Neuroprotective Effect of Aurantio-Obtusin, a Putative Vasopressin V1A Receptor Antagonist, on Transient Forebrain Ischemia Mice Model
Abstract
:1. Introduction
2. Results
2.1. Functional Effect of Test Compounds via cAMP Modulation
2.2. Functional Effect of Test Compounds via Intracellular Ca2+ Ion Modulation
2.3. Molecular Docking Simulation
2.4. Drug-Likeness and ADME Prediction
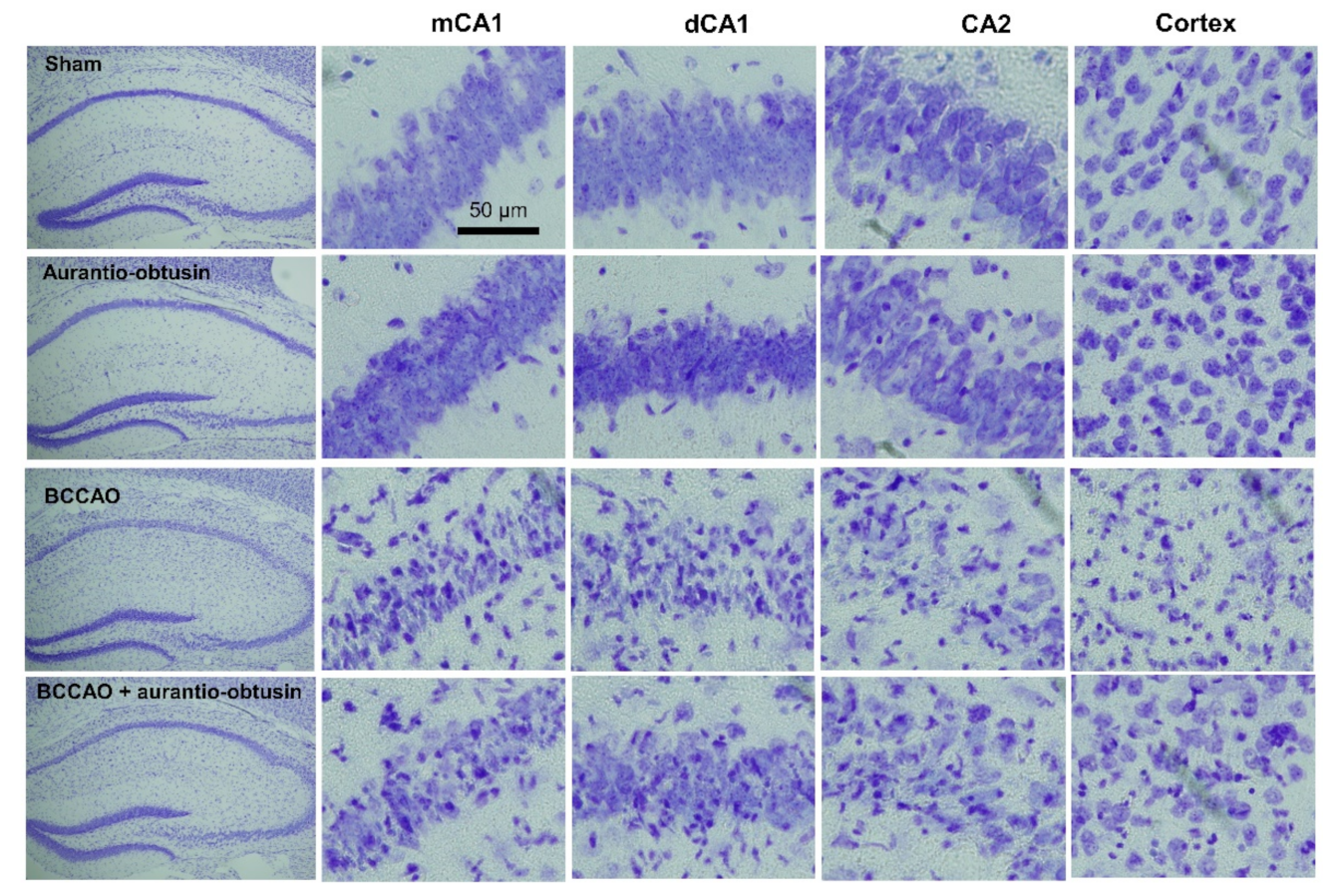
2.5. Neuroprotective Effect of Aurantio-Obtusin
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Plant Material
4.3. Extraction, Fractionation and Isolation of Compounds
4.4. GPCR Functional Assay
4.5. Measurement of cAMP Level
4.6. Measurement of Intracellular Ca2+ Ion Concentration
4.7. Homology Modeling of V1AR
4.8. Molecular Docking
4.9. Drug-Likeness and ADME Prediction
4.10. Animal
4.11. Transient Forebrain Ischemia Surgery
4.12. Passive Avoidance Test
4.13. Slices Preparation and Nissl Staining
4.14. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jobst, A.; Dehning, S.; Ruf, S.; Notz, T.; Buchheim, A.; Henning-Fast, K.; Meißner, D.; Meyer, S.; Bondy, B.; Müller, N. Oxytocin and vasopressin levels are decreased in the plasma of male schizophrenia patients. Acta Neuropsychiatr. 2014, 26, 347–355. [Google Scholar] [CrossRef] [Green Version]
- Yang, S.Y.; Cho, S.-C.; Yoo, H.J.; Cho, I.H.; Park, M.; Kim, B.-N.; Kim, J.-W.; Shin, M.-S.; Park, T.-W.; Son, J.-W. Association study between single nucleotide polymorphisms in promoter region of AVPR1A and Korean autism spectrum disorders. Neurosci. Lett. 2010, 479, 197–200. [Google Scholar] [CrossRef]
- LoParo, D.; Waldman, I. The oxytocin receptor gene (OXTR) is associated with autism spectrum disorder: A meta-analysis. Mol. Psychiatry 2015, 20, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Schnider, P.; Bissantz, C.; Bruns, A.; Dolente, C.; Goetschi, E.; Jakob-Roetne, R.; Künnecke, B.; Mueggler, T.; Muster, W.; Parrott, N.; et al. Discovery of balovaptan, a vasopressin 1a receptor antagonist for the treatment of autism spectrum disorder. J. Med. Chem. 2020, 63, 1511–1525. [Google Scholar] [CrossRef]
- Yuen, K.W.; Garner, J.P.; Carson, D.S.; Keller, J.; Lembke, A.; Hyde, S.A.; Kenna, H.A.; Tennakoon, L.; Schatzberg, A.F.; Parker, K.J. Plasma oxytocin concentrations are lower in depressed vs. healthy control women and are independent of cortisol. J. Psychiatr. Res. 2014, 51, 30–36. [Google Scholar] [CrossRef]
- Simon, N.G.; Guillon, C.; Fabio, K.; Heindel, N.D.; Lu, S.-F.; Miller, M.; Ferris, C.F.; Brownstein, M.J.; Garripa, C.; Koppel, G.A. Vasopressin antagonists as anxiolytics and antidepressants: Recent developments. Recent Pat. CNS Drug Discov. 2008, 3, 77–93. [Google Scholar] [CrossRef]
- Albers, H.E. The regulation of social recognition, social communication and aggression: Vasopressin in the social behavior neural network. Horm. Behav. 2012, 61, 283–292. [Google Scholar] [CrossRef]
- Benarroch, E.E. Oxytocin and vasopressin: Social neuropeptides with complex neuromodulatory functions. Neurology 2013, 80, 1521–1528. [Google Scholar] [CrossRef] [PubMed]
- Bielsky, I.F.; Hu, S.-B.; Szegda, K.L.; Westphal, H.; Young, L.J. Profound impairment in social recognition and reduction in anxiety-like behavior in vasopressin V1a receptor knockout mice. Neuropsychopharmacology 2003, 29, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Ferris, C.F.; Lu, S.-F.; Messenger, T.; Guillon, C.D.; Heindel, N.; Miller, M.; Koppel, G.; Robert Bruns, F.; Simon, N.G. Orally active vasopressin V1a receptor antagonist, SRX251, selectively blocks aggressive behavior. Pharmacol. Biochem. Behav. 2006, 83, 169–174. [Google Scholar] [CrossRef]
- Jorgensen, W.T.; Gulliver, D.W.; Katte, T.A.; Werry, E.L.; Reekie, T.A.; Connor, M.; Kassiou, M. Conformationally rigid derivatives of WAY-267,464: Synthesis and pharmacology at the human oxytocin and vasopressin-1a receptors. Eur. J. Med. Chem. 2018, 143, 1644–1656. [Google Scholar] [CrossRef]
- Vakili, A.; Kataoka, H.; Plesnila, N. Role of arginine vasopressin V1 and V2 receptors for brain damage after transient focal cerebral ischemia. J. Cereb. Blood Flow Metab. 2005, 25, 1012–1019. [Google Scholar] [CrossRef] [Green Version]
- Dou, D.; Chen, L.; Di, H.; Song, Z.; Li, S.; Bu, X.; Dai, Q.; Wang, S.; Li, J.X.; Zhu, X. Vasopressin augments TNBS-induced colitis through enteric neuronal V1a receptor-mediated COX-2-dependent prostaglandin release from mast cells in mice. Neurogastroenterol. Motil. 2019, 31, e13493. [Google Scholar] [CrossRef]
- Linas, S.L.; Anderson, R.J.; Guggenheim, S.J.; Robertson, G.L.; Berl, T.; Dickmann, D.C. Role of vasopressin in impaired water excretion in conscious rats with experimental cirrhosis. Kidney Int. 1981, 20, 173–180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.; Luo, G.; Jiang, J.; Ma, T.; Lin, X.; Jiang, L.; Cheng, J.; Tao, R. Signaling through hepatocyte vasopressin receptor 1 protects mouse liver from ischemia-reperfusion injury. Oncotarget 2016, 7, 69276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Narayen, G.; Mandal, S.N. Vasopressin receptor antagonists and their role in clinical medicine. Indian J. Endocrinol. Metab. 2012, 16, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Share, L. Role of vasopressin in cardiovascular regulation. Physiol. Rev. 1988, 68, 1248–1284. [Google Scholar] [CrossRef]
- Aoyagi, T.; Koshimizu, T.-A.; Tanoue, A. Vasopressin regulation of blood pressure and volume: Findings from V1a receptor–deficient mice. Kidney Int. 2009, 76, 1035–1039. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morel, A.; O’Carroll, A.-M.; Brownstein, M.J.; Lolaft, S.J. Molecular cloning and expression of a rat V1a arginine vasopressin receptor. Nature 1992, 356, 523–526. [Google Scholar] [CrossRef]
- Palin, K.; Moreau, M.L.; Sauvant, J.; Orcel, H.; Nadjar, A.; Duvoid-Guillou, A.; Dudit, J.; Rabié, A.; Moos, F. Interleukin-6 activates arginine vasopressin neurons in the supraoptic nucleus during immune challenge in rats. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E1289–E1299. [Google Scholar] [CrossRef]
- Raber, J.; Bloom, F.E. IL-2 induces vasopressin release from the hypothalamus and the amygdala: Role of nitric oxide-mediated signaling. J. Neurosci. 1994, 14, 6187–6195. [Google Scholar] [CrossRef] [Green Version]
- Bolognani, F.; Del Valle Rubido, M.; Squassante, L.; Wandel, C.; Derks, M.; Murtagh, L.; Sevigny, J.; Khwaja, O.; Umbricht, D.; Fontoura, P. A phase 2 clinical trial of a vasopressin V1a receptor antagonist shows improved adaptive behaviors in men with autism spectrum disorder. Sci. Transl. Med. 2019, 11, eaat7838. [Google Scholar] [CrossRef]
- Koshimizu, T.-A.; Nasa, Y.; Tanoue, A.; Oikawa, R.; Kawahara, Y.; Kiyono, Y.; Adachi, T.; Tanaka, T.; Kuwaki, T.; Mori, T.; et al. V1a vasopressin receptors maintain normal blood pressure by regulating circulating blood volume and baroreflex sensitivity. Proc. Natl. Acad. Sci. USA 2006, 103, 7807–7812. [Google Scholar] [CrossRef] [Green Version]
- Huijuan, S.; Zhuju, W.; Liying, T. Simultaneous determination of 4 major components in semen cassiae obtusifoline by HPLC. Zhongguo Zhong Yao Za Zhi 2011, 36, 1327–1329. [Google Scholar] [CrossRef]
- Kim, M.; Lim, S.J.; Lee, H.-J.; Nho, C.W. Cassia tora seed extract and its active compound aurantio-obtusin inhibit allergic responses in IgE-mediated mast cells and anaphylactic models. J. Agric. Food Chem. 2015, 63, 9037–9046. [Google Scholar] [CrossRef]
- Hyun, S.K.; Lee, H.; Kang, S.S.; Chung, H.Y.; Choi, J.S. Inhibitory activities of Cassia tora and its anthraquinone constituents on angiotensin-converting enzyme. Phytother. Res. 2009, 23, 178–184. [Google Scholar] [CrossRef]
- Byun, E.; Jeong, G.-S.; An, R.-B.; Li, B.; Lee, D.-S.; Ko, E.-K.; Yoon, K.-H.; Kim, Y.-C. Hepatoprotective compounds of Cassiae Semen on tacrine-induced cytotoxicity in Hep G2 cells. Korean J. Pharmacogn. 2007, 38, 400–402. [Google Scholar]
- Choi, J.S.; Lee, H.J.; Park, K.-Y.; Ha, J.-O.; Kang, S.S. In vitro antimutagenic effects of anthraquinone aglycones and naphthopyrone glycosides from Cassia tora. Planta Med. 1997, 63, 11–14. [Google Scholar] [CrossRef]
- Vishnuprasad, C.N.; Tsuchiya, T.; Kanegasaki, S.; Kim, J.H.; Han, S.S. Aurantio-obtusin stimulates chemotactic migration and differentiation of MC3T3-E1 osteoblast cells. Planta Med. 2014, 80, 544–549. [Google Scholar] [CrossRef]
- El-Halawany, A.M.; Chung, M.H.; Nakamura, N.; Ma, C.-M.; Nishihara, T.; Hattori, M. Estrogenic and anti-estrogenic activities of Cassia tora phenolic constituents. Chem. Pharm. Bull. 2007, 55, 1476–1482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, H.A.; Ali, M.Y.; Jung, H.J.; Jeong, H.O.; Chung, H.Y.; Choi, J.S. Inhibitory activities of major anthraquinones and other constituents from Cassia obtusifolia against β-secretase and cholinesterases. J. Ethnopharmacol. 2016, 191, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Yoon, B.H.; Kim, Y.-W.; Lee, S.; Shin, B.Y.; Jung, J.W.; Kim, H.J.; Lee, Y.S.; Choi, J.S.; Kim, S.Y.; et al. The seed extract of Cassia obtusifolia ameliorates learning and memory impairments induced by scopolamine or transient cerebral hypoperfusion in mice. J. Pharmacol. Sci. 2007, 105, 82–93. [Google Scholar] [CrossRef] [Green Version]
- Shrestha, S.; Seong, S.H.; Paudel, P.; Jung, H.A.; Choi, J.S. Structure related inhibition of enzyme systems in cholinesterases and BACE1 in vitro by naturally occurring naphthopyrone and its glycosides isolated from Cassia obtusifolia. Molecules 2017, 23, 69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yi, J.H.; Park, H.J.; Lee, S.; Jung, J.W.; Kim, B.C.; Lee, Y.C.; Ryu, J.H.; Kim, D.H. Cassia obtusifolia seed ameliorates amyloid β-induced synaptic dysfunction through anti-inflammatory and Akt/GSK-3β pathways. J. Ethnopharmacol. 2016, 178, 50–57. [Google Scholar] [CrossRef]
- Ravi, S.K.; Narasingappa, R.B.; Joshi, C.G.; Girish, T.K.; Vincent, B. Neuroprotective effects of Cassia tora against paraquat-induced neurodegeneration: Relevance for Parkinson’s disease. Nat. Prod. Res. 2018, 32, 1476–1480. [Google Scholar] [CrossRef]
- Jung, H.A.; Ali, M.Y.; Choi, J.S. Promising inhibitory effects of anthraquinones, naphthopyrone, and naphthalene glycosides, from Cassia obtusifolia on α-glucosidase and human protein tyrosine phosphatases 1B. Molecules 2017, 22, 28. [Google Scholar] [CrossRef]
- Shrestha, S.; Paudel, P.; Seong, S.H.; Min, B.S.; Seo, E.K.; Jung, H.A.; Choi, J.S. Two new naphthalenic lactone glycosides from Cassia obtusifolia L. seeds. Arch. Pharm. Res. 2018, 41, 737–742. [Google Scholar] [CrossRef]
- Ali, M.Y.; Jannat, S.; Jung, H.A.; Min, B.S.; Paudel, P.; Choi, J.S. Hepatoprotective effect of Cassia obtusifolia seed extract and constituents against oxidative damage induced by tert-butyl hydroperoxide in human hepatic HepG2 cells. J. Food Biochem. 2018, 42, e12439. [Google Scholar] [CrossRef] [Green Version]
- Paudel, P.; Jung, H.A.; Choi, J.S. Anthraquinone and naphthopyrone glycosides from Cassia obtusifolia seeds mediate hepatoprotection via Nrf2-mediated HO-1 activation and MAPK modulation. Arch. Pharm. Res. 2018, 41, 677–689. [Google Scholar] [CrossRef]
- Yen, G.-C.; Chen, H.-W.; Duh, P.-D. Extraction and identification of an antioxidative component from Jue Ming Zi (Cassia tora L.). J. Agri. Food Chem. 1998, 46, 820–824. [Google Scholar] [CrossRef]
- Choi, J.S.; Lee, H.J.; Kang, S.S. Alatemin, cassiaside and rubrofusarin gentiobioside, radical scavenging principles from the seeds of Cassia tora on 1, 1-diphenyl-2-picrylhydrazyl (DPPH) radical. Arch. Pharm. Res. 1994, 17, 462–466. [Google Scholar] [CrossRef]
- Dong, X.; Fu, J.; Yin, X.; Yang, C.; Zhang, X.; Wang, W.; Du, X.; Wang, Q.; Ni, J. Cassiae semen: A review of its phytochemistry and pharmacology. Mol. Med. Rep. 2017, 16, 2331–2346. [Google Scholar] [CrossRef] [Green Version]
- Drever, B.D.; Anderson, W.G.; Riedel, G.; Kim, D.H.; Ryu, J.H.; Choi, D.-Y.; Platt, B. The seed extract of Cassia obtusifolia offers neuroprotection to mouse hippocampal cultures. J. Pharmacol. Sci. 2008, 107, 380–392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paudel, P.; Seong, S.H.; Shrestha, S.; Jung, H.A.; Choi, J.S. In vitro and in silico human monoamine oxidase inhibitory potential of anthraquinones, naphthopyrones, and naphthalenic lactones from Cassia obtusifolia Linn seeds. ACS Omega 2019, 4, 16139–16152. [Google Scholar] [CrossRef] [Green Version]
- Paudel, P.; Seong, S.H.; Fauzi, F.M.; Bender, A.; Jung, H.A.; Choi, J.S. Establishing GPCR targets of hMAO active anthraquinones from Cassia obtusifolia Linn seeds using in silico and in vitro methods. ACS Omega 2020, 5, 7705–7715. [Google Scholar] [CrossRef] [Green Version]
- Oprea, T.I. Property distribution of drug-related chemical databases. J. Comput. Aided Mol. Des. 2000, 14, 251–264. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 1997, 23, 3–25. [Google Scholar] [CrossRef]
- Misra, A.; Ganesh, S.; Shahiwala, A.; Shah, S.P. Drug delivery to the central nervous system: A review. J. Pharm. Pharm. Sci. 2003, 6, 252–273. [Google Scholar]
- Baell, J.B. Feeling Nature’s PAINS: Natural Products, Natural Product Drugs, and Pan Assay Interference Compounds (PAINS). J. Nat. Prod. 2016, 79, 616–628. [Google Scholar] [CrossRef]
- Baell, J.B.; Holloway, G.A. New substructure filters for removal of pan assay interference compounds (PAINS) from screening libraries and for their exclusion in bioassays. J. Med. Chem. 2010, 53, 2719–2740. [Google Scholar] [CrossRef] [Green Version]
- Yu, X.; Wei, L.-H.; Zhang, J.-K.; Chen, T.-R.; Jin, Q.; Wang, Y.-N.; Zhang, S.-J.; Dou, T.-Y.; Cao, Y.-F.; Guo, W.-Z.; et al. Anthraquinones from Cassiae semen as thrombin inhibitors: In vitro and in silico studies. Phytochemistry 2019, 165, 112025. [Google Scholar] [CrossRef]
- Pan, Z.-J.; Lu, Q.; Pan, L.; Xu, X.-X.; Pang, J.-S. Effects of water extracts from Cassia obtusifolia on blood pressure, blood lipid and renal structure in cold-induced hypertensive mice. Chin. J. Exp. Tradit. Med Formulae 2010, 16, 195–198. [Google Scholar]
- Hou, J.; Gu, Y.; Zhao, S.; Huo, M.; Wang, S.; Zhang, Y.; Qiao, Y.; Li, X. Anti-inflammatory effects of aurantio-obtusin from seed of Cassia obtusifolia L. through modulation of the NF-κB pathway. Molecules 2018, 23, 3093. [Google Scholar] [CrossRef] [Green Version]
- Mbatchou, V.C.; Tchouassi, D.P.; Dickson, R.A.; Annan, K.; Mensah, A.Y.; Amponsah, I.K.; Jacob, J.W.; Cheseto, X.; Habtemariam, S.; Torto, B. Mosquito larvicidal activity of Cassia tora seed extract and its key anthraquinones aurantio-obtusin and obtusin. Parasit. Vectors 2017, 10, 562. [Google Scholar] [CrossRef] [Green Version]
- Kwon, K.S.; Lee, J.H.; So, K.S.; Park, B.K.; Lim, H.; Choi, J.S.; Kim, H.P. Aurantio-obtusin, an anthraquinone from cassiae semen, ameliorates lung inflammatory responses. Phytother. Res. 2018, 32, 1537–1545. [Google Scholar] [CrossRef]
- Li, S.; Li, Q.; Lv, X.; Liao, L.; Yang, W.; Li, S.; Lu, P.; Zhu, D. Aurantio-obtusin relaxes systemic arteries through endothelial PI3K/AKT/eNOS-dependent signaling pathway in rats. J. Pharmacol. Sci. 2015, 128, 108–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riegger, G.A.; Liebau, G.; Kochsiek, K. Antidiuretic hormone in congestive heart failure. Am. J. Med. 1982, 72, 49–52. [Google Scholar] [CrossRef]
- Goldsmith, S.R.; Francis, G.S.; Cowley, A.W.; Levine, T.B.; Cohn, J.N. Increased plasma arginine vasopressin levels in patients with congestive heart failure. J. Am. Coll. Cardiol. 1983, 1, 1385–1390. [Google Scholar] [CrossRef] [Green Version]
- Creager, M.A.; Faxon, D.P.; Cutler, S.S.; Kohlmann, O.; Ryan, T.J.; Gavras, H. Contribution of vasopressin to vasoconstriction in patients with congestive heart failure: Comparison with the renin-angiotensin system and the sympathetic nervous system. J. Am. Coll. Cardiol. 1986, 7, 758–765. [Google Scholar] [CrossRef] [Green Version]
- Udelson, J.E.; Smith, W.B.; Hendrix, G.H.; Painchaud, C.A.; Ghazzi, M.; Thomas, I.; Ghali, J.K.; Selaru, P.; Chanoine, F.; Pressler, M.L. Acute hemodynamic effects of conivaptan, a dual V1A and V2 vasopressin receptor antagonist, in patients with advanced heart failure. Circulation 2001, 104, 2417–2423. [Google Scholar] [CrossRef] [Green Version]
- Pulsinelli, W.A. Selective neuronal vulnerability: Morphological and molecular characteristics. Prog. Brain Res. 1985, 63, 29–37. [Google Scholar]
- Kirino, T. Delayed neuronal death in the gerbil hippocampus following ischemia. Brain Res. 1982, 239, 57–69. [Google Scholar] [CrossRef]
- Pulsinelli, W.A.; Brierley, J.B.; Plum, F. Temporal profile of neuronal damage in a model of transient forebrain ischemia. Ann. Neurol. 1982, 11, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Li, J.; Tang, X.; Wang, Y.; Ma, Z.; Gao, Y. Metabolomics of aurantio-obtusin-induced hepatotoxicity in rats for discovery of potential biomarkers. Molecules 2019, 24, 3452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiao, S.-L.; Guan, L.-J.; Jiang, R.-F.; Wang, X.-G.; Li, X.; Cai, W. The metabolism and pharmacokinetics of rhein and aurantio-obtusin. Curr. Drug Metab. 2020, 21, 960–968. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.S.; Kim, Y.J.; Choi, J.S.; Lee, H.J.; Lee, C.J. Obtusifolin isolated from the seeds of Cassia obtusifolia regulates the gene expression and production of MUC5AC mucin in airway epithelial cells via affecting NF-κB pathway. Phytother. Res. 2019, 33, 919–928. [Google Scholar] [CrossRef] [PubMed]
- Paudel, P.; Seong, S.H.; Jung, H.A.; Choi, J.S. Characterizing fucoxanthin as a selective dopamine D3/D4 receptor agonist: Relevance to Parkinson’s disease. Chem. Biol. Interact. 2019, 310, 108757. [Google Scholar] [CrossRef] [PubMed]
- Paudel, P.; Seong, S.H.; Wu, S.; Park, S.; Jung, H.A.; Choi, J.S. Eckol as a potential therapeutic against neurodegenerative diseases targeting dopamine D3/D4 receptors. Mar. Drugs 2019, 17, 108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seong, S.H.; Paudel, P.; Choi, J.-W.; Ahn, D.H.; Nam, T.-J.; Jung, H.A.; Choi, J.S. Probing multi-target action of phlorotannins as new monoamine oxidase inhibitors and dopaminergic receptor modulators with the potential for treatment of neuronal disorders. Mar. Drugs 2019, 17, 377. [Google Scholar] [CrossRef] [Green Version]
- Xu, D.; Zhang, Y. Improving the physical realism and structural accuracy of protein models by a two-step atomic-level energy minimization. Biophys. J. 2011, 101, 2525–2534. [Google Scholar] [CrossRef] [Green Version]
- Goodsell, D.S.; Morris, G.M.; Olson, A.J. Automated docking of flexible ligands: Applications of AutoDock. J. Mol. Recognit. 1996, 9, 1–5. [Google Scholar] [CrossRef]





| Receptors | Rubrofusarin Gentiobioside | Cassiaside | Aurantio-Obtusin | Reference Drugs for Target Receptors | |
|---|---|---|---|---|---|
| % Stimulation a (% Inhibition b) | % Stimulation a (% Inhibition b) | % Stimulation a (% Inhibition b) | Agonist Effect: EC50 c; Antagonist Effect: (IC50 d) | ||
| D3 (h) | 7.05 ± 1.63 (−0.05 ± 0.78) | 14.75 ± 1.20 (−5.05 ± 2.05) | 33.00 ± 1.84 (−2.30 ± 0.57) | Dopamine | 2.7 |
| (+) butaclamol | (25) | ||||
| NK1 (h) | −1.40 ± 0.42 (3.90 ± 5.94) | −1.70 ± 0.14 (20.50 ± 1.84) | −6.40 ± 0.00 (47.60 ± 3.11) | [Sar9, Met(O2)11]-SP | 0.18 |
| L 733,060 | (0.58) | ||||
| 5-HT1A (h) | −0.30 ± 0.28 (4.25 ± 2.47) | 1.75 ± 0.07 (−13.45 ± 0.07) | −6.10 ± 0.00 (10.10 ± 4.10) | Serotonin | 1.6 |
| (S)-WAY-100635 | (5.6) | ||||
| V1A (h) | −4.40 ± 1.41 (−13.60 ± 0.57) | 1.40 ± 2.83 (−9.90 ± 6.65) | −33.20 ± 2.69 (71.80 ± 6.08) | Arginine vasopressin | 0.89 |
| d(CH2)51,Tyr(Me)2]-AVP | (4.5) | ||||
| Ligand | Binding Score (kcal/mol) | Interacting Residues | ||
|---|---|---|---|---|
| H-Bond | Electrostatic | Hydrophobic | ||
| Aurantio-obtusin | −7.58 | Ala101, Lys128, Ser341, Gln131 | − | Val100(π-σ), Ala101(π-alkyl), Trp304(π-π T-shaped, π-alkyl), Ala334(alkyl, π-alkyl), Met135(alkyl, π-aAlkyl), Val100(alkyl), Phe307(π-alkyl) |
| 2-Hydroxyemodin 1-methylether | −7.34 | Ala101, Lys128, Gln131, Gly337 | − | Val100(π-σ), Trp304(π-π T-shaped), Phe307(π-π T-shaped, π-alkyl), Ala334(alkyl, π-alkyl), Met135(alkyl, π-alkyl), Ala101(π-alkyl) |
| d(CH2)51,Tyr(Me)2]-AVP a (antagonist) | −6.07 | Lys128, Asn327, Asp112, Thr331, Gln131 | − | Trp332(π-π stacked), Ile330(alkyl), Tyr115(π-alkyl) |
| AVP a (agonist) | −8.62 | Asp202, Glu54, Asp112, Thr331, Thr198 | Trp204(π-Sulfur), Trp111(π-Sulfur) | Ile330(Alkyl), Val194(π-Alkyl) |
| Compounds | Drug-Likeness g | ADME Characteristics | ||||||
|---|---|---|---|---|---|---|---|---|
| MDDR-like rule | Lipinski’s rule | Log Po/w a | PPB b | HIA c | In vitro MDCK cell permeability (nm/s) d | In vitro Caco2 permeability (nm/s) e | In vivo BBB penetration ([brain]/[blood]) f | |
| Aurantio-obtusin | Mid-structure | Suitable | 2.53 | 86.98 | 84.66 | 113.20 | 19.17 | 0.48 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paudel, P.; Kim, D.H.; Jeon, J.; Park, S.E.; Seong, S.H.; Jung, H.A.; Choi, J.S. Neuroprotective Effect of Aurantio-Obtusin, a Putative Vasopressin V1A Receptor Antagonist, on Transient Forebrain Ischemia Mice Model. Int. J. Mol. Sci. 2021, 22, 3335. https://doi.org/10.3390/ijms22073335
Paudel P, Kim DH, Jeon J, Park SE, Seong SH, Jung HA, Choi JS. Neuroprotective Effect of Aurantio-Obtusin, a Putative Vasopressin V1A Receptor Antagonist, on Transient Forebrain Ischemia Mice Model. International Journal of Molecular Sciences. 2021; 22(7):3335. https://doi.org/10.3390/ijms22073335
Chicago/Turabian StylePaudel, Pradeep, Dong Hyun Kim, Jieun Jeon, Se Eun Park, Su Hui Seong, Hyun Ah Jung, and Jae Sue Choi. 2021. "Neuroprotective Effect of Aurantio-Obtusin, a Putative Vasopressin V1A Receptor Antagonist, on Transient Forebrain Ischemia Mice Model" International Journal of Molecular Sciences 22, no. 7: 3335. https://doi.org/10.3390/ijms22073335







