Fasciola hepatica Gastrodermal Cells Selectively Release Extracellular Vesicles via a Novel Atypical Secretory Mechanism
Abstract
:1. Introduction
2. Results
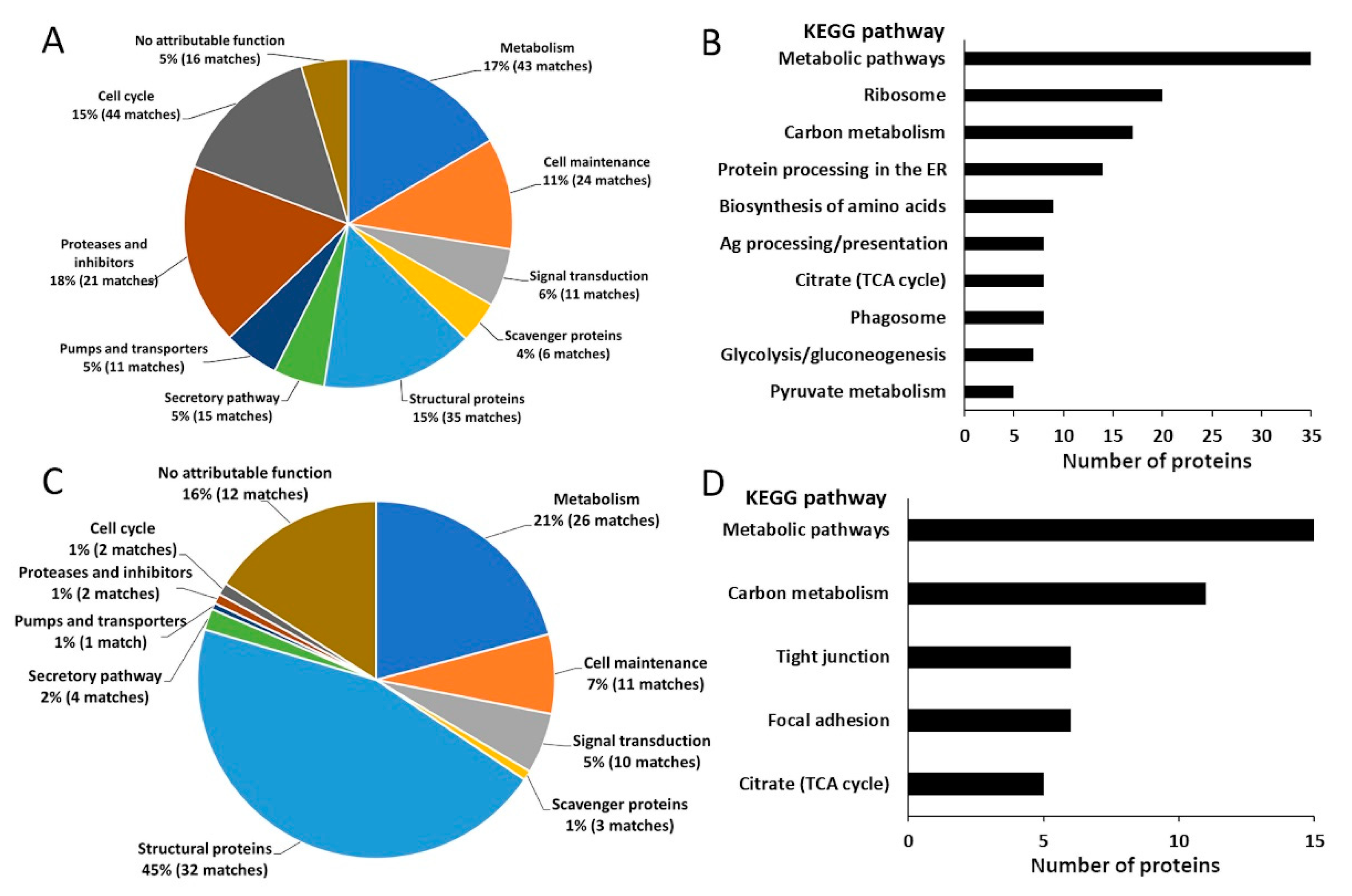
2.1. The Gastrodermal Cell Proteome Reflects Its Role in Secretion and Haematophagy
2.2. The Proteome of the Tegument Supports Its Barrier Function
2.3. Comparisons of the Tegument, Gut, and Secretome Indicate That the Gastrodermal Cells Are the Principal Source of Secreted Proteins
2.4. Microscopical Examination Suggests an Atypical Mechanism of Extracellular Vesicle Release by Some Gastrodermal Cells
3. Discussion
4. Materials and Methods
4.1. Laser Microdissection
4.2. Mass Spectrometry of Laser Microdissected Tissue
4.3. Database Searching
4.4. Criteria for Protein Identification
4.5. Functional Annotation of Proteins
4.6. Isolation and Mass Spectrometry Analysis of Adult F. hepatica 15K Extracellular Vesicles
4.7. Histology and Immunofluorescent Labelling of F. hepatica Tissue Sections
4.8. Processing of F. hepatica for Transmission Electron Microscopy
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Harrington, D.; Lamberton, P.H.L.; McGregor, A. Human Liver Flukes. Lancet Gastroenterol. Hepatol. 2017, 2, 680–689. [Google Scholar] [CrossRef]
- Andrews, S.J. The Life Cycle of Fasciola hepatica. In Fascioliasis; Dalton, J.P., Ed.; CABI: Oxford, UK, 1999; pp. 1–29. [Google Scholar]
- González-Miguel, J.; Becerro-Recio, D.; Siles-Lucas, M. Insights into Fasciola hepatica Juveniles: Crossing the Fasciolosis Rubicon. Trends Parasitol. 2021, 37, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Boray, J.C. Experimental Fascioliasis in Australia. Adv. Parasitol. 1969, 7, 95–210. [Google Scholar] [CrossRef] [PubMed]
- Fairweather, I.; Threadgold, L.T.; Hanna, R.E.B. Development of Fasciola hepatica in the Mammalian Host. In Fascioliasis; Dalton, J.P., Ed.; CABI: Oxford, UK, 1999; pp. 47–103. [Google Scholar]
- Robinson, G.; Threadgold, L.T. Electron microscope studies of Fasciola hepatica: XII. The fine structure of the gastrodermis. Exp. Parasitol. 1975, 37, 20–36. [Google Scholar] [CrossRef]
- Threadgold, L.T. Fasciola hepatica: A Transmission and Scanning Electron Microscopical Study of the Apical Surface of the Gastrodermal Cells. Parasitology 1978, 76, 85–90. [Google Scholar] [CrossRef]
- Robinson, M.W.; Dalton, J.P.; Donnelly, S. Helminth Pathogen Cathepsin Proteases: It’s a Family Affair. Trends Biochem. Sci. 2008, 33, 601–608. [Google Scholar] [CrossRef] [Green Version]
- Hanna, R.E.B. Fasciola hepatica: An Electron Microscope Autoradiographic Study of Protein Synthesis and Secretion by Gut Cells in Tissue Slices. Exp. Parasitol. 1975, 38, 167–180. [Google Scholar] [CrossRef]
- Lowther, J.; Robinson, M.W.; Donnelly, S.M.; Xu, W.; Stack, C.M.; Matthews, J.M.; Dalton, J.P. The Importance of PH in Regulating the Function of the Fasciola hepatica Cathepsin L1 Cysteine Protease. PLoS Negl. Trop. Dis. 2009, 3, e369. [Google Scholar] [CrossRef] [Green Version]
- Cwiklinski, K.; Donnelly, S.; Drysdale, O.; Jewhurst, H.; Smith, D.; Verissimo, C.D.M.; Pritsch, I.C.; O′Neill, S.; Dalton, J.P.; Robinson, M.W. The Cathepsin-like Cysteine Peptidases of Trematodes of the Genus Fasciola. Adv. Parasitol. 2019, 104, 113–164. [Google Scholar] [CrossRef]
- Collins, P.R.; Stack, C.M.; O’Neill, S.M.; Doyle, S.; Ryan, T.; Brennan, G.P.; Mousley, A.; Stewart, M.; Maule, A.G.; Dalton, J.P.; et al. Cathepsin L1, the Major Protease Involved in Liver Fluke (Fasciola hepatica) Virulence: Propeptide Cleavage Sites and Autoactivation of the Zymogen Secreted from Gastrodermal Cells. J. Biol. Chem. 2004, 279, 17038–17046. [Google Scholar] [CrossRef] [Green Version]
- Stack, C.M.; Caffrey, C.R.; Donnelly, S.M.; Seshaadri, A.; Lowther, J.; Tort, J.F.; Collins, P.R.; Robinson, M.W.; Xu, W.; McKerrow, J.H.; et al. Structural and Functional Relationships in the Virulence-Associated Cathepsin L Proteases of the Parasitic Liver Fluke, Fasciola hepatica. J. Biol. Chem. 2008, 283, 9896–9908. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robinson, M.W.; Tort, J.F.; Lowther, J.; Donnelly, S.M.; Wong, E.; Xu, W.; Stack, C.M.; Padula, M.; Herbert, B.; Dalton, J.P. Proteomics and Phylogenetic Analysis of the Cathepsin L Protease Family of the Helminth Pathogen Fasciola hepatica: Expansion of a Repertoire of Virulence-Associated Factors. Mol. Cell. Proteom. 2008, 7, 1111–1123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dalton, J.P.; Skelly, P.; Halton, D.W. Role of the Tegument and Gut in Nutrient Uptake by Parasitic Platyhelminths. Can. J. Zool. 2004, 82, 211–232. [Google Scholar] [CrossRef]
- Bennett, A.P.S.; de la Torre-Escudero, E.; Oliver, N.A.M.; Huson, K.M.; Robinson, M.W. The Cellular and Molecular Origins of Extracellular Vesicles Released by the Helminth Pathogen, Fasciola hepatica. Int. J. Parasitol. 2020, 50, 671–683. [Google Scholar] [CrossRef]
- Cwiklinski, K.; de la Torre-Escudero, E.; Trelis, M.; Bernal, D.; Dufresne, P.J.; Brennan, G.P.; O′Neill, S.; Tort, J.; Paterson, S.; Marcilla, A.; et al. The Extracellular Vesicles of the Helminth Pathogen, Fasciola hepatica: Biogenesis Pathways and Cargo Molecules Involved in Parasite Pathogenesis. Mol. Cell. Proteom. 2015, 14, 3258–3273. [Google Scholar] [CrossRef] [Green Version]
- de la Torre-Escudero, E.; Pérez-Sánchez, R.; Manzano-Román, R.; Oleaga, A. Schistosome Infections Induce Significant Changes in the Host Biliary Proteome. J. Proteom. 2015, 114, 71–82. [Google Scholar] [CrossRef] [Green Version]
- Marcilla, A.; Trelis, M.; Cortés, A.; Sotillo, J.; Cantalapiedra, F.; Minguez, M.T.; Valero, M.L.; Sánchez del Pino, M.M.; Muñoz-Antoli, C.; Toledo, R.; et al. Extracellular Vesicles from Parasitic Helminths Contain Specific Excretory/Secretory Proteins and Are Internalized in Intestinal Host Cells. PLoS ONE 2012, 7, e45974. [Google Scholar] [CrossRef]
- de la Torre-Escudero, E.; Bennett, A.P.S.; Clarke, A.; Brennan, G.P.; Robinson, M.W. Extracellular Vesicle Biogenesis in Helminths: More than One Route to the Surface? Trends Parasitol. 2016, 32, 921–929. [Google Scholar] [CrossRef] [Green Version]
- Haçarız, O.; Baykal, A.T.; Akgün, M.; Kavak, P.; Saǧıroǧlu, M.Ş.; Sayers, G.P. Generating a Detailed Protein Profile of Fasciola hepatica during the Chronic Stage of Infection in Cattle. Proteomics 2014, 14, 1519–1530. [Google Scholar] [CrossRef]
- Wilson, R.A.; Wright, J.M.; de Castro-Borges, W.; Parker-Manuel, S.J.; Dowle, A.A.; Ashton, P.D.; Young, N.D.; Gasser, R.B.; Spithill, T.W. Exploring the Fasciola hepatica Tegument Proteome. Int. J. Parasitol. 2011, 41, 1347–1359. [Google Scholar] [CrossRef] [Green Version]
- Robinson, M.W.; Menon, R.; Donnelly, S.M.; Dalton, J.P.; Ranganathan, S. An Integrated Transcriptomics and Proteomics Analysis of the Secretome of the Helminth Pathogen Fasciola hepatica: Proteins Associated with Invasion and Infection of the Mammalian Host. Mol. Cell. Proteom. 2009, 8, 1891–1907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de la Torre-Escudero, E.; Gerlach, J.Q.; Bennett, A.P.S.; Cwiklinski, K.; Jewhurst, H.L.; Huson, K.M.; Joshi, L.; Kilcoyne, M.; O’Neill, S.; Dalton, J.P.; et al. Surface Molecules of Extracellular Vesicles Secreted by the Helminth Pathogen Fasciola hepatica Direct Their Internalisation by Host Cells. PLoS Negl. Trop. Dis. 2019, 13, e0007087. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Niel, G.; D′Angelo, G.; Raposo, G. Shedding Light on the Cell Biology of Extracellular Vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zheng, W.; Freddolino, P.L.; Zhang, Y. MetaGO: Predicting Gene Ontology of Non-Homologous Proteins through Low-Resolution Protein Structure Prediction and Protein-Protein Network Mapping. J. Mol. Biol. 2018, 430, 2256–2265. [Google Scholar] [CrossRef]
- Le Clainche, C.; Dwivedi, S.P.; Didry, D.; Carlier, M.-F. Vinculin Is a Dually Regulated Actin Filament Barbed End-Capping and Side-Binding Protein. J. Biol. Chem. 2010, 285, 23420–23432. [Google Scholar] [CrossRef] [Green Version]
- Fontao, L.; Favre, B.; Riou, S.; Geerts, D.; Jaunin, F.; Saurat, J.-H.; Green, K.J.; Sonnenberg, A.; Borradori, L. Interaction of the Bullous Pemphigoid Antigen 1 (BP230) and Desmoplakin with Intermediate Filaments Is Mediated by Distinct Sequences within Their COOH Terminus. Mol. Biol. Cell 2003, 14, 1978–1992. [Google Scholar] [CrossRef]
- Rehman, A.A.; Ahsan, H.; Khan, F.H. Alpha-2-Macroglobulin: A Physiological Guardian. J. Cell. Physiol. 2013, 228, 1665–1675. [Google Scholar] [CrossRef]
- Ordway, G.A.; Garry, D.J. Myoglobin: An essential hemoprotein in striated muscle. J. Exp. Biol. 2004, 207, 3441–3446. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Z.; White, K.A.; Polissi, A.; Georgopoulos, C.; Raetz, C.R. Function of Escherichia Coli MsbA, an Essential ABC Family Transporter, in Lipid A and Phospholipid Biosynthesis. J. Biol. Chem. 1998, 273, 12466–12475. [Google Scholar] [CrossRef] [Green Version]
- Kozyraki, R.; Fyfe, J.; Verroust, P.J.; Jacobsen, C.; Dautry-Varsat, A.; Gburek, J.; Willnow, T.E.; Christensen, E.I.; Moestrup, S.K. Megalin-Dependent Cubilin-Mediated Endocytosis Is a Major Pathway for the Apical Uptake of Transferrin in Polarized Epithelia. Proc. Natl. Acad. Sci. USA 2001, 98, 12491–12496. [Google Scholar] [CrossRef] [Green Version]
- Kozyraki, R.; Fyfe, J.; Kristiansen, M.; Gerdes, C.; Jacobsen, C.; Cui, S.; Christensen, E.I.; Aminoff, M.; De La Chapelle, A.; Krahe, R.; et al. The Intrinsic Factor-Vitamin B12 Receptor, Cubilin, Is a High-Affinity Apolipoprotein A-I Receptor Facilitating Endocytosis of High-Density Lipoprotein. Nat. Med. 1999, 5, 656–661. [Google Scholar] [CrossRef] [PubMed]
- Pankao, V.; Sirisriro, A.; Grams, R.; Vichasri-Grams, S.; Meepool, A.; Kangwanrangsan, N.; Wanichanon, C.; Ardseungneon, P.; Viyanant, V.; Upatham, E.S.; et al. Classification of the Parenchymal Cells in Fasciola Gigantica Based on Ultrastructure and Their Expression of Fatty Acid Binding Proteins (FABPs). Vet. Parasitol. 2006, 142, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Ostasiewicz, P.; Zielinska, D.F.; Mann, M.; Wiśniewski, J.R. Proteome, Phosphoproteome, and N-Glycoproteome Are Quantitatively Preserved in Formalin-Fixed Paraffin-Embedded Tissue and Analyzable by High-Resolution Mass Spectrometry. J. Proteome Res. 2010, 9, 3688–3700. [Google Scholar] [CrossRef] [PubMed]
- Morphew, R.M.; Wright, H.A.; LaCourse, E.J.; Woods, D.J.; Brophy, P.M. Comparative Proteomics of Excretory-Secretory Proteins Released by the Liver Fluke Fasciola hepatica in Sheep Host Bile and during in Vitro Culture Ex Host. Mol. Cell. Proteom. 2007, 6, 963–972. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Acosta, D.; Cancela, M.; Piacenza, L.; Roche, L.; Carmona, C.; Tort, J.F. Fasciola hepatica Leucine Aminopeptidase, a Promising Candidate for Vaccination against Ruminant Fasciolosis. Mol. Biochem. Parasitol. 2008, 158, 52–64. [Google Scholar] [CrossRef]
- Magalhães, G.S.; Lopes-Ferreira, M.; Junqueira-De-Azevedo, I.L.M.; Spencer, P.J.; Araújo, M.S.; Portaro, F.C.V.; Ma, L.; Valente, R.H.; Juliano, L.; Fox, J.W.; et al. Natterins, a New Class of Proteins with Kininogenase Activity Characterized from Thalassophryne Nattereri Fish Venom. Biochimie 2005, 87, 687–699. [Google Scholar] [CrossRef]
- Wilson, R.A.; Langermans, J.A.M.; van Dam, G.J.; Vervenne, R.A.; Hall, S.L.; Borges, W.C.; Dillon, G.P.; Thomas, A.W.; Coulson, P.S. Elimination of Schistosoma Mansoni Adult Worms by Rhesus Macaques: Basis for a Therapeutic Vaccine? PLoS Negl. Trop. Dis. 2008, 2, e290. [Google Scholar] [CrossRef] [Green Version]
- Cameron, T.C.; Cooke, I.; Faou, P.; Toet, H.; Piedrafita, D.; Young, N.; Rathinasamy, V.; Beddoe, T.; Anderson, G.; Dempster, R.; et al. A Novel Ex Vivo Immunoproteomic Approach Characterising Fasciola hepatica Tegumental Antigens Identified Using Immune Antibody from Resistant Sheep. Int. J. Parasitol. 2017, 47, 555–567. [Google Scholar] [CrossRef]
- Threadgold, L.T. The Tegument and Associated Structures of Fasciola hepatica. Quart. J. Micr. Sci 1963, 104, 505–512. [Google Scholar] [CrossRef]
- Tielens, A.G.M.; van den Heuvel, J.M.; van den Bergh, S.G. The Energy Metabolism of Fasciola hepatica during Its Development in the Final Host. Mol. Biochem. Parasitol. 1984, 13, 301–307. [Google Scholar] [CrossRef] [Green Version]
- Tielens, A.G.M.; van den Heuvel, J.M.; van den Bergh, S.G. Differences in Intermediary Energy Metabolism between Juvenile and Adult Fasciola hepatica. Mol. Biochem. Parasitol. 1987, 24, 273–281. [Google Scholar] [CrossRef] [Green Version]
- Tielens, A.G.M. Metabolism. In Fascioliasis; Dalton, J.P., Ed.; CABI: Oxford, UK, 1999; pp. 277–306. [Google Scholar]
- Bennett, A.P.S.; de la Torre-Escudero, E.; Robinson, M.W. Helminth Genome Analysis Reveals Conservation of Extracellular Vesicle Biogenesis Pathways but Divergence of RNA Loading Machinery between Phyla. Int. J. Parasitol. 2020, 50, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Anthony, R.M.; Gause, W.C. Immunofluorescent Staining for the Laser Microdissection of Individual Cells. Protoc. Exch. 2006. [Google Scholar] [CrossRef]
- Anthony, R.M.; Urban, J.F.; Alem, F.; Hamed, H.A.; Rozo, C.T.; Boucher, J.-L.; Van Rooijen, N.; Gause, W.C. Memory T(H)2 Cells Induce Alternatively Activated Macrophages to Mediate Protection against Nematode Parasites. Nat. Med. 2006, 12, 955–960. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.N.; Winters, A.; Milic, I.; Devitt, A.; Cookson, A.; Brophy, P.M.; Morphew, R.M. Evidence of Sequestration of Triclabendazole and Associated Metabolites by Extracellular Vesicles of Fasciola hepatica. Sci. Rep. 2020, 10, 13445. [Google Scholar] [CrossRef]
- Hanna, M.G.; Block, S.; Frankel, E.B.; Hou, F.; Johnson, A.; Yuan, L.; Knight, G.; Moresco, J.J.; Yates, J.R.; Ashton, R.; et al. TFG Facilitates Outer Coat Disassembly on COPII Transport Carriers to Promote Tethering and Fusion with ER–Golgi Intermediate Compartments. Proc. Natl. Acad. Sci. USA 2017, 114, E7707–E7716. [Google Scholar] [CrossRef] [Green Version]
- Herron, C.M.; O’Connor, A.; Robb, E.; McCammick, E.; Hill, C.; Marks, N.J.; Robinson, M.W.; Maule, A.G.; McVeigh, P. Developmental Regulation and Functional Prediction of MicroRNAs in an Expanded Fasciola hepatica MiRNome. Front. Cell. Infect. Microbiol. 2022, 12, 811123. [Google Scholar] [CrossRef]
- Fontenla, S.; Langleib, M.; de la Torre-Escudero, E.; Domínguez, M.F.; Robinson, M.W.; Tort, J. Role of Fasciola hepatica Small RNAs in the Interaction with the Mammalian Host. Front. Cell. Infect. Microbiol. 2022, 11, 812141. [Google Scholar] [CrossRef]
- Sotillo, J.; Robinson, M.W.; Kimber, M.J.; Cucher, M.; Ancarola, M.E.; Nejsum, P.; Marcilla, A.; Eichenberger, R.M.; Tritten, L. The Protein and MicroRNA Cargo of Extracellular Vesicles from Parasitic Helminths–Current Status and Research Priorities. Int. J. Parasitol. 2020, 50, 635–645. [Google Scholar] [CrossRef]
- Tang, S.K.Y.; Marshall, W.F. Self-Repairing Cells: How Single Cells Heal Membrane Ruptures and Restore Lost Structures. Science 2017, 356, 1022–1025. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sonnemann, K.J.; Bement, W.M. Wound Repair: Toward Understanding and Integration of Single-Cell and Multicellular Wound Responses. Annu. Rev. Cell Dev. Biol. 2011, 27, 237–263. [Google Scholar] [CrossRef] [Green Version]
- Hanna, R.E.B.; Gordon, A.W.; Moffett, D.; Edgar, H.W.J.; Oliver, L.F.; McConnell, S.; Shaw, L.; Brennan, G.P.; Fairweather, I. Fasciola hepatica: Comparative Effects of Host Resistance and Parasite Intra-Specific Interactions on Size and Reproductive Histology in Flukes from Rats Infected with Isolates Differing in Triclabendazole Sensitivity. Vet. Parasitol. 2011, 178, 251–263. [Google Scholar] [CrossRef] [PubMed]
- Wiśniewski, J.R.; Zougman, A.; Nagaraj, N.; Mann, M. Universal Sample Preparation Method for Proteome Analysis. Nat. Methods 2009, 6, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Cwiklinski, K.; Dalton, J.P.; Dufresne, P.J.; La Course, J.; Williams, D.J.; Hodgkinson, J.; Paterson, S. The Fasciola hepatica Genome: Gene Duplication and Polymorphism Reveals Adaptation to the Host Environment and the Capacity for Rapid Evolution. Genome Biol. 2015, 16, 71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nesvizhskii, A.I.; Keller, A.; Kolker, E.; Aebersold, R. A Statistical Model for Identifying Proteins by Tandem Mass Spectrometry. Anal. Chem. 2003, 75, 4646–4658. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, A.; Chang, H.-Y.; Daugherty, L.; Fraser, M.; Hunter, S.; López, R.; McAnulla, C.; McMenamin, C.; Nuka, G.; Pesseat, S.; et al. The InterPro Protein Families Database: The Classification Resource after 15 Years. Nucleic Acids Res. 2015, 43, D213–D221. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Goto, S.; Sato, Y.; Furumichi, M.; Tanabe, M. KEGG for Integration and Interpretation of Large-Scale Molecular Data Sets. Nucleic Acids Res. 2012, 40, D109–D114. [Google Scholar] [CrossRef] [Green Version]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and Integrative Analysis of Large Gene Lists Using DAVID Bioinformatics Resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Bioinformatics Enrichment Tools: Paths toward the Comprehensive Functional Analysis of Large Gene Lists. Nucleic Acids Res. 2009, 37, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Gallagher, S.S.E.; Threadgold, L.T. Electron-Microscope Studies of Fasciola hepatica II. The Interrelationship of the Parenchyma with Other Organ Systems. Parasitology 1967, 57, 627–632. [Google Scholar] [CrossRef]


| Protein | Identifier | Gastrodermal Cell | LMD Tegument 1 | Previously Detected | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Unique Peptides | Surface Protein Fraction 2 | Tegument 3 | Vomitus 3 | ESP Minus Vomitus 3 | Soluble ESP 4 | 15K EVs 1,5 | 120K EVs 4,5 | ||||||
| R1 | R2 | R3 | R4 | ||||||||||
| Proteases and inhibitors | |||||||||||||
| Proteases | |||||||||||||
| Leucine aminopeptidase 2 | BN1106_s617B000566 | 15 | 11 | 18 | 17 | ● | ● | ● | ● | ● | |||
| Xaa-Pro dipeptidase (M24 family) | BN1106_s468B000343 | 6 | 4 | 5 | 13 | ● | ● | ● | ● | ● | |||
| Leucine aminopeptidase 2 | BN1106_s617B000567 | 5 | 4 | 5 | 6 | ● | ● | ● | ● | ● | |||
| Lysosomal Pro-X carboxypeptidase | BN1106_s1620B000120 | 5 | 4 | 5 | 3 | ● | ● | ● | ● | ● | |||
| Leucine aminopeptidase 1 | BN1106_s7079B000034 | 5 | 4 | 4 | 12 | ● | ● | ● | ● | ● | |||
| Cathepsin B6/8 | BN1106_s793B000177 | 5 | 3 | 4 | 5 | ● | ● | ● | |||||
| Cathepsin L1 | BN1106_s8490B000026 | 4 | 4 | 5 | 5 | ● | ● | ● | ● | ● | ● | ● | |
| Cathepsin L2 | BN1106_s8098B000020 | 3 | 3 | 5 | 7 | ● | ● | ● | ● | ● | ● | ||
| Natterin-4 (DM9 domain-containing protein) | BN1106_s586B000374 | 3 | 3 | 5 | 6 | ● | ● | ● | ● | ||||
| Cathepsin L1 | BN1106_s10332B000010 | 3 | 2 | 3 | 1 | ● | ● | ● | ● | ||||
| Cathepsin L5 | BN1106_s4636B000039 | 2 | 3 | 2 | 5 | ● | ● | ||||||
| Cathepsin B4/5/7 | BN1106_s13444B000002 | 2 | 2 | 3 | 5 | ● | ● | ● | |||||
| Cathepsin L5 | BN1106_s6354B000017 | 2 | 2 | 2 | 2 | ● | ● | ● | ● | ||||
| Cysteine protease-related protein | BN1106_s1772B000188 | 1 | 2 | 2 | 5 | ● | ● | ||||||
| Aspartyl aminopeptidase | BN1106_s2165B000367 | 1 | 2 | 2 | 4 | ● | |||||||
| Legumain-like | BN1106_s1861B000097 | 1 | 1 | 3 | 4 | ● | ● | ● | ● | ● | |||
| Mitochondrial processing peptidase | BN1106_s37B000342 | 1 | 0 | 2 | 2 | ● | |||||||
| Protease inhibitors | |||||||||||||
| Cystatin-1 | BN1106_s4651B000094 | 4 | 3 | 4 | 7 | ● | ● | ● | ● | ● | |||
| Serpin | BN1106_s3864B000104 | 3 | 2 | 5 | 2 | ● | ● | ● | ● | ||||
| FH-KTM kunitz-type proteinase inhibitor | BN1106_s318B000274 | 2 | 2 | 3 | 2 | ● | ● | ● | ● | ● | |||
| FH-KTM kunitz-type proteinase inhibitor | BN1106_s8826B000029 | 1 | 2 | 2 | 2 | ● | ● | ● | ● | ||||
| Structural proteins | |||||||||||||
| Motor/cytoskeletal | |||||||||||||
| Myosin heavy chain | BN1106_s323B000258 | 16 | 12 | 19 | 29 | ● | |||||||
| Paramyosin | BN1106_s1922B000122 | 8 | 4 | 11 | 21 | ● | |||||||
| Filamin | BN1106_s296B000186 | 5 | 0 | 5 | 6 | ● | ● | ● | |||||
| Actin | BN1106_s658B000223 | 4 | 4 | 6 | 9 | ● | ● | ● | ● | ||||
| Filamin-C | BN1106_s1515B000336 | 4 | 3 | 3 | 5 | ● | ● | ||||||
| Myosin heavy chain | BN1106_s323B000257 | 3 | 1 | 4 | 5 | ● | ● | ||||||
| Myosin heavy chain | BN1106_s3182B000117 | 3 | 0 | 2 | 9 | ● | ● | ● | |||||
| Beta-tubulin 4 | BN1106_s1153B000359 | 2 | 2 | 3 | 3 | ● | ● | ● | |||||
| Actin | BN1106_s101B000531 | 2 | 2 | 3 | 2 | ● | ● | ● | |||||
| Alpha-tubulin 2 | BN1106_s925B000543 | 2 | 1 | 2 | 0 | ● | ● | ||||||
| Talin | BN1106_s149B000360 | 2 | 0 | 2 | 1 | ||||||||
| Alpha-actinin sarcomeric | BN1106_s4069B000247 | 1 | 2 | 1 | 4 | ● | ● | ● | ● | ● | |||
| Tropomyosin | BN1106_s647B000405 | 1 | 1 | 5 | 3 | ● | |||||||
| Lamin | BN1106_s1106B000091 | 1 | 1 | 2 | 5 | ||||||||
| Tropomyosin-2 | BN1106_s4130B000080 | 1 | 1 | 1 | 7 | ● | ● | ● | |||||
| Tubulin beta-3 | BN1106_s4860B000047 | 1 | 1 | 0 | 7 | ● | ● | ● | |||||
| Titin | BN1106_s98B000745 | 1 | 0 | 2 | 2 | ● | |||||||
| Cytoskeletal-binding/modifying | |||||||||||||
| Spectrin beta chain, brain 4 | BN1106_s4255B000066 | 7 | 3 | 8 | 8 | ● | ● | ||||||
| Merlin/moesin/ezrin/radixin | BN1106_s1300B000145 | 6 | 8 | 9 | 18 | ● | ● | ● | ● | ||||
| Ezrin-radixin-moesin-binding phosphoprotein 50 | BN1106_s1037B000175 | 4 | 3 | 6 | 6 | ● | ● | ||||||
| Calpain (C02 family) | BN1106_s204B000249 | 4 | 0 | 4 | 7 | ● | ● | ● | |||||
| Rho GDP-dissociation inhibitor | BN1106_s4672B000098 | 2 | 2 | 3 | 1 | ● | |||||||
| Myophilin | BN1106_s3747B000111 | 2 | 1 | 2 | 3 | ● | ● | ● | ● | ||||
| Ras-like GTP-binding protein Rho1 | BN1106_s1908B000177 | 1 | 2 | 2 | 1 | ● | ● | ● | |||||
| Calponin homolog | BN1106_s2140B000163 | 1 | 1 | 4 | 2 | ● | ● | ||||||
| Translationally controlled tumour protein | BN1106_s17035B000006 | 3 | 2 | 3 | 5 | ● | ● | ||||||
| Abnormal long morphology protein 1 | BN1106_s1644B000226 | 0 | 0 | 2 | 2 | ||||||||
| Extracellular matrix/cell adhesion | |||||||||||||
| Heparan sulphate proteoglycan | BN1106_s25B000189 | 5 | 3 | 12 | 10 | ● | ● | ● | |||||
| Alpha 1 type IIA collagen | BN1106_s849B000266 | 3 | 2 | 4 | 4 | ● | |||||||
| Collagen alpha-1(V) chain | BN1106_s457B000392 | 2 | 2 | 2 | 3 | ● | |||||||
| Fasciclin-1 | BN1106_s100B000380 | 2 | 2 | 2 | 2 | ||||||||
| Collagen alpha-1(V) chain | BN1106_s2714B000202 | 2 | 0 | 2 | 3 | ● | |||||||
| Fasciclin I-like protein | BN1106_s1922B000120 | 1 | 1 | 3 | 3 | ● | ● | ||||||
| Collagen alpha-2(I) chain | BN1106_s104B000457 | 1 | 1 | 2 | 2 | ● | |||||||
| Collagen alpha-1(V) chain | BN1106_s849B000265 | 1 | 0 | 2 | 3 | ● | |||||||
| Signal transduction | |||||||||||||
| Signalling | |||||||||||||
| Tyrosine 3-monooxygenase | BN1106_s3172B000053 | 4 | 4 | 6 | 6 | ● | ● | ● | ● | ||||
| 14-3-3 epsilon | BN1106_s686B000273 | 4 | 2 | 4 | 5 | ● | ● | ● | ● | ● | |||
| 14-3-3 protein beta | BN1106_s3904B000042 | 2 | 2 | 1 | 3 | ● | ● | ● | ● | ● | |||
| Nucleoside diphosphate kinase | BN1106_s344B000191 | 1 | 1 | 2 | 2 | ● | ● | ● | |||||
| Protein CBR-FTT-2 | BN1106_s4074B000042 | 1 | 1 | 2 | 2 | ● | |||||||
| Camp-dependent protein kinase type II-alpha | BN1106_s417B000229 | 1 | 0 | 2 | 3 | ● | ● | ||||||
| Calcium-binding | |||||||||||||
| CaBP4 | BN1106_s214B000744 | 6 | 4 | 8 | 11 | ● | ● | ● | ● | ● | ● | ||
| 22.4 kDa tegument protein | BN1106_s214B000748 | 4 | 4 | 4 | 4 | ● | ● | ● | |||||
| Tegumental calcium-binding EF-hand protein | BN1106_s214B000742 | 1 | 1 | 2 | 3 | ● | ● | ● | ● | ● | |||
| Ubiquitination pathway | |||||||||||||
| Ubiquitin-activating enzyme E1 | BN1106_s5276B000036 | 1 | 3 | 1 | 6 | ● | ● | ● | |||||
| Ubiquitin-protein ligase BRE1 | BN1106_s208B000185 | 1 | 1 | 2 | 3 | ● | ● | ● | ● | ||||
| Secretory pathway | |||||||||||||
| ER/Golgi apparatus-related | |||||||||||||
| Venom allergen-like 11 protein | BN1106_s1956B000118 | 5 | 4 | 5 | 6 | ● | |||||||
| Calreticulin | BN1106_s2673B000071 | 2 | 1 | 3 | 6 | ● | |||||||
| Transitional endoplasmic reticulum ATPase | BN1106_s5369B000082 | 2 | 1 | 1 | 2 | ● | ● | ● | |||||
| Preprotein translocase secy subunit Sec61 | BN1106_s2995B000137 | 1 | 1 | 2 | 2 | ||||||||
| Atlastin-1 | BN1106_s3190B000098 | 0 | 1 | 2 | 2 | ||||||||
| Vesicle biogenesis and trafficking | |||||||||||||
| Annexin | BN1106_s819B000364 | 8 | 8 | 9 | 11 | ● | ● | ● | ● | ● | ● | ● | |
| Annexin | BN1106_s945B000218 | 4 | 2 | 5 | 8 | ● | ● | ● | ● | ● | ● | ||
| Coatomer subunit beta | BN1106_s771B000471 | 3 | 0 | 3 | 2 | ● | |||||||
| ADP-ribosylation factor 1 | BN1106_s4512B000085 | 2 | 2 | 2 | 2 | ● | ● | ||||||
| SAR1 gene homolog B | BN1106_s1405B000146 | 2 | 2 | 2 | 0 | ||||||||
| Coatomer protein complex subunit alpha | BN1106_s649B000500 | 2 | 1 | 2 | 3 | ● | |||||||
| Annexin | BN1106_s500B000161 | 1 | 3 | 2 | 5 | ● | ● | ● | ● | ● | ● | ||
| Coatomer subunit gamma | BN1106_s5131B000049 | 1 | 0 | 3 | 7 | ● | |||||||
| Ras-related protein Rab-11B | BN1106_s844B000259 | 1 | 0 | 3 | 3 | ● | ● | ● | |||||
| Protein transport protein Sec23A | BN1106_s2174B000116 | 1 | 0 | 2 | 3 | ● | |||||||
| Pumps and transporters | |||||||||||||
| Smdr2 | BN1106_s274B000296 | 8 | 7 | 12 | 18 | ● | ● | ● | |||||
| ATP synthase subunit beta | BN1106_s1866B000129 | 7 | 5 | 7 | 8 | ● | ● | ● | |||||
| ATP synthase subunit alpha | BN1106_s4332B000087 | 6 | 6 | 5 | 10 | ● | ● | ● | ● | ● | |||
| ATP:ADP antiporter | BN1106_s3313B000078 | 2 | 2 | 5 | 8 | ● | ● | ● | |||||
| V-type H+-transporting ATPase subunit A | BN1106_s2110B000156 | 2 | 2 | 3 | 4 | ● | ● | ||||||
| Innexin | BN1106_s503B000225 | 2 | 2 | 2 | 2 | ||||||||
| Phosphatidylcholine transfer protein | BN1106_s538B000493 | 2 | 2 | 1 | 2 | ● | ● | ||||||
| Sodium potassium transporting ATPase alpha subunit | BN1106_s521B000167 | 2 | 1 | 1 | 3 | ● | ● | ● | |||||
| Vacuolar H+-ATPase SFD subunit | BN1106_s2350B000136 | 1 | 2 | 2 | 2 | ● | ● | ||||||
| Vacuolar ATP synthase subunit e | BN1106_s1399B000513 | 1 | 1 | 2 | 3 | ● | ● | ||||||
| ATP synthase subunit beta vacuolar | BN1106_s1633B000182 | 0 | 0 | 3 | 5 | ● | ● | ||||||
| Protein | Identifier | LMD Tegument | Previously Detected | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gastrodermal Cells 1 | Surface Protein Fraction 2 | Tegument 3 | Vomitus 3 | ESP Minus Vomitus 3 | Soluble ESP 4 | 15K EVs 1,5 | 120K EVs 4,5 | ||||||
| Unique Peptides | |||||||||||||
| R1 | R2 | R3 | R4 | ||||||||||
| Structural proteins | |||||||||||||
| Motor/cytoskeletal | |||||||||||||
| Myosin heavy chain * | BN1106_s323B000258 | 17 | 17 | 23 | 24 | ● | |||||||
| Paramyosin * | BN1106_s1922B000122 | 9 | 10 | 9 | 10 | ● | |||||||
| Filamin | BN1106_s296B000186 | 4 | 3 | 4 | 2 | ● | ● | ● | |||||
| Filamin-C * | BN1106_s1515B000336 | 3 | 3 | 2 | 2 | ● | ● | ||||||
| Myosin heavy chain | BN1106_s323B000257 | 5 | 3 | 2 | 3 | ● | ● | ||||||
| Myosin heavy chain | BN1106_s3182B000117 | 8 | 8 | 0 | 3 | ● | ● | ● | |||||
| Alpha-actinin sarcomeric | BN1106_s4069B000247 | 8 | 10 | 3 | 10 | ● | ● | ● | ● | ● | |||
| Tropomyosin-2 | BN1106_s4130B000080 | 1 | 2 | 1 | 5 | ● | ● | ● | |||||
| Actin | BN1106_s2907B000133 | 4 | 3 | 1 | 3 | ● | ● | ● | ● | ● | |||
| Titin | BN1106_s1119B000202 | 3 | 2 | 3 | 1 | ● | |||||||
| Myosin regulatory light chain | BN1106_s527B000393 | 2 | 0 | 2 | 1 | ● | |||||||
| Dynein heavy chain * | BN1106_s1314B000437 | 2 | 2 | 0 | 0 | ● | |||||||
| Dynein light chain | BN1106_s949B000142 | 2 | 2 | 2 | 2 | ● | ● | ● | |||||
| Dynein light chain | BN1106_s1582B000145 | 1 | 2 | 2 | 1 | ● | ● | ● | |||||
| Dynein light chain | BN1106_s3147B000076 | 2 | 3 | 2 | 1 | ● | ● | ● | |||||
| Cytoskeletal-binding/modifying | |||||||||||||
| Spectrin beta chain, brain 4 | BN1106_s4255B000066 | 3 | 5 | 2 | 1 | ● | ● | ||||||
| Calpain (C02 family) | BN1106_s204B000249 | 8 | 8 | 3 | 2 | ● | ● | ● | |||||
| Calponin homolog | BN1106_s2140B000163 | 4 | 4 | 1 | 1 | ● | ● | ||||||
| Gelsolin | BN1106_s2349B000191 | 1 | 1 | 3 | 6 | ● | ● | ● | ● | ||||
| Lymphocyte cytosolic protein 1 | BN1106_s1403B000129 | 11 | 8 | 8 | 8 | ● | ● | ● | ● | ● | ● | ||
| Gelsolin | BN1106_s2349B000188 | 5 | 5 | 3 | 8 | ● | ● | ● | ● | ||||
| Myophilin | BN1106_s3026B000096 | 2 | 2 | 0 | 0 | ● | ● | ● | |||||
| Adenylyl cyclase-associated protein | BN1106_s4290B000110 | 2 | 2 | 0 | 0 | ● | ● | ||||||
| Extracellular matrix/cell adhesion | |||||||||||||
| Heparan sulphate proteoglycan | BN1106_s25B000189 | 4 | 3 | 8 | 3 | ● | ● | ● | |||||
| Alpha 1 type IIA collagen * | BN1106_s849B000266 | 4 | 4 | 4 | 0 | ● | |||||||
| Collagen alpha-1(V) chain * | BN1106_s457B000392 | 3 | 3 | 1 | 2 | ● | |||||||
| Collagen alpha-1(V) chain * | BN1106_s2714B000202 | 4 | 3 | 3 | 1 | ● | |||||||
| Fasciclin I-like protein * | BN1106_s1922B000120 | 6 | 5 | 4 | 2 | ● | ● | ||||||
| Collagen alpha-2(I) chain * | BN1106_s104B000457 | 4 | 3 | 2 | 2 | ● | |||||||
| Collagen alpha-1(V) chain * | BN1106_s849B000265 | 2 | 3 | 3 | 2 | ● | |||||||
| Collagen alpha-2(V) chain * | BN1106_s1528B000125 | 3 | 3 | 3 | 1 | ||||||||
| Collagen type XV alpha * | BN1106_s26B000447 | 2 | 1 | 2 | 2 | ||||||||
| Signal transduction | |||||||||||||
| Signalling | |||||||||||||
| Tyrosine 3-monooxygenase | BN1106_s3172B000053 | 2 | 2 | 2 | 1 | ● | ● | ● | ● | ||||
| 14-3-3 epsilon | BN1106_s686B000273 | 2 | 3 | 1 | 0 | ● | ● | ● | ● | ● | |||
| cAMP-dependent protein kinase * | BN1106_s2316B000078 | 2 | 2 | 0 | 3 | ● | |||||||
| Bcl-2 homologous antagonist | BN1106_s5167B000050 | 2 | 2 | 0 | 1 | ● | |||||||
| Calcium-binding | |||||||||||||
| CaBP4 | BN1106_s214B000744 | 3 | 5 | 4 | 7 | ● | ● | ● | ● | ● | ● | ||
| Tegumental calcium-binding EF-hand protein | BN1106_s214B000742 | 2 | 2 | 3 | 2 | ● | ● | ● | ● | ● | |||
| Tegumental calcium-binding EF-hand protein | BN1106_s214B000741 | 2 | 2 | 0 | 1 | ● | ● | ● | |||||
| Calmodulin-like protein 2 | BN1106_s2277B000048 | 2 | 3 | 2 | 5 | ● | ● | ● | ● | ||||
| Ubiquitination pathway | |||||||||||||
| Ubiquitin-activating enzyme E1 | BN1106_s5276B000036 | 2 | 2 | 1 | 2 | ● | ● | ● | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bennett, A.P.S.; de la Torre-Escudero, E.; Dermott, S.S.E.; Threadgold, L.T.; Hanna, R.E.B.; Robinson, M.W. Fasciola hepatica Gastrodermal Cells Selectively Release Extracellular Vesicles via a Novel Atypical Secretory Mechanism. Int. J. Mol. Sci. 2022, 23, 5525. https://doi.org/10.3390/ijms23105525
Bennett APS, de la Torre-Escudero E, Dermott SSE, Threadgold LT, Hanna REB, Robinson MW. Fasciola hepatica Gastrodermal Cells Selectively Release Extracellular Vesicles via a Novel Atypical Secretory Mechanism. International Journal of Molecular Sciences. 2022; 23(10):5525. https://doi.org/10.3390/ijms23105525
Chicago/Turabian StyleBennett, Adam P. S., Eduardo de la Torre-Escudero, Susan S. E. Dermott, Lawrence T. Threadgold, Robert E. B. Hanna, and Mark W. Robinson. 2022. "Fasciola hepatica Gastrodermal Cells Selectively Release Extracellular Vesicles via a Novel Atypical Secretory Mechanism" International Journal of Molecular Sciences 23, no. 10: 5525. https://doi.org/10.3390/ijms23105525







