Omics and CRISPR-Cas9 Approaches for Molecular Insight, Functional Gene Analysis, and Stress Tolerance Development in Crops
Abstract
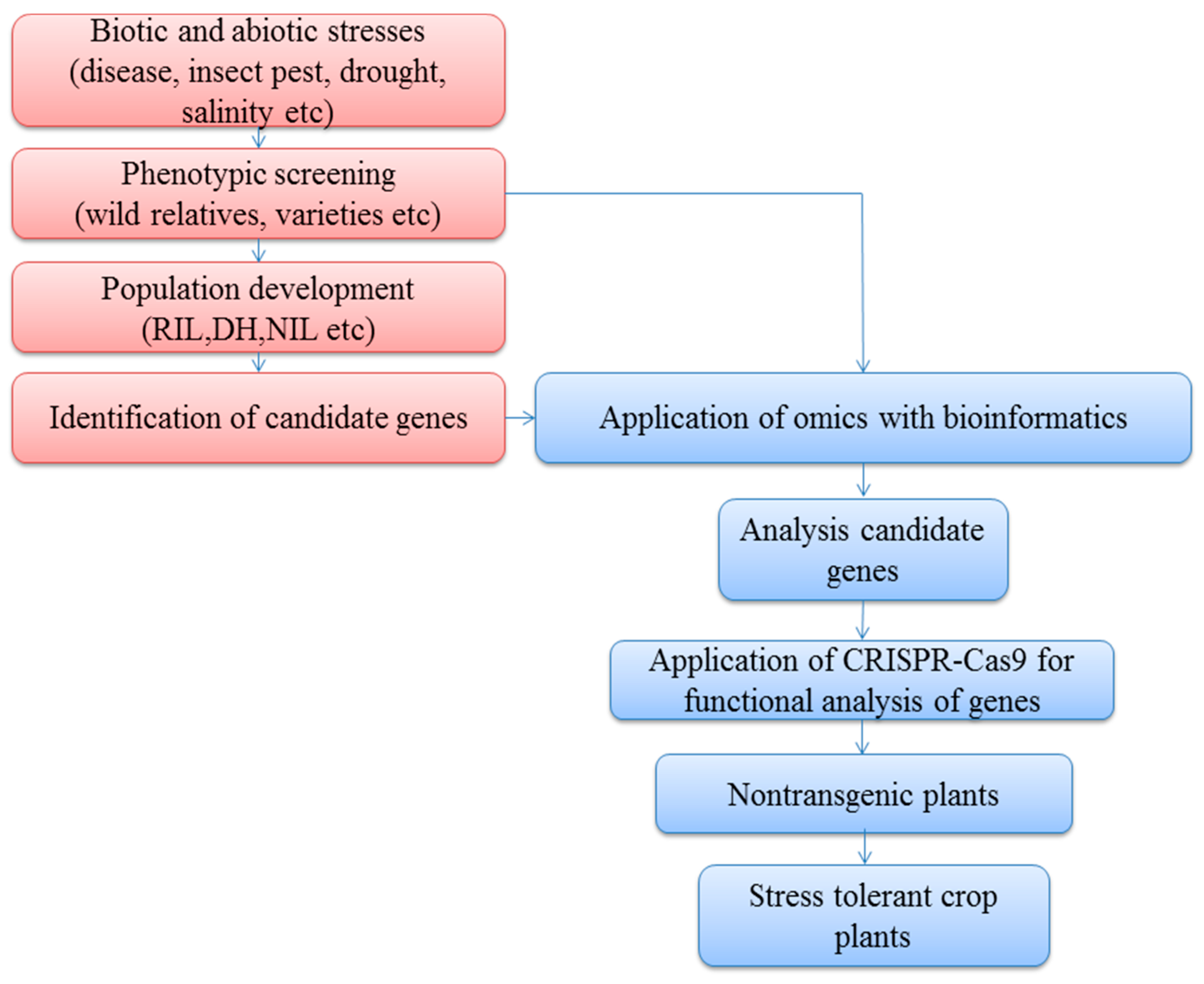
:1. Introduction
2. Multi-Omics Technology
2.1. Genomics
2.2. Transcriptomics
2.3. Proteomics
2.4. Metabolomics
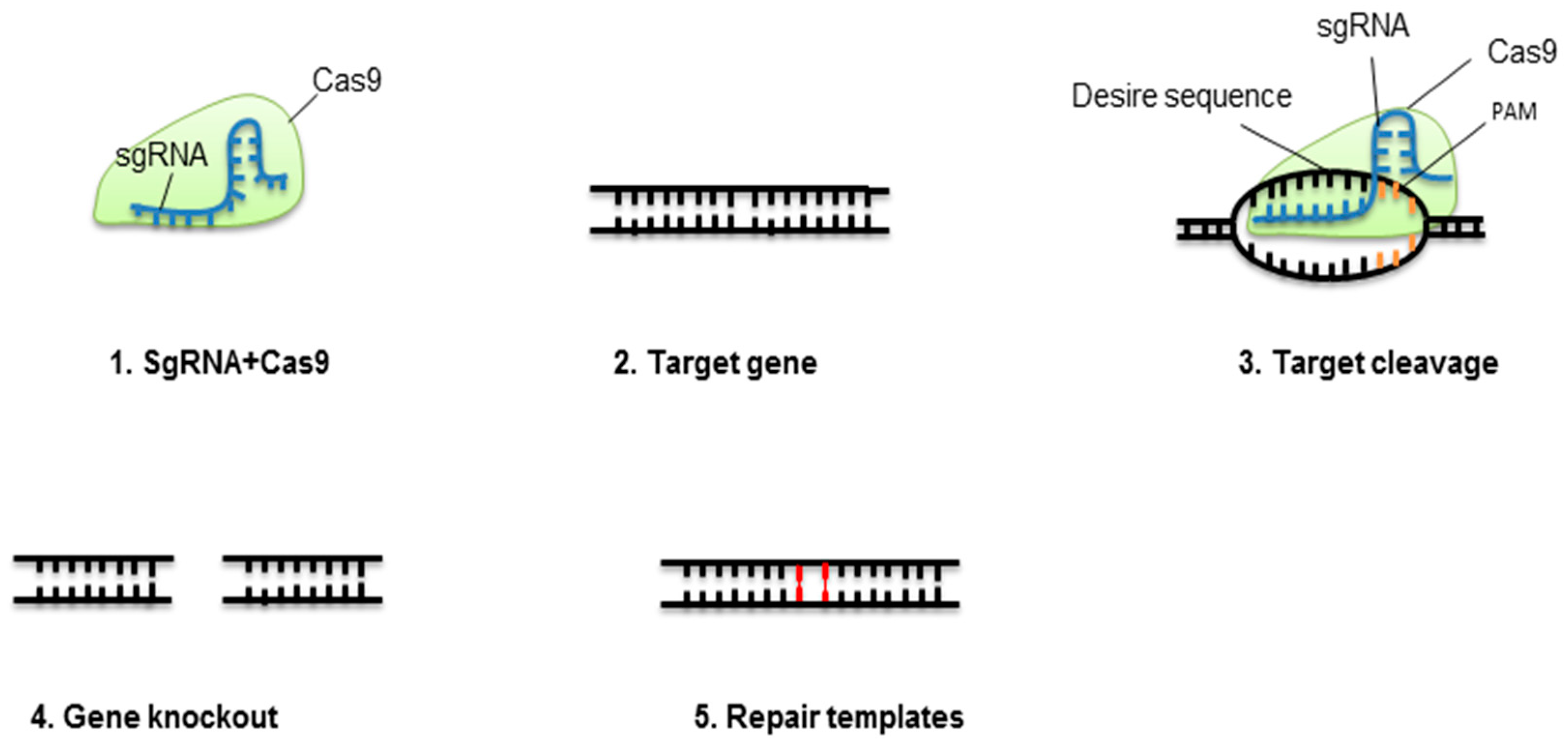
3. CRISPR Technology
3.1. CRISPR-Cas9 Genome Engineering to Biotic Stress Tolerance
3.2. CRISPR-Cas9 Genome Engineering to Abiotic Stress Tolerance
4. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Manavalan, L.P.; Guttikonda, S.K.; Phan Tran, L.-S.; Nguyen, H.T. Physiological and molecular approaches to improve drought resistance in soybean. Plant Cell Physiol. 2009, 50, 1260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Razzaq, M.K.; Rauf, S.; Khurshid, M.; Iqbal, S.; Bhat, J.A.; Farzand, A.; Riaz, A.; Xing, G.; Gai, J. Pollen viability an index of abiotic stresses tolerance and methods for the improved pollen viability. Pak. J. Agric. Res. 2019, 32. [Google Scholar] [CrossRef]
- Hasin, Y.; Seldin, M.; Lusis, A. Multi-omics approaches to disease. Genome Biol. 2017, 18, 1. [Google Scholar] [CrossRef] [PubMed]
- Karahalil, B. Overview of systems biology and omics technologies. Curr. Med. Chem. 2016, 23, 4221. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Pandey, G.K. Genomics and functional genomics of stress-mediated signaling in plants: Volume I. Curr. Genom. 2017, 18, 467. [Google Scholar]
- Singh, B.; Salaria, N.; Thakur, K.; Kukreja, S.; Gautam, S.; Goutam, U. Functional genomic approaches to improve crop plant heat stress tolerance. F1000Research 2019, 8. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A.; Rahman, M.A.; Rathinasabapathi, B.; Babar, M.A. UPLC-HRMS-based untargeted metabolic profiling reveals changes in chickpea (Cicer arietinum) metabolome following long-term drought stress. Plant Cell Environ. 2019, 42, 115. [Google Scholar] [CrossRef] [Green Version]
- Feng, Z.; Zhang, B.; Ding, W.; Liu, X.; Yang, D.-L.; Wei, P.; Cao, F.; Zhu, S.; Zhang, F.; Mao, Y. Efficient genome editing in plants using a CRISPR/Cas system. Cell Res. 2013, 23, 1229. [Google Scholar] [CrossRef] [Green Version]
- Jiang, W.; Zhou, H.; Bi, H.; Fromm, M.; Yang, B.; Weeks, D.P. Demonstration of CRISPR/Cas9/sgRNA-mediated targeted gene modification in Arabidopsis, tobacco, sorghum and rice. Nucleic Acids Res. 2013, 41, e188. [Google Scholar] [CrossRef]
- Li, J.-F.; Norville, J.E.; Aach, J.; McCormack, M.; Zhang, D.; Bush, J.; Church, G.M.; Sheen, J. Multiplex and homologous recombination-mediated genome editing in Arabidopsis and Nicotiana benthamiana using guide RNA and Cas9. Nat. Biotechnol. 2013, 31, 688. [Google Scholar] [CrossRef]
- Shan, Q.; Wang, Y.; Li, J.; Zhang, Y.; Chen, K.; Liang, Z.; Zhang, K.; Liu, J.; Xi, J.J.; Qiu, J.-L. Targeted genome modification of crop plants using a CRISPR-Cas system. Nat. Biotechnol. 2013, 31, 686. [Google Scholar] [CrossRef] [PubMed]
- Chandrasegaran, S.; Carroll, D. Origins of programmable nucleases for genome engineering. J. Mol. Biol. 2016, 428, 963. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, Y.; Zhang, H.; Xu, N.; Zhang, B.; Gou, F.; Zhu, J.-K. Application of the CRISPR–Cas system for efficient genome engineering in plants. Mol. Plant 2013, 6, 2008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, X.; Zhang, Q.; Zhu, Q.; Liu, W.; Chen, Y.; Qiu, R.; Wang, B.; Yang, Z.; Li, H.; Lin, Y. A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants. Mol. Plant 2015, 8, 1274. [Google Scholar] [CrossRef] [PubMed]
- Malzahn, A.; Lowder, L.; Qi, Y. Plant genome editing with TALEN and CRISPR. Cell Biosci. 2017, 7, 21. [Google Scholar] [CrossRef] [Green Version]
- Wang, P.; Su, L.; Gao, H.; Jiang, X.; Wu, X.; Li, Y.; Zhang, Q.; Wang, Y.; Ren, F. Genome-wide characterization of bHLH genes in grape and analysis of their potential relevance to abiotic stress tolerance and secondary metabolite biosynthesis. Front. Plant Sci. 2018, 9, 64. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Li, X.; Ma, B.; Gao, Q.; Du, H.; Han, Y.; Li, Y.; Cao, Y.; Qi, M.; Zhu, Y. The tartary buckwheat genome provides insights into rutin biosynthesis and abiotic stress tolerance. Mol. Plant 2017, 10, 1224. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Jiang, J.; Li, H.; Liu, G. The salt-responsive transcriptome of Populussimonii × Populusnigra via DGE. Gene 2012, 504, 203. [Google Scholar] [CrossRef]
- Gilliham, M.; Able, J.A.; Roy, S.J. Translating knowledge about abiotic stress tolerance to breeding programmes. Plant J. 2017, 90, 898. [Google Scholar] [CrossRef] [Green Version]
- Le, D.T.; Nishiyama, R.; Watanabe, Y.; Tanaka, M.; Seki, M.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Tran, L.-S.P. Differential gene expression in soybean leaf tissues at late developmental stages under drought stress revealed by genome-wide transcriptome analysis. PLoS ONE 2012, 7, e49522. [Google Scholar] [CrossRef] [Green Version]
- Thiry, A.A.; Chavez Dulanto, P.N.; Reynolds, M.P.; Davies, W.J. How can we improve crop genotypes to increase stress resilience and productivity in a future climate? A new crop screening method based on productivity and resistance to abiotic stress. J. Exp. Bot. 2016, 67, 5593. [Google Scholar] [CrossRef] [PubMed]
- Yao, D.; Zhang, X.; Zhao, X.; Liu, C.; Wang, C.; Zhang, Z.; Zhang, C.; Wei, Q.; Wang, Q.; Yan, H. Transcriptome analysis reveals salt-stress-regulated biological processes and key pathways in roots of cotton (Gossypium hirsutum L.). Genomics 2011, 98, 47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rauf, S.; Warburton, M.; Naeem, A.; Kainat, W. Validated markers for sunflower (Helianthus annuus L.) breeding. Oilseeds Fats Crop. Lipids 2020, 27. [Google Scholar] [CrossRef]
- Shen, W.; Li, H.; Teng, R.; Wang, Y.; Wang, W.; Zhuang, J. Genomic and transcriptomic analyses of HD-Zip family transcription factors and their responses to abiotic stress in tea plant. Genomics 2019, 111, 1142. [Google Scholar] [CrossRef]
- Witek, K.; Jupe, F.; Witek, A.I.; Baker, D.; Clark, M.D.; Jones, J.D. Accelerated cloning of a potato late blight resistance gene using RenSeq and SMRT sequencing. Nat. Biotechnol. 2016, 34, 656. [Google Scholar] [CrossRef] [Green Version]
- Steuernagel, B.; Periyannan, S.K.; Hernandez Pinzon, I.; Witek, K.; Rouse, M.N.; Yu, G.; Lagudah, E.S. Rapid cloning of disease resistance genes in plants using mutagenesis and sequence capture. Nat. Biotechnol. 2016, 34, 652. [Google Scholar] [CrossRef]
- Song, L.; Huang, S.S.C.; Wise, A.; Castanon, R.; Nery, J.R.; Chen, H.; Ecker, J.R. A transcription factor hierarchy defines an environmental stress response network. Science 2016, 354, 6312. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.; Izzah, N.K.; Choi, B.; Joh, H.J.; Lee, S.; Perumal, S.; Seo, J.; Ahn, K.; Jo, E.J.; Choi, G.J. Genotyping by sequencing map permits identification of clubroot resistance QTLs and revision of the reference genome assembly in cabbage (Brassica oleracea L.). DNA Res. 2015, 23, 29. [Google Scholar] [CrossRef] [Green Version]
- Leisner, C.P.; Yendrek, C.R.; Ainsworth, E.A. Physiological and transcriptomic responses in the seed coat of field-grown soybean (Glycine max L. Merr.) to abiotic stress. BMC Plant Biol. 2017, 17, 242. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Xu, Y.; Huang, B. Lipidomic reprogramming associated with drought stress priming-enhanced heat tolerance in tall fescue (Festuca arundinacea). Plant Cell Environ. 2019, 42, 947. [Google Scholar] [CrossRef]
- Zhang, X.; Yao, C.; Fu, S.; Xuan, H.; Wen, S.; Liu, C.; Li, F.; Liu, A.; Bi, S.; Zhang, S.; et al. Stress2TF: A manually curated database of TF regulation in plant response to stress. Gene 2018, 638, 36. [Google Scholar] [CrossRef]
- Rizhsky, L.; Liang, H.; Shuman, J.; Shulaev, V.; Davletova, S.; Mittler, R. When defense pathways collide. The response of Arabidopsis to a combination of drought and heat stress. Plant Physiol. 2004, 134, 1683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hewezi, T.; Léger, M.; Gentzbittel, L. A comprehensive analysis of the combined effects of high light and high temperature stresses on gene expression in sunflower. Ann. Bot. 2008, 102, 127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koussevitzky, S.; Suzuki, N.; Huntington, S.; Armijo, L.; Sha, W.; Cortes, D.; Shulaev, V.; Mittler, R. Ascorbate peroxidase 1 plays a key role in the response of Arabidopsis thaliana to stress combination. J. Biol. Chem. 2008, 283, 34197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, D.; Chattopadhyay, D. Analysis of gene expression in response to water deficit of chickpea (Cicer arietinum L.) varieties differing in drought tolerance. BMC Plant Biol. 2010, 10, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, Y.-N.; Shi, D.-Q.; Ruan, M.-B.; Zhang, L.-L.; Meng, Z.-H.; Liu, J.; Yang, W.-C. Transcriptome analysis reveals crosstalk of responsive genes to multiple abiotic stresses in cotton (Gossypium hirsutum L.). PLoS ONE 2013, 8, e80218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, P.; Cao, W.; Fang, H.; Xu, S.; Yin, S.; Zhang, Y.; Lin, D.; Wang, J.; Chen, Y.; Xu, C. Transcriptomic profiling of the maize (Zea mays L.) leaf response to abiotic stresses at the seedling stage. Front. Plant Sci. 2017, 8, 290. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Qin, F. Genome wide association study reveals natural variations contributing to drought resistance in crops. Front. Plant Sci. 2017, 8, 1110. [Google Scholar] [CrossRef] [Green Version]
- Gibbings, J.G.; Cook, B.P.; Dufault, M.R.; Madden, S.L.; Khuri, S.; Turnbull, C.J. Global transcript analysis of rice leaf and seed using SAGE technology. Plant Biotechnol. J. 2003, 1, 271. [Google Scholar] [CrossRef]
- Hilton, I.B.; D’Ippolito, A.M.; Vockley, C.M.; Thakore, P.I.; Crawford, G.E.; Reddy, T.E.; Gersbach, C.A. Epigenome editing by a CRISPR-Cas9-based acetyltransferase activates genes from promoters and enhancers. Nat. Biotechnol. 2015, 33, 510. [Google Scholar] [CrossRef] [Green Version]
- Kearns, N.A.; Pham, H.; Tabak, B.; Genga, R.M.; Silverstein, N.J.; Garber, M.; Maehr, R. Functional annotation of native enhancers with a Cas9-histone demethylase fusion. Nature Methods 2015, 12, 401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luan, H.; Shen, H.; Pan, Y.; Guo, B.; Lv, C.; Xu, R. Elucidating the hypoxic stress response in barley (Hordeum vulgare L.) during waterlogging: A proteomics approach. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Nakagami, H.; Sugiyama, N.; Ishihama, Y.; Shirasu, K. Shotguns in the front line: Phosphoproteomics in plants. Plant Cell Physiol. 2012, 53, 118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Komatsu, S.; Kamal, A.H.; Hossain, Z. Wheat proteomics: Proteome modulation and abiotic stress acclimation. Front. Plant Sci. 2014, 5, 684. [Google Scholar] [CrossRef] [Green Version]
- Shao, S.; Guo, T.; Aebersold, R. Mass spectrometry-based proteomic quest for diabetes biomarkers. Biochimica et Biophysica Acta (BBA) Proteins Proteom. 2015, 1854, 519. [Google Scholar] [CrossRef]
- Yin, X.; Komatsu, S. Quantitative proteomics of nuclear phosphoproteins in the root tip of soybean during the initial stages of flooding stress. J. Proteom. 2015, 119, 183. [Google Scholar] [CrossRef]
- Pandey, A.; Rajamani, U.; Verma, J.; Subba, P.; Chakraborty, N.; Datta, A.; Chakraborty, S.; Chakraborty, N. Identification of extracellular matrix proteins of rice (Oryza sativa L.) involved in dehydration-responsive network: A proteomic approach. J. Proteome Res. 2010, 9, 3443. [Google Scholar] [CrossRef]
- Alvarez, S.; Berla, B.M.; Sheffield, J.; Cahoon, R.E.; Jez, J.M.; Hicks, L.M. Comprehensive analysis of the Brassica juncea root proteome in response to cadmium exposure by complementary proteomic approaches. Proteomics 2009, 9, 2419. [Google Scholar] [CrossRef]
- Hossain, Z.; Hajika, M.; Komatsu, S. Comparative proteome analysis of high and low cadmium accumulating soybeans under cadmium stress. Amino Acids 2012, 43, 2393. [Google Scholar] [CrossRef]
- Hradilová, J.; Řehulka, P.; Řehulková, H.; Vrbová, M.; Griga, M.; Brzobohatý, B. Comparative analysis of proteomic changes in contrasting flax cultivars upon cadmium exposure. Electrophoresis 2010, 31, 421. [Google Scholar] [CrossRef]
- Semane, B.; Dupae, J.; Cuypers, A.; Noben, J.-P.; Tuomainen, M.; Tervahauta, A.; Kärenlampi, S.; Van Belleghem, F.; Smeets, K.; Vangronsveld, J. Leaf proteome responses of Arabidopsis thaliana exposed to mild cadmium stress. J. Plant Physiol. 2010, 167, 247. [Google Scholar] [CrossRef]
- Heidarvand, L.; Maali-Amiri, R. Physio-biochemical and proteome analysis of chickpea in early phases of cold stress. J. Plant Physiol. 2013, 170, 459. [Google Scholar] [CrossRef]
- Zhang, M.; Lv, D.; Ge, P.; Bian, Y.; Chen, G.; Zhu, G.; Li, X.; Yan, Y. Phosphoproteome analysis reveals new drought response and defense mechanisms of seedling leaves in bread wheat (Triticum aestivum L.). J. Proteom. 2014, 109, 290. [Google Scholar] [CrossRef]
- Wang, Y.S.; Yao, H.Y.; Xue, H.W. Lipidomic profiling analysis reveals the dynamics of phospholipid molecules in Arabidopsis thaliana seedling growth. J. Integr. Plant Biol. 2016, 58, 890. [Google Scholar] [CrossRef] [PubMed]
- Tamburino, R.; Vitale, M.; Ruggiero, A.; Sassi, M.; Sannino, L.; Arena, S.; Costa, A.; Batelli, G.; Zambrano, N.; Scaloni, A. Chloroplast proteome response to drought stress and recovery in tomato (Solanum lycopersicum L.). BMC Plant Biol. 2017, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lou, X.; Wang, H.; Ni, X.; Gao, Z.; Iqbal, S. Integrating proteomic and transcriptomic analyses of loquat in response to cold stress. Gene 2018, 677, 57. [Google Scholar] [CrossRef] [PubMed]
- Frederich, M.; Pirotte, B.; Fillet, M.; De Tullio, P. Metabolomics as a challenging approach for medicinal chemistry and personalized medicine. J. Med. Chem. 2016, 59, 8649. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, V.S.; Macedo, F.A.; Do Vale, J.S.; Silva, D.B.; Carollo, C.A. Metabolomics as a tool for understanding the evolution of Tabebuias ensulato. Metabolomics 2017, 13, 72. [Google Scholar] [CrossRef]
- Parida, A.K.; Panda, A.; Rangani, J. Metabolomics-guided elucidation of abiotic stress tolerance mechanisms in plants. In Plant Metabolites and Regulation under Environmental Stress; Elsevier: Amsterdam, The Netherlands, 2018; p. 89. [Google Scholar]
- Kim, H.K.; Choi, Y.H.; Verpoorte, R. NMR-based metabolomic analysis of plants. Nat. Protoc. 2010, 5, 536. [Google Scholar] [CrossRef]
- Vickers, N.J. Animal communication: When I’m calling you, will you answer too? Curr. Biol. 2017, 27, R713. [Google Scholar] [CrossRef]
- Muthuramalingam, P.; Jeyasri, R.; Selvaraj, A.; Pandian, S.K.; Ramesh, M. Integrated transcriptomic and metabolomic analyses of glutamine metabolism genes unveil key players in Oryza sativa (L.) to ameliorate the unique and combined abiotic stress tolerance. Int. J. Biol. Macromol. 2020, 164, 222. [Google Scholar] [CrossRef] [PubMed]
- Muthuramalingam, P.; Krishnan, S.R.; Pandian, S.; Mareeswaran, N.; Aruni, W.; Pandian, S.K.; Ramesh, M. Global analysis of threonine metabolism genes unravel key players in rice to improve the abiotic stress tolerance. Sci. Rep. 2018, 8, 9270. [Google Scholar] [CrossRef] [PubMed]
- Lakshmanan, M.; Cheung, C.; Mohanty, B.; Lee, D.-Y. Modeling rice metabolism: From elucidating environmental effects on cellular phenotype to guiding crop improvement. Front. Plant Sci. 2016, 7, 1795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jwa, N.-S.; Agrawal, G.K.; Tamogami, S.; Yonekura, M.; Han, O.; Iwahashi, H.; Rakwal, R. Role of defense/stress-related marker genes, proteins and secondary metabolites in defining rice self-defense mechanisms. Plant Physiol. Biochem. 2006, 44, 261. [Google Scholar] [CrossRef] [PubMed]
- Urano, K.; Maruyama, K.; Ogata, Y.; Morishita, Y.; Takeda, M.; Sakurai, N.; Suzuki, H.; Saito, K.; Shibata, D.; Kobayashi, M. Characterization of the ABA-regulated global responses to dehydration in Arabidopsis by metabolomics. Plant J. 2009, 57, 1065. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, V.; Obudulu, O.; Bygdell, J.; Löfstedt, T.; Rydén, P.; Nilsson, R.; Ahnlund, M.; Johansson, A.; Jonsson, P.; Freyhult, E. OnPLS integration of transcriptomic, proteomic and metabolomic data shows multi-level oxidative stress responses in the cambium of transgenic hipI-superoxide dismutase Populus plants. BMC Genom. 2013, 14, 893. [Google Scholar] [CrossRef] [Green Version]
- Shen, Q.; Fu, L.; Dai, F.; Jiang, L.; Zhang, G.; Wu, D. Multi-omics analysis reveals molecular mechanisms of shoot adaption to salt stress in Tibetan wild barley. BMC Genom. 2016, 17, 889. [Google Scholar] [CrossRef] [Green Version]
- Hirai, M.Y.; Sugiyama, K.; Sawada, Y.; Tohge, T.; Obayashi, T.; Suzuki, A.; Goda, H. Omics based identification of Arabidopsis Myb transcription factors regulating aliphatic glucosinolate biosynthesis. Proc. Natl. Acad. Sci. USA 2007, 104, 6478. [Google Scholar] [CrossRef] [Green Version]
- Xu, R.; Yang, Y.; Qin, R.; Li, H.; Qiu, C.; Li, L.; Wei, P.; Yang, J. Rapid improvement of grain weight via highly efficient CRISPR/Cas9-mediated multiplex genome editing in rice. J. Genet. Genom. Yi Chuanxuebao 2016, 43, 529. [Google Scholar] [CrossRef]
- Shi, J.; Gao, H.; Wang, H.; Lafitte, H.R.; Archibald, R.L.; Yang, M.; Hakimi, S.M.; Mo, H.; Habben, J.E. ARGOS 8 variants generated by CRISPR-Cas9 improve maize grain yield under field drought stress conditions. Plant Biotechnol. J. 2017, 15, 207. [Google Scholar] [CrossRef] [Green Version]
- Fan, D.; Liu, T.; Li, C.; Jiao, B.; Li, S.; Hou, Y.; Luo, K. Efficient CRISPR/Cas9-mediated targeted mutagenesis in Populus in the first generation. Sci. Rep. 2015, 5, 12217. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.-J.; Xue, L.-J. CRISPRing into the woods. GM Crop. Food 2015, 6, 206. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Feng, F.; Wei, H.; Mei, H.; Xu, K.; Chen, S.; Li, T.; Liang, X.; Liu, H.; Luo, L. Genome-wide association study for plant height and grain yield in rice under contrasting moisture regimes. Front. Plant Sci. 2016, 7, 1801. [Google Scholar] [CrossRef] [Green Version]
- Chang, Z.; Chen, Z.; Yan, W.; Xie, G.; Lu, J.; Wang, N.; Lu, Q.; Yao, N.; Yang, G.; Xia, J. An ABC transporter, OsABCG26, is required for anther cuticle and pollen exine formation and pollen-pistil interactions in rice. Plant Sci. 2016, 253, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, L.; Wu, J.; Liu, Y.; Gong, X.; Xu, J.; Lin, D.; Dong, Y. The rice pentatricopeptide repeat gene TCD10 is needed for chloroplast development under cold stress. Rice 2016, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Bai, Y.; Wu, G.; Zou, S.; Chen, Y.; Gao, C.; Tang, D. Simultaneous modification of three homoeologs of Ta EDR 1 by genome editing enhances powdery mildew resistance in wheat. Plant J. 2017, 91, 714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frye, C.A.; Tang, D.; Innes, R.W. Negative regulation of defense responses in plants by a conserved MAPKK kinase. Proc. Natl. Acad. Sci. USA 2001, 98, 373. [Google Scholar] [CrossRef]
- Chandrasekaran, J.; Brumin, M.; Wolf, D.; Leibman, D.; Klap, C.; Pearlsman, M.; Sherman, A.; Arazi, T.; Gal-On, A. Development of broad virus resistance in non-transgenic cucumber using CRISPR/Cas9 technology. Mol. Plant Pathol. 2016, 17, 1140. [Google Scholar] [CrossRef] [Green Version]
- Pyott, D.E.; Sheehan, E.; Molnar, A. Engineering of CRISPR/Cas9-mediated potyvirus resistance in transgene-free Arabidopsis plants. Mol. Plant Pathol. 2016, 17, 1276. [Google Scholar] [CrossRef] [Green Version]
- Hu, Y.; Zhang, J.; Jia, H.; Sosso, D.; Li, T.; Frommer, W.B.; Yang, B.; White, F.F.; Wang, N.; Jones, J.B. Lateral organ boundaries 1 is a disease susceptibility gene for citrus bacterial canker disease. Proc. Natl. Acad. Sci. USA 2014, 111, E521. [Google Scholar] [CrossRef] [Green Version]
- Jia, H.; Zhang, Y.; Orbović, V.; Xu, J.; White, F.F.; Jones, J.B.; Wang, N. Genome editing of the disease susceptibility gene Cs LOB 1 in citrus confers resistance to citrus canker. Plant Biotechnol. J. 2017, 15, 817. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Humphry, M.; Consonni, C.; Panstruga, R. mlo-based powdery mildew immunity: Silver bullet or simply non-host resistance? Mol. Plant Pathol. 2006, 7, 605. [Google Scholar] [CrossRef] [PubMed]
- Nekrasov, V.; Wang, C.; Win, J.; Lanz, C.; Weigel, D.; Kamoun, S. Rapid generation of a transgene-free powdery mildew resistant tomato by genome deletion. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Cheng, X.; Shan, Q.; Zhang, Y.; Liu, J.; Gao, C.; Qiu, J.-L. Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew. Nat. Biotechnol. 2014, 32, 947. [Google Scholar] [CrossRef]
- Chaparro-Garcia, A.; Kamoun, S.; Nekrasov, V. Boosting plant immunity with CRISPR/Cas. Genome Biol. 2015, 16, 254. [Google Scholar] [CrossRef] [Green Version]
- Ji, X.; Zhang, H.; Zhang, Y.; Wang, Y.; Gao, C. Establishing a CRISPR-Cas-like immune system conferring DNA virus resistance in plants. Nat. Plants 2015, 1, 15144. [Google Scholar] [CrossRef]
- Macovei, A.; Sevilla, N.R.; Cantos, C.; Jonson, G.B.; Slamet-Loedin, I.; Čermák, T.; Voytas, D.F.; Choi, I.R.; Chadha-Mohanty, P. Novel alleles of rice eIF4G generated by CRISPR/Cas9-targeted mutagenesis confer resistance to Rice tungro spherical virus. Plant Biotechnol. J. 2018, 16, 1918. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Tu, M.; Wang, D.; Liu, J.; Li, Y.; Li, Z.; Wang, Y.; Wang, X. CRISPR/Cas9-mediated efficient targeted mutagenesis in grape in the first generation. Plant Biotechnol. J. 2018, 16, 844. [Google Scholar] [CrossRef] [Green Version]
- Zaidi, S.S.-E.-A.; Tashkandi, M.; Mansoor, S.; Mahfouz, M.M. Engineering plant immunity: Using CRISPR/Cas9 to generate virus resistance. Front. Plant Sci. 2016, 7, 1673. [Google Scholar] [CrossRef] [Green Version]
- Zhang, T.; Zheng, Q.; Yi, X.; An, H.; Zhao, Y.; Ma, S.; Zhou, G. Establishing RNA virus resistance in plants by harnessing CRISPR immune system. Plant Biotechnol. J. 2018, 16, 1415. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.; Wang, C.; Liu, P.; Lei, C.; Hao, W.; Gao, Y.; Liu, Y.-G.; Zhao, K. Enhanced rice blast resistance by CRISPR/Cas9-targeted mutagenesis of the ERF transcription factor gene OsERF922. PLoS ONE 2016, 11, e0154027. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Won, S.Y.; Suh, S.C.; Kim, H.; Wing, R.; Jeong, Y.; Hwang, I.; Kim, M. The barley ERF-type transcription factor HvRAF confers enhanced pathogen resistance and salt tolerance in Arabidopsis. Planta 2007, 225, 575. [Google Scholar] [CrossRef] [PubMed]
- Garg, R.; Verma, M.; Agrawal, S.; Shankar, R.; Majee, M.; Jain, M. Deep transcriptome sequencing of wild halophyte rice, Porteresia coarctata, provides novel insights into the salinity and submergence tolerance factors. DNA Res. 2014, 21, 69. [Google Scholar] [CrossRef] [PubMed]
- Mickelbart, M.V.; Hasegawa, P.M.; Bailey-Serres, J. Genetic mechanisms of abiotic stress tolerance that translate to crop yield stability. Nat. Rev. Genet. 2015, 16, 237. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chen, L.; Li, R.; Zhao, R.; Yang, M.; Sheng, J.; Shen, L. Reduced drought tolerance by CRISPR/Cas9-mediated SlMAPK3 mutagenesis in tomato plants. J. Agric. Food Chem. 2017, 65, 8674. [Google Scholar] [CrossRef]
- Lou, D.; Wang, H.; Liang, G.; Yu, D. OsSAPK2 confers abscisic acid sensitivity and tolerance to drought stress in rice. Front. Plant Sci. 2017, 8, 993. [Google Scholar] [CrossRef] [Green Version]
- Jia, Y.; Ding, Y.; Shi, Y.; Zhang, X.; Gong, Z.; Yang, S. The cbfs triple mutants reveal the essential functions of CBF s in cold acclimation and allow the definition of CBF regulons in Arabidopsis. New Phytol. 2016, 212, 345. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.-J.; Su, C.-T.; Lin, C.-H.; Huang, G.-J.; Lin, Y.-H. Expression of sweet potato cysteine protease SPCP2 altered developmental characteristics and stress responses in transgenic Arabidopsis plants. J. Plant Physiol. 2010, 167, 838. [Google Scholar] [CrossRef]
- Liu, H.; Hu, M.; Wang, Q.; Cheng, L.; Zhang, Z. Role of papain-like cysteine proteases in plant development. Front. Plant Sci. 2018, 9, 1717. [Google Scholar] [CrossRef] [Green Version]
- Zang, Q.-W.; Wang, C.-X.; Li, X.-Y.; Guo, Z.-A.; Jing, R.-L.; Zhao, J.; Chang, X.-P. Isolation and characterization of a gene encoding a polyethylene glycol-induced cysteine protease in common wheat. J. Biosci. 2010, 35, 379. [Google Scholar] [CrossRef]
- Antoniou, C.; Chatzimichail, G.; Xenofontos, R.; Pavlou, J.J.; Panagiotou, E.; Christou, A.; Fotopoulos, V. Melatonin systemically ameliorates drought stress-induced damage in Medicago sativa plants by modulating nitro-oxidative homeostasis and proline metabolism. J. Pineal Res. 2017, 62, e12401. [Google Scholar] [CrossRef] [PubMed]
- Byeon, Y.; Back, K. Low melatonin production by suppression of either serotonin N-acetyltransferase or N-acetylserotoninmethyltransferase in rice causes seedling growth retardation with yield penalty, abiotic stress susceptibility, and enhanced coleoptile growth under anoxic conditions. J. Pineal Res. 2016, 60, 348. [Google Scholar] [PubMed]
- Zhou, H.; He, M.; Li, J.; Chen, L.; Huang, Z.; Zheng, S.; Zhu, L.; Ni, E.; Jiang, D.; Zhao, B. Development of commercial thermo-sensitive genic male sterile rice accelerates hybrid rice breeding using the CRISPR/Cas9-mediated TMS5 editing system. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rauf, S.; da Silva, J.T.; Khan, A.A.; Naveed, A. Consequences of plant breeding on genetic diversity. Int. J. Plant Breed. 2010, 4, 1–21. [Google Scholar]
- Abdelrahman, M.; Al-Sadi, A.M.; Pour-Aboughadareh, A.; Burritt, D.J.; Tran, L.-S.P. Genome editing using CRISPR/Cas9–targeted mutagenesis: An opportunity for yield improvements of crop plants grown under environmental stresses. Plant Physiol. Biochem. 2018, 131, 31. [Google Scholar] [CrossRef]
- Tang, L.; Mao, B.; Li, Y.; Lv, Q.; Zhang, L.; Chen, C.; He, H.; Wang, W.; Zeng, X.; Shao, Y. Knockout of OsNramp5 using the CRISPR/Cas9 system produces low Cd-accumulating indica rice without compromising yield. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.-Z.; Chen, M.-X.; Yu, L.-J.; Xie, L.-J.; Yuan, L.-B.; Qi, H.; Xiao, M.; Guo, W.; Chen, Z.; Yi, K. OsARM1, an R2R3 MYB transcription factor, is involved in regulation of the response to arsenic stress in rice. Front. Plant Sci. 2017, 8, 1868. [Google Scholar] [CrossRef] [Green Version]
- Lu, H.P.; Liu, S.M.; Xu, S.L.; Chen, W.Y.; Zhou, X.; Tan, Y.Y.; Huang, J.Z.; Shu, Q.Y. CRISPR-S: An active interference element for a rapid and inexpensive selection of genome-edited, transgene-free rice plants. Plant Biotechnol. J. 2017, 15, 1371. [Google Scholar] [CrossRef]
- Kumar, N.; Galli, M.; Ordon, J.; Stuttmann, J.; Kogel, K.H.; Imani, J. Further analysis of barley MORC 1 using a highly efficient RNA-guided Cas9 gene-editing system. Plant Biotechnol. J. 2018, 16, 1892. [Google Scholar] [CrossRef] [Green Version]
- Peng, A.; Chen, S.; Lei, T.; Xu, L.; He, Y.; Wu, L.; Yao, L.; Zou, X. Engineering canker-resistant plants through CRISPR/Cas9-targeted editing of the susceptibility gene Cs LOB 1 promoter in citrus. Plant Biotechnol. J. 2017, 15, 1509. [Google Scholar] [CrossRef] [Green Version]
- Čermák, T.; Baltes, N.J.; Čegan, R.; Zhang, Y.; Voytas, D.F. High-frequency, precise modification of the tomato genome. Genome Biol. 2015, 16, 232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fiehn, O. Metabolomics—The link between genotypes and phenotypes. In Functional Genomics; Springer: Berlin/Heidelberg, Germany, 2002; p. 155. [Google Scholar]
- Belhaj, K.; Chaparro-Garcia, A.; Kamoun, S.; Patron, N.J.; Nekrasov, V. Editing plant genomes with CRISPR/Cas9. Curr. Opin. Biotechnol. 2015, 32, 76. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.L.; Liu, Y.G. CRISPR/Cas9-based genome editing systems and the analysis of targeted genome mutations in plants. Hereditas 2016, 38, 118. [Google Scholar] [PubMed]
- Ma, X.; Zhu, Q.; Chen, Y.; Liu, Y.G. CRISPR/Cas9 platforms for genome editing in plants: Developments and applications. Mol. Plant 2016, 9, 961. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gilbert, L.A.; Larson, M.H.; Morsut, L.; Liu, Z.; Brar, G.A.; Torres, S.E.; Stern-Ginossar, N.; Brandman, O.; Whitehead, E.H.; Doudna, J.A.; et al. CRISPR-mediated modular RNA-guided regulation of transcription in eukaryotes. Cell 2013, 154, 442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lowder, L.G.; Zhang, D.; Baltes, N.J.; Paul, J.W., 3rd; Tang, X.; Zheng, X.; Voytas, D.F.; Hsieh, T.F.; Zhang, Y.; Qi, Y. A CRISPR/Cas9 toolbox for multiplexed plant genome editing and transcriptional regulation. Plant Physiol. 2015, 169, 971. [Google Scholar] [CrossRef] [Green Version]
- O’Connell, M.R.; Oakes, B.L.; Sternberg, S.H.; East-Seletsky, A.; Kaplan, M.; Doudna, J.A. Programmable RNA recognition and cleavage by CRISPR/Cas9. Nature 2014, 516, 263. [Google Scholar] [CrossRef]
| Name | Species | Database Resource | URL |
|---|---|---|---|
| TAIR | Mainly for Arabidopsis thaliana | Whole genome | http://www.arabidopsis.org |
| 1001genomes | Arabidopsis thaliana | Whole genome | http://www.1001genomes.org |
| Phytozome | Numerous | Whole genome | http://www.phytozome.net |
| NCBI | Numerous | Whole genome | http://www.ncbi.nlm.nih.gov |
| Cottongen | Gossypium spp. | Whole genome and breeding | http://www.cottongen.org |
| Soybean breeders toolbox | Glycine max | Whole genome | http://www.soybase.org |
| MaizeGDB | Zea mays | Whole genome | http://www.maizegdb.org |
| RAP-DB | Oryza sativa | Whole genome | http://rapdb.dna.affrc.go.jp |
| PlantGDB | Numerous | Whole genome | http://www.plantgdb.org |
| IWGSC | Triticum aestivum | Whole genome | http://www.wheatgenome.org |
| Gramene | Numerous | Whole genome | http://www.gramene.org |
| Ensemblplants | Numerous | Whole genome | http://plants.ensembl.org |
| KEGG | Numerous | Whole genome | http://www.genome.jp/kegg/genome/plant.html |
| Graingenes | Numerous | Whole genome | http://wheat.pw.usda.gov/GG2/index.shtm |
| PMN | Numerous | Metabolomics | http://www.plantcyc.org |
| CSB.DB | Arabidopsis thaliana | Metabolomics | http://csbdb.mpimp-golm.mpg.de/csbdb/gmd/gmd.html |
| PRIMe | Arabidopsis thaliana | Metabolomics | http://prime.psc.riken.jp/lcms/ms2tview/ms2tview.html |
| AFGN | Arabidopsis thaliana | Gene expression | https://www.deutsche-botanische-gesellschaft.de/en/about-us/afgn |
| OryzaExpress | Oryza sativa | Gene expression | http://plantomics.mind.meiji.ac.jp/OryzaExpress/ |
| RGAP | Oryza spp. | Gene expression | http://rice.plantbiology.msu.edu |
| CottonFGD | Gossypium spp. | Gene expression | http://www.cottonfgd.org |
| Genevestigator | Numerous | Gene expression | http://genevestigator.com |
| TriFLDB | Triticum aestivum | Gene expression | https://bigd.big.ac.cn/databasecommons/database/id/3452 |
| BAR | Numerous | Gene expression | http://bar.utoronto.ca/welcome.htm |
| NOBLE | Medicago truncatula | Gene expression | http://mtgea.noble.org/v2 |
| Uniprot | Numerous | Proteomics | http://www.uniprot.org/proteomes/ |
| RICE PROTEOME | Oryza sativa | Proteomics | http://gene64.dna.affrc.go.jp/RPD |
| Proteomics database | Arabidopsis thaliana | Proteomics | http://proteomics.arabidopsis.info |
| SUBA | Arabidopsis thaliana | Proteomics | http://www.suba.bcs.uwa.edu.au/ |
| AGRIS | Arabidopsis thaliana | Transcription factor | http://arabidopsis.med.ohio-state.edu |
| PlantTFDB | Numerous | Transcription factor | http://planttfdb.gao-lab.org/ |
| LegumeTFDB | Lotus japonicas, Medicago truncatula, Glycine max | Transcription factor | http://legumetfdb.psc.riken.jp |
| Grassius | Zea mays, Oryza sativa, Sorghum bicolor | Transcription factor | http://grassius.org/ |
| TRIM | Oryza sativa | Mutants | http://trim.sinica.edu.tw |
| RMD | Oryza spp. | Mutants | http://rmd.ncpgr.cn/ |
| ABRC | Arabidopsis thaliana | Mutants | http://abrc.osu.edu |
| NASC | Arabidopsis thaliana | Mutants | http://arabidopsis.org.uk/home.html |
| Fox Hunting | Numerous | Mutants | http://nazunafox.psc.database.riken.jp |
| SIGnAL | Arabidopsis thaliana | Mutants | http://signal.salk.edu |
| Species | Traits | Target Genes | Reference |
|---|---|---|---|
| Abiotic stresses | |||
| Rice | Improved resistance to arsenic stress | ARM1 | [108] |
| Depletion of Cd into grain | LCT1 | [109] | |
| Depletion of Cd into grain | Nramp5 | [107] | |
| Drought tolerance | SAPK2 | [97] | |
| Tomato | Drought tolerance | SIMAPK3 | [96] |
| Maize | Drought tolerance | ARGOS8 | [69] |
| Arabidopsis | Cold tolerance | CBF1 CBF2 | [99] |
| Biotic stresses | |||
| Arabidopsis | Resistance to turnip mosaic virus | eIF (iso)4E | [80] |
| Wheat | Improved resistance to powdery mildew | TaMLO | [85] |
| Improved resistance to powdery mildew | EDR1 | [77] | |
| Rice | Increased resistance to blast fungus | OsERF922 | [92] |
| Increased resistance to tungro spherical virus | eIF4G | [88] | |
| Barley | Improved resistance to fungal pathogens | MORC1 | [110] |
| Orange | Improved resistance to citrus canker | CsLOB1 | [111] |
| Tomato | Improved resistance to powdery mildew | Mlo1 | [84] |
| Anthocyanin biosynthesis | ANT1 | [112] | |
| Grape | Improved resistance to Botrytis cinerea | WRKY52 | [89] |
| Cucumber | Virus resistance | eIF4E | [79] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Razzaq, M.K.; Aleem, M.; Mansoor, S.; Khan, M.A.; Rauf, S.; Iqbal, S.; Siddique, K.H.M. Omics and CRISPR-Cas9 Approaches for Molecular Insight, Functional Gene Analysis, and Stress Tolerance Development in Crops. Int. J. Mol. Sci. 2021, 22, 1292. https://doi.org/10.3390/ijms22031292
Razzaq MK, Aleem M, Mansoor S, Khan MA, Rauf S, Iqbal S, Siddique KHM. Omics and CRISPR-Cas9 Approaches for Molecular Insight, Functional Gene Analysis, and Stress Tolerance Development in Crops. International Journal of Molecular Sciences. 2021; 22(3):1292. https://doi.org/10.3390/ijms22031292
Chicago/Turabian StyleRazzaq, Muhammad Khuram, Muqadas Aleem, Shahid Mansoor, Mueen Alam Khan, Saeed Rauf, Shahid Iqbal, and Kadambot H. M. Siddique. 2021. "Omics and CRISPR-Cas9 Approaches for Molecular Insight, Functional Gene Analysis, and Stress Tolerance Development in Crops" International Journal of Molecular Sciences 22, no. 3: 1292. https://doi.org/10.3390/ijms22031292





