The authors wish to make the following corrections to the original publication [1]. In the original publication, there was a mistake in the legend for Figures 1 and 2. In Figures 1 and 2, the structure of Arg-tRNAArg and ATE1 overlooked the citation of the original work from where it was adapted [x]. The missed citation [x] is, therefore, clarified in the corrected legends (below) as reference [2]. The correct legends for Figure 1 and Figure 2 appear below.
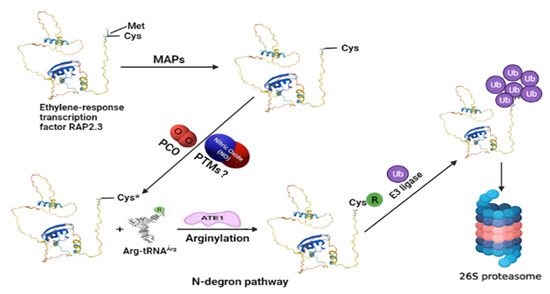
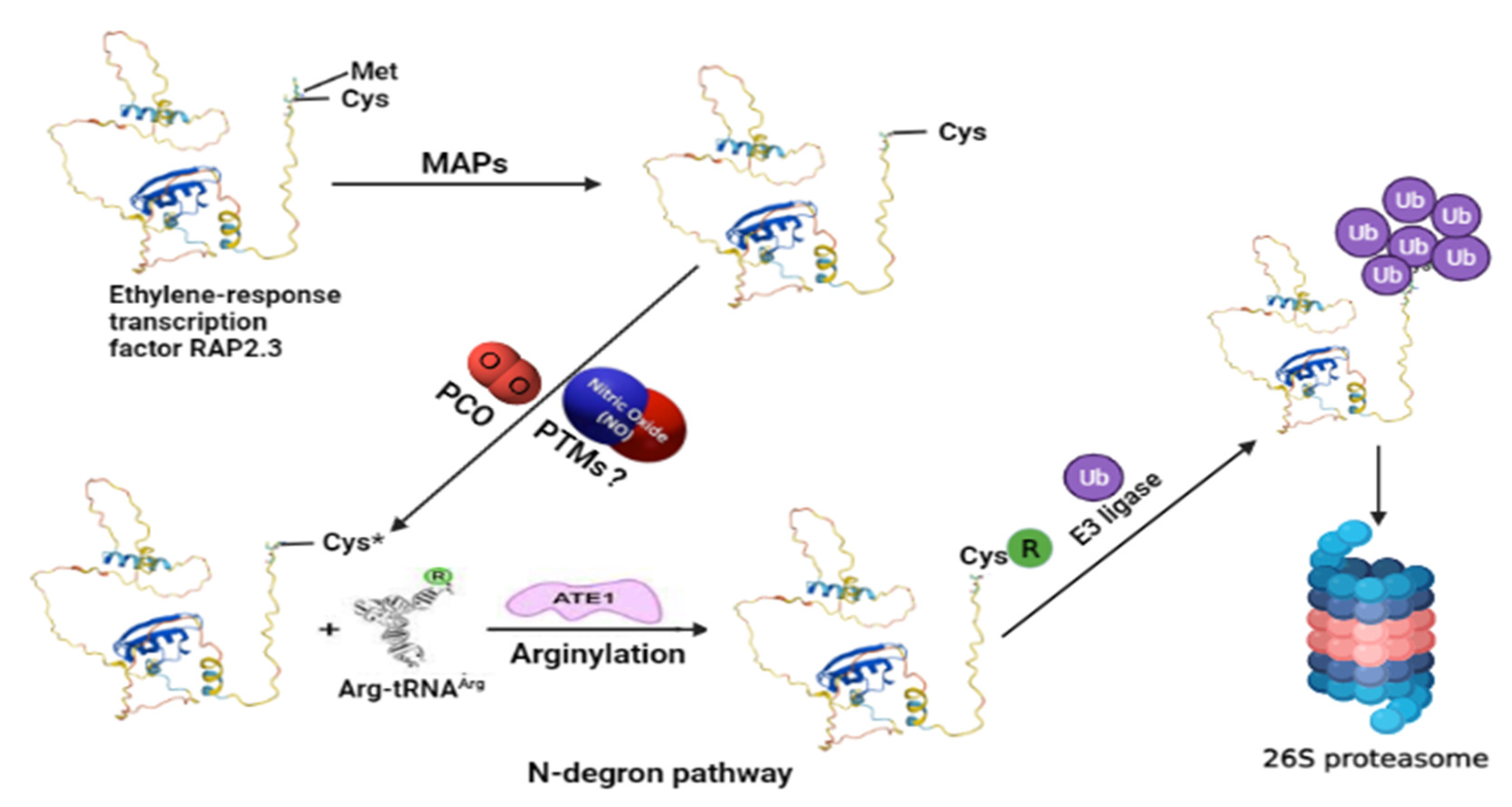
Figure 1.
An exemplified model for understanding the N-degron pathway involving ubiquitin proteasomal degradation of ERF-VII (for example, RELATED TO AP2 3, RAP2.3) TF as a part of the NO sensing mechanism in plants. The N-terminal methionine is cleaved by MAPs exposing the second residue, cysteine (Cys). Cysteine gets oxidized by the action of PCOs. In this step, the role of NO is also reported, so possible NO-mediated oxidation of cysteine residue via PTMs still needs to be explored. The oxidized cysteine (Cys*) of the target protein then undergoes arginylation by Arg-tRNA and is catalyzed by ATE1, which helps in its recognition by PRT6 N-recognin for its ubiquitin-mediated proteolysis. MAPs—aminopeptidases; PCOs—plant cysteine oxidases; PTMs—post-translational modifications; ATE1—arginyl-transferases; PRT6—proteolysis 6 E3 ligases. The model structure of RAP2.3 was obtained from UniProt (https://www.uniprot.org/uniprot/P42736#structure accessed on 19 December 2021). The structure of Arg-tRNAArg and ATE1 is reprinted (adapted) with permission from [2].
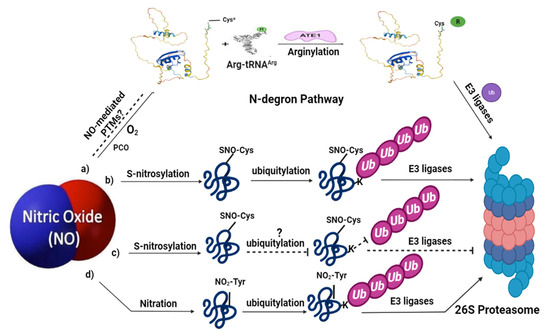
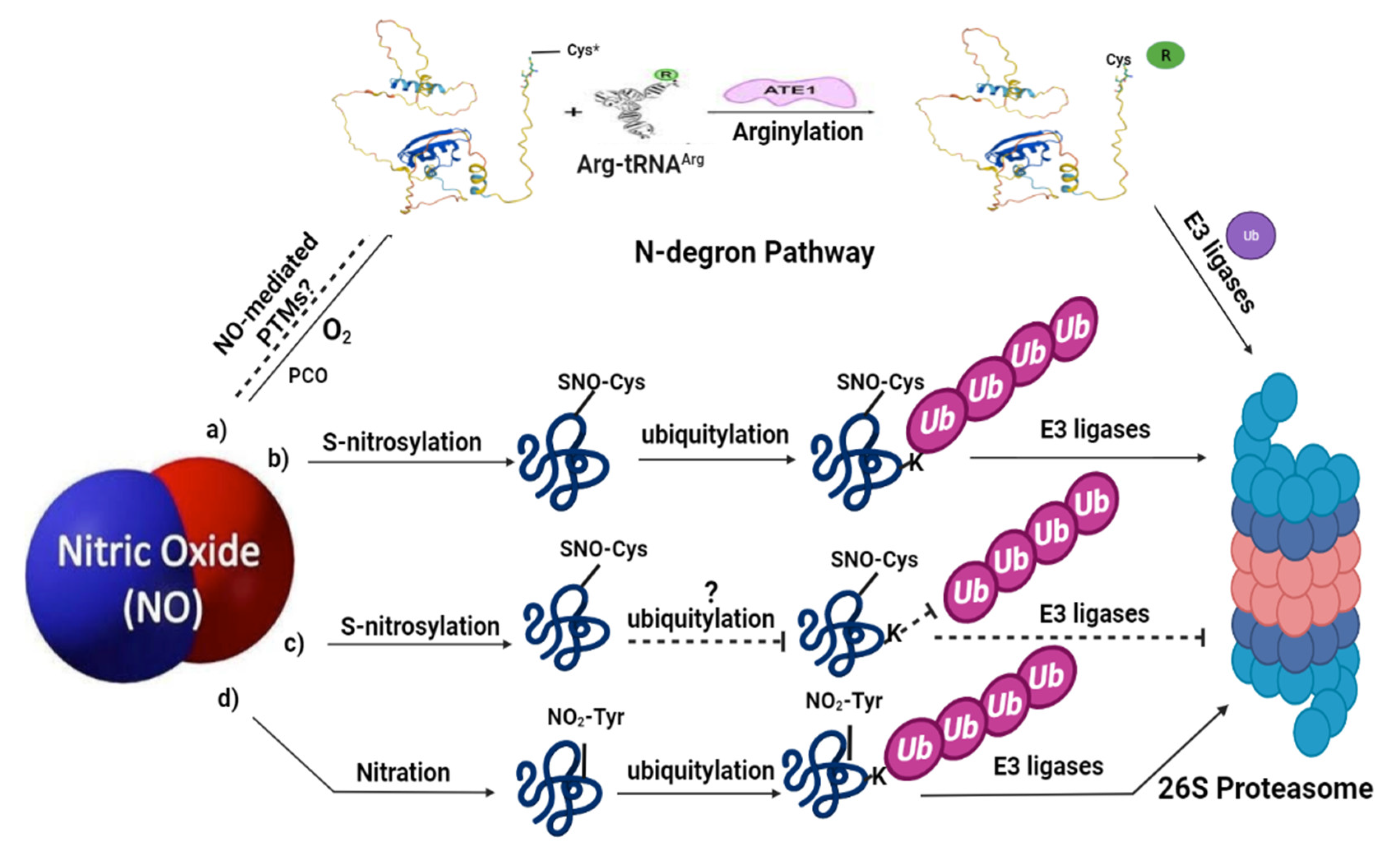
Figure 2.
Nitric oxide signaling associated with ubiquitin-mediated proteolysis in plants: (a) represents the N-degron pathway (already described in detail in Figure 1); (b) under certain environmental conditions, NO can trigger ubiquitin-mediated proteasomal degradation of some proteins via S-nitrosylation, for example, APX1 and ABI5; (c) meanwhile, NO can also protect certain proteins by preventing their degradation via S-nitrosylation; (d) NO-mediated PTMs, such as tyrosine nitration, also leads to the proteolytic degradation via ubiquitin-mediated PTMs. Limited evidence is available for these pathways in plants; therefore, the dashed lines and question marks are used which represent further clarification of these signaling pathways in plants. SNO represents S-nitrosothiol; NO2-Tyr represents tyrosine nitration. The structure of Arg-tRNAArg and ATE1 is reprinted (adapted) with permission from [2].
The authors apologize for any inconvenience caused and state that the scientific conclusions are unaffected. This correction was approved by the Academic Editor. The original publication has also been updated.
References
- Pande, A.; Mun, B.-G.; Khan, M.; Rahim, W.; Lee, D.-S.; Lee, G.-M.; Al Azawi, T.N.I.; Hussain, A.; Yun, B.-W. Nitric Oxide Signaling and Its Association with Ubiquitin-Mediated Proteasomal Degradation in Plants. Int. J. Mol. Sci. 2022, 23, 1657. [Google Scholar] [CrossRef]
- Van, V.; Smith, A.T. ATE1-mediated post-translational arginylation is an essential regulator of eukaryotic cellular homeostasis. ACS Chem. Biol. 2020, 15, 3073–3085. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).