The miRNome of Depression
Abstract
:1. Introduction
2. miRNA Biogenesis, Function and Regulatory Properties
3. Studies Investigating Depression-Related miRNAs in the Brain
3.1. Brodmann Area 9 (BA9)
3.2. Brodmann Area 10 (BA10)
3.3. Brodmann Area 44 (BA44)
3.4. Anterior Cingulate Cortex (ACC)
3.5. Other Brain Regions
4. miRNA Studies of Depression and Antidepressant Treatments Investigating Blood and Body
4.1. Whole Blood
4.2. Serum/Plasma and Cerebrospinal Fluid (CSF)
4.3. Peripheral Blood Mononuclear Cells (PBMCs)
4.4. Extracellular Vesicles
- 1.
- Large extracellular vesicles (L-EVs), including apoptotic bodies, large oncosomes and microvesicles;
- 2.
- Small extracellular vesicles (S-EVs), including exosomes;
- 3.
- Extracellular particles (EPs), including exomeres and chromatimers.
4.5. Brain-Enriched miRNAs Found in Periphery
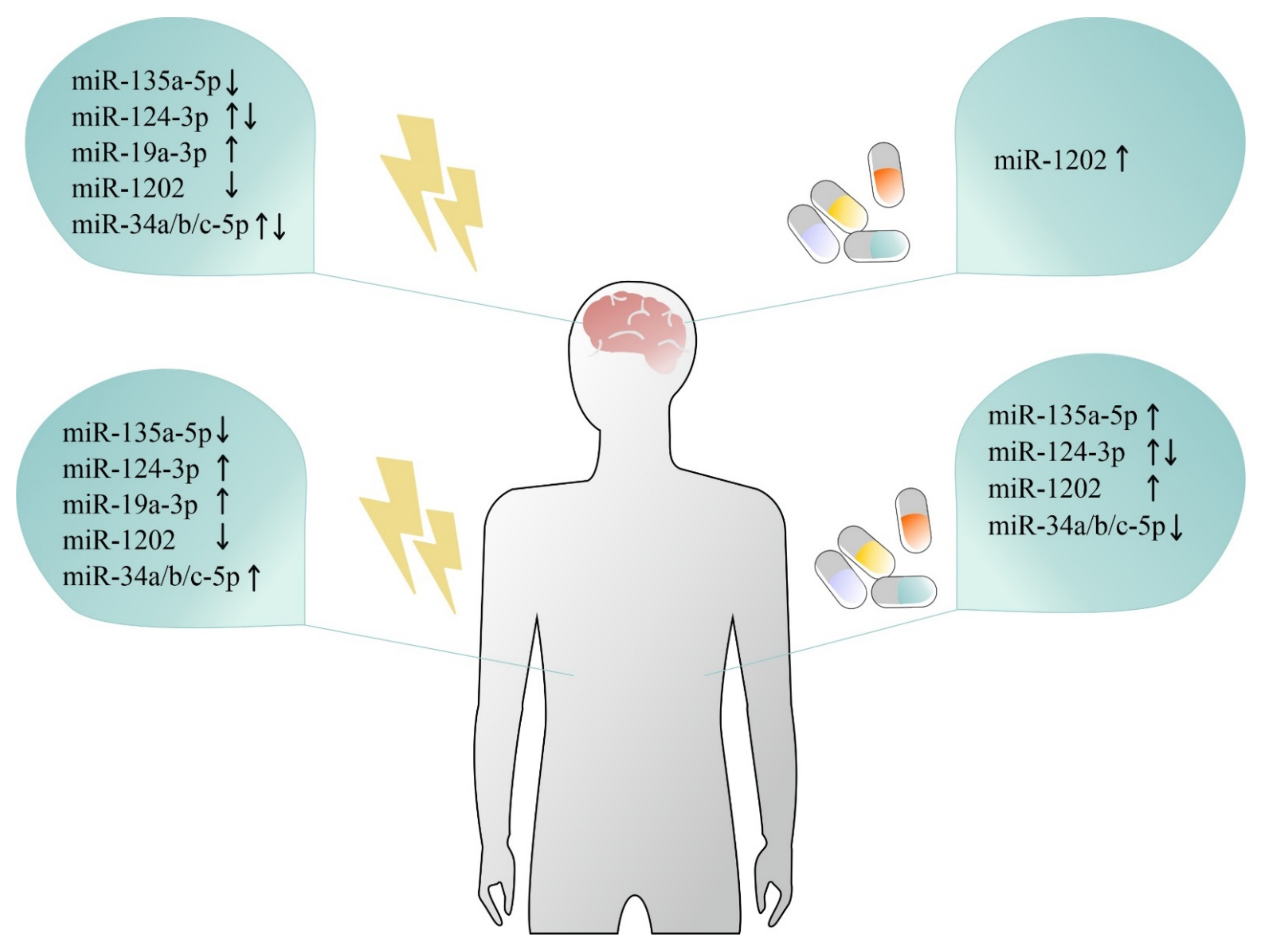
5. From Changes in the Brain to Changes in the Periphery: A Summary
5.1. miR-1202
5.2. miR-124-3p
5.3. miR-19a-3p
5.4. miR-135a-5p
5.5. miR-34 Family
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Manji, H.K.; Drevets, W.C.; Charney, D.S. The Cellular Neurobiology of Depression. Nat. Med. 2001, 7, 541–547. [Google Scholar] [CrossRef]
- Jeon, S.; Kim, Y.-K. Molecular Neurobiology and Promising New Treatment in Depression. Int. J. Mol. Sci. 2016, 17, 381. [Google Scholar] [CrossRef] [Green Version]
- Albert, P.R.; Benkelfat, C.; Descarries, L. The Neurobiology of Depression—Revisiting the Serotonin Hypothesis. I. Cellular and Molecular Mechanisms. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 2378–2381. [Google Scholar] [CrossRef] [Green Version]
- Chandley, M.J.; Ordway, G.A. Noradrenergic Dysfunction in Depression and Suicide. In The Neurobiological Basis of Suicide; Frontiers in Neuroscience; Dwivedi, Y., Ed.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2012. [Google Scholar]
- Belujon, P.; Grace, A.A. Dopamine System Dysregulation in Major Depressive Disorders. Int. J. Neuropsychopharmacol. 2017, 20, 1036–1046. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Zhao, J.; Guo, W. Emotional Roles of Mono-Aminergic Neurotransmitters in Major Depressive Disorder and Anxiety Disorders. Front. Psychol. 2018, 9, 2201. [Google Scholar] [CrossRef]
- Martin-Hernández, D.; Pereira, M.P.; Tendilla-Beltrán, H.; Madrigal, J.L.M.; García-Bueno, B.; Leza, J.C.; Caso, J.R. Modulation of Monoaminergic Systems by Antidepressants in the Frontal Cortex of Rats After Chronic Mild Stress Exposure. Mol. Neurobiol. 2019, 56, 7522–7533. [Google Scholar] [CrossRef]
- Nestler, E.J.; Carlezon, W.A. The Mesolimbic Dopamine Reward Circuit in Depression. Biol. Psychiatry 2006, 59, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Michely, J.; Eldar, E.; Martin, I.M.; Dolan, R.J. A Mechanistic Account of Serotonin’s Impact on Mood. Nat. Commun. 2020, 11, 2335. [Google Scholar] [CrossRef] [PubMed]
- Rush, A.J.; Trivedi, M.H.; Wisniewski, S.R.; Stewart, J.W.; Nierenberg, A.A.; Thase, M.E.; Ritz, L.; Biggs, M.M.; Warden, D.; Luther, J.F.; et al. Bupropion-SR, Sertraline, or Venlafaxine-XR after Failure of SSRIs for Depression. N. Engl. J. Med. 2006, 354, 1231–1242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Carlo, V.; Calati, R.; Serretti, A. Socio-Demographic and Clinical Predictors of Non-Response/Non-Remission in Treatment Resistant Depressed Patients: A Systematic Review. Psychiatry Res. 2016, 240, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Uher, R. The Implications of Gene–Environment Interactions in Depression: Will Cause Inform Cure? Mol. Psychiatry 2008, 13, 1070–1078. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, E.; Tsai, S.-J. Epigenetics and Depression: An Update. Psychiatry Investig. 2019, 16, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Kwong, A.S.F.; López-López, J.A.; Hammerton, G.; Manley, D.; Timpson, N.J.; Leckie, G.; Pearson, R.M. Genetic and Environmental Risk Factors Associated With Trajectories of Depression Symptoms From Adolescence to Young Adulthood. JAMA Netw. Open 2019, 2, e196587. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McIntosh, A.M.; Sullivan, P.F.; Lewis, C.M. Uncovering the Genetic Architecture of Major Depression. Neuron 2019, 102, 91–103. [Google Scholar] [CrossRef]
- McEwen, B.S.; Bowles, N.P.; Gray, J.D.; Hill, M.N.; Hunter, R.G.; Karatsoreos, I.N.; Nasca, C. Mechanisms of Stress in the Brain. Nat. Neurosci. 2015, 18, 1353–1363. [Google Scholar] [CrossRef]
- Howard, D.M.; Adams, M.J.; Clarke, T.-K.; Hafferty, J.D.; Gibson, J.; Shirali, M.; Coleman, J.R.I.; Hagenaars, S.P.; Ward, J.; Wigmore, E.M.; et al. Genome-Wide Meta-Analysis of Depression Identifies 102 Independent Variants and Highlights the Importance of the Prefrontal Brain Regions. Nat. Neurosci. 2019, 22, 343–352. [Google Scholar] [CrossRef] [Green Version]
- Khan, A.R.; Geiger, L.; Wiborg, O.; Czéh, B. Stress-Induced Morphological, Cellular and Molecular Changes in the Brain—Lessons Learned from the Chronic Mild Stress Model of Depression. Cells 2020, 9, 1026. [Google Scholar] [CrossRef] [Green Version]
- Dziedzicka-Wasylewska, M.; Solich, J.; Korlatowicz, A.; Faron-Górecka, A. What Do the Animal Studies of Stress Resilience Teach Us? Cells 2021, 10, 1630. [Google Scholar] [CrossRef]
- Lee, R.C.; Feinbaum, R.L.; Ambros, V. The C. elegans Heterochronic Gene Lin-4 Encodes Small RNAs with Antisense Complementarity to Lin-14. Cell 1993, 75, 843–854. [Google Scholar] [CrossRef]
- Fire, A.; Xu, S.; Montgomery, M.K.; Kostas, S.A.; Driver, S.E.; Mello, C.C. Potent and Specific Genetic Interference by Double-Stranded RNA in Caenorhabditis elegans. Nature 1998, 391, 806–811. [Google Scholar] [CrossRef]
- Kozomara, A.; Griffiths-Jones, S. MiRBase: Annotating High Confidence MicroRNAs Using Deep Sequencing Data. Nucleic Acids Res. 2014, 42, D68–D73. [Google Scholar] [CrossRef] [Green Version]
- Kozomara, A.; Birgaoanu, M.; Griffiths-Jones, S. MiRBase: From MicroRNA Sequences to Function. Nucleic Acids Res. 2019, 47, D155–D162. [Google Scholar] [CrossRef] [PubMed]
- Artigas, F.; Celada, P.; Bortolozzi, A. Can We Increase the Speed and Efficacy of Antidepressant Treatments? Part II. Glutamatergic and RNA Interference Strategies. Eur. Neuropsychopharmacol. 2018, 28, 457–482. [Google Scholar] [CrossRef] [PubMed]
- Xue, Q.; Yu, C.; Wang, Y.; Liu, L.; Zhang, K.; Fang, C.; Liu, F.; Bian, G.; Song, B.; Yang, A.; et al. MiR-9 and MiR-124 Synergistically Affect Regulation of Dendritic Branching via the AKT/GSK3β Pathway by Targeting Rap2a. Sci. Rep. 2016, 6, 26781. [Google Scholar] [CrossRef] [PubMed]
- Friedman, R.C.; Farh, K.K.-H.; Burge, C.B.; Bartel, D.P. Most Mammalian MRNAs Are Conserved Targets of MicroRNAs. Genome Res. 2008, 19, 92–105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, Y.-K.; Kim, B.; Kim, V.N. Re-Evaluation of the Roles of DROSHA, Exportin 5, and DICER in MicroRNA Biogenesis. Proc. Natl. Acad. Sci. USA 2016, 113, E1881–E1889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martinez, N.J.; Gregory, R.I. Argonaute2 Expression Is Post-Transcriptionally Coupled to MicroRNA Abundance. RNA 2013, 19, 605–612. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Brien, J.; Hayder, H.; Zayed, Y.; Peng, C. Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation. Front. Endocrinol. 2018, 9, 402. [Google Scholar] [CrossRef] [Green Version]
- Riffo-Campos, Á.; Riquelme, I.; Brebi-Mieville, P. Tools for Sequence-Based MiRNA Target Prediction: What to Choose? Int. J. Mol. Sci. 2016, 17, 1987. [Google Scholar] [CrossRef] [Green Version]
- O’Connor, R.M.; Dinan, T.G.; Cryan, J.F. Little Things on Which Happiness Depends: MicroRNAs as Novel Therapeutic Targets for the Treatment of Anxiety and Depression. Mol. Psychiatry 2012, 17, 359–376. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Zhang, H.; Zhang, J.; Xing, J. Reciprocal Regulation between MRNA and MicroRNA Enables a Bistable Switch That Directs Cell Fate Decisions. FEBS Lett. 2016, 590, 3443–3455. [Google Scholar] [CrossRef] [Green Version]
- Rajman, M.; Schratt, G. MicroRNAs in Neural Development: From Master Regulators to Fine-Tuners. Development 2017, 144, 2310–2322. [Google Scholar] [CrossRef] [Green Version]
- Venø, M.T.; Reschke, C.R.; Morris, G.; Connolly, N.M.C.; Su, J.; Yan, Y.; Engel, T.; Jimenez-Mateos, E.M.; Harder, L.M.; Pultz, D.; et al. A Systems Approach Delivers a Functional MicroRNA Catalog and Expanded Targets for Seizure Suppression in Temporal Lobe Epilepsy. Proc. Natl. Acad. Sci. USA 2020, 117, 15977–15988. [Google Scholar] [CrossRef]
- Park, C.S.; Tang, S.-J. Regulation of MicroRNA Expression by Induction of Bidirectional Synaptic Plasticity. J. Mol. Neurosci. 2009, 38, 50–56. [Google Scholar] [CrossRef]
- Gurwitz, D. Genomics and the Future of Psychopharmacology: MicroRNAs Offer Novel Therapeutics. Dialogues Clin. Neurosci. 2019, 21, 131–138. [Google Scholar] [CrossRef]
- Babiloni, C.; Ferretti, A.; Del Gratta, C.; Carducci, F.; Vecchio, F.; Romani, G.L.; Rossini, P.M. Human Cortical Responses during One-Bit Delayed-Response Tasks: An FMRI Study. Brain Res. Bull. 2005, 65, 383–390. [Google Scholar] [CrossRef]
- Pirau, L.; Lui, F. Frontal Lobe Syndrome. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Fischer, M.; Moscovitch, M.; Alain, C. A Systematic Review and Meta-analysis of Memory-guided Attention: Frontal and Parietal Activation Suggests Involvement of Fronto-parietal Networks. WIREs Cogn. Sci. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Lane, R.D.; Reiman, E.M.; Bradley, M.M.; Lang, P.J.; Ahern, G.L.; Davidson, R.J.; Schwartz, G.E. Neuroanatomical Correlates of Pleasant and Unpleasant Emotion. Neuropsychologia 1997, 35, 1437–1444. [Google Scholar] [CrossRef]
- Ho, A.M.-C.; Winham, S.J.; Armasu, S.M.; Blacker, C.J.; Millischer, V.; Lavebratt, C.; Overholser, J.C.; Jurjus, G.J.; Dieter, L.; Mahajan, G.; et al. Genome-Wide DNA Methylomic Differences between Dorsolateral Prefrontal and Temporal Pole Cortices of Bipolar Disorder. J. Psychiatr. Res. 2019, 117, 45–54. [Google Scholar] [CrossRef]
- Herwig, U.; Padberg, F.; Unger, J.; Spitzer, M.; Schönfeldt-Lecuona, C. Transcranial Magnetic Stimulation in Therapy Studies: Examination of the Reliability of “Standard” Coil Positioning by Neuronavigation. Biol. Psychiatry 2001, 50, 58–61. [Google Scholar] [CrossRef]
- Smalheiser, N.R.; Lugli, G.; Rizavi, H.S.; Torvik, V.I.; Turecki, G.; Dwivedi, Y. MicroRNA Expression Is Down-Regulated and Reorganized in Prefrontal Cortex of Depressed Suicide Subjects. PLoS ONE 2012, 7, e33201. [Google Scholar] [CrossRef]
- Gorinski, N.; Bijata, M.; Prasad, S.; Wirth, A.; Abdel Galil, D.; Zeug, A.; Bazovkina, D.; Kondaurova, E.; Kulikova, E.; Ilchibaeva, T.; et al. Attenuated Palmitoylation of Serotonin Receptor 5-HT1A Affects Receptor Function and Contributes to Depression-like Behaviors. Nat. Commun. 2019, 10, 3924. [Google Scholar] [CrossRef] [Green Version]
- Wingo, T.S.; Yang, J.; Fan, W.; Min Canon, S.; Gerasimov, E.S.; Lori, A.; Logsdon, B.; Yao, B.; Seyfried, N.T.; Lah, J.J.; et al. Brain MicroRNAs Associated with Late-Life Depressive Symptoms Are Also Associated with Cognitive Trajectory and Dementia. Npj Genomic Med. 2020, 5, 6. [Google Scholar] [CrossRef] [Green Version]
- Babiloni, C.; Vecchio, F.; Bares, M.; Brazdil, M.; Nestrasil, I.; Eusebi, F.; Maria Rossini, P.; Rektor, I. Functional Coupling between Anterior Prefrontal Cortex (BA10) and Hand Muscle Contraction during Intentional and Imitative Motor Acts. NeuroImage 2008, 39, 1314–1323. [Google Scholar] [CrossRef]
- Semendeferi, K.; Armstrong, E.; Schleicher, A.; Zilles, K.; Van Hoesen, G.W. Prefrontal Cortex in Humans and Apes: A Comparative Study of Area 10. Am. J. Phys. Anthropol. 2001, 114, 224–241. [Google Scholar] [CrossRef]
- Katayama, N.; Nakagawa, A.; Umeda, S.; Terasawa, Y.; Kurata, C.; Tabuchi, H.; Kikuchi, T.; Mimura, M. Frontopolar Cortex Activation Associated with Pessimistic Future-Thinking in Adults with Major Depressive Disorder. NeuroImage Clin. 2019, 23, 101877. [Google Scholar] [CrossRef]
- Rogers, R.D.; Owen, A.M.; Middleton, H.C.; Williams, E.J.; Pickard, J.D.; Sahakian, B.J.; Robbins, T.W. Choosing between Small, Likely Rewards and Large, Unlikely Rewards Activates Inferior and Orbital Prefrontal Cortex. J. Neurosci. 1999, 19, 9029–9038. [Google Scholar] [CrossRef] [PubMed]
- Pizzagalli, D.A.; Iosifescu, D.; Hallett, L.A.; Ratner, K.G.; Fava, M. Reduced Hedonic Capacity in Major Depressive Disorder: Evidence from a Probabilistic Reward Task. J. Psychiatr. Res. 2008, 43, 76–87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maussion, G.; Yang, J.; Yerko, V.; Barker, P.; Mechawar, N.; Ernst, C.; Turecki, G. Regulation of a Truncated Form of Tropomyosin-Related Kinase B (TrkB) by Hsa-MiR-185* in Frontal Cortex of Suicide Completers. PLoS ONE 2012, 7, e39301. [Google Scholar] [CrossRef] [PubMed]
- Smalheiser, N.R.; Lugli, G.; Zhang, H.; Rizavi, H.; Cook, E.H.; Dwivedi, Y. Expression of MicroRNAs and Other Small RNAs in Prefrontal Cortex in Schizophrenia, Bipolar Disorder and Depressed Subjects. PLoS ONE 2014, 9, e86469. [Google Scholar] [CrossRef]
- Wang, Q.; Roy, B.; Turecki, G.; Shelton, R.C.; Dwivedi, Y. Role of Complex Epigenetic Switching in Tumor Necrosis Factor-α Upregulation in the Prefrontal Cortex of Suicide Subjects. Am. J. Psychiatry 2018, 175, 262–274. [Google Scholar] [CrossRef] [Green Version]
- Kim, T.; Valera, E.; Desplats, P. Alterations in Striatal MicroRNA-MRNA Networks Contribute to Neuroinflammation in Multiple System Atrophy. Mol. Neurobiol. 2019, 56, 7003–7021. [Google Scholar] [CrossRef]
- Stewart, C.; Riedel, K. Managing Speech and Language Deficits after Stroke. In Stroke Rehabilitation; Mosby Elsevier: Saint Louis, MO, USA, 2016; pp. 673–689. [Google Scholar] [CrossRef]
- Graïc, J.-M.; Peruffo, A.; Corain, L.; Centelleghe, C.; Granato, A.; Zanellato, E.; Cozzi, B. Asymmetry in the Cytoarchitecture of the Area 44 Homolog of the Brain of the Chimpanzee Pan Troglodytes. Front. Neuroanat. 2020, 14, 55. [Google Scholar] [CrossRef]
- Wildgruber, D.; Riecker, A.; Hertrich, I.; Erb, M.; Grodd, W.; Ethofer, T.; Ackermann, H. Identification of Emotional Intonation Evaluated by FMRI. NeuroImage 2005, 24, 1233–1241. [Google Scholar] [CrossRef]
- Lopez, J.P.; Fiori, L.M.; Gross, J.A.; Labonte, B.; Yerko, V.; Mechawar, N.; Turecki, G. Regulatory Role of MiRNAs in Polyamine Gene Expression in the Prefrontal Cortex of Depressed Suicide Completers. Int. J. Neuropsychopharmacol. 2014, 17, 23–32. [Google Scholar] [CrossRef] [Green Version]
- Haramati, S.; Navon, I.; Issler, O.; Ezra-Nevo, G.; Gil, S.; Zwang, R.; Hornstein, E.; Chen, A. MicroRNA as Repressors of Stress-Induced Anxiety: The Case of Amygdalar MiR-34. J. Neurosci. 2011, 31, 14191–14203. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Shen, Y.; Zhang, Y.; Peng, S.; Zhang, R.; Ning, A.; Li, H.; Li, X.; Lin, G.N.; Yu, S. Expression Alteration of MicroRNAs in Nucleus Accumbens Is Associated with Chronic Stress and Antidepressant Treatment in Rats. BMC Med. Inform. Decis. Mak. 2019, 19, 271. [Google Scholar] [CrossRef]
- McKibben, L.A.; Dwivedi, Y. Early-Life Stress Induces Genome-Wide Sex-Dependent MiRNA Expression and Correlation across Limbic Brain Areas in Rats. Epigenomics 2021, 13, 1031–1056. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.-X.; Xie, G.-J.; Mao, X.; Zou, X.-P.; Liao, Y.-J.; Liu, Q.-S.; Wang, H.; Cheng, Y. Exosomes from Patients with Major Depression Cause Depressive-like Behaviors in Mice with Involvement of MiR-139-5p-Regulated Neurogenesis. Neuropsychopharmacology 2020, 45, 1050–1058. [Google Scholar] [CrossRef] [PubMed]
- Lopez, J.P.; Lim, R.; Cruceanu, C.; Crapper, L.; Fasano, C.; Labonte, B.; Maussion, G.; Yang, J.P.; Yerko, V.; Vigneault, E.; et al. MiR-1202 Is a Primate-Specific and Brain-Enriched MicroRNA Involved in Major Depression and Antidepressant Treatment. Nat. Med. 2014, 20, 764–768. [Google Scholar] [CrossRef] [PubMed]
- Torres-Berrío, A.; Lopez, J.P.; Bagot, R.C.; Nouel, D.; Dal Bo, G.; Cuesta, S.; Zhu, L.; Manitt, C.; Eng, C.; Cooper, H.M.; et al. DCC Confers Susceptibility to Depression-like Behaviors in Humans and Mice and Is Regulated by MiR-218. Biol. Psychiatry 2017, 81, 306–315. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Zhao, G.; Yang, Z.; Liu, X.; Xie, P. Downregulation of MicroRNA-124-3p Suppresses the MTOR Signaling Pathway by Targeting DDIT4 in Males with Major Depressive Disorder. Int. J. Mol. Med. 2017, 41, 493–500. [Google Scholar] [CrossRef]
- Stevens, F.L.; Hurley, R.A.; Taber, K.H. Anterior Cingulate Cortex: Unique Role in Cognition and Emotion. J. Neuropsychiatry Clin. Neurosci. 2011, 23, 121–125. [Google Scholar] [CrossRef]
- Gasquoine, P.G. Localization of Function in Anterior Cingulate Cortex: From Psychosurgery to Functional Neuroimaging. Neurosci. Biobehav. Rev. 2013, 37, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Palomero-Gallagher, N.; Mohlberg, H.; Zilles, K.; Vogt, B. Cytology and Receptor Architecture of Human Anterior Cingulate Cortex. J. Comp. Neurol. 2008, 508, 906–926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davis, K.D. Human Anterior Cingulate Cortex Neurons Encode Cognitive and Emotional Demands. J. Neurosci. 2005, 25, 8402–8406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Casey, B.J.; Thomas, K.M.; Welsh, T.F.; Badgaiyan, R.D.; Eccard, C.H.; Jennings, J.R.; Crone, E.A. Dissociation of Response Conflict, Attentional Selection, and Expectancy with Functional Magnetic Resonance Imaging. Proc. Natl. Acad. Sci. USA 2000, 97, 8728–8733. [Google Scholar] [CrossRef] [Green Version]
- Tripp, A.; Oh, H.; Guilloux, J.-P.; Martinowich, K.; Lewis, D.A.; Sibille, E. Brain-Derived Neurotrophic Factor Signaling and Subgenual Anterior Cingulate Cortex Dysfunction in Major Depressive Disorder. Am. J. Psychiatry 2012, 169, 1194–1202. [Google Scholar] [CrossRef] [Green Version]
- Ho, T.C.; Sacchet, M.D.; Connolly, C.G.; Margulies, D.S.; Tymofiyeva, O.; Paulus, M.P.; Simmons, A.N.; Gotlib, I.H.; Yang, T.T. Inflexible Functional Connectivity of the Dorsal Anterior Cingulate Cortex in Adolescent Major Depressive Disorder. Neuropsychopharmacology 2017, 42, 2434–2445. [Google Scholar] [CrossRef] [Green Version]
- Kozel, F.A.; Rao, U.; Lu, H.; Nakonezny, P.A.; Grannemann, B.; McGregor, T.; Croarkin, P.E.; Mapes, K.S.; Tamminga, C.A.; Trivedi, M.H. Functional Connectivity of Brain Structures Correlates with Treatment Outcome in Major Depressive Disorder. Front. Psychiatry 2011, 2, 7. [Google Scholar] [CrossRef] [Green Version]
- Yoshino, Y.; Roy, B.; Dwivedi, Y. Altered MiRNA Landscape of the Anterior Cingulate Cortex Is Associated with Potential Loss of Key Neuronal Functions in Depressed Brain. Eur. Neuropsychopharmacol. 2020, 40, 70–84. [Google Scholar] [CrossRef]
- Fiori, L.M.; Kos, A.; Lin, R.; Théroux, J.-F.; Lopez, J.P.; Kühne, C.; Eggert, C.; Holzapfel, M.; Huettl, R.-E.; Mechawar, N.; et al. MiR-323a Regulates ERBB4 and Is Involved in Depression. Mol. Psychiatry 2020. [Google Scholar] [CrossRef]
- Zucchi, F.C.R.; Yao, Y.; Ward, I.D.; Ilnytskyy, Y.; Olson, D.M.; Benzies, K.; Kovalchuk, I.; Kovalchuk, O.; Metz, G.A.S. Maternal Stress Induces Epigenetic Signatures of Psychiatric and Neurological Diseases in the Offspring. PLoS ONE 2013, 8, e56967. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, J.A.; Carter, B.S.; Meng, F.; Turner, D.L.; Dai, M.; Schatzberg, A.F.; Barchas, J.D.; Jones, E.G.; Bunney, W.E.; Myers, R.M.; et al. The MicroRNA Network Is Altered in Anterior Cingulate Cortex of Patients with Unipolar and Bipolar Depression. J. Psychiatr. Res. 2016, 82, 58–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roy, B.; Wang, Q.; Palkovits, M.; Faludi, G.; Dwivedi, Y. Altered MiRNA Expression Network in Locus Coeruleus of Depressed Suicide Subjects. Sci. Rep. 2017, 7, 4387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maheu, M.; Lopez, J.P.; Crapper, L.; Davoli, M.A.; Turecki, G.; Mechawar, N. MicroRNA Regulation of Central Glial Cell Line-Derived Neurotrophic Factor (GDNF) Signalling in Depression. Transl. Psychiatry 2015, 5, e511. [Google Scholar] [CrossRef] [Green Version]
- Roy, B.; Dunbar, M.; Agrawal, J.; Allen, L.; Dwivedi, Y. Amygdala-Based Altered MiRNome and Epigenetic Contribution of MiR-128-3p in Conferring Susceptibility to Depression-Like Behavior via Wnt Signaling. Int. J. Neuropsychopharmacol. 2020, 23, 165–177. [Google Scholar] [CrossRef]
- Zheng, D.; Sabbagh, J.J.; Blair, L.J.; Darling, A.L.; Wen, X.; Dickey, C.A. MicroRNA-511 Binds to FKBP5 MRNA, Which Encodes a Chaperone Protein, and Regulates Neuronal Differentiation. J. Biol. Chem. 2016, 291, 17897–17906. [Google Scholar] [CrossRef] [Green Version]
- Xu, J.; Wang, R.; Liu, Y.; Wang, W.; Liu, D.; Jiang, H.; Pan, F. Short- and Long-Term Alterations of FKBP5-GR and Specific MicroRNAs in the Prefrontal Cortex and Hippocampus of Male Rats Induced by Adolescent Stress Contribute to Depression Susceptibility. Psychoneuroendocrinology 2019, 101, 204–215. [Google Scholar] [CrossRef]
- Maurel, O.M.; Torrisi, S.A.; Barbagallo, C.; Purrello, M.; Salomone, S.; Drago, F.; Ragusa, M.; Leggio, G.M. Dysregulation of MiR-15a-5p, MiR-497a-5p and MiR-511-5p Is Associated with Modulation of BDNF and FKBP5 in Brain Areas of PTSD-Related Susceptible and Resilient Mice. Int. J. Mol. Sci. 2021, 22, 5157. [Google Scholar] [CrossRef]
- Issler, O.; Haramati, S.; Paul, E.D.; Maeno, H.; Navon, I.; Zwang, R.; Gil, S.; Mayberg, H.S.; Dunlop, B.W.; Menke, A.; et al. MicroRNA 135 Is Essential for Chronic Stress Resiliency, Antidepressant Efficacy, and Intact Serotonergic Activity. Neuron 2014, 83, 344–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roy, B.; Dunbar, M.; Shelton, R.C.; Dwivedi, Y. Identification of MicroRNA-124-3p as a Putative Epigenetic Signature of Major Depressive Disorder. Neuropsychopharmacology 2017, 42, 864–875. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lopez, J.P.; Fiori, L.M.; Cruceanu, C.; Lin, R.; Labonte, B.; Cates, H.M.; Heller, E.A.; Vialou, V.; Ku, S.M.; Gerald, C.; et al. MicroRNAs 146a/b-5 and 425-3p and 24-3p Are Markers of Antidepressant Response and Regulate MAPK/Wnt-System Genes. Nat. Commun. 2017, 8, 15497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aschrafi, A.; Verheijen, J.M.; Gordebeke, P.M.; Loohuis, N.F.O.; Menting, K.; Jager, A.; Palkovits, M.; Geenen, B.; Kos, A.; Martens, G.J.M.; et al. MicroRNA-326 Acts as a Molecular Switch in the Regulation of Midbrain Urocortin 1 Expression. J. Psychiatry Neurosci. 2016, 41, 342–353. [Google Scholar] [CrossRef] [Green Version]
- Yuan, H.; Mischoulon, D.; Fava, M.; Otto, M.W. Circulating MicroRNAs as Biomarkers for Depression: Many Candidates, Few Finalists. J. Affect. Disord. 2018, 233, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Glinge, C.; Clauss, S.; Boddum, K.; Jabbari, R.; Jabbari, J.; Risgaard, B.; Tomsits, P.; Hildebrand, B.; Kääb, S.; Wakili, R.; et al. Stability of Circulating Blood-Based MicroRNAs—Pre-Analytic Methodological Considerations. PLoS ONE 2017, 12, e0167969. [Google Scholar] [CrossRef]
- Mompeón, A.; Ortega-Paz, L.; Vidal-Gómez, X.; Costa, T.J.; Pérez-Cremades, D.; Garcia-Blas, S.; Brugaletta, S.; Sanchis, J.; Sabate, M.; Novella, S.; et al. Disparate MiRNA Expression in Serum and Plasma of Patients with Acute Myocardial Infarction: A Systematic and Paired Comparative Analysis. Sci. Rep. 2020, 10, 5373. [Google Scholar] [CrossRef]
- Das Gupta, S.; Ciszek, R.; Heiskanen, M.; Lapinlampi, N.; Kukkonen, J.; Leinonen, V.; Puhakka, N.; Pitkänen, A. Plasma MiR-9-3p and MiR-136-3p as Potential Novel Diagnostic Biomarkers for Experimental and Human Mild Traumatic Brain Injury. Int. J. Mol. Sci. 2021, 22, 1563. [Google Scholar] [CrossRef]
- He, C.; Bai, Y.; Wang, Z.; Fan, D.; Wang, Q.; Liu, X.; Zhang, H.; Zhang, H.; Zhang, Z.; Yao, H.; et al. Identification of MicroRNA-9 Linking the Effects of Childhood Maltreatment on Depression Using Amygdala Connectivity. NeuroImage 2021, 224, 117428. [Google Scholar] [CrossRef]
- Saeedi, S.; Nagy, C.; Ibrahim, P.; Théroux, J.-F.; Wakid, M.; Fiori, L.M.; Yang, J.; Rotzinger, S.; Foster, J.A.; Mechawar, N.; et al. Neuron-Derived Extracellular Vesicles Enriched from Plasma Show Altered Size and MiRNA Cargo as a Function of Antidepressant Drug Response. Mol. Psychiatry 2021. [Google Scholar] [CrossRef]
- Bocchio-Chiavetto, L.; Maffioletti, E.; Bettinsoli, P.; Giovannini, C.; Bignotti, S.; Tardito, D.; Corrada, D.; Milanesi, L.; Gennarelli, M. Blood MicroRNA Changes in Depressed Patients during Antidepressant Treatment. Eur. Neuropsychopharmacol. 2013, 23, 602–611. [Google Scholar] [CrossRef]
- Li, J.; Meng, H.; Cao, W.; Qiu, T. MiR-335 Is Involved in Major Depression Disorder and Antidepressant Treatment through Targeting GRM4. Neurosci. Lett. 2015, 606, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Lopez, J.P.; Pereira, F.; Richard-Devantoy, S.; Berlim, M.; Chachamovich, E.; Fiori, L.M.; Niola, P.; Turecki, G.; Jollant, F. Co-Variation of Peripheral Levels of MiR-1202 and Brain Activity and Connectivity During Antidepressant Treatment. Neuropsychopharmacology 2017, 42, 2043–2051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fiori, L.M.; Lopez, J.P.; Richard-Devantoy, S.; Berlim, M.; Chachamovich, E.; Jollant, F.; Foster, J.; Rotzinger, S.; Kennedy, S.H.; Turecki, G. Investigation of MiR-1202, MiR-135a, and MiR-16 in Major Depressive Disorder and Antidepressant Response. Int. J. Neuropsychopharmacol. 2017, 20, 619–623. [Google Scholar] [CrossRef] [Green Version]
- Ding, Y.; Zhong, M.; Qiu, B.; Liu, C.; Wang, J.; Liang, J. Abnormal Expression of MiR-135a in Patients with Depression and Its Possible Involvement in the Pathogenesis of the Condition. Exp. Ther. Med. 2021, 22, 726. [Google Scholar] [CrossRef]
- Baudry, A.; Mouillet-Richard, S.; Schneider, B.; Launay, J.-M.; Kellermann, O. MiR-16 Targets the Serotonin Transporter: A New Facet for Adaptive Responses to Antidepressants. Science 2010, 329, 1537–1541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, C.-C.; Tsai, M.-C.; Lee, C.-T.; Sun, M.-H.; Huang, T.-L. Antidepressant Treatment Increased Serum MiR- 183 and MiR-212 Levels in Patients with Major Depressive Disorder. Psychiatry Res. 2018, 270, 232–237. [Google Scholar] [CrossRef]
- Belzeaux, R.; Fiori, L.M.; Lopez, J.P.; Boucekine, M.; Boyer, L.; Blier, P.; Farzan, F.; Frey, B.N.; Giacobbe, P.; Lam, R.W.; et al. Predicting Worsening Suicidal Ideation With Clinical Features and Peripheral Expression of Messenger RNA and MicroRNA During Antidepressant Treatment. J. Clin. Psychiatry 2019, 80. [Google Scholar] [CrossRef]
- Yrondi, A.; Fiori, L.M.; Frey, B.N.; Lam, R.W.; MacQueen, G.M.; Milev, R.; Müller, D.J.; Foster, J.A.; Kennedy, S.H.; Turecki, G. Association Between Side Effects and Blood MicroRNA Expression Levels and Their Targeted Pathways in Patients With Major Depressive Disorder Treated by a Selective Serotonin Reuptake Inhibitor, Escitalopram: A CAN-BIND-1 Report. Int. J. Neuropsychopharmacol. 2020, 23, 88–95. [Google Scholar] [CrossRef]
- Zhao, L.; Yang, X.; Cui, L.; Wei, J.; Ni, P.; Li, M.; Wang, Y.; He, Y.; Li, X.; Liang, S.; et al. Increased Expression of a Novel MiRNA in Peripheral Blood Is Negatively Correlated with Hippocampal Volume in Patients with Major Depressive Disorder. J. Affect. Disord. 2019, 245, 205–212. [Google Scholar] [CrossRef]
- Li, Y.-J.; Xu, M.; Gao, Z.-H.; Wang, Y.-Q.; Yue, Z.; Zhang, Y.-X.; Li, X.-X.; Zhang, C.; Xie, S.-Y.; Wang, P.-Y. Alterations of Serum Levels of BDNF-Related MiRNAs in Patients with Depression. PLoS ONE 2013, 8, e63648. [Google Scholar] [CrossRef] [Green Version]
- Fang, Y.; Qiu, Q.; Zhang, S.; Sun, L.; Li, G.; Xiao, S.; Li, X. Changes in MiRNA-132 and MiR-124 Levels in Non-Treated and Citalopram-Treated Patients with Depression. J. Affect. Disord. 2018, 227, 745–751. [Google Scholar] [CrossRef]
- Li, Y.; Li, S.; Yan, J.; Wang, D.; Yin, R.; Zhao, L.; Zhu, Y.; Zhu, X. MiR-182 (MicroRNA-182) Suppression in the Hippocampus Evokes Antidepressant-like Effects in Rats. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 65, 96–103. [Google Scholar] [CrossRef]
- Wang, X.; Sundquist, K.; Hedelius, A.; Palmér, K.; Memon, A.A.; Sundquist, J. Circulating MicroRNA-144-5p Is Associated with Depressive Disorders. Clin. Epigenetics 2015, 7, 69. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Wang, N.; Pan, J.; Wang, X.; Zhao, Y.; Guo, Z. Hippocampal MiRNA-144 Modulates Depressive-Like Behaviors in Rats by Targeting PTP1B. Neuropsychiatr. Dis. Treat. 2021, 17, 389–399. [Google Scholar] [CrossRef]
- Song, M.-F.; Dong, J.-Z.; Wang, Y.-W.; He, J.; Ju, X.; Zhang, L.; Zhang, Y.-H.; Shi, J.-F.; Lv, Y.-Y. CSF MiR-16 Is Decreased in Major Depression Patients and Its Neutralization in Rats Induces Depression-like Behaviors via a Serotonin Transmitter System. J. Affect. Disord. 2015, 178, 25–31. [Google Scholar] [CrossRef]
- Wan, Y.; Liu, Y.; Wang, X.; Wu, J.; Liu, K.; Zhou, J.; Liu, L.; Zhang, C. Identification of Differential MicroRNAs in Cerebrospinal Fluid and Serum of Patients with Major Depressive Disorder. PLoS ONE 2015, 10, e0121975. [Google Scholar] [CrossRef]
- Kuang, W.-H.; Dong, Z.-Q.; Tian, L.-T.; Li, J. MicroRNA-451a, MicroRNA-34a-5p, and MicroRNA-221-3p as Predictors of Response to Antidepressant Treatment. Braz. J. Med. Biol. Res. 2018, 51, e7212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zurawek, D.; Kusmider, M.; Faron-Gorecka, A.; Gruca, P.; Pabian, P.; Kolasa, M.; Solich, J.; Szafran-Pilch, K.; Papp, M.; Dziedzicka-Wasylewska, M. Time-Dependent MiR-16 Serum Fluctuations Together with Reciprocal Changes in the Expression Level of MiR-16 in Mesocortical Circuit Contribute to Stress Resilient Phenotype in Chronic Mild Stress—An Animal Model of Depression. Eur. Neuropsychopharmacol. 2016, 26, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Zurawek, D.; Kusmider, M.; Faron-Gorecka, A.; Gruca, P.; Pabian, P.; Solich, J.; Kolasa, M.; Papp, M.; Dziedzicka-Wasylewska, M. Reciprocal MicroRNA Expression in Mesocortical Circuit and Its Interplay with Serotonin Transporter Define Resilient Rats in the Chronic Mild Stress. Mol. Neurobiol. 2017, 54, 5741–5751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gheysarzadeh, A.; Sadeghifard, N.; Afraidooni, L.; Pooyan, F.; Mofid, M.; Valadbeigi, H.; Bakhtiari, H.; Keikhavani, S. Serum-Based MicroRNA Biomarkers for Major Depression: MiR-16, MiR-135a, and MiR-1202. J. Res. Med. Sci. 2018, 23, 69. [Google Scholar] [CrossRef]
- Mendes-Silva, A.P.; Fujimura, P.T.; Silva, J.R.d.C.; Teixeira, A.L.; Vieira, E.M.; Guedes, P.H.G.; Barroso, L.S.S.; Nicolau, M.d.S.; Ferreira, J.D.R.; Bertola, L.; et al. Brain-Enriched MicroRNA-184 Is Downregulated in Older Adults with Major Depressive Disorder: A Translational Study. J. Psychiatr. Res. 2019, 111, 110–120. [Google Scholar] [CrossRef]
- Van der Auwera, S.; Ameling, S.; Wittfeld, K.; d’Harcourt Rowold, E.; Nauck, M.; Völzke, H.; Suhre, K.; Najafi-Shoushtari, H.; Methew, J.; Ramachandran, V.; et al. Association of Childhood Traumatization and Neuropsychiatric Outcomes with Altered Plasma Micro RNA-Levels. Neuropsychopharmacology 2019, 44, 2030–2037. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, X.; Chen, J.; Cheng, K.; Bai, S.-J.; Zheng, P.; Zhou, C.; Wang, W.; Wang, H.; Zhong, L.; et al. Circulating MicroRNA 134 Sheds Light on the Diagnosis of Major Depressive Disorder. Transl. Psychiatry 2020, 10, 95. [Google Scholar] [CrossRef] [PubMed]
- Felger, J.C.; Lotrich, F.E. Inflammatory Cytokines in Depression: Neurobiological Mechanisms and Therapeutic Implications. Neuroscience 2013, 246, 199–229. [Google Scholar] [CrossRef] [Green Version]
- Miller, A.H.; Maletic, V.; Raison, C.L. Inflammation and Its Discontents: The Role of Cytokines in the Pathophysiology of Major Depression. Biol. Psychiatry 2009, 65, 732–741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dowlati, Y.; Herrmann, N.; Swardfager, W.; Liu, H.; Sham, L.; Reim, E.K.; Lanctôt, K.L. A Meta-Analysis of Cytokines in Major Depression. Biol. Psychiatry 2010, 67, 446–457. [Google Scholar] [CrossRef] [PubMed]
- Cyranowski, J.M.; Marsland, A.L.; Bromberger, J.T.; Whiteside, T.L.; Chang, Y.; Matthews, K.A. Depressive Symptoms and Production of Proinflammatory Cytokines by Peripheral Blood Mononuclear Cells Stimulated in Vitro. Brain. Behav. Immun. 2007, 21, 229–237. [Google Scholar] [CrossRef]
- Bierhaus, A.; Wolf, J.; Andrassy, M.; Rohleder, N.; Humpert, P.M.; Petrov, D.; Ferstl, R.; von Eynatten, M.; Wendt, T.; Rudofsky, G.; et al. A Mechanism Converting Psychosocial Stress into Mononuclear Cell Activation. Proc. Natl. Acad. Sci. USA 2003, 100, 1920–1925. [Google Scholar] [CrossRef] [Green Version]
- Pace, T.W.W.; Mletzko, T.C.; Alagbe, O.; Musselman, D.L.; Nemeroff, C.B.; Miller, A.H.; Heim, C.M. Increased Stress-Induced Inflammatory Responses in Male Patients With Major Depression and Increased Early Life Stress. Am. J. Psychiatry 2006, 163, 1630–1633. [Google Scholar] [CrossRef]
- Belzeaux, R.; Bergon, A.; Jeanjean, V.; Loriod, B.; Formisano-Tréziny, C.; Verrier, L.; Loundou, A.; Baumstarck-Barrau, K.; Boyer, L.; Gall, V.; et al. Responder and Nonresponder Patients Exhibit Different Peripheral Transcriptional Signatures during Major Depressive Episode. Transl. Psychiatry 2012, 2, e185. [Google Scholar] [CrossRef]
- Fan, H.; Sun, X.; Guo, W.; Zhong, A.; Niu, W.; Zhao, L.; Dai, Y.; Guo, Z.; Zhang, L.; Lu, J. Differential Expression of MicroRNA in Peripheral Blood Mononuclear Cells as Specific Biomarker for Major Depressive Disorder Patients. J. Psychiatr. Res. 2014, 59, 45–52. [Google Scholar] [CrossRef]
- Hung, Y.-Y.; Wu, M.-K.; Tsai, M.-C.; Huang, Y.-L.; Kang, H.-Y. Aberrant Expression of Intracellular Let-7e, MiR-146a, and MiR-155 Correlates with Severity of Depression in Patients with Major Depressive Disorder and Is Ameliorated after Antidepressant Treatment. Cells 2019, 8, 647. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Solich, J.; Kuśmider, M.; Faron-Górecka, A.; Pabian, P.; Kolasa, M.; Zemła, B.; Dziedzicka-Wasylewska, M. Serum Level of MiR-1 and MiR-155 as Potential Biomarkers of Stress-Resilience of NET-KO and SWR/J Mice. Cells 2020, 9, 917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaisvaser, S.; Modai, S.; Farberov, L.; Lin, T.; Sharon, H.; Gilam, A.; Volk, N.; Admon, R.; Edry, L.; Fruchter, E.; et al. Neuro-Epigenetic Indications of Acute Stress Response in Humans: The Case of MicroRNA-29c. PLoS ONE 2016, 11, e0146236. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Liu, X.; Jiang, K.; Peng, D.; Hong, W.; Fang, Y.; Qian, Y.; Yu, S.; Li, H. Alterations of MicroRNA-124 Expression in Peripheral Blood Mononuclear Cells in Pre- and Post-Treatment Patients with Major Depressive Disorder. J. Psychiatr. Res. 2016, 78, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Lei, L.; Wang, Y.; Yang, C.; Liu, Z.; Li, X.; Zhang, K. Preliminary Comparison of Plasma Notch-Associated MicroRNA-34b and -34c Levels in Drug Naive, First Episode Depressed Patients and Healthy Controls. J. Affect. Disord. 2016, 194, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Saeedi, S.; Israel, S.; Nagy, C.; Turecki, G. The Emerging Role of Exosomes in Mental Disorders. Transl. Psychiatry 2019, 9, 122. [Google Scholar] [CrossRef] [PubMed]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal Information for Studies of Extracellular Vesicles 2018 (MISEV2018): A Position Statement of the International Society for Extracellular Vesicles and Update of the MISEV2014 Guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef] [Green Version]
- Scioli, M.G.; Terriaca, S.; Fiorelli, E.; Storti, G.; Fabbri, G.; Cervelli, V.; Orlandi, A. Extracellular Vesicles and Cancer Stem Cells in Tumor Progression: New Therapeutic Perspectives. Int. J. Mol. Sci. 2021, 22, 10572. [Google Scholar] [CrossRef]
- Malkin, E.Z.; Bratman, S.V. Bioactive DNA from Extracellular Vesicles and Particles. Cell Death Dis. 2020, 11, 584. [Google Scholar] [CrossRef]
- Hussain, M.T.; Iqbal, A.J.; Norling, L.V. The Role and Impact of Extracellular Vesicles in the Modulation and Delivery of Cytokines during Autoimmunity. Int. J. Mol. Sci. 2020, 21, 7096. [Google Scholar] [CrossRef] [PubMed]
- Buzas, E.I.; György, B.; Nagy, G.; Falus, A.; Gay, S. Emerging Role of Extracellular Vesicles in Inflammatory Diseases. Nat. Rev. Rheumatol. 2014, 10, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, A.; Kim, H.S.; Bojmar, L.; Gyan, K.E.; Cioffi, M.; Hernandez, J.; Zambirinis, C.P.; Rodrigues, G.; Molina, H.; Heissel, S.; et al. Extracellular Vesicle and Particle Biomarkers Define Multiple Human Cancers. Cell 2020, 182, 1044–1061. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Rai, A.; Chen, M.; Suwakulsiri, W.; Greening, D.W.; Simpson, R.J. Extracellular Vesicles in Cancer—Implications for Future Improvements in Cancer Care. Nat. Rev. Clin. Oncol. 2018, 15, 617–638. [Google Scholar] [CrossRef] [PubMed]
- Goetzl, E.J.; Mustapic, M.; Kapogiannis, D.; Eitan, E.; Lobach, I.V.; Goetzl, L.; Schwartz, J.B.; Miller, B.L. Cargo Proteins of Plasma Astrocyte-derived Exosomes in Alzheimer’s Disease. FASEB J. 2016, 30, 3853–3859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, M.; Liu, C.; Cook, T.J.; Bullock, K.M.; Zhao, Y.; Ginghina, C.; Li, Y.; Aro, P.; Dator, R.; He, C.; et al. Plasma Exosomal α-Synuclein Is Likely CNS-Derived and Increased in Parkinson’s Disease. Acta Neuropathol. 2014, 128, 639–650. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhao, Y.; Tian, C.; Wang, J.; Li, W.; Zhong, C. Differential Exosomal MicroRNA Profile in the Serum of a Patient with Depression. Eur. J. Psychiatry 2018, 32, 105–112. [Google Scholar] [CrossRef]
- Liang, J.-Q.; Liao, H.-R.; Xu, C.-X.; Li, X.-L.; Wei, Z.-X.; Xie, G.-J.; Cheng, Y. Serum Exosome-Derived MiR-139-5p as a Potential Biomarker for Major Depressive Disorder. Neuropsychiatr. Dis. Treat. 2020, 16, 2689–2693. [Google Scholar] [CrossRef]
- Mizohata, Y.; Toda, H.; Koga, M.; Saito, T.; Fujita, M.; Kobayashi, T.; Hatakeyama, S.; Morimoto, Y. Neural Extracellular Vesicle-Derived MiR-17 in Blood as a Potential Biomarker of Subthreshold Depression. Hum. Cell 2021, 34, 1087–1092. [Google Scholar] [CrossRef]
- Makeyev, E.V.; Zhang, J.; Carrasco, M.A.; Maniatis, T. The MicroRNA MiR-124 Promotes Neuronal Differentiation by Triggering Brain-Specific Alternative Pre-MRNA Splicing. Mol. Cell 2007, 27, 435–448. [Google Scholar] [CrossRef] [Green Version]
- Yoo, A.S.; Sun, A.X.; Li, L.; Shcheglovitov, A.; Portmann, T.; Li, Y.; Lee-Messer, C.; Dolmetsch, R.E.; Tsien, R.W.; Crabtree, G.R. MicroRNA-Mediated Conversion of Human Fibroblasts to Neurons. Nature 2011, 476, 228–231. [Google Scholar] [CrossRef]
- Nissan, X.; Blondel, S.; Navarro, C.; Maury, Y.; Denis, C.; Girard, M.; Martinat, C.; De Sandre-Giovannoli, A.; Levy, N.; Peschanski, M. Unique Preservation of Neural Cells in Hutchinson- Gilford Progeria Syndrome Is Due to the Expression of the Neural-Specific MiR-9 MicroRNA. Cell Rep. 2012, 2, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adlakha, Y.K.; Saini, N. Brain MicroRNAs and Insights into Biological Functions and Therapeutic Potential of Brain Enriched MiRNA-128. Mol. Cancer 2014, 13, 33. [Google Scholar] [CrossRef] [Green Version]
- Enatescu, V.R.; Papava, I.; Enatescu, I.; Antonescu, M.; Anghel, A.; Seclaman, E.; Sirbu, I.O.; Marian, C. Circulating Plasma Micro RNAs in Patients with Major Depressive Disorder Treated with Antidepressants: A Pilot Study. Psychiatry Investig. 2016, 13, 549. [Google Scholar] [CrossRef] [Green Version]
- Su, M.; Hong, J.; Zhao, Y.; Liu, S.; Xue, X. MeCP2 Controls Hippocampal Brain-Derived Neurotrophic Factor Expression via Homeostatic Interactions with MicroRNA-132 in Rats with Depression. Mol. Med. Rep. 2015, 12, 5399–5406. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Shi, J.; Liu, H.; Wang, Q.; Chen, X.; Tang, H.; Yan, R.; Yao, Z.; Lu, Q. Plasma MicroRNA Array Analysis Identifies Overexpressed MiR-19b-3p as a Biomarker of Bipolar Depression Distinguishing From Unipolar Depression. Front. Psychiatry 2020, 11, 757. [Google Scholar] [CrossRef]
- Marshe, V.S.; Islam, F.; Maciukiewicz, M.; Fiori, L.M.; Yerko, V.; Yang, J.; Turecki, G.; Foster, J.A.; Kennedy, S.H.; Blumberger, D.M.; et al. Validation Study of MicroRNAs Previously Associated with Antidepressant Response in Older Adults Treated for Late-Life Depression with Venlafaxine. Prog. Neuropsychopharmacol. Biol. Psychiatry 2020, 100, 109867. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Yang, C.; He, X.; Liu, Z.; Liu, S.; Li, X.; Wang, Y.; Jin, R.; Zhang, K. Impact of Expression and Genetic Variation of MicroRNA-34b/c on Cognitive Dysfunction in Patients with Major Depressive Disorder. Neuropsychiatr. Dis. Treat. 2020, 16, 1543–1554. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Wang, M.; Li, M.; Yang, J.; Jia, J.; Liu, L.; Zhou, J.; Zhang, C.; Wang, X. Serum MiR-221-3p as a New Potential Biomarker for Depressed Mood in Perioperative Patients. Brain Res. 2019, 1720, 146296. [Google Scholar] [CrossRef]
- Kim, H.K.; Tyryshkin, K.; Elmi, N.; Dharsee, M.; Evans, K.R.; Good, J.; Javadi, M.; McCormack, S.; Vaccarino, A.L.; Zhang, X.; et al. Plasma MicroRNA Expression Levels and Their Targeted Pathways in Patients with Major Depressive Disorder Who Are Responsive to Duloxetine Treatment. J. Psychiatr. Res. 2019, 110, 38–44. [Google Scholar] [CrossRef]
- Qi, S.; Yang, X.; Zhao, L.; Calhoun, V.D.; Perrone-Bizzozero, N.; Liu, S.; Jiang, R.; Jiang, T.; Sui, J.; Ma, X. MicroRNA132 Associated Multimodal Neuroimaging Patterns in Unmedicated Major Depressive Disorder. Brain 2018, 141, 916–926. [Google Scholar] [CrossRef]
- Wang, X.; Wang, B.; Zhao, J.; Liu, C.; Qu, X.; Li, Y. MiR-155 Is Involved in Major Depression Disorder and Antidepressant Treatment via Targeting SIRT1. Biosci. Rep. 2018, 38, BSR20181139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Volk, N.; Pape, J.C.; Engel, M.; Zannas, A.S.; Cattane, N.; Cattaneo, A.; Binder, E.B.; Chen, A. Amygdalar MicroRNA-15a Is Essential for Coping with Chronic Stress. Cell Rep. 2016, 17, 1882–1891. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maffioletti, E.; Cattaneo, A.; Rosso, G.; Maina, G.; Maj, C.; Gennarelli, M.; Tardito, D.; Bocchio-Chiavetto, L. Peripheral Whole Blood MicroRNA Alterations in Major Depression and Bipolar Disorder. J. Affect. Disord. 2016, 200, 250–258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gururajan, A.; Naughton, M.E.; Scott, K.A.; O’Connor, R.M.; Moloney, G.; Clarke, G.; Dowling, J.; Walsh, A.; Ismail, F.; Shorten, G.; et al. MicroRNAs as Biomarkers for Major Depression: A Role for Let-7b and Let-7c. Transl. Psychiatry 2016, 6, e862. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Yang, X.; Zhao, L.; Zhang, J.; Li, T.; Ma, X. Increased MiR-132 Level Is Associated with Visual Memory Dysfunction in Patients with Depression. Neuropsychiatr. Dis. Treat. 2016, 12, 2905–2911. [Google Scholar] [CrossRef] [Green Version]
- Camkurt, M.A.; Acar, Ş.; Coşkun, S.; Güneş, M.; Güneş, S.; Yılmaz, M.F.; Görür, A.; Tamer, L. Comparison of Plasma MicroRNA Levels in Drug Naive, First Episode Depressed Patients and Healthy Controls. J. Psychiatr. Res. 2015, 69, 67–71. [Google Scholar] [CrossRef]
- Cheng, L.-C.; Pastrana, E.; Tavazoie, M.; Doetsch, F. MiR-124 Regulates Adult Neurogenesis in the Subventricular Zone Stem Cell Niche. Nat. Neurosci. 2009, 12, 399–408. [Google Scholar] [CrossRef] [Green Version]
- Fischbach, S.J.; Carew, T.J. MicroRNAs in Memory Processing. Neuron 2009, 63, 714–716. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.-S.; Mu, R.-H.; Li, C.-F.; Dong, S.-Q.; Geng, D.; Liu, Q.; Yi, L.-T. MicroRNA-124 Targets Glucocorticoid Receptor and Is Involved in Depression-like Behaviors. Prog. Neuropsychopharmacol. Biol. Psychiatry 2017, 79, 417–425. [Google Scholar] [CrossRef]
- Yang, W.; Liu, M.; Zhang, Q.; Zhang, J.; Chen, J.; Chen, Q.; Suo, L. Knockdown of MiR-124 Reduces Depression-like Behavior by Targeting CREB1 and BDNF. Curr. Neurovasc. Res. 2020, 17, 196–203. [Google Scholar] [CrossRef]
- Kozuka, T.; Omori, Y.; Watanabe, S.; Tarusawa, E.; Yamamoto, H.; Chaya, T.; Furuhashi, M.; Morita, M.; Sato, T.; Hirose, S.; et al. MiR-124 Dosage Regulates Prefrontal Cortex Function by Dopaminergic Modulation. Sci. Rep. 2019, 9, 3445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mannironi, C.; Camon, J.; De Vito, F.; Biundo, A.; De Stefano, M.E.; Persiconi, I.; Bozzoni, I.; Fragapane, P.; Mele, A.; Presutti, C. Acute Stress Alters Amygdala MicroRNA MiR-135a and MiR-124 Expression: Inferences for Corticosteroid Dependent Stress Response. PLoS ONE 2013, 8, e73385. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Tang, H.; Gao, L.; Tu, G.-L.; Luo, H.; Xia, Y. LncRNA H19 Aggravates Cerebral Ischemia/Reperfusion Injury by Functioning as a CeRNA for MiR-19a-3p to Target PTEN. Neuroscience 2020, 437, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.-L.; Wang, J.-L.; Liu, X.; Zhang, J.; Liu, C.; Guo, L. Inhibition of MiR-19a Protects Neurons against Ischemic Stroke through Modulating Glucose Metabolism and Neuronal Apoptosis. Cell. Mol. Biol. Lett. 2019, 24, 37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mannironi, C.; Biundo, A.; Rajendran, S.; De Vito, F.; Saba, L.; Caioli, S.; Zona, C.; Ciotti, T.; Caristi, S.; Perlas, E. MiR-135a Regulates Synaptic Transmission and Anxiety-Like Behavior in Amygdala. Mol. Neurobiol. 2018, 55, 3301–3315. [Google Scholar] [CrossRef]
- Jauhari, A.; Singh, T.; Singh, P.; Parmar, D.; Yadav, S. Regulation of MiR-34 Family in Neuronal Development. Mol. Neurobiol. 2018, 55, 936–945. [Google Scholar] [CrossRef]
- Aranha, M.M.; Santos, D.M.; Solá, S.; Steer, C.J.; Rodrigues, C.M.P. MiR-34a Regulates Mouse Neural Stem Cell Differentiation. PLoS ONE 2011, 6, e21396. [Google Scholar] [CrossRef]
- De Antonellis, P.; Medaglia, C.; Cusanelli, E.; Andolfo, I.; Liguori, L.; De Vita, G.; Carotenuto, M.; Bello, A.; Formiggini, F.; Galeone, A.; et al. MiR-34a Targeting of Notch Ligand Delta-Like 1 Impairs CD15+/CD133+ Tumor-Propagating Cells and Supports Neural Differentiation in Medulloblastoma. PLoS ONE 2011, 6, e24584. [Google Scholar] [CrossRef] [Green Version]
- Lo Iacono, L.; Ielpo, D.; Parisi, C.; Napoli, G.; Accoto, A.; Di Segni, M.; Babicola, L.; D’Addario, S.L.; Guzzo, S.M.; Pascucci, T.; et al. MicroRNA-34a Regulates 5-HT2C Expression in Dorsal Raphe and Contributes to the Anti-Depressant-like Effect of Fluoxetine. Neuropharmacology 2021, 190, 108559. [Google Scholar] [CrossRef] [PubMed]
- Andolina, D.; Di Segni, M.; Bisicchia, E.; D’Alessandro, F.; Cestari, V.; Ventura, A.; Concepcion, C.; Puglisi-Allegra, S.; Ventura, R. Effects of Lack of MicroRNA-34 on the Neural Circuitry Underlying the Stress Response and Anxiety. Neuropharmacology 2016, 107, 305–316. [Google Scholar] [CrossRef] [Green Version]
- Koshiol, J.; Wang, E.; Zhao, Y.; Marincola, F.; Landi, M.T. Strengths and Limitations of Laboratory Procedures for MicroRNA Detection: Table 1. Cancer Epidemiol. Biomarkers Prev. 2010, 19, 907–911. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.-M.; Nguyen, D.T.; Lu, L.-F. Progress and Challenge of MicroRNA Research in Immunity. Front. Genet. 2014, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Ref. | Brain Region | miRNA | Regulation MDD vs. HC | Targeted Gene | Validation of Targeted Gene |
|---|---|---|---|---|---|
| [80] | Amygdala | miR-128-3p | Up | DVL1, LEF1, WNT5b | Direct—in vitro |
| [75] | Lateral habenula | miR-320b-3p, miR-331-3p | Up | N/A | N/A |
| miR-323a-3p | Up | ERBB4 | Direct—in vitro | ||
| BA24 | miR-204-5p, miR-331-3p | Up | N/A | N/A | |
| miR-323a-3p | Up | ERBB4 | Direct—in vitro | ||
| [45] | BA9/BA46 | miR-484-5p, miR-26b-5p, miR-30d-5p, miR-197-3p | Down | N/A | N/A |
| [74] | ACC | 117 miRNAs (4.16%) | Up | N/A | N/A |
| 54 miRNAs (2.13%) | Down | N/A | N/A | ||
| [44] | BA9 | miR-30a-5p, miR-30e-5p | Up | ZDHHC21 | Direct—in vitro |
| miR-200a-5p | Down | N/A | N/A | ||
| [65] | BA44 | miR-124-3p | Down * | DDIT | Indirect |
| [53] | BA10 | miR-19a-3p | Up | TNFa | N/A |
| miR-20a-5p, miR-92a-1-3p | Down | N/A | N/A | ||
| [85] | BA46 | miR-124-3p | Up | GRIA3, GRIA4, NR3C1 | Direct—in vitro |
| [78] | Locus coeruleus | miR-17-5p, miR-20b-5p, miR-106a-5p, miR-330-3p, miR-541-3p, miR-582-5p, miR-890, miR-99-3p, miR-550-5p, miR-1179 | Up | RELN, GSK-3β, MAOA, CHRM1, PLCB1,GRIK1 | Indirect |
| miR-409-5p, let-7g-3p, miR-1197 | Down | ||||
| [64] | BA44 | miR-218-5p | Down | DCC | Direct—in vitro |
| [86] | vPFC | miR-146a-5p, miR-146b-5p, miR-24-3p, miR-425-3p | Up | MAPK/Wnt pathway | Direct—in vitro |
| [87] | Midbrain | miR-326 | Down | UCN1 | Direct—in vitro |
| [77] | ACC | miR-184 and miR-34a-5p | Down | PDE4B, NCOA1, NCOR2 | Direct—in vitro |
| [79] | BLA | miR-511, miR-340 | Up | GFRA1 | Direct—in vitro |
| [63] | BA44 | miR-1202 | Down | GRM4 | Direct—in vitro |
| [84] | Raphe nuclei | miR135a-5p | Down | SLC6A4, HTR1A | Direct—in vitro |
| [58] | BA44 | miR-34c-5p, miR-139-5p, miR-195-5p, miR-320c-3p | Up | STAT1, SMOX | Indirect |
| [52] | BA10 | miR-508-3p, miR-152-3p | Down | N/A | N/A |
| [43] | BA9 | miR-142-5p, miR-137, miR-489, miR-148b, miR-101, miR-324-5p, miR-301a, miR-146a, miR-335, miR-494, miR-20b, miR-376a *, miR-190, miR-155, miR-660, miR-130a, miR-27a, miR-497, miR-10a, miR-20a, miR-142-3p | Down | DNMT3b, VEGFa, BCL-2 | Indirect |
| [51] | BA10 | miR-185 *, 491-3p | Up | Trk2- T1 | Direct—in vitro |
| Ref. | Tissue Source | miRNA | Regulation MDD vs. HC | Antidepressant Treatment | miRNA | Regulation by Treatment | Targeted Gene | Validation of Targeted Gene |
|---|---|---|---|---|---|---|---|---|
| [92] | Whole blood | miR-9-5p | Up | - | - | - | - | - |
| [143] | BDEVs in blood | miR-17-5p | & | - | - | - | - | - |
| [93] | BDEVs in blood | - | - | Escitalopram (8 weeks) | miR-30d-5p, miR-486-5p | Up # | NR3C1, SIRT1, SERPINE1, RPS6KB1, ATF6, PSEN1 | Indirect |
| [150] | Plasma | miR-19b-3p, miR-3921 | Down | - | - | - | - | - |
| miR-1180-3p | Up | - | - | - | - | - | ||
| [142] | Serum exosomes | miR-139-5p | Up | - | - | - | - | - |
| [62] | Blood exosomes | miR-139-5p | Up | - | - | - | MAP2 | Direct—in vitro |
| [151] | Buffy coat | - | - | Venlafaxine (12 weeks) | miR-135a-5p | X | - | - |
| [152] | Whole blood | miR-34b-5p, miR-34c-5p | C * | - | - | - | - | - |
| [117] | Plasma | miR-134 | Down | Personalized (8 weeks) | miR-134 | Up | - | - |
| [102] | Whole blood | - | - | Escitalopram (2 weeks) | 33 miRNAs | Up | - | - |
| 12 miRNAs | Down | - | - | |||||
| [153] | Serum | miR-221-3p | Up | - | - | - | IRF2 | Direct—in vitro |
| [103] | Whole blood | pmiR-chr11 | Up | - | - | - | BRPF1 | Direct—in vitro |
| miR-1275 | Down | - | - | - | - | - | ||
| [115] | Plasma | miR-184 | Down | - | - | - | - | - |
| [116] | Plasma | let-7g-5p, miR-103a-3p, miR-107, and miR-142-3p | Down | - | - | - | - | - |
| [126] | PBMC/Monocytes | let-7e-5p, miR-21-5p, miR-146a, miR-155, miR-146a, miR-155 | Down | Personalized (4 weeks) | let-7e-5p, miR-223, miR-146a, miR-155, miR-21-5p, miR-14 | Up | TLR4 | Indirect |
| [154] | Plasma | - | - | Duloxetine (6–8 weeks) | miR-23a-3p, miR-16-5p, miR-146a-5p, miR-21-5p | 10th percentile | - | - |
| [101] | Whole blood | - | - | Duloxetine (8 weeks) | miR-3688, miR-5695 | Up # | - | - |
| [114] | Serum | miR-16-5p, miR-135a-5p, miR-1202 | Down | - | - | - | - | - |
| [155] | Whole blood | miR-132-3p | Up | - | - | - | - | - |
| [156] | Whole blood | miR-132-3p, miR-135b, miR-181b | Up | - | - | - | - | - |
| miR-155 | Up | - | - | - | SIRT1 | Direct—in vitro | ||
| [53] | PBMC | miR-19a-3p | Up | - | - | - | TNFa | Indirect |
| [111] | Serum | miRNA-34a-5p, miRNA-221-3p | Up | Paroxetine (8 weeks) | miRNA-34a-5p, miRNA-221-3p | Down | - | - |
| miRNA-451a | Down | miRNA-451a | Up | - | - | |||
| [105] | Plasma | miR-132-3p, miR-124-3p | Up | Citalopram (8 weeks) | miR-124-3p | Up | - | - |
| miR-132 | Down | - | - | |||||
| [100] | Serum | - | - | SSRI/SNRI (4 weeks) | miR-183, miR-212 | Up | - | - |
| SSRI (4 weeks) | miR-16-5p | Up | - | - | ||||
| [97] | Whole blood | miR-1202 | Down # | Duloxetine (8 weeks) | miR-1202 | Up # | - | - |
| [85] | Serum | miR-124-3p | Up | - | - | - | - | - |
| [86] | Whole blood/ Plasma | - | - | Personalized (8 weeks) | miR-146a-5p, miR-146b-5p, miR-24-3p, miR-425-3p, miR-3074-5p | Down # | MAPK/Wnt | Direct—in vitro |
| [141] | Serum exosomes | miR-1255a, miR-3161, miR-99a-3p, miR-205-5p, miR-26a-1-3p, miR-139-5p, miR-7849-3p, miR-195-5p, miR-125b-2-3p, miR-664a-3p, let-7c-5p, miR-197-3p | Up | - | - | - | - | - |
| miR-499a-5p, miR-4732-3p, miR-222-5p, miR-1291, miR-668-3p, miR-425-3p, miR-6511a-3p, iR-145-3p, miR-200a-3p, miR-143-3p, miR-196b-5p, miR-99a-5p, miR-144-3p, miR-584-5p, miR-210-3p, miR-183-5p, miR-107, miR-130b-5p, miR-589-5p, miR-1910-5p | Down | - | - | - | - | - | ||
| [96] | Whole blood | - | - | Desvenlafaxine (8 weeks) | miR-1202 | Up | - | - |
| [157] | Whole blood | miR-15a-5p | Up | - | - | - | FKBP5 | Direct—in vitro |
| [130] | PBMC | miR-34b-5p, miR-34c-5p | Up | - | - | - | Notch1 | Indirect |
| [158] | Whole blood | miR-199a-5p, miR-345-5p, miR-330-3p, miR-425-3p, miR-24-3p, miR-29c-5p | Up | - | - | - | - | - |
| let-7a-5p, let-7f-5p, let-7d-5p, miR-1915-3p | Down | |||||||
| [129] | PBMC | miR-124-3p | Up | Personalized (8 weeks) | miR-124-3p | Down | - | - |
| [159] | Whole blood | let-7b, let-7c | Down | - | - | - | - | - |
| [128] | PBMC | miR-29c | Up * | - | - | - | - | - |
| [160] | Whole blood | miR-132-3p | Up | - | - | - | - | - |
| [148] | Plasma | - | - | Escitalopram (12 weeks) | 23 miRNAs | Up | - | - |
| 17 miRNAs | Down | - | - | |||||
| [161] | Plasma | miR-451a, miR-17-5p, miR-223-3p | Up | - | - | - | - | - |
| miR-320a | Down | |||||||
| [109] | CSF | miR-16-5p | Down | - | - | - | SLC6A4 | Direct—in vitro |
| Serum | miR-16-5p | Down | - | - | - | |||
| [110] | CSF | miR-34a-5p, miR-221-3p, let-7d-3p | Up | - | - | - | - | - |
| miR-451a | Down | |||||||
| Serum | miR-125a-5p, miR-30a-5p, let-7d-3p, miR-34a-5p, miR-221-3p, miR-29b-3p, miR-10a-5p, miR-375 | Up | ||||||
| miR-155-5p, miR-33a-5p, miR-139-5p, miR-590-5, miR-185-5p, miR-106b-5p, miR-15b-5p, miR-451a | Down | |||||||
| [149] | Whole blood | miR-132 | Up | - | - | - | - | - |
| [107] | Plasma | miR-144-5p | Down | Personalized (8 weeks) | miR-144-5p | Up | - | - |
| [95] | Whole blood | miR-335 | Down | Citalopram (4 weeks) | miR-335 | Up | GRM4 | Direct—in vitro |
| miR-583, mir-650, miR-708, miR-654 | Down | |||||||
| miR-644, miR-450b, mir-328, miR-182 | Up | |||||||
| [125] | PBMC | miRNA-26b, miRNA-1972, miRNA-4485, miRNA-4498, miRNA-4743 | Up | - | - | - | - | - |
| [84] | Whole blood | miR-135a-5p | Down | CBT (3 weeks) | miR-135a-5p | Up | SLC6A4, HTR1A | Direct—in vitro |
| [63] | Whole blood | miR-1202 | Down | Citalopram (8 weeks) | miR-1202 | Up | GRM4 | Direct—in vitro |
| [104] | Serum | miR-182, miR-132 | Up | - | - | - | BDNF | Direct—in vitro |
| [94] | Whole blood | - | - | Escitalopram (12 weeks) | 28 miRNAs | Up | - | |
| miR-34c-5p, miR-770-5p | Down | |||||||
| [124] | PBMC | miR-589, miR-579, miR-941, miR-133a, miR-494, miR-107, miR-148a, miR-652, miR-425-3p | Up | Personalized (8 weeks) | miR-20b-3p, miR-433, miR-409-3p, miR-410, miR-485-3p, miR-133a, miR-145 | Up | - | - |
| miR-517b, miR-636, miR-1243, miR-381, miR-200c | Down | miR-331-5p | Down | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Żurawek, D.; Turecki, G. The miRNome of Depression. Int. J. Mol. Sci. 2021, 22, 11312. https://doi.org/10.3390/ijms222111312
Żurawek D, Turecki G. The miRNome of Depression. International Journal of Molecular Sciences. 2021; 22(21):11312. https://doi.org/10.3390/ijms222111312
Chicago/Turabian StyleŻurawek, Dariusz, and Gustavo Turecki. 2021. "The miRNome of Depression" International Journal of Molecular Sciences 22, no. 21: 11312. https://doi.org/10.3390/ijms222111312






