A TIR-NBS-LRR Gene MdTNL1 Regulates Resistance to Glomerella Leaf Spot in Apple
Abstract
:1. Introduction
2. Results
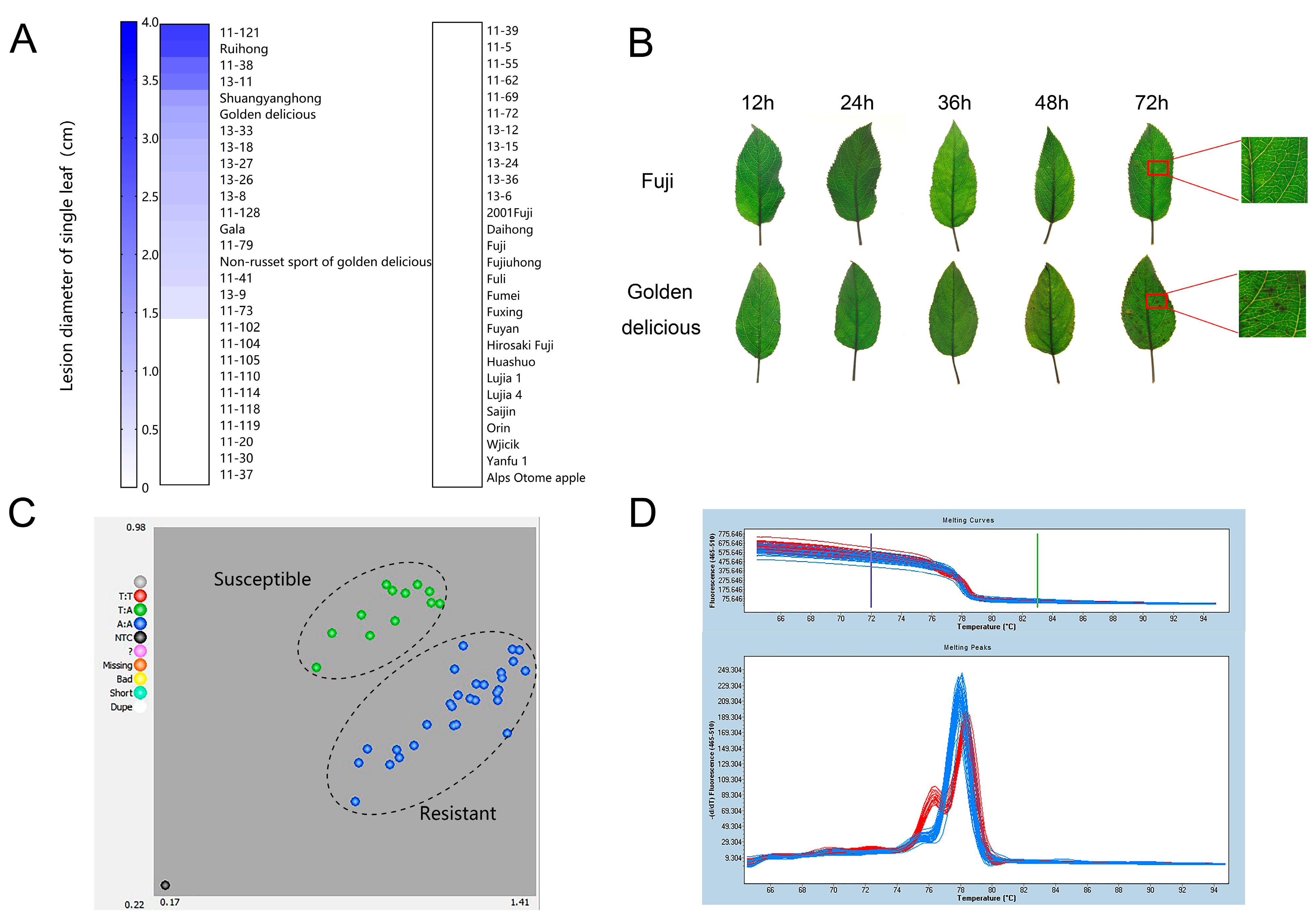
2.1. GLS Resistance Evaluation of Apple Cultivars
2.2. SNP7309212 Sites Show Polymorphism in Resistant and Susceptible Apple Germplasms
2.3. Cloning and Characterization of GLS-Resistant Gene
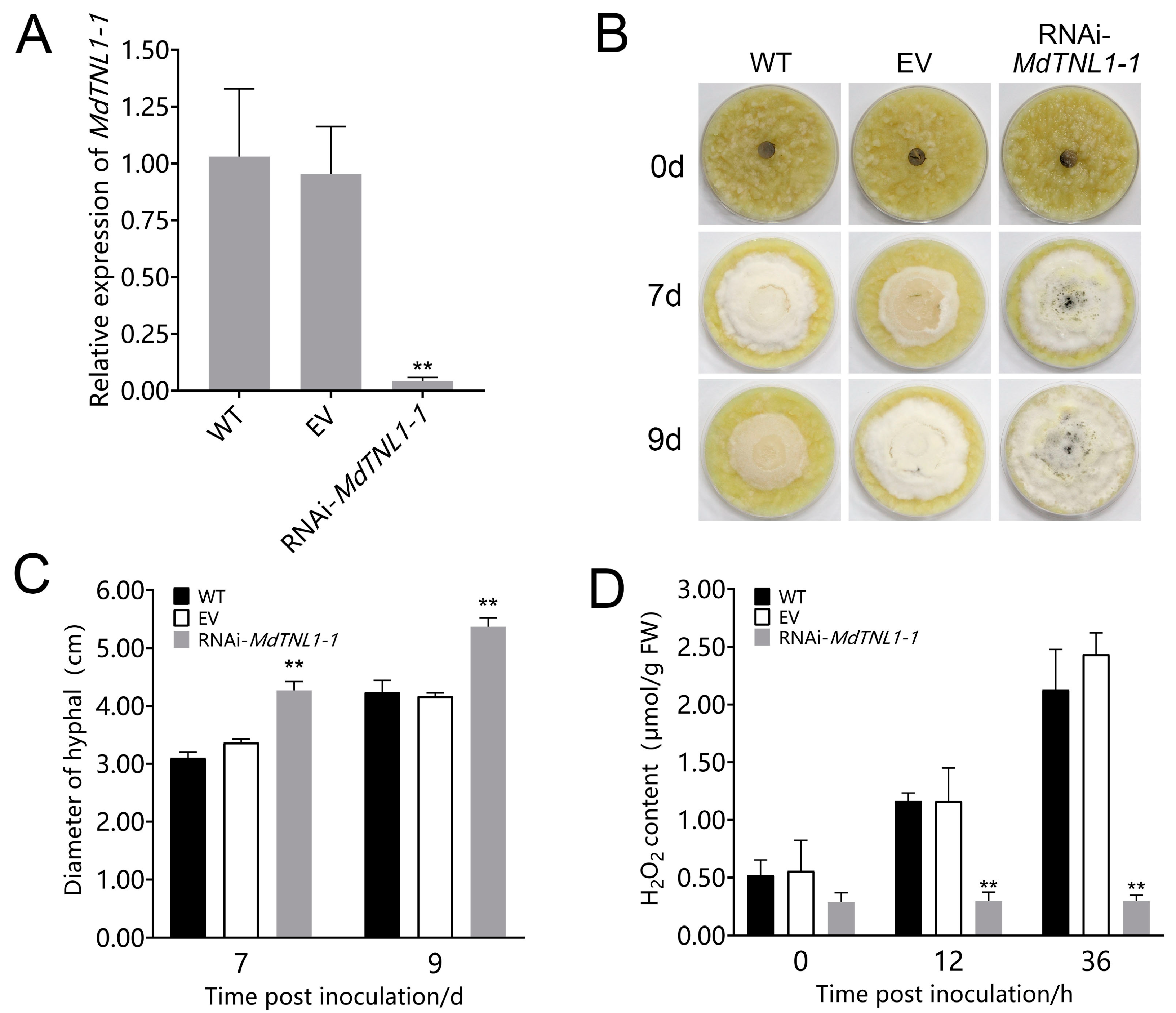
2.4. MdTNL1-1 Was Required for the Resistance of Apple Plant to C. fructicola
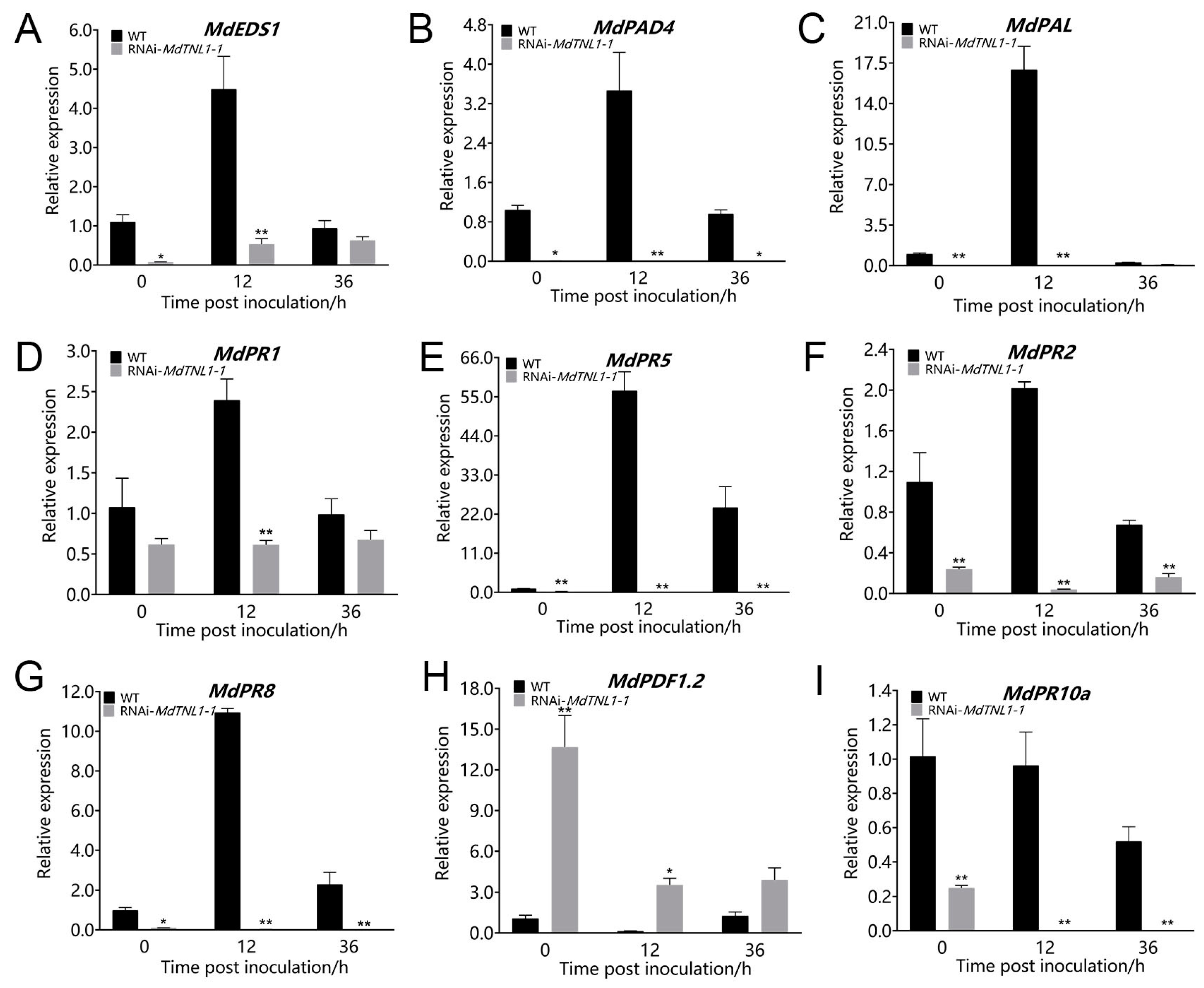
2.5. MdTNL1-1 Influenced the Expression of PR Genes, and SA and JA Signaling
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
4.2. Pathogen Inoculation and Resistance Evaluation
4.3. DNA Extraction and HRM Analysis
4.4. KASP Analysis
4.5. RNA Extraction and qRT-PCR
4.6. Cloning of MdTNL1
4.7. Transient Expression Assay in Tobacco Leaves, Apple Leaves, and Fruits
4.8. Subcellular Localization of MdTNL1-1 and Genetic Transformation of Apple Calli
4.9. Expression Analysis of PR Genes and SA- and JA-Related Genes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, Y.; Lan, J.; Li, Q.; Zhang, Y.; Wang, C.; Dai, H. Rapid location of Glomerella leaf spot resistance gene locus in apple by whole genome re-sequencing. Mol. Breed. 2017, 37, 96. [Google Scholar] [CrossRef]
- Wang, C.X.; Zhang, Z.F.; Li, B.H.; Wang, H.Y.; Dong, X.L. First Report of Glomerella Leaf Spot of Apple Caused by Glomerella cingulata in China. Plant Dis. 2012, 96, 912. [Google Scholar] [CrossRef]
- Liu, Y.X.; Li, B.H.; Wang, C.H.; Liu, C.X.; Kong, X.H.; Zhu, J.; Dao, H.Y. Genetics and molecular marker identification of a resistance to Glomerella leaf spot in apple. Hortic. Plant J. 2016, 2, 121–125. [Google Scholar] [CrossRef] [Green Version]
- Taylor, J. A Necrotic Leaf Blotch and Fruit Rot of Apple Caused by a Strain of Glomerella cingulata. Phytopathology 1971, 61, 221–224. [Google Scholar] [CrossRef]
- Alaniz, S.; Cuozzo, V.; Martínez, V.; Stadnik, M.J.; Mondino, P. Ascospore Infection and Colletotrichum Species Causing Glomerella Leaf Spot of Apple in Uruguay. Plant Pathol. J. 2019, 35, 100–111. [Google Scholar] [CrossRef]
- Wang, B.; Li, B.-H.; Dong, X.-L.; Wang, C.-X.; Zhang, Z.-F. Effects of Temperature, Wetness Duration, and Moisture on the Conidial Germination, Infection, and Disease Incubation Period of Glomerella cingulata. Plant Dis. 2015, 99, 249–256. [Google Scholar] [CrossRef] [Green Version]
- Velho, A.C.; Rockenbach, M.F.; Mondino, P.; Stadnik, M.J. Modulation of oxidative responses by a virulent isolate of Colletotrichum fructicola in apple leaves. Fungal Biol. 2016, 120, 1184–1193. [Google Scholar] [CrossRef] [PubMed]
- Shang, S.; Wang, B.; Zhang, S.; Liu, G.; Liang, X.; Zhang, R.; Gleason, M.L.; Sun, G. A novel effector CfEC92 of Colletotrichum fructicola contributes to glomerella leaf spot virulence by suppressing plant defences at the early infection phase. Mol. Plant Pathol. 2020, 21, 936–950. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Zhang, Q.; Hao, L.; Wang, S.; Wang, S.; Zhang, W.; Xu, C.; Yu, Y.; Li, T. A novel miRNA negatively regulates resistance to Glomerella leaf spot by suppressing expression of an NBS gene in apple. Hortic. Res. 2019, 6, 93. [Google Scholar] [CrossRef] [Green Version]
- Shan, D.; Wang, C.; Zheng, X.; Hu, Z.; Zhu, Y.; Zhao, Y.; Jiang, A.; Zhang, H.; Shi, K.; Bai, Y.; et al. MKK4-MPK3-WRKY17-mediated salicylic acid degradation increases susceptibility to Glomerella leaf spot in apple. Plant Physiol. 2021, 186, 1202–1219. [Google Scholar] [CrossRef] [PubMed]
- Ngou, B.P.M.; Ahn, H.-K.; Ding, P.; Redkar, A.; Brown, H.; Ma, Y.; Youles, M.; Tomlinson, L.; Jones, J.D.G. Estradiol-inducible AvrRps4 expression reveals distinct properties of TIR-NLR-mediated effector-triggered immunity. J. Exp. Bot. 2020, 71, 2186–2197. [Google Scholar] [CrossRef]
- Jung, H.W.; Panigrahi, G.K.; Jung, G.Y.; Lee, Y.J.; Shin, K.H.; Sahoo, A.; Choi, E.S.; Lee, E.; Kim, K.M.; Yang, S.H.; et al. Pathogen-Associated Molecular Pattern-Triggered Immunity Involves Proteolytic Degradation of Core Nonsense-Mediated mRNA Decay Factors During the Early Defense Response. Plant Cell 2020, 32, 1081–1101. [Google Scholar] [CrossRef] [Green Version]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef] [Green Version]
- Funk, A.; Galewski, P.; McGrath, J.M. Nucleotide-binding resistance gene signatures in sugar beet, insights from a new reference genome. Plant J. 2018, 95, 659–671. [Google Scholar] [CrossRef] [PubMed]
- Gassmann, W.; Hinsch, M.E.; Staskawicz, B.J. The Arabidopsis RPS4 bacterial-resistance gene is a member of the TIR-NBS-LRR family of disease-resistance genes. Plant J. 1999, 20, 265–277. [Google Scholar] [CrossRef]
- Li, X.; Zhang, Y.; Yin, L.; Lu, J. Overexpression of pathogen-induced grapevine TIR-NB-LRR gene VaRGA1 enhances disease resistance and drought and salt tolerance in Nicotiana benthamiana. Protoplasma 2017, 254, 957–969. [Google Scholar] [CrossRef]
- Zhao, Y.; Huang, J.; Wang, Z.; Jing, S.; Wang, Y.; Ouyang, Y.; Cai, B.; Xin, X.-F.; Liu, X.; Zhang, C.; et al. Allelic diversity in an NLR gene BPH9 enables rice to combat planthopper variation. Proc. Natl. Acad. Sci. USA 2016, 113, 12850–12855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galli, P.; Patocchi, A.; Broggini, G.A.L.; Gessler, C. The Rvi15 (Vr2) Apple Scab Resistance Locus Contains Three TIR-NBS-LRR Genes. Mol. Plant-Microbe Interact. 2010, 23, 608–617. [Google Scholar] [CrossRef] [Green Version]
- Parravicini, G.; Gessler, C.; Denancé, C.; Lasserre-Zuber, P.; Vergne, E.; Brisset, M.-N.; Patocchi, A.; Durel, C.-E.; Broggini, G. Identification of serine/threonine kinase and nucleotide-binding site-leucine-rich repeat (NBS-LRR) genes in the fire blight resistance quantitative trait locus of apple cultivar ‘Evereste’. Mol. Plant Pathol. 2012, 12, 493–505. [Google Scholar] [CrossRef] [Green Version]
- Dunemann, F.; Peil, A.; Urbanietz, A.; Garcia-Libreros, T. Mapping of the apple powdery mildew resistance gene Pl1 and its genetic association with an NBS-LRR candidate resistance gene. Plant Breed. 2007, 126, 476–481. [Google Scholar] [CrossRef]
- Liu, Y.; Lan, J.; Wang, C.; Li, B.; Zhu, J.; Liu, C.; Dai, H. Investigation and genetic mapping of a Glomerella leaf spot resistance locus in apple. Plant Breed. 2017, 136, 119–125. [Google Scholar] [CrossRef] [Green Version]
- Diaz-Vivancos, P.; Bernal-Vicente, A.; Cantabella, D.; Petri, C.; Hern�Ndez, J.A. Metabolomics and Biochemical Approaches Link Salicylic Acid Biosynthesis to Cyanogenesis in Peach Plants. Plant Cell Physiol. 2017, 58, 2057–2066. [Google Scholar] [CrossRef] [PubMed]
- Rietz, S.; Stamm, A.; Malonek, S.; Wagner, S.; Becker, D.; Medina-Escobar, N.; Vlot, A.C.; Feys, B.J.; Niefind, K.; Parker, J.E. Different roles of Enhanced Disease Susceptibility1 (EDS1) bound to and dissociated from Phytoalexin Deficient4 (PAD4) in Arabidopsis immunity. New Phytol. 2011, 191, 107–119. [Google Scholar] [CrossRef]
- He, X.; Huo, Y.; Liu, X.; Zhou, Q.; Feng, S.; Shen, X.; Li, B.; Wu, S.; Chen, X. Activation of disease resistance against Botryosphaeria dothidea by downregulating the expression of MdSYP121 in apple. Hortic. Res. 2018, 5, 24. [Google Scholar] [CrossRef] [Green Version]
- Schröpfer, S.; Böttcher, C.; Wöhner, T.; Richter, K.; Norelli, J.; Rikkerink, E.H.A.; Hanke, M.-V.; Flachowsky, H. A Single Effector Protein, AvrRpt2EA, from Erwinia amylovora Can Cause Fire Blight Disease Symptoms and Induces a Salicylic Acid–Dependent Defense Response. Mol. Plant-Microbe Interact. 2018, 31, 1179–1191. [Google Scholar] [CrossRef] [Green Version]
- Meyers, B.C.; Kozik, A.; Griego, A.; Kuang, H.; Michelmore, R.W. Genome-Wide Analysis of NBS-LRR–Encoding Genes in Arabidopsis. Plant Cell 2003, 15, 809–834. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, T.; Wang, Y.; Chen, J.-Q.; Araki, H.; Jing, Z.; Jiang, K.; Shen, J.; Tian, D. Genome-wide identification of NBS genes in rice reveals significant expansion of divergent non-TIR NBS genes. Mol. Genet. Genom. 2004, 271, 402–415. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Wang, B.; Yin, C.; Zhang, D.; Wang, D.; Song, J.; Zhou, L.; Kong, Z.; Klosterman, S.J.; Li, J.; et al. The Gossypium hirsutum TIR-NBS-LRR gene GhDSC1 mediates resistance against Verticillium wilt. Mol. Plant Pathol. 2019, 20, 857–876. [Google Scholar] [CrossRef] [Green Version]
- Lapin, D.; Kovacova, V.; Sun, X.; Dongus, J.A.; Bhandari, D.; von Born, P.; Bautor, J.; Guarneri, N.; Rzemieniewski, J.; Stuttmann, J.; et al. A Coevolved EDS1-SAG101-NRG1 Module Mediates Cell Death Signaling by TIR-Domain Immune Receptors. Plant Cell 2021, 31, 2430–2455. [Google Scholar] [CrossRef] [Green Version]
- Buschmann, H. Plant Cell Division Analyzed by Transient Agrobacterium-Mediated Transformation of Tobacco BY-2 Cells. Methods Mol. Biol. 2016, 1370, 17–25. [Google Scholar] [CrossRef]
- Zhou, H.; Bai, S.; Wang, N.; Sun, X.; Zhang, Y.; Zhu, J.; Dong, C. CRISPR/Cas9-Mediated Mutagenesis of MdCNGC2 in Apple Callus and VIGS-Mediated Silencing of MdCNGC2 in Fruits Improve Resistance to Botryosphaeria dothidea. Front. Plant Sci. 2020, 11, 575477. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Pagliarani, G.; Paris, R.; Arens, P.; Tartarini, S.; Ricci, G.; Smulders, M.M.; Van De Weg, W.E. A qRT-PCR assay for the expression of all Mal d 1 isoallergen genes. BMC Plant Biol. 2013, 13, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, O.X.; Tong, M.; Bonardi, V.; El Kasmi, F.; Woloshen, V.; Wünsch, L.K.; Dangl, J.L.; Li, X. TNL-mediated immunity in A rabidopsis requires complex regulation of the redundant ADR 1 gene family. New Phytol. 2016, 210, 960–973. [Google Scholar] [CrossRef]
- Ma, M.; Yuan, Y.; Cheng, C.; Zhang, Y.; Yang, S. The MdXTHB gene is involved in fruit softening in ‘Golden Del. Reinders’ (Malus pumila). J. Sci. Food Agric. 2021, 101, 564–572. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Yu, M.; Wang, Y.; Jia, Q.; Lin, L.; Dong, H. The antagonistic effect of hydroxyl radical on the development of a hypersensitive response in tobacco. FEBS J. 2010, 277, 5097–5111. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Ling, T.; Xue, Y.; Xu, C.; Zhou, W.; Hu, L.; Chen, J.; Shi, Z. Thymol Mitigates Cadmium Stress by Regulating Glutathione Levels and Reactive Oxygen Species Homeostasis in Tobacco Seedlings. Molecules 2016, 21, 1339. [Google Scholar] [CrossRef] [Green Version]
- Sheludko, Y.V.; Sindarovska, Y.R.; Gerasymenko, I.M.; Bannikova, M.; Kuchuk, M. Comparison of severalNicotiana species as hosts for high-scaleAgrobacterium-mediated transient expression. Biotechnol. Bioeng. 2007, 96, 608–614. [Google Scholar] [CrossRef]
- Zhang, W.; Yue, S.; Song, J.; Xun, M.; Han, M.; Yang, H. MhNRAMP1 from Malus hupehensis Exacerbates Cell Death by Accelerating Cd Uptake in Tobacco and Apple Calli. Front. Plant Sci. 2020, 11, 957. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lv, L.; Liu, Y.; Bai, S.; Turakulov, K.S.; Dong, C.; Zhang, Y. A TIR-NBS-LRR Gene MdTNL1 Regulates Resistance to Glomerella Leaf Spot in Apple. Int. J. Mol. Sci. 2022, 23, 6323. https://doi.org/10.3390/ijms23116323
Lv L, Liu Y, Bai S, Turakulov KS, Dong C, Zhang Y. A TIR-NBS-LRR Gene MdTNL1 Regulates Resistance to Glomerella Leaf Spot in Apple. International Journal of Molecular Sciences. 2022; 23(11):6323. https://doi.org/10.3390/ijms23116323
Chicago/Turabian StyleLv, Lingling, Yingshuang Liu, Suhua Bai, Khurshid Sadullaevich Turakulov, Chaohua Dong, and Yugang Zhang. 2022. "A TIR-NBS-LRR Gene MdTNL1 Regulates Resistance to Glomerella Leaf Spot in Apple" International Journal of Molecular Sciences 23, no. 11: 6323. https://doi.org/10.3390/ijms23116323





