Antisense Expression of Apple TFL1-like Gene (MdTFL1) Promotes Early Flowering and Causes Phenotypic Changes in Tobacco
Abstract
:1. Introduction
2. Results
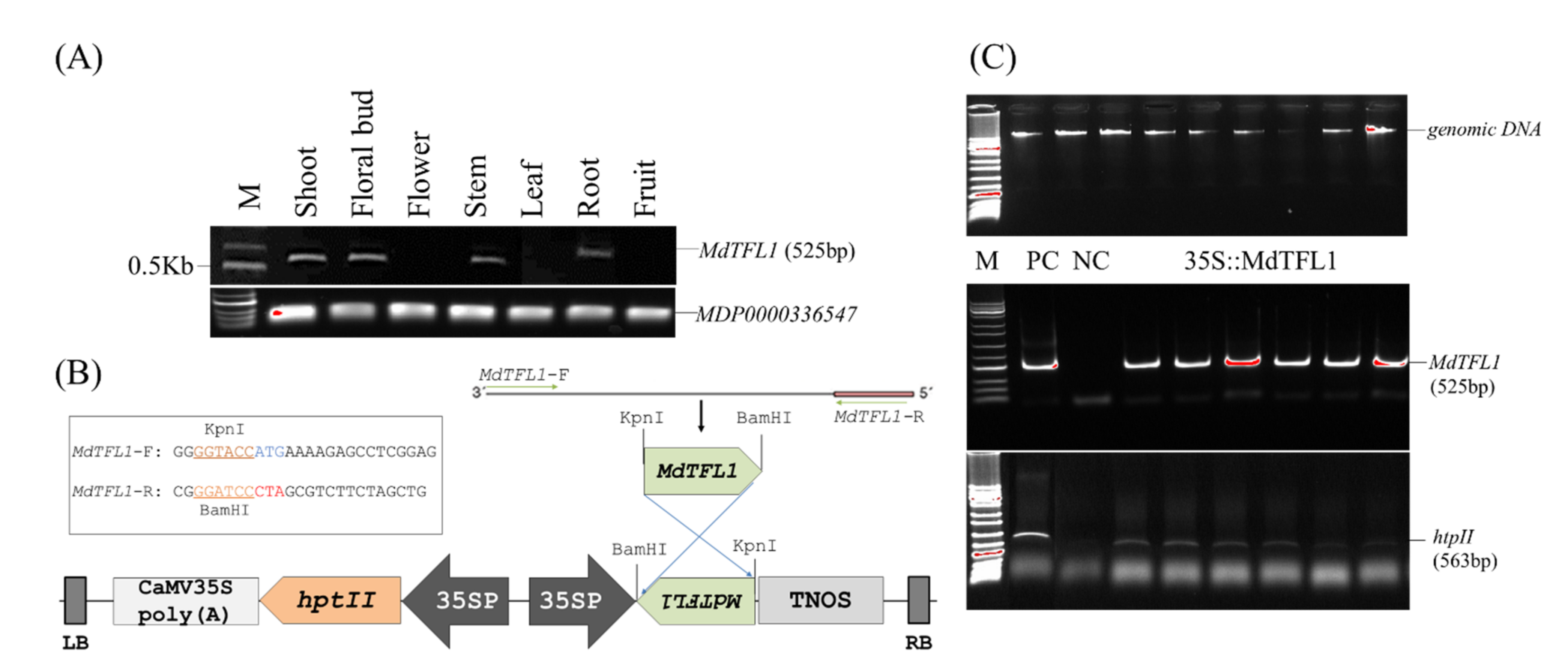
2.1. Construction of Expression Vector
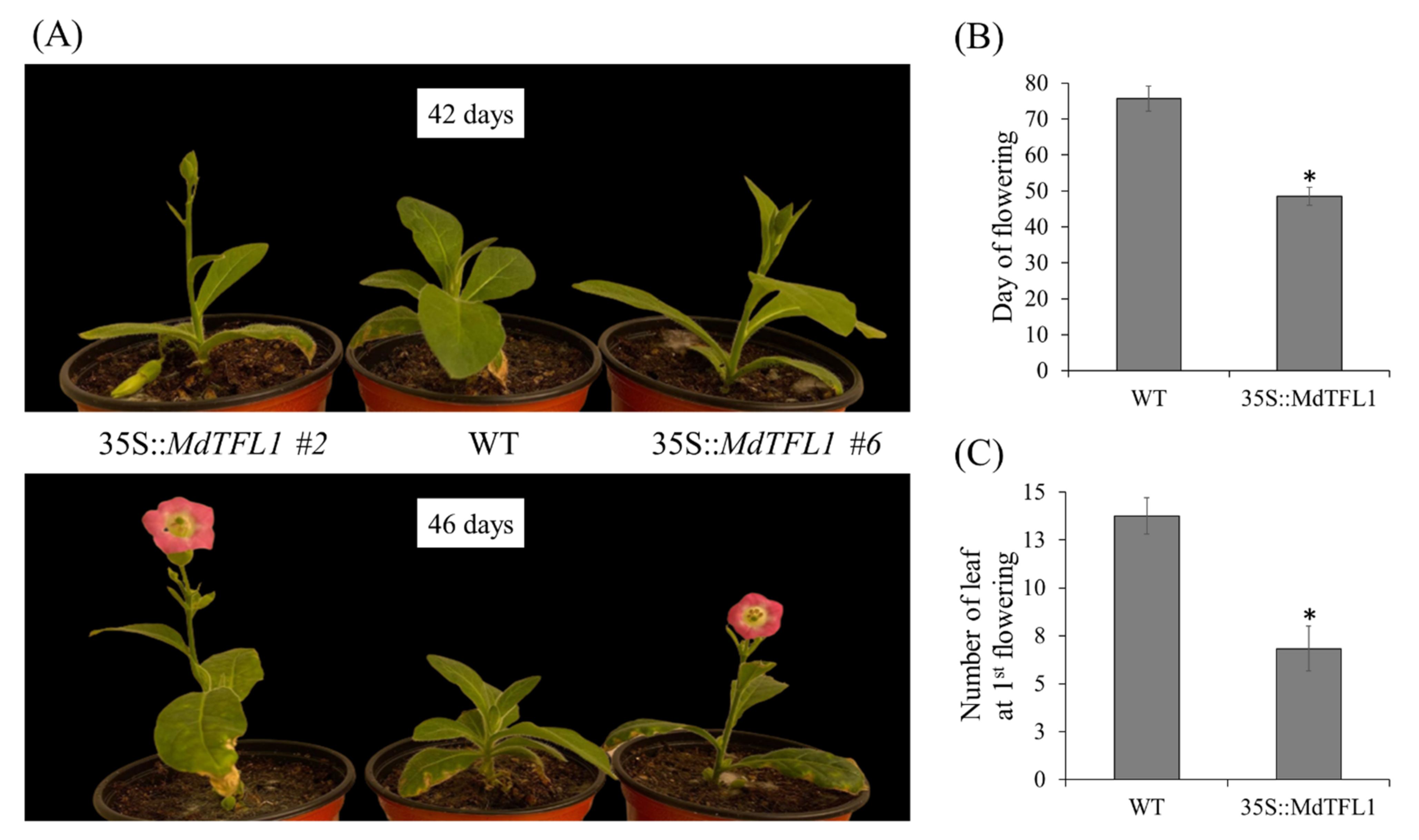
2.2. Antisense Expression of MdTFL1 Promoted Early Flowering
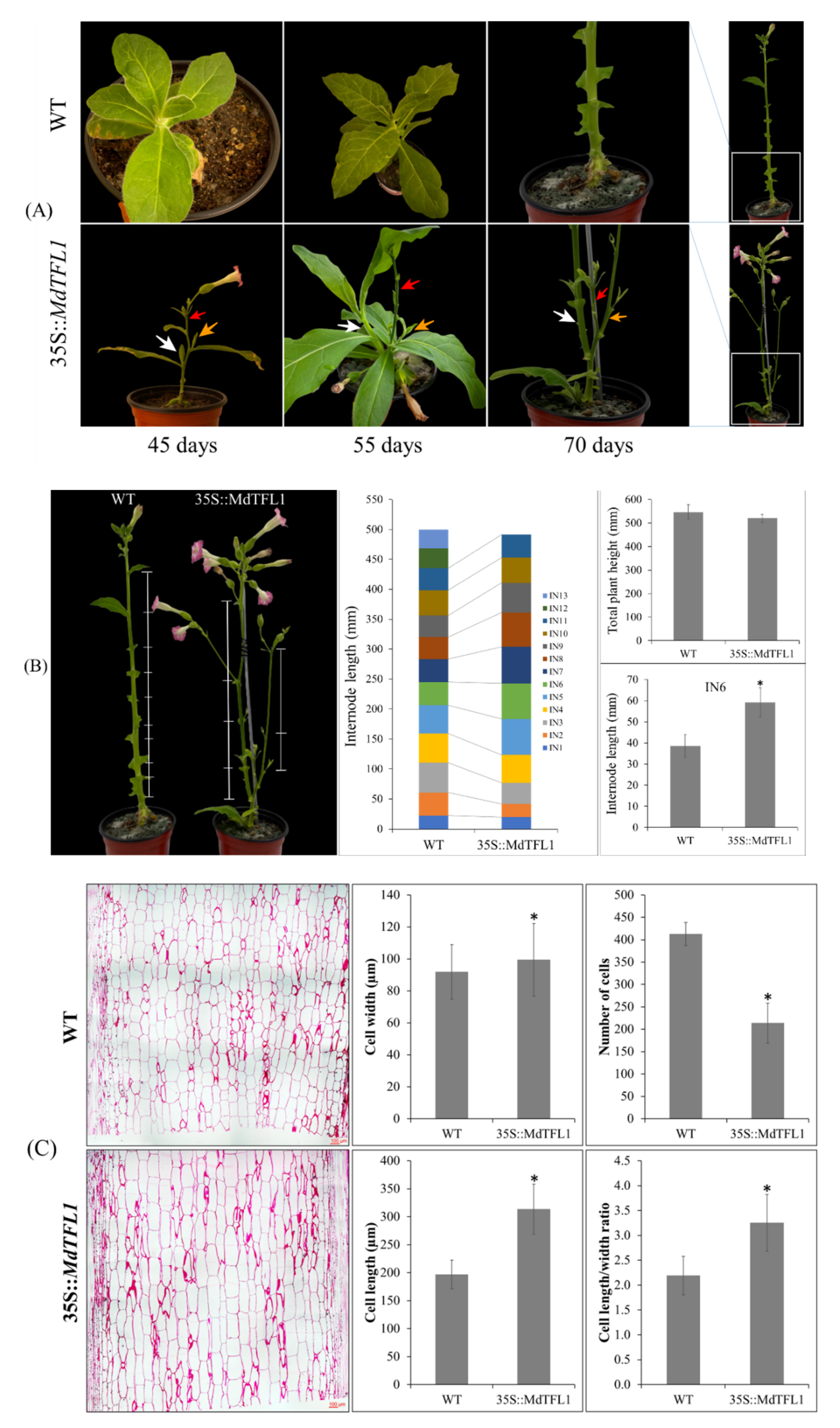
2.3. Antisense Expression of MdTFL1 Induced Lateral Shoot Outgrowth and Affected Plant Architecture
2.4. Antisense Expression of MdTFL1 Influences Leaf Morphology
2.5. Antisense Expression of MdTFL1 Influenced Flower Morphology
2.6. Antisense Expression of MdTFL1 Influenced Set Seed/Fruit in Tobacco
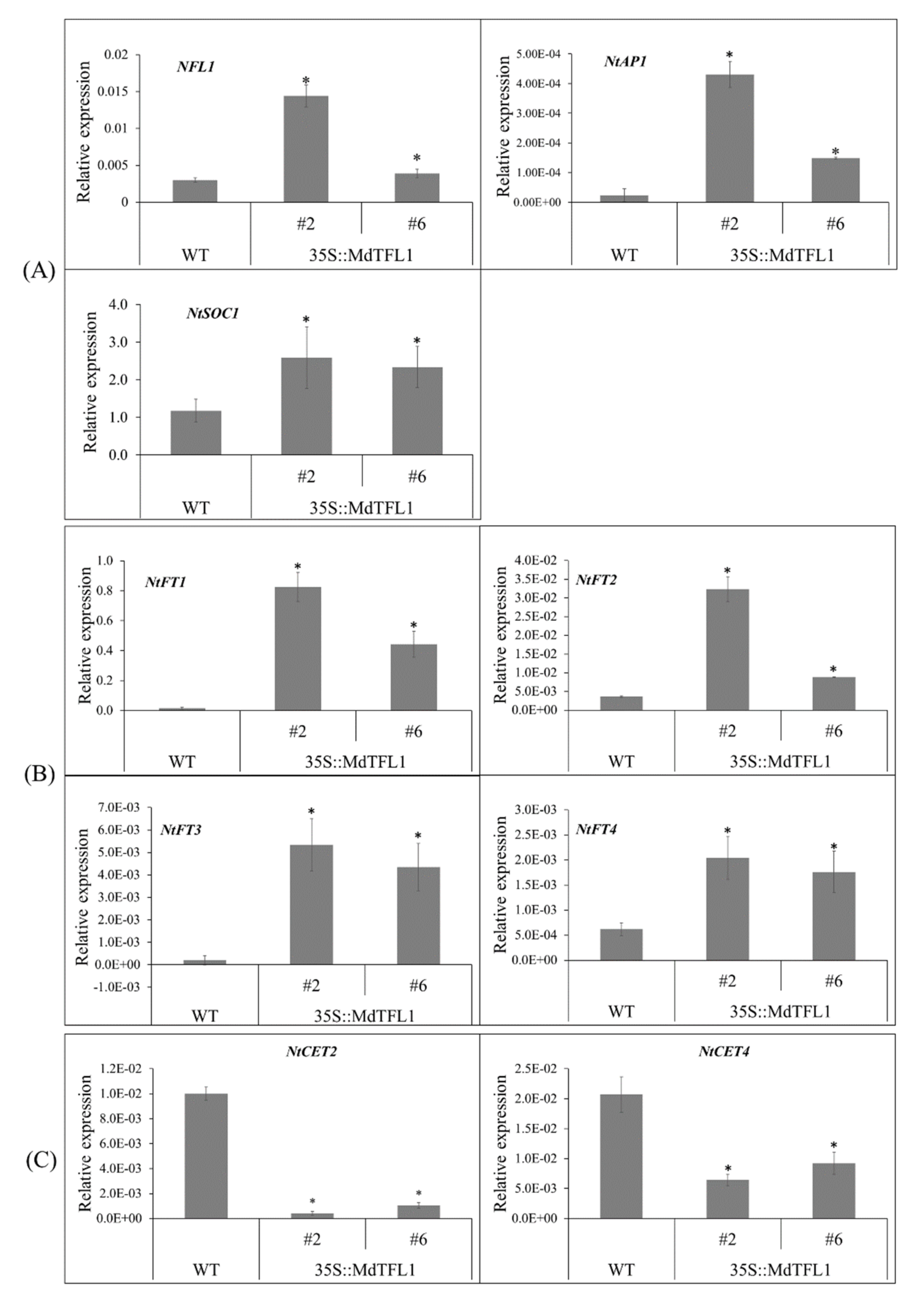
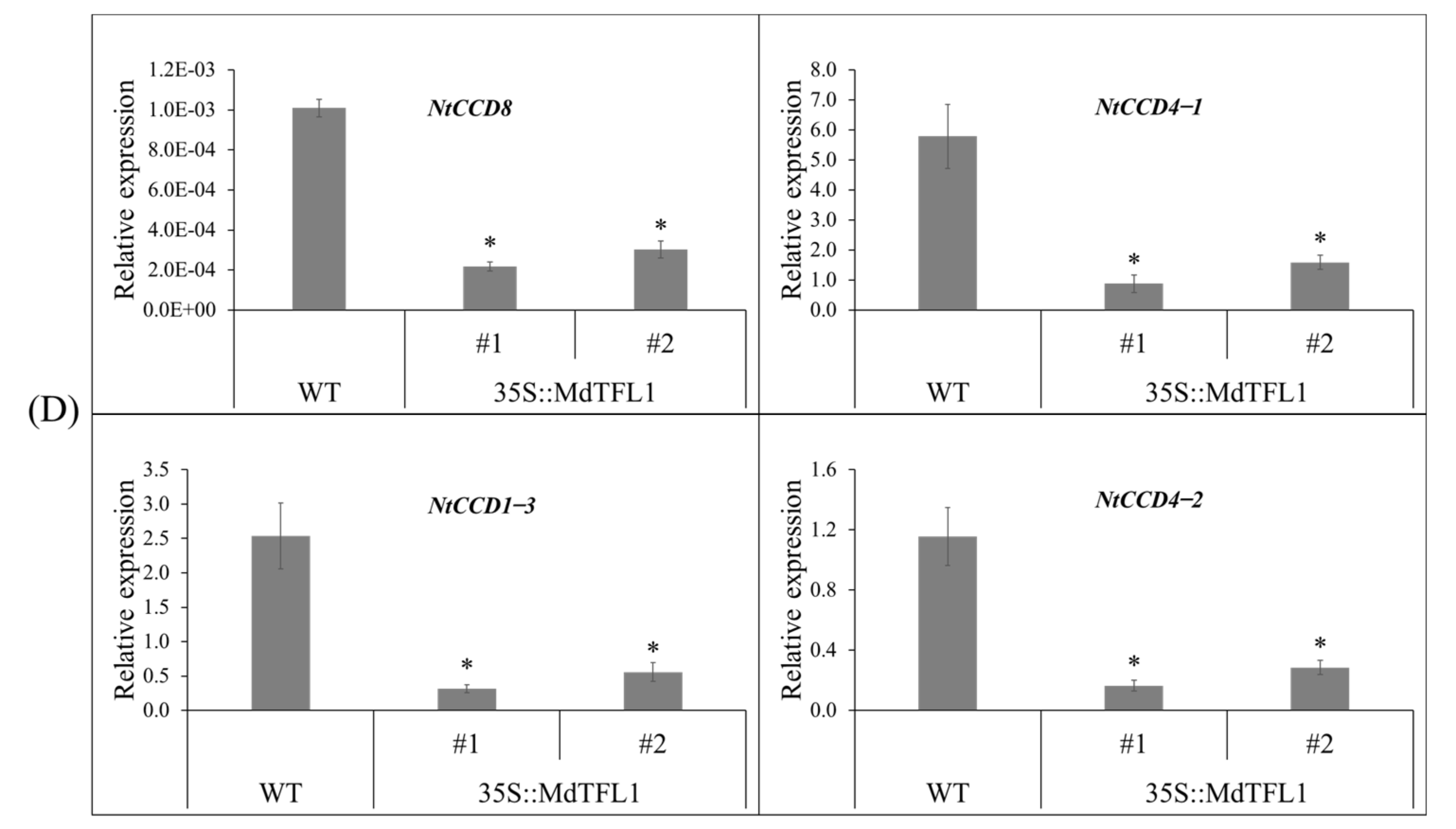
2.7. Antisense Expression of MdTFL1 Altered the Expression of Genes Involved in Metabolic Pathways for Flowering and Branching
3. Discussion
3.1. Antisense Expression of MdTFL1 Promoted Early Flowering by Disturbing the Expression Level of Floral Regulator Genes
3.2. Antisense Expression of MdTFL1 Gene Not Only Promoted Early Flowering but Also Led to Phenotypic Changes in Transgenic Plants
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
4.2. Construction of Overexpression Vectors
4.3. Tobacco Transformation and Establishment of Transgenic Plant
4.4. Genomic DNA Extraction and Genomic DNA PCR
4.5. RNA Extraction and Quantification of Gene Expression
4.6. Measurement of Leaf Area and Leaf Mass per Area (LMA)
4.7. Light Microscopy
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hackett, W.P. Juvenility, maturation, and rejuvenation in woody plants. Hortic. Rev. 2011, 7, 109–154. [Google Scholar]
- Matsoukas, I.G. Attainment of reproductive competence, phase transition, and quantification of juvenility in mutant genetic screens. Front. Plant Sci. 2014, 5, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahn, J.H.; Miller, D.; Winter, V.J.; Banfield, M.J.; Lee, J.H.; Yoo, S.Y.; Henz, S.R.; Brady, R.L.; Weigel, D. A divergent external loop confers antagonistic activity on floral regulators FT and TFL1. EMBO J. 2006, 25, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Putterill, J.; Varkonyi-Gasic, E. FT and florigen long-distance flowering control in plants. Curr. Opin. Plant Biol. 2016, 33, 77–82. [Google Scholar] [CrossRef]
- Hanano, S.; Goto, K. Arabidopsis terminal FLOWER1 is involved in the regulation of flowering time and inflorescence development through transcriptional repression. Plant Cell 2011, 23, 3172–3184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moraes, T.S.; Dornelas, M.C.; Martinelli, A.P. FT/TFL1: Calibrating plant architecture. Front. Plant Sci. 2019, 10, 97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wickland, D.P.; Hanzawa, Y. The FLOWERING LOCUS T/TERMINAL FLOWER 1 gene family: Functional evolution and molecular mechanisms. Mol. Plant 2015, 8, 983–997. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Y.; Klasfeld, S.; Jeong, C.W.; Jin, R.; Goto, K.; Yamaguchi, N.; Wagner, D. Terminal FLOWER 1-FD complex target genes and competition with FLOWERING LOCUS T. Nat. Commun. 2020, 11, 5118. [Google Scholar] [CrossRef]
- Yant, L.; Mathieu, J.; Schmid, M. Just say no: Floral repressors help Arabidopsis bide the time. Curr. Opin. Plant Biol. 2009, 12, 580–586. [Google Scholar] [CrossRef]
- Bradley, D.; Ratcliffe, O.; Vincent, C.; Carpenter, R.; Coen, E. Inflorescence commitment and architecture in Arabidopsis. Science 1997, 275, 80–83. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Hu, W.; Wang, L.; Lin, C.; Cong, B.; Sun, C.; Luo, D. TFL1/CEN-like genes control intercalary meristem activity and phase transition in rice. Plant Sci. 2005, 168, 1393–1408. [Google Scholar] [CrossRef]
- Koskela, E.A.; Sønsteby, A.; Flachowsky, H.; Heide, O.M.; Hanke, M.V.; Elomaa, P.; Hytönen, T. TERMINAL FLOWER 1 is a breeding target for a novel everbearing trait and tailored flowering responses in cultivated strawberry (Fragaria × ananassa Duch.). Plant Biotechnol. J. 2016, 14, 1852–1861. [Google Scholar] [CrossRef] [Green Version]
- Iwata, H.; Gaston, A.; Remay, A.; Thouroude, T.; Jeauffre, J.; Kawamura, K.; Oyant, L.H.S.; Araki, T.; Denoyes, B.; Foucher, F. The TFL1 homologue KSN is a regulator of continuous flowering in rose and strawberry. Plant J. 2012, 69, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Kotoda, N.; Iwanami, H.; Takahashi, S.; Abe, K. Antisense expression of MdTFL1, a TFL1-like gene, reduces the juvenile phase in apple. J. Am. Soc. Hortic. Sci. 2006, 131, 74–81. [Google Scholar] [CrossRef] [Green Version]
- Kotoda, N.; Wada, M. MdTFL1, a TFL1-like gene of apple, retards the transition from the vegetative to reproductive phase in transgenic Arabidopsis. Plant Sci. 2005, 168, 95–104. [Google Scholar] [CrossRef]
- Esumi, T.; Tao, R.; Yonemori, K. Isolation of LEAFY and TERMINAL FLOWER 1 homologues from six fruit tree species in the subfamily Maloideae of the Rosaceae. Sex. Plant Reprod. 2005, 17, 277–287. [Google Scholar] [CrossRef]
- Zuo, X.; Xiang, W.; Zhang, L.; Gao, C.; An, N.; Xing, L.; Ma, J.; Zhao, C.; Zhang, D. Identification of apple TFL1-interacting proteins uncovers an expanded flowering network. Plant Cell Rep. 2021, 40, 2325–2340. [Google Scholar] [CrossRef]
- Patil, V.; McDermott, H.I.; McAllister, T.; Cummins, M.; Silva, J.C.; Mollison, E.; Meikle, R.; Morris, J.; Hedley, P.; Waugh, R.; et al. APETALA2 control of barley internode elongation. Development 2019, 146, dev170373. [Google Scholar] [CrossRef] [Green Version]
- Li, C.; Zhang, Y.; Zhang, K.; Guo, D.; Cui, B.; Wang, X.; Huang, X. Promoting flowering, lateral shoot outgrowth, leaf development, and flower abscission in tobacco plants overexpressing cotton FLOWERING LOCUS T (FT)-like gene GhFT1. Front. Plant Sci. 2015, 6, 454. [Google Scholar] [CrossRef] [Green Version]
- Umehara, M.; Hanada, A.; Yoshida, S.; Akiyama, K.; Arite, T.; Takeda-Kamiya, N.; Magome, H.; Kamiya, Y.; Shirasu, K.; Yoneyama, K.; et al. Inhibition of shoot branching by new terpenoid plant hormones. Nature 2008, 455, 195–200. [Google Scholar] [CrossRef]
- Beveridge, C.A.; Kyozuka, J. New genes in the strigolactone-related shoot branching pathway. Curr. Opin. Plant Biol. 2010, 13, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Ahearn, K.P.; Johnson, H.A.; Weigel, D.; Wagner, D.R. NFL1, a Nicotiana tabacum LEAFY-like gene, controls meristem initiation and floral structure. Plant Cell Physiol. 2001, 42, 1130–1139. [Google Scholar] [CrossRef] [Green Version]
- Kim, N.H.; Han, M.S.; Cho, H.W.; Kim, D.S.; Kim, H.-J.; Kim, B.-D. Molecular cloning of the CaLFY, putative pepper ortholog of FLO/LFY. Mol. Breed. 2008, 22, 443–453. [Google Scholar] [CrossRef]
- Mandel, M.A.; Gustafson-Brown, C.; Savidge, B.; Yanofsky, M.F. Molecular characterization of the Arabidopsis floral homeotic gene APETALA1. Nature 1992, 360, 273–277. [Google Scholar] [CrossRef]
- Zheng, T.; Li, S.; Zang, L.; Dai, L.; Yang, C.; Qu, G.Z. Overexpression of two PsnAP1 genes from Populus simonii × P. nigra causes early flowering in transgenic tobacco and Arabidopsis. PLoS ONE 2014, 9, e111725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smykal, P.; Gennen, J.; De Bodt, S.; Ranganath, V.; Melzer, S. Flowering of strict photoperiodic Nicotiana varieties in non-inductive conditions by transgenic approaches. Plant Mol. Biol. 2007, 65, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Li, X.; Qin, D.; Guo, F.; Wu, C.; Miao, L.; Sun, C. Functional analysis of FLOWERING LOCUS T orthologs from spring orchid (Cymbidium goeringii Rchb. f.) that regulates the vegetative to reproductive transition. Plant Physiol. Biochem. 2012, 58, 98–105. [Google Scholar] [CrossRef]
- Corbesier, L.; Vincent, C.; Jang, S.; Fornara, F.; Fan, Q.; Searle, I.; Giakountis, A.; Farrona, S.; Gissot, L.; Turnbull, C.; et al. FT protein movement contributes to long-distance signaling in floral induction of Arabidopsis. Science 2007, 316, 1030–1033. [Google Scholar] [CrossRef] [Green Version]
- Abe, M.; Kobayashi, Y.; Yamamoto, S.; Daimon, Y.; Yamaguchi, A.; Ikeda, Y.; Ichinoki, H.; Notaguchi, M.; Goto, K.; Araki, T. FD, a bZIP protein mediating signals from the floral pathway integrator FT at the shoot apex. Science 2005, 309, 1052–1056. [Google Scholar] [CrossRef]
- Amaya, I.; Ratcliffe, O.J.; Bradley, D.J. Expression of CENTRORADIALIS (CEN) and CEN-like genes in tobacco reveals a conserved mechanism controlling phase change in diverse species. Plant Cell 1999, 11, 1405–1418. [Google Scholar] [CrossRef] [Green Version]
- Gómez-Soto, D.; Allona, I.; Perales, M. FLOWERING LOCUS T2 promotes shoot apex development and restricts internode elongation via the 13-hydroxylation gibberellin biosynthesis pathway in poplar. Front. Plant Sci. 2021, 12, 814195. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, K.; Yamaguchi, A.; Abe, M.; Araki, T. The florigen genes FT and TSF modulate lateral shoot outgrowth in Arabidopsis thaliana. Plant Cell Physiol. 2013, 54, 352–368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niwa, M.; Endo, M.; Araki, T. Florigen is involved in axillary bud development at multiple stages in Arabidopsis. Plant Signal. Behav. 2013, 8, e27167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teper-Bamnolker, P.; Samach, A. The flowering integrator FT regulates SEPALLATA3 and FRUITFULL accumulation in Arabidopsis leaves. Plant Cell 2005, 17, 2661–2675. [Google Scholar] [CrossRef] [Green Version]
- Irish, V. The ABC model of floral development. Curr. Biol. 2017, 27, R887–R890. [Google Scholar] [CrossRef] [Green Version]
- Pelaz, S.; Ditta, G.S.; Baumann, E.; Wisman, E.; Yanofsky, M.F. B and C floral organ identity functions require SEPALLATA MADS-box genes. Nature 2000, 405, 200–203. [Google Scholar] [CrossRef]
- Kim, S.; Koh, J.; Yoo, M.J.; Kong, H.; Hu, Y.; Ma, H.; Soltis, P.S.; Soltis, D.E. Expression of floral MADS-box genes in basal angiosperms: Implications for the evolution of floral regulators. Plant J. 2005, 43, 724–744. [Google Scholar] [CrossRef]
- Kanno, A.; Saeki, H.; Kameya, T.; Saedler, H.; Theissen, G. Heterotopic expression of class B floral homeotic genes supports a modified ABC model for tulip (Tulipa gesneriana). Plant Mol. Biol. 2003, 52, 831–841. [Google Scholar] [CrossRef]
- Wollmann, H.; Mica, E.; Todesco, M.; Long, J.A.; Weigel, D. On reconciling the interactions between APETALA2, miR172 and AGAMOUS with the ABC model of flower development. Development 2010, 137, 3633–3642. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Sun, Y.; Yu, X.; Li, H.; Bao, M.; He, Y. Functional conservation and divergence of five AP1/FUL-like genes in marigold (Tagetes erecta L.). Genes 2021, 12, 2011. [Google Scholar] [CrossRef]
- Zhang, J.; Yan, G.; Wen, Z.; An, Y.Q.; Singer, S.D.; Liu, Z. Two tobacco AP1-like gene promoters drive highly specific, tightly regulated and unique expression patterns during floral transition, initiation and development. Planta 2014, 239, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.C.; Pan, Z.J.; Su, Y.Y.; Liu, Z.J. New insight into the regulation of floral morphogenesis. Int. Rev. Cell Mol. Biol. 2014, 311, 157–182. [Google Scholar]
- Gwak, Y.S.; Han, J.Y.; Adhikari, P.B.; Ahn, C.H.; Choi, Y.E. Heterologous production of a ginsenoside saponin (compound K) and its precursors in transgenic tobacco impairs the vegetative and reproductive growth. Planta 2017, 245, 1105–1119. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.; Puryear, J.; Cairney, J. A simple and efficient method for isolating RNA from pine trees. Plant Mol. Biol. Rep. 1993, 11, 113–116. [Google Scholar] [CrossRef]
- Horsch, R.B.; Fry, J.E.; Hoffmann, N.L.; Wallroth, M.; Eichholtz, D.; Rogers, S.G.; Fraley, R.T. A simple and general method for transferring genes into plants. Science 1985, 227, 1229–1231. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Do, V.G.; Kim, S.; Kweon, H. Identification of genes associated with nitrogen stress responses in apple leaves. Plants 2021, 10, 2649. [Google Scholar] [CrossRef] [PubMed]
- Clément, C.; Burrus, M.; Audran, J.C. Floral organ growth and carbohydrate content during pollen development in Lilium. Am. J. Bot. 1996, 83, 459–469. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Do, V.G.; Lee, Y.; Kim, S.; Kweon, H.; Do, G. Antisense Expression of Apple TFL1-like Gene (MdTFL1) Promotes Early Flowering and Causes Phenotypic Changes in Tobacco. Int. J. Mol. Sci. 2022, 23, 6006. https://doi.org/10.3390/ijms23116006
Do VG, Lee Y, Kim S, Kweon H, Do G. Antisense Expression of Apple TFL1-like Gene (MdTFL1) Promotes Early Flowering and Causes Phenotypic Changes in Tobacco. International Journal of Molecular Sciences. 2022; 23(11):6006. https://doi.org/10.3390/ijms23116006
Chicago/Turabian StyleDo, Van Giap, Youngsuk Lee, Seonae Kim, Hunjoong Kweon, and Gyungran Do. 2022. "Antisense Expression of Apple TFL1-like Gene (MdTFL1) Promotes Early Flowering and Causes Phenotypic Changes in Tobacco" International Journal of Molecular Sciences 23, no. 11: 6006. https://doi.org/10.3390/ijms23116006





