Regulation of Epigenetic Modifications in the Placenta during Preeclampsia: PPARγ Influences H3K4me3 and H3K9ac in Extravillous Trophoblast Cells
Abstract
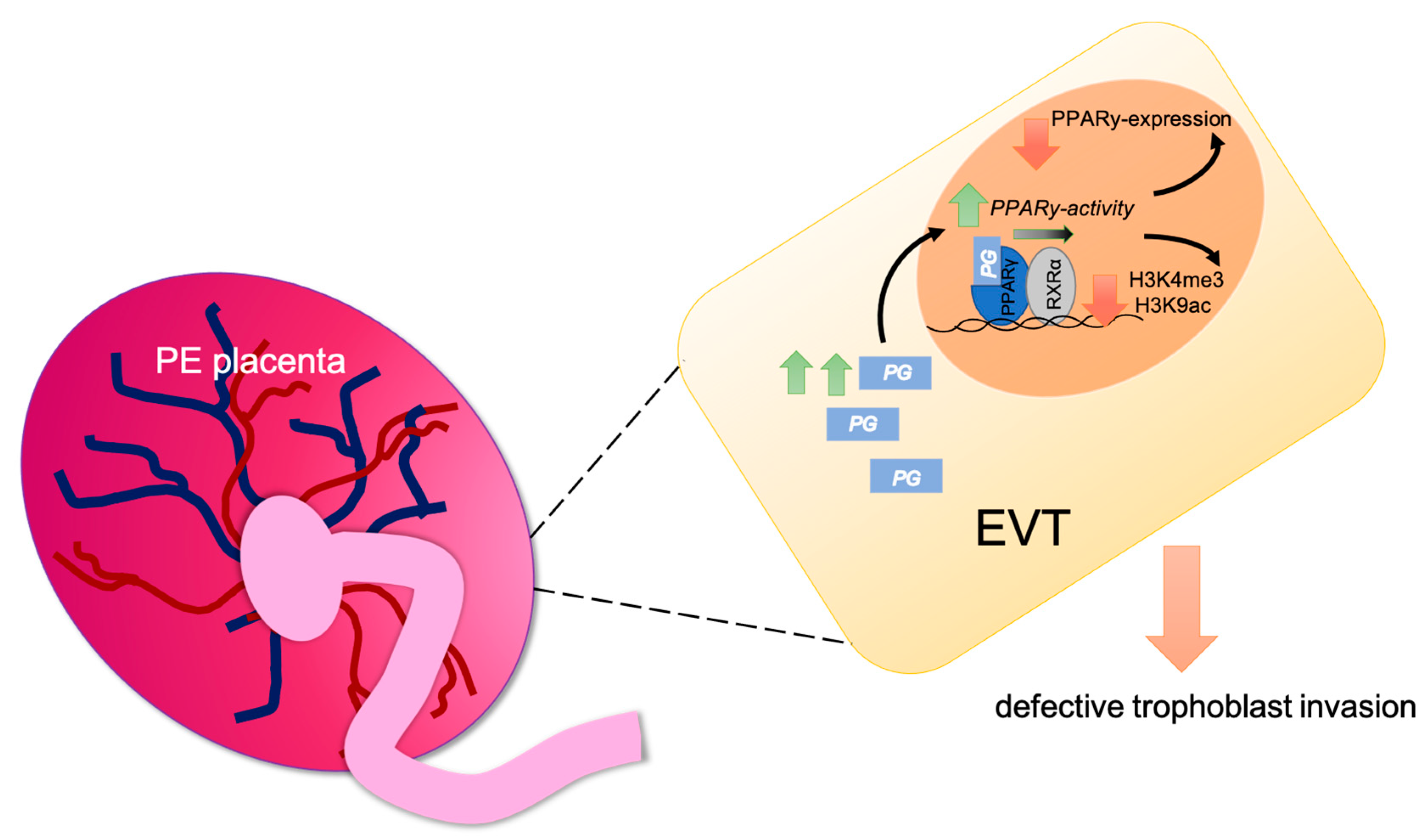
:1. Introduction
2. Results
2.1. Expression of PPARγ and RxRα Is Decreased in the Decidua of PE Placentas
2.2. Correlation of H3K4me3 and H3K9ac with PPARγ and RxRα
2.3. Identification of Decidual Cells Expressing PPARγ/RxRα
2.4. Co-Expression of H3K4me3 and H3K9ac with PPARγ and RxRα
2.5. Specific PPARγ Agonist and Antagonist Regulate H3K4me3 and H3K9ac in Human Trophoblast Cells
3. Discussion
4. Materials and Methods
4.1. Sample—Placental Tissue
4.2. Immunohistochemical Peroxidase Staining
4.3. Double Immunofluorescence Staining of Tissue Slides
4.4. Staining Evaluation
4.5. Isolation of EVT Cells from Placenta Tissue
4.6. FACS of Isolated EVT Cells from Placenta Tissue
4.7. Cell Culture of HVT-Cell Line and Primarily Isolated EVT Cells with PPARγ-Agonist and -Antagonist
4.8. Double Immunofluorescence Staining of Chamber Slides
4.9. Quantitative Analysis of Double Immunofluorescence Staining
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CK 7 | cytokeratin 7 |
| CK 7-PE | phycoerythrin labeled cytokeratin 7 |
| Cy2 | cyanine dyes 2 for fluorescence staining |
| Cy3 | cyanine dyes 3 for fluorescence staining |
| D-PBS | dulbecco’s phosphate buffered saline |
| DAB | chromogenic 3,3′-diaminoenzidine |
| DAPI | 4′,6-Diamino-2-phenylindile |
| DMEM | dulbecco’s Modified Eagle’s Medium |
| EDTA | ethylenediaminetetraacetic acid |
| EVT | extravillous trophoblast |
| FACS | fluorescence-activated cell sorting |
| FBS | fetal bovine serum |
| FCS | fetal calf serum |
| H3K4me3 | trimethylated lysine of the histone H3 |
| H3K9ac | acetylated lysine of the histone H3 |
| HBSS | Hanks’ balanced salt solution Buffer |
| HBSS-HEPES-buffer | Hanks’ balanced salt solution Buffer with 10mM Hepes |
| HEPES | 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid |
| HRP | horseradish peroxidase |
| HVT | human villous trophoblasts, a cell line of cytotrophoblasts |
| IRS | Immunoreactive Score |
| IUGR | intrauterine growth retardation |
| LMU | Ludwig Maximilian University of Munich |
| NaCl | sodium chloride |
| PBS | phosphate-buffered saline |
| PE | preeclampsia (respectively: preeclampsia placentas) |
| Pen | penicillin |
| PFA | perfluoroalkoxy alkane |
| PG | prostaglandins |
| PPARγ | peroxisome proliferator-activated receptor gamma |
| RPMI-1640 | Roswell Park Memorial Institute 1640 |
| RT | room temperature |
| RUPP | reduced uterine perfusion pressure |
| RxRα | retinoid X receptor alpha |
| SD | standard deviation |
| SDS-PAGE | sodium dodecyl sulfate polyacrylamide gel electrophoresis |
| SEM | standard error of the mean |
| Strep | streptomycin |
| T0070907 | antagonist of PPARγ |
| VEGF | vascular endothelial growth factor |
References
- Apicella, C.; Ruano, C.S.M.; Méhats, C.; Miralles, F.; Vaiman, D. The Role of Epigenetics in Placental Development and the Etiology of Preeclampsia. Int. J. Mol. Sci. 2019, 20, 2837. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bakheit, K.H.; Bayoumi, N.K.; Eltom, A.M.; Elbashir, M.I.; Adam, I. Cytokines Profiles in Sudanese Women with Preeclampsia. Hypertens. Pregnancy 2009, 28, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Lee, M.; Jeyabalan, A.; Roberts, J.M. The relationship of hypovitaminosis D and IL-6 in preeclampsia. Am. J. Obstet. Gynecol. 2014, 210, 149.e1–149.e7. [Google Scholar] [CrossRef] [Green Version]
- Weber, M.; Göhner, C.; Martin, S.S.; Vattai, A.; Hutter, S.; Parraga, M.; Jeschke, U.; Schleussner, E.; Markert, U.R.; Fitzgerald, J.S. Unique trophoblast stem cell- and pluripotency marker staining patterns depending on gestational age and placenta-associated pregnancy complications. Cell Adhes. Migr. 2016, 10, 56–65. [Google Scholar] [CrossRef] [Green Version]
- Han, C.; Han, L.; Huang, P.; Chen, Y.; Wang, Y.; Xue, F. Syncytiotrophoblast-Derived Extracellular Vesicles in Pathophysiology of Preeclampsia. Front. Physiol. 2019, 10, 1236. [Google Scholar] [CrossRef] [PubMed]
- Pozharny, Y.; Lambertini, L.; Clunie, G.; Ferrara, L.; Lee, M.-J. Epigenetics in Women’s Health Care. Mt. Sinai J. Med. A J. Transl. Pers. Med. 2010, 77, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, D.; Cui, W.; Rosario, G.X.; Scott, R.L.; Dhakal, P.; Renaud, S.J.; Tachibana, M.; Rumi, M.A.K.; Mason, C.W.; Krieg, A.J.; et al. HIF-KDM3A-MMP12 regulatory circuit ensures trophoblast plasticity and placental adaptations to hypoxia. Proc. Natl. Acad. Sci. USA 2016, 113, E7212–E7221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deng, Z.; Zhang, L.; Tang, Q.; Xu, Y.; Liu, S.; Li, H. Circulating levels of IFN-γ, IL-1, IL-17 and IL-22 in pre-eclampsia: A systematic review and meta-analysis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 248, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Robertson, S.A.; Green, E.S.; Care, A.S.; Moldenhauer, L.M.; Prins, J.R.; Hull, M.L.; Barry, S.C.; Dekker, G. Therapeutic Potential of Regulatory T Cells in Preeclampsia—Opportunities and Challenges. Front. Immunol. 2019, 10. [Google Scholar] [CrossRef]
- Alijotas-Reig, J.; Esteve-Valverde, E.; Olivé, E.L.; Llurba, E.; Gris, J.M. Tumor Necrosis Factor-Alpha and Pregnancy: Focus on Biologics. An Updated and Comprehensive Review. Clin. Rev. Allergy Immunol. 2017, 53, 40–53. [Google Scholar] [CrossRef]
- Scholz, C.; Toth, B.; Santoso, L.; Kuhn, C.; Franz, M.; Mayr, D.; Jeschke, U.; Friese, K.; Schiessl, B. ORIGINAL ARTICLE: Distribution and Maturity of Dendritic Cells in Diseases of Insufficient Placentation. Am. J. Reprod. Immunol. 2008, 60, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Mohammadpour-Gharehbagh, A.; Jahantigh, D.; Eskandari, M.; Eskandari, F.; Rezaei, M.; Zeynali-Moghaddam, S.; Teimoori, B.; Salimi, S. The role of TNF-α and TLR4 polymorphisms in the placenta of pregnant women complicated by preeclampsia and in silico analysis. Int. J. Biol. Macromol. 2019, 134, 1205–1215. [Google Scholar] [CrossRef] [PubMed]
- Ushida, T.; MacDonald-Goodfellow, S.K.; Quadri, A.; Tse, M.Y.; Winn, L.M.; Pang, S.C.; Adams, M.A.; Kotani, T.; Kikkawa, F.; Graham, C.H. Persistence of risk factors associated with maternal cardiovascular disease following aberrant inflammation in rat pregnancy. Biol. Reprod. 2017, 97, 143–152. [Google Scholar] [CrossRef]
- Eddy, A.C.; Chapman, H.; George, E.M. Acute Hypoxia and Chronic Ischemia Induce Differential Total Changes in Placental Epigenetic Modifications. Reprod. Sci. 2019, 26, 766–773. [Google Scholar] [CrossRef]
- Minas, V.; Mylonas, I.; Schiessl, B.; Mayr, D.; Schulze, S.; Friese, K.; Jeschke, U.; Makrigiannakis, A. Expression of the blood-group-related antigens Sialyl Lewis a, Sialyl Lewis x and Lewis y in term placentas of normal, preeclampsia, IUGR- and HELLP-complicated pregnancies. Histochem. Cell Biol. 2007, 128, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Kong, L. LncRNA CRNDE regulates trophoblast cell proliferation, invasion, and migration via modulating miR-1277. Am. J. Transl. Res. 2019, 11, 5905–5918. [Google Scholar]
- Heim, K.; Mulla, M.J.; Potter, J.A.; Han, C.S.; Guller, S.; Abrahams, V.M. Excess glucose induce trophoblast inflammation and limit cell migration through HMGB1 activation of Toll-Like receptor 4. Am. J. Reprod. Immunol. 2018, 80, e13044. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Ye, Y.; Zhang, J.; Ruan, C.-C.; Gao, P.-J. Immune imbalance is associated with the development of preeclampsia. Medicine 2019, 98, e15080. [Google Scholar] [CrossRef] [PubMed]
- Weel, I.; Romão-Veiga, M.; Matias, M.L.; Fioratti, E.G.; Peraçoli, J.C.; Borges, V.T.; Araujo, J.P.; Peraçoli, M.T. Increased expression of NLRP3 inflammasome in placentas from pregnant women with severe preeclampsia. J. Reprod. Immunol. 2017, 123, 40–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levytska, K.; Drewlo, S.; Baczyk, R.; Kingdom, J. PPAR-γ Regulates Trophoblast Differentiation in the BeWo Cell Model. PPAR Res. 2014, 2014, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaufmann, P.; Black, S.; Huppertz, B. Endovascular Trophoblast Invasion: Implications for the Pathogenesis of Intrauterine Growth Retardation and Preeclampsia. Biol. Reprod. 2003, 69, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baines, K.; Renaud, S. Transcription Factors That Regulate Trophoblast Development and Function. Prog. Mol. Biol. Transl. Sci. 2017, 145, 39–88. [Google Scholar] [CrossRef] [PubMed]
- Lyall, F.; Robson, S.C.; Bulmer, J.N. Spiral artery remodeling and trophoblast invasion in preeclampsia and fetal growth restriction: Relationship to clinical outcome. Hypertension 2013, 62, 1046–1054. [Google Scholar] [CrossRef] [Green Version]
- Naicker, T.; Khedun, S.M.; Moodly, J.; Pijnenborg, R. Quantitative analysis of trophoblast invasion in preeclampsia. Acta Obstet. Gynecol. Scand. 2003, 82, 722–729. [Google Scholar] [CrossRef]
- Matthiesen, L.; Berg, G.; Ernerudh, J.; Ekerfelt, C.; Jonsson, Y.; Sharma, S. Immunology of Preeclampsia. Hist. Allergy 2005, 89, 49–61. [Google Scholar] [CrossRef]
- Barak, Y.; Sadovsky, Y.; Shalom-Barak, T. PPAR Signaling in Placental Development and Function. PPAR Res. 2007, 2008, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Evans, R.M.; Barrish, G.D.; Wang, Y.-X. PPARs and the complex journey to obesity. Nat. Med. 2004, 10, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Glass, C.K. Going nuclear in metabolic and cardiovascular disease. J. Clin. Investig. 2006, 116, 556–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Straus, D.S.; Glass, C.K. Anti-inflammatory actions of PPAR ligands: New insights on cellular and molecular mechanisms. Trends Immunol. 2007, 28, 551–558. [Google Scholar] [CrossRef] [PubMed]
- Tache, V.; Ciric, A.; Moretto-Zita, M.; Li, Y.; Peng, J.; Maltepe, E.; Milstone, D.S.; Parast, M.M. Hypoxia and Trophoblast Differentiation: A Key Role for PPARγ. Stem Cells Dev. 2013, 22, 2815–2824. [Google Scholar] [CrossRef] [Green Version]
- He, P.; Chen, Z.; Sun, Q.; Li, Y.; Gu, H.; Ni, X. Reduced Expression of 11β-Hydroxysteroid Dehydrogenase Type 2 in Preeclamptic Placentas Is Associated with Decreased PPARγ but Increased PPARα Expression. Endocrinology 2014, 155, 299–309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruebner, M.; Langbein, M.; Strissel, P.L.; Henke, C.; Schmidt, D.; Goecke, T.W.; Faschingbauer, F.; Schild, R.L.; Beckmann, M.W.; Strick, R. Regulation of the human endogenous retroviral Syncytin-1 and cell-cell fusion by the nuclear hormone receptors PPARγ/RXRα in placentogenesis. J. Cell. Biochem. 2012, 113, 2383–2396. [Google Scholar] [CrossRef]
- Blitek, A.; Szymanska, M. Expression and role of peroxisome proliferator–activated receptors in the porcine early placenta trophoblast. Domest. Anim. Endocrinol. 2018, 67, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Fournier, T.; Pavan, L.; Tarrade, A.; Schoonjans, K.; Auwerx, J.; Rochette-Egly, C.; Evain-Brion, D. The Role of PPAR-γ/RXR-α Heterodimers in the Regulation of Human Trophoblast Invasion. Ann. N. Y. Acad. Sci. 2002, 973, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Tarrade, A.; Schoonjans, K.; Guibourdenche, J.; Bidart, J.M.; Vidaud, M.; Auwerx, J.; Rochette-Egly, C.; Evain-Brion, D. PPARγ/RXRα Heterodimers Are Involved in Human CGβ Synthesis and Human Trophoblast Differentiation. Endocrinology 2001, 142, 4504–4514. [Google Scholar] [CrossRef]
- Fang, S.; Livergood, M.C.; Nakagawa, P.; Wu, J.; Sigmund, C.D. Role of the Peroxisome Proliferator Activated Receptors in Hypertension. Circ. Res. 2021, 128, 1021–1039. [Google Scholar] [CrossRef] [PubMed]
- Permadi, W.; Mantilidewi, K.I.; Khairani, A.F.; Lantika, U.A.; Ronosulistyo, A.R.; Bayuaji, H. Differences in expression of Peroxisome Proliferator-activated Receptor-γ in early-onset preeclampsia and late-onset preeclampsia. BMC Res. Notes 2020, 13, 1–6. [Google Scholar] [CrossRef]
- Holdsworth-Carson, S.; Lim, R.; Mitton, A.; Whitehead, C.; Rice, G.; Permezel, M.; Lappas, M. Peroxisome proliferator-activated receptors are altered in pathologies of the human placenta: Gestational diabetes mellitus, intrauterine growth restriction and preeclampsia. Placenta 2010, 31, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Wiedemann, A.; Vocke, F.; Fitzgerald, J.S.; Markert, U.R.; Jeschke, U.; Lohse, P.; Toth, B. ORIGINAL ARTICLE: Leptin Gene (TTTC)n Microsatellite Polymorphism as well as Leptin Receptor R223Q and PPARγ2 P12A Substitutions are not Associated with Hypertensive Disorders in Pregnancy. Am. J. Reprod. Immunol. 2010, 63, 310–317. [Google Scholar] [CrossRef]
- Waite, L.L.; Louie, R.E.; Taylor, R.N. Circulating Activators of Peroxisome Proliferator-Activated Receptors Are Reduced in Preeclamptic Pregnancy. J. Clin. Endocrinol. Metab. 2005, 90, 620–626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Y.; Guo, S.-W. Peroxisome proliferator-activated receptor-gamma and retinoid X receptor agonists synergistically suppress proliferation of immortalized endometrial stromal cells. Fertil. Steril. 2009, 91, 2142–2147. [Google Scholar] [CrossRef]
- Patel, H.; Truant, R.; Rachubinski, R.A.; Capone, J.P. Activity and subcellular compartmentalization of peroxisome proliferator-activated receptor α are altered by the centrosome-associated protein CAP350. J. Cell Sci. 2005, 118, 175–186. [Google Scholar] [CrossRef] [Green Version]
- Park, H.K.; Kim, H.; Kim, H.-G.; Cho, Y.M.; Jung, W.Y.; Han, H.S.; Hwang, T.S.; Kwon, G.Y.; Lim, S.D. Expression of Peroxisome Proliferator Activated Receptor gamma in Prostatic Adenocarcinoma. J. Korean Med. Sci. 2015, 30, 533–541. [Google Scholar] [CrossRef] [Green Version]
- Shao, W.; Kuhn, C.; Mayr, D.; Ditsch, N.; Kailuwait, M.; Wolf, V.; Harbeck, N.; Mahner, S.; Jeschke, U.; Cavaillès, V.; et al. Cytoplasmic PPARγ is a marker of poor prognosis in patients with Cox-1 negative primary breast cancers. J. Transl. Med. 2020, 18, 94. [Google Scholar] [CrossRef] [PubMed]
- Umemoto, T.; Fujiki, Y. Ligand-dependent nucleo-cytoplasmic shuttling of peroxisome proliferator-activated receptors, PPARα and PPARγ. Genes Cells 2012, 17, 576–596. [Google Scholar] [CrossRef] [PubMed]
- Kamrani, A.; Alipourfard, I.; Ahmadi-Khiavi, H.; Yousefi, M.; Rostamzadeh, D.; Izadi, M.; Ahmadi, M. The role of epigenetic changes in preeclampsia. BioFactors 2019, 45, 712–724. [Google Scholar] [CrossRef]
- Patsouras, M.; Vlachoyiannopoulos, P. Evidence of epigenetic alterations in thrombosis and coagulation: A systematic review. J. Autoimmun. 2019, 104, 102347. [Google Scholar] [CrossRef]
- Rasmussen, L.G.; Lykke, J.A.; Staff, A.C. Angiogenic biomarkers in pregnancy: Defining maternal and fetal health. Acta Obstet. Gynecol. Scand. 2015, 94, 820–832. [Google Scholar] [CrossRef]
- Meister, S.; Hahn, L.; Beyer, S.; Kuhn, C.; Jegen, M.; von Schönfeldt, V.; Corradini, S.; Schulz, C.; Kolben, T.M.; Hester, A.; et al. Epigenetic modification via H3K4me3 and H3K9ac in human placenta is reduced in preeclampsia. J. Reprod. Immunol. 2021, 145, 103287. [Google Scholar] [CrossRef]
- Hutter, S.; Marti, N.; von Schönfeldt, V.; Messner, J.; Kuhn, C.; Hofmann, S.; Andergassen, U.; Knabl, J.; Jeschke, U. Galectin 2 (gal-2) expression is downregulated on protein and mRNA level in placentas of preeclamptic (PE) patients. Placenta 2015, 36, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Dreijerink, K.; Varier, R.A.; van Beekum, O.; Jeninga, E.H.; Höppener, J.W.M.; Lips, C.J.M.; Kummer, J.A.; Kalkhoven, E.; Timmers, H.T.M. The Multiple Endocrine Neoplasia Type 1 (MEN1) Tumor Suppressor Regulates Peroxisome Proliferator-Activated Receptor γ-Dependent Adipocyte Differentiation. Mol. Cell. Biol. 2009, 29, 5060–5069. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cho, Y.-W.; Hong, S.; Jin, Q.; Wang, L.; Lee, J.-E.; Gavrilova, O.; Ge, K. Histone Methylation Regulator PTIP Is Required for PPARγ and C/EBPα Expression and Adipogenesis. Cell Metab. 2009, 10, 27–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sonkar, R.; Powell, C.A.; Choudhury, M. Benzyl butyl phthalate induces epigenetic stress to enhance adipogenesis in mesenchymal stem cells. Mol. Cell. Endocrinol. 2016, 431, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.P.H.; Patrick, C.J.; Parry, L.J.; Familari, M. Using proteomics to advance the search for potential biomarkers for preeclampsia: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0214671. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Irgens, H.U.; Reisæter, L.; Irgens, L.M.; Lie, R.T.; Roberts, J.M. Long term mortality of mothers and fathers after pre-eclampsia: Population based cohort study Pre-eclampsia and cardiovascular disease later in life: Who is at risk? BMJ 2001, 323, 1213–1217. [Google Scholar] [CrossRef] [Green Version]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum: Mahwah, NJ, USA, 1988. [Google Scholar]
- Benyo, D.F.; Smárason, A.; Redman, C.W.G.; Sims, C.; Conrad, K.P. Expression of Inflammatory Cytokines in Placentas from Women with Preeclampsia1. J. Clin. Endocrinol. Metab. 2001, 86, 2505–2512. [Google Scholar] [CrossRef] [Green Version]
- Jang, M.-K.; Yun, Y.-R.; Kim, J.-H.; Park, M.-H.; Jung, M.H. Gomisin N inhibits adipogenesis and prevents high-fat diet-induced obesity. Sci. Rep. 2017, 7, 40345. [Google Scholar] [CrossRef]
- Liu, Y.; Luo, S.; Zhan, Y.; Wang, J.; Zhao, R.; Li, Y.; Zeng, J.; Lu, Q. Increased Expression of PPAR-γ Modulates Monocytes Into a M2-Like Phenotype in SLE Patients: An Implicative Protective Mechanism and Potential Therapeutic Strategy of Systemic Lupus Erythematosus. Front. Immunol. 2021, 11, 3384. [Google Scholar] [CrossRef]
- Emamgholipour, S.; Ebrahimi, R.; Bahiraee, A.; Niazpour, F.; Meshkani, R. Acetylation and insulin resistance: A focus on metabolic and mitogenic cascades of insulin signaling. Crit. Rev. Clin. Lab. Sci. 2020, 57, 196–214. [Google Scholar] [CrossRef]
- Choi, J.H.; Lee, H. Histone demethylase KDM4D cooperates with NFIB and MLL1 complex to regulate adipogenic differentiation of C3H10T1/2 mesenchymal stem cells. Sci. Rep. 2020, 10, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Lemberger, T.; Desvergne, B.; Wahli, W. PEROXISOME PROLIFERATOR-ACTIVATED RECEPTORS: A Nuclear Receptor Signaling Pathway in Lipid Physiology. Annu. Rev. Cell Dev. Biol. 1996, 12, 335–363. [Google Scholar] [CrossRef] [PubMed]
- Lane, S.L.; Dodson, R.B.; Doyle, A.S.; Park, H.; Rathi, H.; Matarrazo, C.J.; Moore, L.G.; Lorca, R.A.; Wolfson, G.H.; Julian, C.G. Pharmacological activation of peroxisome proliferator-activated receptor γ (PPAR-γ) protects against hypoxia-associated fetal growth restriction. FASEB J. 2019, 33, 8999–9007. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, F.P.; Drewlo, S.; Kingdom, J.; Johns, E.J.; Walsh, S.K.; Kenny, L.C. Peroxisome Proliferator-Activated Receptor-γ as a Potential Therapeutic Target in the Treatment of Preeclampsia. J. Hypertens. 2011, 58, 280–286. [Google Scholar] [CrossRef] [Green Version]
- Schaiff, W.T.; Carlson, M.G.; Smith, S.D.; Levy, R.; Nelson, D.M.; Sadovsky, Y. Peroxisome Proliferator-Activated Receptor-γ Modulates Differentiation of Human Trophoblast in a Ligand-Specific Manner 1. J. Clin. Endocrinol. Metab. 2000, 85, 3874–3881. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barak, Y.; Nelson, M.C.; Ong, E.S.; Jones, Y.Z.; Ruiz-Lozano, P.; Chien, K.R.; Koder, A.; Evans, R.M. PPARγ Is Required for Placental, Cardiac, and Adipose Tissue Development. Mol. Cell 1999, 4, 585–595. [Google Scholar] [CrossRef]
- Fournier, T.; Handschuh, K.; Tsatsaris, V.; Evain-Brion, D. Involvement of PPARγ in Human Trophoblast Invasion. Placenta 2007, 28, S76–S81. [Google Scholar] [CrossRef] [PubMed]
- Nadra, K.; Quignodon, L.; Sardella, C.; Joye, E.; Mucciolo, A.; Chrast, R.; Desvergne, B. PPARγ in Placental Angiogenesis. Endocrinology 2010, 151, 4969–4981. [Google Scholar] [CrossRef]
- Mashayekhi, S.; Yousefi, B.; Tohidi, E.; Darband, S.G.; Mirza-Aghazadeh-Attari, M.; Sadighparvar, S.; Kaviani, M.; Shafiei-Irannejad, V.; Kafil, H.S.; Karimian, A.; et al. Overexpression of tensin homolog deleted on chromosome ten (PTEN) by ciglitazone sensitizes doxorubicin-resistance leukemia cancer cells to treatment. J. Cell. Biochem. 2019, 120, 15719–15729. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.W.; Choi, C.H.; Park, J.Y.; Kang, S.K.; Kim, Y.K. Ciglitazone Induces Caspase-Independent Apoptosis through Down-Regulation of XIAP and Survivin in Human Glioma Cells. Neurochem. Res. 2008, 33, 551–561. [Google Scholar] [CrossRef]
- An, Z.; Muthusami, S.; Yu, J.-R.; Park, W.-Y. T0070907, a PPAR γ Inhibitor, Induced G2/M Arrest Enhances the Effect of Radiation in Human Cervical Cancer Cells through Mitotic Catastrophe. Reprod. Sci. 2014, 21, 1352–1361. [Google Scholar] [CrossRef] [Green Version]
- Chan, S.L.; Cipolla, M.J. Determination of PPARgamma activity in adipose tissue and spleen. J. Immunoass. Immunochem 2012, 33, 314–324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jianhua, L.; Xueqin, M.; Jifen, H. Expression and clinical significance of LXRalpha and SREBP-1c in placentas of preeclampsia. Open Med. 2016, 11, 292–296. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.D.; Sadovsky, Y.; Graham, C.; Anteby, E.Y.; Polakoski, K.L.; Huang, X.; Nelson, D.M. The Expression and Activity of Prostaglandin H Synthase-2 Is Enhanced in Trophoblast from Women with Preeclampsia. J. Clin. Endocrinol. Metab. 1997, 82, 3059–3062. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J.; Guo, C.; Wu, J. 15-Deoxy—(12,14)-Prostaglandin J2 (15d-PGJ2), an Endogenous Ligand of PPAR-gamma: Function and Mechanism. PPAR Res. 2019, 2019, 7242030. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plissonnier, M.-L.; Fauconnet, S.; Bittard, H.; Mougin, C.; Rommelaere, J.; Lascombe, I. Cell death and restoration of TRAIL-sensitivity by ciglitazone in resistant cervical cancer cells. Oncotarget 2017, 8, 107744–107762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- An, Z.; Yu, J.-R.; Park, W.-Y. T0070907 inhibits repair of radiation-induced DNA damage by targeting RAD51. Toxicol. In Vitro 2016, 37, 1–8. [Google Scholar] [CrossRef] [PubMed]







| H3K4me3 | H3K9ac | |||
|---|---|---|---|---|
| Syncytium | Decidua | Syncytium | Decidua | |
| RxRα syncytium | 0.448 ** 0.002 | 0.513 ** 0.000 | 0.428 ** 0.003 | 0.577 ** 0.000 |
| RxRα decidua | 0.406 ** 0.007 | 0.327 * 0.032 | 0.389 ** 0.010 | 0.464 ** 0.002 |
| PPARγ syncytium | 0.216 0.131 | 0.249 0.084 | 0.284 * 0.046 | 0.379 ** 0.007 |
| PPARγ decidua | 0.357 * 0.012 | 0.227 0.121 | 0.347 * 0.014 | 0.399 ** 0.004 |
| Antibody | Species Isotype | Manufacturer | Dilution | |
|---|---|---|---|---|
| H3K9ac | Rabbit IgG monoclonal (Clone: Y28) | Abcam | 1:200 | IH, IF, IC |
| H3K4me3 | Rabbit IgG polyclonal | Abcam | 1:100 | IH, IF, IC |
| RxRα | Mouse IgG2a monoclonal (Clone: K8508) | Perseus Proteomics | 1:200 | IH, IF, IC |
| PPARγ | Rabbit IgG1 polyclonal | Abcam | 1:100 | IH |
| PPARγ | Mouse IgG1 monoclonal (Clone: 8D1H8F4) | Abnova | 1:500 | IC |
| PPARγ | Rabbit IgG polyclonal | LSBio | 1:500 | IF |
| CK 7 | Mouse IgG1 monoclonal (Clone: NCL-L-CK7-OVTL) | Novocastra | 1:30 | IF |
| Cy2 | Goat-Anti-Mouse IgG | Dianova | 1:100 | IF, IC |
| Cy3 | Goat-Anti-Rabbit IgG | Dianova | 1:500 | IF, IC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meister, S.; Hahn, L.; Beyer, S.; Paul, C.; Mitter, S.; Kuhn, C.; von Schönfeldt, V.; Corradini, S.; Sudan, K.; Schulz, C.; et al. Regulation of Epigenetic Modifications in the Placenta during Preeclampsia: PPARγ Influences H3K4me3 and H3K9ac in Extravillous Trophoblast Cells. Int. J. Mol. Sci. 2021, 22, 12469. https://doi.org/10.3390/ijms222212469
Meister S, Hahn L, Beyer S, Paul C, Mitter S, Kuhn C, von Schönfeldt V, Corradini S, Sudan K, Schulz C, et al. Regulation of Epigenetic Modifications in the Placenta during Preeclampsia: PPARγ Influences H3K4me3 and H3K9ac in Extravillous Trophoblast Cells. International Journal of Molecular Sciences. 2021; 22(22):12469. https://doi.org/10.3390/ijms222212469
Chicago/Turabian StyleMeister, Sarah, Laura Hahn, Susanne Beyer, Corinna Paul, Sophie Mitter, Christina Kuhn, Viktoria von Schönfeldt, Stefanie Corradini, Kritika Sudan, Christian Schulz, and et al. 2021. "Regulation of Epigenetic Modifications in the Placenta during Preeclampsia: PPARγ Influences H3K4me3 and H3K9ac in Extravillous Trophoblast Cells" International Journal of Molecular Sciences 22, no. 22: 12469. https://doi.org/10.3390/ijms222212469







