1. Introduction
The placenta, which functions as a fetomaternal organ, is composed of both fetal tissues and maternal tissues by structure. The amnion membrane, chorion membrane, and umbilical cord are the fetal parts of the placenta. These tissues have been shown to support restoration of several tissue types, including bone, tendon, and skin tissue [
1,
2,
3]. The observed plethora of biological effects are due to the activation of epithelialization and neovascularization, suppression of inflammation and scarring, pain reduction, and inherent antimicrobial properties [
4,
5,
6,
7,
8,
9,
10]. The mesenchymal stromal cells (MSCs), rich extracellular matrix, key cytokines, and growth factors in the tissues contribute to these functional properties. There is wide acceptance that the extracellular matrix, cytokines, and growth factors are mainly derived from the secretion of the MSCs within the tissues. Therefore, the MSCs residing in the tissues at least partially dominate the functional properties of the amnion membrane, chorion membrane, and umbilical cord.
MSCs have the potential for use in regenerative medicine and have been investigated in a number of clinical trials for currently untreatable diseases, such as myocardial infarction, stroke, graft-versus-host disease, bone and cartilage defects, and autoimmune diseases [
11,
12,
13,
14]. Since bone-marrow-derived MSCs (BM-MSCs) were first reported, a number of studies have shown similar cell types in a wide range of tissues, including the placental fetal membrane, umbilical cord, adipose tissue, dental tissue, skin, hair follicles, and tonsils. Recently, comparative studies showed that MSCs derived from fetal placental tissues are more prone to differentiating into osteoblasts than BM-MSCs are. BM-MSCs and adipose-tissue-derived MSCs (AD-MSCs) represent the optimal stem cell source for tissue engineering and regenerative medicine [
15,
16]. However, as sources of MSCs, placental tissues present several advantages for clinical usage compared with other MSC sources [
17,
18,
19,
20,
21,
22]. These include the absence of ethical concerns, their uncomplicated harvest protocol, avoidance any invasive procedure on the donors, and, importantly, the young cellular age of the donors. In addition, the factors secreted by placental MSCs are more adept at angiogenesis, cell proliferation, differentiation, cell survival, immunomodulation, and collagen degradation than those of AD-MSCs [
23,
24].
MSCs can be isolated from all layers of a full-term placenta, including the amniotic membrane, chorionic membrane, chorionic villi, decidua, and umbilical cord [
25,
26,
27,
28]. Comparing MSCs from different parts of the placenta will facilitate the selection of appropriate MSCs for clinical applications. Several studies have isolated and investigated the characteristics of these cells after in vitro culture. However, controversial results between different research groups have rendered the outcomes inconclusive. Soncini et al. (2007) showed that both amnion MSCs and chorion MSCs are comparable with regard to multi-lineage differentiation [
29]. However, Choi et al. (2017) showed that chorion MSCs have higher multi-lineage differentiation potential, and both chorion MSCs and chorionic villi MSCs tend to have higher proliferative potentials than amnion MSCs [
30]. Yamahara et al. (2014) showed that amnion and chorion MSCs secrete differential soluble factors and showed differences in their angiogenic and immune-suppressive functions [
31]. However, Wu et al. (2018) showed that MSCs from the umbilical cord and amnion exhibited superior growth potential and higher anti-inflammatory properties than chorion MSCs [
32], whereas, chorion MSCs displayed potential pro-angiogenic activity due to the higher secretion of HGF and VCAM-1 [
32]. Moreover, Huang et al. (2019) showed that chorion MSCs demonstrated significantly stronger proliferation ability and immunomodulatory properties in comparison with MSCs derived from other layers of the placenta [
26]. Kim et al. (2011) concluded that chorion MSCs, rather than chorionic villi MSCs, are useful sources of cells for appropriate clinical applications in the treatment of various degenerative diseases [
33]. Kannaiyan et al. (2018) showed that MSCs from the amnion and chorionic villi were capable of differentiating into cardiac progenitor MSCs and had comparable angiogenic capacity [
34]. Taken together, further investigation is needed to better understand the functional properties of MSCs derived from different sources of placental tissues, and this will potentially impact future clinical applications. This may be achieved by identifying the molecular pathways and cytokine profiles of these MSCs.
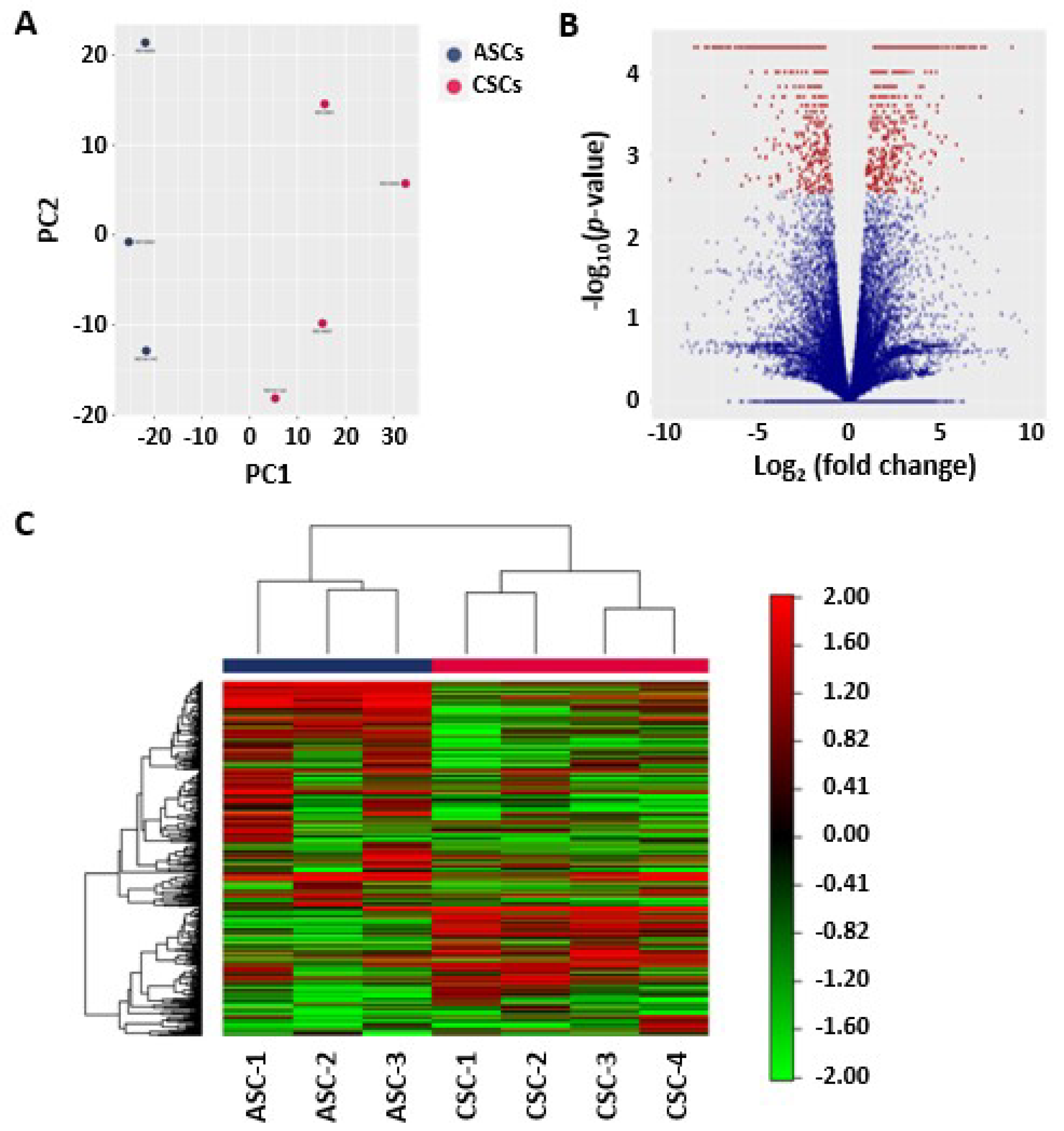
Among the MSCs derived from different layers of the placental tissue, amnion and chorion MSCs have advantages as cell sources, as a large quantity of cells can be obtained because of their size and the minimal contamination with the original maternal cells. To comprehensively compare amnion and chorion MSCs at the molecular and signaling levels, in the present study, we used next-generation sequencing technology to explore the differences between freshly isolated amnion MSCs and chorion MSCs at the molecular level. The morphology and immunophenotype of the isolated MSCs were analyzed. The cytokine secretion profiles were quantitatively evaluated with an enzyme-linked immunosorbent assay and multiplex cytokine arrays. We further investigated the anti-inflammatory and pro-angiogenic functions of the MSCs derived from both the amnion and chorion. Our data suggest that amnion MSCs exhibit strong anti-inflammatory functions, whereas chorion MSCs show increased pro-angiogenic signaling and increased secretion of angiogenic factors.
3. Discussion
The human placenta contains non-immunogenic cells, growth factors, cytokines, and an extracellular matrix (ECM), making it a potent solution for a variety of indications due to its angiogenic, anti-inflammatory, anti-oxidative, anti-microbial, and anti-fibrotic properties. The MSCs, rich extracellular matrix, key cytokines, and growth factors in the tissues contribute to the functions of these properties. It is believed that the extracellular matrix, cytokines, and growth factors are mainly from the secretions of the MSCs within the tissues. Therefore, the MSCs residing in the tissues at least partially dominate the functional properties of the amnion membrane, chorion membrane, and umbilical cord. MSCs have been shown to have regenerative functions, including angiogenic, anti-inflammatory, anti-oxidative, anti-microbial, and anti-fibrotic properties. MSCs can be isolated from all layers of a full-term placenta, including from the amniotic membrane, chorionic membrane, chorionic villi, decidua, and umbilical cord. Comparing MSCs from different parts of the placenta will facilitate the selection of appropriate MSCs for clinical applications. Several studies have isolated and investigated the characteristics of these cells after in vitro culture. However, a comparison of the freshly isolated cells before putting them in culture has not been reported [
36]. Here, for the first time, we systematically characterized the gene expression profiles of directly isolated human amniotic stromal cells and human chorionic stromal cells from term pregnancies.
We identified differentially expressed genes, including the highly expressed genes in ASCs,
LMO3 and
UTY, and the highly expressed genes in CSCs,
NAA11, BIRC7, APOD, HOXA11, HOXA13, MKRN4P, RGPD1, and
NR2F1. Among these genes,
LMO3 is a member of the LIM-domain-only (LMO) protein family. Studies have reported that
LMO3 is involved in the transcriptional regulation of specific target genes in collaboration with other transcription factors, such as p53, as well as the regulation of cell invasion and proliferation [
37,
38,
39].
UTY is the human ubiquitously transcribed tetratricopeptide repeat gene, and it encodes histone demethylase, which is involved in protein–protein interactions, cell proliferation, and differentiation [
40]. Evidence of the UTY protein at the protein level predicted intracellular and secreted proteins. Interestingly, both the
LMO3 and
UTY genes are related to gene epigenetic regulations, and their high expression in ACSs but not in CSCs suggests that distinct machineries of epigenetic regulation exist in ACSs and CSCs.
NAA11 expression was detected only in the testis and placenta obtained from normal human subjects [
41]. Here, we showed that
NAA11 is only expressed in CSCs; however, no detectable expression was found in ASCs. Therefore,
NAA11 may serve as a biomarker for identifying the origin of CSCs from ASCs. It has been shown that
Hoxa11 and
Hoxa13 suppression result in a significant reduction of Sox9 and collagen type 2 expression, suggesting a reduction in chondrogenic potential [
42,
43,
44]. High expression of
HOXA11 and
HOXA13 in CSCs demonstrates that these cells have higher chondrogenic potential than ASCs. It is important to note that during our data analysis, long non-coding RNA (lncRNA) identification and annotation were at a preliminary stage [
45,
46]. Therefore, further investigation is necessary in order to identify potential novel lncRNAs.
Alternative splicing is an important posttranscriptional process that enables a single gene to produce multiple distinct transcripts, namely isoforms. These isoforms carry different biological properties that are different in catalytic ability, subcellular localization, or protein interaction. We identified the 10 most significantly differentially expressed isoforms, including both known and novel isoforms. These isoforms included
CXCL6, FOXF1-AS1, HGF, MAEL, NPTX1, HGF, ATP2A3, DPT, POSTN, and a novel isoform (Isoform ID: XLOC_040015). Among them,
HGF, MAEL, NPTX1, ATP2A3, and
DPT were highly expressed in CSCs, and
CXCL6, FOXF1-AS1, POSTN, and the novel isoform (Isoform ID: XLOC_040015) were highly expressed in ASCs. The naturally occurring isoforms of HGF, NK1, and NK2 are expressed in human tissues during development and in normal adults [
47]. Studies in cell culture and in transgenic animals have suggested that NK1 is capable of recapitulating normal HGF signaling and biological activities, while NK2 appears to be an antagonist for HGF-induced cellular proliferation. NK2 expression is increased relative to full-length HGF in human fibrotic organ diseases, and it is possible that NK2 may play a role in the failure of normal repair [
47]. The normal biological roles of the truncated HGF isoforms remain to be determined. Further understanding of the normal functions of these isoforms, similarly to the novel isoforms identified in this study, may provide insight into the characteristic properties of CSCs and ASCs.
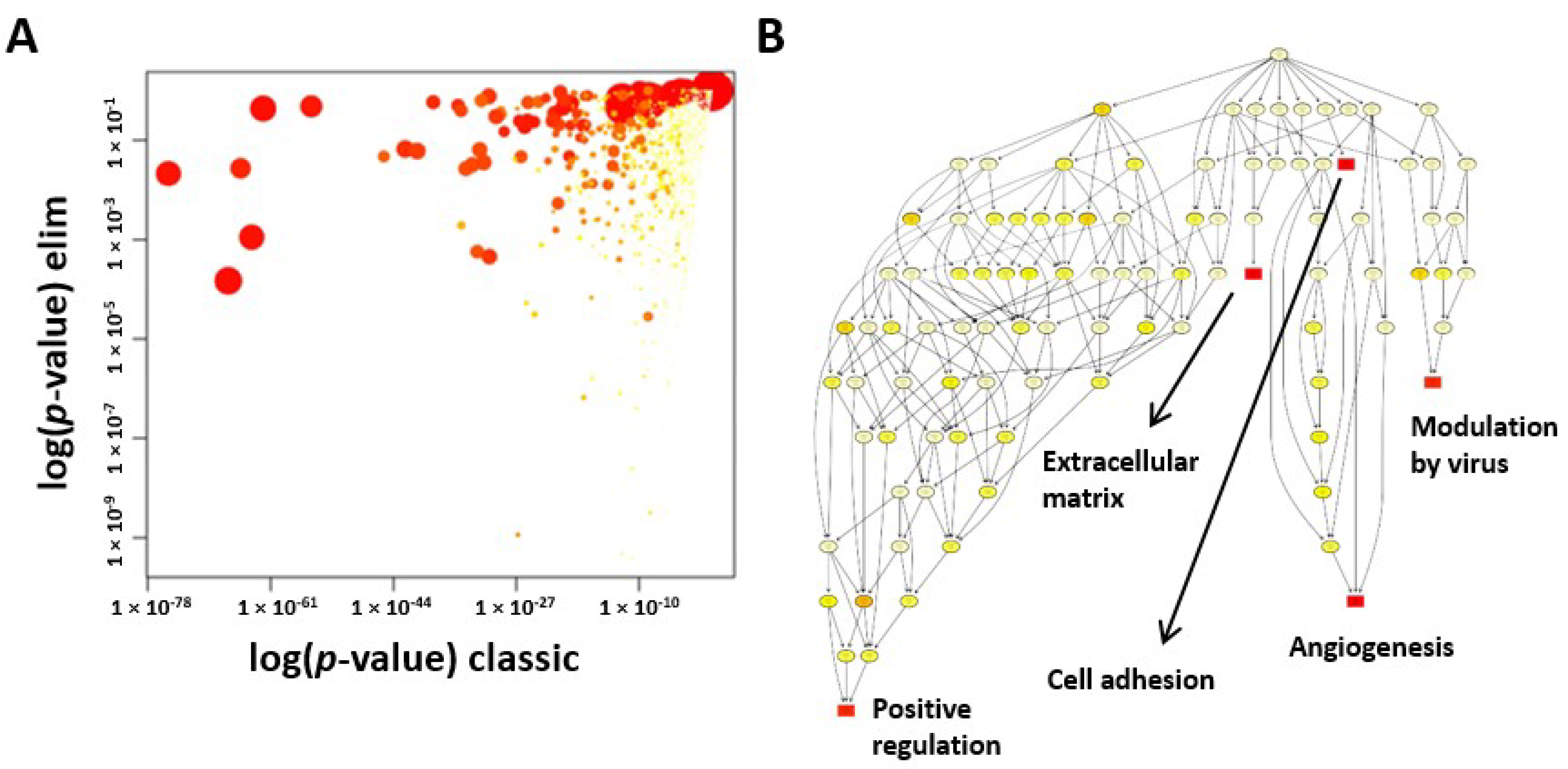
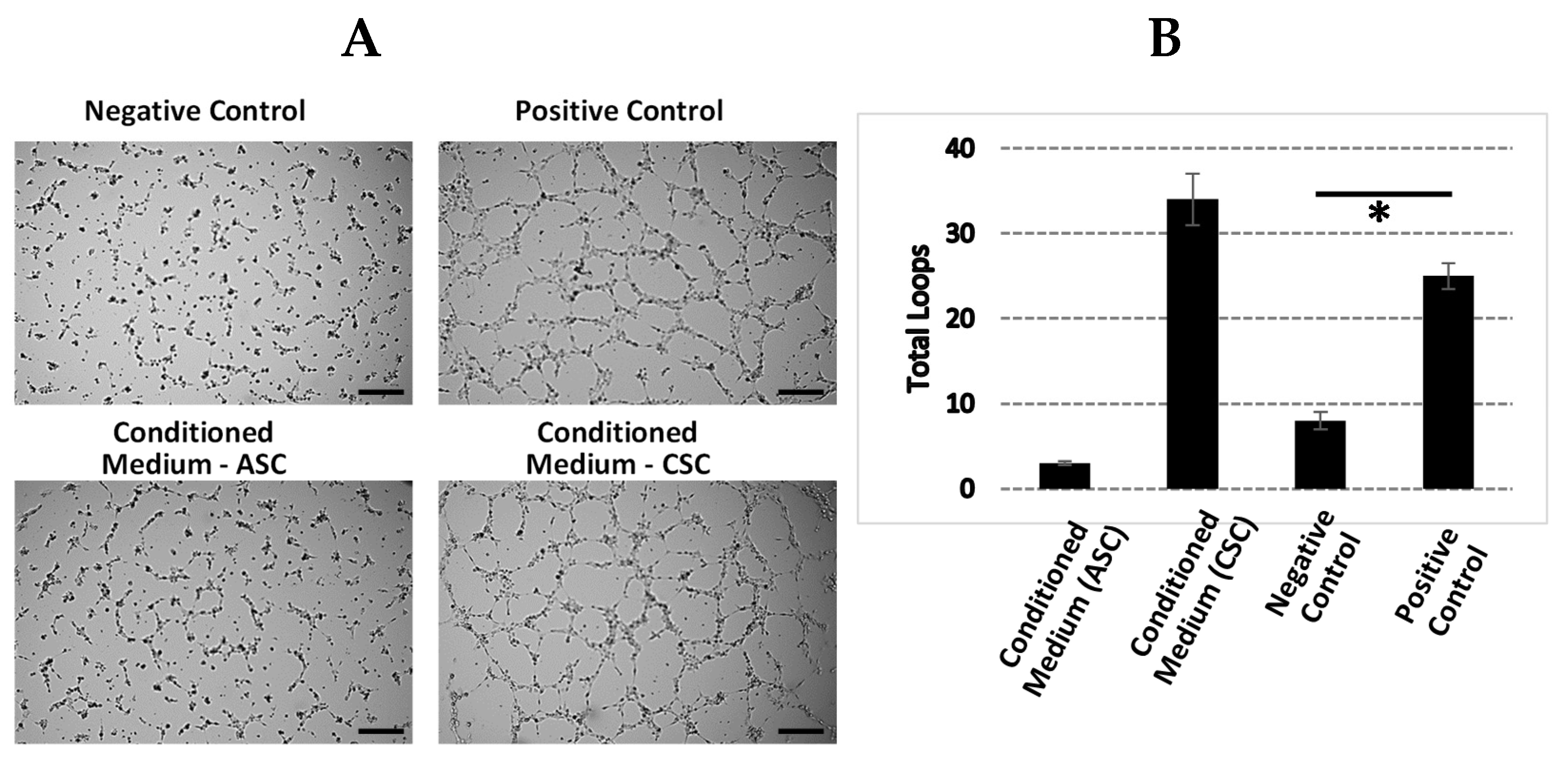
Our GO analysis showed that the GO terms of extracellular matrix, angiogenesis, and cell adhesion were significantly enriched in CSCs. We further explored the angiogenic function of CSCs due to its importance in regenerative medicine. Increased levels of angiogenin, VEGFA, HGF, and bFGF protein and enhanced tube formation in vitro proved that CSCs exhibited a strong angiogenic function. Although inflammation did not fall into the top 10 GO terms, we realized that many key genes and/or their isoforms (
Table 1,
Table 2 and
Table 3) that were involved in inflammation were significantly expressed between ASCs and CSCs, such as CXCL6, IL-1B, HOXA11, HOXA13, HGF, CSF1, and NR2F1, suggesting that the different regulation machineries for the secretion of anti-inflammatory factors exist in these two populations. Therefore, we investigated the anti-inflammatory response in this study in terms of inhibition of an inflammatory factor, TNF-a, and enhancement of secretion of an anti-inflammatory factor, TSG-6. Interestingly, ASCs induced two-fold higher TSG-6 secretion than CSCs. Furthermore, among the GO terms that were found to be different, there were also negative regulators of cell proliferation, although our preliminary observation did not show different proliferation rates between ASCs and CSCs. Further investigation is warranted in order to confirm these observations due to the importance of the physiology of these cells, such as in cell proliferation, senescence, and apoptosis.
It is important to note that freshly isolated heterogenous cell populations were used in this study. Further in vitro and in vivo experiments with phenotypic and morphologic characteristics, as well as differential capacities, are warranted in order to characterize the MSCs isolated from amnions and chorions. It will be of special interest to describe the differentiation and progenitor potential of these amnion and chorion MSCs, as well as the full spectrum of secreted factors.
Mesenchymal stromal cells derived from the fetal placenta have emerged as promising resources for regenerative medicine. Our study demonstrated that CSCs have superior angiogenic potential, but ASCs exhibit increased anti-inflammatory properties. Therefore, both CSCs and ASCs could potentially be the sources of MSCs for regenerative medicine based on the required functional properties. The current results add to the rapidly expanding field of interest in the potential research and therapeutic applications of the MSCs derived from fetal placenta membranes.
4. Materials and Methods
4.1. Tissue Procurement and Ethics Statement
Placental tissue with procurement and ethics statements that were collected from eligible donors after obtaining written consent was purchased from The National Disease Research Interchange (Philadelphia, PA, USA) and Cord Blood America, Inc. (Las Vegas, NV, USA) [
48].
4.2. Placental Tissue Processing and Isolation of the Amniotic Membrane and the Chorionic Membrane
Placental research tissues were aseptically processed as described previously [
49,
50]. The amniotic membrane was physically separated from the chorionic membrane. The chorionic membrane (CM) was separated from the decidua via blunt dissection, followed by a wash in anticoagulant citrate dextrose, solution A (ACD-A) (Fenwal Inc., Lake Zurich, IL, USA). The CM was then subjected to enzymatic treatment with dispase solution (1:20 dilution) (Corning Inc, Corning, NY, USA) to allow for separation of the stromal layer of CM from the trophoblast layer, the choriodecidua, and the decidua. The stromal layer of CM was washed with saline and mechanically cleaned to remove residual blood and trophoblasts. The membrane was then incubated for 24–48 h in an antibiotic cocktail solution as described before [
48]. The CM was rinsed in Dulbecco’s phosphate buffered saline (DPBS), and then CSCs were isolated through enzymatic membrane digestion.
4.3. Isolation of ASCs and CSCs from the Amniotic Membrane and Chorionic Membrane
To isolate CSCs from the chorionic membrane, the membrane was incubated in 1 mg/mL type II collagenase (Worthington Biochemical Cporporation, Lakewood, NJ, USA) at 37 °C, followed by filtering in 100, 70, and 30 μm nylon strainers. The cell digest was centrifuged (Beckman Coulter GS-6R) at 1500 rpm to pellet the CSCs, and a cell count was obtained using a Cellometer (Nexcelom Bioscience, Lawrence, MA, USA). To isolate ASCs from the amniotic membrane, the amniotic membrane was first treated with 0.25% trypsin for 25 min to remove amniotic epithelial cells. After the membrane was washed twice with DPBS, ASC isolation was performed in the same way as the CSC isolation mentioned above. Freshly isolated, uncultured ASCs and CSCs were used for RNA-seq, and cells with passages below five were used for the entire in vitro study.
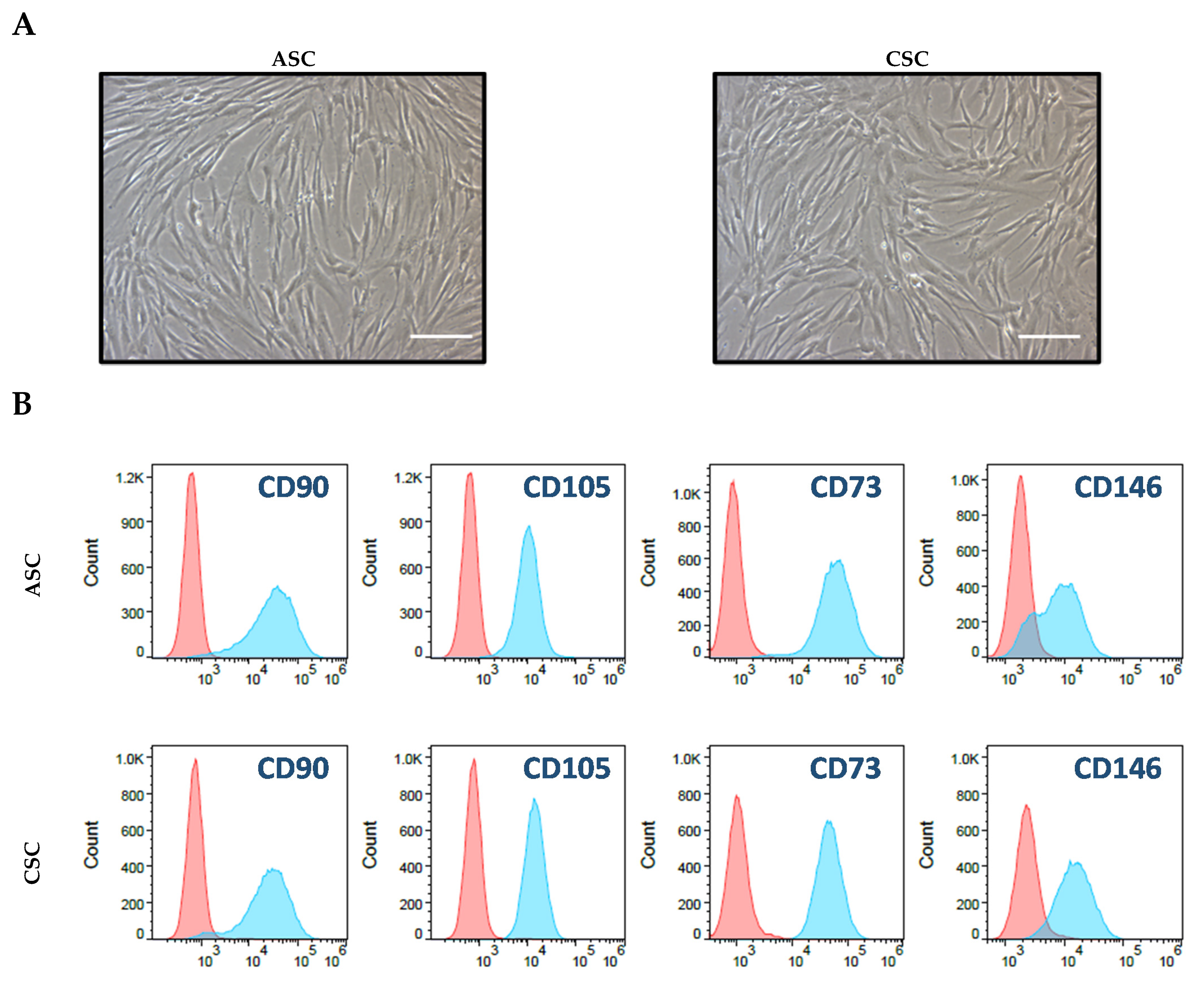
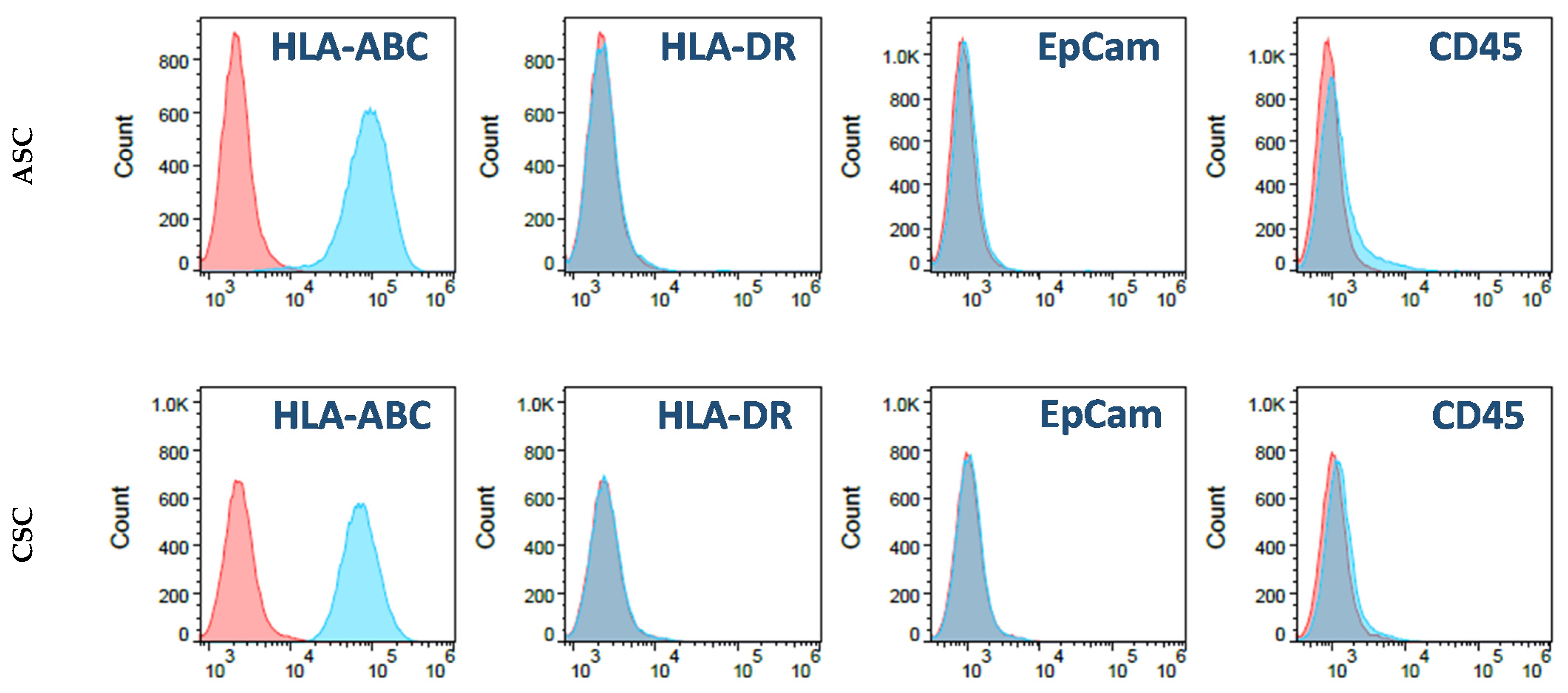
4.4. Cell Culture and Characteristic Analysis through Flow Cytometry
A total 5 × 105 of ASCs and CSCs were cultured in T175 tissue-culture-treated plates containing 30 mL low glucose DMEM medium plus 5% FBS. The culture medium was changed every other day. For the flow cytometry analysis, first-passage ASCs and CSCs were digested with TypLE. After washing with PBS, the cells were resuspended in a cell-staining solution (PBS plus 0.5% Bovine serum albumin (BSA)). The cells were then stained with fluorochrome-conjugated antibodies: IgG isotype-FITC, IgG isotype-PE, IgG isotype-PerCP, IgG isotype-APC, HLA-ABC-FITC, HLA-DR-FITC, CD73-PE, CD45-PE, EpCam-PE, CD146-PerCP, CD90-APC, and CD105-APC (BD Bioscience, San Jose, CA, USA). After 30 min, the stained cells were washed twice with the staining solution. Then, flow cytometry data were acquired by Accuri C6 (BD Bioscience) and analyzed with the FlowJo software (Version 10.7, BD Bioscience, San Jose, CA, USA).
4.5. RNA-Seq and Data Analysis
ASCs isolated from three donors and CSCs from four donors were immediately preserved in TRIzol reagent at –80 °C. RNA extraction and RNA-seq were carried out at Qiagen Genomic Services. The library preparation was done using a TruSeq
® Stranded mRNA Sample preparation kit (Illumina Inc., San Diego, CA, USA). The starting material (100 ng) of the total RNA was mRNA enriched using the oligo dT bead system. The isolated mRNA was subsequently fragmented using enzymatic fragmentation. Then, first-strand synthesis and second-strand synthesis were performed, and the double-stranded cDNA was purified (AMPure XP, Beckman Coulter, Sykesville, MD, USA). The cDNA was end repaired and 3′ adenylated, Illumina sequencing adaptors were ligated onto the fragments’ ends, and the library was purified (AMPure XP). The mRNA stranded libraries were pre-amplified with polymerase chain reaction (PCR) and purified (AMPure XP). The libraries’ size distribution was validated, and the quality was inspected on a Bioanalyzer 2100 or BioAnalyzer 4200 tapeStation (Agilent Technologies). High-quality libraries were pooled based on equimolar concentrations based on the Bioanalyzer Smear Analysis tool (Agilent Technologies, Wilmington, DE, USA). The library pools were quantified using quantitative PCR (qPCR), and the optimal concentration of the library pool was used to generate the clusters on the surface of a flow cell before sequencing on a NextSeq500 instrument (76 cycles) according to the manufacturer’s instructions (Illumina Inc.). To inspect the mapping in detail, we used the Integrative Genomics Viewer software to browse the BAM files [
51,
52]. The Tuxedo software package, including Bowtie2 [
53], Tophat [
54], and Cufflinks [
55,
56,
57], was used for the data analysis. In addition, we performed fragment bias correction, which sought to correct for sequence bias during library preparation [
58,
59,
60]. On average, 64.1 million reads were generated for each sample, and the average genome mapping rate was 96.3%. Only bases with an O-score above 30 were included in the downstream analysis. The mapped reads of the 500 genes that had the largest coefficients of variation based on the fragments per kilobase of transcripts per million (FPKM) [
61] were used for principal component analysis (PCA) and heat mapping with unsupervised clustering. Supervised clustering was used to generate a volcano plot and identify differently expressed genes and novel isoforms. Gene Ontology (GO) enrichment analysis for biological processes was used to investigate specific GO terms.
4.6. Characterization of Cytokine and Growth Factor Profiles in ASCs and CSCs
ASC and CSC lysates or culture supernatants, which were prepared through an overnight cell culture in a growth medium, were obtained from three donors for the growth factor and cytokine profile determinations. Cells were plated in six-well culture plates (3 × 105 cells per well) in DMEM growth media (supplemented with 10% fetal bovine serum (FBS) and 1% antibiotic/antimycotic). The supernatants were collected and analyzed using a Human Inflammation Panel, 37-Plex (Bio-Rad, Hercules, CA, USA) on a Bio-Plex MAGPIX plate reader (Bio-Rad, Hercules, CA). The test panels included growth factors that are important for anti-inflammation, wound healing, angiogenesis, antimicrobial activity, and osteogenesis: APRIL, BAFF, sCD30, sCD136, chitinase-3-like 1, sIL-6Rβ, IFNα2, IFNβ, IFN-γ, IL-2, sIL-6Rα, IL-8, IL-10, IL-11, IL12 (p40), IL-12 (p70), IL-19, IL-20, IL-22, IL-26, IL-27 (p28), IFNγ2, IFNγ1, IL-32, IL-34, IL-35, LIGHT, MMP-1, MMP-2, MMP-3, osteocalcin, osteopontin, pentraxin, sTNF-R1, sTNF-R2, TSLP, and TWEAK. PGE2, angiogenin, FGF-2, VEGFA, and TSG-6 were analyzed with ELISA (R&D Systems, Menneapolis, MN, USA).
4.7. Secretion of Inflammatory Factors by Activated THP-1 cells
The anti-inflammatory capabilities of CSCs and ASCs were characterized with an inhibition assay of the TNF-α produced by LPS-activated THP-1 cells. A total of 500,000 THP-1 cells were plated per well in a 24-well plate, stimulated with 1 ug/mL of LPS in DMEM growth media, and co-cultured with either CSCs or conditioned media from an overnight culture of CSCs and ASCs in growth media. The untreated THP-1 and LPS-treated THP-1 cells represented the positive control and negative control, respectively. The culture was incubated overnight at 37 °C with 5% CO2. After overnight stimulation and treatment, the cell media were collected, and the amount of TNF-α produced by the THP-1 cells after LPS treatment was compared to those of the CSC and ASC treatments and reported as percent inhibition vs. the control. TNF-α was quantified with Luminex and/or ELISA (R&D Systems).
4.8. HUVEC Tube Formation Induced by ASC and CSC Cell Culture Supernatants
The ASC and CSC culture supernatant dilutions were evaluated in an angiogenesis tube formation assay through stimulation of human umbilical vein endothelial cells (HUVECs; Lonza, Walkersville, MD, USA) to form closed, vessel-like structures. HUVECs were seeded with 1.2 × 104 cells/well on Matrigel (Corning, NY, USA)-coated u-Plate 96-culture wells (Ibidi, Grafelfing, Bayern, Germany). The negative and positive control wells contained endothelial basal medium 2 (EBM-2; Lonza, Walkersville, MD, USA) and endothelial growth medium 2 (EGM-2; Lonza), respectively. The HUVECs were treated with 70 ul of ASC and CSC culture supernatants at 1:4 dilutions in DMEM only, DMEM growth media, and DMEM only. The HUVEC cultures were incubated for 6 h at 37 °C and 5% CO2, and images of representative fields were taken at 4× magnification. The magnitude of the tube formation was quantified with the Wimasis software by the number of closed structures.
4.9. Statistical Analysis
The results are presented as mean ± standard deviation (SD) for one representative experiment consisting of three donors and two biological replicates. Student’s T-test was used to determine the significance of differences between groups, whereby p < 0.05 was considered significant.