Phytosterols in Seaweeds: An Overview on Biosynthesis to Biomedical Applications
Abstract
:1. Introduction
2. Biosynthesis of Phytosterols
2.1. Biosynthesis of Isoprene
2.2. Condensation of Isoprene into Triterpenoids and Their Epoxidation
2.3. Phytosterol Synthesis
3. Extraction and Characterization of Phytosterols
4. Health Benefits and Biomedical Applications of Phytosterols
4.1. Phytosterols as Antioxidative Agents
4.2. Phytosterols as Antimicrobial Agents
4.3. Phytosterols as Anti-Inflammatory Agents
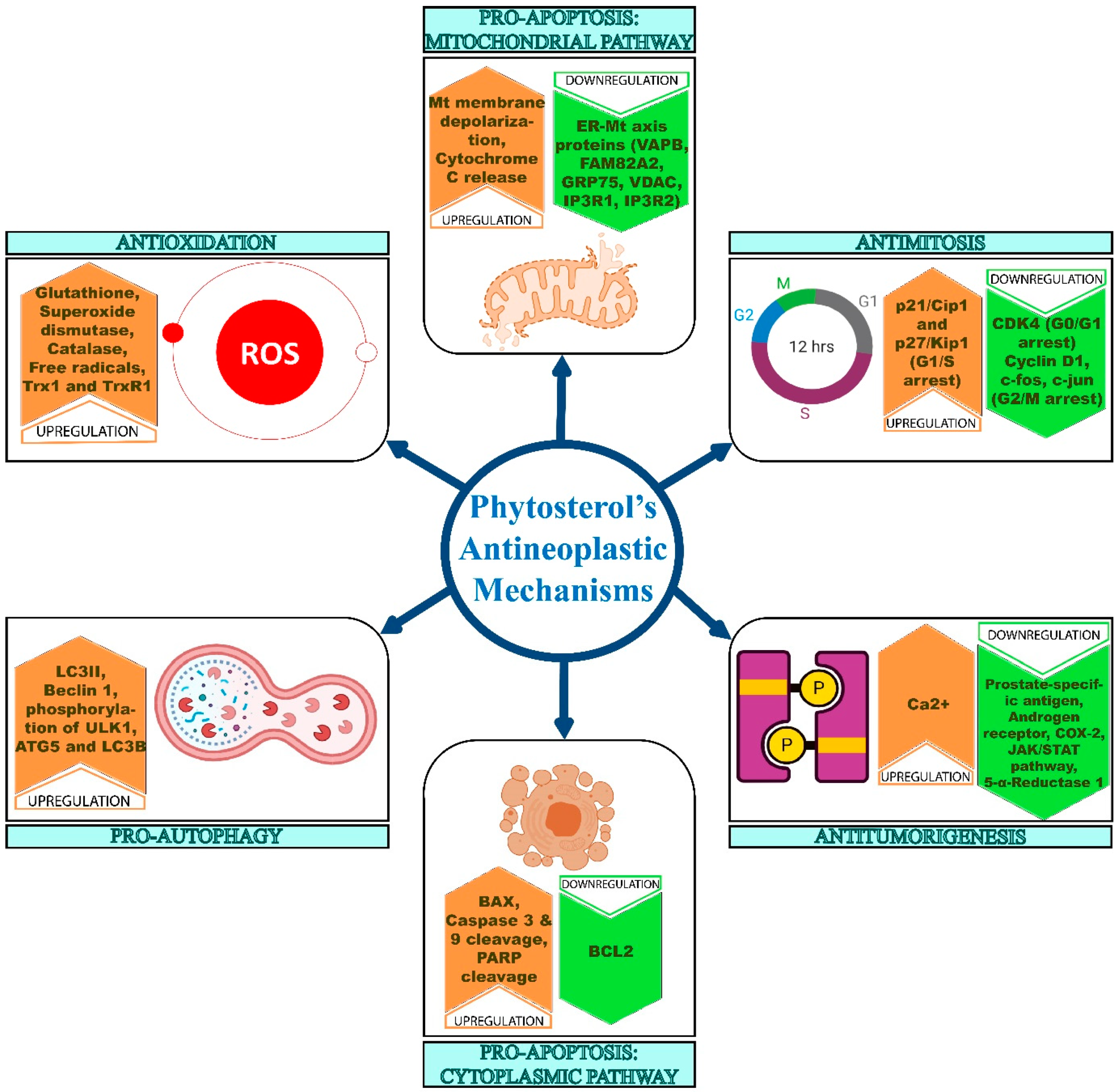
4.4. Phytosterols as Anticancer Agents
5. Safety of Phytosterols
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Shirosaki, M.; Koyama, T. Laminaria japonica as a food for the prevention of obesity and diabetes. Adv. Food Nutr. Res. 2011, 64, 199–212. [Google Scholar] [PubMed]
- Chen, Z.; Liu, J.; Fu, Z.; Ye, C.; Zhang, R.; Song, Y.; Zhang, Y.; Li, H.; Ying, H.; Liu, H. 24 (S)-Saringosterol from edible marine seaweed Sargassum fusiforme is a novel selective LXRβ agonist. J. Agric. Food Chem. 2014, 62, 6130–6137. [Google Scholar] [CrossRef]
- Kılınç, B.; Cirik, S.; Turan, G.; Tekogul, H.; Koru, E. Seaweeds for Food and Industrial Applications. In Food Industry; IntechOpen: London, UK, 2013. [Google Scholar]
- Ling, W.H.; Jones, P.J.H. Dietary phytosterols: A review of metabolism, benefits and side effects. Life Sci. 1995, 57, 195–206. [Google Scholar] [CrossRef]
- Lopes, G.; Sousa, C.; Valentão, P.; Andrade, P.B. Sterols in Algae and Health. In Bioactive Compounds from Marine Foods: Plant and Animal Sources; Wiley Online Library: Hoboken, NJ, USA, 2013; pp. 173–191. [Google Scholar]
- Pereira, C.M.; Nunes, C.F.; Zambotti-Villela, L.; Streit, N.M.; Dias, D.; Pinto, E.; Colepicolo, P. Extraction of sterols in brown macroalgae from Antarctica and their identification by liquid chromatography coupled with tandem mass spectrometry. J. Appl. Phycol. 2017, 29, 751–757. [Google Scholar] [CrossRef]
- Whittaker, M.H.; Frankos, V.H.; Wolterbeek, A.M.P.; Waalkens-Berendsen, D.H. Effects of dietary phytosterols on cholesterol metabolism and atherosclerosis: Clinical and experimental evidence. Am. J. Med. 2000, 109, 600–601. [Google Scholar] [CrossRef]
- Sánchez-Machado, D.I.; López-Hernández, J.; Paseiro-Losada, P.; López-Cervantes, J. An HPLC method for the quantification of sterols in edible seaweeds. Biomed. Chromatogr. 2004, 18, 183–190. [Google Scholar] [CrossRef]
- Dutta, P.C. Phytosterols as Functional Food Components and Nutraceuticals; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Nes, W.D. Biosynthesis of cholesterol and other sterols. Chem. Rev. 2011, 111, 6423–6451. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.H.; Yin, X.; Welander, P.V. Sterol Synthesis in Diverse Bacteria. Front. Microbiol. 2016, 7, 990. [Google Scholar] [CrossRef] [Green Version]
- Hannan, M.A.; Sohag, A.A.M.; Dash, R.; Haque, M.N.; Mohibbullah, M.; Oktaviani, D.F.; Moon, I.S. Phytosterols of marine algae: Insights into the potential health benefits and molecular pharmacology. Phytomedicine 2020, 69, 153201. [Google Scholar] [CrossRef]
- Schepers, M.; Martens, N.; Tiane, A.; Vanbrabant, K.; Liu, H.B.; Lütjohann, D.; Mulder, M.; Vanmierlo, T. Edible seaweed-derived constituents: An undisclosed source of neuroprotective compounds. Neural. Regen. Res. 2020, 15, 790–795. [Google Scholar] [CrossRef] [PubMed]
- Calegario, G.; Pollier, J.; Arendt, P.; de Oliveira, L.S.; Thompson, C.; Soares, A.R.; Pereira, R.C.; Goossens, A.; Thompson, F.L. Cloning and Functional Characterization of Cycloartenol Synthase from the Red Seaweed Laurencia dendroidea. PLoS ONE. 2016, 11, e0165954. [Google Scholar] [CrossRef] [Green Version]
- Lohr, M.; Schwender, J.; Polle, J.E. Isoprenoid biosynthesis in eukaryotic phototrophs: A spotlight on algae. Plant Sci. 2012, 185, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Chatzivasileiou, A.O.; Ward, V.; Edgar, S.M.; Stephanopoulos, G. Two-step pathway for isoprenoid synthesis. Proc. Natl. Acad. Sci. USA 2019, 116, 506–511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harder, J. Isoprene, Isoprenoids and Sterols. In Handbook of Hydrocarbon and Lipid Microbiology; Timmis, K.N., Ed.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 127–131. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. Evolution of Carotenoid and Isoprenoid Biosynthesis in Photosynthetic and Nonphotosynthetic Organisms. In Proceedings of the 16th International Plant Lipid Symposium, Budapest, Hungary, 1–4 June 2004. [Google Scholar]
- Pulido, P.; Perello, C.; Rodriguez-Concepcion, M. New insights into plant isoprenoid metabolism. Mol. Plant 2012, 5, 964–967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vranová, E.; Coman, D.; Gruissem, W. Network analysis of the MVA and MEP pathways for isoprenoid synthesis. Annu. Rev. Plant Biol. 2013, 64, 665–700. [Google Scholar] [CrossRef]
- Weber, H. Fatty acid-derived signals in plants. Trends Plant Sci. 2002, 7, 217–224. [Google Scholar] [CrossRef]
- Dewick, P.M. The biosynthesis of C5-C25 terpenoid compounds. Nat. Prod. Rep. 2002, 19, 181–222. [Google Scholar] [CrossRef]
- Perreca, E.; Rohwer, J.; González-Cabanelas, D.; Loreto, F.; Schmidt, A.; Gershenzon, J.; Wright, L.P. Effect of Drought on the Methylerythritol 4-Phosphate (MEP) Pathway in the Isoprene Emitting Conifer Picea glauca. Front. Plant Sci. 2020, 11, 1535. [Google Scholar] [CrossRef]
- Sonawane, P.D.; Pollier, J.; Panda, S.; Szymanski, J.; Massalha, H.; Yona, M.; Unger, T.; Malitsky, S.; Arendt, P.; Pauwels, L.; et al. Plant cholesterol biosynthetic pathway overlaps with phytosterol metabolism. Nat. Plants. 2016, 3, 16205. [Google Scholar] [CrossRef]
- Cabianca, A.; Müller, L.; Pawlowski, K.; Dahlin, P. Changes in the Plant β-Sitosterol/Stigmasterol Ratio Caused by the Plant Parasitic Nematode Meloidogyne incognita. Plants 2021, 10, 292. [Google Scholar] [CrossRef]
- Ohyama, K.; Suzuki, M.; Kikuchi, J.; Saito, K.; Muranaka, T. Dual biosynthetic pathways to phytosterol via cycloartenol and lanosterol in Arabidopsis. Proc. Natl. Acad. Sci. USA 2009, 106, 725–730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grosso, C.; Valentão, P.; Ferreres, F.; Andrade, P.B. Alternative and efficient extraction methods for marine-derived compounds. Mar. Drugs 2015, 13, 3182–3230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lagarda, M.J.; Garcia-Llatas, G.; Farré, R. Analysis of phytosterols in foods. Rev. J. Pharm. Biomed. Anal. 2006, 41, 1486–1496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, A.; Naraniwal, M.; Kothari, V. Modern extraction methods for preparation of bioactive plant extracts. Int. J. Appl. Nat. Sci. 2012, 1, 8–26. [Google Scholar]
- Smith, R.M. Before the injection-modern methods of sample preparation for separation techniques. Rev. J. Chromatogr. A 2003, 1000, 3–27. [Google Scholar] [CrossRef]
- Uddin, M.S.; Ferdosh, S.; Haque Akanda, M.J.; Ghafoor, K.; Ah, R.; Ali, M.E.; Kamaruzzaman, B.Y.; Mb, F.; Shaarani, S.; Islam Sarker, M.Z. Techniques for the extraction of phytosterols and their benefits in human health: A review. Sep. Sci. Technol. 2018, 53, 2206–2223. [Google Scholar]
- Poulose, N.; Sajayan, A.; Ravindran, A.; Chandran, A.; Priyadharshini, G.B.; Selvin, J.; Kiran, G.S. Anti-diabetic Potential of a Stigmasterol from the Seaweed Gelidium spinosum and Its Application in the Formulation of Nanoemulsion Conjugate for the Development of Functional Biscuits. Front. Nutr. 2021, 8, 694362. [Google Scholar] [CrossRef]
- De Santi, I.I.; Pacheco, B.S.; Venzke, D.; Freitag, R.A.; de Almeida, L.S.; Colepicolo, P.; Pereira, C.M. Sterols in red macroalgae from antarctica: Extraction and quantification by Gas Chromatography–Mass spectrometry. Polar Biol. 2021, 44, 987–995. [Google Scholar] [CrossRef]
- Gololo, S.S.; Shai, L.J.; Sethoga, L.; Agyei, N.; Bassey, K.E.; Mogale, M.A. Isolation of a mixture of Phytosterol compounds from the n-Hexane extract of Jatropha lagarinthoides (Sond) collected from Zebediela sub-region in Limpopo province, South Africa. J. Chem. Pharm. Sci. 2016, 9, 3084–3087. [Google Scholar]
- De Jong, D.L.; Timmermans, K.R.; de Winter, J.M.; Derksen, G.C. Effects of nutrient availability and light intensity on the sterol content of Saccharina latissima (Laminariales, Phaeophyceae). J. Appl. Phycol. 2021, 33, 1101–1113. [Google Scholar] [CrossRef]
- Saini, R.K.; Mahomoodally, M.F.; Sadeer, N.B.; Keum, Y.S.; Rr Rengasamy, K. Characterization of nutritionally important lipophilic constituents from brown kelp Ecklonia radiata (C. Ag.). J. Agardh. Food Chem. 2021, 340, 127897. [Google Scholar] [CrossRef]
- Akbary, P.; Liao, L.M.; Aminikhoei, Z.; Tavabe, K.R.; Hobbi, M.; Erfanifar, E. Sterol and fatty acid profiles of three macroalgal species collected from the Chabahar coasts, southeastern Iran. Aquac. Int. 2021, 29, 155–165. [Google Scholar] [CrossRef]
- Berneira, L.M.; de Santi, I.I.; da Silva, C.C.; Venzke, D.; Colepicolo, P.; Vaucher, R.A.; Dos Santos, M.; de Pereira, C. Bioactivity and composition of lipophilic metabolites extracted from Antarctic macroalgae. Braz. J. Microbiol. 2021, 52, 1275–1285. [Google Scholar] [CrossRef] [PubMed]
- Cvitković, D.; Dragović-Uzelac, V.; Dobrinčić, A.; Čož-Rakovac, R.; Balbino, S. The effect of solvent and extraction method on the recovery of lipid fraction from Adriatic Sea macroalgae. Algal Res. 2021, 56, 102291. [Google Scholar] [CrossRef]
- Ahmed, S.A.; Rahman, A.A.; Elsayed, K.; Abd El-Mageed, H.R.; Mohamed, H.S.; Ahmed, S.A. Cytotoxic activity, molecular docking, pharmacokinetic properties and quantum mechanics calculations of the brown macroalga Cystoseira trinodis compounds. J. Biomol. Struct. Dyn. 2021, 39, 3855–3873. [Google Scholar] [CrossRef]
- Xia, M.; Liu, C.; Gao, L.; Lu, Y. One-Step Preparative Separation of Phytosterols from Edible Brown Seaweed Sargassum horneri by High-Speed Countercurrent Chromatography. Mar. Drugs 2019, 17, 691. [Google Scholar] [CrossRef] [Green Version]
- Bogie, J.; Hoeks, C.; Schepers, M.; Tiane, A.; Cuypers, A.; Leijten, F.; Mulder, M. Dietary Sargassum fusiforme improves memory and reduces amyloid plaque load in an Alzheimer’s disease mouse model. Sci. Rep. 2019, 9, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Milović, S.; Stanković, I.; Nikolić, D.; Radović, J.; Kolundžić, M.; Nikolić, V.; Kundaković-Vasović, T. Chemical analysis of selected seaweeds and seagrass from the Adriatic Coast of Montenegro. Chem. Biodivers. 2019, 16, e1900327. [Google Scholar] [CrossRef]
- Magura, J.; Moodley, R.; Jonnalagadda, S.B. Toxic metals (As and Pb) in Sargassum elegans Suhr (1840) and its bioactive compounds. Int. J. Environ. Health Res. 2019, 29, 266–275. [Google Scholar] [CrossRef]
- Agatonovic-Kustrin, S.; Kustrin, E.; Gegechkori, V.; Morton, D.W. High-performance thin-layer chromatography hyphenated with microchemical and biochemical derivatizations in bioactivity profiling of marine species. Mar. Drugs 2019, 17, 148. [Google Scholar] [CrossRef] [Green Version]
- Teixeira, T.R.; Santos, G.S.; Turatti, I.C.C.; Paziani, M.H.; von Zeska Kress, M.R.; Colepicolo, P.; Debonsi, H.M. Characterization of the lipid profile of Antarctic brown seaweeds and their endophytic fungi by gas chromatography–mass spectrometry (GC–MS). Polar Biol. 2019, 42, 1431–1444. [Google Scholar] [CrossRef]
- Oh, J.H.; Choi, J.S.; Nam, T.J. Fucosterol from an edible brown alga Ecklonia stolonifera prevents soluble amyloid beta-induced cognitive dysfunction in aging rats. Mar. Drugs 2018, 16, 368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ito, M.; Koba, K.; Hikihara, R.; Ishimaru, M.; Shibata, T.; Hatate, H.; Tanaka, R. Analysis of functional components and radical scavenging activity of 21 algae species collected from the Japanese coast. Food Chem. 2018, 255, 147–156. [Google Scholar] [CrossRef]
- Terasaki, M.; Kawagoe, C.; Ito, A.; Kumon, H.; Narayan, B.; Hosokawa, M.; Miyashita, K. Spatial and seasonal variations in the biofunctional lipid substances (fucoxanthin and fucosterol) of the laboratory-grown edible Japanese seaweed (Sargassum horneri Turner) cultured in the open sea. Saudi J. Biol. Sci. 2017, 24, 1475–1482. [Google Scholar] [CrossRef]
- Park, S.Y.; Hwang, E.; Shin, Y.K.; Lee, D.G.; Yang, J.E.; Park, J.H.; Yi, T.H. Immunostimulatory Effect of Enzyme-Modified Hizikia fusiformein a Mouse Model In Vitro and Ex Vivo. Mar. Biotechnol. 2017, 19, 65–75. [Google Scholar] [CrossRef]
- Kazłowska, K.; Lin, H.T.; Chang, S.H.; Tsai, G.J. In Vitro and In Vivo Anticancer Effects of Sterol Fraction from Red Algae Porphyra dentata. Evid.-Based Complementary Altern. Med. 2013, 2013, 493869. [Google Scholar] [CrossRef] [Green Version]
- Xiao, H.X.; Zhi, Q.Y.; Gong, K.L. Preparation of phytosterols and phytol from edible marine algae by microwave-assisted extraction and high-speed counter-current chromatography. Sep. Purif. Technol 2013, 104, 284–289. [Google Scholar] [CrossRef]
- Roiaini, M.; Seyed, H.M.; Jinap, S.; Norhayati, H. Effect of extraction methods on yield, oxidative value, phytosterols and antioxidant content of cocoa butter. Int. Food Res. J. 2016, 23, 47–54. [Google Scholar]
- Salkowski, F. The reaction of cholesterol with sulfuric acid. Arch. Dtsch. Ges. Physiol. 1872, 6, 207–222. [Google Scholar] [CrossRef]
- Andrade, P.B.; Barbosa, M.; Matos, R.P.; Lopes, G.; Vinholes, J.; Mouga, T.; Valentão, P. Valuable compounds in macroalgae extracts. Food Chem. 2013, 138, 1819–1828. [Google Scholar] [CrossRef]
- Kendel, M.; Wielgosz-Collin, G.; Bertrand, S.; Roussakis, C.; Bourgougnon, N.; Bedoux, G. Lipid composition, fatty acids and sterols in the seaweeds Ulva armoricana, and Solieria chordalis from Brittany (France): An analysis from nutritional, chemotaxonomic, and antiproliferative activity perspectives. Mar. Drugs 2015, 13, 5606–5628. [Google Scholar] [CrossRef]
- Bouzidi, N.; Viano, Y.; Ortalo, M.A.; Seridi, H.; Alliche, Z.; Daghbouche, Y.; Culioli, G.; Hattab, M. Sterols from the brown alga Cystoseira foeniculacea: Degradation of fucosterol into saringosterol epimers. Arab. J. Chem. 2019, 12, 1474–1478. [Google Scholar] [CrossRef] [Green Version]
- Cabral, C.E.; Klein, M.R.S.T. Phytosterols in the treatment of hypercholesterolemia and prevention of cardiovascular diseases. Arq. Cardiol. 2017, 109, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, I.; Tanaka, K.; Sugano, M.; Vahouny, G.V.; Gallo, L.L. Inhibition of cholesterol absorption in rats by plant sterols. J. Lipid Res. 1988, 29, 1573–1582. [Google Scholar] [CrossRef]
- Zhen, X.H.; Quan, Y.C.; Jiang, H.Y.; Wen, Z.S.; Qu, Y.L.; Guan, L.P. Fucosterol, a sterol extracted from Sargassum fusiforme, shows antidepressant and anticonvulsant effects. Eur. J. Pharmacol. 2015, 768, 131–138. [Google Scholar] [CrossRef]
- Lee, Y.S.; Shin, K.H.; Kim, B.K.; Lee, S. Anti-diabetic activities of fucosterol from Pelvetia siliquosa. Arch. Pharmacal Res. 2004, 27, 1120–1122. [Google Scholar]
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free radicals: Properties, sources, targets, and their implication in various diseases. Indian J. Clin. Biochem. 2015, 30, 11–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Q. Role of nrf2 in oxidative stress and toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef] [Green Version]
- Loboda, A.; Damulewicz, M.; Pyza, E.; Jozkowicz, A.; Dulak, J. Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: An evolutionarily conserved mechanism. Cell. Mol. Life Sci. 2016, 73, 3221–3247. [Google Scholar] [CrossRef] [Green Version]
- Jung, H.A.; Jin, S.E.; Ahn, B.R.; Lee, C.M.; Choi, J.S. Anti-inflammatory activity of edible brown alga Eisenia bicyclis and its constituents fucosterol and phlorotannins in LPS-stimulated RAW264. 7 macrophages. Food Chem. Toxicol. 2013, 59, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Jayawardena, T.U.; Sanjeewa, K.; Lee, H.G.; Nagahawatta, D.P.; Yang, H.W.; Kang, M.C.; Jeon, Y.J. Particulate Matter-Induced Inflammation/Oxidative Stress in Macrophages: Fucosterol from Padina boryana as a Potent Protector, Activated via NF-κB/MAPK Pathways and Nrf2/HO-1 Involvement. Mar. Drugs 2020, 18, 628. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.H.; Gan, S.Y.; Tan, S.C.; Gany, S.A.; Ying, T.; Gray, A.I.; Igoli, J.; Chan, E.W.L.; Phang, S.M. Fucosterol inhibits the cholinesterase activities and reduces the release of pro-inflammatory mediators in lipopolysaccharide and amyloid-induced microglial cells. J. Appl. Phycol. 2018, 30, 3261–3270. [Google Scholar] [CrossRef]
- Murugan, K.; Iyer, V.V. Antioxidant Activity and Gas Chromatographic-Mass Spectrometric Analysis of Extracts of the Marine Algae, Cauler papeltata and Padina gymnospora. Indian J. Pharm. Sci. 2014, 76, 548–552. [Google Scholar]
- Fernando, I.; Jayawardena, T.U.; Kim, H.S.; Vaas, A.; De Silva, H.; Nanayakkara, C.M.; Abeytunga, D.; Lee, W.; Ahn, G.; Lee, D.S.; et al. A keratinocyte and integrated fibroblast culture model for studying particulate matter-induced skin lesions and therapeutic intervention of fucosterol. Life Sci. 2019, 233, 116714. [Google Scholar] [CrossRef]
- Fernando, I.; Jayawardena, T.U.; Kim, H.S.; Lee, W.W.; Vaas, A.; De Silva, H.; Abayaweera, G.S.; Nanayakkara, C.M.; Abeytunga, D.; Lee, D.S.; et al. Beijing urban particulate matter-induced injury and inflammation in human lung epithelial cells and the protective effects of fucosterol from Sargassum binderi (Sonder ex J. Agardh). Environ. Res. 2019, 172, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Han, Y.R.; Byeon, J.S.; Choung, S.Y.; Sohn, H.S.; Jung, H.A. Protective effect of fucosterol isolated from the edible brown algae, Ecklonia stolonifera and Eisenia bicyclis, on tert-butyl hydroperoxide- and tacrine-induced HepG2 cell injury. J. Pharm. Pharmacol. 2015, 67, 1170–1178. [Google Scholar] [CrossRef]
- Jiang, H.; Li, J.; Chen, A.; Li, Y.; Xia, M.; Guo, P.; Yao, S.; Chen, S. Fucosterol exhibits selective antitumor anticancer activity against HeLa human cervical cell line by inducing mitochondrial mediated apoptosis, cell cycle migration inhibition and downregulation of m-TOR/PI3K/Akt signalling pathway. Oncol. Lett. 2018, 15, 3458–3463. [Google Scholar] [CrossRef]
- Ko, W.; Lee, H.; Kim, N.; Jo, H.G.; Woo, E.R.; Lee, K.; Han, Y.S.; Park, S.R.; Ahn, G.; Cheong, S.H.; et al. The Anti-Oxidative and Anti-Neuroinflammatory Effects of Sargassum horneri by Heme Oxygenase-1 Induction in BV2 and HT22 Cells. Antioxidants 2021, 10, 859. [Google Scholar] [CrossRef]
- Ibrahim, E.A.; Aly, H.F.; Baker, D.H.A.; Mahmoud, K.H.A.L.E.D.; El-Baz, F.K. Marine algal sterol hydrocarbon with anti-inflammatory, anticancer and anti-oxidant properties. Int. J. Pharma Bio Sci. 2016, 7, 392–398. [Google Scholar]
- Vanbrabant, K.; Van Meel, D.; Kerksiek, A.; Friedrichs, S.; Dubbeldam, M.; Schepers, M.; Zhan, N.; Gutbrod, K.; Dörmann, P.; Liu, H.B.; et al. 24(R, S)-Saringosterol—From artefact to a biological medical agent. J. Steroid Biochem. Mol. Biol. 2021, 212, 105942. [Google Scholar] [CrossRef] [PubMed]
- Santos, S.; Trindade, S.S.; Oliveira, C.; Parreira, P.; Rosa, D.; Duarte, M.F.; Ferreira, I.; Cruz, M.T.; Rego, A.M.; Abreu, M.H.; et al. Lipophilic Fraction of Cultivated Bifurcaria bifurcata, R. Ross: Detailed Composition and In Vitro Prospection of Current Challenging Bioactive Properties. Mar. Drugs 2017, 15, 340. [Google Scholar] [CrossRef] [Green Version]
- Kavita, K.; Singh, V.K.; Jha, B. 24-Branched Δ5 sterols from Laurencia papillosa red seaweed with antibacterial activity against human pathogenic bacteria. Microbiol. Res. 2014, 169, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Stabili, L.; Fraschetti, S.; Acquaviva, M.I.; Cavallo, R.A.; De Pascali, S.A.; Fanizzi, F.P.; Gerardi, C.; Narracci, M.; Rizzo, L. The Potential Exploitation of the Mediterranean Invasive Alga Caulerpa cylindracea: Can the Invasion Be Transformed into a Gain? Mar. Drugs 2016, 14, 210. [Google Scholar] [CrossRef]
- Bakar, K.; Mohamad, H.; Tan, H.S.; Latip, J. Sterols compositions, antibacterial, and antifouling properties from two Malaysian seaweeds: Dictyota dichotoma and Sargassum granuliferum. J. Appl. Pharm. Sci. 2019, 9, 47–53. [Google Scholar] [CrossRef] [Green Version]
- Rahelivao, M.P.; Gruner, M.; Andriamanantoanina, H.; Bauer, I.; Knölker, H.J. Brown Algae (Phaeophyceae) from the Coast of Madagascar: Preliminary Bioactivity Studies and Isolation of Natural Products. Nat. Prod. Bioprospecting 2015, 5, 223–235. [Google Scholar] [CrossRef] [Green Version]
- Elsayed, K.N.; Radwan, M.M.; Hassan, S.H.; Abdelhameed, M.S.; Ibraheem, I.B.; Ross, S.A. Phytochemical and biological studies on some Egyptian seaweeds. Nat. Prod. Commun. 2012, 7, 1209–1210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wächter, G.A.; Franzblau, S.G.; Montenegro, G.; Hoffmann, J.J.; Maiese, W.M.; Timmermann, B.N. Inhibition of Mycobacterium tuberculosis growth by saringosterol from Lessonia nigrescens. J. Nat. Prod. 2001, 64, 1463–1464. [Google Scholar] [CrossRef]
- Santoyo, S.; Plaza, M.; Jaime, L.; Ibañez, E.; Reglero, G.; Señorans, J. Pressurized liquids as an alternative green process to extract antiviral agents from the edible seaweed Himanthalia elongata. J. Appl. Phycol. 2011, 23, 909–917. [Google Scholar] [CrossRef] [Green Version]
- Tyśkiewicz, K.; Tyśkiewicz, R.; Konkol, M.; Rój, E.; Jaroszuk-Ściseł, J.; Skalicka-Woźniak, K. Antifungal Properties of Fucus vesiculosus, L. Supercritical Fluid Extract Against Fusarium culmorum and Fusarium oxysporum. Molecules 2019, 24, 3518. [Google Scholar] [CrossRef] [Green Version]
- Perumal, P.; Sowmiya, R.; Prasanna Kumar, S.; Ravikumar, S.; Deepak, P.; Balasubramani, G. Isolation, structural elucidation and antiplasmodial activity of fucosterol compound from brown seaweed, Sargassum linearifolium against malarial parasite Plasmodium falciparum. Nat. Prod. Res. 2018, 32, 1316–1319. [Google Scholar] [CrossRef] [PubMed]
- Budiman, A.; Rusnawan, D.W.; Yuliana, A. Antibacterial activity of Piper betle L. extract in cream dosage forms against Staphylococcus aureus and Propionibacterium acne. J. Pharm. Sci. Res. 2018, 10, 493–496. [Google Scholar]
- Geethalakshmi, R.; Sarada, V.D. In vitro and in silico antimicrobial activity of sterol and flavonoid isolated from Trianthema decandra L. Microb. Pathog. 2018, 121, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Ferrero-Miliani, L.; Nielsen, O.; Andersen, P.; Girardin, S. Chronic inflammation: Importance of NOD2 and NALP3 in interleukin-1β generation. Clin. Exp. Immunol. 2007, 147, 227–235. [Google Scholar] [CrossRef]
- Muller, W.A. Getting leukocytes to the site of inflammation. Vet. Pathol. 2013, 50, 7–22. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.M.; An, J. Cytokines, inflammation, and pain. Int. Anesthesiol. Clin. 2007, 45, 27–37. [Google Scholar] [CrossRef] [Green Version]
- Clark, M.; Kroger, C.J.; Tisch, R.M. Type 1 Diabetes: A Chronic Anti-Self-Inflammatory Response. Front. Immunol. 2017, 8, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Libby, P. Inflammatory mechanisms: The molecular basis of inflammation and disease. Nutr. Rev. 2007, 65, S140–S146. [Google Scholar] [CrossRef]
- Barbalace, M.C.; Malaguti, M.; Giusti, L.; Lucacchini, A.; Hrelia, S.; Angeloni, C. Anti-inflammatory activities of marine algae in neurodegenerative diseases. Int. J. Mol. Sci. 2019, 20, 3061. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Ni, L.; Fu, X.; Wang, L.; Duan, D.; Huang, L.; Xu, J.; Gao, X. Molecular Mechanism of Anti-Inflammatory Activities of a Novel Sulfated Galactofucan from Saccharina japonica. Mar. Drugs 2021, 19, 430. [Google Scholar] [CrossRef]
- Jin, D.Q.; Lim, C.S.; Sung, J.Y.; Choi, H.G.; Ha, I.; Han, J.S. Ulva conglobata, a marine algae, has neuroprotective and anti-inflammatory effects in murine hippocampal and microglial cells. Neurosci. Lett. 2006, 402, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.K.; Lee, B.; Kwon, M.; Yoon, N.; Shin, T.; Kim, N.G.; Choi, J.S.; Kim, H.R. Phlorofucofuroeckol B suppresses inflammatory responses by down-regulating nuclear factor κB activation via Akt, ERK, and JNK in LPS-stimulated microglial cells. Int. Immunopharmacol. 2015, 28, 1068–1075. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Chen, L.; Shen, L.; Chen, Z.; Xu, L.; Zhang, J.; Yu, X. Trans-astaxanthin attenuates lipopolysaccharide-induced neuroinflammation and depressive-like behavior in mice. Brain Res. 2016, 1649, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Besednova, N.N.; Zaporozhets, T.S.; Kuznetsova, T.A.; Makarenkova, I.D.; Kryzhanovsky, S.P.; Fedyanina, L.N.; Ermakova, S.P. Extracts and Marine Algae Polysaccharides in Therapy and Prevention of Inflammatory Diseases of the Intestine. Mar Drugs. 2020, 18, 289. [Google Scholar] [CrossRef] [PubMed]
- Vilahur, G.; Ben-Aicha, S.; Diaz-Riera, E.; Badimon, L.; Padro, T. Phytosterols and Inflammation. Curr. Med. Chem. 2019, 26, 6724–6734. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Mohamed, M.A.A.; Park, S.Y.; Yi, T.H. Fucosterol protects cobalt chloride induced inflammation by the inhibition of hypoxia-inducible factor through PI3K/Akt pathway. Int. Immunopharmacol. 2015, 29, 642–647. [Google Scholar] [CrossRef]
- Li, Y.; Li, X.; Liu, G.; Sun, R.; Wang, L.; Wang, J.; Wang, H. Fucosterol attenuates lipopolysaccharide-induced acute lung injury in mice. J. Surg. Res. 2015, 195, 515–521. [Google Scholar] [CrossRef]
- Lawrence, T. The nuclear factor NF-κB pathway in inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a001651. [Google Scholar] [CrossRef] [Green Version]
- Johnson, G.V.; Bailey, C.D. The p38 MAP kinase signaling pathway in Alzheimer’s disease. Exp. Neurol. 2003, 183, 263–268. [Google Scholar] [CrossRef]
- Hollenbach, E.; Neumann, M.; Vieth, M.; Roessner, A.; Malfertheiner, P.; Naumann, M. Inhibition of p38 MAP kinase-and RICK/NF-κB-signaling suppresses inflammatory bowel disease. FASEB J. 2004, 18, 1550–1552. [Google Scholar] [CrossRef]
- Schett, G.; Zwerina, J.; Firestein, G. The p38 mitogen-activated protein kinase (MAPK) pathway in rheumatoid arthritis. Ann. Rheum. Dis. 2008, 67, 909–916. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zarubin, T.; Jiahuai, H.A.N. Activation and signaling of the p38 MAP kinase pathway. Cell Res. 2005, 15, 11–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoo, M.S.; Shin, J.S.; Choi, H.E.; Cho, Y.W.; Bang, M.H.; Baek, N.I.; Lee, K.T. Fucosterol isolated from Undaria pinnatifida inhibits lipopolysaccharide-induced production of nitric oxide and pro-inflammatory cytokines via the inactivation of nuclear factor-κB and p38 mitogen-activated protein kinase in RAW264. 7 macrophages. Food Chem. 2012, 135, 967–975. [Google Scholar] [CrossRef]
- O’Brien, R.J.; Wong, P.C. Amyloid precursor protein processing and Alzheimer’s disease. Annu. Rev. Neurosci. 2011, 34, 185–204. [Google Scholar] [CrossRef] [Green Version]
- Kheiri, G.; Dolatshahi, M.; Rahmani, F.; Rezaei, N. Role of p38/MAPKs in Alzheimer’s disease: Implications for amyloid beta toxicity targeted therapy. Rev. Neurosci. 2019, 30, 9–30. [Google Scholar] [CrossRef] [PubMed]
- Mandrekar-Colucci, S.; Landreth, G.E. Microglia and inflammation in Alzheimer’s disease. CNS Neurol. Disord.-Drug Targets 2010, 9, 156–167. [Google Scholar] [CrossRef]
- Heneka, M.T.; Carson, M.J.; El Khoury, J.; Landreth, G.E.; Brosseron, F.; Feinstein, D.L.; Jacobs, A.H.; Wyss-Coray, T.; Vitorica, J.; Ransohoff, R.M.; et al. Neuroinflammation in Alzheimer’s disease. Lancet Neurol. 2015, 14, 388–405. [Google Scholar] [CrossRef] [Green Version]
- Jung, H.A.; Ali, M.Y.; Choi, R.J.; Jeong, H.O.; Chung, H.Y.; Choi, J.S. Kinetics and molecular docking studies of fucosterol and fucoxanthin, BACE1 inhibitors from brown algae Undaria pinnatifida and Ecklonia stolonifera. Food Chem. Toxicol. 2016, 89, 104–111. [Google Scholar] [CrossRef]
- Mandrekar, S.; Jiang, Q.; Lee, C.D.; Koenigsknecht-Talboo, J.; Holtzman, D.M.; Landreth, G.E. Microglia mediate the clearance of soluble Aβ through fluid phase macropinocytosis. J. Neurosci. 2009, 29, 4252–4262. [Google Scholar] [CrossRef] [Green Version]
- Alghazwi, M.; Smid, S.; Musgrave, I.; Zhang, W. In vitro studies of the neuroprotective activities of astaxanthin and fucoxanthin against amyloid beta (Aβ1-42) toxicity and aggregation. Neurochem. Int. 2019, 124, 215–224. [Google Scholar] [CrossRef] [Green Version]
- Jiang, L.; Zhao, X.; Xu, J.; Li, C.; Yu, Y.; Wang, W.; Zhu, L. The protective effect of dietary phytosterols on cancer risk: A systematic meta-analysis. J. Oncol. 2019, 2019, 7479518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, K.; Yuan, D.; Yan, R.; Meng, L.; Zhang, Y.; Zhu, K. Stigmasterol exhibits potent antitumor effects in human gastric cancer cells mediated via inhibition of cell migration, cell cycle arrest, mitochondrial mediated apoptosis and inhibition of JAK/STAT signaling pathway. J. BUON 2018, 23, 1420–1425. [Google Scholar] [PubMed]
- Ali, H.; Dixit, S.; Ali, D.; Alqahtani, S.M.; Alkahtani, S.; Alarifi, S. Isolation and evaluation of anticancer efficacy of stigmasterol in a mouse model of DMBA-induced skin carcinoma. Drug Des. Dev. Ther. 2015, 9, 2793. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, H.; Zhang, X.; Wang, M.; Lin, Y.; Zhou, S. Stigmasterol Simultaneously Induces Apoptosis and Protective Autophagy by Inhibiting Akt/mTOR Pathway in Gastric Cancer Cells. Front. Oncol. 2021, 11, 43. [Google Scholar] [CrossRef]
- Tanida, I.; Ueno, T.; Kominami, E. LC3 and Autophagy. In Autophagosome and Phagosome; Humana Press: Totowa, NJ, USA, 2008; pp. 77–88. [Google Scholar]
- Pattingre, S.; Espert, L.; Biard-Piechaczyk, M.; Codogno, P. Regulation of macroautophagy by mTOR and Beclin 1 complexes. Biochimie 2008, 90, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Bae, H.; Park, S.; Yang, C.; Song, G.; Lim, W. Disruption of endoplasmic reticulum and ROS production in human ovarian cancer by campesterol. Antioxidants 2021, 10, 379. [Google Scholar] [CrossRef]
- Nishida, N.; Yano, H.; Nishida, T.; Kamura, T.; Kojiro, M. Angiogenesis in cancer. Vasc. Health Risk Manag. 2016, 2, 213. [Google Scholar] [CrossRef]
- Choi, J.M.; Lee, E.O.; Lee, H.J.; Kim, K.H.; Ahn, K.S.; Shim, B.S.; Kim, S.H. Identification of campesterol from Chrysanthemum coronarium L. and its antiangiogenic activities. Phytother. Res. 2007, 21, 954–959. [Google Scholar] [CrossRef]
- Xu, Y.; Ryu, S.; Lee, Y.K.; Lee, H.J. Brassicasterol from Edible Aquacultural Hippocampus abdominalis Exerts an Anti-Cancer Effect by Dual-Targeting AKT and AR Signaling in Prostate Cancer. Biomedicines 2020, 8, 370. [Google Scholar] [CrossRef]
- Wagner, U.; Süess, K.; Luginbühl, T.; Schmid, U.; Ackermann, D.; Zellweger, T.; Sauter, G. Cyclin D1 overexpression lacks prognostic significance in superficial urinary bladder cancer. J. Pathol. 1999, 188, 44–50. [Google Scholar] [CrossRef]
- Thanan, R.; Murata, M.; Ma, N.; Hammam, O.; Wishahi, M.; El Leithy, T.; Kawanishi, S. Nuclear localization of COX-2 in relation to the expression of stemness markers in urinary bladder cancer. Mediat. Inflamm. 2012, 2012, 165879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ikarashi, N.; Ono, T.; Hoshino, M.; Toda, T.; Yazawa, Y.; Sugiyama, K. Mechanistic Analysis of Bladder Cancer Suppression by Brassicasterol in a Rat Model of N-butyl-N-(4-Hydroxybutyl) Nitrosamine-Induced Bladder Cancer. In Traditional & Kampo Medicine; Wiley Online Library: Hoboken, NJ, USA, 2021. [Google Scholar]
- Baskar, A.A.; Ignacimuthu, S.; Paulraj, G.M.; Al Numair, K.S. Chemopreventive potential of β-sitosterol in experimental colon cancer model-an in vitro and in vivo study. BMC Complementary Altern. Med. 2010, 10, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vundru, S.S.; Kale, R.K.; Singh, R.P. β-Sitosterol induces G1 arrest and causes depolarization of mitochondrial membrane potential in breast carcinoma MDA-MB-231 cells. BMC Complementary Altern. Med. 2013, 13, 280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajavel, T.; Mohankumar, R.; Archunan, G.; Ruckmani, K.; Devi, K.P. Beta sitosterol and Daucosterol (phytosterols identified in Grewia tiliaefolia) perturbs cell cycle and induces apoptotic cell death in A549 cells. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.B.; Han, L.B.; Wang, H.Y.; Zhang, J.; Sun, S.T.; Feng, D.Q.; Xia, G.X. The thioredoxin GbNRX1 plays a crucial role in homeostasis of apoplastic reactive oxygen species in response to Verticillium dahliae infection in cotton. Plant Physiol. 2016, 170, 2392–2406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajavel, T.; Packiyaraj, P.; Suryanarayanan, V.; Singh, S.K.; Ruckmani, K.; Devi, K.P. β-Sitosterol targets Trx/Trx1 reductase to induce apoptosis in A549 cells via ROS mediated mitochondrial dysregulation and p53 activation. Sci. Rep. 2018, 8, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Mofeed, J.; Deyab, M.; Sabry, A.E.N.; Ward, F. In Vitro Anticancer Activity of Five Marine Seaweeds Extract from Egypt Against Human Breast and Colon Cancer Cell Lines. Res. Sq. 2021. [Google Scholar] [CrossRef]
- Pan, T.J.; Li, L.X.; Zhang, J.W.; Yang, Z.S.; Shi, D.M.; Yang, Y.K.; Wu, W.Z. Antimetastatic Effect of Fucoidan-Sargassum against Liver Cancer Cell Invadopodia Formation via Targeting Integrin αVβ3 and Mediating αVβ3/Src/E2F1 Signaling. J. Cancer 2019, 10, 4777–4792. [Google Scholar] [CrossRef]
- Alghazeer, R.; Howell, N.K.; El-Naili, M.B.; Awayn, N. Anticancer and Antioxidant Activities of Some Algae from Western Libyan Coast. Nat. Sci. 2008, 10, 232–246. [Google Scholar] [CrossRef] [Green Version]
- Henry, M.M.; Wilson, M.N.; Martin, O.O.; Ntevheleni, T.; Wilfred, T.M. Phlorotannins and a sterol isolated from a brown alga Ecklonia maxima, and their cytotoxic activity against selected cancer cell lines HeLa, H157 and MCF7. Interdiscip. J. Chem. 2017, 2, 1–6. [Google Scholar]
- Isnansetyo, A.; Lutfia, F.N.L.; Nursid, M.; Trijoko Susidarti, R.A. Cytotoxicity of Fucoidan from Three Tropical Brown Algae Against Breast and Colon Cancer Cell Lines. Pharmacogn. J. 2017, 9, 14–20. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.M.; Liu, P.Y.; Chen, Y.A.; Tseng, H.Y.; Shen, P.C.; Hwang, P.A.; Hsu, H.L. Oligo-Fucoidan prevents IL-6 and CCL2 production and cooperates with p53 to suppress ATM signaling and tumor progression. Sci. Rep. 2017, 7, 11864. [Google Scholar] [CrossRef] [Green Version]
- Arsianti, A.; Fadilah, F.; Wibisino, L.K.; Kusmardi, S.; Putrianingsih, R.I.S.T.A.; Murniasih, T.U.T.I.K.; Rasyid, A.; Pangestuti, R. Phytochemical composition and anticancer activity of seaweeds Ulva lactuca and Eucheuma cottonii against breast MCF-7 and colon HCT-116 cells. Asian J. Pharm. Clin. Res. 2016, 11, 115–119. [Google Scholar] [CrossRef]
- Vizetto-Duarte, C.; Custódio, L.; Gangadhar, K.N.; Lago, J.H.G.; Dias, C.; Matos, A.M.; Neng, N.; Nogueira, J.M.F.; Barreira, L.; Albericio, F.; et al. Isololiolide, a carotenoid metabolite isolated from the brown alga Cystoseira tamariscifolia, is cytotoxic and able to induce apoptosis in hepatocarcinoma cells through caspase-3 activation, decreased Bcl-2 levels, increased p53 expression and PARP cleavage. Phytomedicine 2016, 23, 550–557. [Google Scholar] [PubMed]
- Sun, J.; Sun, J.; Song, B.; Zhang, L.; Shao, Q.; Liu, Y.; Yuan, D.; Zhang, Y.; Qu, X. Fucoidan inhibits CCL22 production through NF-κB pathway in M2 macrophages: A potential therapeutic strategy for cancer. Sci. Rep. 2016, 6, 35855. [Google Scholar] [CrossRef]
- Kim, R.K.; Uddin, N.; Hyun, J.W.; Kim, C.; Suh, Y.; Lee, S.J. Novel anticancer activity of phloroglucinol against breast cancer stem-like cells. Toxicol. Appl. Pharmacol. 2015, 286, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Wang, P.; Wang, H.; Li, Q.; Teng, H.; Liu, Z.; Yang, W.; Hou, L.; Zou, X. Fucoidan Derived from Undaria pinnatifida Induces Apoptosis in Human Hepatocellular Carcinoma SMMC-7721 Cells via the ROS-Mediated Mitochondrial Pathway. Mar. Drugs 2013, 11, 1961–1976. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boo, H.J.; Hong, J.Y.; Kim, S.C.; Kang, J.I.; Kim, M.K.; Kim, E.J.; Hyun, J.W.; Koh, Y.S.; Yoo, E.S.; Kwon, J.M.; et al. The anticancer effect of fucoidan in PC-3 prostate cancer cells. Mar. Drugs 2013, 11, 2982–2999. [Google Scholar] [CrossRef] [Green Version]
- Park, H.K.; Kim, J.; Nam, T.J. Induction of apoptosis and regulation of ErbB signaling by laminarin in HT-29 human colon cancer cells. Int. J. Mol. Med. 2013, 32, 291–295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, H.K.; Kim, I.H.; Kim, J.; Nam, T.J. Induction of apoptosis by laminarin, regulating the insulin-like growth factor-IR signaling pathways in HT-29 human colon cells. Int. J. Mol. Med. 2012, 30, 734–738. [Google Scholar] [CrossRef] [Green Version]
- Namvar, F.; Mahamad, R.; Baharara, J.; Balnejah, S.Z.; Fargahi, F.; Rahman, H.S. Antioxidant, antiproliferative, and antiangiogenesis effects of polyphenol-rich seaweed (Sargassum muticum). Biomed Res. Int. 2013, 2013, 604787. [Google Scholar] [CrossRef] [Green Version]
- Mary, J.S.; Vinotha, P.; Pradeep, A.M. Screening for in vitro cytotoxic activity of seaweed, Sargassum sp. against Hep-2 and MCF-7 cancer cell lines. Asian Pac. J. Cancer Prev. 2012, 13, 6073–6076. [Google Scholar] [CrossRef]
- Boo, H.J.; Hyun, J.H.; Kim, S.C.; Kang, J.I.; Kim, M.K.; Kim, S.Y.; Cho, H.; Yoo, E.S.; Kang, H.K. Fucoidan from Undaria pinnatifida induces apoptosis in A549 human lung carcinoma cells. Phytother. Res. 2011, 25, 1082–1086. [Google Scholar] [CrossRef] [PubMed]
- Zandi, K.; Ahmadzadeh, S.; Tajbakhsh, S.; Rastian, Z.; Yousefi, F.; Farshadpour, F.; Sartavi, K. Anticancer activity of Sargassum oligocystum water extract against human cancer cell lines. Eur. Rev. Med Pharmacol. Sci. 2010, 14, 669–673. [Google Scholar] [PubMed]
- Maruyama, H.; Tamauchi, H.; Iizuka, M.; Nakano, T. The role of NK cells in antitumor activity of dietary fucoidan from Undaria pinnatifidasporophylls (Mekabu). Planta Med. 2006, 72, 1415–1417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harada, H.; Yamashita, U.; Kurihara, H.; Fukushi, E.; Kawabata, J.; Kamei, Y. Antitumor activity of palmitic acid found as a selective cytotoxic substance in a marine red alga. Anticancer Res. 2020, 22, 2587–2590. [Google Scholar]
- Baker, V.A.; Hepburn, P.A.; Kennedy, S.J.; Jones, P.A.; Lea, L.J.; Sumpter, J.P.; Ashby, J. Safety evaluation of phytosterol esters. Part 1. Assessment of oestrogenicity using a combination of in vivo and in vitro assays. Food Chem. Toxicol. 1999, 37, 13–22. [Google Scholar] [CrossRef]
- Weststrate, J.A.; Ayesh, R.; Bauer-Plank, C.; Drewitt, P.N. Safety evaluation of phytosterol esters. Part 4. Faecal concentrations of bile acids and neutral sterols in healthy normolipidaemic volunteers consuming a controlled diet either with or without a phytosterol ester-enriched margarine. Food Chem. Toxicol. 1999, 37, 1063–1071. [Google Scholar] [CrossRef]
- Wolfreys, A.M.; Hepburn, P.A. Safety evaluation of phytosterol esters. Part 7. Assessment of mutagenic activity of phytosterols, phytosterol esters and the cholesterol derivative, 4-cholesten-3-one. Food Chem. Toxicol. 2002, 40, 461–470. [Google Scholar] [CrossRef]
- St-Onge, M.P.; Jones, P.J. Phytosterols and human lipid metabolism: Efficacy, safety, and novel foods. Lipids 2003, 38, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Ostlund, R.E., Jr. Phytosterols in human nutrition. Annu. Rev. Nutr. 2002, 22, 533–549. [Google Scholar] [CrossRef] [PubMed]
- Saikia, S.; Mahnot, N.K.; Sahu, R.K.; Kalita, J. Edible Seaweeds as Potential Source of Nutraceuticals. In Marine Niche: Applications in Pharmaceutical Sciences; Springer: Singapore, 2020; pp. 183–201. [Google Scholar]
- Lu, B.Y.; Zhao, Y.J. Photooxidation of phytochemicals in food and control: A review. Ann. N. Y. Acad. Sci. 2017, 1398, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Belwal, T.; Li, L.; Limwachiranon, J.; Liu, X.; Luo, Z. Phytosterols and their derivatives: Potential health-promoting uses against lipid metabolism and associated diseases, mechanism, and safety issues. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1243–1267. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, Y.; Kenny, O.; O’Connell, N.M.; Maguire, A.R.; McCarthy, F.O.; O’Brien, N.M. Synthesis and assessment of the relative toxicity of the oxidised derivatives of campesterol and dihydrobrassicasterol in U937 and HepG2 cells. Biochimie 2013, 95, 496–503. [Google Scholar] [CrossRef]

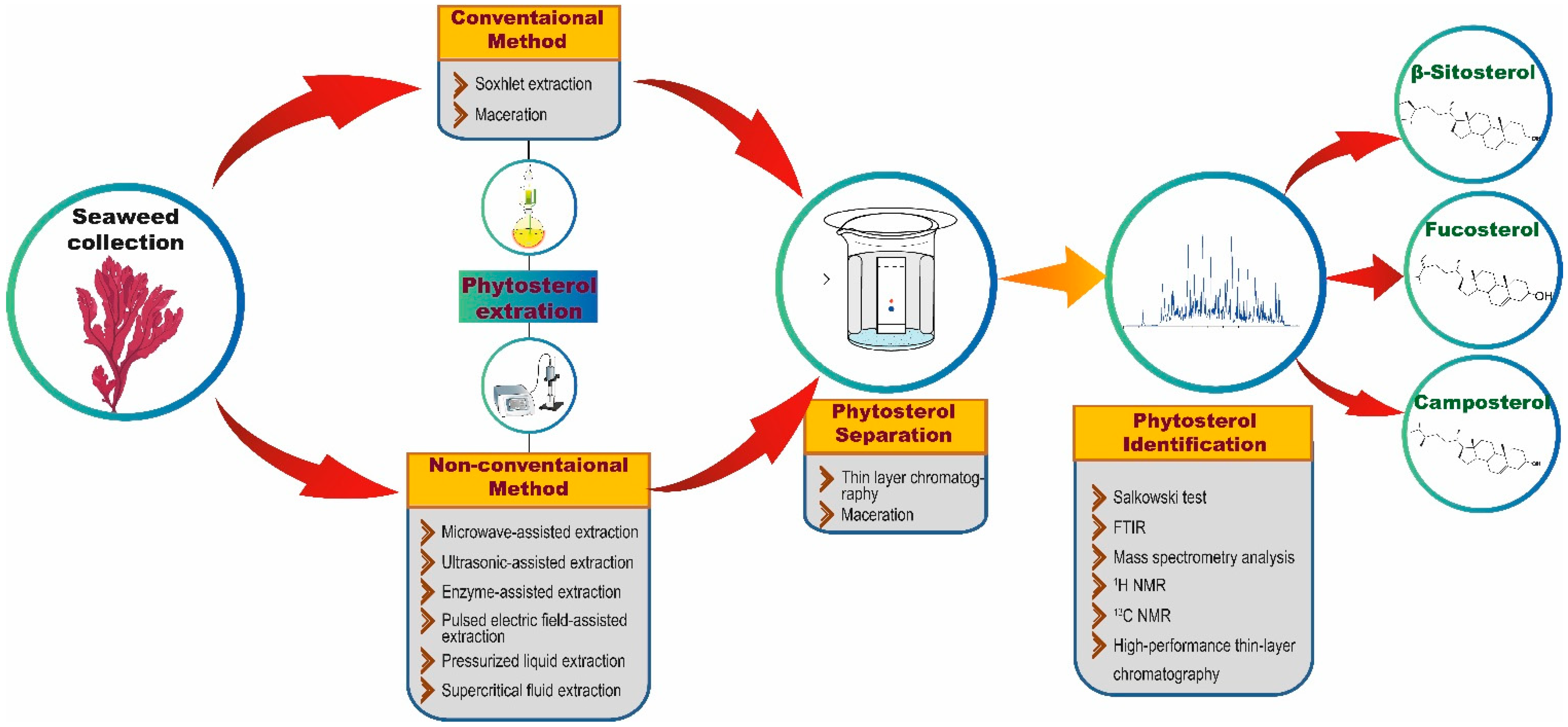


| Source | Extraction Method | Methods of Analysis | Identified Phytosterols | References |
|---|---|---|---|---|
| Gelidium spinosum | Soxhlet method | FTIR and GC–MS | Stigmasterol | [32] |
| Saccharina latissima | saponified extract | GC–MS | cholesterol, desmosterol, 24-methylenecholesterol, fucosterol, cycloartenol, and unknown ∆5-sterol | [35] |
| Palmaria decipiens, Plocamium cartilagineum, Iridaea cordata, and Pyropia endiviifolia | alkaline hydrolysis | GC–MS | cholesterol, brassicasterol, campesterol, stigmasterol, β-sitosterol, and fucosterol | [33] |
| Ecklonia radiata | alkaline saponification | LC-MS/MS and GC–MS | fucosterol, Sitostanol, 24α-methyl cholesterol, and 24α-ethyl cholesterol | [36] |
| Padina australis and Stoechospermum marginatum, and Ahnfeltiopsis pygmaea | acid and alkaline hydrolysis followed by solvent extraction, derivatization, and GC determination | gas chromatography coupled with a flame ionization detection system (GC–FID) | sitostanol, campestanol ergosterol, campesterol, delta-5-avenasterol, stigmasterol, sistenol, cholesterol, and 24-methylenecholesterol | [37] |
| Adenocystis utricularis, Desmarestia confervoides, Curdiea racovitzae, Myriogramme manginii, and Ulva intestinalis | Soxhlet method | GC–MS and FT-IR | fucosterol, cholesterol, and hydroxymethylcholesterol | [38] |
| Phaeophyta (Cystoseira barbata, Cystoseira compressa, Fucus virsoides) and chlorophyta (Codium bursa) | agitation-assisted extraction and pressurized liquid extraction | TLC | cholesterol, brassicasterol, campesterol, campestanol, stigmasterol, β-sitosterol, fucosterol, and isofucosterol | [39] |
| Cystoseira trinodis | solvent extraction and column chromatography | 1H, 13C NMR, heteronuclear multiple-bond correlation (HMBC), heteronuclear single-quantum coherence (HSQC), GC–MS, and electron ionization-mass spectra (EI-MS) | saringosterol, β-sitosterol | [40] |
| Sargassum horneri | high-speed countercurrent chromatography | NMR | fucosterol and saringosterol | [41] |
| S. fusiforme | Folch method | GC–MS | 24(S)-Saringosterol | [42] |
| Halimeda tuna, Codium bursa, and Cystoseira barbata | solvent extraction | GC and GC–MS | fucosterol, campesterol and β-sitosterol | [43] |
| Sargassum elegans | solvent extraction and column chromatography | NMR (1H and 13C), IR and mass spectral data | β-sitosterol, fucosterol | [44] |
| Phaeophyta (Cystophora pectinata, Pyllospora comoasa, Scytothalia dorycarpa, Carpoglossum confluens, E. radiata, Sargassum lacerifolium, Perithalia caudata, Codium harveyi, Scytothalia dorycarpa, Hypnea valida, Cystophora monilifera, Hormosira banksia, Myriodesma integrifolium, Epiphytic algae sp., Cystophora subfarcinata), Rhodophyta (Austrophyllis harveyana, Rhodophyllis membaneacea), and chlorophyta (Codium fragile) | maceration | post-chromatographic derivatization and HPTLC | β-sitosterol | [45] |
| Ascoseira mirabilis, A. utricularis, Desmarestia anceps, and Phaeurus antarcticus | saponification | GC–MS | cholesterol, desmosterol, brassicasterol, campesterol, stigmasterol, fucosterol, and β-sitosterol | [46] |
| Ecklonia stolonifera | silica gel column chromatography | 1H and 13C NMR | fucosterol | [47] |
| Rhodophyta (Gracilaria vermiculophylla, Pterocladiella tenuis, Palisada intermedia, Chrysymenia wrightii, Gracilaria elegans, Grateloupia asiatica, Laurencia okamurae) and Phaeophyta (Eckloniopsi radicosa, Sargassum thunbergia, Ecklonia kurome, Eisenia arborea, Sargassum piluliferum, S. fusiforme, U. pinnatifida, Ecklonia cava) | saponification | HPLC with fluorescence detection | cholesterol, β-sitosterol, ergosterol, stigmasterol, and fucosterol | [48] |
| S. horneri | total lipid extraction using methanol | RP-HPLC | fucosterol | [49] |
| Hizikia fusiformein | ethanol extraction and chromatographic separation | LC/ electrospray ionization (ESI)-MS | fucosterol | [50] |
| A. utricularis, Ascoseira mirabilis, Cystosphaera jacquinotii, D. anceps, Durvillaea antarctica, and Himantothallus grandifolius | ultrasound irradiation | LC-MS/MS | ergosterol, brassicasterol, fucosterol, β-sitosterol, campesterol, cholesterol, and stigmasterol) | [6] |
| Porphyra dentata | methanol extraction and silica gel column chromatography | HPLC- evaporative light scattering detector (ELSD) | cholesterol, β -sitosterol, and campesterol | [51] |
| Seaweed Names | Cell Lines Used | Therapeutic Compounds | Anticancer Activity | References |
|---|---|---|---|---|
| Ulva fasciata, Ulva lactuca, Amphiroa anceps, Corallina mediterranea, and Sargassum filipendula | human breast adenocarcinoma cell line (MCF-7) and colorectal carcinoma cell line (HCT-116) | palmitic acid, oleic acid, retinoic acid, dihydroactinidiolide, thiosemicarbazide, diisobutyl phthalate, and phytol | anticancer agents against human breast and colon cancers | [134] |
| Sargassum spp. | SMMC-7721, Huh7, and HCCLM3 liver | fucoidan | deactivates the integrin αVβ3/SRC/E2F1 signaling pathway; Antimetastatic | [135] |
| Ulva lactuca, Codium tomentosum, Cystoseira crinita, Cystoseira stricta, Sargassum vulgare, Gelidium latifolium, Hypnea musciformis and Jania rubens | human colorectal carcinoma (Caco2) and human corneal epithelial cells (HCEC) | polyphenols and flavonoids | human colorectal carcinoma | [136] |
| Ecklonia maxima | HeLa, H157 and MCF7 cancer cell lines | phlorotannins and sterol | cytotoxic activity | [137] |
| Sargassum spp., Turbinaria spp. and Padina spp. | breast cancer (MCF-7) and colon cancer cells (WiDr) | fucoidan | showed potential selective cytotoxicity | [138] |
| Sargassum hemiphyllum | HCT116 | oligo-fucoidan | DNA damage; cell cycle checkpoint; prevents HCT116 tumorigenicity and regulate the cancer cell death | [139] |
| Ulva lactuca and Eucheuma cottonii | breast MCF-7 and colorectal HCT-116 cancer cells | steroids, glycosides, flavonoids, and tannins | anti-breast and anticolorectal cancer agents | [140] |
| Carpodesmia tamariscifolia | hepatocellular carcinoma Hep G2, AGS and HCT-15 cell lines | isololiolide | caspase-3 activation, decreased Bcl-2 levels, increased p53 expression and PARP cleavage | [141] |
| Fucus vesiculosus | human hepatoma cell line MHCC-97H | fucoidan | macrophages M2 anti-inflammatory reduction; inhibition of tumor cell migration | [142] |
| Brown algae spp. | MCF-7 cell line | phloroglucinol | decreased CD44+ cancer cell population, expression of CSC regulators such as Sox2, CD44, Oct4, Notch2, and β-catenin; inhibited KRAS and its downstream PI3K/AKT and RAF-1/ERK signaling pathway | [143] |
| U. pinnatifida | human hepatocellular carcinoma SMMC-7721 cells | fucoidan | apoptosis via the ROS mediated mitochondrial pathway | [144] |
| U. pinnatifida | PC-3 human prostate cancer cells | fucoidan | induced intrinsic and extrinsic apoptosis pathways | [145] |
| Laminaria digitata | HT-29 colon cancer cells | laminarin | induction of apoptosis; affected insulin-like growth factor (IGF-IR); decreased MAPK and ERK phosphorylation; decreased IGF-IR-dependent proliferation | [146,147] |
| Sargassum muticum | MCF-7 cells | Sargassum muticum methanol extract (SMME) | induced apoptosis; showed antiangiogenic activity in the chorioallantoic membrane (CAM) assay; antioxidant effects | [148] |
| Porphyra dentata | 4T1 cancer cells | cholesterol, β-sitosterol, and campesterol | induced apoptosis; decreased the ROS and arginase activity of MDSCs in tumor-bearing mice | [51] |
| Sargassum spp. | MCF-7 (breast cancer) and Hep-2 (liver cancer) cell line | ethanol extract | induced cell shrinkage, cell membrane blebbing and formation of apoptotic bodies | [149] |
| U. pinnatifida | A549 human lung carcinoma cells | fucoidan | induced apoptosis through down-regulation of p38, PI3K/Akt, and the activation of the ERK1/2 MAPK pathway | [150] |
| Sargassum oligocystum | K562 and Daudi human cancer cell lines | fucoidans | antitumor activity | [151] |
| U. pinnatifidasporophylls | leukemia A20 cells | fucoidan | T-cell mediated and NK cell response; tumor destruction by immune cells | [152] |
| Amphiroa zonata | human leukemic cells | palmitic acid | showed selective cytotoxicity | [153] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sohn, S.-I.; Rathinapriya, P.; Balaji, S.; Jaya Balan, D.; Swetha, T.K.; Durgadevi, R.; Alagulakshmi, S.; Singaraj, P.; Pandian, S. Phytosterols in Seaweeds: An Overview on Biosynthesis to Biomedical Applications. Int. J. Mol. Sci. 2021, 22, 12691. https://doi.org/10.3390/ijms222312691
Sohn S-I, Rathinapriya P, Balaji S, Jaya Balan D, Swetha TK, Durgadevi R, Alagulakshmi S, Singaraj P, Pandian S. Phytosterols in Seaweeds: An Overview on Biosynthesis to Biomedical Applications. International Journal of Molecular Sciences. 2021; 22(23):12691. https://doi.org/10.3390/ijms222312691
Chicago/Turabian StyleSohn, Soo-In, Periyasamy Rathinapriya, Sekaran Balaji, Devasahayam Jaya Balan, Thirukannamangai Krishnan Swetha, Ravindran Durgadevi, Selvaraj Alagulakshmi, Patchiappan Singaraj, and Subramani Pandian. 2021. "Phytosterols in Seaweeds: An Overview on Biosynthesis to Biomedical Applications" International Journal of Molecular Sciences 22, no. 23: 12691. https://doi.org/10.3390/ijms222312691








