Anti-Müllerian Hormone, Growth Hormone, and Insulin-Like Growth Factor 1 Modulate the Migratory and Secretory Patterns of GnRH Neurons
Abstract
:1. Introduction
2. Results
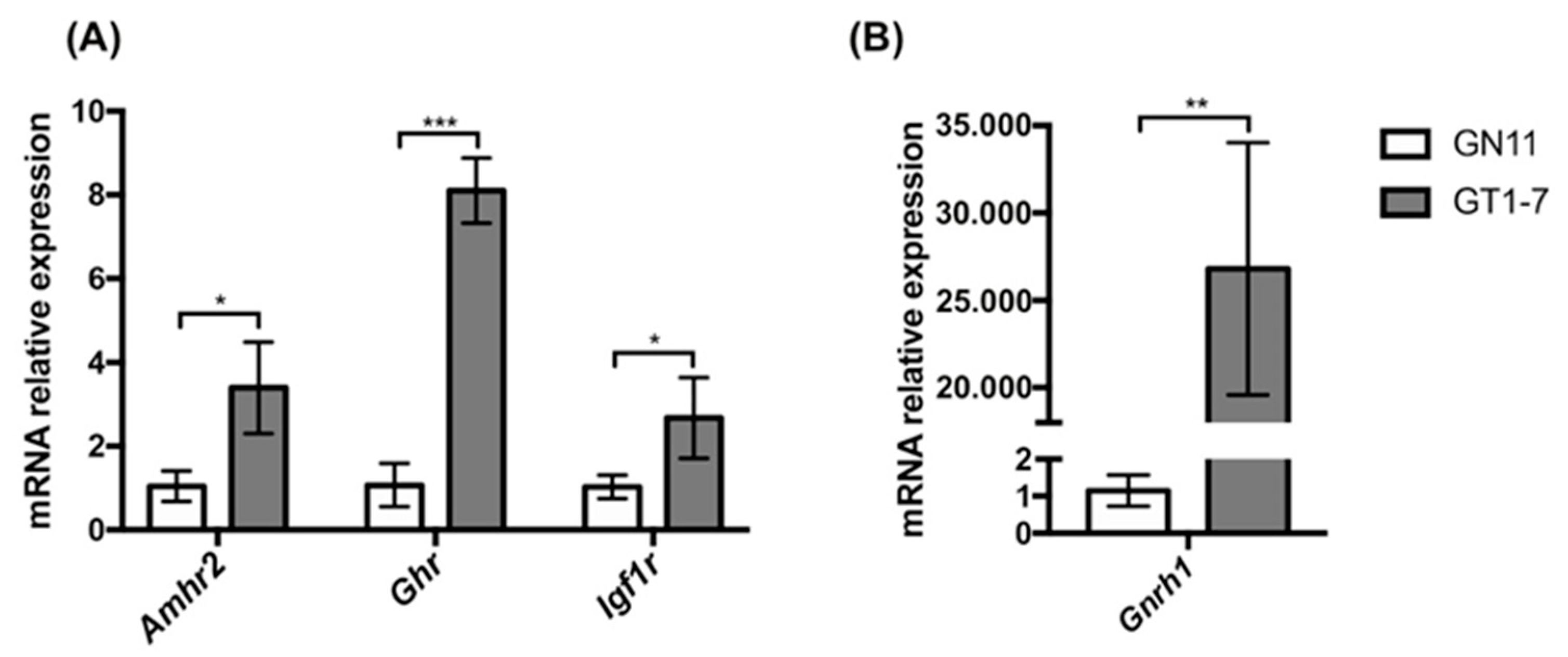
2.1. Immortalized GnRH Neurons (GN11 and GT1-7 Cell Lines) Express Different mRNA Levels for Amhr2, Ghr, and Igf1r
2.2. Effect of Treatment with AMH, GH, and IGF1 on GN11 Cell Migration
2.3. Effects of AMH, GH, and IGF1 on GN11 Cell Morphology
2.4. Effect of Treatment with AMH, GH, and IGF1 on GnRH Release by GT1-7 Cells
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Cell Cultures
4.3. RNA Extraction and Gene Expression Analyses
4.4. Chemomigration Assays
4.5. Cytoskeleton Labeling
4.6. GnRH Secretion Assay
4.7. Analysis of the Data
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Festa, A.; Umano, G.R.; Miraglia Del Giudice, E.; Grandone, A. Genetic Evaluation of Patients with Delayed Puberty and Congenital Hypogonadotropic Hypogonadism: Is it Worthy of Consideration? Front. Endocrinol. 2020, 11, 253. [Google Scholar] [CrossRef] [PubMed]
- Cate, R.L.; Mattaliano, R.J.; Hession, C.; Tizard, R.; Farber, N.M.; Cheung, A.; Ninfa, E.G.; Frey, A.Z.; Gash, D.J.; Chow, E.P. Isolation of the bovine and human genes for Müllerian inhibiting substance and expression of the human gene in animal cells. Cell 1986, 45, 685–698. [Google Scholar] [CrossRef]
- Condorelli, R.A.; Cannarella, R.; Calogero, A.E.; La Vignera, S. Evaluation of testicular function in prepubertal children. Endocrine 2018, 62, 274–280. [Google Scholar] [CrossRef]
- Cimino, I.; Casoni, F.; Liu, X.; Messina, A.; Parkash, J.; Jamin, S.P.; Catteau-Jonard, S.; Collier, F.; Baroncini, M.; Dewailly, D.; et al. Novel role for anti-Müllerian hormone in the regulation of GnRH neuron excitability and hormone secretion. Nat. Commun. 2016, 7, 10055. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kereilwe, O.; Pandey, K.; Borromeo, V.; Kadokawa, H. Anti-Müllerian hormone receptor type 2 is expressed in gonadotrophs of postpubertal heifers to control gonadotrophin secretion. Reprod. Fertil. Dev. 2018, 30, 1192–1203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garrel, G.; Denoyelle, C.; L’Hôte, D.; Picard, J.Y.; Teixeira, J.; Kaiser, U.B.; Laverrière, J.N.; Cohen-Tannoudji, J. GnRH Transactivates Human AMH Receptor Gene via Egr1 and FOXO1 in Gonadotrope Cells. Neuroendocrinology 2019, 108, 65–83. [Google Scholar] [CrossRef]
- Juul, A.; Skakkebæk, N.E. Why Do Normal Children Have Acromegalic Levels of IGF-I During Puberty? J. Clin. Endocrinol. Metab. 2019, 104, 2770–2776. [Google Scholar] [CrossRef] [PubMed]
- Daftary, S.S.; Gore, A.C. The hypothalamic insulin-like growth factor-1 receptor and its relationship to gonadotropin-releasing hormones neurones during postnatal development. J. Neuroendocrinol. 2004, 16, 160–169. [Google Scholar] [CrossRef]
- Anderson, R.A.; Zwain, I.H.; Arroyo, A.; Mellon, P.L.; Yen, S.S. The insulin-like growth factor system in the GT1-7 GnRH neuronal cell line. Neuroendocrinology 1999, 70, 353–359. [Google Scholar] [CrossRef]
- Longo, K.M.; Sun, Y.; Gore, A.C. Insulin-like growth factor-I effects on gonadotropin-releasing hormone biosynthesis in GT1-7 cells. Endocrinology 1998, 139, 1125–1132. [Google Scholar] [CrossRef]
- Onuma, T.A.; Ding, Y.; Abraham, E.; Zohar, Y.; Ando, H.; Duan, C. Regulation of temporal and spatial organization of newborn GnRH neurons by IGF signaling in zebrafish. J. Neurosci. 2011, 31, 11814–11824. [Google Scholar] [CrossRef] [PubMed]
- Magni, P.; Vettor, R.; Pagano, C.; Calcagno, A.; Beretta, E.; Messi, E.; Zanisi, M.; Martini, L.; Motta, M. Expression of a leptin receptor in immortalized gonadotropin-releasing hormone-secreting neurons. Endocrinology 1999, 140, 1581–1585. [Google Scholar] [CrossRef]
- Dozio, E.; Watanobe, H.; Ruscica, M.; Maggi, R.; Motta, M.; Magni, P. Expression of functional ciliary neurotrophic factor receptors in immortalized gonadotrophin-releasing hormone-secreting neurones. J. Neuroendocrinol. 2005, 17, 286–291. [Google Scholar] [CrossRef]
- Magni, P.; Dozio, E.; Ruscica, M.; Watanobe, H.; Cariboni, A.; Zaninetti, R.; Motta, M.; Maggi, R. Leukemia inhibitory factor induces the chemomigration of immortalized gonadotropin-releasing hormone neurons through the independent activation of the Janus kinase/signal transducer and activator of transcription 3, mitogen-activated protein kinase/extracellularly regulated kinase 1/2, and phosphatidylinositol 3-kinase/Akt signaling pathways. Mol. Endocrinol. 2007, 21, 1163–1174. [Google Scholar] [CrossRef] [Green Version]
- Olson, B.R.; Scott, D.C.; Wetsel, W.C.; Elliot, S.J.; Tomic, M.; Stojilkovic, S.; Nieman, L.K.; Wray, S. Effects of insulin-like growth factors I and II and insulin on the immortalized hypothalamic GTI-7 cell line. Neuroendocrinology 1995, 62, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Zhen, S.; Zakaria, M.; Wolfe, A.; Radovick, S. Regulation of gonadotropin-releasing hormone (GnRH) gene expression by insulin-like growth factor I in a cultured GnRH-expressing neuronal cell line. Mol. Endocrinol. 1997, 11, 1145–1155. [Google Scholar] [CrossRef] [PubMed]
- Ochoa, A.; Domenzáin, C.; Clapp, C.; Martínez de la Escalera, G. Differential effects of basic fibroblast growth factor, epidermal growth factor, transforming growth factor-alpha, and insulin-like growth factor-I on a hypothalamic gonadotropin-releasing hormone neuronal cell line. J. Neurosci. Res. 1997, 49, 739–749. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, H.; Gao, P.; Chen, J.; Yu, C.; Zong, C.; Lu, S.; Li, X.; Ma, X.; Liu, Y.; et al. The Effect of Growth Hormone on Lipid Accumulation or Maturation in Adipocytes. Cell. Physiol. Biochem. 2016, 39, 2135–2148. [Google Scholar] [CrossRef]
- Malone, S.A.; Papadakis, G.E.; Messina, A.; Mimouni, N.E.H.; Trova, S.; Imbernon, M.; Allet, C.; Cimino, I.; Acierno, J.; Cassatella, D.; et al. Defective AMH signaling disrupts GnRH neuron development and function and contributes to hypogonadotropic hypogonadism. Elife 2019, 8, e47198. [Google Scholar] [CrossRef]
- Imbeaud, S.; Belville, C.; Messika-Zeitoun, L.; Rey, R.; di Clemente, N.; Josso, N.; Picard, J.Y. A 27 base-pair deletion of the anti-müllerian type II receptor gene is the most common cause of the persistent müllerian duct syndrome. Hum. Mol. Genet. 1996, 5, 1269–1277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chemes, H.E. Infancy is not a quiescent period of testicular development. Int. J. Androl. 2001, 24, 2–7. [Google Scholar] [CrossRef]
- Edelsztein, N.Y.; Grinspon, R.P.; Schteingart, H.F.; Rey, R.A. Anti-Müllerian hormone as a marker of steroid and gonadotropin action in the testis of children and adolescents with disorders of the gonadal axis. Int. J. Pediatr. Endocrinol. 2016, 2016, 20. [Google Scholar] [CrossRef] [Green Version]
- van Rooij, I.A.; Broekmans, F.J.; te Velde, E.R.; Fauser, B.C.; Bancsi, L.F.; de Jong, F.H.; Themmen, A.P. Serum anti-Müllerian hormone levels: a novel measure of ovarian reserve. Hum. Reprod. 2002, 17, 3065–3071. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peñarrubia, J.; Fábregues, F.; Manau, D.; Creus, M.; Casals, G.; Casamitjana, R.; Carmona, F.; Vanrell, J.A.; Balasch, J. Basal and stimulation day 5 anti-Mullerian hormone serum concentrations as predictors of ovarian response and pregnancy in assisted reproductive technology cycles stimulated with gonadotropin-releasing hormone agonist--gonadotropin treatment. Hum. Reprod. 2005, 20, 915–922. [Google Scholar] [CrossRef] [PubMed]
- Barbotin, A.L.; Peigné, M.; Malone, S.A.; Giacobini, P. Emerging Roles of Anti-Müllerian Hormone in Hypothalamic-Pituitary Function. Neuroendocrinology 2019, 109, 218–229. [Google Scholar] [CrossRef]
- Coutant, R.; Biette-Demeneix, E.; Bouvattier, C.; Bouhours-Nouet, N.; Gatelais, F.; Dufresne, S.; Rouleau, S.; Lahlou, N. Baseline inhibin B and anti-Mullerian hormone measurements for diagnosis of hypogonadotropic hypogonadism (HH) in boys with delayed puberty. J. Clin. Endocrinol. Metab. 2010, 95, 5225–5232. [Google Scholar] [CrossRef] [Green Version]
- Adan, L.; Lechevalier, P.; Couto-Silva, A.C.; Boissan, M.; Trivin, C.; Brailly-Tabard, S.; Brauner, R. Plasma inhibin B and antimüllerian hormone concentrations in boys: discriminating between congenital hypogonadotropic hypogonadism and constitutional pubertal delay. Med. Sci. Monit. 2010, 16, CR511–CR517. [Google Scholar]
- Rohayem, J.; Nieschlag, E.; Kliesch, S.; Zitzmann, M. Inhibin B, AMH, but not INSL3, IGF1 or DHEAS support differentiation between constitutional delay of growth and puberty and hypogonadotropic hypogonadism. Andrology 2015, 3, 882–887. [Google Scholar] [CrossRef]
- Albanese, A.; Stanhope, R. Treatment of growth delay in boys with isolated growth hormone deficiency. Eur. J. Endocrinol. 1994, 130, 65–69. [Google Scholar] [CrossRef]
- Laron, Z.; Sarel, R. Penis and testicular size in patients with growth hormone insufficency. Acta Endocrinol. 1970, 63, 625–633. [Google Scholar] [CrossRef]
- Laron, Z.; Sarel, R.; Pertzelan, A. Puberty in Laron type dwarfism. Eur. J. Pediatr. 1980, 134, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Guevara-Aguirre, J.; Rosenbloom, A.L.; Fielder, P.J.; Diamond, F.B.; Rosenfeld, R.G. Growth hormone receptor deficiency in Ecuador: clinical and biochemical phenotype in two populations. J. Clin. Endocrinol. Metab. 1993, 76, 417–423. [Google Scholar] [CrossRef] [Green Version]
- Zhou, C.; Niu, Y.; Xu, H.; Li, Z.; Wang, T.; Yang, W.; Wang, S.; Wang, D.W.; Liu, J. Mutation profiles and clinical characteristics of Chinese males with isolated hypogonadotropic hypogonadism. Fertil. Steril. 2018, 110, 486–495.e5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oleari, R.; Andrè, V.; Lettieri, A.; Tahir, S.; Roth, L.; Paganoni, A.; Eberini, I.; Parravicini, C.; Scagliotti, V.; Cotellessa, L.; et al. A novel SEMA3G mutation in two siblings affected by syndromic GnRH deficiency. Neuroendocrinology 2020. [Google Scholar] [CrossRef] [PubMed]
- Lettieri, A.; Borgo, C.; Zanieri, L.; D’Amore, C.; Oleari, R.; Paganoni, A.; Pinna, L.A.; Cariboni, A.; Salvi, M. Protein Kinase CK2 Subunits Differentially Perturb the Adhesion and Migration of GN11 Cells: A Model of Immature Migrating Neurons. Int. J. Mol. Sci. 2019, 20, 5951. [Google Scholar] [CrossRef] [PubMed] [Green Version]









| Treatment | Percentage of Cells with an Elongated Morphology |
|---|---|
| PBS | 8.3% |
| PBS HCl | 5.9% |
| AMH (50 ng/mL) | 29.2% |
| AMH (150 ng/mL) | 38.6% |
| GH (100 ng/mL) | 36.3% |
| GH (300 ng/mL) | 52.3% |
| IGF1 (30 ng/mL) | 35.8% |
| IGF1 (100 ng/mL) | 48.2% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cannarella, R.; Paganoni, A.J.J.; Cicolari, S.; Oleari, R.; Condorelli, R.A.; La Vignera, S.; Cariboni, A.; Calogero, A.E.; Magni, P. Anti-Müllerian Hormone, Growth Hormone, and Insulin-Like Growth Factor 1 Modulate the Migratory and Secretory Patterns of GnRH Neurons. Int. J. Mol. Sci. 2021, 22, 2445. https://doi.org/10.3390/ijms22052445
Cannarella R, Paganoni AJJ, Cicolari S, Oleari R, Condorelli RA, La Vignera S, Cariboni A, Calogero AE, Magni P. Anti-Müllerian Hormone, Growth Hormone, and Insulin-Like Growth Factor 1 Modulate the Migratory and Secretory Patterns of GnRH Neurons. International Journal of Molecular Sciences. 2021; 22(5):2445. https://doi.org/10.3390/ijms22052445
Chicago/Turabian StyleCannarella, Rossella, Alyssa J. J. Paganoni, Stefania Cicolari, Roberto Oleari, Rosita A. Condorelli, Sandro La Vignera, Anna Cariboni, Aldo E. Calogero, and Paolo Magni. 2021. "Anti-Müllerian Hormone, Growth Hormone, and Insulin-Like Growth Factor 1 Modulate the Migratory and Secretory Patterns of GnRH Neurons" International Journal of Molecular Sciences 22, no. 5: 2445. https://doi.org/10.3390/ijms22052445











