Docosahexaenoic Acid Modulates Paracellular Absorption of Testosterone and Claudin-1 Expression in a Tissue-Engineered Skin Model
Abstract
:1. Introduction
2. Results
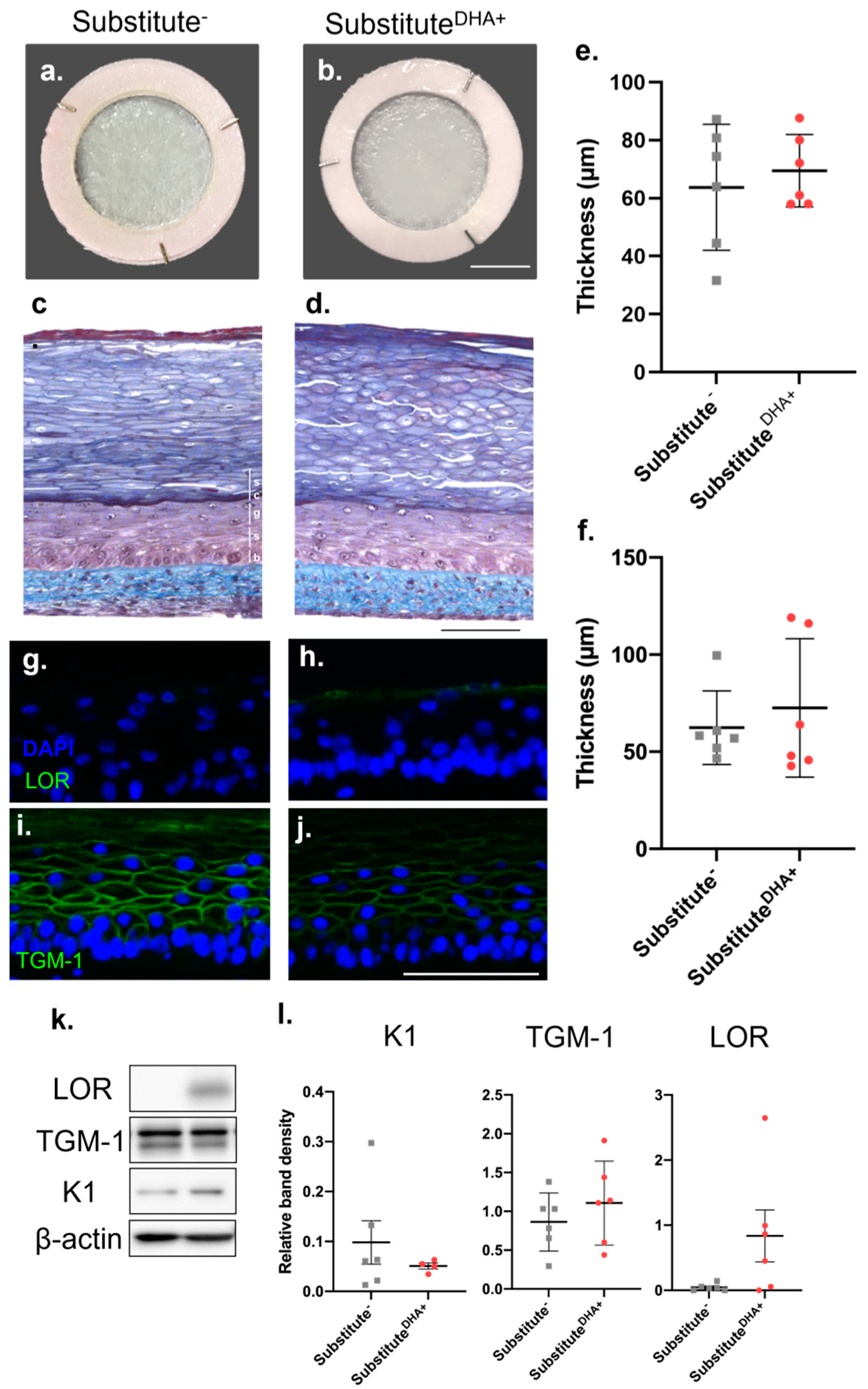
2.1. Impact of DHA on the Morphology of the Skin Substitutes
2.2. Impact of DHA on the Permeability of the Skin Substitutes
2.3. Impact of DHA on the Fatty Acid Phospholipid Profile of the Skin Substitutes
2.4. Impact of DHA on Epidermal Expression of TJ Proteins
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Production of Tissue-Engineered Skin Substitutes
4.3. Fatty Acid Supplementation of Cell Culture Media
4.4. Histological Analysis
4.5. Percutaneous Absorption
4.6. Gas Chromatography
4.7. Western Blot Analysis
4.8. Immunofluorescence Staining
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Madison, K.C. Barrier Function of the Skin: “La Raison d’Être” of the Epidermis. J. Investig. Dermatol. 2003, 121, 231–241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feingold, K.R. The outer frontier: The importance of lipid metabolism in the skin. J. Lipid. Res. 2009, 50, S417–S422. [Google Scholar] [CrossRef] [Green Version]
- Candi, E.; Schmidt, R.; Melino, G. The cornified envelope: A model of cell death in the skin. Nat. Rev. Mol. Cell Biol. 2005, 6, 328–340. [Google Scholar] [CrossRef] [PubMed]
- van Smeden, J.; Bouwstra, J.A. Stratum Corneum Lipids: Their Role for the Skin Barrier Function in Healthy Subjects and Atopic Dermatitis Patients. Curr. Probl. Dermatol. 2016, 49, 8–26. [Google Scholar] [CrossRef] [PubMed]
- Furuse, M.; Hata, M.; Furuse, K.; Yoshida, Y.; Haratake, A.; Sugitani, Y.; Noda, T.; Kubo, A.; Tsukita, S. Claudin-based tight junctions are crucial for the mammalian epidermal barrier: A lesson from claudin-1-deficient mice. J. Cell Biol. 2002, 156, 1099–1111. [Google Scholar] [CrossRef] [PubMed]
- Brandner, J.M.; Kief, S.; Grund, C.; Rendl, M.; Houdek, P.; Kuhn, C.; Tschachler, E.; Franke, W.W.; Moll, I. Organization and formation of the tight junction system in human epidermis and cultured keratinocytes. Eur. J. Cell Biol. 2002, 81, 253–263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuki, T.; Haratake, A.; Koishikawa, H.; Morita, K.; Miyachi, Y.; Inoue, S. Tight junction proteins in keratinocytes: Localization and contribution to barrier function. Exp. Dermatol. 2007, 16, 324–330. [Google Scholar] [CrossRef]
- Gruber, R.; Börnchen, C.; Rose, K.; Daubmann, A.; Volksdorf, T.; Wladykowski, E.; Vidal-y-Sy, S.; Peters, E.M.; Danso, M.; Bouwstra, J.A.; et al. Diverse Regulation of Claudin-1 and Claudin-4 in Atopic Dermatitis. Am. J. Pathol. 2015, 185, 2777–2789. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, S.; von Buenau, B.; Vidal-y-Sy, S.; Haftek, M.; Wladykowski, E.; Houdek, P.; Lezius, S.; Duplan, H.; Bäsler, K.; Dähnhardt-Pfeiffer, S.; et al. Claudin-1 decrease impacts epidermal barrier function in atopic dermatitis lesions dose-dependently. Sci. Rep. 2020, 10, 2024. [Google Scholar] [CrossRef]
- Saito, A.C.; Higashi, T.; Fukazawa, Y.; Otani, T.; Tauchi, M.; Higashi, A.Y.; Furuse, M.; Chiba, H. Occludin and tricellulin facilitate formation of anastomosing tight-junction strand network to improve barrier function. Mol. Biol. Cell 2021, 32, 722–738. [Google Scholar] [CrossRef]
- Pummi, K.; Malminen, M.; Aho, H.; Karvonen, S.L.; Peltonen, J.; Peltonen, S. Epidermal tight junctions: ZO-1 and occludin are expressed in mature, developing, and affected skin and in vitro differentiating keratinocytes. J. Investig. Dermatol. 2001, 117, 1050–1058. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hartsock, A.; Nelson, W.J. Adherens and tight junctions: Structure, function and connections to the actin cytoskeleton. Biochim. Biophys. Acta 2008, 1778, 660–669. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kruse, V.; Neess, D.; Færgeman, N.J. The Significance of Epidermal Lipid Metabolism in Whole-Body Physiology. Trends Endocrinol. Metab. 2017, 28, 669–683. [Google Scholar] [CrossRef] [PubMed]
- Ziboh, V.A.; Miller, C.C.; Cho, Y. Metabolism of polyunsaturated fatty acids by skin epidermal enzymes: Generation of antiinflammatory and antiproliferative metabolites. Am. J. Clin. Nutr. 2000, 71, 361s–366s. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jia, T.; Qiao, W.; Yao, Q.; Wu, W.; Kaku, K. Treatment with Docosahexaenoic Acid Improves Epidermal Keratinocyte Differentiation and Ameliorates Inflammation in Human Keratinocytes and Reconstructed Human Epidermis Models. Molecules 2019, 24, 3156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kendall, A.C.; Pilkington, S.M.; Massey, K.A.; Sassano, G.; Rhodes, L.E.; Nicolaou, A. Distribution of Bioactive Lipid Mediators in Human Skin. J. Investig. Dermatol. 2015, 135, 1510–1520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burr, G.O.; Burr, M.M. Nutrition classics from The Journal of Biological Chemistry 82:345-67, 1929. A new deficiency disease produced by the rigid exclusion of fat from the diet. Nutr. Rev. 1973, 31, 248–249. [Google Scholar] [CrossRef] [PubMed]
- Simard, M.; Julien, P.; Fradette, J.; Pouliot, R. Modulation of the Lipid Profile of Reconstructed Skin Substitutes after Essential Fatty Acid Supplementation Affects Testosterone Permeability. Cells 2019, 8, 1142. [Google Scholar] [CrossRef] [Green Version]
- Tanojo, H.; Bouwstra, J.A.; Junginger, H.E.; Boddé, H.E. In vitro human skin barrier modulation by fatty acids: Skin permeation and thermal analysis studies. Pharm. Res. 1997, 14, 42–49. [Google Scholar] [CrossRef]
- Willemsen, L.E.; Koetsier, M.A.; Balvers, M.; Beermann, C.; Stahl, B.; van Tol, E.A. Polyunsaturated fatty acids support epithelial barrier integrity and reduce IL-4 mediated permeability in vitro. Eur. J. Nutr. 2008, 47, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Popa, I. The concept of sphingolipid rheostat in skin: A driving force for new active ingredients in cosmetic applications. OCL 2018, 25, D507. [Google Scholar] [CrossRef]
- Clark, C.C.T.; Taghizadeh, M.; Nahavandi, M.; Jafarnejad, S. Efficacy of ω-3 supplementation in patients with psoriasis: A meta-analysis of randomized controlled trials. Clin. Rheumatol. 2019, 38, 977–988. [Google Scholar] [CrossRef] [PubMed]
- Morin, A.; Simard, M.; Rioux, G.; Grenier, A.; Morin, S.; Pouliot, R. Application of an In Vitro Psoriatic Skin Model to Study Cutaneous Metabolization of Tazarotene. Processes 2019, 7, 871. [Google Scholar] [CrossRef] [Green Version]
- Bélanger, A.; Grenier, A.; Simard, F.; Gendreau, I.; Pichette, A.; Legault, J.; Pouliot, R. Dihydrochalcone Derivatives from Populus balsamifera L. Buds for the Treatment of Psoriasis. Int. J. Mol. Sci. 2020, 21, 256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grenier, A.; Legault, J.; Pichette, A.; Jean, L.; Bélanger, A.; Pouliot, R. Antioxidant, Anti-Inflammatory, and Anti-Aging Potential of a Kalmia angustifolia Extract and Identification of Some Major Compounds. Antioxidants 2021, 10, 1373. [Google Scholar] [CrossRef] [PubMed]
- Grenier, A.; Gendreau, I.; Pouliot, R. Optimization of the Self-Assembly Method for the Production of Psoriatic Skin Substitutes; IntechOpen: London, UK, 2019. [Google Scholar]
- Jean, J.; Lapointe, M.; Soucy, J.; Pouliot, R. Development of an in vitro psoriatic skin model by tissue engineering. J. Dermatol. Sci. 2009, 53, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Larouche, D.; Paquet, C.; Fradette, J.; Carrier, P.; Auger, F.A.; Germain, L. Regeneration of Skin and Cornea by Tissue Engineering. In Stem Cells in Regenerative Medicine; Audet, J., Stanford, W.L., Eds.; Humana Press: Totowa, NJ, USA, 2009; pp. 233–256. [Google Scholar]
- Balic, A.; Vlasic, D.; Zuzul, K.; Marinovic, B.; Bukvic Mokos, Z. Omega-3 Versus Omega-6 Polyunsaturated Fatty Acids in the Prevention and Treatment of Inflammatory Skin Diseases. Int. J. Mol. Sci. 2020, 21, 741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niehues, H.; Bouwstra, J.A.; El Ghalbzouri, A.; Brandner, J.M.; Zeeuwen, P.; van den Bogaard, E.H. 3D skin models for 3R research: The potential of 3D reconstructed skin models to study skin barrier function. Exp. Dermatol. 2018, 27, 501–511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- OCDE. Test No. 428: Skin Absorption: In Vitro Method; Éditions OCDE: Paris, France, 2004. [Google Scholar]
- Flaten, G.E.; Palac, Z.; Engesland, A.; Filipović-Grčić, J.; Vanić, Ž.; Škalko-Basnet, N. In vitro skin models as a tool in optimization of drug formulation. Eur. J. Pharm. Sci. 2015, 75, 10–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lorthois, I.; Simard, M.; Morin, S.; Pouliot, R. Infiltration of T Cells into a Three-Dimensional Psoriatic Skin Model Mimics Pathological Key Features. Int. J. Mol. Sci. 2019, 20, 1670. [Google Scholar] [CrossRef] [Green Version]
- Batheja, P.; Song, Y.; Wertz, P.; Michniak-Kohn, B. Effects of growth conditions on the barrier properties of a human skin equivalent. Pharm. Res. 2009, 26, 1689–1700. [Google Scholar] [CrossRef] [PubMed]
- Carriel, V.; Garzón, I.; Jiménez, J.M.; Oliveira, A.C.; Arias-Santiago, S.; Campos, A.; Sánchez-Quevedo, M.C.; Alaminos, M. Epithelial and stromal developmental patterns in a novel substitute of the human skin generated with fibrin-agarose biomaterials. Cells Tissues Organs 2012, 196, 682. [Google Scholar] [CrossRef] [PubMed]
- Mieremet, A.; García, V.A.; Boiten, W.; van Dijk, R.; Gooris, G.; Bouwstra, J.A.; El Ghalbzouri, A. Human skin equivalents cultured under hypoxia display enhanced epidermal morphogenesis and lipid barrier formation. Sci. Rep. 2019, 9, 7811. [Google Scholar] [CrossRef]
- Helder, R.W.J.; Rousel, J.; Boiten, W.A.; Gooris, G.S.; Nadaban, A.; El Ghalbzouri, A.; Bouwstra, J.A. Improved organotypic skin model with reduced quantity of monounsaturated ceramides by inhibiting stearoyl-CoA desaturase-1. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2021, 1866, 158885. [Google Scholar] [CrossRef]
- Thakoersing, V.S.; van Smeden, J.; Mulder, A.A.; Vreeken, R.J.; El Ghalbzouri, A.; Bouwstra, J.A. Increased presence of monounsaturated fatty acids in the stratum corneum of human skin equivalents. J. Investig. Dermatol. 2013, 133, 59–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elias, P.M.; Brown, B.E.; Ziboh, V.A. The Permeability Barrier in Essential Fatty Acid Deficiency: Evidence for a Direct Role for Linoleic Acid in Barrier Function. J. Investig. Dermatol. 1980, 74, 230–233. [Google Scholar] [CrossRef] [Green Version]
- Mbarik, M.; Biam, R.S.; Robichaud, P.P.; Surette, M.E. The impact of PUFA on cell responses: Caution should be exercised when selecting PUFA concentrations in cell culture. Prostaglandins Leukot. Essent. Fat. Acids 2020, 155, 102083. [Google Scholar] [CrossRef]
- Xiao, K.; Liu, C.; Tu, Z.; Xu, Q.; Chen, S.; Zhang, Y.; Wang, X.; Zhang, J.; Hu, C.-A.A.; Liu, Y. Activation of the NF-κB and MAPK Signaling Pathways Contributes to the Inflammatory Responses, but Not Cell Injury, in IPEC-1 Cells Challenged with Hydrogen Peroxide. Oxidative Med. Cell. Longev. 2020, 2020, 5803639. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.; Shi, P.; Sun, Y.; Sun, J.; Dong, J.-N.; Wang, H.-G.; Zuo, L.-G.; Gong, J.-F.; Li, Y.; Gu, L.-L.; et al. DHA protects against experimental colitis in IL-10-deficient mice associated with the modulation of intestinal epithelial barrier function. Br. J. Nutr. 2015, 114, 181–188. [Google Scholar] [CrossRef] [Green Version]
- Simard, M.; Rioux, G.; Morin, S.; Martin, C.; Guérin, S.L.; Flamand, N.; Julien, P.; Fradette, J.; Pouliot, R. Investigation of n-3 Polyunsaturated Fatty Acid Biological Activity in a Tissue-Engineered Skin Model Involving Psoriatic cells: Omega-3 and psoriatic skin. J. Investig. Dermatol. 2021, 141, 2391–2401. [Google Scholar] [CrossRef]
- Schuchardt, J.P.; Ostermann, A.I.; Stork, L.; Fritzsch, S.; Kohrs, H.; Greupner, T.; Hahn, A.; Schebb, N.H. Effect of DHA supplementation on oxylipin levels in plasma and immune cell stimulated blood. Prostaglandins Leukot. Essent. Fat. Acids 2017, 121, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Morin, S.; Simard, M.; Flamand, N.; Pouliot, R. Biological action of docosahexaenoic acid in a 3D tissue-engineered psoriatic skin model: Focus on the PPAR signaling pathway. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2021, 1866, 159032. [Google Scholar] [CrossRef] [PubMed]
- Kendall, A.C.; Pilkington, S.M.; Murphy, S.A.; Del Carratore, F.; Sunarwidhi, A.L.; Kiezel-Tsugunova, M.; Urquhart, P.; Watson, R.E.B.; Breitling, R.; Rhodes, L.E.; et al. Dynamics of the human skin mediator lipidome in response to dietary ω-3 fatty acid supplementation. Faseb J. 2019, 33, 13014–13027. [Google Scholar] [CrossRef] [Green Version]
- Tsukita, S.; Furuse, M. Claudin-based barrier in simple and stratified cellular sheets. Curr. Opin. Cell Biol. 2002, 14, 531–536. [Google Scholar] [CrossRef]
- Kirschner, N.; Houdek, P.; Fromm, M.; Moll, I.; Brandner, J.M. Tight junctions form a barrier in human epidermis. Eur. J. Cell Biol. 2010, 89, 839–842. [Google Scholar] [CrossRef]
- Morita, K.; Miyachi, Y.; Furuse, M. Tight junctions in epidermis: From barrier to keratinization. Eur. J. Dermatol. 2011, 21, 12–17. [Google Scholar] [CrossRef]
- Hintsala, H.-R.; Siponen, M.; Haapasaari, K.-M.; Karihtala, P.; Soini, Y. Claudins 1, 2, 3, 4, 5 and 7 in solar keratosis and squamocellular carcinoma of the skin. Int. J. Clin. Exp. Pathol. 2013, 6, 2855–2863. [Google Scholar] [PubMed]
- Tu, Y.; Wu, W.; Guo, Y.; Lu, F.; Xu, D.; Li, X.; Zhao, Y.; He, L. Upregulation of hsa-miR-31-3p induced by ultraviolet affects keratinocytes permeability barrier by targeting CLDN1. Biochem. Biophys. Res. Commun. 2020, 532, 626–632. [Google Scholar] [CrossRef]
- Hadj-Rabia, S.; Baala, L.; Vabres, P.; Hamel-Teillac, D.; Jacquemin, E.; Fabre, M.; Lyonnet, S.; de Prost, Y.; Munnich, A.; Hadchouel, M.; et al. Claudin-1 gene mutations in neonatal sclerosing cholangitis associated with ichthyosis: A tight junction disease. Gastroenterology 2004, 127, 1386–1390. [Google Scholar] [CrossRef]
- De Benedetto, A.; Rafaels, N.M.; McGirt, L.Y.; Ivanov, A.I.; Georas, S.N.; Cheadle, C.; Berger, A.E.; Zhang, K.; Vidyasagar, S.; Yoshida, T.; et al. Tight junction defects in patients with atopic dermatitis. J. Allergy Clin. Immunol. 2011, 127, 773–786.e7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tokumasu, R.; Yamaga, K.; Yamazaki, Y.; Murota, H.; Suzuki, K.; Tamura, A.; Bando, K.; Furuta, Y.; Katayama, I.; Tsukita, S. Dose-dependent role of claudin-1 in vivo in orchestrating features of atopic dermatitis. Proc. Natl. Acad. Sci. USA 2016, 113, E4061–E4068. [Google Scholar] [CrossRef] [Green Version]
- Jung, Y.O.; Jeong, H.; Cho, Y.; Lee, E.O.; Jang, H.W.; Kim, J.; Nam, K.; Lim, K.M. Lysates of a Probiotic, Lactobacillus rhamnosus, Can Improve Skin Barrier Function in a Reconstructed Human Epidermis Model. Int. J. Mol. Sci. 2019, 20, 4289. [Google Scholar] [CrossRef] [Green Version]
- Strüver, K.; Friess, W.; Hedtrich, S. Development of a Perfusion Platform for Dynamic Cultivation of in vitro Skin Models. Ski. Pharmacol. Physiol. 2017, 30, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Beguin, P.; Errachid, A.; Larondelle, Y.; Schneider, Y.J. Effect of polyunsaturated fatty acids on tight junctions in a model of the human intestinal epithelium under normal and inflammatory conditions. Food Funct. 2013, 4, 923–931. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, Q.; Zhang, M.; Wang, C.; Zhu, Z.; Li, N.; Li, J. Effect of n-3 polyunsaturated fatty acids on membrane microdomain localization of tight junction proteins in experimental colitis. FEBS J. 2008, 275, 411–420. [Google Scholar] [CrossRef]
- Whiting, C.V.; Bland, P.W.; Tarlton, J.F. Dietary n-3 polyunsaturated fatty acids reduce disease and colonic proinflammatory cytokines in a mouse model of colitis. Inflamm. Bowel Dis. 2005, 11, 340–349. [Google Scholar] [CrossRef]
- Reed, J.T.; Ghadially, R.; Elias, P.M. Skin type, but neither race nor gender, influence epidermal permeability barrier function. Arch. Dermatol. 1995, 131, 1134–1138. [Google Scholar] [CrossRef]
- Darlenski, R.; Fluhr, J.W. Influence of skin type, race, sex, and anatomic location on epidermal barrier function. Clin. Dermatol. 2012, 30, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Xue, Z.; Yang, H.; Zhao, F.; Liu, C.; Chen, J.; Lu, S.; Zou, Z.; Zhou, Y.; Zhang, X. Differential effects of EPA and DHA on DSS-induced colitis in mice and possible mechanisms involved. Food Funct. 2021, 12, 1803–1817. [Google Scholar] [CrossRef]
- Shiozaki, A.; Bai, X.H.; Shen-Tu, G.; Moodley, S.; Takeshita, H.; Fung, S.Y.; Wang, Y.; Keshavjee, S.; Liu, M. Claudin 1 mediates TNFalpha-induced gene expression and cell migration in human lung carcinoma cells. PLoS ONE 2012, 7, e38049. [Google Scholar] [CrossRef] [Green Version]
- Zhu, L.; Han, J.; Li, L.; Wang, Y.; Li, Y.; Zhang, S. Claudin Family Participates in the Pathogenesis of Inflammatory Bowel Diseases and Colitis-Associated Colorectal Cancer. Front. Immunol. 2019, 10, 1441. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Yokouchi, M.; Nagao, K.; Ishii, K.; Amagai, M.; Kubo, A. Functional tight junction barrier localizes in the second layer of the stratum granulosum of human epidermis. J. Dermatol. Sci. 2013, 71, 89–99. [Google Scholar] [CrossRef]
- Ogasawara, N.; Kojima, T.; Go, M.; Ohkuni, T.; Koizumi, J.; Kamekura, R.; Masaki, T.; Murata, M.; Tanaka, S.; Fuchimoto, J.; et al. PPARgamma agonists upregulate the barrier function of tight junctions via a PKC pathway in human nasal epithelial cells. Pharmacol. Res. 2010, 61, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Iitaka, D.; Moodley, S.; Shimizu, H.; Bai, X.H.; Liu, M. PKCδ-iPLA2-PGE2-PPARγ signaling cascade mediates TNF-α induced Claudin 1 expression in human lung carcinoma cells. Cell. Signal. 2015, 27, 568–577. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, Q.; Wang, M.; Zhao, S.; Xu, G.; Li, J. n-3 polyunsaturated fatty acids prevent disruption of epithelial barrier function induced by proinflammatory cytokines. Mol. Immunol. 2008, 45, 1356–1365. [Google Scholar] [CrossRef]
- Yamamoto, T.; Saeki, Y.; Kurasawa, M.; Kuroda, S.; Arase, S.; Sasaki, H. Effect of RNA interference of tight junction-related molecules on intercellular barrier function in cultured human keratinocytes. Arch. Dermatol. Res. 2008, 300, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Polyak, K.; Halushka, M.K.; Nassar, H.; Kouprina, N.; Iacobuzio-Donahue, C.; Wu, X.; Sukumar, S.; Hicks, J.; De Marzo, A.; et al. Serial analysis of gene expression of lobular carcinoma in situ identifies down regulation of claudin 4 and overexpression of matrix metalloproteinase 9. Breast Cancer Res. 2008, 10, R91. [Google Scholar] [CrossRef] [Green Version]
- Sobel, G.; Paska, C.; Szabo, I.; Kiss, A.; Kadar, A.; Schaff, Z. Increased expression of claudins in cervical squamous intraepithelial neoplasia and invasive carcinoma. Hum. Pathol. 2005, 36, 162–169. [Google Scholar] [CrossRef]
- Cunningham, S.C.; Kamangar, F.; Kim, M.P.; Hammoud, S.; Haque, R.; Iacobuzio-Donahue, C.A.; Maitra, A.; Ashfaq, R.; Hustinx, S.; Heitmiller, R.E.; et al. Claudin-4, mitogen-activated protein kinase kinase 4, and stratifin are markers of gastric adenocarcinoma precursor lesions. Cancer Epidemiol. Biomark. Prev. 2006, 15, 281–287. [Google Scholar] [CrossRef] [Green Version]
- Montgomery, E.; Mamelak, A.J.; Gibson, M.; Maitra, A.; Sheikh, S.; Amr, S.S.; Yang, S.; Brock, M.; Forastiere, A.; Zhang, S.; et al. Overexpression of claudin proteins in esophageal adenocarcinoma and its precursor lesions. Appl. Immunohistochem. Mol. Morphol. 2006, 14, 24–30. [Google Scholar] [CrossRef]
- Brandner, J.M. Tight junctions and tight junction proteins in mammalian epidermis. Eur. J. Pharm. Biopharm. 2009, 72, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Germain, L.; Rouabhia, M.; Guignard, R.; Carrier, L.; Bouvard, V.; Auger, F.A. Improvement of human keratinocyte isolation and culture using thermolysin. Burns 1993, 19, 99–104. [Google Scholar] [CrossRef]
- Franz, T.J. Percutaneous absorption on the relevance of in vitro data. J. Investig. Dermatol. 1975, 64, 190–195. [Google Scholar] [CrossRef] [Green Version]
- Duque-Fernandez, A.; Gauthier, L.; Simard, M.; Jean, J.; Gendreau, I.; Morin, A.; Soucy, J.; Auger, M.; Pouliot, R. A 3D-psoriatic skin model for dermatological testing: The impact of culture conditions. Biochem. Biophys. Rep. 2016, 8, 268–276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Julien, C.; Berthiaume, L.; Hadj-Tahar, A.; Rajput, A.H.; Bédard, P.J.; Paolo, T.D.; Julien, P.; Calon, F. Postmortem brain fatty acid profile of levodopa-treated Parkinson disease patients and parkinsonian monkeys. Neurochem. Int. 2006, 48, 404–414. [Google Scholar] [CrossRef] [PubMed]
- Rioux, G.; Pouliot-Bérubé, C.; Simard, M.; Benhassine, M.; Soucy, J.; Guérin, S.L.; Pouliot, R. The Tissue-Engineered Human Psoriatic Skin Substitute: A Valuable In Vitro Model to Identify Genes with Altered Expression in Lesional Psoriasis. Int. J. Mol. Sci. 2018, 19, 2923. [Google Scholar] [CrossRef] [PubMed] [Green Version]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tremblay, A.; Simard, M.; Morin, S.; Pouliot, R. Docosahexaenoic Acid Modulates Paracellular Absorption of Testosterone and Claudin-1 Expression in a Tissue-Engineered Skin Model. Int. J. Mol. Sci. 2021, 22, 13091. https://doi.org/10.3390/ijms222313091
Tremblay A, Simard M, Morin S, Pouliot R. Docosahexaenoic Acid Modulates Paracellular Absorption of Testosterone and Claudin-1 Expression in a Tissue-Engineered Skin Model. International Journal of Molecular Sciences. 2021; 22(23):13091. https://doi.org/10.3390/ijms222313091
Chicago/Turabian StyleTremblay, Andréa, Mélissa Simard, Sophie Morin, and Roxane Pouliot. 2021. "Docosahexaenoic Acid Modulates Paracellular Absorption of Testosterone and Claudin-1 Expression in a Tissue-Engineered Skin Model" International Journal of Molecular Sciences 22, no. 23: 13091. https://doi.org/10.3390/ijms222313091








