Effect of Cationic (Na+) and Anionic (F−) Co-Doping on the Structural and Electrochemical Properties of LiNi1/3Mn1/3Co1/3O2 Cathode Material for Lithium-Ion Batteries
Abstract
:1. Introduction
2. Results and Discussion
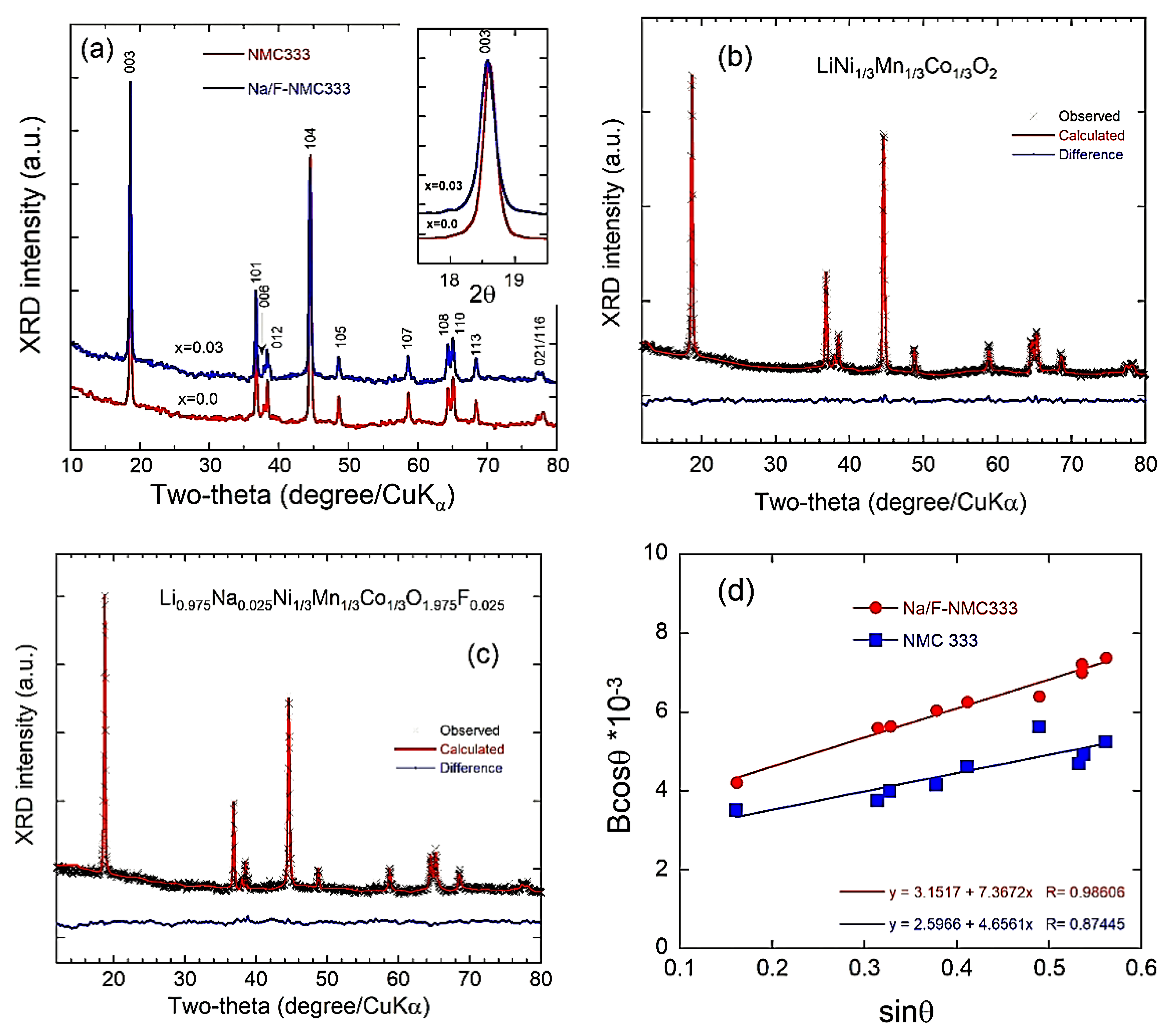
2.1. Structure and Morphology
2.2. Electrochemical Behavior
3. Conclusions
4. Materials and Methods
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Julien, C.M.; Mauger, A.; Vijh, A.; Zaghib, K. Lithium Batteries: Science and Technology; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Manthiram, A. A reflection on lithium-ion battery cathode chemistry. Nat. Commun. 2020, 11, 1550. [Google Scholar] [CrossRef] [PubMed]
- Julien, C.M.; Mauger, A.; Zaghib, K.; Groult, H. Comparative issues of cathode materials for Li-ion batteries. Inorganics 2014, 2, 132–154. [Google Scholar] [CrossRef] [Green Version]
- Ohzuku, T.; Makimura, Y. Layered lithium insertion material of LiNi1/3Mn1/3Co1/3O2 for lithium-ion batteries. J. Chem. Lett. 2001, 7, 642–643. [Google Scholar] [CrossRef]
- Shaju, K.M.; Subba Rao, G.V.; Chowdari, B.V.R. Performance of layered LiNi1/3Mn1/3Co1/3O2 as cathode for Li-ion batteries. J. Electrochim. Acta 2002, 48, 145–151. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, J.; Chao, D.; Baikie, T.; Bai, L.; Chen, S.; Zhao, Y.; Sum, T.C.; Lin, J.; Shen, Z. Hierarchical porous LiNi1/3Co1/3Mn1/3O2 nano-/micro spherical cathode material: Minimized cation mixing and improved Li+ mobility for enhanced electrochemical performance. Sci. Rep. 2016, 6, 25771. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fey, G.T.K.; Chang, C.-S.; Prem Kumar, T. Synthesis and surface treatment of LiNi1/3Co1/3Mn1/3O2 cathode materials for Li-ion batteries. J. Solid State Electrochem. 2010, 14, 17–26. [Google Scholar]
- Li, J.B.; Xu, Y.L.; Xiong, L.L.; Wang, J.P. Improvement of LiNi1/3Co1/3Mn1/3O2 cathode materials by nano-MgO doping. Acta Phys. Chim. Sin. 2011, 27, 2593–2599. [Google Scholar]
- Hashem, A.M.; Abdel-Ghany, A.E.; Scheuermann, M.; Indris, S.; Ehrenberg, H.; Mauger, A.; Julien, C.M. Doped nanoscale NMC333 as cathode materials for Li-ion batteries. Materials 2019, 12, 2899. [Google Scholar] [CrossRef] [Green Version]
- Ding, Y.; Zhang, P.; Long, Z.; Jiang, Y.; Xu, F. Morphology and electrochemical properties of Al doped LiNi1/3Co1/3Mn1/3O2 nanofibers prepared by electrospinning. J. Alloys Compd. 2009, 487, 507–510. [Google Scholar] [CrossRef]
- Liu, L.; Sun, K.; Zhang, N.; Yang, T. Improvement of high-voltage cycling behavior of Li(Ni1/3Co1/3Mn1/3)O2 cathodes by Mg, Cr, and Al substitution. J. Solid State Electrochem. 2009, 13, 1381–1386. [Google Scholar] [CrossRef]
- Xiao, W.; Nie, Y.; Miao, C.; Wang, J.; Tan, Y.; Wen, M. Structural design of high-performance Ni-rich LiNi0.83Co0.11Mn0.06O2 cathode materials enhanced by Mg2+ doping and Li3PO4 coating for lithium ion battery. J. Colloid Interface Sci. 2022, 607, 1071–1082. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.X.; Bai, Y.C.; Feng, X.Y.; Chen, C.H. Improvement of electrochemical properties of layered LiNi1/3Co1/3Mn1/3O2 positive electrode material by zirconium doping. Solid State Ion. 2011, 189, 69–73. [Google Scholar] [CrossRef]
- Wang, J.; Nie, Y.; Miao, C.; Tan, Y.; Wen, M.; Xiao, W. Enhanced electrochemical properties of Ni-rich layered cathode materials via Mg2+ and Ti4+ co-doping for lithium-ion batteries. J. Colloid Interface Sci. 2021, 601, 853–862. [Google Scholar] [CrossRef]
- Gong, C.; Lv, W.; Qu, L.; Bankole, O.E.; Li, G.; Zhang, R.; Hu, M.; Lei, L. Synthesis and electrochemical properties of layered Li0.95Na0.05Ni1/3Co1/3Mn1/3O2 and LiNi1/3Co1/3Mn1/3O2. J. Power Sources 2014, 247, 151–155. [Google Scholar] [CrossRef]
- Chen, Z.; Xie, T.; Li, L.; Xu, M.; Zhu, H.; Wan, W. Characterization of Na-substituted LiNi1/3Co1/3Mn1/3O2 cathode materials for lithium-ion battery. Ionics 2014, 20, 629–634. [Google Scholar] [CrossRef]
- Li, Y.; Liu, J.; Lei, Y.; Lai, C.; Xu, Q. Enhanced electrochemical performances of Na-doped cathode material LiNi1/3Co1/3Mn1/3O2 for lithium-ion batteries. J. Mater. Sci. 2017, 52, 13596–13605. [Google Scholar] [CrossRef]
- Guo, L.-F.; Xie, Y.-L. Na-doped LiNi1/3Co1/3Mn1/3O2 with enhanced rate performance as a cathode for Li-ion batteries. Ionics 2022, 28, 2117–2123. [Google Scholar] [CrossRef]
- Yang, Z.G.; Guo, X.D.; Xiang, W.; Hua, W.B.; Zhang, J.; He, F.R.; Wang, K.; Xiao, Y.; Zhong, B. K-doped layered LiNi0.5Co0.2Mn0.3O2 cathode material: Towards the superior rate capability and cycling performance. J. Alloys Compd. 2017, 699, 358–365. [Google Scholar] [CrossRef]
- Kageyama, M.; Li, D.; Kobayakawa, K.; Sato, Y.; Lee, Y.-S. Structural and electrochemical properties of LiNi1/3Mn1/3Co1/3O2-xFx prepared by solid state reaction. J. Power Sources 2006, 157, 494–500. [Google Scholar] [CrossRef]
- He, Y.-S.; Pei, L.; Liao, X.-Z.; Ma, Z.-F. Synthesis of LiNi1/3Co1/3Mn1/3O2-xFx cathode material from oxalate precursors for lithium ion battery. J. Fluor. Chem. 2007, 128, 139–143. [Google Scholar] [CrossRef]
- Liao, L.; Wang, X.; Luo, X.; Wang, X.; Gamboa, S.; Sebastian, P.J. Synthesis and electrochemical properties of layered Li[Ni0.333Co0.333Mn0.333Al0.04]O2-xFx cathode materials prepared by the sol–gel method. J. Power Sources 2006, 160, 657–661. [Google Scholar] [CrossRef]
- Ye, S.; Xia, Y.; Zhang, P.; Qiao, Z. Al, B, and F doped LiNi1/3Co1/3Mn1/3O2 as cathode material of lithium-ion batteries. J. Solid State Electrochem. 2007, 11, 805–810. [Google Scholar] [CrossRef]
- Zhang, H.-L.; Liu, S. Synthesis and characterization of LiNi1/3Co1/3Mn1/3O2-xClx as cathode materials for lithium ion batteries at 55 °C. Adv. Mater. Sci. Eng. 2013, 2013, 375074. [Google Scholar] [CrossRef] [Green Version]
- Cong, L.; Zhao, Q.; Wang, Z.; Zhang, Y.; Wu, X.; Zhang, J.; Wang, R.; Xie, H.; Sun, L. (PO4)3− polyanions doped LiNi1/3Co1/3Mn1/3O2: An ultrafast-rate, long-life and high-voltage cathode material for Li-ion rechargeable batteries. Electrochim. Acta 2016, 201, 8–19. [Google Scholar] [CrossRef]
- Gao, Y.; Shen, K.; Liu, L.; Chi, F.; Hou, X.; Yang, W. First-principles investigation on electrochemical performance of Na-doped LiNi1/3Co1/3Mn1/3O2. Front. Phys. 2021, 8, 616066. [Google Scholar] [CrossRef]
- Vanaphuti, P.; Chen, J.; Cao, J.; Bigham, K.; Chen, B.; Yang, L.; Chen, H.; Wang, Y. Enhanced electrochemical performance of the lithium-manganese-rich cathode for Li-ion batteries with Na and F cooping. ACS Appl. Mater. Interfaces 2019, 11, 37842–37849. [Google Scholar] [CrossRef]
- Wang, J.; Liu, C.; Xu, G.; Miao, C.; Wen, M.; Xu, M.; Wang, C.; Xiao, W. Strengthened the structural stability of in-situ F-doping Ni-rich LiNi0.8Co0.15Al0.05O2 cathode materials for lithium-ion batteries. Chem. Eng. J. 2022, 438, 135537. [Google Scholar] [CrossRef]
- Wang, D.; Wang, Z.; Li, X.; Guo, H.; Xu, Y.; Fan, Y.; Pan, W. Effects of surface fluorine substitution on high voltage electrochemical performances of layered LiNi0.5Co0.2Mn0.3O2 cathode materials. Appl. Surf. Sci. 2016, 371, 172–179. [Google Scholar] [CrossRef]
- Pang, W.K.; Lin, H.-F.; Peterson, V.K.; Lu, C.-Z.; Liu, C.-E.; Liao, S.-C.; Chen, J.-M. Effects of fluorine and chromium doping on the performance of lithium-rich Li1+xMO2 (M = Ni, Mn, Co) positive electrodes. Chem. Mater. 2017, 29, 10299–10311. [Google Scholar] [CrossRef] [Green Version]
- Xiang, W.; Zhu, C.-Q.; Zhang, J.; Shi, H.; Liang, Y.-T.; Yu, M.-H.; Zhu, X.-M.; He, F.-R.; Lv, G.-P.; Guo, X.-D. Synergistic coupling effect of sodium and fluorine co-substitution on enhancing rate capability and cycling performance of Ni-rich cathode for lithium ion battery. J. Alloys Compd. 2019, 786, 56–64. [Google Scholar] [CrossRef]
- Vanaphuti, P.; Bai, J.; Ma, L.; Ehrlich, S.; Kisslinger, K.; Wang, F.; Wang, Y. Unraveling Na and F coupling effects in stabilizing Li, Mn-rich layered oxide cathodes via ordering modification. Energy Storage Mater. 2020, 31, 459–469. [Google Scholar] [CrossRef]
- Liu, D.; Fan, X.; Li, Z.; Liu, T.; Sun, M.; Qian, C.; Ling, M.; Liu, Y.; Liang, C. A cation/anion co-doped Li1.12Na0.08Ni0.2Mn0.6O1.95F0.05 cathode for lithium ion batteries. Nano Energy 2019, 58, 786–796. [Google Scholar] [CrossRef]
- Hashem, A.M.; El-Taweel, R.S.; Abuzeid, H.M.; Abdel-Ghany, A.E.; Eid, A.E.; Groult, H.; Mauger, A.; Julien, C.M. Structural and electrochemical properties of LiNi1/3Co1/3Mn1/3O2 material prepared by a two-step synthesis via oxalate precursor. Ionics 2012, 18, 1–9. [Google Scholar] [CrossRef]
- Ohzuku, T.; Ueda, A.; Nagayama, M. Electrochemistry and structural chemistry of LiNiO2 (R3m) for 4 volt secondary lithium cells. J. Electrochem. Soc. 1993, 140, 1862–1870. [Google Scholar] [CrossRef]
- Chen, Z.; Gong, X.; Zhu, H.; Cao, K.; Liu, Q.; Liu, J.; Li, L.; Duan, J. High performance and structural stability of K and Cl co-doped LiNi0.5Co0.2Mn0.3O2 cathode materials in 4.6 voltage. Front. Chem. 2019, 6, 643. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G.K.; Hall, W.H. X-ray line broadening from filed aluminium and wolfram. Acta Metall. 1953, 1, 22–31. [Google Scholar] [CrossRef]
- Zheng, J.; Gu, M.; Genc, A.; Xiao, J.; Xu, P.; Chen, X.; Zhu, Z.; Zhao, W.; Pullan, L.; Wang, C.; et al. Mitigating voltage fade in cathode materials by improving the atomic level uniformity of elemental distribution. Nano Lett. 2014, 14, 2628–2635. [Google Scholar] [CrossRef]
- Zheng, J.; Wu, X.; Yang, Y. Improved electrochemical performance of Li[Li0.2Mn0.54Ni0.13Co0.13]O2 cathode material by fluorine incorporation. Electrochim. Acta 2013, 105, 200–208. [Google Scholar] [CrossRef]
- Guo, B.; Zhao, J.; Fan, X.; Zhang, W.; Li, S.; Yang, Z.; Chen, Z.; Zang, W. Aluminum and fluorine co-doping for promotion of stability and safety of lithium-rich layered cathode material. Electrochim. Acta 2017, 236, 171–179. [Google Scholar] [CrossRef]
- Sotomayor, F.; Cychosz, K.A.; Thommes, M. Characterization of micro/mesoporous materials by physisorption: Concepts and case studies. Acc. Mater. Surf. Res. 2018, 3, 34–50. [Google Scholar]
- Thiele, G.; Poston, M.; Brown, R. A Case Study in Sizing Nanoparticles. Micromeritics Instrument Corporation. Available online: http://www.particletesting.com/library (accessed on 1 January 2019).
- Ben-Kamel, K.; Amdouni, N.; Mauger, A.; Julien, C.M. Study of the local structure of LiNi0.33+δMn0.33+δCo0.33−2δO2 (0.025 ≤ δ ≤ 0.075) oxides. J. Alloys Compd. 2012, 528, 91–98. [Google Scholar] [CrossRef]
- Julien, C.M.; Mauger, A. In situ Raman analyses of electrode materials for Li-ion batteries. AIMS Mater. Sci. 2018, 5, 650–698. [Google Scholar] [CrossRef]
- Deng, C.; Liu, L.; Zhou, W.; Sun, K.; Sun, D. Characterization of Li[Ni1/3Co1/3Mn1/3]O2 synthesized via salvolatile coprecipitation for lithium-ion batteries. Electrochem. Solid-State Lett. 2007, 10, A279–A282. [Google Scholar] [CrossRef]
- Luo, X.F.; Wang, X.Y.; Liao, L.; Gamboa, S.; Sebastian, P.J. Synthesis and characterization of high tap-density layered LiNi1/3Co1/3Mn1/3O2 cathode material via hydroxide co-precipitation. J. Power Sources 2006, 158, 654–658. [Google Scholar] [CrossRef]
- Li, L.; Wang, L.; Zhang, X.; Xie, M.; Wu, F.; Chen, R. Structural and electrochemical study of hierarchical LiNi1/3Co1/3Mn1/3O2 cathode material for lithium-ion batteries. ACS Appl. Mater. Interfaces 2015, 7, 21939–21947. [Google Scholar] [CrossRef]
- Yun, S.H.; Park, K.S.; Park, Y.J. The electrochemical property of ZrFx-coated Li[Ni1/3Co1/3Mn1/3]O2 cathode material. J. Power Sources 2010, 195, 6108–6115. [Google Scholar] [CrossRef]
- Huang, B.; Wang, M.; Yang, X.; Gu, Y. Facile synthesis of fluorine doped single crystal Ni-rich cathode material for lithium-ion batteries. Solid State Ion. 2019, 342, 115065. [Google Scholar]
- Woo, S.-U.; Park, B.-C.; Yoon, C.S.; Myung, S.-T.; Prakash, J.; Sun, Y.-K. Improvement of electrochemical performances of Li[Ni0.8Co0.1Mn0.1]O2 cathode material by fluorine substitution. J. Electrochem. Soc. 2007, 154, A649–A655. [Google Scholar] [CrossRef]
- Li, X.; Xie, Z.; Liu, W.; Ge, W.; Wang, H.; Qu, M. Effects of fluorine doping on structure surface chemistry, and electrochemical performance of LiNi0.8Co0.15Al0.05O2. Electrochim. Acta 2015, 174, 1122–1130. [Google Scholar] [CrossRef]
- Kim, H.; Kim, S.-B.; Park, D.-H.; Park, K.-W. Fluorine-doped LiNi0.8Mn0.1Co0.1O2 cathode for high-performance lithium-ion batteries. Energies 2020, 13, 4808. [Google Scholar] [CrossRef]
- Breddemann, U.; Krossing, I. Review on synthesis, characterization, and electrochemical properties of fluorinated nickel-cobalt-manganese cathode active materials for lithium-ion batteries. ChemElectroChem 2020, 7, 1389–1430. [Google Scholar] [CrossRef]
- Shi, S.J.; Tu, J.P.; Tang, Y.Y.; Zhang, Y.Q.; Liu, X.Y.; Wang, X.L.; Gu, C.D. Enhanced electrochemical performance of LiF-modified LiNi1/3Co1/3Mn1/3O2 cathode materials for Li-ion batteries. J. Power Sources 2013, 225, 338–346. [Google Scholar] [CrossRef]
- Yue, P.; Wang, Z.; Guo, H.; Xiong, X.; Li, X. A low temperature fluorine substitution on the electrochemical performance of layered LiNi0°8Co0.1Mn0.1O2−zFz cathode materials. Electrochim. Acta 2013, 92, 1–8. [Google Scholar] [CrossRef]
- He, R.; Zhang, L.; Yan, M.; Gao, Y.; Liu, Z. Effects of Cr2O3-modified LiNi1/3Co1/3Mn1/3O2 cathode materials on the electrochemical performance of lithium-ion batteries. J. Mater. Sci. 2016, 52, 4599–4607. [Google Scholar] [CrossRef]
- Baboo, J.P.; Park, H.; Song, J.; Kim, S.; Jo, J.; Pham, D.T.; Mathew, V.; Xiu, Z.; Kim, J. Facile redox synthesis of layered LiNi1/3Co1/3Mn1/3O2 for rechargeable Li-ion batteries. Electrochim. Acta 2017, 224, 243–250. [Google Scholar] [CrossRef]
- Nayak, P.K.; Grinblat, J.; Levi, M.; Wu, Y.; Powell, B.; Aurbach, D. TEM and Raman spectroscopy evidence of layered to spinle phase transformation in layered LiNi1/3Mn1/3Co1/3O2 upon cycling to high voltages. J. Electroanal. Chem. 2014, 733, 6–19. [Google Scholar] [CrossRef]
- Julien, C.M.; Mauger, A. Functional behavior of AlF3 coatings for high-performance cathode materials for lithium-ion batteries. AIMS Mater. Sci. 2019, 6, 406–440. [Google Scholar] [CrossRef]
- Zhang, P.; Zhai, X.; Huang, H.; Zhou, J.; Li, X.; He, Y.; Guo, Z. Synergistic Na+ and F− co-doping modification strategy to improve the electrochemical performance of Li-rich Li1.20Mn0.54Ni0.13Co0.13O2 cathode. Ceram. Int. 2020, 46, 24723–24736. [Google Scholar] [CrossRef]
- Ho, C.; Raistrick, I.D.; Huggins, R.A. Application of a-c techniques to the study of lithium diffusion in tungsten trioxide thin films. J. Electrochem. Soc. 1980, 127, 343–350. [Google Scholar] [CrossRef]
- Bard, A.J.; Faulkner, L.R. Electrochemical Methods: Fundamentals and Applications; Wiley & Sons, Inc.: New York, NY, USA, 2001; pp. 226–260. [Google Scholar]
- Ko, J.Y.; Varini, M.; Klett, M.; Ekström, H.; Lindbergh, G. Porous electrode model with particle stress effects for Li(Ni1/3Co1/3Mn1/3)O2 electrode. J. Electrochem. Soc. 2019, 166, A2939–A2949. [Google Scholar] [CrossRef] [Green Version]
- Belharouak, I.; Sun, Y.-K.; Liu, J.; Amine, K. Li(Ni1/3Co1/3Mn1/3)O2 as a suitable cathode for high power applications. J. Power Sources 2003, 123, 247–252. [Google Scholar] [CrossRef]
- Oh, S.W.; Park, S.H.; Park, C.W.; Sun, Y.K. Structural and electrochemical properties of layered Li[Ni0.5Mn0.5]1-xCoxO2 positive materials synthesized by ultrasonic spray pyrolysis method. Solid State Ion. 2004, 171, 167–172. [Google Scholar] [CrossRef]












| Crystal Data | NMC333 | Na/F-NMC333 |
|---|---|---|
| Lattice parameters | ||
| a (Å) | 2.857(3) | 2.859(7) |
| c(Å) | 14.205(3) | 14.221(6) |
| V (Å3) | 100.43 | 100.72 |
| c/a | 4.971(9) | 4.974(1) |
| Lc (nm) | 42 | 34 |
| ε × 10−2 (rd) | 1.84 | 1.16 |
| I(003)/I(104) | 1.24 | 1.32 |
| Reliability factors | ||
| Rp (%) | 9.8 | 7.8 |
| Rwp (%) | 11.9 | 12.2 |
| χ2 | 1.22 | 1.61 |
| Occupancy (Occ) | ||
| Ni2+ on Li-site% | 2.08 | 1.28 |
| Na on Li-site% | 0 | 2.47 |
| F on O-site% | 0 | 2.39 |
| Zoxy | 0.24056 | 0.24499 |
| S(MO2) (Å) a | 2.636 | 2.513 |
| I(LiO2) (Å) b | 2.099 | 2.251 |
| Nominal Formula | Mole Fraction | |||||
|---|---|---|---|---|---|---|
| Na | F | Ni | Mn | Co | O | |
| LiNi0.33Mn0.33Co0.33 O2 | - | - | 0.3411 | 0.3305 | 0.3284 | 1.998 |
| Li0.975Na0.025Ni0.33Mn0.33Co0.33O1.975F0.025 | 0.028 | 0.026 | 0.3240 | 0.3310 | 0.3454 | 1.972 |
| Material | Specific Surface Area (m2 g−1) | Average Pore Radius (nm) | Pore Volume (cm3 g−1) | LBET (nm) |
|---|---|---|---|---|
| NMC333 | 6.7 | 2.8 | 0.0075 | 196 |
| Na/F-NMC333 | 7.1 | 3.0 | 0.0086 | 185 |
| Modes | Band Position (cm−1) | Band Width (cm−1) | Band Area | |
|---|---|---|---|---|
| NMC333 | ||||
| ν1 | Eg (Ni) | 477 | 52 | 5678.4 |
| ν2 | A1g Ni) | 539 | 31 | 3226.7 |
| ν3 | Eg (Co) | 512 | 37 | 2371.7 |
| ν4 | A1g (Co) | 600 | 45 | 25,298.9 |
| ν5 | Eg (Mn) | 566 | 41 | 5950.3 |
| ν6 | A1g (Mn) | 640 | 51 | 9213.5 |
| Na/F-NMC333 | ||||
| ν1 | Eg (Ni) | 480 | 45 | 6151.7 |
| ν2 | A1g (Ni) | 544 | 27 | 4840.6 |
| ν3 | Eg (Co) | 514 | 34 | 3556.9 |
| ν4 | A1g (Co) | 601 | 44 | 23,604.8 |
| ν5 | Eg (Mn) | 571 | 39 | 6241.5 |
| ν6 | A1g (Mn) | 638 | 49 | 12,607.1 |
| Element | Core Level | Binding Energy (eV) | |
|---|---|---|---|
| NMC333 | Na/F-NMC333 | ||
| Ni 2p | 2p3/2 | 845.4 | 845.4 |
| 2p1/2 | 871.8 | 871.6 | |
| Satellite | 861.6 | 876.7 | |
| ΔBE | 17.4 | 17.2 | |
| Co 2p | 2p3/2 | 780.0 | 780.0 |
| 2p1/2 | 795.0 | 795.0 | |
| Satellite | 789.4 | 790.0 | |
| ΔBE | 15.0 | 15.0 | |
| Mn 2p | 2p3/2 | 742.8 | 742.8 |
| 2p1/2 | 754.4 | 754.4 | |
| ΔBE | 11.6 | 11.6 | |
| O 1s | 1s | 529.6 | 529.3 |
| 531.3 | 531.0 | ||
| F 1s | 1s | - | 685.4 |
| Na 1s | 1s | - | 1071.8 |
| Dopant | Specific Capacity (mAh g−1) | Cycles | Decay per Cycle (%) | Ref. |
|---|---|---|---|---|
| 0.05 Na | 120 @ 1C | 10 | 2.51 | [15] |
| 0.03 Na | 133 @ 0.5C | 50 | 0.09 | [16] |
| 0.05 Na | 177 @ 0.1C | 30 | 0.42 | [17] |
| 0.1 Na | 95 @ 0.2C | 100 | 0.37 | [18] |
| 0.08 F | 136 @ 1.2 mA cm−2 | 50 | 0.21 | [20] |
| 0.04 F | 172 @ 0.2C | 30 | 0.15 | [21] |
| 0.04 Al + 0.05 F | 148 @ 0.1C | 20 | 0.3 | [22] |
| 0.02 Al + 0.02 B + 0.02 F | 157 @ 1C | 30 | 0.02 | [23] |
| 0.025 Na + 0.025 F | 70 @ 1C | 1000 | 0.05 | this work |
| Electrode | Specific Capacity (mAh g−1) | Coulombic Efficiency 200th Cycle (%) | |||
|---|---|---|---|---|---|
| 1st Cycle | 500th Cycle | Capacity Decay | 1000th Cycle | ||
| NMC333 | 136 | 17 | 0.24 | 2.5 | 99.4 |
| Na/F-NMC333 | 142 | 86 | 0.11 | 70 | 99.5 |
| Parameters | NMC333 | Na/F-NMC333 | ||
|---|---|---|---|---|
| Fresh Cell | After Cycling | Fresh Cell | After Cycling | |
| Rs (Ω) | 45.4 | 18.1 | 29.4 | 51.7 |
| RSEI (Ω) | - | 164.7 | - | 95 |
| CPESEI-T | - | 1.2 × 10−5 | - | 1.4 × 10−5 |
| CPESEI-P | - | 0.82 | - | 0.81 |
| Rct (Ω) | 61.6 | 321.3 | 62.8 | 242.5 |
| CPEdl-T | 1.5 × 10−5 | 3 × 10−4 | 6.8 × 10−5 | 8.9 × 10−5 |
| CPEdl-P | 0.85 | 0.65 | 0.74 | 0.73 |
| Zw-R | 51.1 | 2073 | 46 | 950.6 |
| Zw-T | 0.097 | 31.6 | 0.123 | 12.8 |
| Zw-P | 0.58 | 0.56 | 0.78 | 0.57 |
| DLi+ (cm2 s−1) | 1.3 × 10−11 | 8.3 × 10−15 | 1.7 × 10−11 | 4.0 × 10−14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Hashem, A.M.; Abdel-Ghany, A.E.; Abbas, S.M.; El-Tawil, R.S.; Li, T.; Li, X.; El-Mounayri, H.; Tovar, A.; Zhu, L.; et al. Effect of Cationic (Na+) and Anionic (F−) Co-Doping on the Structural and Electrochemical Properties of LiNi1/3Mn1/3Co1/3O2 Cathode Material for Lithium-Ion Batteries. Int. J. Mol. Sci. 2022, 23, 6755. https://doi.org/10.3390/ijms23126755
Wang H, Hashem AM, Abdel-Ghany AE, Abbas SM, El-Tawil RS, Li T, Li X, El-Mounayri H, Tovar A, Zhu L, et al. Effect of Cationic (Na+) and Anionic (F−) Co-Doping on the Structural and Electrochemical Properties of LiNi1/3Mn1/3Co1/3O2 Cathode Material for Lithium-Ion Batteries. International Journal of Molecular Sciences. 2022; 23(12):6755. https://doi.org/10.3390/ijms23126755
Chicago/Turabian StyleWang, Hua, Ahmed M. Hashem, Ashraf E. Abdel-Ghany, Somia M. Abbas, Rasha S. El-Tawil, Tianyi Li, Xintong Li, Hazim El-Mounayri, Andres Tovar, Likun Zhu, and et al. 2022. "Effect of Cationic (Na+) and Anionic (F−) Co-Doping on the Structural and Electrochemical Properties of LiNi1/3Mn1/3Co1/3O2 Cathode Material for Lithium-Ion Batteries" International Journal of Molecular Sciences 23, no. 12: 6755. https://doi.org/10.3390/ijms23126755
APA StyleWang, H., Hashem, A. M., Abdel-Ghany, A. E., Abbas, S. M., El-Tawil, R. S., Li, T., Li, X., El-Mounayri, H., Tovar, A., Zhu, L., Mauger, A., & Julien, C. M. (2022). Effect of Cationic (Na+) and Anionic (F−) Co-Doping on the Structural and Electrochemical Properties of LiNi1/3Mn1/3Co1/3O2 Cathode Material for Lithium-Ion Batteries. International Journal of Molecular Sciences, 23(12), 6755. https://doi.org/10.3390/ijms23126755












