Allicin as a Volatile or Nebulisable Antimycotic for the Treatment of Pulmonary Mycoses: In Vitro Studies Using a Lung Flow Test Rig
Abstract
:1. Introduction
2. Results
2.1. Identification and Molecular Characterisation of a Suitable Thermophilic Fungus
2.2. Allicin Inhibits Spore Germination upon Direct Contact and via the Gas Phase
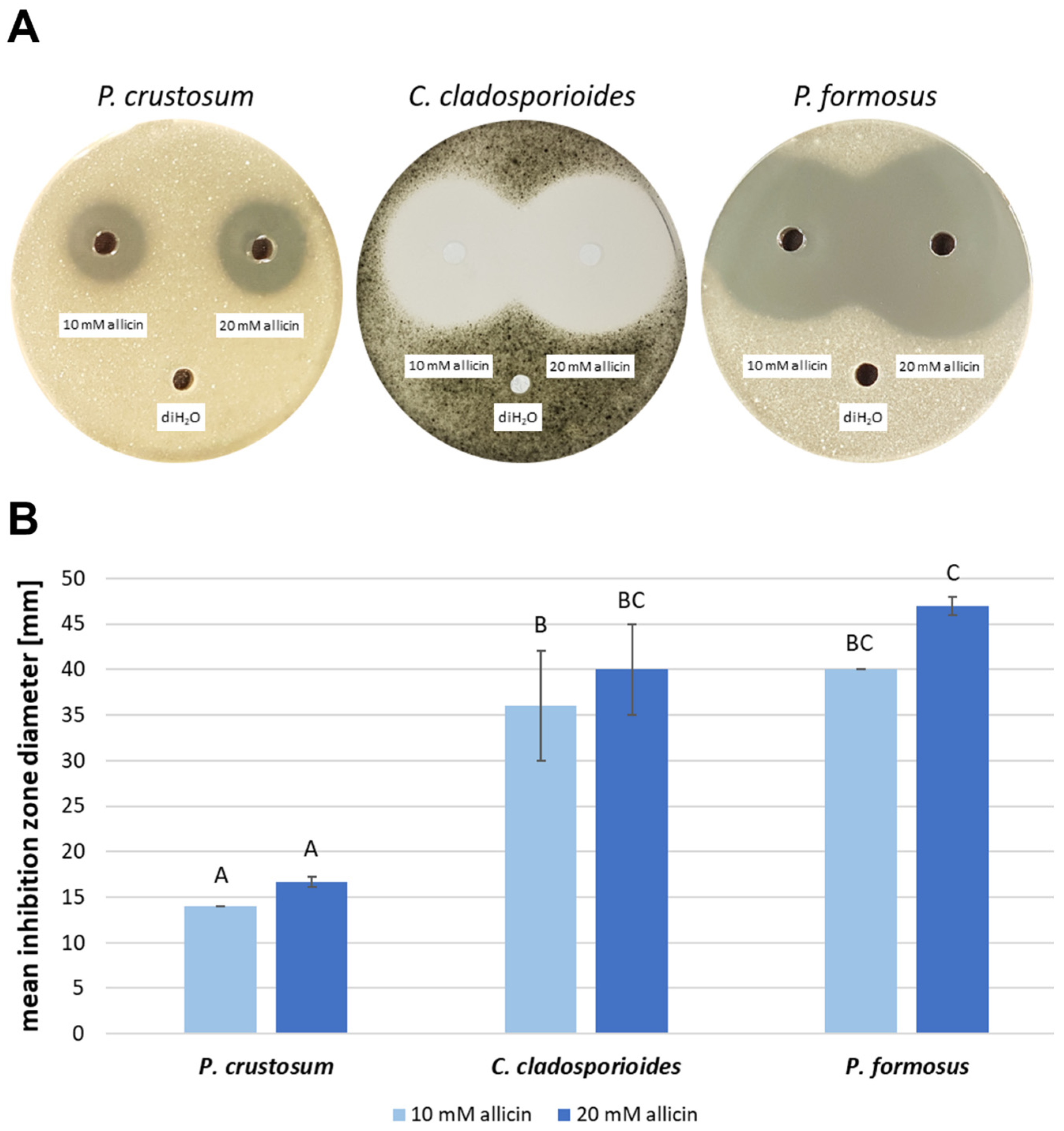
2.3. Comparison of Allicin Efficacy with That of Amphotericin B
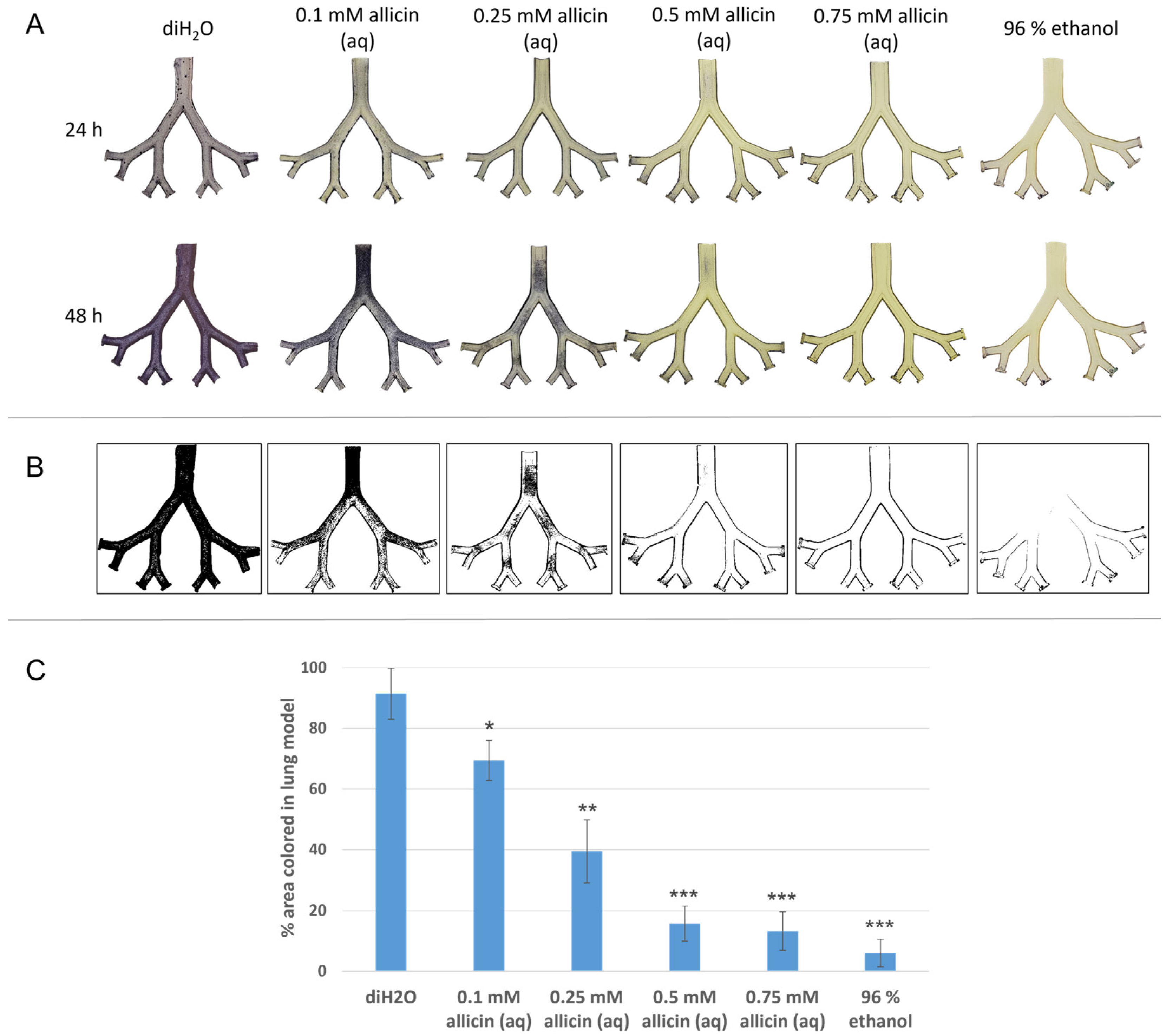
2.4. Allicin Aerosol Inhibits Fungal Growth in an Artificial Lung Model
2.5. Enhancement of Allicin Vapour Efficacy Using Ethanol as Solvent
3. Discussion
4. Materials and Methods
4.1. Allicin Synthesis
4.2. Isolation and Characterisation of Fungi
4.3. Sequencing and Species Identification
4.4. Cultivation Methods
4.5. Susceptibility Assays
4.6. MIC and EC50 Determination
4.7. Lung Model Experiments
4.8. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. List of Culture Media
| Potato dextrose agar | PDA (39 g potato glucose agar (AppliChem, Darmstadt, Germany) |
| Czapek agar | CZA (30 g saccharose, 2.5 g NaNO3, 1 g K2HPO4, 0.5 g KCl, 0.5 g MgSO4 × 7H2O, 0.01 g FeSO4 × 7H2O) |
| Malt extract agar | MEA (15 g malt extract (Carl Roth, Karlsruhe, Germany)) |
| Sabouraud agar | Sabo (40 g glucose/maltose monohydrate, 10 g peptone) |
| Oatmeal agar | OA (30 g of oat flakes were brought to a boil in 1 L deionised water and allowed to simmer gently for 2 h. The oat flakes were then suspended. The suspension was filtered and agar was added to the filtrate.) |
Appendix B. Macroscopic and Microscopic Characteristics of the Isolated Fungi
Appendix B.1. Penicillium crustosum

Appendix B.2. Paecilomyces formosus
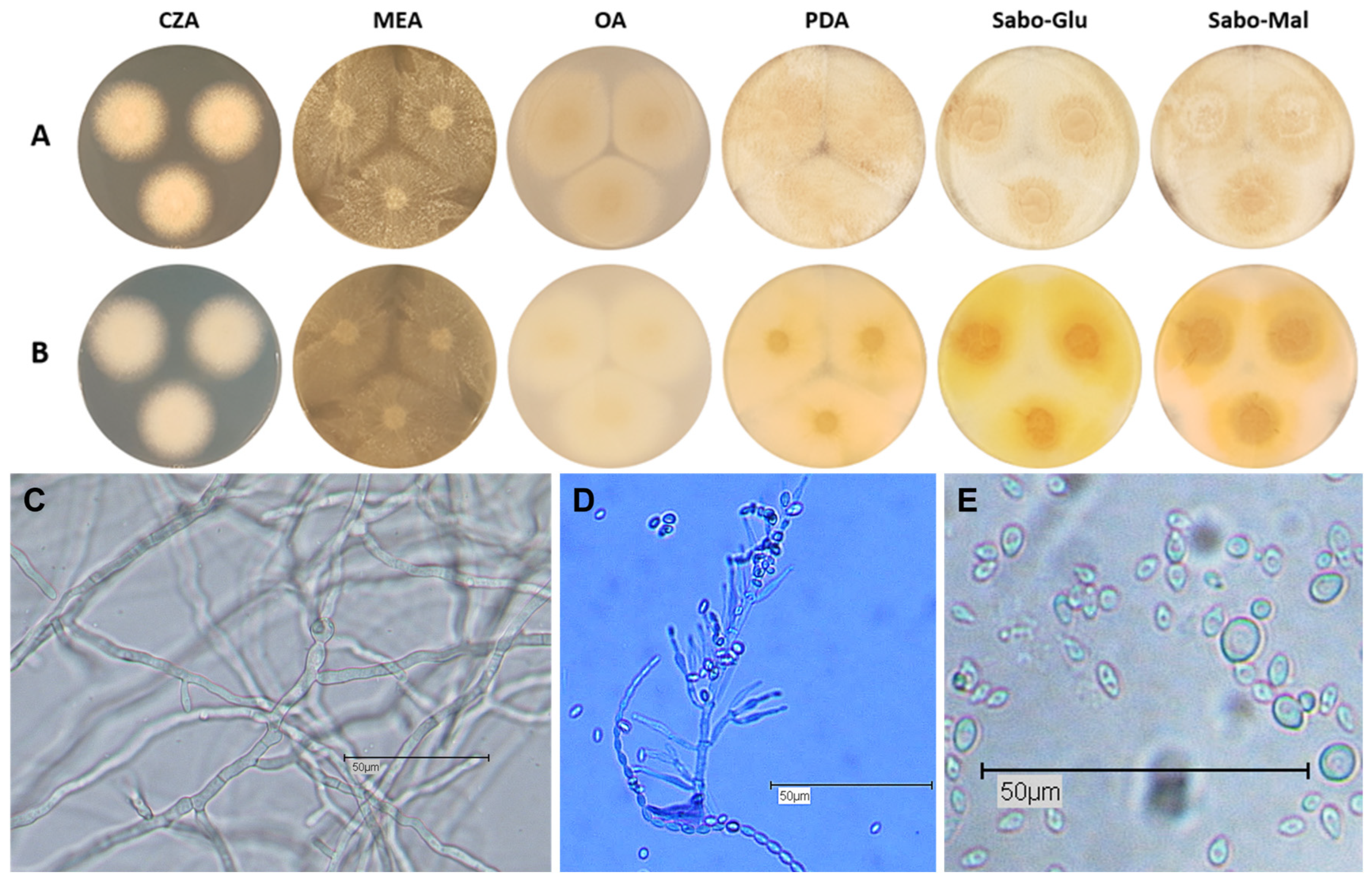
Appendix B.3. Cladosporium cladosporioides

References
- Köhler, J.R.; Casadevall, A.; Perfect, J. The spectrum of fungi that infects humans. Cold Spring Harb. Perspect. Med. 2014, 5, a019273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.; Sudbery, P. Candida albicans, a major human fungal pathogen. J. Microbiol. 2011, 49, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Solano Aquilar, E.; Negroni, R.; Meinhof, W.; Padilha-Gonçalves, A.; Pappagianis, D.; Iwata, K.; González Mendoza, A.; Graybill, J.R. Systemic Mycosis. In Dermatology in Five Continents; Orfanos, C.E., Stadler, R., Gollnick, H., Eds.; Springer: Berlin/Heidelberg, Germany, 1988; pp. 355–376. [Google Scholar] [CrossRef]
- Burns, T.; Breathnach, S.M.; Cox, N.; Griffiths, C. Rook’s Textbook of Dermatology, 8th ed.; Wiley-Blackwell: New York, NY, USA, 2013. [Google Scholar]
- Malcolm, T.R.; Chin-Hong, P.V. Endemic mycoses in immunocompromised hosts. Curr. Infect. Dis. Rep. 2013, 15, 536–543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Latgé, J.P. Aspergillus fumigatus and aspergillosis. Clin. Microbiol. Rev. 1999, 12, 310–350. [Google Scholar] [CrossRef] [Green Version]
- Youssef, C.; Widlus, D.M. Imaging diagnosis of aspergilloma. J. Community Hosp. Intern. Med. Perspect. 2012, 2, 17276. [Google Scholar] [CrossRef]
- Gostinčar, C.; Zajc, J.; Lenassi, M.; Plemenitaš, A.; de Hoog, S.; Al-Hatmi, A.M.S.; Gunde-Cimerman, N. Fungi between extremotolerance and opportunistic pathogenicity on humans. Fungal Div. 2018, 93, 195–213. [Google Scholar] [CrossRef] [Green Version]
- Pemán, J.; Ruiz-Gaitán, A.; García-Vidal, C.; Salavert, M.; Ramírez, P.; Puchades, F.; García-Hita, M.; Alastruey-Izquierdo, A.; Quindós, G. Fungal co-infection in COVID-19 patients: Should we be concerned? Rev. Iberoam. Micol. 2020, 37, 41–46. [Google Scholar] [CrossRef]
- Lansbury, L.; Lim, B.; Baskaran, V.; Lim, W.S. Co-infections in people with COVID-19: A systematic review and meta-analysis. J. Infect. 2020, 81, 266–275. [Google Scholar] [CrossRef]
- Garg, D.; Muthu, V.; Sehgal, I.S.; Ramachandran, R.; Kaur, H.; Bhalla, A.; Puri, G.D.; Chakrabarti, A.; Agarwal, R. Coronavirus Disease (Covid-19) Associated Mucormycosis (CAM): Case Report and Systematic Review of Literature. Mycopathologia 2021, 186, 289–298. [Google Scholar] [CrossRef]
- Mahalaxmi, I.; Jayaramayya, K.; Venkatesan, D.; Subramaniam, M.D.; Renu, K.; Vijayakumar, P.; Narayanasamy, A.; Gopalakrishnan, A.V.; Kumar, N.S.; Sivaprakash, P.; et al. Mucormycosis: An opportunistic pathogen during COVID-19. Environ. Res. 2021, 201, 111643. [Google Scholar] [CrossRef]
- García-Carnero, L.C.; Mora-Montes, H.M. Mucormycosis and COVID-19-Associated Mucormycosis: Insights of a Deadly but Neglected Mycosis. J. Fungi 2022, 8, 445. [Google Scholar] [CrossRef] [PubMed]
- Prakash, H.; Chakrabarti, A. Global epidemiology of mucormycosis. J. Fungi 2019, 5, 26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prakash, H.; Chakrabarti, A. Epidemiology of mucormycosis in India. Microorganisms 2021, 9, 523. [Google Scholar] [CrossRef] [PubMed]
- Hoenigl, M.; Seidel, D.; Carvalho, A.; Rudramurthy, S.M.; Arastehfar, A.; Gangneux, J.-P.; Nasir, N.; Bonifaz, A.; Araiza, J.; Klimko, N.; et al. The emergence of COVID-19 associated mucormycosis: A review of cases from 18 countries. Lancet Microbe 2022, 5247, 1–10. [Google Scholar] [CrossRef]
- Scorzoni, L.; de Paula e Silva, A.C.A.; Marcos, C.M.; Assato, P.A.; de Melo, W.C.M.A.; de Oliveira, H.C.; Costa-Orlandi, C.B.; Mendes-Giannini, M.J.S.; Fusco-Almeida, A.M. Antifungal Therapy: New Advances in the Understanding and Treatment of Mycosis. Front. Microbiol. 2017, 8, 36. [Google Scholar] [CrossRef] [Green Version]
- Mori, T.; Matsumura, M.; Ebe, T.; Takahashi, M.; Kohara, T.; Inagaki, M.; Isonuma, H.; Hibiya, I.; Hamamoto, T.; Watanabe, K. Fluconazole treatment of systemic mycoses. Jpn. J. Antibiot. 1989, 42, 55–62. [Google Scholar]
- Marichal, P.; Vanden Bossche, H. Mechanisms of resistance to azole antifungals. Acta Biochim. Pol. 1995, 42, 509–516. [Google Scholar] [CrossRef] [Green Version]
- Sanglard, D.; Ischer, F.; Bille, J. Role of ATP-binding-cassette transporter genes in high-frequency acquisition of resistance to azole antifungals in Candida glabrata. Antimicrob. Agents Chemother. 2001, 45, 1174–1183. [Google Scholar] [CrossRef] [Green Version]
- Khandelwal, N.K.; Chauhan, N.; Sarkar, P.; Esquivel, B.D.; Coccetti, P.; Singh, A.; Coste, A.T.; Gupta, M.; Sanglard, D.; White, T.C.; et al. Azole resistance in a Candida albicans mutant lacking the ABC transporter CDR6/ROA1 depends on TOR signaling. J. Biol. Chem. 2018, 293, 412–432. [Google Scholar] [CrossRef] [Green Version]
- Gray, K.C.; Palacios, D.S.; Dailey, I.; Endo, M.M.; Uno, B.E.; Wilcock, B.C.; Burke, M.D. Amphotericin primarily kills yeast by simply binding ergosterol. Proc. Natl. Acad. Sci. USA 2012, 109, 2234–2239. [Google Scholar] [CrossRef] [Green Version]
- Baginski, M.; Sternal, K.; Czub, J.; Borowski, E. Molecular modelling of membrane activity of amphotericin B, a polyene macrolide antifungal antibiotic. Acta Biochim. Pol. 2005, 52, 655–658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barrett, J.P.; Vardulaki, K.A.; Conlon, C.; Cooke, J.; Daza-Ramirez, P.; Glyn, E.; Evans, V.; Hawkey, P.M.; Herbrecht, R.; Marks, D.I.; et al. A systematic review of the antifungal effectiveness and tolerability of amphotericin B formulations. Clin. Ther. 2003, 25, 1295–1320. [Google Scholar] [CrossRef]
- Esposito, E.; Bortolotti, F.; Menegatti, E.; Cortesi, R. Amphiphilic association systems for Amphotericin B delivery. Int. J. Pharm. 2003, 260, 249–260. [Google Scholar] [CrossRef]
- Sabra, R.; Branch, R.A. Amphotericin B nephrotoxicity. Drug Saf. 1990, 5, 94–108. [Google Scholar] [CrossRef]
- Borlinghaus, J.; Albrecht, F.; Gruhlke, M.C.; Nwachukwu, I.D.; Slusarenko, A.J. Allicin: Chemistry and biological properties. Molecules 2014, 19, 12591–12618. [Google Scholar] [CrossRef] [Green Version]
- Borlinghaus, J.; Bolger, A.; Schier, C.; Vogel, A.; Usadel, B.; Gruhlke, M.C.H.; Slusarenko, A.J. Genetic and molecular characterization of multicomponent resistance of Pseudomonas against allicin. Life Sci. Alliance 2020, 3, e202000670. [Google Scholar] [CrossRef] [Green Version]
- Cavallito, C.J.; Bailey, J.H. Allicin, the Antibacterial Principle of Allium sativum. I. Isolation, Physical Properties and Antibacterial Action. J. Am. Chem. Soc. 1944, 66, 1950–1951. [Google Scholar] [CrossRef]
- Pasteur, L. Memoire sur la fermentation appelee lactique. Mem. Soc. Imp. Sci. Agric. 1858, 5, 13–26. [Google Scholar]
- Borlinghaus, J.; Foerster, J.; Kappler, U.; Antelmann, H.; Noll, U.; Gruhlke, M.C.H.; Slusarenko, A.J. Allicin, the Odor of Freshly Crushed Garlic: A Review of Recent Progress in Understanding Allicin’s Effects on Cells. Molecules 2021, 26, 1505. [Google Scholar] [CrossRef]
- Curtis, H.; Noll, U.; Störmann, J.; Slusarenko, A.J. Broad-spectrum activity of the volatile phytoanticipin allicin in extracts of garlic (Allium sativum L.) against plant pathogenic bacteria, fungi and Oomycetes. Physiol. Mol. Plant Pathol. 2004, 65, 79–89. [Google Scholar] [CrossRef]
- Weber, N.D.; Andersen, D.O.; North, J.A.; Murray, B.K.; Lawson, L.D.; Hughes, B.G. In vitro virucidal effects of Allium sativum (garlic) extract and compounds. Planta Med. 1992, 58, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Gruhlke, M.C.; Nicco, C.; Batteux, F.; Slusarenko, A.J. The Effects of Allicin, a Reactive Sulfur Species from Garlic, on a Selection of Mammalian Cell Lines. Antioxidants 2017, 6, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gruhlke, M.C.H.; Antelmann, H.; Bernhardt, J.; Kloubert, V.; Rink, L.; Slusarenko, A.J. The human allicin-proteome: S-thioallylation of proteins by the garlic defence substance allicin and its biological effects. Free Radic. Biol. Med. 2019, 131, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Wills, E.D. Enzyme inhibition by allicin, the active principle of garlic. Biochem. J. 1956, 63, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Prager-Khoutorsky, M.; Goncharov, I.; Rabinkov, A.; Mirelman, D.; Geiger, B.; Bershadsky, A.D. Allicin inhibits cell polarization, migration and division via its direct effect on microtubules. Cell Motil. Cytoskel. 2007, 64, 321–337. [Google Scholar] [CrossRef] [PubMed]
- Noll, U.; Schreiber, M.; Hermanns, M.; Mertes, C.A.; Slusarenko, A.J.; Gruhlke, M.C.H. Allicin From Garlic Disrupts the Cytoskeleton in Plants. bioRxiv 2021. [Google Scholar] [CrossRef]
- Loi, V.V.; Huyen, N.T.T.; Busche, T.; Tung, Q.N.; Gruhlke, M.C.H.; Kalinowski, J.; Bernhardt, J.; Slusarenko, A.J.; Antelmann, H. Staphylococcus aureus responds to allicin by global S-thioallylation—Role of the Brx/BSH/YpdA pathway and the disulfide reductase MerA to overcome allicin stress. Free Radic. Biol. Med. 2019, 139, 55–69. [Google Scholar] [CrossRef]
- Mösbauer, K.; Fritsch, V.N.; Adrian, L.; Bernhardt, J.; Gruhlke, M.C.H.; Slusarenko, A.J.; Niemeyer, D.; Antelmann, H. The Effect of Allicin on the Proteome of SARS-CoV-2 Infected Calu-3 Cells. Front. Microbiol. 2021, 12, 746795. [Google Scholar] [CrossRef]
- Reiter, J.; Levina, N.; Van der Linden, M.; Gruhlke, M.; Martin, C.; Slusarenko, A.J. Diallylthiosulfinate (Allicin), a Volatile Antimicrobial from Garlic (Allium sativum), Kills Human Lung Pathogenic Bacteria, Including MDR Strains, as a Vapor. Molecules 2017, 22, 1711. [Google Scholar] [CrossRef] [Green Version]
- Reiter, J.; Borlinghaus, J.; Dörner, P.; Schröder, W.; Gruhlke, M.C.H.; Klaas, M.; Slusarenko, A.J. Investigation of the deposition behaviour and antibacterial effectivity of allicin aerosols and vapour using a lung model. Exp. Ther. Med. 2020, 19, 1541–1549. [Google Scholar] [CrossRef] [Green Version]
- Dörner, P.; Müller, P.M.; Reiter, J.; Gruhlke, M.C.; Slusarenko, A.J.; Schröder, W.; Klaas, M. Feasibility study of a surface-coated lung model to quantify active agent deposition for preclinical studies. Clin. Biomech. 2020, 76, 105029. [Google Scholar] [CrossRef] [PubMed]
- Leontiev, R.; Hohaus, N.; Jacob, C.; Gruhlke, M.C.H.; Slusarenko, A.J. A Comparison of the Antibacterial and Antifungal Activities of Thiosulfinate Analogues of Allicin. Sci. Rep. 2018, 8, 6763. [Google Scholar] [CrossRef] [PubMed]
- Perelló, A.; Noll, U.; Slusarenko, A.J. In vitro efficacy of garlic extract to control fungal pathogens of wheat. J. Med. Plants Res. 2013, 7, 1809–1817. [Google Scholar]
- Lai, C.C.; Chen, S.Y.; Ko, W.C.; Hsueh, P.R. Increased antimicrobial resistance during the COVID-19 pandemic. Int. J. Antimicrob. Agents 2021, 57, 106324. [Google Scholar] [CrossRef]
- Burks, C.; Darby, A.; Gómez Londoño, L.; Momany, M.; Brewer, M.T. Azole-resistant Aspergillus fumigatus in the environment: Identifying key reservoirs and hotspots of antifungal resistance. PLoS Pathog. 2021, 17, e1009711. [Google Scholar] [CrossRef]
- Zhang, J.; Debets, A.J.M.; Verweij, P.E.; Snelders, E. Azole-Resistance Development; How the Aspergillus fumigatus Lifecycle Defines the Potential for Adaptation. J. Fungi 2021, 7, 599. [Google Scholar] [CrossRef]
- Ma, X.; Hu, J.; Yu, Y.; Wang, C.; Gu, Y.; Cao, S.; Huang, X.; Wen, Y.; Zhao, Q.; Wu, R.; et al. Assessment of the pulmonary adaptive immune response to Cladosporium cladosporioides infection using an experimental mouse model. Sci. Rep. 2021, 11, 909. [Google Scholar] [CrossRef]
- Heshmatnia, J.; Marjani, M.; Mahdaviani, S.A.; Adimi, P.; Pourabdollah, M.; Tabarsi, P.; Mahdavi, F.; Jamaati, H.; Adcock, I.M.; Garssen, J.; et al. Paecilomyces formosus Infection in an Adult Patient with Undiagnosed Chronic Granulomatous Disease. J. Clin. Immun. 2017, 37, 342–346. [Google Scholar] [CrossRef]
- Batarseh, R.Y.; Shehata, M.; Becker, M.D.; Sigdel, S.; He, P.; Shweihat, Y.R. Paecilomyces in an immune competent host. IDCases 2020, 21, e00885. [Google Scholar] [CrossRef]
- Hohenadel, I.A.; Kiworr, M.; Genitsariotis, R.; Zeidler, D.; Lorenz, J. Role of bronchoalveolar lavage in immunocompromised patients with pneumonia treated with a broad spectrum antibiotic and antifungal regimen. Thorax 2001, 56, 115. [Google Scholar] [CrossRef] [Green Version]
- Albrecht, F.; Leontiev, R.; Jacob, C.; Slusarenko, A.J. An Optimized Facile Procedure to Synthesize and Purify Allicin. Molecules 2017, 22, 770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raja, H.A.; Miller, A.N.; Pearce, C.J.; Oberlies, N.H. Fungal Identification Using Molecular Tools: A Primer for the Natural Products Research Community. J. Nat. Prod. 2017, 80, 756–770. [Google Scholar] [CrossRef] [PubMed]
- EPPO Standards. PM 7/129 (1) DNA barcoding as an identification tool for a number of regulated pests. EPPO Bull. 2016, 46, 501–537. [Google Scholar] [CrossRef] [Green Version]



| Treatment | Aerosol | Vapour | |||||
|---|---|---|---|---|---|---|---|
| Allicin concentration [mM] | 0.1 | 0.25 | 0.5 | 0.75 | 20 | 40 | 20 |
| Solvent | deionised water | deionised water | 96% ethanol | ||||
| Mean treatment volume [mL] | 4.581 (±0.020) | 4.974 (±0.041) | 5.001 (±0.073) | 4.935 (±0.175) | 0.273 (±0.033) | 0.454 (±0.021) | 1.379 (±0.003) |
| Mean allicin consumed [µmol] | 0.46 (±0.002) | 1.2 (±0.01) | 2.5 (±0.04) | 3.7 (±0.13) | 5.5 (±0.7) | 18 (±1.4) | 28 (±0.1) |
| Amplicon | Primer Name | Primer Sequence (5′-3′) | Source |
|---|---|---|---|
| ITS1 | ITS1F | CTT GGT CAT TTA GAG GAA GTA A | [54] |
| ITS2 | GCT GCG TTC TTC ATC GAT GC | ||
| TUB2 | TUB2Fd | GTB CAC CTY CAR ACC GGY CAR TG | [55] |
| TUB4Rd | CCR GAY TGR CCR AAR ACR AAG TTG TC | ||
| CALM | CAL-228F | GAG TTC AAG GAG GCC TTC TCC C | [55] |
| CAL-737R | CAT CTT TCT GGC CAT CAT GG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schier, C.; Foerster, J.; Heupel, M.; Dörner, P.; Klaas, M.; Schröder, W.; Rink, L.; Slusarenko, A.J.; Gruhlke, M.C.H. Allicin as a Volatile or Nebulisable Antimycotic for the Treatment of Pulmonary Mycoses: In Vitro Studies Using a Lung Flow Test Rig. Int. J. Mol. Sci. 2022, 23, 6607. https://doi.org/10.3390/ijms23126607
Schier C, Foerster J, Heupel M, Dörner P, Klaas M, Schröder W, Rink L, Slusarenko AJ, Gruhlke MCH. Allicin as a Volatile or Nebulisable Antimycotic for the Treatment of Pulmonary Mycoses: In Vitro Studies Using a Lung Flow Test Rig. International Journal of Molecular Sciences. 2022; 23(12):6607. https://doi.org/10.3390/ijms23126607
Chicago/Turabian StyleSchier, Christina, Jana Foerster (née Reiter), Monika Heupel, Philipp Dörner, Michael Klaas, Wolfgang Schröder, Lothar Rink, Alan J. Slusarenko, and Martin C. H. Gruhlke. 2022. "Allicin as a Volatile or Nebulisable Antimycotic for the Treatment of Pulmonary Mycoses: In Vitro Studies Using a Lung Flow Test Rig" International Journal of Molecular Sciences 23, no. 12: 6607. https://doi.org/10.3390/ijms23126607






