Cytokines and Venous Leg Ulcer Healing—A Systematic Review
Abstract
:1. Introduction
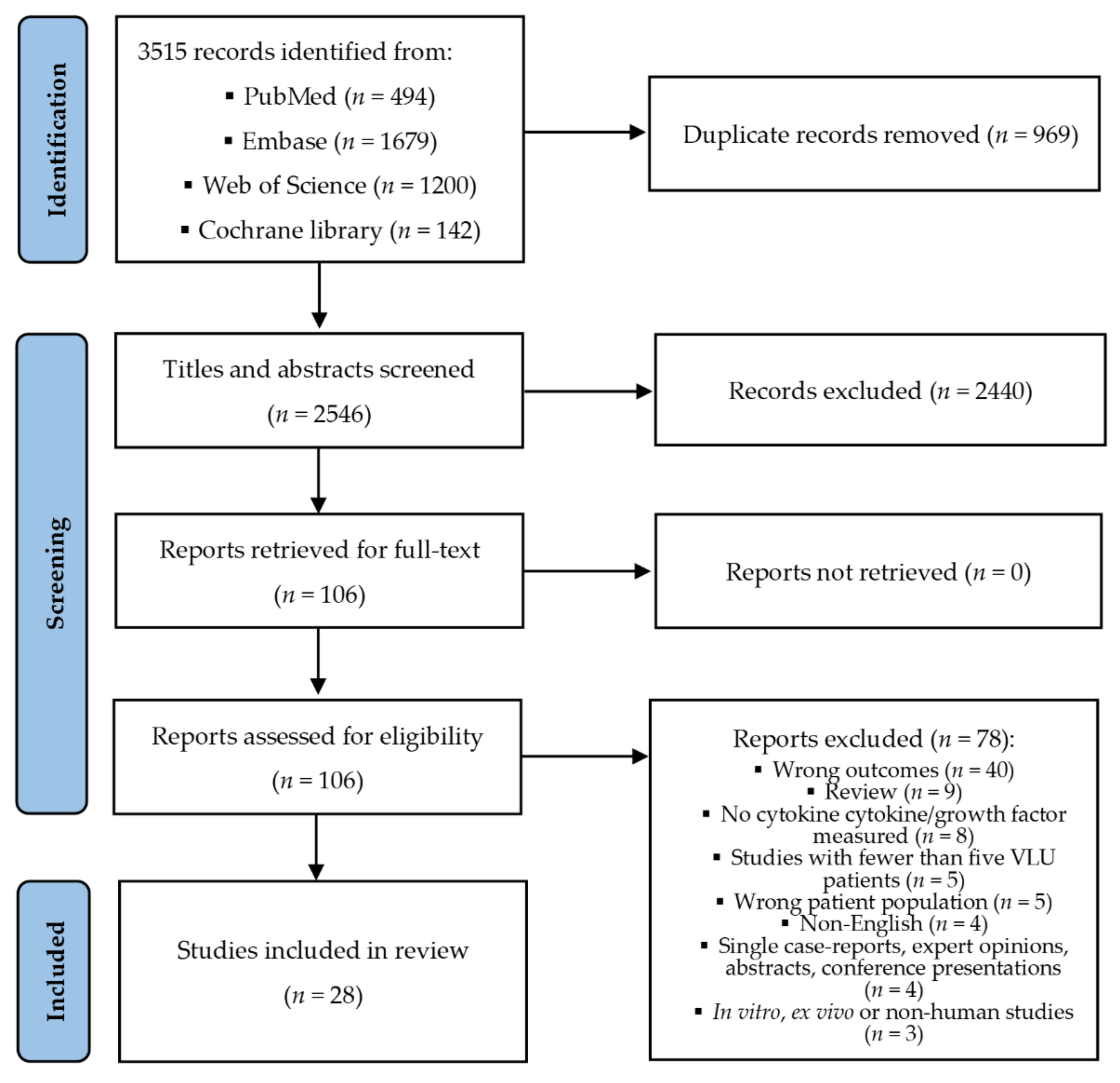
2. Materials and Methods
2.1. Study Types
2.2. Target Population
2.3. Cytokines Selected for the Search
2.4. Search Strategy
2.5. Data Collection and Extraction
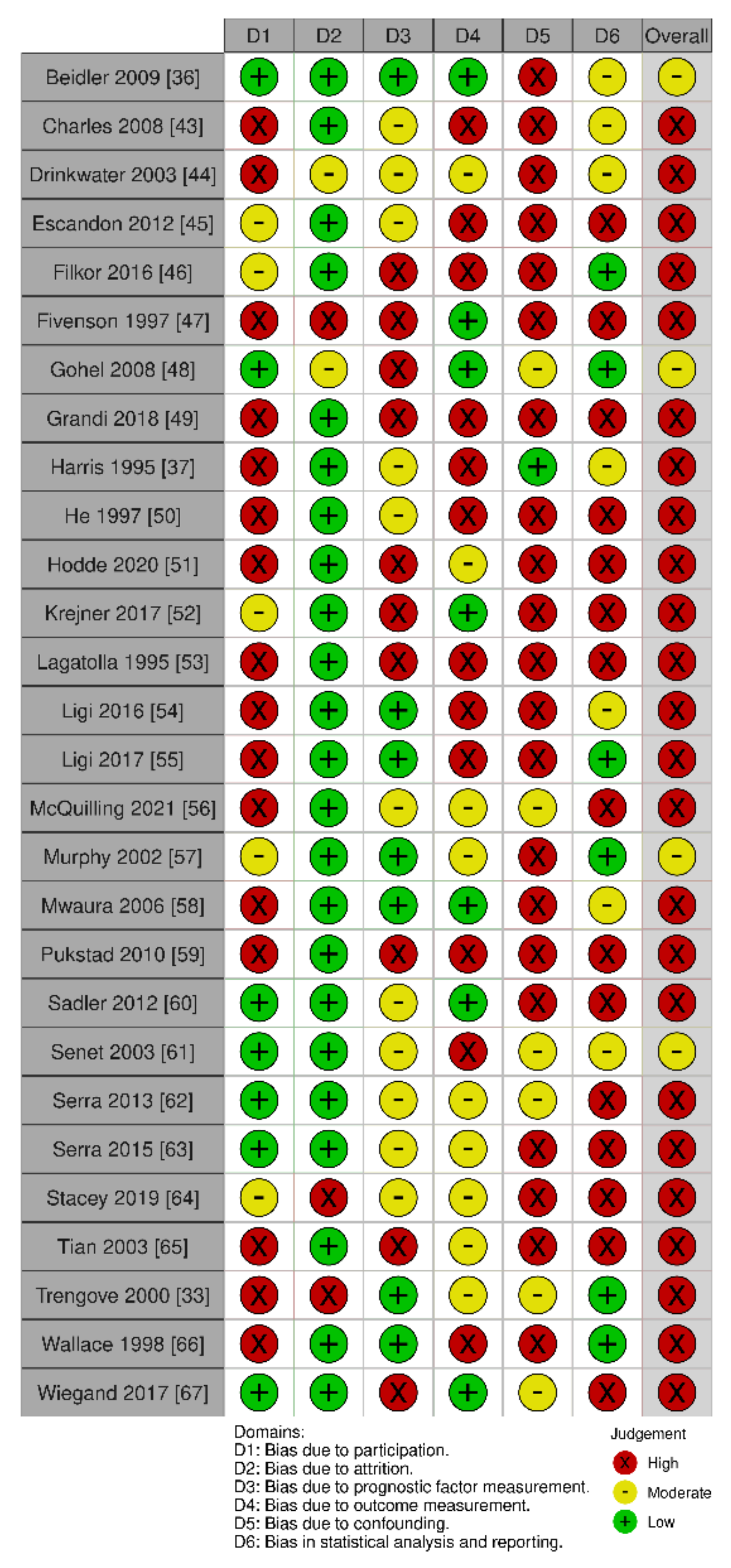
2.6. Risk of Bias (Quality) Assessment
2.7. Measures of Association, Data Synthesis and Pooling of Data
2.8. Missing Data
3. Results
3.1. Characteristics of Included Studies and Participants
3.2. Collection of Specimen, Analysis, and Processing
3.3. Definitions of Healing
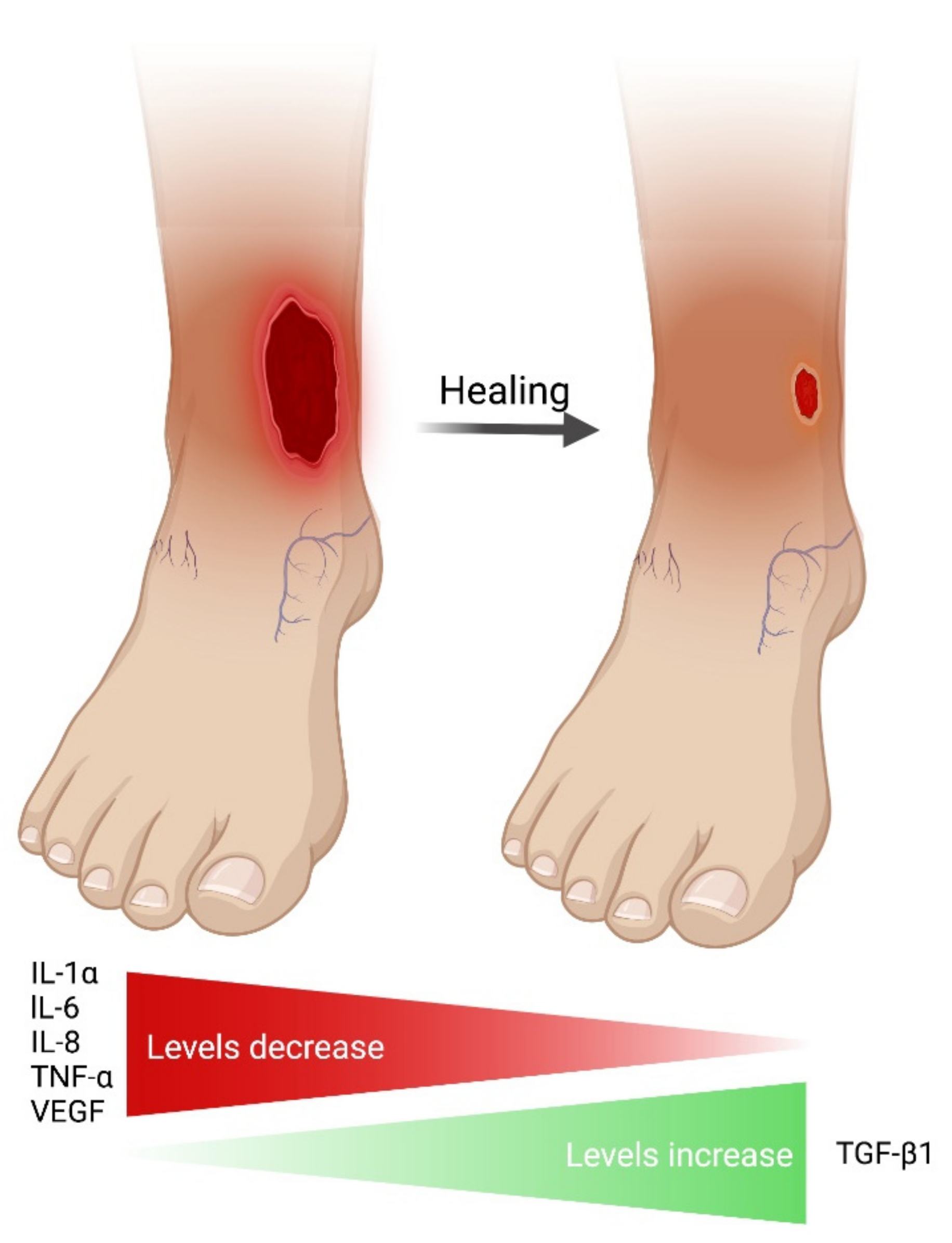
3.4. Cytokines in Non-Healing/Healing VLUs
3.5. Influence of Background Factors on Cytokine Levels
3.5.1. Compression Therapy
3.5.2. Other Factors
3.6. Bioactivity of Cytokines and Cytokine Receptors
3.7. Quality Assessment of Included Studies
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABPI | ankle-brachial pressure index |
| cDNA | complementary DNA |
| EGF | epidermal growth factor |
| ELISA | enzyme-linked immunosorbent assay |
| FGF | fibroblast growth factor |
| G-CSF | granulocyte colony-stimulating factor |
| GM-CSF | granulocyte-macrophage colony-stimulating factor |
| HB-EGF | heparin-binding epidermal growth factor |
| HGF | hepatocyte growth factor |
| IFN | interferon |
| IGF | insulin-like growth factor |
| IHC | immunohistochemistry |
| IL | interleukin |
| IL-1Ra | interleukin-1 receptor antagonist |
| KGF | keratinocyte growth factor |
| M-CSF | macrophage colony-stimulating factor |
| NA | not applicable |
| NGF | nerve growth factor |
| NR | not reported |
| PBMC | peripheral blood mononuclear cells |
| PDGF | platelet-derived growth factor |
| qRT-PCR | real-time quantitative reverse transcription polymerase chain reaction |
| RCT | randomized controlled trial |
| TGF | transforming growth factor |
| TNF | tumor necrosis factor |
| TNF-R | tumor necrosis factor receptor |
| VEGF | vascular endothelial growth factor |
| VEGF-R | vascular endothelial growth factor receptor |
| VLU | venous leg ulcer |
| WF | wound fluid |
References
- Nelzén, O.; Bergqvist, D.; Lindhagen, A. Venous and non-venous leg ulcers: Clinical history and appearance in a population study. Br. J. Surg. 1994, 81, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Margolis, D.J.; Bilker, W.; Santanna, J.; Baumgarten, M. Venous leg ulcer: Incidence and prevalence in the elderly. J. Am. Acad. Dermatol. 2002, 46, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Guest, J.F.; Fuller, G.W.; Vowden, P. Cohort study evaluating the burden of wounds to the UK’s National Health Service in 2017/2018: Update from 2012/2013. BMJ Open 2020, 10, e045253. [Google Scholar] [CrossRef] [PubMed]
- Van den Oever, R.; Hepp, B.; Debbaut, B.; Simon, I. Socio-economic impact of chronic venous insufficiency. An underestimated public health problem. Int. Angiol. 1998, 17, 161–167. [Google Scholar]
- Gottrup, F.; Henneberg, E.; Trangbaek, R.; Baekmark, N.; Zøllner, K.; Sørensen, J. Point prevalence of wounds and cost impact in the acute and community setting in Denmark. J. Wound Care 2013, 22, 413–422. [Google Scholar] [CrossRef]
- Olsson, M.; Järbrink, K.; Divakar, U.; Bajpai, R.; Upton, Z.; Schmidtchen, A.; Car, J. The humanistic and economic burden of chronic wounds: A systematic review. Wound Repair. Regen. 2019, 27, 114–125. [Google Scholar] [CrossRef]
- Menke, N.B.; Ward, K.R.; Witten, T.M.; Bonchev, D.G.; Diegelmann, R.F. Impaired wound healing. Clin. Dermatol. 2007, 25, 19–25. [Google Scholar] [CrossRef]
- Gottrup, F.; Apelqvist, J.; Price, P. Outcomes in controlled and comparative studies on non-healing wounds: Recommendations to improve the quality of evidence in wound management. J. Wound Care 2010, 19, 237–268. [Google Scholar] [CrossRef]
- Eming, S.A.; Martin, P.; Tomic-Canic, M. Wound repair and regeneration: Mechanisms, signaling, and translation. Sci. Transl. Med. 2014, 6, 265sr266. [Google Scholar] [CrossRef]
- Nelson, E.A.; Bell-Syer, S.E. Compression for preventing recurrence of venous ulcers. Cochrane Database Syst. Rev. 2014, 9, CD002303. [Google Scholar] [CrossRef]
- O’Meara, S.; Cullum, N.; Nelson, E.A.; Dumville, J.C. Compression for venous leg ulcers. Cochrane Database Syst. Rev. 2012, 11, CD000265. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Dumville, J.C.; Cullum, N.; Connaughton, E.; Norman, G. Compression bandages or stockings versus no compression for treating venous leg ulcers. Cochrane Database Syst. Rev. 2021, 7, CD013397. [Google Scholar] [CrossRef] [PubMed]
- Gohel, M.S.; Taylor, M.; Earnshaw, J.J.; Heather, B.P.; Poskitt, K.R.; Whyman, M.R. Risk factors for delayed healing and recurrence of chronic venous leg ulcers—An analysis of 1324 legs. Eur. J. Vasc. Endovasc. Surg. 2005, 29, 74–77. [Google Scholar] [CrossRef] [PubMed]
- Guest, M.; Smith, J.J.; Sira, M.S.; Madden, P.; Greenhalgh, R.M.; Davies, A.H. Venous ulcer healing by four-layer compression bandaging is not influenced by the pattern of venous incompetence. Br. J. Surg. 1999, 86, 1437–1440. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.; Weaver, C. Janeway’s Immunobiology, 9th ed.Garland Science: New York, NY, USA; Taylor & Francis Group: Oxfordshire, UK, 2017. [Google Scholar]
- Dinarello, C.A. Historical insights into cytokines. Eur. J. Immunol. 2007, 37 (Suppl. 1), S34–S45. [Google Scholar] [CrossRef] [PubMed]
- Miyajima, A.; Kitamura, T.; Harada, N.; Yokota, T.; Arai, K. Cytokine receptors and signal transduction. Annu. Rev. Immunol. 1992, 10, 295–331. [Google Scholar] [CrossRef]
- Zlotnik, A. Perspective: Insights on the nomenclature of cytokines and chemokines. Front. Immunol. 2020, 11, 908. [Google Scholar] [CrossRef]
- Stenken, J.A.; Poschenrieder, A.J. Bioanalytical chemistry of cytokines—A review. Anal. Chim. Acta 2015, 853, 95–115. [Google Scholar] [CrossRef]
- Liu, C.; Chu, D.; Kalantar-Zadeh, K.; George, J.; Young, H.A.; Liu, G. Cytokines: From clinical significance to quantification. Adv. Sci. 2021, 8, e2004433. [Google Scholar] [CrossRef]
- Commins, S.P.; Borish, L.; Steinke, J.W. Immunologic messenger molecules: Cytokines, interferons, and chemokines. J. Allergy Clin. Immunol. 2010, 125, S53–S72. [Google Scholar] [CrossRef]
- Landén, N.X.; Li, D.; Ståhle, M. Transition from inflammation to proliferation: A critical step during wound healing. Cell. Mol. Life Sci. 2016, 73, 3861–3885. [Google Scholar] [CrossRef] [PubMed]
- Nuutila, K.; Siltanen, A.; Peura, M.; Bizik, J.; Kaartinen, I.; Kuokkanen, H.; Nieminen, T.; Harjula, A.; Aarnio, P.; Vuola, J.; et al. Human skin transcriptome during superficial cutaneous wound healing. Wound Repair Regen. 2012, 20, 830–839. [Google Scholar] [CrossRef] [PubMed]
- Vogt, P.M.; Lehnhardt, M.; Wagner, D.; Jansen, V.; Krieg, M.; Steinau, H.U. Determination of endogenous growth factors in human wound fluid: Temporal presence and profiles of secretion. Plast. Reconstr. Surg. 1998, 102, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Rennekampff, H.O.; Hansbrough, J.F.; Kiessig, V.; Dore, C.; Sticherling, M.; Schröder, J.M. Bioactive interleukin-8 is expressed in wounds and enhances wound healing. J. Surg. Res. 2000, 93, 41–54. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, B.C.F.; Campos, A.K.; Alves, R.S.; Sarandy, M.M.; Novaes, R.D.; Esposito, D.; Gonçalves, R.V. What is the impact of depletion of immunoregulatory genes on wound healing? A systematic review of preclinical evidence. Oxid. Med. Cell. Longev. 2020, 2020, 8862953. [Google Scholar] [CrossRef]
- McFarland-Mancini, M.M.; Funk, H.M.; Paluch, A.M.; Zhou, M.; Giridhar, P.V.; Mercer, C.A.; Kozma, S.C.; Drew, A.F. Differences in wound healing in mice with deficiency of IL-6 versus IL-6 receptor. J. Immunol. 2010, 184, 7219–7228. [Google Scholar] [CrossRef]
- Gallucci, R.M.; Simeonova, P.P.; Matheson, J.M.; Kommineni, C.; Guriel, J.L.; Sugawara, T.; Luster, M.I. Impaired cutaneous wound healing in interleukin-6-deficient and immunosuppressed mice. FASEB J. 2000, 14, 2525–2531. [Google Scholar] [CrossRef]
- Fang, Y.; Gong, S.J.; Xu, Y.H.; Hambly, B.D.; Bao, S. Impaired cutaneous wound healing in granulocyte/macrophage colony-stimulating factor knockout mice. Br. J. Dermatol. 2007, 157, 458–465. [Google Scholar] [CrossRef]
- Ågren, M.S.; Gottrup, F. Causation of venous leg ulcers. In Leg Ulcers: A Problem-Based Learning Approach; Morison, M.J., Moffatt, C.J., Franks, P.J., Eds.; Mosby: Edinburgh, UK, 2007; pp. 141–154. [Google Scholar]
- Mansilha, A.; Sousa, J. Pathophysiological mechanisms of chronic venous disease and implications for venoactive drug therapy. Int. J. Mol. Sci. 2018, 19, 1669. [Google Scholar] [CrossRef]
- Liu, Y.C.; Margolis, D.J.; Isseroff, R.R. Does inflammation have a role in the pathogenesis of venous ulcers? A critical review of the evidence. J. Invest. Dermatol. 2011, 131, 818–827. [Google Scholar] [CrossRef]
- Trengove, N.J.; Bielefeldt-Ohmann, H.; Stacey, M.C. Mitogenic activity and cytokine levels in non-healing and healing chronic leg ulcers. Wound Repair Regen. 2000, 8, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Loots, M.A.; Lamme, E.N.; Zeegelaar, J.; Mekkes, J.R.; Bos, J.D.; Middelkoop, E. Differences in cellular infiltrate and extracellular matrix of chronic diabetic and venous ulcers versus acute wounds. J. Invest. Dermatol. 1998, 111, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Trøstrup, H.; Lundquist, R.; Christensen, L.H.; Jorgensen, L.N.; Karlsmark, T.; Haab, B.B.; Ågren, M.S. S100A8/A9 deficiency in nonhealing venous leg ulcers uncovered by multiplexed antibody microarray profiling. Br. J. Dermatol. 2011, 165, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Beidler, S.K.; Douillet, C.D.; Berndt, D.F.; Keagy, B.A.; Rich, P.B.; Marston, W.A. Inflammatory cytokine levels in chronic venous insufficiency ulcer tissue before and after compression therapy. J. Vasc. Surg. 2009, 49, 1013–1020. [Google Scholar] [CrossRef] [PubMed]
- Harris, I.R.; Yee, K.C.; Walters, C.E.; Cunliffe, W.J.; Kearney, J.N.; Wood, E.J.; Ingham, E. Cytokine and protease levels in healing and non-healing chronic venous leg ulcers. Exp. Dermatol. 1995, 4, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Burian, E.A.; Franks, P.J.; Moffatt, C.J.; Kirketerp-Møller, K.; Karlsmark, T.; Thyssen, J.P.; Ågren, M.S. The Role of Cytokines and Growth Factors as Biomarkers for Wound Healing in Venous Leg Ulcers—A Systematic Review. Available online: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42021262687 (accessed on 1 April 2022).
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Hayden, J.A.; van der Windt, D.A.; Cartwright, J.L.; Côté, P.; Bombardier, C. Assessing bias in studies of prognostic factors. Ann. Intern. Med. 2013, 158, 280–286. [Google Scholar] [CrossRef]
- Hayden, J.A.; Wilson, M.N.; Riley, R.D.; Iles, R.; Pincus, T.; Ogilvie, R. Individual recovery expectations and prognosis of outcomes in non-specific low back pain: Prognostic factor review. Cochrane Database Syst. Rev. 2019, 9, CD011284. [Google Scholar] [CrossRef]
- Westby, M.J.; Dumville, J.C.; Stubbs, N.; Norman, G.; Wong, J.K.; Cullum, N.; Riley, R.D. Protease activity as a prognostic factor for wound healing in venous leg ulcers. Cochrane Database Sys. Rev. 2018, 9, CD012841. [Google Scholar] [CrossRef]
- Charles, C.A.; Tomic-Canic, M.; Vincek, V.; Nassiri, M.; Stojadinovic, O.; Eaglstein, W.H.; Kirsner, R.S. A gene signature of nonhealing venous ulcers: Potential diagnostic markers. J. Am. Acad. Dermatol. 2008, 59, 758–771. [Google Scholar] [CrossRef]
- Drinkwater, S.L.; Burnand, K.G.; Ding, R.; Smith, A. Increased but ineffectual angiogenic drive in nonhealing venous leg ulcers. J. Vasc. Surg. 2003, 38, 1106–1112. [Google Scholar] [CrossRef]
- Escandon, J.; Vivas, A.C.; Perez, R.; Kirsner, R.; Davis, S. A prospective pilot study of ultrasound therapy effectiveness in refractory venous leg ulcers. Int. Wound J. 2012, 9, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Filkor, K.; Németh, T.; Nagy, I.; Kondorosi, É.; Urbán, E.; Kemény, L.; Szolnoky, G. The expression of inflammatory cytokines, TAM tyrosine kinase receptors and their ligands is upregulated in venous leg ulcer patients: A novel insight into chronic wound immunity. Int. Wound J. 2016, 13, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Fivenson, D.P.; Faria, D.T.; Nickoloff, B.J.; Poverini, P.J.; Kunkel, S.; Burdick, M.; Strieter, R.M. Chemokine and inflammatory cytokine changes during chronic wound healing. Wound Repair Regen. 1997, 5, 310–322. [Google Scholar] [CrossRef]
- Gohel, M.S.; Windhaber, R.A.; Tarlton, J.F.; Whyman, M.R.; Poskitt, K.R. The relationship between cytokine concentrations and wound healing in chronic venous ulceration. J. Vasc. Surg. 2008, 48, 1272–1277. [Google Scholar] [CrossRef]
- Grandi, V.; Bacci, S.; Corsi, A.; Sessa, M.; Puliti, E.; Murciano, N.; Scavone, F.; Cappugi, P.; Pimpinelli, N. ALA-PDT exerts beneficial effects on chronic venous ulcers by inducing changes in inflammatory microenvironment, especially through increased TGF-beta release: A pilot clinical and translational study. Photodiagn. Photodyn. Ther. 2018, 21, 252–256. [Google Scholar] [CrossRef]
- He, C.F.; Cherry, G.W.; Arnold, F. Postural vasoregulation and mediators of reperfusion injury in venous ulceration. J. Vasc. Surg. 1997, 25, 647–653. [Google Scholar] [CrossRef]
- Hodde, J.P.; Hiles, M.C.; Metzger, D.W. Characterization of the local wound environment following treatment of chronic leg ulcers with SIS wound matrix. J. Tissue Viability 2020, 29, 42–47. [Google Scholar] [CrossRef]
- Krejner, A.; Litwiniuk, M.; Grzela, T. LL-37 but not 25-hydroxy-vitamin D serum level correlates with healing of venous leg ulcers. Arch. Immunol. Ther. Exp. 2017, 65, 455–461. [Google Scholar] [CrossRef]
- Lagattolla, N.R.; Stacey, M.C.; Burnand, K.G.; Gaffney, P.G. Growth factors, tissue and urokinase-type plasminogen activators in venous ulcers. Ann. Cardiol. Angeiol. 1995, 44, 299–303. [Google Scholar]
- Ligi, D.; Mosti, G.; Croce, L.; Raffetto, J.D.; Mannello, F. Chronic venous disease—Part I: Inflammatory biomarkers in wound healing. Biochim. Biophys. Acta 2016, 1862, 1964–1974. [Google Scholar] [CrossRef] [PubMed]
- Ligi, D.; Croce, L.; Mosti, G.; Raffetto, J.D.; Mannello, F. Chronic venous insufficiency: Transforming growth factor-beta isoforms and soluble endoglin concentration in different states of wound healing. Int. J. Mol. Sci. 2017, 18, 2206. [Google Scholar] [CrossRef] [PubMed]
- McQuilling, J.P.; Carter, M.J.; Fulton, J.A.; Patel, K.; Doner, B.; Serena, T.E.; Mowry, K.C. A prospective clinical trial evaluating changes in the wound microenvironment in patients with chronic venous leg ulcers treated with a hypothermically stored amniotic membrane. Int. Wound J. 2021. [Google Scholar] [CrossRef]
- Murphy, M.A.; Joyce, W.P.; Condron, C.; Bouchier-Hayes, D. A reduction in serum cytokine levels parallels healing of venous ulcers in patients undergoing compression therapy. Eur. J. Vasc. Endovasc. Surg. 2002, 23, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Mwaura, B.; Mahendran, B.; Hynes, N.; Defreitas, D.; Avalos, G.; Adegbola, T.; Adham, M.; Connolly, C.E.; Sultan, S. The impact of differential expression of extracellular matrix metalloproteinase inducer, matrix metalloproteinase-2, tissue inhibitor of matrix metalloproteinase-2 and PDGF-AA on the chronicity of venous leg ulcers. Eur. J. Vasc. Endovasc. Surg. 2006, 31, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Pukstad, B.S.; Ryan, L.; Flo, T.H.; Stenvik, J.; Moseley, R.; Harding, K.; Thomas, D.W.; Espevik, T. Non-healing is associated with persistent stimulation of the innate immune response in chronic venous leg ulcers. J. Dermatol. Sci. 2010, 59, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Sadler, G.M.; Wallace, H.J.; Stacey, M.C. Oral doxycycline for the treatment of chronic leg ulceration. Arch. Dermatol. Res. 2012, 304, 487–493. [Google Scholar] [CrossRef]
- Senet, P.; Bon, F.X.; Benbunan, M.; Bussel, A.; Traineau, R.; Calvo, F.; Dubertret, L.; Dosquet, C. Randomized trial and local biological effect of autologous platelets used as adjuvant therapy for chronic venous leg ulcers. J. Vasc. Surg. 2003, 38, 1342–1348. [Google Scholar] [CrossRef]
- Serra, R.; Grande, R.; Buffone, G.; Gallelli, L.; De Franciscis, S. The effects of minocycline on extracellular matrix in patients with chronic venous leg ulcers. Acta Phlebol. 2013, 14, 99–107. [Google Scholar]
- Serra, R.; Gallelli, L.; Buffone, G.; Molinari, V.; Stillitano, D.M.; Palmieri, C.; de Franciscis, S. Doxycycline speeds up healing of chronic venous ulcers. Int. Wound J. 2015, 12, 179–184. [Google Scholar] [CrossRef]
- Stacey, M.C.; Phillips, S.A.; Farrokhyar, F.; Swaine, J.M. Evaluation of wound fluid biomarkers to determine healing in adults with venous leg ulcers: A prospective study. Wound Repair Regen. 2019, 27, 509–518. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.W.; Stacey, M.C. Cytokines and growth factors in keratinocytes and sweat glands in chronic venous leg ulcers. An immunohistochemical study. Wound Repair Regen. 2003, 11, 316–325. [Google Scholar] [CrossRef]
- Wallace, H.J.; Stacey, M.C. Levels of tumor necrosis factor-alpha (TNF-alpha) and soluble TNF receptors in chronic venous leg ulcers--correlations to healing status. J. Invest. Dermatol. 1998, 110, 292–296. [Google Scholar] [CrossRef] [PubMed]
- Wiegand, C.; Bittenger, K.; Galiano, R.D.; Driver, V.R.; Gibbons, G.W. Does noncontact low-frequency ultrasound therapy contribute to wound healing at the molecular level? Wound Repair Regen. 2017, 25, 871–882. [Google Scholar] [CrossRef] [PubMed]
- Serra, R.; Grande, R.; Buffone, G.; Molinari, V.; Perri, P.; Perri, A.; Amato, B.; Colosimo, M.; de Franciscis, S. Extracellular matrix assessment of infected chronic venous leg ulcers: Role of metalloproteinases and inflammatory cytokines. Int. Wound J. 2016, 13, 53–58. [Google Scholar] [CrossRef]
- McInnes, R.L.; Cullen, B.M.; Hill, K.E.; Price, P.E.; Harding, K.G.; Thomas, D.W.; Stephens, P.; Moseley, R. Contrasting host immuno-inflammatory responses to bacterial challenge within venous and diabetic ulcers. Wound Repair Regen. 2014, 22, 58–69. [Google Scholar] [CrossRef]
- McGuinness, L.A.; Higgins, J.P.T. Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res. Synth. Methods 2020, 12, 55–61. [Google Scholar] [CrossRef]
- Carvalho, M.R.; Silveira, I.A.; Oliveira, B. Treatment of venous ulcers with growth factors: Systematic review and meta-analysis. Rev. Bras. Enferm. 2019, 72, 200–210. [Google Scholar] [CrossRef]
- Lee, Y.; Lee, M.H.; Phillips, S.A.; Stacey, M.C. Growth factors for treating chronic venous leg ulcers: A systematic review and meta-analysis. Wound Repair Regen. 2022, 30, 117–125. [Google Scholar] [CrossRef]
- Kantor, J.; Margolis, D.J. A multicentre study of percentage change in venous leg ulcer area as a prognostic index of healing at 24 weeks. Br. J. Dermatol. 2000, 142, 960–964. [Google Scholar] [CrossRef]
- Cardinal, M.; Eisenbud, D.E.; Phillips, T.; Harding, K. Early healing rates and wound area measurements are reliable predictors of later complete wound closure. Wound Repair Regen. 2008, 16, 19–22. [Google Scholar] [CrossRef] [PubMed]
- Marston, W.A.; Ennis, W.J.; Lantis, J.C., 2nd; Kirsner, R.S.; Galiano, R.D.; Vanscheidt, W.; Eming, S.A.; Malka, M.; Cargill, D.I.; Dickerson, J.E., Jr.; et al. Baseline factors affecting closure of venous leg ulcers. J. Vasc. Surg. Venous Lymphat. Disord. 2017, 5, 829–835.e821. [Google Scholar] [CrossRef] [PubMed]
- Grimstad, Ø.; Sandanger, Ø.; Ryan, L.; Otterdal, K.; Damaas, J.K.; Pukstad, B.; Espevik, T. Cellular sources and inducers of cytokines present in acute wound fluid. Wound Repair Regen. 2011, 19, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Bao, P.; Kodra, A.; Tomic-Canic, M.; Golinko, M.S.; Ehrlich, H.P.; Brem, H. The role of vascular endothelial growth factor in wound healing. J. Surg. Res. 2009, 153, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Ambrosch, A.; Lobmann, R.; Pott, A.; Preissler, J. Interleukin-6 concentrations in wound fluids rather than serological markers are useful in assessing bacterial triggers of ulcer inflammation. Int. Wound J. 2008, 5, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Zillmer, R.; Trøstrup, H.; Karlsmark, T.; Ifversen, P.; Ågren, M.S. Duration of wound fluid secretion from chronic venous leg ulcers is critical for interleukin-1α, interleukin-1β, interleukin-8 levels and fibroblast activation. Arch. Dermatol. Res. 2011, 303, 601–606. [Google Scholar] [CrossRef] [PubMed]
- Nylander-Lundqvist, E.; Egelrud, T. Formation of active IL-1 beta from pro-IL-1 beta catalyzed by stratum corneum chymotryptic enzyme in vitro. Acta Derm. Venereol. 1997, 77, 203–206. [Google Scholar]
- Baggiolini, M.; Dewald, B.; Moser, B. Interleukin-8 and related chemotactic cytokines–CXC and CC chemokines. Adv. Immunol. 1994, 55, 97–179. [Google Scholar]
- Krzyszczyk, P.; Schloss, R.; Palmer, A.; Berthiaume, F. The role of macrophages in acute and chronic wound healing and interventions to promote pro-wound healing phenotypes. Front. Physiol. 2018, 9, 419. [Google Scholar] [CrossRef]
- Shapouri-Moghaddam, A.; Mohammadian, S.; Vazini, H.; Taghadosi, M.; Esmaeili, S.A.; Mardani, F.; Seifi, B.; Mohammadi, A.; Afshari, J.T.; Sahebkar, A. Macrophage plasticity, polarization, and function in health and disease. J. Cell. Physiol. 2018, 233, 6425–6440. [Google Scholar] [CrossRef]
- Tecchio, C.; Micheletti, A.; Cassatella, M.A. Neutrophil-derived cytokines: Facts beyond expression. Front. Immunol. 2014, 5, 508. [Google Scholar] [CrossRef] [PubMed]
- Wilgus, T.A.; Roy, S.; McDaniel, J.C. Neutrophils and wound repair: Positive actions and negative reactions. Adv. Wound Care 2013, 2, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Cañedo-Dorantes, L.; Cañedo-Ayala, M. Skin acute wound healing: A comprehensive review. Int. J. Inflam. 2019, 2019, 3706315. [Google Scholar] [CrossRef] [PubMed]
- Piipponen, M.; Li, D.; Landén, N.X. The immune functions of keratinocytes in skin wound healing. Int. J. Mol. Sci. 2020, 21, 8790. [Google Scholar] [CrossRef]
- Kondo, T.; Ohshima, T.; Eisenmenger, W. Immunohistochemical and morphometrical study on the temporal expression of interleukin-1alpha (IL-1alpha) in human skin wounds for forensic wound age determination. Int. J. Legal Med. 1999, 112, 249–252. [Google Scholar] [CrossRef]
- Pulido, T.; Velarde, M.C.; Alimirah, F. The senescence-associated secretory phenotype: Fueling a wound that never heals. Mech. Ageing Dev. 2021, 199, 111561. [Google Scholar] [CrossRef]
- Short, W.D.; Wang, X.; Keswani, S.G. The role of T lymphocytes in cutaneous scarring. Adv. Wound Care 2022, 11, 121–131. [Google Scholar] [CrossRef]
- Sachdev, U.; Vodovotz, L.; Bitner, J.; Barclay, D.; Zamora, R.; Yin, J.; Simmons, R.L.; Vodovotz, Y. Suppressed networks of inflammatory mediators characterize chronic venous insufficiency. J. Vasc. Surg. Venous Lymphat. Disord. 2018, 6, 358–366. [Google Scholar] [CrossRef]
- Herrick, S.E.; Sloan, P.; McGurk, M.; Freak, L.; McCollum, C.N.; Ferguson, M.W. Sequential changes in histologic pattern and extracellular matrix deposition during the healing of chronic venous ulcers. Am. J. Pathol. 1992, 141, 1085–1095. [Google Scholar]
- Bosanquet, D.C.; Rangaraj, A.; Richards, A.J.; Riddell, A.; Saravolac, V.M.; Harding, K.G. Topical steroids for chronic wounds displaying abnormal inflammation. Ann. R. Coll. Surg. Engl. 2013, 95, 291–296. [Google Scholar] [CrossRef]
- Hofman, D.; Moore, K.; Cooper, R.; Eagle, M.; Cooper, S. Use of topical corticosteroids on chronic leg ulcers. J. Wound Care 2007, 16, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.D.; Baquerizo-Nole, K.L.; Keegan, B.R.; Macquhae, F.; Escandon, J.; Espinosa, A.; Perez, C.; Romanelli, P.; Kirsner, R.S. Adalimumab treatment leads to reduction of tissue tumor necrosis factor-alpha correlated with venous leg ulcer improvement: A pilot study. Int. Wound J. 2016, 13, 963–966. [Google Scholar] [CrossRef] [PubMed]
- Streit, M.; Beleznay, Z.; Braathen, L.R. Topical application of the tumour necrosis factor-alpha antibody infliximab improves healing of chronic wounds. Int. Wound J. 2006, 3, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Bootun, R. Effects of immunosuppressive therapy on wound healing. Int. Wound J. 2013, 10, 98–104. [Google Scholar] [CrossRef]
- Tekin, H.G.; Eskandarani, H.A.; Iversen, L.; Juel, J. Immunosuppressive therapy and post-operative wound healing. Ugeskr. Laeger. 2021, 183, V07200544. [Google Scholar]
- Yang, Z.P.; Hong, L.; Wu, Q.; Wu, K.C.; Fan, D.M. Preoperative infliximab use and postoperative complications in Crohn’s disease: A systematic review and meta-analysis. Int. J. Surg. 2014, 12, 224–230. [Google Scholar] [CrossRef]
- Parameswaran, N.; Patial, S. Tumor necrosis factor-α signaling in macrophages. Crit. Rev. Eukaryot. Gene Expr. 2010, 20, 87–103. [Google Scholar] [CrossRef]
- Brooklyn, T.N.; Dunnill, M.G.; Shetty, A.; Bowden, J.J.; Williams, J.D.; Griffiths, C.E.; Forbes, A.; Greenwood, R.; Probert, C.S. Infliximab for the treatment of pyoderma gangrenosum: A randomised, double blind, placebo controlled trial. Gut 2006, 55, 505–509. [Google Scholar] [CrossRef]
- Ashcroft, G.S.; Jeong, M.J.; Ashworth, J.J.; Hardman, M.; Jin, W.; Moutsopoulos, N.; Wild, T.; McCartney-Francis, N.; Sim, D.; McGrady, G.; et al. Tumor necrosis factor-alpha (TNF-α) is a therapeutic target for impaired cutaneous wound healing. Wound Repair Regen. 2012, 20, 38–49. [Google Scholar] [CrossRef]
| Author | N 1 | Study Type | Follow-Up Time | Specimen | Analysis | Cytokine 2 |
|---|---|---|---|---|---|---|
| Beidler 2009 [36] | 29 | Cohort | 4 weeks | Biopsy | Luminex, ELISA | G-CSF, GM-CSF, IFN-γ, IL-1α, IL-1β, IL-1Ra, IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL12p40, IL-12p70, IL-13, IL-15, IL-17, TGF-β1, TNF-α |
| Charles 2008 [43] | 10 | Cross-sectional | 0 | Biopsy | cDNA microarray | HB-EGF, PDGFRα, S100A7 |
| Drinkwater 2003 [44] | 35 | Cohort | ~1 year | WF, biopsy | ELISA, RT-PCR | VEGF-121, VEGF-165, VEGF-189, VEGF-RI, VEGF-RII |
| Escandon 2012 [45] | 10 | Clinical trial 3 | 4 weeks | Biopsy | RT-PCR | IL-1α, IL-6, IL-8, IL-10, IL-11, TNF-α, VEGF |
| Filkor 2016 [46] | 69 | Cohort | 4 weeks | PBMC | qRT-PCR | IL-1α, IL-8, IL-10, TNF-α |
| Fivenson 1997 [47] | 14 | Cohort | 8 weeks | WF | ELISA | IL-8, IL-10 |
| Gohel 2008 [48] | 80 | Cohort | 5 weeks | WF, serum | ELISA | FGF-2, IL-1β, TGF-β1, TNF-α, VEGF |
| Grandi 2018 [49] | 19 | Clinical trial 4 | ~3 weeks | Biopsy | IHC | TGF-β, TNF-α |
| Harris 1995 [37] | 18 | Cross-sectional | 0 | WF | ELISA, bioassay | FGF-2, GM-CSF, IL-1α, IL-1α bio, IL-1β bio, IL-6 bio, PDGF-AB |
| He 1997 [50] | 10 | Experimental 5 | 200 min | Serum | ELISA | IL-1β, IL-1Ra, IL-6, TNF-α |
| Hodde 2020 [51] | 12 | Clinical trial 6 | 12 weeks | WF | Luminex, ELISA | GM-CSF, IFN-γ, IL-1β, IL-2, IL-4, IL-5, IL-8, IL-10, IL-12(p70), IL-13, TGF-β1, TNF-α |
| Krejner 2017 [52] | 19 | Cohort, retrospective | -4 weeks | Serum | ELISA | IL-6, IL-8, TNF |
| Lagattolla 1995 [53] | 19 | Cohort 7 | 6 months | Biopsy | ELISA | FGF-2, PDGF-AB, TGF-β1, |
| Ligi 2016 [54] | 34 | Cross-sectional, cohort | During admission | WF | Luminex | FGF-2, G-CSF, GM-CSF, IFN-γ, IL-1β, IL-1Ra, IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, IL-9, IL-10, IL-12(p70), IL-13, IL-15, IL-17, PDGF-BB, TNF-α, VEGF |
| Ligi 2017 [55] | 30 | Cross-sectional, cohort | 0 | WF | Luminex | TGF-β1, TGF-β2, TGF-β3 |
| McQuilling 2021 [56] | 15 | Clinical trial 8 | 12 weeks | WF | Multiplex MAP arrays | EGF, FGF-2, G-CSF, GM-CSF, IFN-γ, IL-1α, IL-1β, IL-1Ra, IL-2, IL-3, IL-4, IL-5, IL-6, IL-7, IL-8, IL-9, IL-10, IL-12p40, IL-12p70, IL-13, IL-15, IL-17, PDGF-AA, PDGF-BB, TGF-α, TGF-β1, TGF-β2, TGF-β3, TNF-α, TNF-β, VEGF |
| Murphy 2002 [57] | 8 | Cohort | 12 weeks | Serum | ELISA | TNF-α, VEGF |
| Mwaura 2006 [58] | 40 | Cohort | 8 weeks | WF, biopsy | ELISA, IHC | PDGF-AA |
| Pukstad 2010 [59] | 8 | Cohort | 8 weeks | WF | Antibody array, bioassay | IL-1α, IL-1β, IL-6R, IL-8, TNF-α, sTNF-RI, sTNF-RII |
| Sadler 2012 [60] | 20 | Clinical trial 9 | 4 weeks | WF | ELISA | TNF-α |
| Senet 2003 [61] | 15 | RCT 10 | 16 weeks | WF | ELISA | IL-8, KGF, VEGF |
| Serra 2013 [62] | 60 | RCT 11 | ~13 months | WF, plasma | ELISA | VEGF |
| Serra 2015 [63] | 64 | RCT 9 | ~13 months | WF, plasma | ELISA | VEGF |
| Stacey 2019 [64] | 42 | Cohort | 13 weeks | WF | Multiplex ELISA | G-CSF, GM-CSF, IFN-γ, IL-1α, IL-1β, IL-1Ra, IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-11, IL-12p40, IL-12p70, IL-13, IL-15, IL-16, IL-17, M-CSF, PDGF-BB, TNF-α, TNF-β, TNF-RI, TNF-RII |
| Tian 2003 [65] | 21 | Cohort | 2 weeks | Biopsy | IHC | EGF, FGF-2, IL-1α, IL-6, PDGF-A, TGF-β1, TNF-α, TNF-RI, VEGF |
| Trengove 2000 [33] | 26 | Cohort | 2 weeks | WF | ELISA, bioassay | EGF, FGF-2, IL-1α, IL-1β, IL-1 bio, IL-6, IL-6 bio, TGF-β1, TNF-α, PDGF |
| Wallace 1998 [66] | 21 | Cohort | 2 weeks | WF | ELISA, bioassay | TNF-α, TNF-α bio, sTNF-RI, sTNF-RII |
| Wiegand 2017 [67] | 42 | RCT 12 | 4 weeks | WF, biopsy | Luminex, IHC | IL-1β, IL-6, IL-8, IL-10, TGF-β, TNF-α |
| Cytokine | Non-Healing VLUs | vs. | Healing VLUs |
|---|---|---|---|
| EGF | ↑ Biopsy [65] | ↑ WF [56] | |
| FGF-2 | ↑ Biopsy [65] | ↑ Biopsy [53] | |
| GM-CSF | ↑ WF [54,64] | ↑ Biopsy [36] 1 | |
| HB-EGF | ↑ Biopsy [43] | ||
| IFN-γ | ↑ Biopsy [36] 1 | ||
| IL-1α | ↑ WF [33,56], ↑ Biopsy [45,65], ↑ Blood [46] | ↑ Biopsy [36] 1 | |
| IL-1β | ↑ WF [33,54] | ↑ Biopsy [36] 1 | |
| IL-1 bio 3 | ↑ WF [33] | ||
| IL-1Ra | ↑ WF [56] | ↑ Biopsy [36] 2 | |
| IL-2 | ↑ WF [56] | ||
| IL-3 | ↑ WF [56] | ||
| IL-6 | ↑ WF [67], ↑ Biopsy [45,65] | ↑ WF [64] 4 | |
| IL-6 bio 3 | ↑ WF [33] | ||
| IL-7 | ↑ WF [56] | ||
| IL-8 | ↑ WF [51,54,56,61], ↑ Biopsy [45], ↑ Blood [46] | ↑ WF [47] | |
| IL-9 | ↑ WF [56] | ||
| IL-10 | ↑ WF [54] | ||
| IL-11 | ↑ Biopsy [45] | ||
| IL-12p40 | ↑ Biopsy [36] 1 | ||
| IL-12p70 | ↑ WF [54] | ||
| IL-16 | ↑ WF [64] 4 | ||
| PDGF-AA | ↑ Biopsy [65] | ↑ WF [58], ↑ Biopsy [58] | |
| PDGF-BB | ↑ WF [54,64] 4 | ||
| PDGFRα | ↑ Biopsy [43] | ||
| S100A7 | ↑ Biopsy [43] | ||
| TGF-β1 | ↑ Biopsy [65] | ↑ WF [48,51], ↑ Biopsy [49,53] | |
| TGF-β3 | ↑ WF [55] | ||
| TNF-α | ↑ WF [33,51,66], ↑ Biopsy [45,65], ↑ Blood [57] 2 | ||
| TNF-β | ↑ WF [56] | ||
| TNF-RI | ↑ Biopsy [65] | ||
| TNF-RII | ↑ WF [66] | ||
| VEGF | ↑ WF [44,54,62,63], ↑ Biopsy [45,65], ↑ Blood [57,62,63] 2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burian, E.A.; Sabah, L.; Karlsmark, T.; Kirketerp-Møller, K.; Moffatt, C.J.; Thyssen, J.P.; Ågren, M.S. Cytokines and Venous Leg Ulcer Healing—A Systematic Review. Int. J. Mol. Sci. 2022, 23, 6526. https://doi.org/10.3390/ijms23126526
Burian EA, Sabah L, Karlsmark T, Kirketerp-Møller K, Moffatt CJ, Thyssen JP, Ågren MS. Cytokines and Venous Leg Ulcer Healing—A Systematic Review. International Journal of Molecular Sciences. 2022; 23(12):6526. https://doi.org/10.3390/ijms23126526
Chicago/Turabian StyleBurian, Ewa A., Lubna Sabah, Tonny Karlsmark, Klaus Kirketerp-Møller, Christine J. Moffatt, Jacob P. Thyssen, and Magnus S. Ågren. 2022. "Cytokines and Venous Leg Ulcer Healing—A Systematic Review" International Journal of Molecular Sciences 23, no. 12: 6526. https://doi.org/10.3390/ijms23126526
APA StyleBurian, E. A., Sabah, L., Karlsmark, T., Kirketerp-Møller, K., Moffatt, C. J., Thyssen, J. P., & Ågren, M. S. (2022). Cytokines and Venous Leg Ulcer Healing—A Systematic Review. International Journal of Molecular Sciences, 23(12), 6526. https://doi.org/10.3390/ijms23126526







