemiology-clinical-manifestations-and-diagnosis (accessed on 27 September 2021).
- Cabral, F.C.; Krajewski, K.M.; Rosenthal, M.H.; Hirsch, M.S.; Howard, S.A. Teratoma with malignant transformation: Report of three cases and review of the literature. Clin. Imaging 2014, 38, 589–593. [Google Scholar] [CrossRef]
- Berney, D.M.; Stoneham, S.; Arora, R.; Shamash, J.; Lockley, M. Ovarian germ cell tumour classification: Views from the testis. Histopathology 2019, 76, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Euscher, E.D. Germ Cell Tumors of the Female Genital Tract. Surg. Pathol. Clin. 2019, 12, 621–649. [Google Scholar] [CrossRef] [PubMed]
- Nogales, F.F.; Dulcey, I.; Preda, O. Germ Cell Tumors of the Ovary: An Update. Arch. Pathol. Lab. Med. 2014, 138, 351–362. [Google Scholar] [CrossRef] [Green Version]
- Ayhan, A.; Bukulmez, O.; Genc, C.; Karamursel, B.S.; Ayhan, A. Mature cystic teratomas of the ovary: Case series from one institution over 34 years. Eur. J. Obstet. Gynecol. Reprod. Biol. 2000, 88, 153–157. [Google Scholar] [CrossRef]
- Pekar-Zlotin, M.; Rabinovich, I.; Goldrat, I.; Vaknin, Z.; Gidoni, Y.; Zur-Naaman, H.; Maymon, R.; Smorgick, N. Ovarian Dermoid Cysts Associated with Paraneoplastic Syndrome N-methyl-D-aspartic Acid Receptor Antibodies Encephalitis. J. Minim. Invasive Gynecol. 2021, 28, 1190–1193. [Google Scholar] [CrossRef]
- Westhoff, C.; Pike, M.; Vessey, M. Benign ovarian teratomas: A population-based case-control study. Br. J. Cancer 1988, 58, 93–98. [Google Scholar] [CrossRef] [Green Version]
- Comerci, J.T.; Licciardi, F.; Bergh, P.A.; Gregori, C.; Breen, J.L. Mature cystic teratoma: A clinicopathologic evaluation of 517 cases and review of the literature. Obstet. Gynecol. 1994, 84, 22–28. [Google Scholar] [PubMed]
- Singh, P.; Yordan, E.L.; Wilbanks, G.D.; Miller, A.W.; Wee, A. Malignancy associated with benign cystic teratomas (dermoid cysts) of the ovary. Singap. Med. J. 1988, 29, 30–34. [Google Scholar]
- Smith, H.O.; Berwick, M.; Verschraegen, C.F.; Wiggins, C.; Lansing, L.; Muller, C.Y.; Qualls, C.R. Incidence and Survival Rates for Female Malignant Germ Cell Tumors. Obstet. Gynecol. 2006, 107, 1075–1085. [Google Scholar] [CrossRef] [PubMed]
- Hackethal, A.; Brueggmann, D.; Bohlmann, M.K.; Franke, F.E.; Tinneberg, H.-R.; Münstedt, K. Squamous-cell carcinoma in mature cystic teratoma of the ovary: Systematic review and analysis of published data. Lancet Oncol. 2008, 9, 1173–1180. [Google Scholar] [CrossRef]
- Dos Santos, L.; Mok, E.; Iasonos, A.; Park, K.; Soslow, R.; Aghajanian, C.; Alektiar, K.; Barakat, R.R.; Abu-Rustum, N.R. Squamous cell carcinoma arising in mature cystic teratoma of the ovary: A case series and review of the literature. Gynecol. Oncol. 2007, 105, 321–324. [Google Scholar] [CrossRef]
- Kato, N.; Sakamoto, K.; Murakami, K.; Iwasaki, Y.; Kamataki, A.; Kurose, A. Genetic zygosity of mature ovarian teratomas, struma ovarii, and ovarian carcinoids. Virchows Archiv 2018, 473, 177–182. [Google Scholar] [CrossRef]
- Soga, J.; Osaka, M.; Yakuwa, Y. Carcinoids of the ovary: An analysis of 329 reported cases. J. Exp. Clin. Cancer Res. 2000, 19, 271–280. [Google Scholar]
- Zaborowski, M.P.; Spaczynski, M.; Nowak-Markwitz, E.; Michalak, S. Paraneoplastic neurological syndromes associated with ovarian tumors. J. Cancer Res. Clin. Oncol. 2015, 141, 99–108. [Google Scholar] [CrossRef] [Green Version]
- Höftberger, R.; Rosenfeld, M.; Dalmau, J. Update on neurological paraneoplastic syndromes. Curr. Opin. Oncol. 2015, 27, 489–495. [Google Scholar] [CrossRef]
- Overview of Paraneoplastic Syndromes of the Nervous System. Available online: https://www.uptodate.com/contents/overview-of-paraneoplastic-syndromes-of-the-nervous-system (accessed on 27 September 2021).
- Iorio, R.; Spagni, G.; Masi, G. Paraneoplastic neurological syndromes. Semin. Diagn. Pathol. 2019, 36, 279–292. [Google Scholar] [CrossRef] [PubMed]
- Graus, F.; Delattre, J.Y.; Antoine, J.C.; Dalmau, J.; Giometto, B.; Grisold, W.; Honnorat, J.; Smitt, P.S.; Vedeler, C.; Verschuuren, J.; et al. Recommended diagnostic criteria for paraneoplastic neurological syndromes. J. Neurol. Neurosurg. Psychiatry 2004, 75, 1135–1140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Höftberger, R.; Lassmann, H. Immune-mediated disorders. Handb. Clin. Neurol. 2018, 145, 285–299. [Google Scholar] [CrossRef]
- Adam, J.K.; Odhav, B.; Bhoola, K.D. Immune responses in cancer. Pharmacol. Ther. 2003, 99, 113–132. [Google Scholar] [CrossRef]
- Dalmau, J.; Geis, C.; Graus, F. Autoantibodies to Synaptic Receptors and Neuronal Cell Surface Proteins in Autoimmune Diseases of the Central Nervous System. Physiol. Rev. 2017, 97, 839–887. [Google Scholar] [CrossRef]
- Mueller, S.H.; Färber, A.; Prüss, H.; Melzer, N.; Golombeck, K.S.; Kümpfel, T.; Thaler, F.; Elisak, M.; Lewerenz, J.; Kaufmann, M.; et al. Genetic predisposition in anti-LGI1 and anti-NMDA receptor encephalitis. Ann. Neurol. 2018, 83, 863–869. [Google Scholar] [CrossRef]
- Dalmau, J.; Gleichman, A.J.; Hughes, E.G.; Rossi, J.E.; Peng, X.; Lai, M.; Dessain, S.K.; Rosenfeld, M.R.; Balice-Gordon, R.; Lynch, D.R. Anti-NMDA-receptor encephalitis: Case series and analysis of the effects of antibodies. Lancet Neurol. 2008, 7, 1091–1098. [Google Scholar] [CrossRef] [Green Version]
- Dalmau, J.; Armangue, T.; Planagumà, J.; Radosevic, M.; Mannara, F.; Leypoldt, F.; Geis, C.; Lancaster, E.; Titulaer, M.J.; Rosenfeld, M.R.; et al. An update on anti-NMDA receptor encephalitis for neurologists and psychiatrists: Mechanisms and models. Lancet Neurol. 2019, 18, 1045–1057. [Google Scholar] [CrossRef]
- Paraneoplastic and Autoimmune Encephalitis. Available online: https://www.uptodate.com/contents/paraneoplastic-and-autoimmune-encephalitis (accessed on 27 September 2021).
- Dalmau, J.; Tüzün, E.; Wu, H.-Y.; Masjuan, J.; Ba, J.E.R.; Voloschin, A.; Baehring, J.M.; Shimazaki, H.; Koide, R.; King, D.; et al. Paraneoplastic anti-N-methyl-D-aspartate receptor encephalitis associated with ovarian teratoma. Ann. Neurol. 2007, 61, 25–36. [Google Scholar] [CrossRef] [Green Version]
- Dai, Y.; Zhang, J.; Ren, H.; Zhou, X.; Chen, J.; Cui, L.; Lang, J.; Guan, H.; Sun, D. Surgical outcomes in patients with anti-N-methyl D-aspartate receptor encephalitis with ovarian teratoma. Am. J. Obstet. Gynecol. 2019, 221, 485.e1–485.e10. [Google Scholar] [CrossRef]
- Florance, N.R.; Ba, R.L.D.; Lam, C.; Szperka, C.; Zhou, L.; Ahmad, S.; Campen, C.J.; Moss, H.; Peter, N.; Bs, A.J.G.; et al. Anti-N-methyl-D-aspartate receptor (NMDAR) encephalitis in children and adolescents. Ann. Neurol. 2009, 66, 11–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Titulaer, M.; McCracken, L.; Gabilondo, I.; Armangue, T.; Glaser, C.; Iizuka, T.; Honig, L.S.; Benseler, S.M.; Kawachi, I.; Martinez-Hernandez, E.; et al. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: An observational cohort study. Lancet Neurol. 2013, 12, 157–165. [Google Scholar] [CrossRef] [Green Version]
- Bost, C.; Chanson, E.; Picard, G.; Meyronet, D.; Mayeur, M.-E.; Ducray, F.; Rogemond, V.; Psimaras, D.; Antoine, J.-C.; Delattre, J.-Y.; et al. Malignant tumors in autoimmune encephalitis with anti-NMDA receptor antibodies. J. Neurol. 2018, 265, 2190–2200. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Lu, Y.; Xu, L.; Liu, L.; Wu, X.; Zhang, Y.; Zhu, G.; Hong, Z. Anti-N-methyl-D-aspartate receptor encephalitis with accompanying ovarian teratoma in female patients from East China: Clinical features, treatment, and prognostic outcomes. Seizure 2020, 75, 55–62. [Google Scholar] [CrossRef]
- Yaguchi, H.; Tsuji, T.; Yabe, I.; Hirayama, E.; Nomura, T.; Ohashi, I.; Mito, Y.; Tanaka, K.; Tajima, Y. Incidence of anti-NMDAR encephalitis in patients undergoing resection of ovarian teratoma in a single institution. J. Neurol. Sci. 2020, 409, 116608. [Google Scholar] [CrossRef]
- Xu, X.; Lu, Q.; Huang, Y.; Fan, S.; Zhou, L.; Yuan, J.; Yang, X.; Ren, H.; Sun, D.; Dai, Y.; et al. Anti-NMDAR encephalitis. Neurol. Neuroimmunol. Neuroinflammation 2020, 7, e633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Acién, P.; Acién, M.; Ruiz-Maciá, E.; Martín-Estefanía, C. Ovarian teratoma-associated anti-NMDAR encephalitis: A systematic review of reported cases. Orphanet J. Rare Dis. 2014, 9, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiu, H.-C.; Su, Y.-C.; Huang, S.-C.; Chiang, H.-L.; Huang, P.-S. Anti-NMDAR encephalitis with ovarian teratomas: Review of the literature and two case reports. Taiwan. J. Obstet. Gynecol. 2019, 58, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Yan, B.; Wang, Y.; Zhang, Y.; Lou, W. Teratoma-associated anti-N-methyl-D-aspartate receptor encephalitis. Medicine 2019, 98, e15765. [Google Scholar] [CrossRef]
- Yu, M.; Li, S.; Cheng, J.; Zhou, L.; Jiang, Z.; Di, W. Ovarian teratoma-associated anti-NMDAR encephalitis: A single-institute series of six patients from China. Arch. Gynecol. Obstet. 2021, 303, 1283–1294. [Google Scholar] [CrossRef]
- Ahmad, J.; Sohail, M.S.; Khan, A.; Qavi, A.H.; Gaudel, P.; Zahid, M.; Assad, S. Anti-n-Methyl-d-Aspartate-Receptor (NMDAR) Encephalitis in Association with Ovarian Teratoma. Cureus 2017, 9, 1425. [Google Scholar] [CrossRef] [Green Version]
- Omata, T.; Kodama, K.; Watanabe, Y.; Iida, Y.; Furusawa, Y.; Takashima, A.; Takahashi, Y.; Sakuma, H.; Tanaka, K.; Fujii, K.; et al. Ovarian teratoma development after anti-NMDA receptor encephalitis treatment. Brain Dev. 2017, 39, 448–451. [Google Scholar] [CrossRef]
- Mitra, A.D.; Afifya, A. Ovarian teratoma associated Anti N-methyl-D-aspartate receptor encephalitis: A difficult diagnosis with a favorable prognosis. Autops. Case Rep. 2018, 8, e2018019. [Google Scholar] [CrossRef] [Green Version]
- Lwin, S.; Yi, M.S.; Mardiana, K.; Woon, S.Y.; Nwe, T.M. Ovarian teratoma-associated anti-NMDAR encephalitis in a 12-year-old girl. Med. J. Malaysia 2020, 75, 731–733. [Google Scholar]
- Lee, C.H.; Kim, E.J.; Lee, M.H.; Yim, G.W.; Kim, K.J.; Kim, K.K.; Kim, E.J.; Roh, J.-W. Anti-N-methyl-D-aspartate Receptor Encephalitis: A Rare Complication of Ovarian Teratoma. J. Korean Med. Sci. 2020, 35, 207. [Google Scholar] [CrossRef]
- Chernyshkova, I.; Estefan, B.; Hoque, R.; Lee, A. Neurologic Presentation of Probable Seronegative Paraneoplastic Encephalitis in a Woman with an Ovarian Teratoma. Cureus 2020, 12, e8485. [Google Scholar] [CrossRef]
- Li, W.; Jia, D.; Tong, L.; Lun, Z.; Li, H. Anti-N-methyl-d-aspartate receptor encephalitis induced by bilateral ovarian teratomas with distinct histopathologic types. Medicine 2019, 98, e18148. [Google Scholar] [CrossRef] [PubMed]
- Iemura, Y.; Yamada, Y.; Hirata, M.; Kataoka, T.R.; Minamiguchi, S.; Haga, H. Histopathological characterization of the neuroglial tissue in ovarian teratoma associated with anti-N-methyl-D-aspartate (NMDA) receptor encephalitis. Pathol. Int. 2018, 68, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Chefdeville, A.; Treilleux, I.; Mayeur, M.-E.; Couillault, C.; Picard, G.; Bost, C.; Mokhtari, K.; Vasiljevic, A.; Meyronet, D.; Rogemond, V.; et al. Immunopathological characterization of ovarian teratomas associated with anti-N-methyl-D-aspartate receptor encephalitis. Acta Neuropathol. Commun. 2019, 7, 38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Broderick, J.P.; Adeoye, O.; Elm, J. Evolution of the Modified Rankin Scale and Its Use in Future Stroke Trials. Stroke 2017, 48, 2007–2012. [Google Scholar] [CrossRef]
- Gabilondo, I.; Saiz, A.; Galan, L.; Gonzalez, V.; Jadraque, R.; Sabater, L.; Sans, A.; Sempere, A.; Vela, A.; Villalobos, F.; et al. Analysis of relapses in anti-NMDAR encephalitis. Neurology 2011, 77, 996–999. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.-Y.; Lei, S.; Zhang, L.; Liu, X.; Lin, M.-T.; Blumcke, I.; Piao, Y.-S.; Zhou, D.; Li, J.-M. Co-expression of NMDA-receptor subunits NR1, NR2A, and NR2B in dysplastic neurons of teratomas in patients with paraneoplastic NMDA-receptor-encephalitis: A retrospective clinico-pathology study of 159 patients. Acta Neuropathol. Commun. 2020, 8, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Nosadini, M.; Mohammad, S.; Corazza, F.; Ruga, E.M.; Kothur, K.; Perilongo, G.; Frigo, A.C.; Toldo, I.; Dale, R.C.; Sartori, S. Herpes simplex virus-induced anti-N-methyl-d-aspartate receptor encephalitis: A systematic literature review with analysis of 43 cases. Dev. Med. Child. Neurol. 2017, 59, 796–805. [Google Scholar] [CrossRef] [Green Version]
- Lynch, D.R.; Rattelle, A.; Na Dong, Y.; Roslin, K.; Gleichman, A.J.; Panzer, J.A. Anti-NMDA Receptor Encephalitis: Clinical Features and Basic Mechanisms. Adv. Pharmacol. 2018, 82, 235–260. [Google Scholar] [CrossRef]
- Damjanov, I.; Andrews, P.W. Teratomas produced from human pluripotent stem cells xenografted into immunodeficient mice—A histopathology atlas. Int. J. Dev. Biol. 2016, 60, 337–419. [Google Scholar] [CrossRef] [Green Version]
- Nolan, A.; Buza, N.; Margeta, M.; Rabban, J.T. Ovarian Teratomas in Women with Anti-N-methyl-D-Aspartate Receptor Encephalitis. Am. J. Surg. Pathol. 2019, 43, 949–964. [Google Scholar] [CrossRef]
- Makuch, M.; Wilson, R.; Al-Diwani, A.; Varley, J.; Kienzler, A.-K.; Taylor, J.; Berretta, A.; Fowler, D.; Lennox, B.; Leite, M.I.; et al. N-methyl-D-aspartate receptor antibody production from germinal center reactions: Therapeutic implications. Ann. Neurol. 2018, 83, 553–561. [Google Scholar] [CrossRef]
- Sautès-Fridman, C.; Petitprez, F.; Calderaro, J.; Fridman, W.H. Tertiary lymphoid structures in the era of cancer immunotherapy. Nat. Rev. Cancer 2019, 19, 307–325. [Google Scholar] [CrossRef]
- Bien, C.G.; Vincent, A.; Barnett, M.H.; Becker, A.J.; Blümcke, I.; Graus, F.; Jellinger, K.A.; Reuss, D.E.; Ribalta, T.; Schlegel, J.; et al. Immunopathology of autoantibody-associated encephalitides: Clues for pathogenesis. Brain 2012, 135, 1622–1638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martinez-Hernandez, E.; Horvath, J.; Shiloh-Malawsky, Y.; Sangha, N.; Martinez-Lage, M.; Dalmau, J. Analysis of complement and plasma cells in the brain of patients with anti-NMDAR encephalitis. Neurology 2011, 77, 589–593. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gleichman, A.J.; Spruce, L.A.; Dalmau, J.; Seeholzer, S.H.; Lynch, D.R. Anti-NMDA Receptor Encephalitis Antibody Binding Is Dependent on Amino Acid Identity of a Small Region within the GluN1 Amino Terminal Domain. J. Neurosci. 2012, 32, 11082–11094. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kunchok, A.; Zekeridou, A.; McKeon, A. Autoimmune glial fibrillary acidic protein astrocytopathy. Curr. Opin. Neurol. 2019, 32, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Ding, Y.; Zheng, D.; Wang, Z.; Pan, S.; Ji, T.; Shen, H.-Y.; Wang, H. Elevation of YKL-40 in the CSF of Anti-NMDAR Encephalitis Patients Is Associated with Poor Prognosis. Front. Neurol. 2018, 9, 727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, Y.-W.; Pan, S.-Y.; Xie, W.; Shen, H.-Y.; Wang, H.-H. Elevated Soluble Fas and FasL in Cerebrospinal Fluid and Serum of Patients with Anti-N-methyl-D-aspartate Receptor Encephalitis. Front. Neurol. 2018, 9, 904. [Google Scholar] [CrossRef] [Green Version]
- Yu, Y.; Wu, Y.; Cao, X.; Li, J.; Liao, X.; Wei, J.; Huang, W. The Clinical Features and Prognosis of Anti-NMDAR Encephalitis Depends on Blood Brain Barrier Integrity. Mult. Scler. Relat. Disord. 2021, 47, 102604. [Google Scholar] [CrossRef]
- Shiomi, A.; Usui, T. Pivotal Roles of GM-CSF in Autoimmunity and Inflammation. Mediat. Inflamm. 2015, 2015, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Wesselingh, R.; Butzkueven, H.; Buzzard, K.; Tarlinton, D.; O’Brien, T.; Monif, M. Innate Immunity in the Central Nervous System: A Missing Piece of the Autoimmune Encephalitis Puzzle? Front. Immunol. 2019, 10, 2066. [Google Scholar] [CrossRef] [PubMed]
- Aubé, B.; Lévesque, S.A.; Paré, A.; Chamma, É.; Kébir, H.; Gorina, R.; Lécuyer, M.-A.; Alvarez, J.I.; De Koninck, Y.; Engelhardt, B.; et al. Neutrophils Mediate Blood–Spinal Cord Barrier Disruption in Demyelinating Neuroinflammatory Diseases. J. Immunol. 2014, 193, 2438–2454. [Google Scholar] [CrossRef] [Green Version]
- Galli, J.; Clardy, S.; Piquet, A.L. NMDAR Encephalitis Following Herpes Simplex Virus Encephalitis. Curr. Infect. Dis. Rep. 2017, 19, 1. [Google Scholar] [CrossRef] [PubMed]
- Armangue, T.; Spatola, M.; Vlagea, A.; Mattozzi, S.; Cárceles-Cordon, M.; Martinez-Heras, E.; Llufriu, S.; Muchart, J.; Erro, M.E.; Abraira, L.; et al. Frequency, symptoms, risk factors, and outcomes of autoimmune encephalitis after herpes simplex encephalitis: A prospective observational study and retrospective analysis. Lancet Neurol. 2018, 17, 760–772. [Google Scholar] [CrossRef] [Green Version]
- Van Sonderen, A.; Arends, S.; Tavy, D.L.J.; Bastiaansen, A.; Bruijn, M.A.A.M.D.; Schreurs, M.W.J.; Smitt, P.A.E.S.; Titulaer, M.J. Predictive value of electroencephalography in anti-NMDA receptor encephalitis. J. Neurol. Neurosurg. Psychiatry 2018, 89, 1101–1106. [Google Scholar] [CrossRef]
- Paoletti, P.; Bellone, C.; Zhou, Q. NMDA receptor subunit diversity: Impact on receptor properties, synaptic plasticity and disease. Nat. Rev. Neurosci. 2013, 14, 383–400. [Google Scholar] [CrossRef] [PubMed]
- Franchini, L.; Carrano, N.; Di Luca, M.; Gardoni, F. Synaptic GluN2A-Containing NMDA Receptors: From Physiology to Pathological Synaptic Plasticity. Int. J. Mol. Sci. 2020, 21, 1538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hansen, K.B.; Yi, F.; Perszyk, R.; Furukawa, H.; Wollmuth, L.P.; Gibb, A.; Traynelis, S.F. Structure, function, and allosteric modulation of NMDA receptors. J. Gen. Physiol. 2018, 150, 1081–1105. [Google Scholar] [CrossRef]
- Nevian, T. Single Spine Ca2+ Signals Evoked by Coincident EPSPs and Backpropagating Action Potentials in Spiny Stellate Cells of Layer 4 in the Juvenile Rat Somatosensory Barrel Cortex. J. Neurosci. 2004, 24, 1689–1699. [Google Scholar] [CrossRef] [Green Version]
- Volianskis, A.; France, G.; Jensen, M.S.; Bortolotto, Z.A.; Jane, D.E.; Collingridge, G.L. Long-term potentiation and the role of N -methyl- d -aspartate receptors. Brain Res. 2015, 1621, 5–16. [Google Scholar] [CrossRef] [Green Version]
- Morris, R.G. NMDA receptors and memory encoding. Neuropharmacology 2013, 74, 32–40. [Google Scholar] [CrossRef]
- Mikasova, L.; De Rossi, P.; Bouchet, D.; Georges, F.; Rogemond, V.; Didelot, A.; Meissirel, C.; Honnorat, J.; Groc, L. Disrupted surface cross-talk between NMDA and Ephrin-B2 receptors in anti-NMDA encephalitis. Brain 2012, 135, 1606–1621. [Google Scholar] [CrossRef] [Green Version]
- Nolt, M.J.; Lin, Y.; Hruska, M.; Murphy, J.; Sheffler-Colins, S.I.; Kayser, M.S.; Passer, J.; Bennett, M.V.L.; Zukin, R.S.; Dalva, M.B. EphB Controls NMDA Receptor Function and Synaptic Targeting in a Subunit-Specific Manner. J. Neurosci. 2011, 31, 5353–5364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosch, R.E.; Wright, S.; Cooray, G.; Papadopoulou, M.; Goyal, S.; Lim, M.; Vincent, A.; Upton, A.L.; Baldeweg, T.; Friston, K.J. NMDA-receptor antibodies alter cortical microcircuit dynamics. Proc. Natl. Acad. Sci. USA 2018, 115, E9916–E9925. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malviya, M.; Barman, S.; Golombeck, K.S.; Planagumà, J.; Mannara, F.; Strutz-Seebohm, N.; Wrzos, C.; Demir, F.; Baksmeier, C.; Steckel, J.; et al. NMDAR encephalitis: Passive transfer from man to mouse by a recombinant antibody. Ann. Clin. Transl. Neurol. 2017, 4, 768–783. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Planaguma, J.; Haselmann, H.; Mannara, F.; Petit-Pedrol, M.; Grunewald, B.; Aguilar, E.; Ropke, L.; Martin-Garcia, E.; Titulaer, M.J.; Jercog, P.; et al. Ephrin-B2 prevents N-methyl-D-aspartate receptor antibody effects on memory and neuroplasticity. Ann. Neurol. 2016, 80, 388–400. [Google Scholar] [CrossRef]
- Jones, B.E.; Tovar, K.R.; Goehring, A.; Jalali-Yazdi, F.; Okada, N.J.; Gouaux, E.; Westbrook, G.L. Autoimmune receptor encephalitis in mice induced by active immunization with conformationally stabilized holoreceptors. Sci. Transl. Med. 2019, 11, eaaw0044. [Google Scholar] [CrossRef]
- Planagumà, J.; Leypoldt, F.; Mannara, F.; Gutiérrez-Cuesta, J.; Martín-García, E.; Aguilar, E.; Titulaer, M.; Petit-Pedrol, M.; Jain, A.; Balice-Gordon, R.; et al. Human N-methyl D-aspartate receptor antibodies alter memory and behaviour in mice. Brain 2015, 138, 94–109. [Google Scholar] [CrossRef] [PubMed]
- Meisel, J.L.; Woo, K.M.; Sudarsan, N.; Eng, J.; Patil, S.; Jacobsen, E.P.; Murali, R.; Gardner, G.J.; Bosl, G.; Aghajanian, C.; et al. Development of a risk stratification system to guide treatment for female germ cell tumors. Gynecol. Oncol. 2015, 138, 566–572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guan, H.-Z.; Ren, H.-T.; Cui, L.-Y. Autoimmune Encephalitis. Chin. Med. J. 2016, 129, 1122–1127. [Google Scholar] [CrossRef]
- Delangle, R.; Demeret, S.; Canlorbe, G.; Chelon, L.; Belghiti, J.; Gonthier, C.; Nikpayam, M.; Uzan, C.; Azaïs, H. Anti-NMDA receptor encephalitis associated with ovarian tumor: The gynecologist point of view. Arch. Gynecol. Obstet. 2020, 302, 315–320. [Google Scholar] [CrossRef]
- Mizutamari, E.; Matsuo, Y.; Namimoto, T.; Ohba, T.; Yamashita, Y.; Katabuchi, H. Successful outcome following detection and removal of a very small ovarian teratoma associated with anti- NMDA receptor encephalitis during pregnancy. Clin. Case Rep. 2016, 4, 223–225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tu, N.P.; Nha, P.B.; Hung, N.D.; Minh, N.H.; Anh, H.N.; Dinh, T.C. Treatment of Anti–NMDA Receptor Encephalitis with Ovarian Teratoma Removal: A Literature Review and Two Case Reports. Open Access Maced. J. Med. Sci. 2019, 7, 4378–4382. [Google Scholar] [CrossRef] [Green Version]
- Lee, W.-J.; Lee, S.-T.; Moon, J.; Sunwoo, J.-S.; Byun, J.-I.; Lim, J.-A.; Kim, T.-J.; Shin, Y.-W.; Lee, K.-J.; Jun-Sang, S.; et al. Tocilizumab in Autoimmune Encephalitis Refractory to Rituximab: An Institutional Cohort Study. Neurotherapeutics 2016, 13, 824–832. [Google Scholar] [CrossRef] [Green Version]
- Guasp, M.; Dalmau, J. Encefalitis por anticuerpos contra el receptor de NMDA. Med. Clínica 2018, 151, 71–79. [Google Scholar] [CrossRef]
- Crimì, F.; Camporese, G.; Lacognata, C.; Fanelli, G.N.; Cecchin, D.; Zoccarato, M. Ovarian Teratoma or Uterine Malformation? PET/MRI as a Novel Useful Tool in NMDAR Encephalitis. In Vivo 2018, 32, 1231–1233. [Google Scholar] [CrossRef] [Green Version]
- Thomas, A.G.; Mohan, M.; Thomas, R.; Baby, F.M. Ovarian Teratoma presenting as Anti NMDAR Antibody Negative Limbic Encephalitis. J. Assoc. Physicians India 2019, 67, 94–95. [Google Scholar] [PubMed]
- Balu, R.; McCracken, L.; Lancaster, E.; Graus, F.; Dalmau, J.; Titulaer, M.J. A score that predicts 1-year functional status in patients with anti-NMDA receptor encephalitis. Neurology 2019, 92, e244–e252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uchida, Y.; Kato, D.; Yamashita, Y.; Ozaki, Y.; Matsukawa, N. Failure to improve after ovarian resection could be a marker of recurrent ovarian teratoma in anti-NMDAR encephalitis: A case report. Neuropsychiatr. Dis. Treat. 2018, 14, 339–342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, X.; Li, J.; Zhu, Q.; Liang, G.; Xia, W.; He, X.; Zhu, C.; Qi, H.; Deng, B.; Chen, X.; et al. HLA-A and HLA-DRB1 may play a unique role in ovarian teratoma-associated anti-N-methyl-D-aspartate receptor encephalitis. Reprod. Biol. Endocrinol. 2020, 18, 1–12. [Google Scholar] [CrossRef]
- Cho, E.H.; Byun, J.M.; Park, H.Y.; Kang, M.R.; Jung, Y.J.; Jeong, C.H.; Kim, Y.N.; Lee, K.B.; Sung, M.S.; Jeong, D.H. The first case of Anti-N-methyl-d-aspartate receptor encephalitis (Anti-NMDAR encephalitis) associated with ovarian mucinous cystadenoma: A case report. Taiwan. J. Obstet. Gynecol. 2019, 58, 557–559. [Google Scholar] [CrossRef] [PubMed]
- Webster, E.M.; Tymon-Rosario, J.; D’Addario, J.; Zeybek, B.; Ratner, E.S. Opsoclonus-ataxia syndrome and mature ovarian teratoma in an adolescent. Gynecol. Oncol. Rep. 2020, 33, 100601. [Google Scholar] [CrossRef]
| Antigen | Autoantibody | Main Neurological Syndromes | Cancer Types |
|---|---|---|---|
| HuD | Hu-IgG (ANNA1) | Sensory neuronopathy, limbic encephalitis, and cerebellar ataxia. | SCLC, NSCLC, and extra-thoracic cancers. |
| Cdr-2 | Yo-IgG (PCA1) | PCD (majority), brainstem encephalitis, and myelopathy. | Ovarian, breast and fallopian tube carcinoma; gastrointestinal cancer in males. |
| SOX1 | SOX1-IgG | LEMS, PCD, and limbic encephalitis | SCLC, NSCLC, and extra-thoracic cancers. |
| Unknown | ANNA-3 | Limbic encephalitis, neuropathies, cerebellar ataxia, myelopathy, and brain stem encephalitis. | SCLC, NSCLC, and other tobacco-related airway cancers. |
| NOVA1 and NOVA2 | Ri-IgG (ANNA2) | Brainstem encephalitis, opsoclonus, laryngospasm, and jaw dystonia | Breast, lung, and neuroblastoma. |
| Ma1 and Ma2 | Ma and/or Ma2-IgG | Limbic encephalitis and brain stem encephalitis | Testicular, lung, and others (mainly gastrointestinal). |
| ZIC4 | ZIC4-IgG | PCD, and others. | SCLC, and ovarian adenocarcinoma. |
| Antigen | Autoantibody | Main Neurological Syndromes | Tumor Types | Frequency of Tumor |
|---|---|---|---|---|
| DNER | DNER-IgG (PCA-Tr) | PCD. | Hodgkin lymphoma. | >95% |
| GluA1, GluA2 | AMPAR-IgG | Limbic encephalitis. | SCLC, NSCLC, breast, and thymoma. | 60–70% |
| P/Q-type VGCC | P/Q-type VGCC-IgG | LEMS, and PCD. | SCLC. | 60% |
| β1 subunits | GABAbR-IgG | Limbic encephalitis, isolated status epilepticus, cerebellar ataxia, and opsoclonus myoclonus. | SCLC, thymoma, and extra-thoracic cancers. | 60% |
| GluD2 | GluD2-IgG | Opsoclonus myoclonus ataxia syndrome. | Neuroblastoma, and ovarian teratoma. | 50% |
| α1, β3, γ2 subunits | GABAaR-IgG | Encephalitis with seizures, cognitive impairment, and behavior changes. | Thymoma, and Hodgkin lymphoma. | 40% |
| GluN1 | NMDAR-IgG | Encephalitis with initial psychiatric disturbances, followed by catatonia, dystonia, seizures, aphasia, coma, and central hypoventilation. | Ovarian teratoma. | 20–40% |
| Muscle AChR | Anti-AChR | Myasthenia gravis. | Thymoma. | 15% |
| mGluR1 | mGluR1-IgG | Cerebellar ataxia. | Hematologic malignancies, and prostate adenocarcinoma. | 10% |
| DPPX/Kv4.2 | DPPX-IgG | Diarrhea, weight loss, cognitive dysfunction, and CNS hyperexcitability. | B-cell neoplasms. | 10% |
| Aquaporin-4 | Aquaporin-4-IgG | Neuromyelitis optica spectrum disorders (optic neuritis, longitudinally extensive transverse myelitis, and area postrema syndrome). | Thymoma, breast, and lung. | 5% |
| Criteria | ||
|---|---|---|
| Probable Case | Rapid onset (<3 months) of at least 4 of the 6 major groups of symptoms: | Major groups of symptoms:
|
| Additionally, at least one of the laboratory studies: |
| |
| Or 3 of the above groups of symptoms and identification of a systemic teratoma | ||
| Exclude the recent history of herpes simplex virus encephalitis or Japanese B encephalitis, resulting in relapsing immune-mediated neurological symptoms. | ||
| Definite Case | One or more of the 6 major groups of symptoms and IgG GluN1 antibodies (antibody testing should include CSF); if only serum is available, confirmatory tests should be included (e.g., live neurons or tissue immunohistochemistry, in addition to a cell-based assay) | |
| Exclude the recent history of herpes simplex virus encephalitis or Japanese B encephalitis, resulting in relapsing immune-mediated neurological symptoms. | ||
| Author/Reference | Study Design | GCT Case Number/Age | Ovarian GCT | Size and Laterality/ Time to Diagnosis | Prodromal Symptoms and PNS | Encephalitis Relapse Rate | GCT Incidence Rate | Note | |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Florance et al. [31] | Single institutional observational study. | 32 patients (>18 y/o: 24 cases; ≤18 y/o: 8 cases) | Teratomas, subtype not mentioned. | N/A | Prodromal symptoms: 48%$ Autonomic instability: 86%$ Movement disorder: 84%$Seizures: 77%$ Behavior/personality: 59%$ | 0% | 32/69 (46.4%) | 69 female patients with anti-NMDAR encephalitis (>18 y/o: 43 cases; ≤18 y/o: 26 cases). $: the percentages were based on cases assessable in the original study. |
| 2 | Titulaer et al. [32] | Multi-institutional observational study. | 207 patients (<12 y/o: 4 cases; 12–44 y/o: 199 cases; ≥45 y/o: 4 cases) | Teratomas, subtype not mentioned. | N/A | Prodromal symptoms: N/A Behavior/cognition: >95% Movement disorder: ~80% Seizures/memory deficits/ speech disorder: 70~80% | 12/207 (5.8%) | 207/468 (44.2%) | Among 468 female patients with anti-NMDAR encephalitis, 207 had ovarian teratomas, 4 had extraovarian teratomas, and 9 had other tumors. |
| 3 | Bost et al. [33] | Single-center retrospective observational study. | 51 patients; MT: the median age of 25 years (range: 15–45) IT: the median age of 22 years (range: 12–38) | MTs: 45 cases; ITs: 6 cases (grade 1: 2 cases, grade 2: 3 cases, grade 3: 1 case). | Median (range):MT: 7 days (−26~643); IT: 0 day (−6~131). | Prodromal symptoms: N/A Behavior/personality: 82%- Cognition: 32~100%- Movement disorder: 12~78%- Autonomic instability: 2~74%- Seizures: 8~64%- | 1/6 (16.7%)+ | 51/195 (26.2%) | There were 195 female patients with anti-NMDAR encephalitis (169 patients were above 12 years of age). +: among the 6 ITs patients −: percentages range from first and subsequent symptoms |
| 4 | Dai et al. [30] | A single-center prospective study. | 29 patients (mean age: 23.1, range: 10–36) | MTs: 28 cases; ITs: 1 case (grade 1). | Mean size: 4.6 cm (1–12 cm). | Prodromal symptoms: 53%; Mental/behavioral disorder: 89.7%; Seizures: 79.3%; Decreased consciousness: 65.5%; Hyperhidrosis: 62.1%; Speech disorder: 55.2%. | 4/29 (13.8%) | 29/108 (26.9%) | There were 108 female patients with anti-NMDAR encephalitis and a mean age of 23.4 years (range: 5–72). |
| 5 | Zhang et al. [34] | Multi-institutional observational study. | 26 patients (mean age: 23.3, range: 14–36) | MTs: 23 cases ITs: 3 cases | N/A | Prodromal symptoms: 38%; Psychiatric symptoms: 92.3%; Speech dysfunction: 84.6%; Seizures: 80.8%; Movement disorder: 76.9%; Decreased consciousness: 61.5%. | 3/26 (11.5%) | 26/56 (46.4%) | There were 56 female patients with anti-NMDAR encephalitis |
| 6 | Yaguchi et al. [35] | Case series. | 4 patients (mean age: 28, range: 23–31) | MTs: 3 cases. ITs: 1 case. | N/A | Prodromal symptoms: N/A; Psychosis: 100%; Seizures: 100%; Status epilepticus: 100%. (present 4 out of 4) | 0% | 4/343 (1.17%)# | All ovarian teratomas: 343 patients; MTs: 327 cases; ITs: 16 cases; (131 of 343 ovarian teratomas had neuroectodermal tissue.) #: incidence of encephalitis among patients with teratomas. |
| 4/6 (66.7%) | There were 6 female patients with anti-NMDAR encephalitis. Four of them had ovarian teratomas. | ||||||||
| 7 | Xu et al. [36] | Single-center prospective study. | 42 patients (>18 y/o: 33 cases; ≤18 y/o: 9 cases) | Teratomas, subtype not mentioned. | N/A | Prodromal symptoms: 48.2%; Psychosis: 82.7%; Seizures: 80.9%; Decreased consciousness: 53.2%; Memory deficit: 48.2%; Speech disturbance: 45.5%. | 5/42 (11.9%) | 42/143 (29.4%) | There were 143 female patients with anti-NMDAR encephalitis (>18 y/o: 102 cases; ≤18 y/o: 41 cases). |
| 8 | Acién et al. [37] | A systematic review of reported cases. | 174 patients (mean age: 23.9, range: 7–54) | MTs: 99 cases; ITs: 29 cases; Mixed MTs and ITs: 6 cases; Unknown: 40 cases. | Mean size: 6.7 cm (range: 0.1–22 cm); Right: 56 cases; Left: 46 cases; Bilateral: 20 case; Unknown: 52 cases; Mean time: 28 days (3 to 455 days). | N/A | N/A | N/A | Collected cases and data before 2014. |
| 9 | Chiu et al. [38] | Case series. | 5 patients (mean age: 18.6, range: 7–28) | MTs: 5 cases; Ovarian fibroma: 1 case. | Mean size: 2.65 cm (2–3.3 cm); Right side: 1 case; Left side: 1 case; Not marked: 3 cases; Mean time: 59.6 days (7 to 150 days). | Prodromal symptoms: 60%; Autonomic dysfunction: 80%; Psychosis: 80%; Seizures: 80%; Decreased consciousness: 60%; Impaired speech: 60%. | 1/5 (20%) | 5/13 (38.5%) | 13 female patients with anti-NMDAR encephalitis (mean age: 19.9, range: 7–28). |
| 10 | Yan et al. [39] | Case report and literature review. | 15 patients (mean age: 21, range: 7–33) | MTs: 10 cases; ITs: 2 cases; Unknown: 3 cases. (5 of 10 MTs had mature brain tissues.) | Right: 5 cases; Left: 2 cases; Bilateral: 1 case; Unknown: 7 cases. | Prodromal symptoms: 66.7%; Nervous and mental symptoms: 93.3%; Seizures: 53.3%; Dyskinesia: 40%; Autonomic instability: 38.5%. | N/A | N/A | 14 published case reports during 2010 to 2019 and one presented case. NMDAR-Ab was positive in CSF. |
| 11 | Yu et al. [40] | Case series. | 6 patients (mean age: 25, range: 21–27) | MTs: 6 cases (4 of 6 had mature brain tissues). | Mean size: 1.73 cm; Right side: 3 cases; Left side: 3 cases; Mean time: 22.5 days (5 days to 3 months). | Prodromal symptoms: 50%; Psychotic symptoms: 100%; Cognitive decline: 100%; Abnormal movement: 100%; Seizures: 100%; Autonomic dysfunction: 66.7%. | N/A | N/A | NMDAR-Ab was positive in CSF. |
| 12 | Dalmau et al. [29] | Case series. | 12 patients (mean age: 24, range: 14–44) | MTs: 8 cases; ITs: 4 cases. | Mean size: 6.4 cm (1.5–22 cm); Right side: 4 cases; Left side: 6 cases; Bilateral: 1 case; Mediastium: 1 case; Mean time: 8 weeks (3 weeks to 5 months). | Prodromal symptoms: 83.3%; Seizures: 91.7%; Psychiatric symptoms: 83.3%; Memory deficit: 58.3%; Movement disorder: 50%; Decreased consciousness: 41.7%; Impaired speech: 41.7%; Autonomic dysfunction: 33.3%. | 0% | N/A | - |
| 13 | Ahmad et al. [41] | Case report. | 1 patient, 26 y/o | MT: 1 case. | Size: 2.5 cm; Right side. | Prodromal symptoms: 100% Psychosis: 100% Seizures: 100% Movement disorder: 100% Decreased consciousness: 100% | 0% | N/A | NMDAR-Ab was positive in CSF. |
| 14 | Omata et al. [42] | Case report. | 2 patients (mean age: 12.5, range: 11–14) | MTs: 2 cases. | Mean size: 2.8 cm (1.3–5 cm); Left side: 1 case; Bilateral: 1 case. | Prodromal symptoms: N/A Psychotic symptoms: 100% Movement disorder: 100% Abnormal speech: 50% | 0% | N/A | NMDAR-Ab was positive in CSF. |
| 15 | Mitra et al. [43] | Case report. | 1 patient, 22 y/o | MT: 1 case with neural elements resembling white matter. | Size: 1.4 cm; Right side. | Prodromal symptoms: N/A Altered mental status: 100% Psychosis: 100% Autonomic dysfunction: 100% Movement disorder: 100% | 0% | N/A | NMDAR-Ab was positive in CSF. |
| 16 | Lwin et al. [44] | Case report. | 1 patient, 12 y/o | MT: 1 case. | Size: 5 cm; Left side. | Prodromal symptoms: N/A Psychosis/Abnormal behavior: 100% Seizures: 100% | N/A | N/A | NMDAR-Ab was positive in CSF. |
| 17 | Lee et al. [45] | Case report. | 1 patient, 24 y/o | MT: 1 case with mature brain tissue. | Size: 1 cm; Right side. | Prodromal symptoms: 100% Abnormal movement: 100% Decreased mental status: 100% Autonomic dysfunction: 100% | N/A | N/A | NMDAR-Ab was positive in CSF. |
| 18 | Chernyshkova et al. [46] | Case report. | 1 patient, 55 y/o | MT: 1 case. | Left side. | Prodromal symptoms: N/A Altered mental status: 100% Autonomic dysfunction: 100% Seizures: 100% Movement disorder: 100% | N/A | N/A | This is a probable case based on Table 3 since autoantibodies in CSF were not detected. |
| Total conclusion (for assessable cases) | Mean age: 23.97; (age range: 7~55) | MTs: 234 cases; ITs: 46 cases; Unknown: 330 cases. | Mean size: 3.48 cm; Right side: 72 cases; Left side: 61 cases; Bilateral: 23 cases. | Prodromal symptoms: 64.7%; Behavior/personality/ Psychosis: 89.8%; Seizures: 82.4%; Movement disorder: 79.6%; Decreased consciousness: 77.1%; Autonomic dysfunction: 70.5%; Speech disorder/memory deficit: 60.4%. | 26/367 (7.1%) | 396/1058 (37.4%) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, C.-Y.; Wu, J.-D.; Chen, C.-C. The Association of Ovarian Teratoma and Anti-N-Methyl-D-Aspartate Receptor Encephalitis: An Updated Integrative Review. Int. J. Mol. Sci. 2021, 22, 10911. https://doi.org/10.3390/ijms222010911
Wu C-Y, Wu J-D, Chen C-C. The Association of Ovarian Teratoma and Anti-N-Methyl-D-Aspartate Receptor Encephalitis: An Updated Integrative Review. International Journal of Molecular Sciences. 2021; 22(20):10911. https://doi.org/10.3390/ijms222010911
Chicago/Turabian StyleWu, Cheng-Yang, Jiann-Der Wu, and Chien-Chin Chen. 2021. "The Association of Ovarian Teratoma and Anti-N-Methyl-D-Aspartate Receptor Encephalitis: An Updated Integrative Review" International Journal of Molecular Sciences 22, no. 20: 10911. https://doi.org/10.3390/ijms222010911







