MicroRNA Methylome Signature and Their Functional Roles in Colorectal Cancer Diagnosis, Prognosis, and Chemoresistance
Abstract
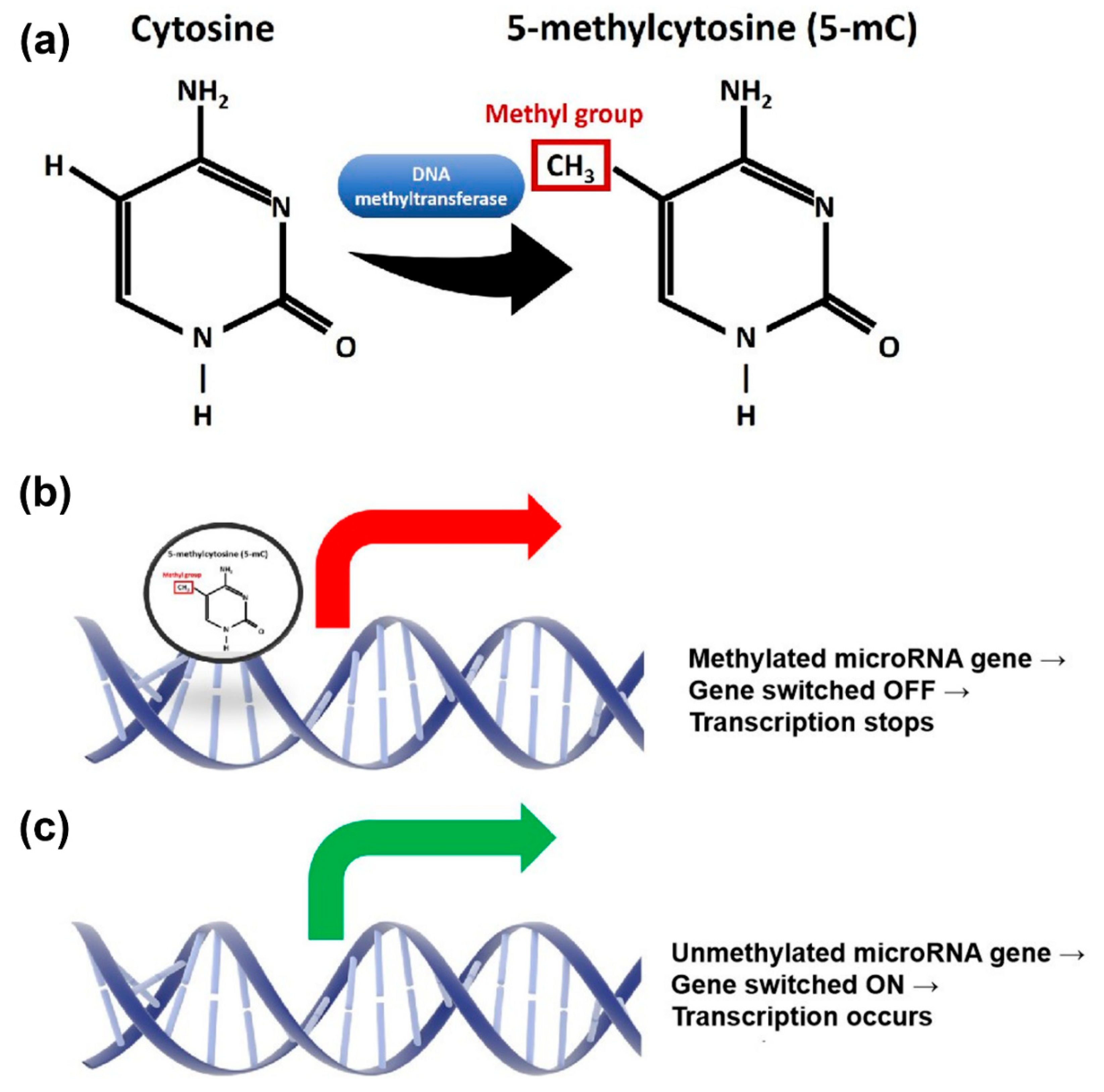
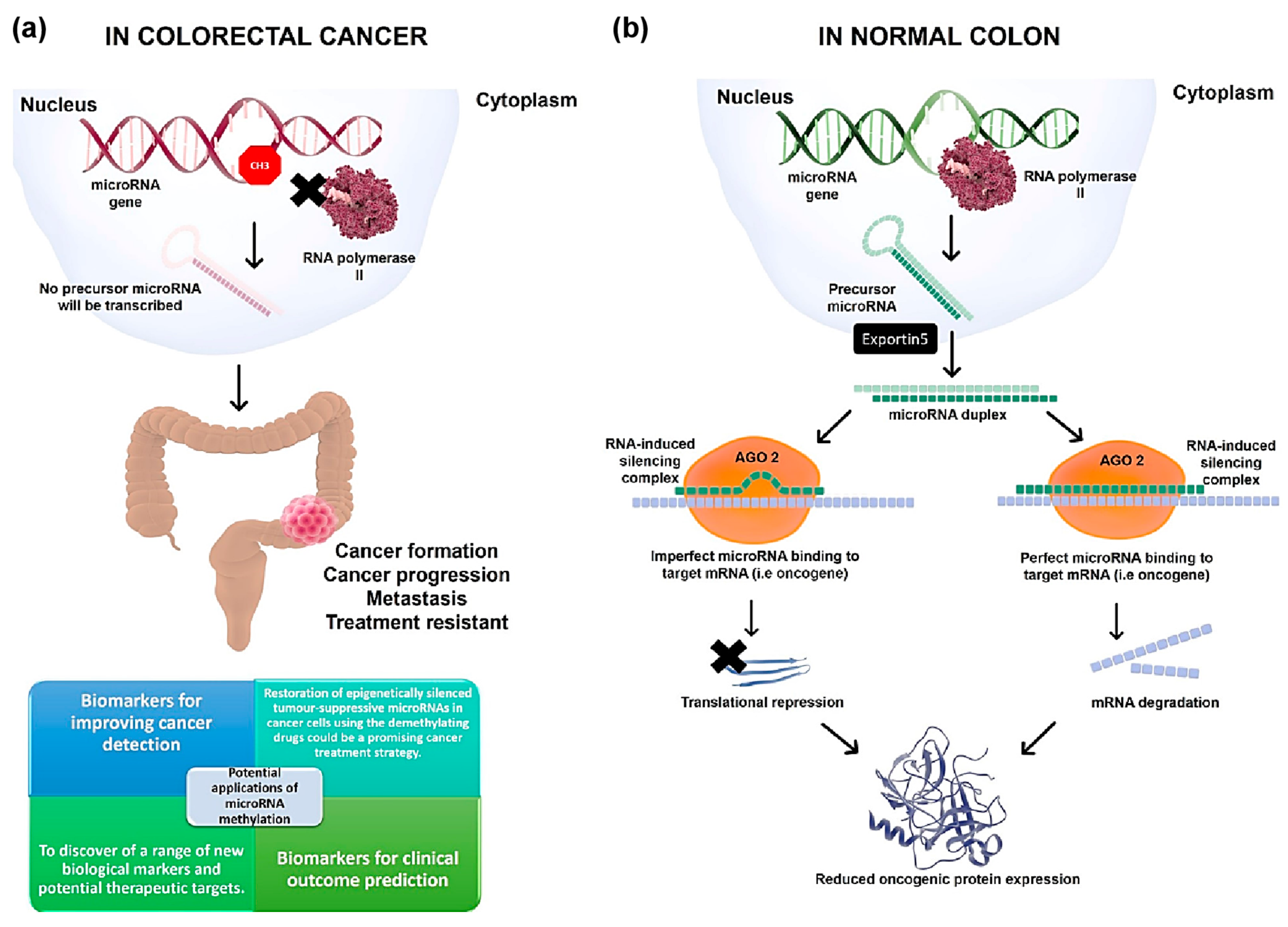
:1. Introduction
2. MicroRNA Methylation with Diagnostic and Prognostic Markers
2.1. MiRNA-124a
2.2. MiRNA-137
2.3. MiRNA-34
2.4. Other microRNA Genes
| MicroRNA(s) and Reference | MicroRNA Methylation Detection Method | Types of Specimens | Key Findings |
|---|---|---|---|
| miR-124a [17] Known targets: STAT3, IASPP PRRX1, KITENIN, PRPS1, RPIA PTB1/PKM1/PKM2, DNMT3B, DNMT1, ROCK1, PRRX1, PLCB1 | Methylation-specific PCR (MSP) and bisulfite sequencing | Cell line model with disrupted DNA methyltransferase |
|
| miR-34b/c [19] Known targets: SATB2 | Methylation-specific PCR (MSP) and bisulfite sequencing | CRC cell lines |
|
| miR-133b [20] Known targets: CXCR4, HOXA9 | Methylation-specific PCR (MSP) and combined bisulfite restriction analysis (COBRA) | Screening using CRC cell lines and validation in the tissues (6 CRCs, 2 adjacent non-tumors, and 2 healthy colorectal tissues) |
|
| miR-324 [21] Known targets: ELAVL1 | Methylation-specific PCR (MSP) and bisulfite sequencing | 42 CRCs, 9 colorectal adenomas, and 16 normal mucosae in patients with and without CRC |
|
| miR-137, miR- 342 [36] Known targets: miR-137: TCF4, FMNL2, Aurora-A miR-342: DNMT1, FOXM1, FOXQ1 | Methylation-specific PCR (MSP) | Fresh-frozen tissues (51 polyps, 8 tumors, and 14 normal mucosa) |
|
| miR-9, miR-129, miR-137 [52] Known targets: miR-9: TM4SF1, FOXP2, ANO1 miR-129: MALAT1 miR-137: TCF4, FMNL2, Aurora-A | Methylation-specific PCR (MSP) and bisulfite sequencing | CRC cell lines and 50 primary CRCs with adjacent normal tissues |
|
| miR-345 [54] No known target | Methylation-specific PCR (MSP) and bisulfite sequencing | CRC cell lines and 31 CRC patients |
|
| miR-129-2, miR-345, miR-132 [56] Known targets: miR-129: MALAT1 miR-345: No known target miR-132: ZEB2, ERK1 | Bisulfite sequencing and Methylation-Specific Multiplex Ligation-Dependent Probe Amplification (MS-MLPA) | CRC cell lines treated with 5-aza-2′deoxycytidine followed by validation in 205 CRCs |
|
| miR-132 [57] Known targets: miR-132: ZEB2, ERK1 | Methylation-specific PCR (MSP) and bisulfite sequencing | CRC cell lines and 36 CRCs with adjacent normal tissues |
|
| miR-1, miR-9, miR-124, miR-137 [29] Known targets: miR-1: SMAD3 miR-9: TM4SF1, FOXP2, ANO1 miR-124: STAT3, IASPP PRRX1, KITENIN, PRPS1, RPIA. PTB1/PKM1/PKM2, DNMT3B, DNMT1, ROCK1, PRRX1, PLCB1. miR-137: TCF4, FMNL2, Aurora-A | Quantitative bisulfite pyrosequencing | 387 colorectal epithelial specimens (362 non-neoplastic and 25 neoplastic tissues) |
|
| miR-125 [58] Known targets: BCL2, BCL2L12, MCL1, SMURF1, VEGFA, TAZ, CXCL12/CXCR4 | Bisulfite sequencing PCR | CRC tissues and adjacent normal tissues from 68 CRC patients |
|
| miR-941 [59] No known target | Bisulfite sequencing | CRC cell lines |
|
| miR-1237 [59] No known target | Bisulfite sequencing | CRC cell lines |
|
| miR-1247 [60] No known target | Methylation-specific PCR (MSP) and bisulfite sequencing | CRC cell lines and patients (hypermethylated and non-methylated CRCs) |
|
| miR-128 [61] Known targets: IRS1, Galectin-3 | Bisulfite sequencing PCR | CRC cell lines and patients |
|
| miR-148a [62] Known targets: BCL2, ERBB3 | Bisulfite pyrosequencing | 273 CRC patients (76 stage II, 125 stage III, 72 stage IV) |
|
| miR-126 [63] Known targets: CXCR4 | Methylation-specific PCR (MSP) and bisulfite sequencing | CRC cell lines and patients |
|
| miR-27b [64] Known targets: RAB3D | Methylation-specific PCR (MSP) | CRC cell lines |
|
| miR-149 [65] Known targets: FOXM1, EPHB3 | Methylation-specific PCR (MSP) | CRC cell lines |
|
| miR-497/195 [66] Known targets: IGF1R, NRDP1, KSR1, FRA-1, PTPN3, CARMA3, FGF2 | Combined bisulfite restriction analysis (COBRA) and bisulfite genomic sequencing (BGS) | CRC cell lines and patients |
|
| miR-212 [67] Known targets: MnSOD | Methylation-specific PCR (MSP) and bisulfite sequencing | CRC cell lines and tissues |
|
| miR-200c/141 [68] Known targets: ZEB1, DLC1, TRAF5 | Methylation-specific PCR (MSP) | CRC tissues |
|
| miR-373 [69] No known targets | Methylation-specific PCR (MSP) and bisulfite sequencing | CRC cell lines and 40 CRC patients |
|
3. MicroRNA Methylation in CRC Chemoresistance
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ezat, S.W.; Natrah, M.S.; Aljunid, S.; Rizal, M.A.; Saperi, S.; Ismail, S.; Fuad, I.; Azrif, M.A. Economic evaluation of monoclonal antibody in the management of colorectal cancer. J. Cancer Res. Ther. 2013, 1, 34–39. [Google Scholar]
- Azzani, M.; Dahlui, M.; Ishak, W.Z.W.; Roslani, A.C.; Su, T.T. Provider costs of treating colorectal cancer in government hospital of Malaysia. Malays J. Med. Sci. 2019, 26, 73–86. [Google Scholar] [CrossRef] [PubMed]
- Azizah, A.M.; Nor Saleha, I.T.; Noor Hashimah, A.; Asmah, Z.A.; Mastulu, W. Malaysian National Cancer Registry Report 2007–2011; National Cancer Institute: Putrajaya, Malaysia, 2016. [Google Scholar]
- The Cancer Genome Atlas (TCGA) Research Network. Comprehensive molecular characterization of human colon and rectal cancer. Nature 2012, 487, 330–337. [Google Scholar] [CrossRef] [Green Version]
- Naumov, V.A.; Generozov, E.V.; Zaharjevskaya, N.B.; Matushkina, D.S.; Larin, A.K.; Chernyshov, S.V.; Alekseev, M.V.; Shelygin, Y.A.; Govorun, V.M. Genome-scale analysis of DNA methylation in colorectal cancer using infinium humanmethylation450 beadchips. Epigenetics 2013, 8, 921–934. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baharudin, R.; Ab Mutalib, N.-S.; Othman, S.N.; Sagap, I.; Rose, I.M.; Mohd Mokhtar, N.; Jamal, R. Identification of predictive DNA methylation biomarkers for chemotherapy response in colorectal cancer. Front. Pharmacol. 2017, 8, 47. [Google Scholar] [CrossRef] [Green Version]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Wang, J. MicroRNAs are important regulators of drug resistance in colorectal cancer. Biol. Chem. 2017, 398, 929–938. [Google Scholar] [CrossRef] [Green Version]
- Al-Akhrass, H.; Christou, N. The clinical assessment of microRNA diagnostic, prognostic, and theranostic value in colorectal cancer. Cancers 2021, 13, 2916. [Google Scholar] [CrossRef]
- Imedio, L.; Cristóbal, I.; Rubio, J.; Santos, A.; Rojo, F.; García-Foncillas, J. MicroRNAs in rectal cancer: Functional significance and promising therapeutic value. Cancers 2020, 12, 2040. [Google Scholar] [CrossRef]
- Pidíková, P.; Herichová, I. MiRNA clusters with up-regulated expression in colorectal cancer. Cancers 2021, 13, 2979. [Google Scholar] [CrossRef]
- Svoronos, A.A.; Engelman, D.M.; Slack, F.J. OncomiR or tumor suppressor? The duplicity of MicroRNAs in cancer. Cancer Res. 2016, 76, 3666–3670. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaur, S.; Lotsari-Salomaa, J.E.; Seppänen-Kaijansinkko, R.; Peltomäki, P. MicroRNA methylation in colorectal cancer. In Non-Coding RNAs in Colorectal Cancer; Slaby, O., Calin, G.A., Eds.; Springer International Publishing: Cham, Switzerland, 2016; Volume 937, pp. 109–122. ISBN 978-3-319-42057-8. [Google Scholar]
- Wang, S.; Wu, W.; Claret, F.X. Mutual regulation of micrornas and DNA methylation in human cancers. Epigenetics 2017, 12, 187–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stark, V.A.; Facey, C.O.B.; Viswanathan, V.; Boman, B.M. The role of MiRNAs, MiRNA clusters, and isomirs in development of cancer stem cell populations in colorectal cancer. Int. J. Mol. Sci. 2021, 22, 1424. [Google Scholar] [CrossRef] [PubMed]
- Lujambio, A.; Calin, G.A.; Villanueva, A.; Ropero, S.; Sanchez-Cespedes, M.; Blanco, D.; Montuenga, L.M.; Rossi, S.; Nicoloso, M.S.; Faller, W.J.; et al. A MicroRNA DNA methylation signature for human cancer metastasis. Proc. Natl. Acad. Sci. USA 2008, 105, 13556–13561. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lujambio, A.; Ropero, S.; Ballestar, E.; Fraga, M.F.; Cerrato, C.; Setién, F.; Casado, S.; Suarez-Gauthier, A.; Sanchez-Cespedes, M.; Git, A.; et al. Genetic unmasking of an epigenetically silenced microrna in human cancer cells. Cancer Res. 2007, 67, 1424–1429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patil, N.; Abba, M.L.; Zhou, C.; Chang, S.; Gaiser, T.; Leupold, J.H.; Allgayer, H. Changes in methylation across structural and MicroRNA genes relevant for progression and metastasis in colorectal cancer. Cancers 2021, 13, 5951. [Google Scholar] [CrossRef]
- Toyota, M.; Suzuki, H.; Sasaki, Y.; Maruyama, R.; Imai, K.; Shinomura, Y.; Tokino, T. Epigenetic silencing of MicroRNA-34b/c and B-cell translocation gene 4 is associated with cpg island methylation in colorectal cancer. Cancer Res. 2008, 68, 4123–4132. [Google Scholar] [CrossRef] [Green Version]
- Lv, L.V.; Zhou, J.; Lin, C.; Hu, G.; Yi, L.U.; Du, J.; Gao, K.; Li, X. DNA methylation is involved in the aberrant expression of mir-133b in colorectal cancer cells. Oncol. Lett. 2015, 10, 907–912. [Google Scholar] [CrossRef] [Green Version]
- Grady, W.M.; Parkin, R.K.; Mitchell, P.S.; Lee, J.H.; Kim, Y.-H.; Tsuchiya, K.D.; Washington, M.K.; Paraskeva, C.; Willson, J.K.V.; Kaz, A.M.; et al. Epigenetic silencing of the intronic MicroRNA Hsa-MiR-342 and Its host gene EVL in colorectal cancer. Oncogene 2008, 27, 3880–3888. [Google Scholar] [CrossRef] [Green Version]
- Oltra, S.S.; Peña-Chilet, M.; Vidal-Tomas, V.; Flower, K.; Martinez, M.T.; Alonso, E.; Burgues, O.; Lluch, A.; Flanagan, J.M.; Ribas, G. Methylation deregulation of MiRNA promoters identifies MiR124-2 as a survival biomarker in breast cancer in very young women. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, K.; Huang, W.; Weng, W.; Zhang, Z.; Guo, Y.; Deng, T.; Xiang, Y.; Ni, X.; Chen, B.; et al. The DNA methylation profile of non-coding RNAs improves prognosis prediction for pancreatic adenocarcinoma. Cancer Cell Int. 2019, 19, 107. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.; Chatterjee, A.; Das, D.; Ray, A.; Singh, R.; Chattopadhyay, E.; Sarkar, N.D.; Eccles, M.; Pal, M.; Maitra, A.; et al. Genome-wide MiRNA methylome analysis in oral cancer: Possible biomarkers associated with patient survival. Epigenomics 2019, 11, 473–487. [Google Scholar] [CrossRef] [PubMed]
- Lujambio, A.; Esteller, M. CpG island hypermethylation of tumor suppressor MicroRNAs in human cancer. Cell Cycle 2007, 6, 1455–1459. [Google Scholar] [CrossRef]
- Hibner, G.; Kimsa-Furdzik, M.; Francuz, T. Relevance of MicroRNAs as potential diagnostic and prognostic markers in colorectal cancer. Int. J. Mol. Sci. 2018, 19, 2944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harada, T.; Yamamoto, E.; Yamano, H.; Nojima, M.; Maruyama, R.; Kumegawa, K.; Ashida, M.; Yoshikawa, K.; Kimura, T.; Harada, E.; et al. Analysis of DNA methylation in bowel lavage fluid for detection of colorectal cancer. Cancer Prev. Res. 2014, 7, 1002–1010. [Google Scholar] [CrossRef] [Green Version]
- Ueda, Y.; Ando, T.; Nanjo, S.; Ushijima, T.; Sugiyama, T. DNA methylation of MicroRNA-124a is a potential risk marker of colitis-associated cancer in patients with ulcerative colitis. Dig. Dis. Sci. 2014, 59, 2444–2451. [Google Scholar] [CrossRef]
- Toiyama, Y.; Okugawa, Y.; Tanaka, K.; Araki, T.; Uchida, K.; Hishida, A.; Uchino, M.; Ikeuchi, H.; Hirota, S.; Kusunoki, M.; et al. A panel of methylated MicroRNA biomarkers for identifying high-risk patients with ulcerative colitis-associated colorectal cancer. Gastroenterology 2017, 153, 1634.e8–1646.e8. [Google Scholar] [CrossRef] [Green Version]
- Wang, M.-J.; Li, Y.; Wang, R.; Wang, C.; Yu, Y.-Y.; Yang, L. Downregulation of MicroRNA-124 is an independent prognostic factor in patients with colorectal cancer. Int. J. Colorectal. Dis. 2013, 28, 183–189. [Google Scholar] [CrossRef] [Green Version]
- Jinushi, T.; Shibayama, Y.; Kinoshita, I.; Oizumi, S.; Jinushi, M.; Aota, T.; Takahashi, T.; Horita, S.; Dosaka-Akita, H.; Iseki, K. Low expression levels of MicroRNA-124-5p correlated with poor prognosis in colorectal cancer via targeting of SMC4. Cancer Med. 2014, 3, 1544–1552. [Google Scholar] [CrossRef]
- Zhou, L.; Xu, Z.; Ren, X.; Chen, K.; Xin, S. MicroRNA-124 (MiR-124) inhibits cell proliferation, metastasis and invasion in colorectal cancer by downregulating rho-associated protein kinase 1(ROCK1). CPB 2016, 38, 1785–1795. [Google Scholar] [CrossRef]
- Mahmoudi, E.; Cairns, M.J. MiR-137: An important player in neural development and neoplastic transformation. Mol. Psychiatry 2017, 22, 44–55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, Y.-C.; Lee, C.-T.; Lee, J.-C.; Liu, Y.-W.; Chen, Y.-J.; Tseng, J.T.; Kang, J.-W.; Sheu, B.-S.; Lin, B.-W.; Hung, L.-Y. Epigenetic silencing of MiR-137 contributes to early colorectal carcinogenesis by impaired aurora—A inhibition. Oncotarget 2016, 7, 76852–76866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balaguer, F.; Link, A.; Lozano, J.J.; Cuatrecasas, M.; Nagasaka, T.; Boland, C.R.; Goel, A. Epigenetic silencing of MiR-137 is an early event in colorectal carcinogenesis. Cancer Res. 2010, 70, 6609–6618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kashani, E.; Hadizadeh, M.; Chaleshi, V.; Mirfakhraie, R.; Young, C.; Savabkar, S. The differential DNA hypermethylation patterns of MicroRNA-137 and MicroRNA-342 locus in early colorectal lesions and tumours. Biomolecules 2019, 9, 519. [Google Scholar] [CrossRef] [Green Version]
- Bahnassy, A.A.; El-Sayed, M.; Ali, N.M.; Khorshid, O.; Hussein, M.M.; Yousef, H.F.; Mohanad, M.A.; Zekri, A.-R.N.; Salem, S.E. Aberrant expression of MiRNAs predicts recurrence and survival in stage-II colorectal cancer patients from Egypt. Appl. Cancer Res. 2017, 37, 39. [Google Scholar] [CrossRef] [Green Version]
- Sakaguchi, M.; Hisamori, S.; Oshima, N.; Sato, F.; Shimono, Y.; Sakai, Y. MiR-137 regulates the tumorigenicity of colon cancer stem cells through the inhibition of DCLK1. Mol. Cancer Res. 2016, 14, 354–362. [Google Scholar] [CrossRef] [Green Version]
- Chen, T.; Cai, S.-L.; Li, J.; Qi, Z.-P.; Li, X.-Q.; Ye, L.-C.; Xie, X.-F.; Hou, Y.-Y.; Yao, L.-Q.; Xu, M.-D.; et al. Mecp2-Mediated epigenetic silencing of MiR-137 contributes to colorectal adenoma-carcinoma sequence and tumor progression via relieving the suppression of c-met. Sci. Rep. 2017, 7, 44543. [Google Scholar] [CrossRef]
- Navarro, F.; Lieberman, J. MiR-34 and P53: New insights into a complex functional relationship. PLoS ONE 2015, 10, e0132767. [Google Scholar] [CrossRef]
- Zhang, L.; Liao, Y.; Tang, L. MicroRNA-34 family: A potential tumor suppressor and therapeutic candidate in cancer. J. Exp. Clin. Cancer Res. 2019, 38, 53. [Google Scholar] [CrossRef] [Green Version]
- Wu, X.; Song, Y.-C.; Cao, P.-L.; Zhang, H.; Guo, Q.; Yan, R.; Diao, D.-M.; Cheng, Y.; Dang, C.-X. Detection of MiR-34a and MiR-34b/c in stool sample as potential screening biomarkers for noninvasive diagnosis of colorectal cancer. Med. Oncol. 2014, 31, 894. [Google Scholar] [CrossRef]
- Kalimutho, M.; Di Cecilia, S.; Del Vecchio Blanco, G.; Roviello, F.; Sileri, P.; Cretella, M.; Formosa, A.; Corso, G.; Marrelli, D.; Pallone, F.; et al. Epigenetically silenced MiR-34b/c as a novel faecal-based screening marker for colorectal cancer. Br. J. Cancer 2011, 104, 1770–1778. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, J.; Li, N.; Dong, Y.; Li, S.; Xu, L.; Li, X.; Li, Y.; Li, Z.; Ng, S.S.; Sung, J.J.; et al. MiR-34a-5p suppresses colorectal cancer metastasis and predicts recurrence in patients with stage II/III colorectal cancer. Oncogene 2015, 34, 4142–4152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siemens, H.; Neumann, J.; Jackstadt, R.; Mansmann, U.; Horst, D.; Kirchner, T.; Hermeking, H. Detection of MiR-34a promoter methylation in combination with elevated expression of c-met and β-catenin predicts distant metastasis of colon cancer. Clin. Cancer Res. 2013, 19, 710–720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rapti, S.-M.; Kontos, C.K.; Christodoulou, S.; Papadopoulos, I.N.; Scorilas, A. MiR-34a overexpression predicts poor prognostic outcome in colorectal adenocarcinoma, independently of clinicopathological factors with established prognostic value. Clin. Biochem. 2017, 50, 918–924. [Google Scholar] [CrossRef]
- Hasakova, K.; Reis, R.; Vician, M.; Zeman, M.; Herichova, I. Expression of MiR-34a-5p is up-regulated in human colorectal cancer and correlates with survival and clock gene PER2 expression. PLoS ONE 2019, 14, e0224396. [Google Scholar] [CrossRef] [Green Version]
- Li, D.; Xia, L.; Chen, M.; Lin, C.; Wu, H.; Zhang, Y.; Pan, S.; Li, X. MiR-133b, a particular member of myomirs, coming into playing its unique pathological role in human cancer. Oncotarget 2017, 8, 50193–50208. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Bu, J.; Liu, X.; Wang, W.; Mai, W.; Lv, B.; Zou, J.; Mo, X.; Li, X.; Wang, J.; et al. MiR-133b suppresses metastasis by targeting HOXA9 in human colorectal cancer. Oncotarget 2017, 8, 63935–63948. [Google Scholar] [CrossRef] [Green Version]
- Datta, J.; Kutay, H.; Nasser, M.W.; Nuovo, G.J.; Wang, B.; Majumder, S.; Liu, C.-G.; Volinia, S.; Croce, C.M.; Schmittgen, T.D.; et al. Methylation mediated silencing of MicroRNA-1 gene and its role in hepatocellular carcinogenesis. Cancer Res. 2008, 68, 5049–5058. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.-S.; Leung, C.-M.; Pan, H.-W.; Hu, L.-Y.; Li, S.-C.; Ho, M.-R.; Tsai, K.-W. Silencing of MiR-1-1 and MiR-133a-2 cluster expression by DNA hypermethylation in colorectal cancer. Oncol. Rep. 2012, 28, 1069–1076. [Google Scholar] [CrossRef] [Green Version]
- Bandres, E.; Agirre, X.; Bitarte, N.; Ramirez, N.; Zarate, R.; Roman-Gomez, J. Epigenetic regulation of MicroRNA expression in colorectal cancer. Int. J. Cancer 2009, 125, 2737–2743. [Google Scholar] [CrossRef]
- Cekaite, L.; Rantala, J.K.; Bruun, J.; Guriby, M.; Ågesen, T.H.; Danielsen, S.A.; Lind, G.E.; Nesbakken, A.; Kallioniemi, O.; Lothe, R.A.; et al. MiR-9, -31, and -182 deregulation promote proliferation and tumor cell survival in colon cancer. Neoplasia 2012, 14, 868–881. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, J.-T.; Wang, J.-L.; Du, W.; Hong, J.; Zhao, S.-L.; Wang, Y.-C.; Xiong, H.; Chen, H.-M.; Fang, J.-Y. MicroRNA 345, a methylation-sensitive MicroRNA is involved in cell proliferation and invasion in human colorectal cancer. Carcinogenesis 2011, 32, 1207–1215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Wu, J.; Meng, X.; Ying, X.; Zuo, Y.; Liu, R.; Pan, Z.; Kang, T.; Huang, W. MicroRNA-342 inhibits colorectal cancer cell proliferation and invasion by directly targeting DNA methyltransferase 1. Carcinogenesis 2011, 32, 1033–1042. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaur, S.; Lotsari, J.E.; Al-Sohaily, S.; Warusavitarne, J.; Kohonen-Corish, M.R.; Peltomäki, P. Identification of subgroup-specific MiRNA patterns by epigenetic profiling of sporadic and lynch syndrome-associated colorectal and endometrial carcinoma. Clin. Epigenetics 2015, 7, 20. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Qin, J.; Ke, J.; Wang, F.; Zhou, Y.; Jiang, Y.; Xu, J. Downregulation of MicroRNA-132 by DNA hypermethylation is associated with cell invasion in colorectal cancer. OncoTargets Ther. 2015, 3639. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Xu, Z. Hypermethylation-associated silencing of MiR-125a and MiR-125b: A potential marker in colorectal cancer. Dis. Markers 2015, 2015, 345080. [Google Scholar] [CrossRef] [Green Version]
- Yan, H.; Choi, A.; Lee, B.H.; Ting, A.H. Identification and functional analysis of epigenetically silenced microRNAs in colorectal cancer cells. PLoS ONE 2011, 6, e20628. [Google Scholar] [CrossRef] [Green Version]
- Liang, J.; Zhou, W.; Sakre, N.; DeVecchio, J.; Ferrandon, S.; Ting, A.H.; Bao, S.; Bissett, I.; Church, J.; Kalady, M.F. Epigenetically regulated MiR-1247 functions as a novel tumour suppressor via MYCBP2 in methylator colon cancers. Br. J. Cancer 2018, 119, 1267–1277. [Google Scholar] [CrossRef]
- Takahashi, Y.; Iwaya, T.; Sawada, G.; Kurashige, J.; Matsumura, T.; Uchi, R.; Ueo, H.; Takano, Y.; Eguchi, H.; Sudo, T.; et al. Up-regulation of NEK2 by MicroRNA-128 methylation is associated with poor prognosis in colorectal cancer. Ann. Surg. Oncol. 2014, 21, 205–212. [Google Scholar] [CrossRef]
- Takahashi, M.; Cuatrecasas, M.; Balaguer, F.; Hur, K.; Toiyama, Y.; Castells, A.; Boland, C.R.; Goel, A. The clinical significance of MiR-148a as a predictive biomarker in patients with advanced colorectal cancer. PLoS ONE 2012, 7, e46684. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.; Xu, B.; Wang, B.; Wang, Z.; Liang, Y.; Zhou, J.; Hu, J.; Jiang, B. Epigenetic silencing of MiR-126 contributes to tumor invasion and angiogenesis in colorectal cancer. Oncol. Rep. 2013, 30, 1976–1984. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ye, J.; Wu, X.; Wu, D.; Wu, P.; Ni, C.; Zhang, Z.; Chen, Z.; Qiu, F.; Xu, J.; Huang, J. MiRNA-27b targets vascular endothelial growth factor C to inhibit tumor progression and angiogenesis in colorectal cancer. PLoS ONE 2013, 8, e60687. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shan, S.; Lu, Y.; Zhang, X.; Shi, J.; Li, H.; Li, Z. Inhibitory effect of bound polyphenol from foxtail millet bran on MiR-149 methylation increases the chemosensitivity of human colorectal cancer HCT-8/Fu cells. Mol. Cell Biochem. 2021, 476, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Menigatti, M.; Staiano, T.; Manser, C.; Bauerfeind, P.; Komljenovic, A.; Robinson, M.; Jiricny, J.; Buffoli, F.; Marra, G. Epigenetic silencing of monoallelically methylated MiRNA loci in precancerous colorectal lesions. Oncogenesis 2013, 2, e56. [Google Scholar] [CrossRef] [Green Version]
- Meng, X.; Wu, J.; Pan, C.; Wang, H.; Ying, X.; Zhou, Y.; Yu, H.; Zuo, Y.; Pan, Z.; Liu, R.-Y.; et al. Genetic and epigenetic down-regulation of MicroRNA-212 promotes colorectal tumor metastasis via dysregulation of MnSOD. Gastroenterology 2013, 145, 426.e1–6–436.e1–6. [Google Scholar] [CrossRef]
- Taheri, Z.; Asadzadeh Aghdaei, H.; Irani, S.; Modarressi, M.H.; Noormohammadi, Z. Clinical correlation of MiR-200c/141 cluster DNA methylation and MiR-141 expression with the clinicopathological features of colorectal primary lesions/tumors. Rep. Biochem. Mol. Biol. 2019, 8, 208–215. [Google Scholar]
- Tanaka, T.; Arai, M.; Wu, S.; Kanda, T.; Miyauchi, H.; Imazeki, F.; Matsubara, H.; Yokosuka, O. Epigenetic silencing of MicroRNA-373 plays an important role in regulating cell proliferation in colon cancer. Oncol. Rep. 2011, 26, 1329–1335. [Google Scholar] [CrossRef] [Green Version]
- Xie, B.; Ding, Q.; Han, H.; Wu, D. MiRCancer: A microRNA–Cancer association database constructed by text mining on literature. Bioinformatics 2013, 29, 638–644. [Google Scholar] [CrossRef]
- Glaich, O.; Parikh, S.; Bell, R.E.; Mekahel, K.; Donyo, M.; Leader, Y.; Shayevitch, R.; Sheinboim, D.; Yannai, S.; Hollander, D.; et al. DNA methylation directs MicroRNA biogenesis in mammalian cells. Nat. Commun. 2019, 10, 5657. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, H.; Maruyama, R.; Yamamoto, E.; Kai, M. DNA methylation and microRNA dysregulation in cancer. Mol. Oncol. 2012, 6, 567–578. [Google Scholar] [CrossRef] [Green Version]
- Kok-Sin, T.; Mohktar, N.M.; Hassan, N.Z.A.; Sagap, I.; Rose, I.M.; Harun, R.; Jamal, R. Identification of diagnostic markers in colorectal cancer via integrative epigenomics and genomics data. Oncol. Rep. 2015, 34, 22–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garofalo, M.; Croce, C.M. MicroRNAs: Master regulators as potential therapeutics in cancer. Annu. Rev. Pharmacol. Toxicol. 2011, 51, 25–43. [Google Scholar] [CrossRef] [PubMed]
- André, T.; Boni, C.; Mounedji-Boudiaf, L.; Navarro, M.; Tabernero, J.; Hickish, T.; Topham, C.; Zaninelli, M.; Clingan, P.; Bridgewater, J.; et al. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N. Engl. J. Med. 2004, 350, 2343–2351. [Google Scholar] [CrossRef] [Green Version]
- Hon, K.W.; Abu, N.; Ab Mutalib, N.-S.; Jamal, R. MiRNAs and LncRNAs as predictive biomarkers of response to FOLFOX therapy in colorectal cancer. Front. Pharmacol. 2018, 9, 846. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Lu, F.-Y.; Shi, R.-H.; Feng, Y.-D.; Zhao, X.-D.; Lu, Z.-P.; Xiao, L.; Zhou, G.-Q.; Qiu, J.-M.; Cheng, C.-E. MiR-26b regulates 5-FU-resistance in human colorectal cancer via down-regulation of pgp. Am. J. Cancer Res. 2018, 8, 2518–2527. [Google Scholar]
- Shi, L.; Li, X.; Wu, Z.; Li, X.; Nie, J.; Guo, M.; Mei, Q.; Han, W. DNA methylation-mediated repression of MiR-181a/135a/302c expression promotes the microsatellite-unstable colorectal cancer development and 5-FU resistance via targeting PLAG1. J. Genet. Genom. 2018, 45, 205–214. [Google Scholar] [CrossRef]
- Wang, F.; Ma, Y.-L.; Zhang, P.; Shen, T.-Y.; Shi, C.-Z.; Yang, Y.-Z.; Moyer, M.-P.; Zhang, H.-Z.; Chen, H.-Q.; Liang, Y.; et al. SP1 mediates the link between methylation of the tumour suppressor MiR-149 and outcome in colorectal cancer. J. Pathol. 2013, 229, 12–24. [Google Scholar] [CrossRef]
- Liu, X.; Xie, T.; Mao, X.; Xue, L.; Chu, X.; Chen, L. MicroRNA-149 increases the sensitivity of colorectal cancer cells to 5-fluorouracil by targeting forkhead box transcription factor FOXM1. CPB 2016, 39, 617–629. [Google Scholar] [CrossRef]
- Liang, Y.; Hou, L.; Li, L.; Li, L.; Zhu, L.; Wang, Y.; Huang, X.; Hou, Y.; Zhu, D.; Zou, H.; et al. Dichloroacetate restores colorectal cancer chemosensitivity through the P53/MiR-149-3p/PDK2-mediated glucose metabolic pathway. Oncogene 2020, 39, 469–485. [Google Scholar] [CrossRef]
- Senfter, D.; Holzner, S.; Kalipciyan, M.; Staribacher, A.; Walzl, A.; Huttary, N.; Krieger, S.; Brenner, S.; Jäger, W.; Krupitza, G.; et al. Loss of MiR-200 family in 5-fluorouracil resistant colon cancer drives lymphendothelial invasiveness in vitro. Hum. Mol. Genet. 2015, 24, 3689–3698. [Google Scholar] [CrossRef] [Green Version]
- Fang, L.; Li, H.; Wang, L.; Hu, J.; Jin, T.; Wang, J.; Yang, B.B. MicroRNA-17-5p promotes chemotherapeutic drug resistance and tumour metastasis of colorectal cancer by repressing PTEN expression. Oncotarget 2014, 5, 2974–2987. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Z.; Liu, S.; Tian, L.; Wu, M.; Ai, F.; Tang, W.; Zhao, L.; Ding, J.; Zhang, L.; Tang, A. MiR-124 and MiR-506 inhibit colorectal cancer progression by targeting DNMT3B and DNMT1. Oncotarget 2015, 6, 38139–38150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qian, X.; Yu, J.; Yin, Y.; He, J.; Wang, L.; Li, Q.; Zhang, L.-Q.; Li, C.-Y.; Shi, Z.-M.; Xu, Q.; et al. MicroRNA-143 inhibits tumor growth and angiogenesis and sensitizes chemosensitivity to oxaliplatin in colorectal cancers. Cell Cycle 2013, 12, 1385–1394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, L.-L.; Xie, F.-J.; Tang, C.-H.; Xu, W.-R.; Ding, X.-S.; Liang, J. MiR-340 suppresses tumor growth and enhances chemosensitivity of colorectal cancer by targeting RLIP76. Eur. Rev. Med. Pharmacol. Sci 2017, 21, 2875–2886. [Google Scholar]
- O’Brien, S.J.; Carter, J.V.; Burton, J.F.; Oxford, B.G.; Schmidt, M.N.; Hallion, J.C.; Galandiuk, S. The role of the MiR-200 family in epithelial–mesenchymal transition in colorectal cancer: A systematic review. Int. J. Cancer 2018, 142, 2501–2511. [Google Scholar] [CrossRef] [PubMed]
- Konno, M.; Koseki, J.; Asai, A.; Yamagata, A.; Shimamura, T.; Motooka, D.; Okuzaki, D.; Kawamoto, K.; Mizushima, T.; Eguchi, H.; et al. Distinct methylation levels of mature microRNAs in gastrointestinal cancers. Nat. Commun. 2019, 10, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Ng, E.K.O.; Tsang, W.P.; Ng, S.S.M.; Jin, H.C.; Yu, J.; Li, J.J.; Röcken, C.; Ebert, M.P.A.; Kwok, T.T.; Sung, J.J.Y. MicroRNA-143 targets DNA methyltransferases 3A in colorectal cancer. Br. J. Cancer 2009, 101, 699–706. [Google Scholar] [CrossRef] [Green Version]
- Huang, Z.; Li, Q.; Luo, K.; Zhang, Q.; Geng, J.; Zhou, X.; Xu, Y.; Qian, M.; Zhang, J.; Ji, L.; et al. MiR-340-FHL2 axis inhibits cell growth and metastasis in ovarian cancer. Cell Death Dis. 2019, 10, 372. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baharudin, R.; Rus Bakarurraini, N.Q.; Ismail, I.; Lee, L.-H.; Ab Mutalib, N.S. MicroRNA Methylome Signature and Their Functional Roles in Colorectal Cancer Diagnosis, Prognosis, and Chemoresistance. Int. J. Mol. Sci. 2022, 23, 7281. https://doi.org/10.3390/ijms23137281
Baharudin R, Rus Bakarurraini NQ, Ismail I, Lee L-H, Ab Mutalib NS. MicroRNA Methylome Signature and Their Functional Roles in Colorectal Cancer Diagnosis, Prognosis, and Chemoresistance. International Journal of Molecular Sciences. 2022; 23(13):7281. https://doi.org/10.3390/ijms23137281
Chicago/Turabian StyleBaharudin, Rashidah, Nurul Qistina Rus Bakarurraini, Imilia Ismail, Learn-Han Lee, and Nurul Syakima Ab Mutalib. 2022. "MicroRNA Methylome Signature and Their Functional Roles in Colorectal Cancer Diagnosis, Prognosis, and Chemoresistance" International Journal of Molecular Sciences 23, no. 13: 7281. https://doi.org/10.3390/ijms23137281







