Functions of PPR Proteins in Plant Growth and Development
Abstract
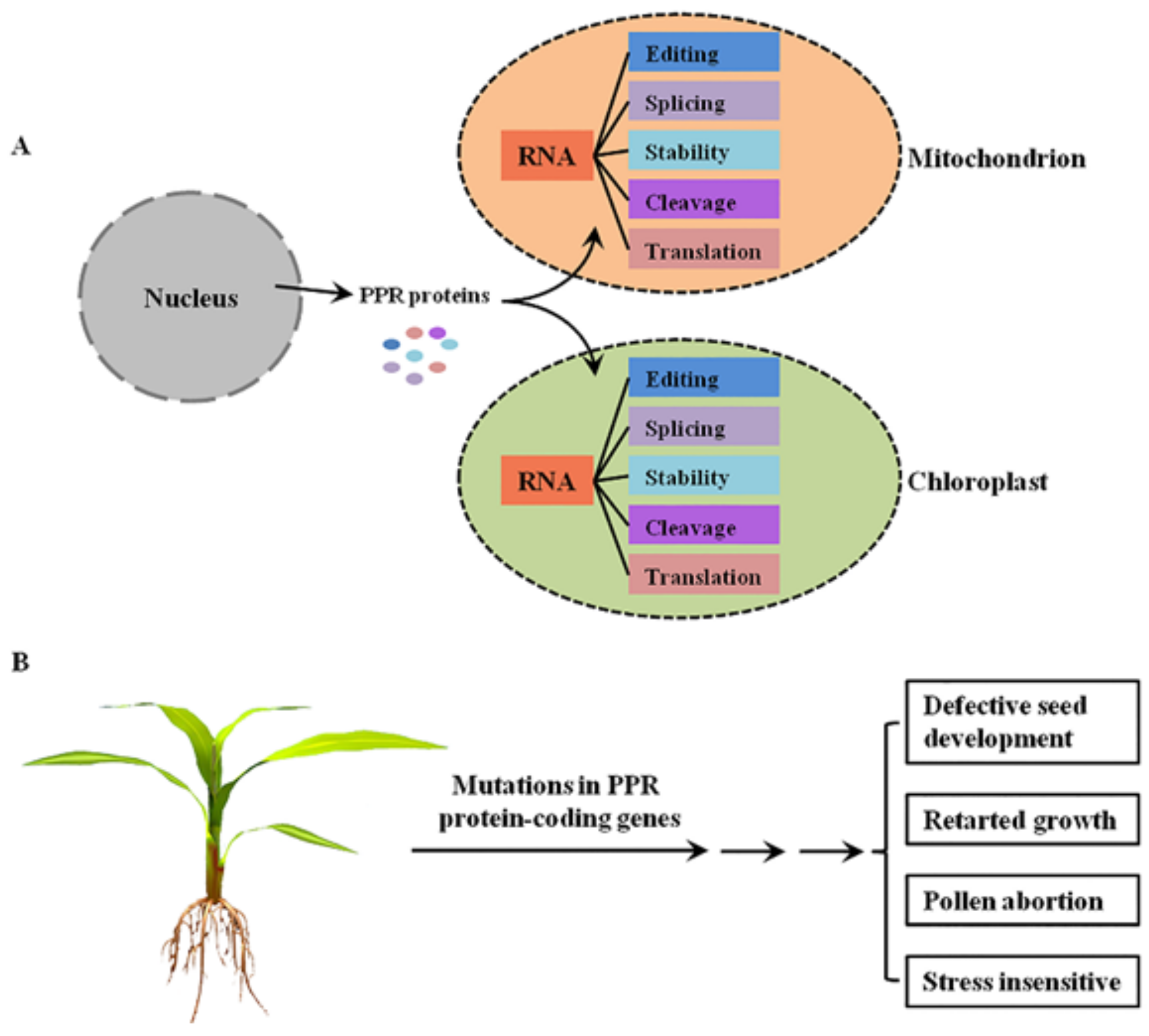
:1. Introduction
2. Functions of PPR Proteins in Cytoplasmic Male Sterility
3. Functions of PPR Proteins in Plant Responses to Biotic and Abiotic Stresses
4. Functions of PPR Proteins in Seed Development
5. Mechanisms of PPR Protein-Mediated RNA Processing in Plant Organelles
5.1. Molecular Mechanisms of PPR Proteins in C-to-U RNA Editing
5.2. Molecular Mechanisms of PPR Proteins in Group II Intron Splicing
6. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Small, I.D.; Peeters, N. The PPR motif-a TPR-related motif prevalent in plant organellar proteins. Trends Biochem. Sci. 2000, 25, 45–47. [Google Scholar] [CrossRef]
- Lurin, C.; Andrés, C.; Aubourg, S.; Bellaoui, M.; Bitton, F.; Bruyère, C.; Caboche, M.; Debast, C.; Gualberto, J.; Hoffmann, B.; et al. Genome-wide analysis of Arabidopsis pentatricopeptide repeat proteins reveals their essential role in organelle biogenesis. Plant Cell 2004, 16, 2089–2103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, S.F.; Gutmann, B.; Zhong, X.; Ye, Y.T.; Fisher, M.F.; Bai, F.Q.; Castleden, I.; Song, Y.; Song, B.; Huang, J.Y.; et al. Redefining the structural motifs that determine RNA binding and RNA editing by pentatricopeptide repeat proteins in land plants. Plant J. 2016, 85, 532–547. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barkan, A.; Small, I. Pentatricopeptide repeat proteins in plants. Annu. Rev. Plant Biol. 2014, 65, 415–442. [Google Scholar] [CrossRef] [PubMed]
- Schmitz-Linneweber, C.; Small, I. Pentatricopeptide repeat proteins: A socket set for organelle gene expression. Trends Plant Sci. 2008, 13, 663–670. [Google Scholar] [CrossRef]
- Chase, C.D. Cytoplasmic male sterility: A window to the world of plant mitochondrial-nuclear interactions. Trends Genet. 2007, 23, 81–90. [Google Scholar] [CrossRef]
- Bohra, A.; Jha, U.C.; Adhimoolam, P.; Bisht, D.; Singh, N.P. Cytoplasmic male sterility (CMS) in hybrid breeding in field crops. Plant Cell Rep. 2016, 35, 967–993. [Google Scholar] [CrossRef]
- Linke, B.; Börner, T. Mitochondrial effects on flower and pollen development. Mitochondrion 2005, 5, 389–402. [Google Scholar] [CrossRef] [PubMed]
- Eckardt, N.A. Cytoplasmic male sterility and fertility restoration. Plant Cell 2006, 18, 515–517. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.H.; Zou, Y.J.; Li, X.Y.; Zhang, Q.Y.; Chen, L.T.; Wu, H.; Su, D.H.; Chen, Y.L.; Guo, J.X.; Luo, D.; et al. Cytoplasmic male sterility of rice with Boro II cytoplasm is caused by a cytotoxic peptide and is restored by two related PPR motif genes via distinct modes of mRNA silencing. Plant Cell 2006, 18, 676–687. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kazama, T.; Nakamura, T.; Watanabe, M.; Sugita, M.; Toriyama, K. Suppression mechanism of mitochondrial ORF79 accumulation by Rf1 protein in BT-type cytoplasmic male sterile rice. Plant J. 2008, 55, 619–628. [Google Scholar] [CrossRef]
- Luo, D.P.; Xu, H.; Liu, Z.L.; Guo, J.X.; Li, H.Y.; Chen, L.T.; Fang, C.; Zhang, Q.Y.; Bai, M.; Yao, N.; et al. A detrimental mitochondrial-nuclear interaction causes cytoplasmic male sterility in rice. Nat. Genet. 2013, 45, 573–577. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Luo, D.; Zhou, D.; Zhang, Q.; Tian, D.; Zheng, X.; Chen, L.; Liu, Y.G. The rice restorer Rf4 for wild-abortive cytoplasmic male sterility encodes a mitochondrial-localized PPR protein that functions in reduction of WA352 transcripts. Mol. Plant 2014, 7, 1497–1500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, J.; Wang, K.; Huang, W.C.; Liu, G.; Gao, Y.; Wang, J.M.; Huang, Q.; Ji, Y.X.; Qin, X.J.; Wan, L.; et al. The rice pentatricopeptide repeat protein RF5 restores fertility in Hong-Lian cytoplasmic male-sterile lines via a complex with the glycine-rich protein GRP162. Plant Cell 2012, 24, 109–122. [Google Scholar] [CrossRef] [Green Version]
- Huang, W.C.; Yu, C.C.; Hu, J.; Wang, L.L.; Dan, Z.W.; Zhou, W.; He, C.L.; Zeng, Y.F.; Yao, G.X.; Qi, J.Z.; et al. Pentatricopeptide-repeat family protein RF6 functions with hexokinase 6 to rescue rice cytoplasmic male sterility. Proc. Natl. Acad. Sci. USA 2015, 112, 14984–14989. [Google Scholar] [CrossRef] [Green Version]
- Brown, G.G.; Formanová, N.; Jin, H.; Wargachuk, R.; Dendy, C.; Patil, P.; Laforest, M.; Zhang, J.F.; Cheung, W.Y.; Landry, B.S. The radish Rfo restorer gene of Ogura cytoplasmic male sterility encodes a protein with multiple pentatricopeptide repeats. Plant J. 2003, 35, 262–272. [Google Scholar] [CrossRef]
- Uyttewaal, M.; Arnal, N.; Quadrado, M.; Martin-Canadell, A.; Vrielynck, N.; Hiard, S.; Gherbi, H.; Bendahmane, A.; Budar, F.; Mireau, H. Characterization of Raphanus sativus pentatricopeptide repeat proteins encoded by the fertility restorer locus for Ogura cytoplasmic male sterility. Plant Cell 2008, 20, 3331–3345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.W.; De Wang, C.D.; Mei, S.Y.; Gao, L.; Zhou, Y.; Wang, T. An insertion-deletion at a pentatricopeptide repeat locus linked to fertility transition to cytoplasmic male sterility in radish (Raphanus sativus L.). Mol. Breed. 2015, 35, 108–112. [Google Scholar] [CrossRef]
- Imai, R.; Koizuka, N.; Fujimoto, H.; Hayakawa, T.; Sakai, T.; Imamura, J. Delimitation of the fertility restorer locus Rfk1 to a 43-kb contig in Kosena radish (Raphanus sativus L.). Mol. Genet. Genom. 2003, 269, 388–394. [Google Scholar] [CrossRef]
- Klein, R.R.; Klein, P.E.; Mullet, J.E.; Minx, P.; Rooney, W.L.; Schertz, K.F. Fertility restorer locus Rf1 of sorghum (Sorghum bicolor L.) encodes a pentatricopeptide repeat protein not present in the colinear region of rice chromosome 12. Theor. Appl. Genet. 2005, 111, 994–1012. [Google Scholar] [CrossRef]
- Madugula, P.; Uttam, A.G.; Tonapi, V.A.; Ragimasalawada, M. Fine mapping of Rf2, a major locus controlling pollen fertility restoration in sorghum A1 cytoplasm, encodes a PPR gene and its validation through expression analysis. Plant Breed. 2018, 137, 148–161. [Google Scholar] [CrossRef]
- Liu, Z.; Yang, Z.; Wang, X.; Li, K.; An, H.; Liu, J.; Yang, G.S.; Fu, T.; Yi, B.; Hong, D.F. A mitochondria-targeted PPR protein restores pol cytoplasmic male sterility by reducing orf224 transcript levels in oilseed rape. Mol. Plant 2016, 9, 1082–1084. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Z.; Dong, F.M.; Wang, X.; Wang, T.; Su, R.; Hong, D.F.; Yang, G.S. A pentatricopeptide repeat protein restores nap cytoplasmic male sterility in Brassica napus. J. Exp. Bot. 2017, 68, 4115–4123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gillman, J.D.; Bentolila, S.; Hanson, M.R. The petunia restorer of fertility protein is part of a large mitochondrial complex that interacts with transcripts of the CMS-associated locus. Plant J. 2007, 49, 217–227. [Google Scholar] [CrossRef]
- Zhang, H.M.; Wu, J.Q.; Dai, Z.H.; Qin, M.L.; Hao, L.Y.; Ren, Y.J.; Li, Q.F.; Zhang, L.G. Allelism analysis of BrRfp locus in different restorer lines and map-based cloning of a fertility restorer gene, BrRfp1, for pol CMS in Chinese cabbage (Brassica rapa L.). Theor. Appl. Genet. 2017, 130, 539–547. [Google Scholar] [CrossRef]
- Rizzolatti, C.; Bury, P.; Tatara, E.; Pin, P.A.; Rodde, N.; Bergès, H.; Budar, F.; Mireau, H.; Gielen, J.J.L. Map-based cloning of the fertility restoration locus Rfm1 in cultivated barley (Hordeum vulgare). Euphytica 2017, 213, 276–287. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.T.; Liu, Y.G. Male sterility and fertility restoration in crops. Annu. Rev. Plant Biol. 2014, 65, 579–606. [Google Scholar] [CrossRef]
- Jiang, S.C.; Mei, C.; Liang, S.; Yu, Y.T.; Lu, K.; Wu, Z.; Wang, X.F.; Zhang, D.P. Crucial roles of the pentatricopeptide repeat protein SOAR1 in Arabidopsis response to drought, salt and cold stresses. Plant Mol. Biol. 2015, 88, 369–385. [Google Scholar] [CrossRef] [Green Version]
- Wu, G.Z.; Chalvin, C.; Hoelscher, M.; Meyer, E.H.; Wu, X.N.; Bock, R. Control of retrograde signaling by rapid turnover of GENOMES UNCOUPLED1. Plant Physiol. 2018, 176, 2472–2495. [Google Scholar] [CrossRef] [Green Version]
- Mochizuki, N.; Susek, R.; Chory, J. An intracellular signal transduction pathway between the chloroplast and nucleus is involved in de-etiolation. Plant Physiol. 1996, 112, 1465–1469. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, K.; Suzuki, M.; Tang, J.W.; Nagata, N.; Ohyama, K.; Seki, H.; Kiuchi, R.; Kaneko, Y.; Nakazawa, M.; Matsui, M.; et al. LOVASTATIN INSENSITIVE 1, a novel pentatricopeptide repeat protein, is a potential regulatory factor of isoprenoid biosynthesis in Arabidopsis. Plant Cell Physiol. 2007, 48, 322–331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zsigmond, L.; Rigoó, G.; Szarka, A.; Szeékely, G.; Ötvös, K.; Darula, Z.; Medzihradszky, K.F.; Koncz, C.; Koncz, Z.; Szabados, L. Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport. Plant Physiol. 2008, 146, 1721–1737. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; He, J.N.; Chen, Z.Z.; Ren, X.Z.; Hong, X.H.; Gong, Z.Z. ABA overly-sensitive 5 (ABO5), encoding a pentatricopeptide repeat protein required for cis-splicing of mitochondrial nad2 intron 3, is involved in the abscisic acid response in Arabidopsis. Plant J. 2010, 63, 749–765. [Google Scholar] [CrossRef]
- Laluk, K.; AbuQamar, S.; Mengiste, T. The Arabidopsis mitochondria-localized pentatricopeptide repeat protein PGN functions in defense against necrotrophic fungi and abiotic stress tolerance. Plant Physiol. 2011, 156, 2053–2068. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murayama, M.; Hayashi, S.; Nishimura, N.; Ishide, M.; Kobayashi, K.; Yagi, Y.; Asami, T.; Nakamura, T.; Shinozaki, K.; Hirayama, T. Isolation of Arabidopsis ahg11, a weak ABA hypersensitive mutant defective in nad4 RNA editing. J. Exp. Bot. 2012, 63, 5301–5310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, H.; Liu, D. Functional disruption of the pentatricopeptide protein SLG1 affects mitochondrial RNA editing, plant development, and responses to abiotic stresses in Arabidopsis. Plant J. 2012, 70, 432–444. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Dugardeyn, J.; Zhang, C.Y.; Mühlenbock, P.; Eastmond, P.J.; Valcke, R.; De Coninck, B.; Öden, S.; Karampelias, M.; Cammue, B.P.A.; et al. The Arabidopsis thaliana RNA editing factor SLO2, which affects the mitochondrial electron transport chain, participates in multiple stress and hormone responses. Mol. Plant 2014, 7, 290–310. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.M.; Zhao, J.Y.; Lu, P.P.; Chen, M.; Guo, C.H.; Xu, Z.S.; Ma, Y.Z. The E-subgroup pentatricopeptide repeat protein family in Arabidopsis thaliana and confirmation of the responsiveness PPR96 to abiotic stresses. Front. Plant Sci. 2016, 7, 1825. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Emami, H.; Kumar, A.; Kempken, F. Transcriptomic analysis of poco1, a mitochondrial pentatricopeptide repeat protein mutant in Arabidopsis thaliana. BMC Plant Biol. 2020, 20, 209. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.D.; Su, Q.Q.; Lin, D.Z.; Jiang, Q.; Xu, J.L.; Zhang, J.H.; Teng, S.; Dong, Y.J. The rice OsV4 encoding a novel pentatricopeptide repeat protein is required for chloroplast development during the early leaf stage under cold stress. J. Integr. Plant Biol. 2014, 56, 400–410. [Google Scholar] [CrossRef]
- Wu, L.L.; Wu, J.; Liu, Y.X.; Gong, X.D.; Xu, J.L.; Lin, D.Z.; Dong, Y.J. The rice pentatricopeptide repeat gene TCD10 is needed for chloroplast development under cold stress. Rice 2016, 9, 67–79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, J.J.; Tan, Z.H.; Wu, F.Q.; Sheng, P.K.; Heng, Y.Q.; Wang, X.H.; Ren, Y.L.; Wang, J.L.; Guo, X.P.; Zhang, X.; et al. A novel chloroplast-localized pentatricopeptide repeat protein involved in splicing affects chloroplast development and abiotic stress response in rice. Mol. Plant 2014, 7, 1329–1349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, T.C.; Zhao, X.S.; Feng, H.J.; Qi, L.L.; Yang, J.; Peng, Y.L.; Zhao, W.S. OsNBL3, a mitochondrion-localized pentatricopeptide repeat protein, is involved in splicing nad5 intron 4 and its disruption causes lesion mimic phenotype with enhanced resistance to biotic and abiotic stresses. Plant Biotechnol. J. 2021. [Google Scholar] [CrossRef] [PubMed]
- Tadini, L.; Pesaresi, P.; Kleine, T.; Rossi, F.; Guljamow, A.; Sommer, F.; Mühlhaus, T.; Schroda, M.; Masiero, S.; Pribil, M.; et al. GUN1 controls accumulation of the plastid ribosomal protein S1 at the protein level and interacts with proteins involved in plastid protein homeostasis. Plant Physiol. 2016, 170, 1817–1830. [Google Scholar] [CrossRef] [Green Version]
- Takenaka, M.; Zehrmann, A.; Verbitskiy, D.; Kugelmann, M.; Hartel, B.; Brennicke, A. Multiple organellar RNA editing factor (MORF) family proteins are required for RNA editing in mitochondria and plastids of plants. Proc. Natl. Acad. Sci. USA 2012, 109, 5104–5109. [Google Scholar] [CrossRef] [Green Version]
- Tadini, L.; Peracchio, C.; Trotta, A.; Colombo, M.; Mancini, I.; Jeran, N.; Costa, A.; Faoro, F.; Marsoni, M.; Vannini, C.; et al. GUN1 influences the accumulation of NEP-dependent transcripts and chloroplast protein import in Arabidopsis cotyledons upon perturbation of chloroplast protein homeostasis. Plant J. 2020, 101, 1198–1220. [Google Scholar] [CrossRef]
- Zhao, X.B.; Huang, J.Y.; Chory, J. GUN1 interacts with MORF2 to regulate plastid RNA editing during retrograde signaling. Proc. Natl. Acad. Sci. USA 2019, 116, 10162–10167. [Google Scholar] [CrossRef] [Green Version]
- Verbitskiy, D.; Zehrmann, A.; van der Merwe, J.A.; Brennicke, A.; Takenaka, M. The PPR protein encoded by the LOVASTATIN INSENSITIVE1 gene is involved in RNA editing at three sites in mitochondria of Arabidopsis thaliana. Plant J. 2010, 61, 446–455. [Google Scholar] [CrossRef]
- Goldberg, R.B.; de Paiva, G.; Yadegari, R. Plant embryogenesis: Zygote to seed. Science 1994, 266, 605–614. [Google Scholar] [CrossRef]
- Linkies, A.; Graeber, K.; Knight, C.; Leubner-Metzger, G. The evolution of seeds. New Phytol. 2010, 186, 817–831. [Google Scholar] [CrossRef]
- Dai, D.W.; Ma, Z.Y.; Song, R.T. Maize kernel development. Mol. Breed. 2021, 41, 2. [Google Scholar] [CrossRef]
- Qi, W.W.; Yang, Y.; Feng, X.Z.; Zhang, M.L.; Song, R.T. Mitochondrial function and maize kernel development requires Dek2, a pentatricopeptide repeat protein involved in nad1 mRNA splicing. Genetics 2017, 205, 239–249. [Google Scholar] [CrossRef] [Green Version]
- Qi, W.W.; Tian, Z.R.; Lu, L.; Chen, X.Z.; Chen, X.Z.; Zhang, W.; Song, R. Editing of mitochondrial transcripts nad3 and cox2 by Dek10 is essential for mitochondrial function and maize plant development. Genetics 2017, 205, 1489–1501. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.Z.; Feng, F.; Qi, W.W.; Xu, L.M.; Yao, D.S.; Wang, Q.; Song, R.T. Dek35 encodes a PPR protein that affects cis-splicing of mitochondrial nad4 intron 1 and seed development in maize. Mol. Plant 2017, 10, 427–441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, G.; Zhong, M.Y.; Shuai, B.L.; Song, J.D.; Zhang, J.; Han, L.; Ling, H.L.; Tang, Y.P.; Wang, G.F.; Song, R.T. E+ subgroup PPR protein defective kernel 36 is required for multiple mitochondrial transcripts editing and seed development in maize and Arabidopsis. New Phytol. 2017, 214, 1563–1578. [Google Scholar] [CrossRef] [Green Version]
- Dai, D.W.; Luan, S.C.; Chen, X.Z.; Wang, Q.; Feng, Y.; Zhu, C.G.; Qi, W.W.; Song, R.T. Maize Dek37 encodes a P-type PPR protein that affects cis-splicing of mitochondrial nad2 intron 1 and seed development. Genetics 2018, 208, 1069–1082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.J.; Gu, W.; Sun, S.L.; Chen, Z.L.; Chen, J.; Song, W.B.; Zhao, H.M.; Lai, J.S. Defective Kernel 39 encodes a PPR protein required for seed development in maize. J. Integr. Plant Biol. 2018, 60, 45–64. [Google Scholar] [CrossRef] [Green Version]
- Ren, R.C.; Lu, X.D.; Zhao, Y.J.; Wei, Y.M.; Wang, L.L.; Zhang, L.; Zhang, W.T.; Zhang, C.Y.; Zhang, X.S.; Zhao, X.Y. Pentatricopeptide repeat protein DEK40 is required for mitochondrial function and kernel development in maize. J. Exp. Bot. 2019, 70, 6163–6179. [Google Scholar] [CrossRef]
- Ren, R.C.; Wang, L.L.; Zhang, L.; Zhao, Y.J.; Wu, J.W.; Wei, Y.M.; Zhang, X.S.; Zhao, X.Y. DEK43 is a P-type pentatricopeptide repeat (PPR) protein responsible for the cis-splicing of nad4 in maize mitochondria. J. Integr. Plant Biol. 2020, 62, 299–313. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.G.; Jin, G.P.; Fang, P.; Zhang, Y.; Feng, X.Z.; Tang, Y.P.; Qi, W.W.; Song, R.T. Maize pentatricopeptide repeat protein DEK41 affects cis-splicing of mitochondrial nad4 intron 3 and is required for normal seed development. J. Exp. Bot. 2019, 70, 3795–3808. [Google Scholar] [CrossRef] [Green Version]
- Xu, C.H.; Song, S.; Yang, Y.Z.; Lu, F.; Zhang, M.D.; Sun, F.; Jia, R.X.; Song, R.L.; Tan, B.C. DEK46 performs C-to-U editing of a specific site in mitochondrial nad7 introns that is critical for intron splicing and seed development in maize. Plant J. 2020, 103, 1767–1782. [Google Scholar] [CrossRef] [PubMed]
- Dai, D.W.; Jin, L.F.; Huo, Z.Z.; Yan, S.M.; Ma, Z.Y.; Qi, W.W.; Song, R.T. Maize pentatricopeptide repeat protein DEK53 is required for mitochondrial RNA editing at multiple sites and seed development. J. Exp. Bot. 2020, 71, 6246–6261. [Google Scholar] [CrossRef] [PubMed]
- Ren, R.C.; Yan, X.W.; Zhao, Y.J.; Wei, Y.M.; Lu, X.D.; Zang, J.; Wu, J.W.; Zheng, G.M.; Ding, X.H.; Zhang, X.S.; et al. The novel E-subgroup pentatricopeptide repeat protein DEK55 is responsible for RNA editing at multiple sites and for the splicing of nad1 and nad4 in maize. BMC Plant Biol. 2020, 20, 553. [Google Scholar] [CrossRef]
- Fan, K.J.; Peng, Y.X.; Ren, Z.J.; Li, D.L.; Zhen, S.H.; Hey, S.; Cui, Y.; Fu, J.J.; Gu, R.L.; Wang, J.H.; et al. Maize defective kernel605 encodes a canonical DYW-Type PPR protein that edits a conserved site of nad1 and is essential for seed nutritional quality. Plant Cell Physiol. 2020, 61, 1954–1966. [Google Scholar] [CrossRef]
- Li, X.J.; Zhang, Y.F.; Hou, M.M.; Sun, F.; Shen, Y.; Xiu, Z.H.; Wang, X.M.; Chen, Z.L.; Sun, S.S.M.; Small, I.; et al. Small kernel 1 encodes a pentatricopeptide repeat protein required for mitochondrial nad7 transcript editing and seed development in maize (Zea mays) and rice (Oryza sativa). Plant J. 2014, 79, 797–809. [Google Scholar] [CrossRef]
- Wang, H.C.; Sayyed, A.; Liu, X.Y.; Yang, Y.Z.; Sun, F.; Wang, Y.; Wang, M.D.; Tan, B.C. SMALL KERNEL4 is required for mitochondrial cox1 transcript editing and seed development in maize. J. Integr. Plant Biol. 2020, 62, 777–792. [Google Scholar] [CrossRef]
- Ding, S.; Liu, X.Y.; Wang, H.C.; Wang, Y.; Tang, J.J.; Yang, Y.Z.; Tan, B.C. SMK6 mediates the C-to-U editing at multiple sites in maize mitochondria. J. Plant Physiol. 2019, 240, 152992. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.Y.; Liu, M.; Xiao, Z.Y.; Ren, X.M.; Zhao, H.L.; Gong, D.M.; Liang, K.; Tan, Z.D.; Shao, Y.Q.; Qiu, F.Z. ZmSMK9, a pentatricopeptide repeat protein, is involved in the cis-splicing of nad5, kernel development and plant architecture in maize. Plant Sci. 2019, 288, 110205. [Google Scholar] [CrossRef]
- Sosso, D.; Mbelo, S.; Vernoud, V.; Gendrot, G.; Dedieu, A.; Chambrier, P.; Dauzat, M.; Heurtevin, L.; Guyon, V.; Takenaka, M.; et al. PPR2263, a DYW-subgroup pentatricopeptide repeat protein, is required for mitochondrial nad5 and cob transcript editing, mitochondrion biogenesis, and maize growth. Plant Cell 2012, 24, 676–691. [Google Scholar] [CrossRef] [Green Version]
- Manavski, N.; Guyon, V.; Meurer, J.; Wienand, U.; Brettschneider, R. An essential pentatricopeptide repeat protein facilitates 5′ maturation and translation initiation of rps3 mRNA in maize mitochondria. Plant Cell 2012, 24, 3087–3105. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.Z.; Ding, S.; Wang, Y.; Wang, H.C.; Liu, X.Y.; Sun, F.; Xu, C.H.; Liu, B.H.; Tan, B.C. PPR20 is required for the cis-splicing of mitochondrial nad2 intron 3 and seed development in maize. Plant Cell Physiol. 2020, 61, 370–380. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.F.; Suzuki, M.; Sun, F.; Tan, B.C. The mitochondrion-targeted PENTATRICOPEPTIDE REPEAT78 protein is required for nad5 mature mRNA stability and seed development in maize. Mol. Plant 2017, 10, 1321–1333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gutierrez-Marcos, J.F.; Dal Pra, M.; Giulini, A.; Costa, L.M.; Gavazzi, G.; Cordelier, S.; Sellam, O.; Tatout, C.; Paul, W.; Perez, P.; et al. Empty pericarp4 encodes a mitochondrion-targeted pentatricopeptide repeat protein necessary for seed development and plant growth in maize. Plant Cell 2007, 19, 196–210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.J.; Xiu, Z.H.; Meeley, R.; Tan, B.C. Empty pericarp5 encodes a pentatricopeptide repeat protein that is required for mitochondrial RNA editing and seed development in maize. Plant Cell 2013, 25, 868–883. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, F.; Wang, X.M.; Bonnard, G.; Shen, Y.; Xiu, Z.H.; Li, X.J.; Gao, D.H.; Zhang, Z.H.; Tan, B.C. Empty pericarp7 encodes a mitochondrial E-subgroup pentatricopeptide repeat protein that is required for ccmFN editing, mitochondrial function and seed development in maize. Plant J. 2015, 84, 283–295. [Google Scholar] [CrossRef]
- Sun, F.; Zhang, X.Y.; Shen, Y.; Wang, H.C.; Liu, R.; Wang, X.M.; Gao, D.H.; Yang, Y.Z.; Liu, Y.W.; Tan, B.C. The pentatricopeptide repeat protein EMPTY PERICARP8 is required for the splicing of three mitochondrial introns and seed development in maize. Plant J. 2018, 95, 919–932. [Google Scholar] [CrossRef]
- Yang, Y.Z.; Ding, S.; Wang, H.C.; Sun, F.; Huang, W.L.; Song, S.; Xu, C.H.; Tan, B.C. The pentatricopeptide repeat protein EMP9 is required for mitochondrial ccmB and rps4 transcript editing, mitochondrial complex biogenesis and seed development in maize. New Phytol. 2017, 214, 782–795. [Google Scholar] [CrossRef] [Green Version]
- Cai, M.J.; Li, S.Z.; Sun, F.; Sun, Q.; Zhao, H.L.; Ren, X.M.; Zhao, Y.X.; Tan, B.C.; Zhang, Z.X.; Qiu, F.Z. Emp10 encodes a mitochondrial PPR protein that affects the cis-splicing of nad2 intron 1 and seed development in maize. Plant J. 2017, 91, 132–144. [Google Scholar] [CrossRef] [Green Version]
- Ren, X.M.; Pan, Z.Y.; Zhao, H.L.; Zhao, J.L.; Cai, M.J.; Li, J.; Zhang, Z.X.; Qiu, F.Z. EMPTY PERICARP11 serves as a factor for splicing of mitochondrial nad1 intron and is required to ensure proper seed development in maize. J. Exp. Bot. 2017, 68, 4571–4581. [Google Scholar] [CrossRef] [Green Version]
- Sun, F.; Xiu, Z.H.; Jiang, R.C.; Liu, Y.W.; Zhang, X.Y.; Yang, Y.Z.; Li, X.J.; Zhang, X.; Wang, Y.; Tan, B.C. The mitochondrial pentatricopeptide repeat protein EMP12 is involved in the splicing of three nad2 introns and seed development in maize. J. Exp. Bot. 2019, 70, 963–972. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, J.; Lu, G.; Liu, L.; Raihan, M.S.; Xu, J.T.; Jian, L.M.; Zhao, L.X.; Tran, T.M.; Zhang, Q.H.; Liu, J.; et al. The kernel size-related quantitative trait locus qKW9 encodes a pentatricopeptide repeat protein that affects photosynthesis and grain filling. Plant Physiol. 2020, 183, 1696–1709. [Google Scholar] [CrossRef]
- Xiu, Z.H.; Sun, F.; Shen, Y.; Zhang, X.Y.; Jiang, R.C.; Bonnard, G.; Zhang, J.H.; Tan, B.C. EMPTY PERICARP16 is required for mitochondrial nad2 intron 4 cis-splicing, complex I assembly and seed development in maize. Plant J. 2016, 85, 507–519. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, X.Y.; Huang, Z.Q.; Li, Y.Y.; Yang, Y.Z.; Sayyed, A.; Sun, F.; Gu, Z.Q.; Wang, X.M.; Tan, B.C. PPR-DYW protein EMP17 is required for mitochondrial RNA editing, complex III biogenesis, and seed development in maize. Front. Plant Sci. 2021, 12, 693272. [Google Scholar]
- Li, X.L.; Huang, W.L.; Yang, H.H.; Jiang, R.C.; Sun, F.; Wang, H.C.; Zhao, J.; Xu, C.H.; Tan, B.C. EMP18 functions in mitochondrial atp6 and cox2 transcript editing and is essential to seed development in maize. New Phytol. 2019, 221, 896–907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Liu, X.Y.; Yang, Y.Z.; Huang, J.; Sun, F.; Lin, J.S.; Gu, Z.Q.; Sayyed, A.; Xu, C.H.; Tan, B.C. Empty Pericarp21 encodes a novel PPR-DYW protein that is required for mitochondrial RNA editing at multiple sites, complexes I and V biogenesis, and seed development in maize. PLoS Genet. 2019, 15, e1008305. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.Z.; Ding, S.; Liu, X.Y.; Tang, J.J.; Wang, Y.; Sun, F.; Xu, C.H.; Tan, B.C. EMP32 is required for the cis-splicing of nad7 intron 2 and seed development in maize. RNA Biol. 2021, 18, 499–509. [Google Scholar] [CrossRef]
- Ren, Z.J.; Fan, K.J.; Fang, T.; Zhang, J.J.; Yang, L.; Wang, J.H.; Wang, G.Y.; Liu, Y.J. Maize Empty pericarp602 encodes a P-type PPR protein that is essential for seed development. Plant Cell Physiol. 2019, 60, 1734–1746. [Google Scholar] [CrossRef]
- Fan, K.J.; Ren, Z.J.; Zhang, X.F.; Liu, Y.; Fu, J.J.; Qi, C.L.; Tatar, W.; Rasmusson, A.G.; Wang, G.Y.; Liu, Y.J. The pentatricopeptide repeat protein EMP603 is required for the splicing of mitochondrial Nad1 intron 2 and seed development in maize. J. Exp. Bot. 2021, erab339. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.C.; Chen, Z.L.; Yang, Y.Z.; Sun, F.; Ding, S.; Li, X.L.; Xu, C.H.; Tan, B.C. PPR14 interacts with PPR-SMR1 and CRM protein Zm-mCSF1 to facilitate mitochondrial intron splicing in maize. Front. Plant Sci. 2020, 11, 732. [Google Scholar] [CrossRef]
- Liu, R.; Cao, S.K.; Sayyed, A.; Xu, C.H.; Sun, F.; Wang, X.M.; Tan, B.C. The mitochondrial pentatricopeptide repeat protein PPR18 is required for the cis-splicing of nad4 intron 1 and essential to seed development in maize. Int. J. Mol. Sci. 2020, 21, 4047. [Google Scholar]
- Liu, R.; Cao, S.K.; Sayyed, A.; Yang, H.H.; Zhao, J.; Wang, X.M.; Jia, R.X.; Sun, F.; Tan, B.C. The DYW-subgroup pentatricopeptide repeat protein PPR27 functions on editing of multiple mitochondrial transcripts and interacts with ZmMORF1 in maize. J. Exp. Bot. 2020, 71, 5495–5505. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.H.; Xiu, Z.H.; Wang, L.; Cao, S.K.; Li, X.L.; Sun, F.; Tan, B.C. Two pentatricopeptide repeat proteins are required for the splicing of nad5 introns in maize. Front. Plant Sci. 2020, 11, 732. [Google Scholar] [CrossRef]
- Chen, Z.L.; Wang, H.C.; Shen, J.Y.; Sun, F.; Wang, M.D.; Xu, C.H.; Tan, B.C. PPR-SMR1 is required for the splicing of multiple mitochondrial introns, interacts with Zm-mCSF1, and is essential for seed development in maize. J. Exp. Bot. 2019, 70, 5245–5258. [Google Scholar] [CrossRef]
- Sosso, D.; Canut, M.; Gendrot, G.; Dedieu, A.; Chambrier, P.; Barkan, A.; Consonni, G.; Rogowsky, P.M. PPR8522 encodes a chloroplast-targeted pentatricopeptide repeat protein necessary for maize embryogenesis and vegetative development. J. Exp. Bot. 2012, 63, 5843–5857. [Google Scholar] [CrossRef] [Green Version]
- Yuan, N.N.; Wang, J.C.; Zhou, Y.; An, D.; Xiao, Q.; Wang, W.Q.; Wu, Y.R. EMB-7L is required for embryogenesis and plant development in maize involved in RNA splicing of multiple chloroplast genes. Plant Sci. 2019, 287, 110203. [Google Scholar] [CrossRef] [PubMed]
- De Longevialle, A.F.; Meyer, E.H.; Andres, C.; Taylor, N.L.; Lurin, C.; Millar, A.H.; Small, I.D. The pentatricopeptide repeat gene OTP43 is required for trans-splicing of the mitochondrial nad1 intron 1 in Arabidopsis thaliana. Plant Cell 2007, 19, 3256–3265. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.; Han, J.H.; Park, Y.I.; Colas des Francs-Small, C.; Small, I.; Kang, H. The mitochondrial pentatricopeptide repeat protein PPR19 is involved in the stabilization of NADH dehydrogenase 1 transcripts and is crucial for mitochondrial function and Arabidopsis thaliana development. New Phytol. 2017, 215, 202–216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, Y.; Huang, J.Y.; Zhong, S.; Gu, H.Y.; He, S.; Qu, L.J. Novel DYW-type pentatricopeptide repeat (PPR) protein BLX controls mitochondrial RNA editing and splicing essential for early seed development of Arabidopsis. J. Genet. Genom. 2018, 45, 155–168. [Google Scholar] [CrossRef]
- Lu, Y.Q.; Li, C.; Wang, H.; Chen, H.; Berg, H.; Xia, Y.J. AtPPR2, an Arabidopsis pentatricopeptide repeat protein, binds to plastid 23S rRNA and plays an important role in the first mitotic division during gametogenesis and in cell proliferation during embryogenesis. Plant J. 2011, 67, 13–25. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.W.; An, Y.Q.; Qi, Z.; Xiao, J.W. PPR protein early chloroplast development 2 is essential for chloroplast development at the early stage of Arabidopsis development. Plant Sci. 2021, 308, 110908. [Google Scholar] [CrossRef]
- Aryamanesh, N.; Ruwe, H.; Sanglard, L.V.P.; Eshraghi, L.; Bussell, J.D.; Howell, K.A.; Small, I.; Colas des Francs-Small, C. The pentatricopeptide repeat protein EMB2654 is essential for trans-splicing of a chloroplast small ribosomal subunit transcript. Plant Physiol. 2017, 173, 1164–1176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marchetti, F.; Cainzos, M.; Shevtsov, S.; Córdoba, J.P.; Sultan, L.D.; Brennicke, A.; Takenaka, M.; Pagnussat, G.; Ostersetzer-Biran, O.; Zabaleta, E. Mitochondrial pentatricopeptide repeat protein, EMB2794, plays a pivotal role in NADH dehydrogenase subunit nad2 mRNA maturation in Arabidopsis thaliana. Plant Cell Physiol. 2020, 61, 1080–1094. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.H.; Liu, N.Y.; Tang, Z.S.; Liu, J.; Yang, W.C. Arabidopsis GLUTAMINE-RICH PROTEIN23 is essential for early embryogenesis and encodes a novel nuclear PPR motif protein that interacts with RNA polymerase II subunit III. Plant Cell 2006, 18, 815–830. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, P.; Wang, F.; Li, N.; Shi, D.Q.; Yang, W.C. Pentatricopeptide repeat protein MID1 modulates nad2 intron 1 splicing and Arabidopsis development. Sci Rep. 2020, 10, 2008. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Xiao, J.W.; Li, Y.Q.; Su, B.D.; Xu, H.M.; Shan, X.Y.; Song, C.W.; Xie, J.B.; Li, R.L. PDM3, a pentatricopeptide repeat-containing protein, affects chloroplast development. J. Exp. Bot. 2017, 68, 5615–5627. [Google Scholar] [CrossRef]
- Zhang, L.; Qi, Y.Z.; Wu, M.M.; Zhao, L.; Zhao, Z.C.; Lei, C.L.; Hao, Y.Y.; Yu, X.W.; Sun, Y.L.; Zhang, X.; et al. Mitochondrion-targeted PENTATRICOPEPTIDE REPEAT5 is required for cis-splicing of nad4 intron 3 and endosperm development in rice. Crop J. 2021, 9, 282–296. [Google Scholar] [CrossRef]
- Leu, K.C.; Hsieh, M.H.; Wang, H.J.; Hsieh, H.L.; Jauh, G.Y. Distinct role of Arabidopsis mitochondrial P-type pentatricopeptide repeat protein-modulating editing protein, PPME, in nad1 RNA editing. RNA Biol. 2016, 13, 593–604. [Google Scholar] [CrossRef] [Green Version]
- Small, I.D.; Schallenberg-Rudinger, M.; Takenaka, M.; Mireau, H.; Ostersetzer-Biran, O. Plant organellar RNA editing: What 30 years of research has revealed. Plant J. 2020, 101, 1040–1056. [Google Scholar] [CrossRef] [PubMed]
- Blanc, V.; Litvak, S.; Araya, A. RNA editing in wheat mitochondria proceeds by a deamination mechanism. FEBS Lett. 1995, 373, 56–60. [Google Scholar] [CrossRef] [Green Version]
- Bhattacharya, S.; Navaratnam, N.; Morrison, J.R.; Scott, J.; Taylor, W.R. Cytosine nucleoside/nucleotide deaminases and apolipoprotein B mRNA editing. Trends Biochem. Sci. 1994, 19, 105–106. [Google Scholar] [CrossRef]
- Boussardon, C.; Avon, A.; Kindgren, P.; Bond, C.S.; Challenor, M.; Lurin, C.; Small, I. The cytidine deaminase signature HxE(x)nCxxC of DYW1 binds zinc and is necessary for RNA editing of ndhD-1. New Phytol. 2014, 203, 1090–1095. [Google Scholar] [CrossRef]
- Hayes, M.L.; Dang, K.N.; Diaz, M.F.; Mulligan, R.M. A conserved glutamate residue in the C-terminal deaminase domain of pentatricopeptide repeat proteins is required for RNA editing activity. J. Biol. Chem. 2015, 290, 10136–10142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oldenkott, B.; Yang, Y.Y.; Lesch, E.; Knoop, V.; Schallenberg-Rüdinger, M. Plant-type pentatricopeptide repeat proteins with a DYW domain drive C-to-U RNA editing in Escherichia coli. Commun. Biol. 2019, 2, 85. [Google Scholar] [CrossRef] [PubMed]
- Hayes, M.L.; Santibanez, P.I. A plant pentatricopeptide repeat protein with a DYW-deaminase domain is sufficient for catalyzing C-to-U RNA editing in vitro. J. Biol. Chem. 2020, 295, 3497–3505. [Google Scholar] [CrossRef] [PubMed]
- Takenaka, M.; Takenaka, S.; Barthel, T.; Frink, B.; Haag, S.; Verbitskiy, D.; Oldenkott, B.; Schallenberg-Rüdinger, M.; Feiler, C.G.; Weiss, M.S.; et al. DYW domain structures imply an unusual regulation principle in plant organellar RNA editing catalysis. Nat. Catal. 2021, 4, 510–522. [Google Scholar] [CrossRef]
- Boussardon, C.; Salone, V.; Avon, A.; Berthomé, R.; Hammani, K.; Okuda, K.; Shikanai, T.; Small, I.; Lurin, C. Two interacting proteins are necessary for the editing of the NdhD-1 site in Arabidopsis plastids. Plant Cell 2012, 24, 3684–3694. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andres-Colas, N.; Zhu, Q.; Takenaka, M.; De Rybel, B.; Weijers, D.; Van Der Straeten, D. Multiple PPR protein interactions are involved in the RNA editing system in Arabidopsis mitochondria and plastids. Proc. Natl. Acad. Sci. USA 2017, 114, 8883–8888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malbert, B.; Burger, M.; Lopez-Obando, M.; Baudry, K.; Launay-Avon, A.; Härtel, B.; Verbitskiy, D.; Jörg, A.; Berthomé, R.; Lurin, C.; et al. The analysis of the editing defects in the dyw2 mutant provides new clues for the prediction of RNA targets of Arabidopsis E+-class PPR proteins. Plants 2020, 9, 280. [Google Scholar] [CrossRef] [Green Version]
- Guillaumot, D.; Lopez-Obando, M.; Baudry, K.; Avon, A.; Rigaill, G.; Falcon de Longevialle, A.; Broche, B.; Takenaka, M.; Berthomé, R.; De Jaeger, G.; et al. Two interacting PPR proteins are major Arabidopsis editing factors in plastid and mitochondria. Proc. Natl. Acad. Sci. USA 2017, 114, 8877–8882. [Google Scholar] [CrossRef] [Green Version]
- Bentolila, S.; Heller, W.P.; Sun, T.; Babina, A.M.; Friso, G.; van Wijk, K.J.; Hanson, M.R. RIP1, a member of an Arabidopsis protein family, interacts with the protein RARE1 and broadly affects RNA editing. Proc. Natl. Acad. Sci. USA 2012, 109, e1453–e1461. [Google Scholar] [CrossRef] [Green Version]
- Härtel, B.; Zehrmann, A.; Verbitskiy, D.; van der Merwe, J.A.; Brennicke, A.; Takenaka, M. MEF10 is required for RNA editing at nad2-842 in mitochondria of Arabidopsis thaliana and interacts with MORF8. Plant Mol. Biol. 2013, 81, 337–346. [Google Scholar] [CrossRef]
- Brehme, N.; Bayer-Császár, E.; Glass, F.; Takenaka, M. The DYW subgroup PPR protein MEF35 targets RNA editing sites in the mitochondrial rpl16, nad4 and cob mRNAs in Arabidopsis thaliana. PLoS ONE 2015, 10, e0140680. [Google Scholar] [CrossRef] [Green Version]
- Barkan, A.; Rojas, M.; Fujii, S.; Yap, A.; Chong, Y.S.; Bond, C.S.; Small, I. A combinatorial amino acid code for RNA recognition by pentatricopeptide repeat proteins. PLoS Genet. 2012, 8, e1002910. [Google Scholar] [CrossRef]
- Huang, C.; Li, Z.R.; Yu, Q.B.; Ye, L.S.; Cui, Y.L.; Molloy, D.P.; Yang, Z.N. MORF2 tightly associates with MORF9 to regulate chloroplast RNA editing in Arabidopsis. Plant Sci. 2019, 278, 64–69. [Google Scholar] [CrossRef]
- Yan, J.J.; Zhang, Q.X.; Guan, Z.Y.; Wang, Q.; Li, L.; Ruan, F.Y.; Lin, R.C.; Zou, T.T.; Yin, P. MORF9 increases the RNA-binding activity of PLS-type pentatricopeptide repeat protein in plastid RNA editing. Nat. Plants 2017, 3, 17037. [Google Scholar] [CrossRef]
- Brown, G.G.; Colas des Francs-Small, C.; Ostersetzer-Biran, O. Group II intron splicing factors in plant mitochondria. Front. Plant Sci. 2014, 5, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmitz-Linneweber, C.; Lampe, M.K.; Sultan, L.D.; Ostersetzer-Biran, O. Organellar maturases: A window into the evolution of the spliceosome. Biochim. Biophys. Acta BBA Bioenerg. 2015, 1847, 798–808. [Google Scholar] [CrossRef] [Green Version]
- Colas des Francs-Small, C.; Falcon de Longevialle, A.; Li, Y.H.; Lowe, E.; Tanz, S.K.; Smith, C.; Bevan, M.W.; Small, I. The pentatricopeptide repeat proteins TANG2 and ORGANELLE TRANSCRIPT PROCESSING439 are involved in the splicing of the multipartite nad5 transcript encoding a subunit of mitochondrial complex I. Plant Physiol. 2014, 165, 1409–1416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hsieh, W.Y.; Liao, J.C.; Chang, C.Y.; Harrison, T.; Boucher, C.; Hsieh, M.H. The SLOW GROWTH3 pentatricopeptide repeat protein is Required for the splicing of mitochondrial NADH dehydrogenase subunit7 intron 2 in Arabidopsis. Plant Physiol. 2015, 168, 490–501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haïli, N.; Planchard, N.; Arnal, N.; Quadrado, M.; Vrielynck, N.; Dahan, J.; Colas des Francs-Small, C.; Mireau, H. The MTL1 pentatricopeptide repeat protein is required for both translation and splicing of the mitochondrial NADH DEHYDROGENASE SUBUNIT7 mRNA in Arabidopsis. Plant Physiol. 2016, 170, 354–366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiu, Z.H.; Peng, L.; Wang, Y.; Yang, H.H.; Sun, F.; Wang, X.M.; Cao, S.K.; Jiang, R.C.; Wang, L.; Chen, B.Y.; et al. Empty pericarp24 and Empty pericarp25 are required for the splicing of mitochondrial introns, complex I assembly, and seed development in maize. Front. Plant Sci. 2020, 11, 608550. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.Y.; Wang, Y.L.; Wu, M.M.; Zhu, X.P.; Teng, X.; Sun, Y.L.; Zhu, J.P.; Zhang, Y.Y.; Jing, R.N.; Lei, J.; et al. The nuclear-localized PPR protein OsNPPR1 is important for mitochondrial function and endosperm development in rice. J. Exp. Bot. 2019, 70, 4705–4720. [Google Scholar] [CrossRef] [Green Version]
- Wu, M.M.; Ren, Y.L.; Cai, M.H.; Wang, Y.L.; Zhu, S.S.; Zhu, J.P.; Hao, Y.Y.; Teng, X.; Zhu, X.P.; Jing, R.N.; et al. Rice FLOURY ENDOSPERM 10 encodes a pentatricopeptide repeat protein that is essential for the trans-splicing of mitochondrial nad1 intron 1 and endosperm development. New Phytol. 2020, 223, 736–750. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.Y.; Liu, L.L.; Yu, Y.F.; Zhu, J.P.; Gao, H.; Wang, Y.H.; Wan, J.M. Lose-of-function of a rice nucleolus-localized pentatricopeptide repeat protein is responsible for the floury endosperm14 mutant phenotypes. Rice 2019, 12, 100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weißenberger, S.; Soll, J.; Carrie, C. The PPR protein SLOW GROWTH 4 is involved in editing of nad4 and affects the splicing of nad2 intron 1. Plant Mol. Biol. 2016, 93, 355–368. [Google Scholar] [CrossRef]
- Ichinose, M.; Tasaki, E.; Sugita, C.; Sugita, M. A PPR-DYW protein is required for splicing of a group II intron of cox1 pre-mRNA in Physcomitrella patens. Plant J. 2012, 70, 271–278. [Google Scholar] [CrossRef]
- Schmitz-Linneweber, C.; Williams-Carrier, R.E.; Williams-Voelker, P.M.; Kroeger, T.S.; Vichas, A.; Barkan, A. A pentatricopeptide repeat protein facilitates the trans-splicing of the maize chloroplast rps12 pre-mRNA. Plant Cell 2006, 18, 2650–2663. [Google Scholar] [CrossRef] [Green Version]
- Tadini, L.; Ferrari, R.; Lehniger, M.K.; Mizzotti, C.; Moratti, F.; Resentini, F.; Colombo, M.; Costa, A.; Masiero, S.; Pesaresi, P. Trans-splicing of plastid rps12 transcripts, mediated by AtPPR4, is essential for embryo patterning in Arabidopsis thaliana. Planta 2018, 248, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Ostheimer, G.J.; Williams-Carrier, R.; Belcher, S.; Osborne, E.; Gierke, J.; Barkan, A. Group II intron splicing factors derived by diversification of an ancient RNA-binding domain. EMBO J. 2003, 22, 3919–3929. [Google Scholar] [CrossRef] [Green Version]
- Jenkins, B.D.; Barkan, A. Recruitment of a peptidyl-tRNA hydrolase as a facilitator of group II intron splicing in chloroplasts. EMBO J. 2001, 20, 872–879. [Google Scholar] [CrossRef]
- Asakura, Y.; Galarneau, E.; Watkins, K.P.; Barkan, A.; van Wijk, K.J. Chloroplast RH3 DEAD Box RNA helicases in maize and Arabidopsis function in splicing of specific group II introns and affect chloroplast ribosome biogenesis. Plant Physiol. 2012, 159, 961–974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, C.D.; Aubé, F.; Quadrado, M.; Dargel-Graffin, C.; Mireau, H. Three new pentatricopeptide repeat proteins facilitate the splicing of mitochondrial transcripts and complex I biogenesis in Arabidopsis. J. Exp. Bot. 2018, 69, 5131–5140. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Fourdin, R.; Quadrado, M.; Dargel-Graffin, C.; Tollete, D. Rerouting of ribosomal proteins into splicing in plant organelles. Proc. Natl. Acad. Sci. USA 2020, 117, 29979–29987. [Google Scholar] [CrossRef] [PubMed]
- Khrouchtchova, A.; Monde, R.A.; Barkan, A. A short PPR protein required for the splicing of specific group II introns in angiosperm chloroplasts. RNA 2012, 18, 1197–1209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ke, J.Y.; Chen, R.Z.; Ban, T.; Zhou, X.E.; Gu, X.; Tan, M.H.E.; Chen, C.; Kang, Y.Y.; Brunzelle, J.S.; Zhu, J.K.; et al. Structural basis for RNA recognition by a dimeric PPR-protein complex. Nat. Struct. Mol. Biol. 2013, 20, 1377–1382. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Park, S.J.; Colas des Francs-Small, C.; Whitby, M.; Small, I.; Kang, H. The coordinated action of PPR4 and EMB 2654 on each intron half mediates trans-splicing of rps12 transcripts in plant chloroplasts. Plant J. 2019, 100, 1193–1207. [Google Scholar] [CrossRef]
- Fedorova, O.; Solem, A.; Pyle, A.M. Protein-facilitated folding of group II intron ribozymes. J. Mol. Biol. 2010, 397, 799–813. [Google Scholar] [CrossRef] [Green Version]
- Yan, J.J.; Yao, Y.Y.; Hong, S.X.; Yang, Y.; Shen, C.C.; Zhang, Q.X.; Zhang, D.L.; Zou, T.T.; Yin, P. Delineation of pentatricopeptide repeat codes for target RNA prediction. Nucleic Acids Res. 2019, 47, 3728–3738. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Species | Subcellular Localization | Mutant Phenotype | Protein Name | PPR Class | Function(s) | References |
|---|---|---|---|---|---|---|
| Maize | Mitochondrion | dek | DEK2 | P | RNA splicing, nad1 intron 1 | [52] |
| Mitochondrion | dek | DEK10 | PLS | RNA editing, nad3-61, 62, and cox2-550 | [53] | |
| Mitochondrion | dek | DEK35 | P | RNA splicing, nad4 intron 1 | [54] | |
| Mitochondrion | dek | DEK36 | PLS | RNA editing, atp4-59, nad7-383, and ccmFN-302 | [55] | |
| Mitochondrion | dek | DEK37 | P | RNA splicing, nad2 intron 1 | [56] | |
| Mitochondrion | dek | DEK39 | PLS | RNA editing, nad3-247 and nad3-275 | [57] | |
| Mitochondrion | dek | DEK40 | PLS | RNA editing, cox3-314, nad2-26, and nad5-1916 | [58] | |
| Mitochondrion | dek | DEK41/ DEK43 | P | RNA splicing, nad4 intron 1 and 3 | [59,60] | |
| Mitochondrion | dek | DEK46 | PLS | RNA editing, D5-C22 of nad7 intron 3 and 4 | [61] | |
| Mitochondrion | dek | DEK53 | PLS | RNA editing, multiples sites | [62] | |
| Mitochondrion | dek | DEK55 | PLS | RNA splicing, nad4 intron 1 and 3;RNA editing, multiple sites | [63] | |
| Mitochondrion | dek | DEK605 | PLS | RNA editing, nad1-608 | [64] | |
| Mitochondrion | smk | SMK1 | PLS | RNA editing, nad7-836 | [65] | |
| Mitochondrion | smk | SMK4 | PLS | RNA editing, cox1-1489 | [66] | |
| Mitochondrion | smk | SMK6 | PLS | RNA editing, nad1-740, nad4L-110, nad7-739, and mttB-138, 139 | [67] | |
| Mitochondrion | smk | ZmSMK9 | P | RNA splicing, nad5 intron 1 and 4 | [68] | |
| Mitochondrion | smk | PPR2263 | PLS | RNA editing, nad5-1550 and cob-908 | [69] | |
| Mitochondrion | smk | MPPR6 | P | Translation, rps3 mRNA | [70] | |
| Mitochondrion | dek/smk | PPR20 | P | RNA splicing, nad2 intron 3 | [71] | |
| Mitochondrion | smk | PPR78 | P | RNA stabilization, nad5 mature mRNA | [72] | |
| Mitochondrion | emp | EMP4 | P | Expression of mitochondrial transcripts | [73] | |
| Mitochondrion | emp | EMP5 | PLS | RNA editing, multiple sites | [74] | |
| Mitochondrion | emp | EMP7 | PLS | RNA editing, ccmFN-1553 | [75] | |
| Mitochondrion | emp | EMP8 | P | RNA splicing, nad1 intron 4, nad2 intron 1, and nad4 intron 1 | [76] | |
| Mitochondrion | emp | EMP9 | PLS | RNA editing, ccmB-43 and rps4-335 | [77] | |
| Mitochondrion | emp | EMP10 | P | RNA splicing, nad2 intron 1 | [78] | |
| Mitochondrion | emp | EMP11 | P | RNA splicing, nad1 intron 1, 2, 3, and 4 | [79] | |
| Mitochondrion | emp | EMP12 | P | RNA splicing, nad2 intron 1, 2, and 4 | [80] | |
| Chloroplast | smk | qKW9 | PLS | RNA editing, NdhB-246 | [81] | |
| Mitochondrion | emp | EMP16 | P | RNA splicing, nad2 intron 4 | [82] | |
| Mitochondrion | emp | EMP17 | PLS | RNA editing, ccmFC-799 and nad2-677 | [83] | |
| Mitochondrion | emp | EMP18 | PLS | RNA editing, atp6-635 and cox2-449 | [84] | |
| Mitochondrion | emp | EMP21 | PLS | RNA editing, multiple sites | [85] | |
| Mitochondrion | emp | EMP32 | P | RNA splicing, nad7 intron 2 | [86] | |
| Mitochondrion | emp | EMP602 | P | RNA splicing, nad4 intron 1 and 3 | [87] | |
| Mitochondrion | emp | EMP603 | P | RNA splicing, nad1 intron 2 | [88] | |
| Mitochondrion | emp | PPR14 | P | RNA splicing, nad2 intron 3, nad7 intron 1 and 2 | [89] | |
| Mitochondrion | emp | PPR18 | P | RNA splicing, nad4 intron 1 | [90] | |
| Mitochondrion | emp | PPR27 | PLS | RNA editing, multiple sites | [91] | |
| Mitochondrion | emp | PPR101 | P | RNA splicing, nad5 intron 1 and 2 | [92] | |
| Mitochondrion | emp | PPR231 | P | RNA splicing, nad5 intron 1, 2, 3 and nad2 intron 3 | [92] | |
| Mitochondrion | emp | PPR-SMR1 | P | RNA splicing, multiple introns | [93] | |
| Chloroplast | emb | PPR8522 | P | RNA transcription, nearly all chloroplast-encoded genes | [94] | |
| Chloroplast | emb | EMB-7L | P | RNA splicing, multiple introns | [95] | |
| Arabidopsis | Mitochondrion | dek | OTP43 | P | RNA splicing, nad1 intron 1 | [96] |
| Mitochondrion | smk | PPR19 | P | RNA stabilization, nad1 mature mRNA | [97] | |
| Mitochondrion | emp | BLX | PLS | RNA editing, multiple sites; RNA splicing, nad1 intron 4 and nad2 intron 1 | [98] | |
| Chloroplast | emb | AtPPR2 | P | RNA translation | [99] | |
| Chloroplast | emb | ECD2 | P | RNA splicing, ndhA, ycf3 intron 1, rps12 intron 2 and clpp intron 2 | [100] | |
| Chloroplast | emb | EMB2654 | P | RNA splicing, rps12 intron 1 | [101] | |
| Mitochondrion | emb | EMB2794 | P | RNA splicing, nad2 intron 2 | [102] | |
| Nucleus | emb | GRP23 | P | RNA transcription | [103] | |
| Mitochondrion | emb | MID1 | P | RNA splicing, nad2 intron 1 | [104] | |
| Chloroplast | emb | PMD3 | P | RNA splicing, trnA, ndhB, and clpP-1 | [105] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Sun, M.; Liu, S.; Teng, Q.; Li, S.; Jiang, Y. Functions of PPR Proteins in Plant Growth and Development. Int. J. Mol. Sci. 2021, 22, 11274. https://doi.org/10.3390/ijms222011274
Li X, Sun M, Liu S, Teng Q, Li S, Jiang Y. Functions of PPR Proteins in Plant Growth and Development. International Journal of Molecular Sciences. 2021; 22(20):11274. https://doi.org/10.3390/ijms222011274
Chicago/Turabian StyleLi, Xiulan, Mengdi Sun, Shijuan Liu, Qian Teng, Shihui Li, and Yueshui Jiang. 2021. "Functions of PPR Proteins in Plant Growth and Development" International Journal of Molecular Sciences 22, no. 20: 11274. https://doi.org/10.3390/ijms222011274





