Beneficial Effects of Theaflavins on Metabolic Syndrome: From Molecular Evidence to Gut Microbiome
Abstract
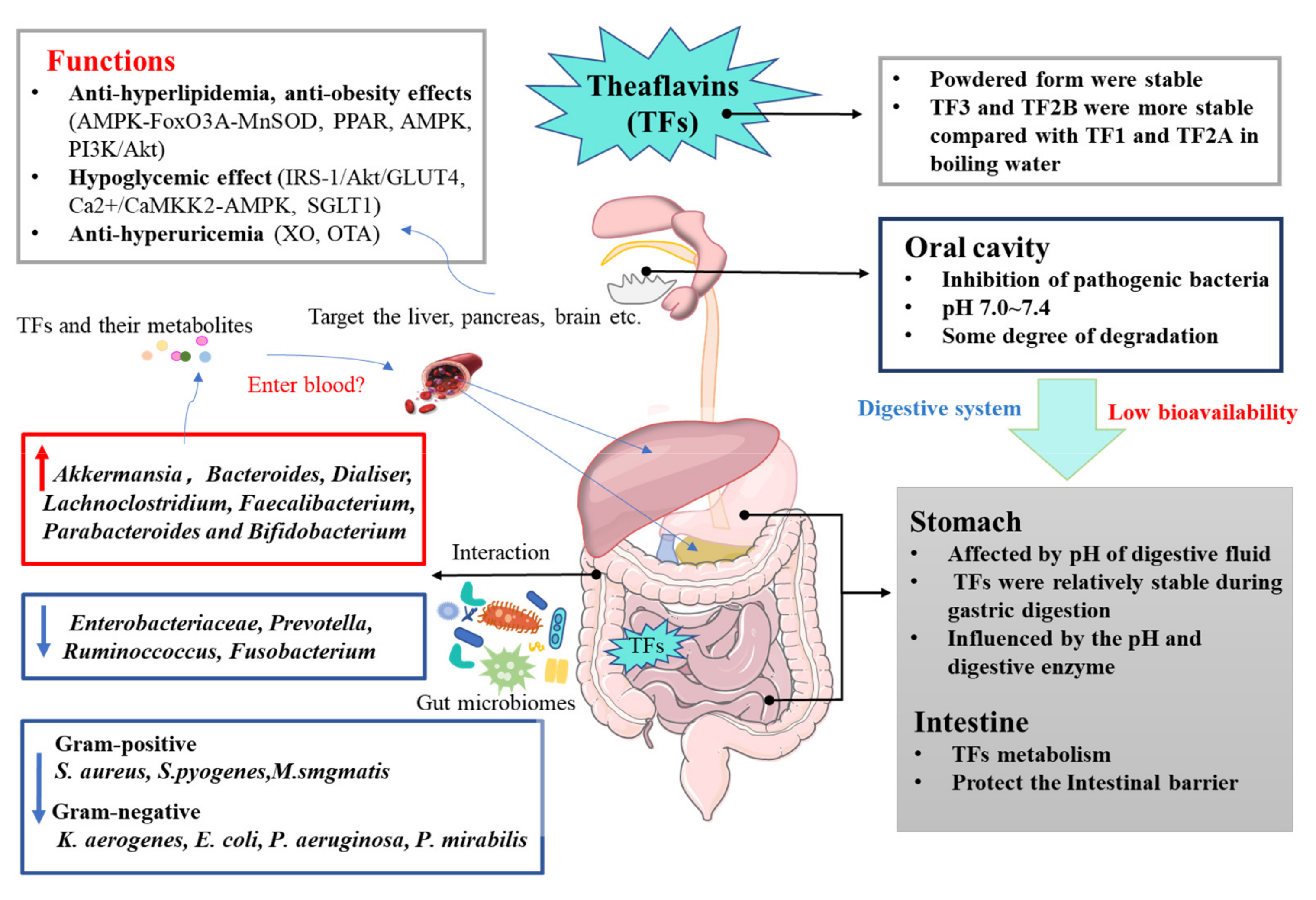
:1. Introduction
2. Stability and Bioavailability of Theaflavins
3. Metabolic Syndrome and Theaflavins
3.1. Antiobesity and Lipid-Lowering Effects
| Type | Related Disease | Cell Line/Animal Model | Treatment | Effects | References |
|---|---|---|---|---|---|
| Hyperlipidemia | Obesity | Mouse 3T3-L1 fibroblast | 0, 25, 50 μM TF3, 48 h | ↓ FAS expression | [38] |
| ↓ upregulation of CD36 and ACS | |||||
| ↑ gene expression of lipid catabolism and β-oxidation | |||||
| ↑ CPT-1L, CAD and HSL transcript levels | |||||
| ↑ UCP-1, UCP-2 | |||||
| ↑ Akt (Ser473) | |||||
| ↑ PPARα gene expression | |||||
| ↓ PPARγ upregulation | |||||
| ↓ phosphorylated FoxO3A | |||||
| ↓ inactive FoxO3A protein level | |||||
| ↑ MnSOD | |||||
| ↑ GFP intensity | |||||
| ICR mice | 0.5, 1.0, or 2.0 g/kg Y9 BTI for two weeks, | ↓ diet consumption | [41] | ||
| ↓ abdominal adipose weight | |||||
| ↑ fecal triglyceride | |||||
| ↓ lipid absorption | |||||
| ↑ Protein intake | |||||
| ↑ LKB1 and AMPK | |||||
| ↑ FAS | |||||
| ↑ phosphorylation of ACC. | |||||
| ↓ l IL-1β, iNOS, and Cox-2 | |||||
| C57BL/6 mice with HFD-induced obesity | 150, 300 mg/kg/day black tea extract for 9 weeks, orally | ↓ Body weight | [42] | ||
| ↓ food intake and body weight | |||||
| ↓ Liver and kidney weight | |||||
| ↓ WAT lipid accumulation | |||||
| ↓ total WAT mass | |||||
| ↓ adipocyte hypertrophy | |||||
| ↓ BCAAs and AAAs content | |||||
| ↑ PPP metabolites | |||||
| ↓ PPARα, Cpt1a, Ehhadhm and Acox1 | |||||
| ↓ FAS, Acc1 and Srebp1 | |||||
| ↑ p-Acc1 levels | |||||
| ↓ p-Irs1 (Ser 318) and PI3K-p85 levels | |||||
| ↑ Akt phosphorylation | |||||
| ↑ p-AMPK levels | |||||
| ↑ insulin signalling synergistically | |||||
| ↑ EDRs | |||||
| ↓ phospho-elF2α (Ser52) | |||||
| ↓ chol | |||||
| ↓ hepatotoxicity | |||||
| ↑ mRNA level (WAT lipolysis) | |||||
| fatty liver | HepG2 | 5 μM TF3, 4 h | ↓ SREBP-1c | [47] | |
| ↓ FAS | |||||
| ↑ CPT1 activity | |||||
| ↑ ACC phosphorylation | |||||
| ↓ PK activity | |||||
| ↓ hepatic lipid accumulation | |||||
| ↓ liver steatosis | |||||
| Dyslipidemia | Atherosclerosis | HUVEC (CRL-1730) | 5, 10 μmol/L TF1, 2 h | ↓ ROS | [45] |
| ↓ MDA | |||||
| ↑ SOD, CAT, and GSH-Px | |||||
| ↑ Nrf2 | |||||
| ↑ down-stream protein HO-1 | |||||
| ↑ miR-24 | |||||
| ApoE-/-mice, C57BL/6J mice | 5, 10 mg/kg TF for 12 weeks, intragastrically | ↓ serum TG, TC, and LDL-C elevation | |||
| ↑ HDL-C | |||||
| ↓ vacuoles size and number | |||||
| ↓ atherosclerotic lesion area | |||||
| ↓ MMP-2 | |||||
| ↓ MMP-9 | |||||
| ↓ ROS | |||||
| ↓ MDA | |||||
| ↑ antioxidant enzymes activities | |||||
| Dysglycemia | type 2 diebete | C2C12(T2D) | 20 μM TF1, 48 h | ↑ Ca2+ abundance | [49] |
| ↑ mitochondrial abundance | |||||
| ↑ CaMKK2 | |||||
| ↑ AMPK | |||||
| ↑ PGC-1α | |||||
| ↑ SIRT1 | |||||
| ↑ mitochondrial metabolic activity | |||||
| ↑ 2-NBDG uptake | |||||
| ↑ total GLUT4 | |||||
| HepG2 | 2.5, 5, 10 µg/mL TFs, 24 h | ↑ membrane bound GLUT4 | [50] | ||
| ↓ IRS-1 (Ser307) | |||||
| ↑ Akt (Ser473) | |||||
| ↑ glucose uptake | |||||
| ↑ insulin sensitivity | |||||
| ↑ mtDNA copy number | |||||
| ↓ PGC-1β | |||||
| ↑ PRC | |||||
| ↓ TC uptake | |||||
| ↓ blood glucose level | |||||
| HFD-induced mice | TF1, TF2a, TF3 100 mg kg/d, and TFs 200 mg kg/d for 9 weeks | ↓ serum glucose | [51] | ||
| ↓ TC, TG, LDL and HLD | |||||
| ↑ SIRT6 expression | |||||
| ↓ SREBP-1 and FASN expression | |||||
| ↓ Serum glucose | |||||
| ↑ glucose tolerance | |||||
| SDT rats | 2 mL theaflavin extract in 0.5% CMC, 25 mg/kg/day for 10-, 16-, 22-, 24- and 28-wk, orally | ↑ plasma insulin levels | [52] | ||
| ↑ GLP and GLP1 | |||||
| ↑ incretin secretion | |||||
| the development of pre-diabetes in control, affect glucose transporter expression | |||||
| ↓ blood glucose levels | |||||
| ↑ plasma insulin | |||||
| streptozotocin-induced diabetic rats | theaflavin (25, 50 and 100 mg/kg b.wt.) in 0.5 mL water for 30 days, intra- gastrically | ↓ HOMA-IR index | [53] | ||
| ↑ total hemoglobin | |||||
| ↓ HbA1C | |||||
| ↓ hexose, hexosamine, fucose, | |||||
| and sialic acid in plasma | |||||
| ↓ TCA cycle key enzymes activities | |||||
| ↑ plasma insulin level | |||||
| ↓ TG | |||||
| ↓ FFA | |||||
| 1 µM alloxan and 4% glucose induced diabetic Zebrafish model. | TF3 (0.5, 2, 4, 6.7, 10, and 20 µg/mL) or metformin hydrochloride (10 µg/mL) for 24 h | ↓ glucose level | [54] | ||
| ↓ PEPCK level | |||||
| ↑ GCK expression | |||||
| ↑ β cell regeneration rates | |||||
| uric acid metabolism | Hyperuricemia | Kunming male mice of SPF einjected with PO-induced Hyperuricemia | 20, 50 and 100 mg/kg/day TF, TF-3-G and TFDG for 7 days, intragastrically | ↓ SUA values | [55] |
| ↓ serum Cr values | |||||
| ↓ ADA | |||||
| ↓ XOD | |||||
| ↓ URAT1 | |||||
| ↓ GLUT9 | |||||
| ↑ ABCG2 mRNA | |||||
| ↓ OAT1/2 | |||||
| ↑ OCTN1, OAT1 and OAT2 mRNA | |||||
| ↓ inflammatory cells | |||||
| ↑ Nrf2 and HO-1 |
3.2. Hypoglycemic Activity
3.3. Uric Acid Lowering Effect
4. The Interactions of Theaflavins and Gut Microbiota
5. Conclusions and Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, X.; Li, S.; Chen, M.; Wang, J.; Xie, B.; Sun, Z. (−)-Epigallocatechin-3-gallate (EGCG) inhibits starch digestion and improves glucose homeostasis through direct or indirect activation of PXR/CAR-mediated phase II metabolism in diabetic mice. Food Funct. 2018, 9, 4651–4663. [Google Scholar] [CrossRef]
- Gregory, K.; Panagiota, P.; Eva, K.; George, C. Metabolic syndrome: Definitions and controversies. BMC Med. 2011, 9, 48. [Google Scholar]
- Cardiology Research and Practice. Retracted: A Comprehensive Review on Metabolic Syndrome. Cardiol. Res. Pract. 2019, 2019, 4301528. [Google Scholar]
- Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z. The metabolic syndrome. Lancet 2005, 365, 1415–1428. [Google Scholar] [CrossRef]
- Vogiatzoglou, A.; Mulligan, A.A.; Luben, R.N.; Lentjes, M.A.H.; Heiss, C.; Kelm, M.; Merx, M.W.; Spencer, J.P.E.; Schroeter, H.; Kuhnle, G.G.C. Assessment of the dietary intake of total flavan-3-ols, monomeric flavan-3-ols, proanthocyanidins and theaflavins in the European Union. Br. J. Nutr. 2014, 111, 1463–1473. [Google Scholar] [CrossRef] [Green Version]
- Derrick, S.A.; Kristo, A.S.; Reaves, S.K.; Sikalidis, A.K. Effects of Dietary Red Raspberry Consumption on Pre-Diabetes and Type 2 Diabetes Mellitus Parameters. Int. J. Environ. Res. Public Health 2021, 18, 9364. [Google Scholar] [CrossRef]
- Pascual-Teresa, S.; Santos-Buelga, C.; Rivas-Gonzalo, J.C. Quantitative analysis of flavan-3-ols in Spanish foodstuffs and beverages. J. Agric. Food Chem. 2000, 48, 5331–5337. [Google Scholar] [CrossRef]
- Sharma, V.; Rao, L.J.M. A Thought on the Biological Activities of Black Tea. Crit. Rev. Food Sci. Nutr. 2009, 49, 379–404. [Google Scholar] [CrossRef]
- Bhuyan, L.P.; Borah, P.; Sabhapondit, S.; Gogoi, R.; Bhattacharyya, P. Spatial variability of theaflavins and thearubigins fractions and their impact on black tea quality. J. Food Sci. Technol. 2015, 52, 7984–7993. [Google Scholar] [CrossRef] [Green Version]
- Sang, S.; Lambert, J.D.; Tian, S.; Hong, J.; Hou, Z.; Ryu, J.-H.; Stark, R.E.; Rosen, R.T.; Huang, M.-T.; Yang, C.S. Enzymatic synthesis of tea theaflavin derivatives and their anti-inflammatory and cytotoxic activities. Bioorg. Med. Chem. 2004, 12, 459–467. [Google Scholar] [CrossRef]
- Beecher, G.R. Overview of dietary flavonoids: Nomenclature, occurrence and intake. J. Nutr. 2003, 133, 3248S–3254S. [Google Scholar] [CrossRef]
- Teng, J.; Gong, Z.; Deng, Y.; Chen, L.; Li, Q.; Shao, Y.; Lin, L.; Xiao, W. Purification, characterization and enzymatic synthesis of theaflavins of polyphenol oxidase isozymes from tea leaf (Camellia sinensis). LWT 2017, 84, 263–270. [Google Scholar] [CrossRef]
- Bag, S.; Mondal, A.; Majumder, A.; Banik, A. Tea and its phytochemicals: Hidden health benefits & modulation of signaling cascade by phytochemicals. Food Chem. 2022, 371, 131098. [Google Scholar] [PubMed]
- Rahim, R.A.; Jayusman, P.A.; Muhammad, N.; Ahmad, F.; Mokhtar, N.; Mohamed, I.N.; Mohamed, N.; Shuid, A.N. Recent Advances in Nanoencapsulation Systems Using PLGA of Bioactive Phenolics for Protection against Chronic Diseases. Int. J. Environ. Res. Public Health 2019, 16, 4962. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lun, S.Y.; Kwok, L.L.; Yu, H.; Zhen-Yu, C. Stability of tea theaflavins and catechins. Food Chem. 2003, 83, 189–195. [Google Scholar] [CrossRef]
- Li, S.M.; Lo, C.Y.; Pan, M.H.; Lai, C.S.; Ho, C.T. Black tea: Chemical analysis and stability. Food Funct. 2013, 4, 10–18. [Google Scholar] [CrossRef]
- Jhoo, J.-W.; Lo, C.-Y.; Li, S.; Sang, S.; Ang, C.Y.W.; Heinze, T.M.; Ho, C.-T. Stability of black tea polyphenol, theaflavin, and identification of theanaphthoquinone as its major radical reaction product. J. Agric. Food Chem. 2005, 53, 6146–6150. [Google Scholar] [CrossRef]
- Garcia-Arieta, A. Interactions between active pharmaceutical ingredients and excipients affecting bioavailability: Impact on bioequivalence. Eur. J. Pharm. Sci. 2014, 65, 89–97. [Google Scholar] [CrossRef]
- Gomez-Mejia, E.; Rosales-Conrado, N.; Leon-Gonzalez, M.E.; Valverde, A.; Madrid, Y. A combined analytical-chemometric approach for the in vitro determination of polyphenol bioaccessibility by simulated gastrointestinal digestion. Anal. Bioanal. Chem. 2022, 414, 2739–2755. [Google Scholar] [CrossRef]
- Qu, F.; Ai, Z.; Liu, S.; Zhang, H.; Chen, Y.; Wang, Y.; Ni, D. Study on mechanism of low bioavailability of black tea theaflavins by using Caco-2 cell monolayer. Drug Deliv. 2021, 28, 1737–1747. [Google Scholar] [CrossRef]
- Flowers, S.A.; Bhat, S.; Lee, J.C. Potential Implications of Gut Microbiota in Drug Pharmacokinetics and Bioavailability. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2020, 40, 704–712. [Google Scholar] [CrossRef] [PubMed]
- Henning, S.M.; Aronson, W.; Niu, Y.; Conde, F.; Lee, N.H.; Seeram, N.P.; Lee, R.P.; Lu, J.; Harris, D.M.; Moro, A. Tea polyphenols and theaflavins are present in prostate tissue of humans and mice after green and black tea consumption. J. Nutr. 2006, 136, 1839. [Google Scholar] [CrossRef]
- Kondo, A.; Narumi, K.; Okuhara, K.; Takahashi, Y.; Iseki, K. Black tea extract and theaflavin derivatives affect the pharmacokinetics of rosuvastatin by modulating organic anion transporting polypeptide (OATP) 2B1 activity. Biopharm. Drug Dispos. 2019, 40, 302–306. [Google Scholar] [CrossRef] [PubMed]
- Kondo, A.; Narumi, K.; Ogura, J.; Sasaki, A.; Yabe, K.; Kobayashi, T.; Furugen, A.; Kobayashi, M.; Iseki, K. Organic anion-transporting polypeptide (OATP) 2B1 contributes to the cellular uptake of theaflavin. Drug Metab. Pharmacokinet. 2017, 32, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Barrueta, T.; Martínez-Bustos, F.; Castaño-Tostado, E.; Lee, Y.; Miller, M.J.; Amaya-Llano, S.L. Encapsulation of probiotics in whey protein isolate and modified huauzontle’s starch: An approach to avoid fermentation and stabilize polyphenol compounds in a ready-to-drink probiotic green tea. LWT 2020, 124, 109131. [Google Scholar] [CrossRef]
- Yin, Z.Y.; Zheng, T.; Ho, C.T.; Huang, Q.R.; Wu, Q.L.; Zhang, M. Improving the stability and bioavailability of tea polyphenols by encapsulations: A review. Food Sci. Hum. Wellness 2022, 11, 537–556. [Google Scholar] [CrossRef]
- Xia, Y.A.; Xc, B.; Shuang, L.C.; Lla, D.; Jie, W.E.; Min, W.B.; Liang, Z. Studies on the interactions of theaflavin-3,3′-digallate with bovine serum albumin: Multi-spectroscopic analysis and molecular docking. Food Chem. 2021, 366, 130422. [Google Scholar]
- Ye, Q.; Li, T.; Li, J.; Liu, L.; Zhang, X. Development and evaluation of tea polyphenols loaded water in oil emulsion with zein as stabilizer. J. Drug Deliv. Sci. Technol. 2020, 56, 101528. [Google Scholar] [CrossRef]
- Hu, J.; Yang, J.; Guo, H.; Yao, X.; Qiu, H.; Gao, P.; Wang, Q. Effect of Theaflavin on Inflammatory and Remolding of Airway in the Asthma Mice. J. Biomater. Tissue Eng. 2021, 11, 1091–1098. [Google Scholar] [CrossRef]
- Yu, X.; Cai, X.H.; Li, S.; Luo, L.Y.; Wang, J.; Wang, M.; Zeng, L. Studies on the interactions of theaflavin-3,3′-digallate with bovine serum albumin: Multi-spectroscopic analysis and molecular docking. Food Chem. 2022, 366, 130422. [Google Scholar] [CrossRef]
- Ding, Y.; Zou, L.; Lu, C.; Tong, H.; Chen, B. In situ enzymatic synthesis and purification of theaflavin-3,3′-digallate monomer and incorporation into nanoliposome. Int. J. Food Sci. Technol. 2018, 53, 2552–2559. [Google Scholar] [CrossRef]
- Srivastava, A.K.; Bhatnagar, P.; Singh, M. Synthesis of PLGA nanoparticles of tea polyphenols and their strong in vivo protective effect against chemically induced DNA damage. Int. J. Nanomed. 2013, 8, 1451, Erratum in Int. J. Nanomed. 2019, 14, 7001–7002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, Y.K.; Zheng, T.; Jin, W.P.; Shi, Y.X.; Huang, Q.R. Enhancing Intestinal Permeability of Theaflavin-3,3’-digallate by Chitosan-Caseinophosphopeptides Nanocomplexes. J. Agric. Food Chem. 2022, 70, 2029–2041. [Google Scholar] [CrossRef] [PubMed]
- He, H.F. Research progress on theaflavins: Efficacy, formation, and preparation. Food Nutr. Res. 2017, 61, 1344521. [Google Scholar] [CrossRef] [Green Version]
- Kun, Z.; Jie, O.; Jianan, H.; Zhonghua, L. Research progress of black tea thearubigins: A review. Crit. Rev. Food Sci. Nutr. 2021, 61, 1556–1566. [Google Scholar]
- Chooi, Y.C.; Ding, C.; Magkos, F. The epidemiology of obesity. Metabolism 2019, 92, 6–10. [Google Scholar] [CrossRef] [Green Version]
- Engin, A. The definition and prevalence of obesity and metabolic syndrome. In Obesity and Lipotoxicity; Springer: Cham, Switzerland, 2017; pp. 1–17. [Google Scholar]
- Ko, H.-J.; Lo, C.-Y.; Wang, B.-J.; Chiou, R.Y.-Y.; Lin, S.-M. Theaflavin-3,3′-digallate, a black tea polyphenol, stimulates lipolysis associated with the induction of mitochondrial uncoupling proteins and AMPK-FoxO3A-MnSOD pathway in 3T3-L1 adipocytes. J. Funct. Foods 2015, 17, 271–282. [Google Scholar] [CrossRef]
- Zhang, L.; Wen, S.P.; Da-Hai, G.U.; Zhi-Gang, L.I.; Cao, Z.H.; Rong, H.; Chang-Rong, G.E.; Jia, J.J. The Polymorphisms of UCPs Genes Associated with Fat Metabolism. China Anim. Husb. Vet. Med. 2010, 37, 1513–1522. [Google Scholar]
- Berardi, M.; Chou, J. Fatty Acid Flippase Activity of UCP2 Is Essential for Its Proton Transport in Mitochondria. Cell Metab. 2014, 20, 541–552. [Google Scholar] [CrossRef] [Green Version]
- Pan, S.; Deng, X.; Sun, S.; Lai, X.; Sun, L.; Li, Q.; Xiang, L.; Zhang, L.; Huang, Y. Black tea affects obesity by reducing nutrient intake and activating AMP-activated protein kinase in mice. Mol. Biol. Rep. 2018, 45, 689–697. [Google Scholar] [CrossRef]
- Xu, J.; Li, M.; Zhang, Y.; Chu, S.; Huo, Y.; Zhao, J.; Wan, C. Huangjinya Black Tea Alleviates Obesity and Insulin Resistance via Modulating Fecal Metabolome in High-Fat Diet-Fed Mice. Molecular Nutr. Food Res. 2020, 64, e2000353. [Google Scholar] [CrossRef] [PubMed]
- Glisan, S.L.; Grove, K.A.; Yennawar, N.H.; Lambert, J.D. Inhibition of pancreatic lipase by black tea theaflavins: Comparative enzymology and in silico modeling studies. Food Chem. 2017, 216, 296–300. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Yu, X.H.; Ou, X.; Ouyang, X.P.; Tang, C.K. Hepatic cholesterol transport and its role in non-alcoholic fatty liver disease and atherosclerosis. Prog. Lipid Res. 2021, 83, 101109. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Deng, Z.; Zou, Y.; Liu, C.; Fu, H.; Gu, Y.; Chang, H. Theaflavin alleviates oxidative injury and atherosclerosis progress via activating microRNA-24-mediated Nrf2/HO-1 signal. Phytother. Res. 2021, 35, 3418–3427. [Google Scholar] [CrossRef]
- Arroyave-Ospina, J.C.; Wu, Z.; Geng, Y.; Han, M. Role of Oxidative Stress in the Pathogenesis of Non-Alcoholic Fatty Liver Disease: Implications for Prevention and Therapy. Antioxidants 2021, 10, 174. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; An, R.; Li, Q.; Sun, L.; Lai, X.; Chen, R.; Li, D.; Sun, S. Theaflavin TF3 Relieves Hepatocyte Lipid Deposition through Activating an AMPK Signaling Pathway by targeting Plasma Kallikrein. J. Agric. Food Chem. 2020, 68, 2673–2683. [Google Scholar] [CrossRef]
- Luo, X.Y.; Takahara, T.; Hou, J.; Kawai, K.; Sugiyama, T.; Tsukada, K.; Takemoto, M.; Takeuchi, M.; Zhong, L.; Li, X.K. Theaflavin attenuates ischemia-reperfusion injury in a mouse fatty liver model. Biochem. Biophys. Res. Commun. 2012, 417, 287–293. [Google Scholar] [CrossRef]
- Qu, Z.; Liu, A.; Liu, C.; Tang, Q.; Zhan, L.; Xiao, W.; Huang, J.; Liu, Z.; Zhang, S. Theaflavin Promotes Mitochondrial Abundance and Glucose Absorption in Myotubes by Activating the CaMKK2-AMPK Signal Axis via Calcium-Ion Influx. J. Agric. Food Chem. 2021, 69, 8144–8159. [Google Scholar] [CrossRef]
- Tong, T.; Ren, N.; Soomi, P.; Wu, J.; Guo, N.; Kang, H.; Kim, E.; Wu, Y.; He, P.; Tu, Y.; et al. Theaflavins Improve Insulin Sensitivity through Regulating Mitochondrial Biosynthesis in Palmitic Acid-Induced HepG2 Cells. Molecules 2018, 23, 3382. [Google Scholar] [CrossRef] [Green Version]
- Cai, X.; Liu, Z.; Dong, X.; Wang, Y.; Zhu, L.; Li, M.; Xu, Y. Hypoglycemic and lipid lowering effects of theaflavins in high-fat diet-induced obese mice. Food Funct. 2021, 12, 9922–9931. [Google Scholar] [CrossRef]
- Li, B.; Fu, L.; Kojima, R.; Yamamoto, A.; Matsui, T. Theaflavins prevent the onset of diabetes through ameliorating glucose tolerance mediated by promoted incretin secretion in spontaneous diabetic Torii rats. J. Funct. Foods 2021, 86, 104702. [Google Scholar] [CrossRef]
- Gothandam, K.; Ganesan, V.S.; Ayyasamy, T.; Ramalingam, S. Protective effect of theaflavin on glycoprotein components and TCA cycle enzymes in high-fat diet and streptozotocin-induced diabetic rats. J. Basic Appl. Zool. 2019, 80, 43. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y. Simultaneous Tests of Theaflavin-3,3′-digallate as an Anti-Diabetic Drug in Human Hepatoma G2 Cells and Zebrafish (Danio rerio). Nutrients 2021, 13, 4379. [Google Scholar]
- Tai, L.; Liu, Z.; Sun, M.; Xie, Q.; Cai, X.; Wang, Y.; Dong, X.; Xu, Y. Anti-hyperuricemic effects of three theaflavins isolated from black tea in hyperuricemic mice. J. Funct. Foods 2020, 66, 103803. [Google Scholar] [CrossRef]
- Chatterjee, S.; Khunti, K.; Davies, M.J. Type 2 diabetes. Lancet 2017, 389, 2239–2251. [Google Scholar] [CrossRef]
- Maykish, A.; Sikalidis, A.K. Utilization of Hydroxyl-Methyl Butyrate, Leucine, Glutamine and Arginine Supplementation in Nutritional Management of Sarcopenia-Implications and Clinical Considerations for Type 2 Diabetes Mellitus Risk Modulation. J. Pers. Med. 2020, 10, 19. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.I.; Dominy, J.E.; Sikalidis, A.K.; Hirschberger, L.L.; Wang, W.; Stipanuk, M.H. HepG2/C3A cells respond to cysteine deprivation by induction of the amino acid deprivation/integrated stress response pathway. Physiol. Genom. 2008, 33, 218–229. [Google Scholar] [CrossRef] [Green Version]
- He, J.H.; Chen, L.X.; Li, H. Progress in the discovery of naturally occurring anti-diabetic drugs and in the identification of their molecular targets. Fitoterapia 2019, 134, 270–289. [Google Scholar] [CrossRef]
- Li, S.; Yin, L.; Yi, J.; Zhang, L.; Yang, L. Insight into interaction mechanism between theaflavin-3-gallate and α-glucosidase using spectroscopy and molecular docking analysis. J. Food Biochem. 2021, 45, e13550. [Google Scholar] [CrossRef]
- Sun, L.; Warren, F.J.; Netzel, G.; Gidley, M.J. 3 or 3′-Galloyl substitution plays an important role in association of catechins and theaflavins with porcine pancreatic α-amylase: The kinetics of inhibition of α-amylase by tea polyphenols. J. Funct. Foods 2016, 26, 144–156. [Google Scholar] [CrossRef]
- Hana, A.; Wang, Y.; Liu, D. Dietary Flavonoids in the Prevention of T2D: An Overview. Nutrients 2018, 10, 438. [Google Scholar]
- Bl, A.; Lei, F.A.; Ca, A.; Amn, A.; Ay, B.; Tm, A. Theaflavins inhibit glucose transport across Caco-2 cells through the downregulation of the Ca2+/AMP-activated protein kinase-mediated glucose transporter SGLT1. J. Funct. Foods 2020, 75, 104273. [Google Scholar]
- Hudish, L.I.; Reusch, J.; Sussel, L. β Cell dysfunction during progression of metabolic syndrome to type 2 diabetes. J. Clin. Investig. 2019, 129, 4001–4008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.; Luo, L.; Hu, S.; Gan, R.; Zeng, L. The chemistry, processing, and preclinical anti-hyperuricemia potential of tea: A comprehensive review. Crit. Rev. Food Sci. Nutr. 2022, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Li, Q.; Ye, Y.; Ran, M.; Jin, N. Inhibition of xanthine oxidase by theaflavin: Possible mechanism for anti-hyperuricaemia effect in mice. Process Biochem. 2020, 97, 11–18. [Google Scholar] [CrossRef]
- Dabke, K.; Hendrick, G.; Devkota, S. The gut microbiome and metabolic syndrome. J. Clin. Investig. 2019, 129, 4050–4057. [Google Scholar] [CrossRef]
- Herrema, H.; Niess, J.H. Intestinal microbial metabolites in human metabolism and type 2 diabetes. Diabetologia 2020, 63, 2533–2547. [Google Scholar] [CrossRef]
- Yin, R.; Kuo, H.-C.; Hudlikar, R.; Sargsyan, D.; Li, S.; Wang, L.; Wu, R.; Kong, A.-N. Gut microbiota, dietary phytochemicals and benefits to human health. Curr. Pharmacol. Rep. 2019, 5, 332–344. [Google Scholar] [CrossRef]
- Aravind, S.M.; Wichienchot, S.; Tsao, R.; Ramakrishnan, S.; Chakkaravarthi, S. Role of dietary polyphenols on gut microbiota, their metabolites and health benefits. Food Res. Int. 2021, 142, 110189. [Google Scholar] [CrossRef]
- Mulder, T.P.; van Platerink, C.J.; Schuyl, P.J.W.; van Amelsvoort, J.M. Analysis of theaflavins in biological fluids using liquid chromatography-electrospray mass spectrometry. J. Chromatogr. B Biomed. Sci. Appl. 2001, 760, 271–279. [Google Scholar] [CrossRef]
- Chen, H.D.; Parks, T.A.; Chen, X.X.; Gillitt, N.D.; Jobin, C.; Sang, S.M. Structural identification of mouse fecal metabolites of theaflavin 3,3′-digallate using liquid chromatography tandem mass spectrometry. J. Chromatogr. A 2011, 1218, 7297–7306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, H.; Hu, Y.Z.; Zhang, B.W.; Shao, Z.P.; Roura, E.G.; Wang, S. Tea polyphenol-gut microbiota interactions: Hints on improving the metabolic syndrome in a multi-element and multi-target manner. Food Sci. Hum. Wellness 2022, 11, 11–21. [Google Scholar] [CrossRef]
- Chen, H.D.; Sang, S.M. Biotransformation of tea polyphenols by gut microbiota. J. Funct. Foods 2014, 7, 26–42. [Google Scholar] [CrossRef]
- Chen, H.D.; Hayek, S.; Guzman, J.R.; Gillitt, N.D.; Ibrahim, S.A.; Jobin, C.; Sang, S.M. The Microbiota Is Essential for the Generation of Black Tea Theaflavins-Derived Metabolites. PLoS ONE 2012, 7, 10. [Google Scholar] [CrossRef] [Green Version]
- Pereira-Caro, G.; Moreno-Rojas, J.M.; Brindani, N.; Del Rio, D.; Lean, M.E.J.; Hara, Y.; Crozier, A. Bioavailability of Black Tea Theaflavins: Absorption, Metabolism, and Colonic Catabolism. J. Agric. Food Chem. 2017, 65, 5365–5374. [Google Scholar] [CrossRef]
- Liu, Z.; Bruijn, W.; Bruins, M.E.; Vincken, J.P. Microbial Metabolism of Theaflavin-3,3′-digallate and Its Gut Microbiota Composition Modulatory Effects. J. Agric. Food Chem. 2020, 69, 232–245. [Google Scholar] [CrossRef]
- Shuwei, Z.; Ohland, C.; Jobin, C.; Sang, S. Degradation of black tea theaflavin through C-ring cleavage by gut microbiota. Food Sci. Hum. Wellness 2022, 11, 598–605. [Google Scholar]
- Anandhan, A.; Tamilselvam, K.; Radhiga, T.; Rao, S.; Essa, M.M.; Manivasagam, T. Theaflavin, a black tea polyphenol, protects nigral dopaminergic neurons against chronic MPTP/probenecid induced Parkinson’s disease. Brain Res. 2012, 1433, 104–113. [Google Scholar] [CrossRef]
- Owuor, P.O.; Obanda, M. The use of green tea (Camellia sinensis) leaf flavan-3-ol composition in predicting plain black tea quality potential. Food Chem. 2007, 100, 873–884. [Google Scholar] [CrossRef]
- Chen, T.; Liu, A.B.; Sun, S.; Ajami, N.J.; Ross, M.C.; Wang, H.; Zhang, L.; Reuhl, K.; Kobayashi, K.; Onishi, J.C. Green tea polyphenols modify the gut microbiome in db/db mice as Co-abundance groups correlating with the blood glucose lowering effect. Mol. Nutr. Food Res. 2019, 63, 1801064. [Google Scholar] [CrossRef]
- Meiyan, W.; Jianying, L.; Ting, H.; Hui, Z. Metabolic fate of tea polyphenols and their crosstalk with gut microbiota. Food Sci. Hum. Wellness 2022, 11, 455–466. [Google Scholar]
- Cai, Q.S.; Ji, S.M.; Li, M.W.; Zheng, S.; Zhou, X.H.; Guo, H.M.; Deng, S.Y.; Zhu, J.Y.; Li, D.X.; Xie, Z.W. Theaflavin-regulated Imd condensates control Drosophila intestinal homeostasis and aging. iScience 2021, 24, 35. [Google Scholar] [CrossRef]
- Yussof, A.; Cammalleri, B.; Fayemiwo, O.; Lopez, S.; Chu, T.C. Antibacterial and Sporicidal Activity Evaluation of Theaflavin-3,3′-digallate. Int. J Mol. Sci. 2022, 23, 21. [Google Scholar] [CrossRef] [PubMed]
- Appice, G.M. Comparative Evaluation of the Antibacterial, Anti-biofilm and Anti-spore Effects of Theaflavins and Palmitoyl-EGCG. Master’s Thesis, Seton Hall University, South Orange, NJ, USA, 2017. [Google Scholar]
- Kong, J.H.; Zhang, G.Q.; Xia, K.; Diao, C.H.; Yang, X.F.; Zuo, X.B.; Li, Y.D.; Liang, X.L. Tooth brushing using toothpaste containing theaflavins reduces the oral pathogenic bacteria in healthy adults. 3 Biotech 2021, 11, 11. [Google Scholar] [CrossRef] [PubMed]
- Lagha, A.B.; Grenier, D. Tea polyphenols inhibit the activation of NF-κB and the secretion of cytokines and matrix metalloproteinases by macrophages stimulated with Fusobacterium nucleatum. Sci. Rep. 2016, 6, 34520. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Ohland, C.; Jobin, C.; Sang, S. Black Tea Theaflavin Detoxifies Metabolic Toxins in the Intestinal Tract of Mice. Mol. Nutr. Food Res. 2021, 65, 2000887. [Google Scholar] [CrossRef]
- Sun, C.P.; Tian, X.G.; Feng, L.; Wang, C.; Li, J.X.; Huo, X.K.; Zhao, W.Y.; Ning, J.; Yu, Z.L.; Deng, S.; et al. Inhibition of gut bacterial beta-glucuronidase by chemical components from black tea: Inhibition interactions and molecular mechanism. Arab. J. Chem. 2021, 14, 11. [Google Scholar] [CrossRef]
- Sun, L.; Xu, H.; Ye, J.; Gaikwad, N.W. Comparative effect of black, green, oolong, and white tea intake on weight gain and bile acid metabolism. Nutrition 2019, 65, 208–215. [Google Scholar] [CrossRef]
- Sung-Bum, C.; Kang-Jin, P.; Ho-Seok, K.; Dae-Ho, C.; Gi-Hoon, L.; Cho-Yun, C.; Kyu-Yeol, K.; Young-Lan, P.; Song, Y.A.; Wan-Sik, L. Black tea extract prevents lipopolysaccharide-induced NF-κB signaling and attenuates dextran sulfate sodium-induced experimental colitis. BMC Complementary Altern. Med. 2011, 11, 91. [Google Scholar]
- Angiletta, C.J.; Griffin, L.E.; Steele, C.N.; Baer, D.J.; Novotny, J.A.; Davy, K.P.; Neilson, A.P. Impact of short-term flavanol supplementation on fasting plasma trimethylamine N-oxide concentrations in obese adults. Food Funct. 2018, 9, 5350–5361. [Google Scholar] [CrossRef] [Green Version]
- Ma, Y.; Liu, G.; Tang, M.; Fang, J.; Jiang, H. Epigallocatechin Gallate Can Protect Mice from Acute Stress Induced by LPS While Stabilizing Gut Microbes and Serum Metabolites Levels. Front. Immunol. 2021, 12, 640305. [Google Scholar] [CrossRef] [PubMed]
- Gadaleta, R.M.; Erpecum, K.V.; Oldenburg, B.; Willemsen, E.; Renooij, W.; Murzilli, S.; Klomp, L.; Siersema, P.D.; Schipper, M.; Danese, S. Farnesoid X receptor activation inhibits inflammation and preserves the intestinal barrier in inflammatory bowel disease. Gut 2011, 60, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Shanahan, F. Therapeutic Manipulation of Gut Flora. (research on inflammatory bowel disease). Science 2000, 289, 1311–1312. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.A.; Park, Y.L.; Yoon, S.H.; Kim, K.Y.; Cho, S.B.; Lee, W.S.; Chung, I.J.; Joo, Y.E. Black tea polyphenol theaflavin suppresses LPS-induced ICAM-1 and VCAM-1 expression via blockage of NF-κB and JNK activation in intestinal epithelial cells. Inflamm. Res. 2011, 60, 493–500. [Google Scholar] [CrossRef]
- Yan, R.; Ho, C.; Zhang, X. Interaction Between Tea Polyphenols and Intestinal Microbiota in Host Metabolic Diseases from the Perspective of the Gut-Brain Axis. Mol. Nutr. Food Res. 2020, 64, 2000187. [Google Scholar] [CrossRef]
- Hu, S.S.; Luo, L.Y.; Zeng, L. Tea combats circadian rhythm disorder syndrome via the gut-liver-brain axis: Potential mechanisms speculated. Crit. Rev. Food Sci. Nutr. 2022, 1–22. [Google Scholar] [CrossRef]
- Guo, T.; Song, D.; Ho, C.T.; Zhang, X.; Wu, Z. Omics Analyses of Gut Microbiota in a Circadian Rhythm Disorder Mouse Model Fed with Oolong Tea Polyphenols. J. Agric. Food Chem. 2019, 67, 8847–8854. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, M.; Lu, Y.; Wu, J.; Zheng, Z.; Lv, C.; Ye, J.; Qin, S.; Zeng, C. Beneficial Effects of Theaflavins on Metabolic Syndrome: From Molecular Evidence to Gut Microbiome. Int. J. Mol. Sci. 2022, 23, 7595. https://doi.org/10.3390/ijms23147595
Shi M, Lu Y, Wu J, Zheng Z, Lv C, Ye J, Qin S, Zeng C. Beneficial Effects of Theaflavins on Metabolic Syndrome: From Molecular Evidence to Gut Microbiome. International Journal of Molecular Sciences. 2022; 23(14):7595. https://doi.org/10.3390/ijms23147595
Chicago/Turabian StyleShi, Meng, Yuting Lu, Junling Wu, Zhibing Zheng, Chenghao Lv, Jianhui Ye, Si Qin, and Chaoxi Zeng. 2022. "Beneficial Effects of Theaflavins on Metabolic Syndrome: From Molecular Evidence to Gut Microbiome" International Journal of Molecular Sciences 23, no. 14: 7595. https://doi.org/10.3390/ijms23147595
APA StyleShi, M., Lu, Y., Wu, J., Zheng, Z., Lv, C., Ye, J., Qin, S., & Zeng, C. (2022). Beneficial Effects of Theaflavins on Metabolic Syndrome: From Molecular Evidence to Gut Microbiome. International Journal of Molecular Sciences, 23(14), 7595. https://doi.org/10.3390/ijms23147595








