Effectiveness of SARS-CoV-2 Vaccines for Short- and Long-Term Immunity: A General Overview for the Pandemic Contrast
Abstract
:1. Introduction
2. COVID-19 Variants and Antibodies’ Response
2.1. Variants of Concern (VOC)
2.2. Variants of Interest (VOI)
2.3. Variants under Monitoring (VUM, Formerly Called “Alerts for Further Monitoring”)
3. Mechanisms of Relationships between ABO Blood Groups and COVID-19
4. Spike Protein Interactions
5. Serological IgM, IgG, and IgA
- The antigenic test is performed by a swab on nasal mucosa or oropharyngeal region by identifying SARS-CoV2-specific Spike glycoproteins (S). Nevertheless, this technique may provide false negatives. In many cases, the test should be subjected to a repetition in the following days [130].
- The molecular test, instead, identifies SARS-CoV-2 genomes in the substance and are present in swabs [130].
6. Anti-Spike and Anti-N Difference
7. COVID-19 Syndrome: Diagnosis and Treatment Procedure at Home and in Emergency
- The less severe clinical form, which includes the paucisymptomatic condition characterized by anosmia and ageusia; the oligosymptomatic form, which includes minor respiratory problems, nasal congestion, conjunctivitis, pharyngodynia, cough, and gastrointestinal problems (abdominal aches, vomiting, and diarrhea), and neurological symptoms such as dizziness, syncope, and headache [155];
- Clinical variants characterized by greater concerns and different stages. At the beginning, there are rapidly evolving pictures of the minor forms, described as follows: interstitial alveolar pneumonia with acute hypoxemic-hypercapnic pneumonia that may evolve to the most serious forms including sepsis, hypovolemic shock, and eventually multi-organ appearance syndrome (MODS).
- High risk clinical pictures are essential criteria for emergency clinical diagnosis, and they are as follows: alteration of the sensory, tachypnea HR > 30 beats/min, dyspnea, dehydration, oligo-anuria, very high fever (>39 °C) and protracted > 7 days, persistent dry cough, hoarse voice, anosmia/ageusia, intense, exhaustion, extreme fatigue, and body aches [156,157,158].
| COVID-19 SYNDROME TREATMENT | |
|---|---|
RISK FACTORS
| CLINICAL SEVERITY LEVELS OF ARDS
|
LABORATORY RESULTS
| EMERGENCY THERAPY Respiratory stabilization On the territory
|
PHARMACOLOGICAL THERAPY
| HOME THERAPY
|
8. Key Points in COVID-19 Patient Management
9. Major Complications of COVID-19
- Patient with P/F < 350 mmHg with CPAP or PEEP > 5 cm H2O should undergo NIMV;
- Patient with P/F < 200 mmHg with CPAP or PEEP > 5 cm H2O should be evaluated for early intubation;
- Patient with P/F < 100 mmHg with CPAP or PEEP > 5 cm H2O must be immediately intubated and subjected to cycles of pronation;
10. Anti-SARS-CoV-2 Vaccines
10.1. SARS-CoV-2 Vaccine Candidate Phase III Clinical Trials
10.1.1. Vaccine BBIBP-CorV; Developer: Beijing Institute of Biological Prodducts/Sinopharm
10.1.2. Vaccine Untitled; Developer: Wuhan Institute of Biological Products/Sinopharm
10.1.3. Vaccine: Ad5-nCoV; Developer: CanSino Biological Inc./Beijing Institute of Biotechnology
10.1.4. Vaccine: CoronaVac (PiCoVacc); Developer: Sinovac
10.1.5. Vaccine: Sputnik V; Developer: Gamaleya Research Institute
10.1.6. Vaccine: NVX-CoV2373; Developer: Novavax
10.2. SARS-CoV-2 Vaccines Phase IV Trials
10.2.1. Vaccine: BNT162b2; Developer: Pfizer/BioNTech/Fosun
10.2.2. Vaccine: mRNA-1273; Developer: Moderna/NIAID
10.2.3. Vaccine: AZD1222 (ChAdOx1 nCoV-19); Developer: Oxford University and AstraZeneca
10.2.4. Vaccine: Ad26.COV2-S; Developer: Janssen Pharmaceutical
10.2.5. Mucosal Vaccine Platform for COVID-19 Vaccine Development
10.2.6. Vaxart Vaccine—Developer: Vaxart
10.2.7. IosBio’s (Sabilitech’s) OraPro-COVID-19 Vaccine
10.3. COVID-19 Vaccine and Antibody Response in Children
- In individuals 16 years of age and older;
- In cases of emergency (EUA):
- -
- Prevent COVID-19 in individuals 12 through 15 years;
- -
- Administer a third dose to immunocompromised individuals over 12 years of age.
10.4. COVID-19 Vaccines for Moderately to Severely Immunocompromised People
- People being treated for cancer;
- People being treated with immunosuppressive drugs following organ transplantation;
- People being treated with immunosuppressive drugs following stem-cell transplantation (within 2 years);
- People with immunodeficiency as a result of syndromes such as DiGeorge syndrome and Wiskott–Aldrich syndrome;
- People with advanced or untreated HIV infections;
- Patients with suppressed immune responses following high-dose corticosteroid therapy.
11. Vaccine’s Implications in Pregnancy and Fertility
12. Side Effects and Post-Vaccination Pathologies Related to Antibody Titers
13. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
List of Abbreviations
| ACE2 | angiotensin-converting enzyme-2 |
| ACE | angiotensin-converting enzyme |
| ACE1 | angiotensin-converting enzyme 1 |
| AIFA | Agenzia Italiana del Farmaco |
| Alpha | English variant B.1.1.7 |
| Anti-RBD IgG | Immunogloublin G anti receptor-binding domain |
| Anti-Spike | test igg anti-Spike |
| BAU | unità arbitrarie vincolanti |
| Beta variant | (former of South Africa) |
| BMI | body mass index |
| CI | intervallo di confidenza |
| CLIAs | chemiluminescence immunoassay |
| CRP | C-reactive protein |
| Delta | Indian variant B.1.617.2 |
| ELISA | enzyme-linked immunosorbent assay |
| EMA | European Medicines Agency |
| ETA | variant B.1.525; date of designation Mar-2021 |
| Gamma | Brasilian variant P.1 |
| hACE2 receptor | human angiotensin I-converting enzyme 2 receptor |
| IFN | Interferon |
| IgA | Immunoglobulins A |
| IgG | Immunoglobulins G |
| IgM | Immunoglobulins M |
| IOTA | variant B.1.526; earliest documented samples USA (Nov-2020), date of designation Mar-2021 |
| IQR | interquartile range |
| KAPPA | Indian variant B.1.617.1 |
| LAMBA | variant C.37; earliest documented samples Peru (Aug-2020), date of designation June 2021 |
| LFIAs | lateral flow immunoassays. |
| MERS | Middle East Respiratory Syndrome |
| MMF | mycophenolate mofetil |
| MPA | mycophenolic acid |
| MPPDH | inosine-5′-monophosphate dehydrogenase |
| NAAT | nucleic acid amplification test |
| NGS | Next-Generation Sequencing |
| bNAbs | Broadly neutralizing antibodies |
| N-IgG | anti-N-IgG |
| PRD | viral prion-like domain |
| RBD | receptor-binding domain |
| RBDs | receptor-binding domains |
| RDB-IgG | receptor-binding domain neutralizing antibodies |
| RT-PCR | real-time PCR Polymerase chain reaction |
| S | Spike glycoprotein |
| SARS-CoV-1 | Severe Acute Respiratory Syndrome Coronavirus 1 |
| SARS-CoV-2 | Severe Acute Respiratory Syndrome Coronavirus 2 (COVID-19) |
| SARSr-CoV Rp3 | salivar protein similar to fused 8a and 8b SARS-CoV Beta Coronavirus |
| S-IgG | anti-Spike IgG |
| thio-NAD | thionicotinamide-adenine dinucleotide |
| TNF | tumor necrosis factor |
| VIPIT | prothrombotic immune thrombocytopenia |
| VOC | variants of concern |
| VOI | variants of interest |
| ZETA | variant P.2 |
References
- Cao, W.; Fang, Z.; Hou, G.; Han, M.; Xu, X.; Dong, J.; Zheng, J. The Psychological Impact of the COVID-19 Epidemic on College Students in China. Psychiatry Res. 2020, 287, 112934. [Google Scholar] [CrossRef] [PubMed]
- CDC. CDC 2020-2021 Flu Vaccine Campaign Kickoff. Available online: https://www.cdc.gov/flu/spotlights/2020-2021/2020-21-campaign-kickoff.htm (accessed on 11 July 2022).
- Han, Q.; Lin, Q.; Ni, Z.; You, L. Uncertainties about the Transmission Routes of 2019 Novel Coronavirus. Influenza Other Respir. Viruses 2020, 14, 470–471. [Google Scholar] [CrossRef] [PubMed]
- Klompas, M.; Baker, M.A.; Rhee, C. Airborne Transmission of SARS-CoV-2: Theoretical Considerations and Available Evidence. JAMA 2020, 324, 441. [Google Scholar] [CrossRef]
- Jiang, F.; Deng, L.; Zhang, L.; Cai, Y.; Cheung, C.W.; Xia, Z. Review of the Clinical Characteristics of Coronavirus Disease 2019 (COVID-19). J. Gen. Intern. Med. 2020, 35, 1545–1549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spagnolo, L.; Vimercati, L.; Caputi, A.; Benevento, M.; De Maria, L.; Ferorelli, D.; Solarino, B. Role and Tasks of the Occupational Physician during the COVID-19 Pandemic. Medicina 2021, 57, 479. [Google Scholar] [CrossRef]
- Boffetta, P.; Violante, F.; Durando, P.; De Palma, G.; Pira, E.; Vimercati, L.; Cristaudo, A.; Icardi, G.; Sala, E.; Coggiola, M. Determinants of SARS-CoV-2 Infection in Italian Healthcare Workers: A Multicenter Study. Sci. Rep. 2021, 11, 5788. [Google Scholar] [CrossRef] [PubMed]
- Vimercati, L.; De Maria, L.; Quarato, M.; Caputi, A.; Stefanizzi, P.; Gesualdo, L.; Migliore, G.; Fucilli, F.I.M.; Cavone, D.; Delfino, M.C.; et al. COVID-19 Hospital Outbreaks: Protecting Healthcare Workers to Protect Frail Patients. An Italian Observational Cohort Study. Int. J. Infect. Dis. 2021, 102, 532–537. [Google Scholar] [CrossRef]
- Luyten, J.; Beutels, P. The Social Value Of Vaccination Programs: Beyond Cost-Effectiveness. Health Aff. 2016, 35, 212–218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scarano, A.; Inchingolo, F.; Lorusso, F. Facial Skin Temperature and Discomfort When Wearing Protective Face Masks: Thermal Infrared Imaging Evaluation and Hands Moving the Mask. Int. J. Environ. Res. Public Health 2020, 17, 4624. [Google Scholar] [CrossRef]
- Charitos, I.A.; Del Prete, R.; Inchingolo, F.; Mosca, A.; Carretta, D.; Ballini, A.; Santacroce, L. What We Have Learned for the Future about COVID-19 and Healthcare Management of It? Acta Biomed. 2020, 91, e2020126. [Google Scholar]
- Dohan Ehrenfest, D.M.; Del Corso, M.; Inchingolo, F.; Charrier, J.-B. Selecting a Relevant in Vitro Cell Model for Testing and Comparing the Effects of a Choukroun’s Platelet-Rich Fibrin (PRF) Membrane and a Platelet-Rich Plasma (PRP) Gel: Tricks and Traps. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2010, 110, 409–411, author reply 411–413. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Inchingolo, F.; Lorusso, F. Environmental Disinfection of a Dental Clinic during the COVID-19 Pandemic: A Narrative Insight. Biomed. Res. Int. 2020, 2020, 8896812. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Inchingolo, F.; Rapone, B.; Festa, F.; Tari, S.R.; Lorusso, F. Protective Face Masks: Effect on the Oxygenation and Heart Rate Status of Oral Surgeons during Surgery. Int. J. Environ. Res. Public Health 2021, 18, 2363. [Google Scholar] [CrossRef] [PubMed]
- Inchingolo, A.D.; Inchingolo, A.M.; Bordea, I.R.; Malcangi, G.; Xhajanka, E.; Scarano, A.; Lorusso, F.; Farronato, M.; Tartaglia, G.M.; Isacco, C.G.; et al. SARS-CoV-2 Disease Adjuvant Therapies and Supplements Breakthrough for the Infection Prevention. Microorganisms 2021, 9, 525. [Google Scholar] [CrossRef] [PubMed]
- Satarker, S.; Nampoothiri, M. Structural Proteins in Severe Acute Respiratory Syndrome Coronavirus-2. Arch. Med. Res. 2020, 51, 482–491. [Google Scholar] [CrossRef]
- Yadav, R.; Chaudhary, J.K.; Jain, N.; Chaudhary, P.K.; Khanra, S.; Dhamija, P.; Sharma, A.; Kumar, A.; Handu, S. Role of Structural and Non-Structural Proteins and Therapeutic Targets of SARS-CoV-2 for COVID-19. Cells 2021, 10, 821. [Google Scholar] [CrossRef]
- Balzanelli, M.G.; Distratis, P.; Lazzaro, R.; D’Ettorre, E.; Nico, A.; Inchingolo, F.; Dipalma, G.; Tomassone, D.; Serlenga, E.M.; Dalagni, G. New Translational Trends in Personalized Medicine: Autologous Peripheral Blood Stem Cells and Plasma for COVID-19 Patient. J. Pers. Med. 2022, 12, 85. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Yang, C.; Xu, X.-F.; Xu, W.; Liu, S.-W. Structural and Functional Properties of SARS-CoV-2 Spike Protein: Potential Antivirus Drug Development for COVID-19. Acta Pharm. Sin. 2020, 41, 1141–1149. [Google Scholar] [CrossRef]
- Yang, J.; Petitjean, S.J.; Koehler, M.; Zhang, Q.; Dumitru, A.C.; Chen, W.; Derclaye, S.; Vincent, S.P.; Soumillion, P.; Alsteens, D. Molecular Interaction and Inhibition of SARS-CoV-2 Binding to the ACE2 Receptor. Nat. Commun. 2020, 11, 4541. [Google Scholar] [CrossRef]
- Bošnjak, B.; Stein, S.C.; Willenzon, S.; Cordes, A.K.; Puppe, W.; Bernhardt, G.; Ravens, I.; Ritter, C.; Schultze-Florey, C.R.; Gödecke, N.; et al. Low Serum Neutralizing Anti-SARS-CoV-2 S Antibody Levels in Mildly Affected COVID-19 Convalescent Patients Revealed by Two Different Detection Methods. Cell. Mol. Immunol. 2021, 18, 936–944. [Google Scholar] [CrossRef]
- Arashkia, A.; Jalilvand, S.; Mohajel, N.; Afchangi, A.; Azadmanesh, K.; Salehi-Vaziri, M.; Fazlalipour, M.; Pouriayevali, M.H.; Jalali, T.; Mousavi Nasab, S.D.; et al. Severe Acute Respiratory Syndrome-coronavirus-2 Spike (S) Protein Based Vaccine Candidates: State of the Art and Future Prospects. Rev. Med. Virol. 2021, 31. [Google Scholar] [CrossRef]
- Tracking SARS-CoV-2 Variants. Available online: https://www.who.int/en/activities/tracking-SARS-CoV-2-variants/ (accessed on 7 January 2022).
- Chavda, V.P.; Apostolopoulos, V. Global Impact of Delta plus Variant and Vaccination. Expert Rev. Vaccines 2022, 21, 597–600. [Google Scholar] [CrossRef] [PubMed]
- Classification of Omicron (B.1.1.529): SARS-CoV-2 Variant of Concern. Available online: https://www.who.int/news/item/26-11-2021-classification-of-omicron-(b.1.1.529)-sars-cov-2-variant-of-concern (accessed on 7 January 2022).
- Pulliam, J.R.; van Schalkwyk, C.; Govender, N.; von Gottberg, A.; Cohen, C.; Groome, M.J.; Dushoff, J.; Mlisana, K.; Moultrie, H. Increased Risk of SARS-CoV-2 Reinfection Associated with Emergence of the Omicron Variant in South Africa. MedRxiv 2021, 376, eabn4947. [Google Scholar]
- Callaway, E.; Ledford, H. How Bad Is Omicron? What Scientists Know so Far. Nature 2021, 600, 197–199. [Google Scholar] [CrossRef]
- Xiang, F.; Wang, X.; He, X.; Peng, Z.; Yang, B.; Zhang, J.; Zhou, Q.; Ye, H.; Ma, Y.; Li, H.; et al. Antibody Detection and Dynamic Characteristics in Patients With Coronavirus Disease 2019. Clin. Infect. Dis. 2020, 71, 1930–1934. [Google Scholar] [CrossRef] [PubMed]
- WHO. Prevention SARS-CoV-2 Variant Classifications and Definitions. 2021. Available online: https://www.who.int/activities/tracking-SARS-CoV-2-variants (accessed on 7 January 2022).
- Preliminary Genomic Characterisation of an Emergent SARS-CoV-2 Lineage in the UK Defined by a Novel Set of Spike Mutations—SARS-CoV-2 Coronavirus/NCoV-2019 Genomic Epidemiology. Available online: https://virological.org/t/preliminary-genomic-characterisation-of-an-emergent-sars-cov-2-lineage-in-the-uk-defined-by-a-novel-set-of-spike-mutations/563 (accessed on 12 July 2022).
- Horby, P.; Barclay, W. NERVTAG Paper Brief Update on SARS-CoV-2 Variants [New Text in Red]. 5; Scientific Advisory Group for Emergencies: London, UK, 2021. [Google Scholar]
- The COVID-19 Genomics UK (COG-UK) consortium; Volz, E.; Mishra, S.; Chand, M.; Barrett, J.C.; Johnson, R.; Geidelberg, L.; Hinsley, W.R.; Laydon, D.J.; Dabrera, G.; et al. Assessing Transmissibility of SARS-CoV-2 Lineage B.1.1.7 in England. Nature 2021, 593, 266–269. [Google Scholar] [CrossRef] [PubMed]
- Krause, P.R.; Fleming, T.R.; Longini, I.M.; Peto, R.; Briand, S.; Heymann, D.L.; Beral, V.; Snape, M.D.; Rees, H.; Ropero, A.-M.; et al. SARS-CoV-2 Variants and Vaccines. N. Engl. J. Med. 2021, 385, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Lauring, A.S.; Hodcroft, E.B. Genetic Variants of SARS-CoV-2—What Do They Mean? JAMA 2021, 325, 529. [Google Scholar] [CrossRef]
- Lorusso, F.; Inchingolo, F.; Scarano, A. The Impact of COVID-19 on the Scientific Production Spread: A Five-Month Bibliometric Report of the Worldwide Research Community. Acta Med. Mediterr. 2020, 36, 3357–3360. [Google Scholar] [CrossRef]
- Callaway, E. The Coronavirus Is Mutating—Does It Matter? Nature 2020, 585, 174–177. [Google Scholar] [CrossRef]
- Dao, M.H.; Phan, L.T.; Cao, T.M.; Luong, Q.C.; Pham, H.T.T.; Vu, N.H.P.; Khuu, N.V.; Nguyen, T.V.; Nguyen, L.T.; Nguyen, H.T.; et al. Genome-wide Analysis of SARS-CoV-2 Strains Circulating in Vietnam: Understanding the Nature of the Epidemic and Role of the D614G Mutation. J. Med. Virol. 2021, 93, 5660–5665. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Pham, T.N.; Van, T.D.; Nguyen, T.T.; Nguyen, D.T.N.; Le, H.N.M.; Eden, J.-S.; Rockett, R.J.; Nguyen, T.T.H.; Vu, B.T.N.; et al. Genetic Diversity of SARS-CoV-2 and Clinical, Epidemiological Characteristics of COVID-19 Patients in Hanoi, Vietnam. PLoS ONE 2020, 15, e0242537. [Google Scholar] [CrossRef] [PubMed]
- Aleem, A.; Akbar Samad, A.B.; Slenker, A.K. Emerging Variants of SARS-CoV-2 And Novel Therapeutics Against Coronavirus (COVID-19). In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Kim, Y.J.; Jang, U.S.; Soh, S.M.; Lee, J.-Y.; Lee, H.-R. The Impact on Infectivity and Neutralization Efficiency of SARS-CoV-2 Lineage B.1.351 Pseudovirus. Viruses 2021, 13, 633. [Google Scholar] [CrossRef] [PubMed]
- Yurkovetskiy, L.; Wang, X.; Pascal, K.E.; Tomkins-Tinch, C.; Nyalile, T.; Wang, Y.; Baum, A.; Diehl, W.E.; Dauphin, A.; Carbone, C.; et al. Structural and Functional Analysis of the D614G SARS-CoV-2 Spike Protein Variant. Microbiology 2020, 183, 739–751. [Google Scholar]
- Korber, B.; Fischer, W.M.; Gnanakaran, S.; Yoon, H.; Theiler, J.; Abfalterer, W.; Hengartner, N.; Giorgi, E.E.; Bhattacharya, T.; Foley, B.; et al. Tracking Changes in SARS-CoV-2 Spike: Evidence That D614G Increases Infectivity of the COVID-19 Virus. Cell 2020, 182, 812–827.e19. [Google Scholar] [CrossRef] [PubMed]
- Yurkovetskiy, L.; Pascal, K.E.; Tomkins-Tinch, C.; Nyalile, T.; Wang, Y.; Baum, A.; Diehl, W.E.; Dauphin, A.; Carbone, C.; Veinotte, K. SARS-CoV-2 Spike Protein Variant D614G Increases Infectivity and Retains Sensitivity to Antibodies That Target the Receptor Binding Domain. BioRxiv 2020. [Google Scholar] [CrossRef]
- England, P.H. SARS-CoV-2 Variants of Concern and Variants under Investigation in England. Public Health Engl. 2021, 11, 1–61. [Google Scholar]
- Yang, Q.; Meyerson, N.R.; Clark, S.K.; Paige, C.L.; Fattor, W.T.; Gilchrist, A.R.; Barbachano-Guerrero, A.; Healy, B.G.; Worden-Sapper, E.R.; Wu, S.S.; et al. Saliva TwoStep for Rapid Detection of Asymptomatic SARS-CoV-2 Carriers. eLife 2021, 10, e65113. [Google Scholar] [CrossRef]
- Sandoval Torrientes, M.; Castelló Abietar, C.; Boga Riveiro, J.; Álvarez-Argüelles, M.E.; Rojo-Alba, S.; Abreu Salinas, F.; Costales González, I.; Pérez Martínez, Z.; Martín Rodríguez, G.; Gómez de Oña, J.; et al. A Novel Single Nucleotide Polymorphism Assay for the Detection of N501Y SARS-CoV-2 Variants. J. Virol. Methods 2021, 294, 114143. [Google Scholar] [CrossRef]
- Vega-Magaña, N.; Sánchez-Sánchez, R.; Hernández-Bello, J.; Venancio-Landeros, A.A.; Peña-Rodríguez, M.; Vega-Zepeda, R.A.; Galindo-Ornelas, B.; Díaz-Sánchez, M.; García-Chagollán, M.; Macedo-Ojeda, G.; et al. RT-QPCR Assays for Rapid Detection of the N501Y, 69-70del, K417N, and E484K SARS-CoV-2 Mutations: A Screening Strategy to Identify Variants with Clinical Impact. Front. Cell. Infect. Microbiol. 2021, 11, 672562. [Google Scholar] [CrossRef] [PubMed]
- Yaniv, K.; Ozer, E.; Shagan, M.; Lakkakula, S.; Plotkin, N.; Bhandarkar, N.S.; Kushmaro, A. Direct RT-QPCR Assay for SARS-CoV-2 Variants of Concern (Alpha, B.1.1.7 and Beta, B.1.351) Detection and Quantification in Wastewater. Environ. Res. 2021, 201, 111653. [Google Scholar] [CrossRef] [PubMed]
- Vogels, C.B.F.; Breban, M.I.; Ott, I.M.; Alpert, T.; Petrone, M.E.; Watkins, A.E.; Kalinich, C.C.; Earnest, R.; Rothman, J.E.; Goes de Jesus, J.; et al. Multiplex QPCR Discriminates Variants of Concern to Enhance Global Surveillance of SARS-CoV-2. PLoS Biol. 2021, 19, e3001236. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, G.; Mancini, P.; Bonanno Ferraro, G.; Veneri, C.; Iaconelli, M.; Lucentini, L.; Bonadonna, L.; Brusaferro, S.; Brandtner, D.; Fasanella, A.; et al. Rapid Screening for SARS-CoV-2 Variants of Concern in Clinical and Environmental Samples Using Nested RT-PCR Assays Targeting Key Mutations of the Spike Protein. Water Res. 2021, 197, 117104. [Google Scholar] [CrossRef]
- Vircell. Available online: https://en.vircell.com/ (accessed on 12 July 2022).
- New VIASURE SARS-CoV-2 Variant I & Variant II RT-PCR Kits—CERTEST Biotec IVD Diagnostic Products. Available online: https://www.certest.es/news/new-viasure-sars-cov-2-variant-i-variant-ii-rt-pcr-kits/ (accessed on 12 July 2022).
- Maglione, M.; Bevilacqua, L.; Dotto, F.; Costantinides, F.; Lorusso, F.; Scarano, A. Observational Study on the Preparation of the Implant Site with Piezosurgery vs. Drill: Comparison between the Two Methods in Terms of Postoperative Pain, Surgical Times, and Operational Advantages. BioMed Res. Int. 2019, 2019, 8483658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inchingolo, A.D.; Dipalma, G.; Inchingolo, A.M.; Malcangi, G.; Santacroce, L.; D’oria, M.T.; Isacco, C.G.; Bordea, I.R.; Candrea, S.; Scarano, A.; et al. The 15-Months Clinical Experience of SARS-CoV-2: A Literature Review of Therapies and Adjuvants. Antioxidants 2021, 10, 881. [Google Scholar] [CrossRef] [PubMed]
- Pham, V.H.; Gargiulo Isacco, C.; Nguyen, K.C.D.; Le, S.H.; Tran, D.K.; Nguyen, Q.V.; Pham, H.T.; Aityan, S.; Pham, S.T.; Cantore, S.; et al. Rapid and Sensitive Diagnostic Procedure for Multiple Detection of Pandemic Coronaviridae Family Members SARS-CoV-2, SARS-CoV, MERS-CoV and HCoV: A Translational Research and Cooperation between the Phan Chau Trinh University in Vietnam and University of Bari “Aldo Moro” in Italy. Eur. Rev. Med. Pharm. Sci. 2020, 24, 7173–7191. [Google Scholar] [CrossRef]
- GISAID—HCov19 Variants. Available online: https://www.gisaid.org/hcov19-variants/ (accessed on 19 August 2021).
- Kimura, I.; Kosugi, Y.; Wu, J.; Yamasoba, D.; Butlertanaka, E.P.; Tanaka, Y.L.; Liu, Y.; Shirakawa, K.; Kazuma, Y.; Nomura, R. SARS-CoV-2 Lambda Variant Exhibits Higher Infectivity and Immune Resistance. bioRxiv 2021. [Google Scholar] [CrossRef]
- Acevedo, M.L.; Alonso-Palomares, L.; Bustamante, A.; Gaggero, A.; Paredes, F.; Cortés, C.P.; Valiente-Echeverría, F.; Soto-Rifo, R. Infectivity and Immune Escape of the New SARS-CoV-2 Variant of Interest Lambda. medRxiv 2021. [Google Scholar] [CrossRef]
- KFF. COVID-19 Vaccine Breakthrough Cases: Data from the States. Available online: https://www.kff.org/policy-watch/covid-19-vaccine-breakthrough-cases-data-from-the-states/ (accessed on 19 August 2021).
- Dhama, K.; Khan, S.; Tiwari, R.; Sircar, S.; Bhat, S.; Malik, Y.S.; Singh, K.P.; Chaicumpa, W.; Bonilla-Aldana, D.K.; Rodriguez-Morales, A.J. Coronavirus Disease 2019–COVID-19. Clin. Microbiol. Rev. 2020, 33, e00028-20. [Google Scholar] [CrossRef]
- Christie, A.; Brooks, J.T.; Hicks, L.A.; Sauber-Schatz, E.K.; Yoder, J.S.; Honein, M.A. CDC COVID-19 Response Team Guidance for Implementing COVID-19 Prevention Strategies in the Context of Varying Community Transmission Levels and Vaccination Coverage. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1044–1047. [Google Scholar] [CrossRef]
- Steensels, D.; Pierlet, N.; Penders, J.; Mesotten, D.; Heylen, L. Comparison of SARS-CoV-2 Antibody Response Following Vaccination With BNT162b2 and MRNA-1273. JAMA 2021, 326, 1533. [Google Scholar] [CrossRef] [PubMed]
- Latin America DW. Coronavirus Lambda Variant Spreads across Latin America. 28 July 2021. Available online: https://www.dw.com/en/coronavirus-lambda-variant-spreads-across-latin-america/a-58035249 (accessed on 12 July 2022).
- Nikolopoulou, G.B.; Maltezou, H.C. COVID-19 in Children: Where Do We Stand? Arch. Med. Res. 2021, 53, 1–8. [Google Scholar] [CrossRef]
- Tipnis, S.R.; Hooper, N.M.; Hyde, R.; Karran, E.; Christie, G.; Turner, A.J. A Human Homolog of Angiotensin-Converting Enzyme. J. Biol. Chem. 2000, 275, 33238–33243. [Google Scholar] [CrossRef] [Green Version]
- Balzanelli, M.G.; Distratis, P.; Catucci, O.; Cefalo, A.; Lazzaro, R.; Inchingolo, F.; Tomassone, D.; Aityan, S.K.; Ballini, A.; Nguyen, K.C. Mesenchymal Stem Cells: The Secret Children’s Weapons against the SARS-CoV-2 Lethal Infection. Appl. Sci. 2021, 11, 1696. [Google Scholar] [CrossRef]
- Dohan Ehrenfest, D.M.; de Peppo, G.M.; Doglioli, P.; Sammartino, G. Slow Release of Growth Factors and Thrombospondin-1 in Choukroun’s Platelet-Rich Fibrin (PRF): A Gold Standard to Achieve for All Surgical Platelet Concentrates Technologies. Growth Factors 2009, 27, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Cheng, G.; Chui, C.H.; Lau, F.Y.; Chan, P.K.; Ng, M.H.; Sung, J.J.; Wong, R.S. ABO Blood Group and Susceptibility to Severe Acute Respiratory Syndrome. JAMA 2005, 293, 1447. [Google Scholar] [CrossRef]
- Breiman, A.; Ruvën-Clouet, N.; Le Pendu, J. Harnessing the Natural Anti-Glycan Immune Response to Limit the Transmission of Enveloped Viruses Such as SARS-CoV-2. PLoS Pathog. 2020, 16, e1008556. [Google Scholar] [CrossRef]
- Wu, S.-C.; Arthur, C.M.; Wang, J.; Verkerke, H.; Josephson, C.D.; Kalman, D.; Roback, J.D.; Cummings, R.D.; Stowell, S.R. The SARS-CoV-2 Receptor-Binding Domain Preferentially Recognizes Blood Group A. Blood Adv. 2021, 5, 1305–1309. [Google Scholar] [CrossRef]
- Cooling, L. Blood Groups in Infection and Host Susceptibility. Clin. Microbiol. Rev. 2015, 28, 801–870. [Google Scholar] [CrossRef] [Green Version]
- Anstee, D.J. The Relationship between Blood Groups and Disease. Blood 2010, 115, 4635–4643. [Google Scholar] [CrossRef] [Green Version]
- Pillay, T.S. Gene of the Month: The 2019-NCoV/SARS-CoV-2 Novel Coronavirus Spike Protein. J. Clin. Pathol. 2020, 73, 366–369. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Du, L.; Shi, Z. An Emerging Coronavirus Causing Pneumonia Outbreak in Wuhan, China: Calling for Developing Therapeutic and Prophylactic Strategies. Emerg. Microbes Infect. 2020, 9, 275–277. [Google Scholar] [CrossRef]
- Bosch, B.J.; van der Zee, R.; de Haan, C.A.M.; Rottier, P.J.M. The Coronavirus Spike Protein Is a Class I Virus Fusion Protein: Structural and Functional Characterization of the Fusion Core Complex. J. Virol. 2003, 77, 8801–8811. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inchingolo, A.D.; Inchingolo, A.M.; Bordea, I.R.; Malcangi, G.; Xhajanka, E.; Scarano, A.; Lorusso, F.; Farronato, M.; Tartaglia, G.M.; Isacco, C.G.; et al. SARS-CoV-2 Disease through Viral Genomic and Receptor Implications: An Overview of Diagnostic and Immunology Breakthroughs. Microorganisms 2021, 9, 793. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Zhu, Y.; Liu, M.; Lan, Q.; Xu, W.; Wu, Y.; Ying, T.; Liu, S.; Shi, Z.; Jiang, S.; et al. Fusion Mechanism of 2019-NCoV and Fusion Inhibitors Targeting HR1 Domain in Spike Protein. Cell Mol. Immunol. 2020, 17, 765–767. [Google Scholar] [CrossRef]
- Fung, T.S.; Liu, D.X. Post-Translational Modifications of Coronavirus Proteins: Roles and Function. Future Virol. 2018, 13, 405–430. [Google Scholar] [CrossRef] [Green Version]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L.; et al. A Pneumonia Outbreak Associated with a New Coronavirus of Probable Bat Origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Zheng, W.; Huang, X.; Bell, E.W.; Zhou, X.; Zhang, Y. Protein Structure and Sequence Reanalysis of 2019-NCoV Genome Refutes Snakes as Its Intermediate Host and the Unique Similarity between Its Spike Protein Insertions and HIV-1. J. Proteome Res. 2020, 19, 1351–1360. [Google Scholar] [CrossRef] [Green Version]
- Zhang, T.; Wu, Q.; Zhang, Z. Probable Pangolin Origin of SARS-CoV-2 Associated with the COVID-19 Outbreak. Curr. Biol. 2020, 30, 1346–1351.e2. [Google Scholar] [CrossRef]
- Guruprasad, L. Human SARS-CoV-2 Spike Protein Mutations. Proteins: Struct. Funct. Bioinform. 2021, 89, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhang, Y.; Wu, L.; Niu, S.; Song, C.; Zhang, Z.; Lu, G.; Qiao, C.; Hu, Y.; Yuen, K.-Y.; et al. Structural and Functional Basis of SARS-CoV-2 Entry by Using Human ACE2. Cell 2020, 181, 894–904.e9. [Google Scholar] [CrossRef] [PubMed]
- Li, F. Structure, Function, and Evolution of Coronavirus Spike Proteins. Annu. Rev. Virol. 2016, 3, 237–261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weissenhorn, W.; Dessen, A.; Calder, L.J.; Harrison, S.C.; Skehel, J.J.; Wiley, D.C. Structural Basis for Membrane Fusion by Enveloped Viruses. Mol. Membr. Biol. 1999, 16, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Balzanelli, M.G.; Distratis, P.; Lazzaro, R.; Cefalo, A.; Catucci, O.; Aityan, S.K.; Dipalma, G.; Vimercati, L.; Inchingolo, A.D.; Maggiore, M.E.; et al. The Vitamin D, IL-6 and the EGFR Markers a Possible Way to Elucidate the Lung–Heart–Kidney Cross-Talk in COVID-19 Disease: A Foregone Conclusion. Microorganisms 2021, 9, 1903. [Google Scholar] [CrossRef]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.-L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM Structure of the 2019-NCoV Spike in the Prefusion Conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.-H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280.e8. [Google Scholar] [CrossRef]
- Tay, M.Z.; Poh, C.M.; Rénia, L.; MacAry, P.A.; Ng, L.F.P. The Trinity of COVID-19: Immunity, Inflammation and Intervention. Nat Rev Immunol 2020, 20, 363–374. [Google Scholar] [CrossRef]
- Millet, J.K.; Whittaker, G.R. Host Cell Proteases: Critical Determinants of Coronavirus Tropism and Pathogenesis. Virus Res. 2015, 202, 120–134. [Google Scholar] [CrossRef]
- Walls, A.C.; Park, Y.-J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020, 181, 281–292.e6. [Google Scholar] [CrossRef]
- Matsuyama, S.; Nagata, N.; Shirato, K.; Kawase, M.; Takeda, M.; Taguchi, F. Efficient Activation of the Severe Acute Respiratory Syndrome Coronavirus Spike Protein by the Transmembrane Protease TMPRSS2. J. Virol. 2010, 84, 12658–12664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eckert, D.M.; Kim, P.S. Mechanisms of Viral Membrane Fusion and Its Inhibition. Annu. Rev. Biochem. 2001, 70, 777–810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Masters, P.S. The Molecular Biology of Coronaviruses. In Advances in Virus Research; Elsevier: Amsterdam, The Netherlands, 2006; Volume 66, pp. 193–292. ISBN 978-0-12-039869-0. [Google Scholar]
- Hussain, M.; Jabeen, N.; Raza, F.; Shabbir, S.; Baig, A.A.; Amanullah, A.; Aziz, B. Structural Variations in Human ACE2 May Influence Its Binding with SARS-CoV-2 Spike Protein. J. Med. Virol. 2020, 92, 1580–1586. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, W.; Moore, M.J.; Vasilieva, N.; Sui, J.; Wong, S.K.; Berne, M.A.; Somasundaran, M.; Sullivan, J.L.; Luzuriaga, K.; Greenough, T.C.; et al. Angiotensin-Converting Enzyme 2 Is a Functional Receptor for the SARS Coronavirus. Nature 2003, 426, 450–454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuba, K.; Imai, Y.; Ohto-Nakanishi, T.; Penninger, J.M. Trilogy of ACE2: A Peptidase in the Renin–Angiotensin System, a SARS Receptor, and a Partner for Amino Acid Transporters. Pharmacol. Ther. 2010, 128, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Verdecchia, P.; Cavallini, C.; Spanevello, A.; Angeli, F. The Pivotal Link between ACE2 Deficiency and SARS-CoV-2 Infection. Eur. J. Intern. Med. 2020, 76, 14–20. [Google Scholar] [CrossRef]
- Tetz, G.; Tetz, V. SARS-CoV-2 Prion-Like Domains in Spike Proteins Enable Higher Affinity to ACE2. Microorganisms 2020, 10, 280. [Google Scholar] [CrossRef]
- Buzhdygan, T.P.; DeOre, B.J.; Baldwin-Leclair, A.; Bullock, T.A.; McGary, H.M.; Khan, J.A.; Razmpour, R.; Hale, J.F.; Galie, P.A.; Potula, R.; et al. The SARS-CoV-2 Spike Protein Alters Barrier Function in 2D Static and 3D Microfluidic in-Vitro Models of the Human Blood–Brain Barrier. Neurobiol. Dis. 2020, 146, 105131. [Google Scholar] [CrossRef]
- Daneman, R.; Prat, A. The Blood–Brain Barrier. Cold Spring Harb. Perspect. Biol. 2015, 7, a020412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daniels, B.P.; Holman, D.W.; Cruz-Orengo, L.; Jujjavarapu, H.; Durrant, D.M.; Klein, R.S. Viral Pathogen-Associated Molecular Patterns Regulate Blood-Brain Barrier Integrity via Competing Innate Cytokine Signals. mBio 2014, 5, e01476-14. [Google Scholar] [CrossRef] [Green Version]
- Grifoni, A.; Weiskopf, D.; Ramirez, S.I.; Mateus, J.; Dan, J.M.; Moderbacher, C.R.; Rawlings, S.A.; Sutherland, A.; Premkumar, L.; Jadi, R.S.; et al. Targets of T Cell Responses to SARS-CoV-2 Coronavirus in Humans with COVID-19 Disease and Unexposed Individuals. Cell 2020, 181, 1489–1501.e15. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Su, B.; Guo, X.; Sun, W.; Deng, Y.; Bao, L.; Zhu, Q.; Zhang, X.; Zheng, Y.; Geng, C.; et al. Potent Neutralizing Antibodies against SARS-CoV-2 Identified by High-Throughput Single-Cell Sequencing of Convalescent Patients’ B Cells. Cell 2020, 182, 73–84.e16. [Google Scholar] [CrossRef] [PubMed]
- Ju, B.; Zhang, Q.; Ge, J.; Wang, R.; Sun, J.; Ge, X.; Yu, J.; Shan, S.; Zhou, B.; Song, S.; et al. Human Neutralizing Antibodies Elicited by SARS-CoV-2 Infection. Nature 2020, 584, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Wang, P.; Nair, M.S.; Yu, J.; Rapp, M.; Wang, Q.; Luo, Y.; Chan, J.F.-W.; Sahi, V.; Figueroa, A.; et al. Potent Neutralizing Antibodies Directed to Multiple Epitopes on SARS-CoV-2 Spike. Microbiology 2020, 584, 450–456. [Google Scholar]
- Brouwer, P.J.M.; Caniels, T.G.; van der Straten, K.; Snitselaar, J.L.; Aldon, Y.; Bangaru, S.; Torres, J.L.; Okba, N.M.A.; Claireaux, M.; Kerster, G.; et al. Potent Neutralizing Antibodies from COVID-19 Patients Define Multiple Targets of Vulnerability. Science 2020, 369, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Shen, J.; Dai, E.; Li, H.; Tang, G.; Zhang, L.; Hou, X.; Lu, M.; Wu, X.; Duan, S.; et al. The Seroprevalence and Kinetics of IgM and IgG in the Progression of COVID-19. BMC Immunol. 2021, 22, 14. [Google Scholar] [CrossRef]
- Attolico, I.; Tarantini, F.; Carluccio, P.; Schifone, C.P.; Delia, M.; Gagliardi, V.P.; Perrone, T.; Gaudio, F.; Longo, C.; Giordano, A.; et al. Serological Response Following BNT162b2 Anti-SARS-CoV-2 MRNA Vaccination in Haematopoietic Stem Cell Transplantation Patients. Br. J. Haematol. 2021. [Google Scholar] [CrossRef]
- Qu, J.; Wu, C.; Li, X.; Zhang, G.; Jiang, Z.; Li, X.; Zhu, Q.; Liu, L. Profile of Immunoglobulin G and IgM Antibodies Against Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Clin. Infect. Dis. 2020, 71, 2255–2258. [Google Scholar] [CrossRef]
- Vimercati, L.; Stefanizzi, P.; De Maria, L.; Caputi, A.; Cavone, D.; Quarato, M.; Gesualdo, L.; Lopalco, P.L.; Migliore, G.; Sponselli, S.; et al. Large-Scale IgM and IgG SARS-CoV-2 Serological Screening among Healthcare Workers with a Low Infection Prevalence Based on Nasopharyngeal Swab Tests in an Italian University Hospital: Perspectives for Public Health. Environ. Res 2021, 195, 110793. [Google Scholar] [CrossRef]
- Nuccetelli, M.; Pieri, M.; Gisone, F.; Bernardini, S. Combined Anti-SARS-CoV-2 IgA, IgG, and IgM Detection as a Better Strategy to Prevent Second Infection Spreading Waves. Immunol. Investig. 2020, 51, 1–13. [Google Scholar] [CrossRef]
- Rhorer, J.; Ambrose, C.S.; Dickinson, S.; Hamilton, H.; Oleka, N.A.; Malinoski, F.J.; Wittes, J. Efficacy of Live Attenuated Influenza Vaccine in Children: A Meta-Analysis of Nine Randomized Clinical Trials. Vaccine 2009, 27, 1101–1110. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Wang, D.; Wang, H.; Zhang, X.; Liang, T.; Dai, J.; Li, M.; Zhang, J.; Zhang, K.; Xu, D.; et al. COVID-19 Diagnostic Testing: Technology Perspective. Clin. Transl. Med. 2020, 10. [Google Scholar] [CrossRef]
- Kowitdamrong, E.; Puthanakit, T.; Jantarabenjakul, W.; Prompetchara, E.; Suchartlikitwong, P.; Putcharoen, O.; Hirankarn, N. Antibody Responses to SARS-CoV-2 in Patients with Differing Severities of Coronavirus Disease 2019. PLoS ONE 2020, 15, e0240502. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Xu, Y.; Sun, C.; Wang, X.; Guo, Y.; Qiu, S.; Ma, K. A Systematic Review of Asymptomatic Infections with COVID-19. J. Microbiol. Immunol. Infect. 2021, 54, 12–16. [Google Scholar] [CrossRef]
- Fox, A.; Marino, J.; Amanat, F.; Krammer, F.; Hahn-Holbrook, J.; Zolla-Pazner, S.; Powell, R.L. Robust and Specific Secretory IgA Against SARS-CoV-2 Detected in Human Milk. iScience 2020, 23, 101735. [Google Scholar] [CrossRef] [PubMed]
- Mahmoodpoor, A.; Nader, N.D. Immune Responses to the Novel Coronavirus-2: Friend or Foe? Immunol. Investig. 2021, 50, 949–951. [Google Scholar] [CrossRef]
- Agarwal, A.; Chen, A.; Ravindran, N.; To, C.; Thuluvath, P.J. Gastrointestinal and Liver Manifestations of COVID-19. J. Clin. Exp. Hepatol. 2020, 10, 263–265. [Google Scholar] [CrossRef] [PubMed]
- Ballini, A.; Dipalma, G.; Isacco, C.G.; Boccellino, M.; Di Domenico, M.; Santacroce, L.; Nguyễn, K.C.D.; Scacco, S.; Calvani, M.; Boddi, A.; et al. Oral Microbiota and Immune System Crosstalk: A Translational Research. Biology 2020, 9, 131. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Ren, L.; Yang, S.; Xiao, M.; Chang, D.; Yang, F.; Dela Cruz, C.S.; Wang, Y.; Wu, C.; Xiao, Y.; et al. Profiling Early Humoral Response to Diagnose Novel Coronavirus Disease (COVID-19). Clin. Infect. Dis. 2020, 71, 778–785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tetz, G.; Tetz, V. Prion-Like Domains in Spike Protein of SARS-CoV-2 Differ across Its Variants and Enable Changes in Affinity to ACE2. Microorganisms 2022, 10, 280. [Google Scholar] [CrossRef] [PubMed]
- Ballini, A.; Santacroce, L.; Cantore, S.; Bottalico, L.; Dipalma, G.; Vito, D.D.; Saini, R.; Inchingolo, F. Probiotics Improve Urogenital Health in Women. Open Access Maced. J. Med. Sci. 2018, 6, 1845–1850. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Padoan, A.; Sciacovelli, L.; Basso, D.; Negrini, D.; Zuin, S.; Cosma, C.; Faggian, D.; Matricardi, P.; Plebani, M. IgA-Ab Response to Spike Glycoprotein of SARS-CoV-2 in Patients with COVID-19: A Longitudinal Study. Clin. Chim. Acta 2020, 507, 164–166. [Google Scholar] [CrossRef]
- Padoan, A.; Cosma, C.; Sciacovelli, L.; Faggian, D.; Plebani, M. Analytical Performances of a Chemiluminescence Immunoassay for SARS-CoV-2 IgM/IgG and Antibody Kinetics. Clin. Chem. Lab. Med. (CCLM) 2020, 58, 1081–1088. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, L.; Zhao, J.; Peng, J.; Li, X.; Deng, X.; Geng, Z.; Shen, Z.; Guo, F.; Zhang, Q.; Jin, Y.; et al. Detection of SARS-CoV-2 in Saliva and Characterization of Oral Symptoms in COVID-19 Patients. Cell Prolif. 2020, 53. [Google Scholar] [CrossRef]
- Rapone, B.; Ferrara, E.; Santacroce, L.; Topi, S.; Gnoni, A.; Dipalma, G.; Mancini, A.; Di Domenico, M.; Tartaglia, G.M.; Scarano, A. The Gaseous Ozone Therapy as a Promising Antiseptic Adjuvant of Periodontal Treatment: A Randomized Controlled Clinical Trial. Int. J. Environ. Res. Public Health 2022, 19, 985. [Google Scholar] [CrossRef] [PubMed]
- Phan, D.Q.; Nguyen, L.D.N.; Pham, S.T.; Nguyen, T.; Pham, P.T.T.; Nguyen, S.T.H.; Pham, D.T.; Pham, H.T.; Tran, D.K.; Le, S.H.; et al. The Distribution of Dengue Virus Serotype in Quang Nam Province (Vietnam) during the Outbreak in 2018. Int. J. Environ. Res. Public Health 2022, 19, 1285. [Google Scholar] [CrossRef] [PubMed]
- Indonesia First to Greenlight Novavax COVID-19 Vaccine | AP News. Available online: https://apnews.com/article/coronavirus-pandemic-technology-science-business-health-7e75808d4675e0e7597f52aeed1b8d5a (accessed on 12 July 2022).
- Algaissi, A.; Alfaleh, M.A.; Hala, S.; Abujamel, T.S.; Alamri, S.S.; Almahboub, S.A.; Alluhaybi, K.A.; Hobani, H.I.; Alsulaiman, R.M.; AlHarbi, R.H.; et al. SARS-CoV-2 S1 and N-Based Serological Assays Reveal Rapid Seroconversion and Induction of Specific Antibody Response in COVID-19 Patients. Sci. Rep. 2020, 10, 16561. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.-W.; Schmitz, J.E.; Persing, D.H.; Stratton, C.W. Laboratory Diagnosis of COVID-19: Current Issues and Challenges. J. Clin. Microbiol. 2020, 58, e00512-20. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Yi, Y.; Luo, X.; Xiong, N.; Liu, Y.; Li, S.; Sun, R.; Wang, Y.; Hu, B.; Chen, W.; et al. Development and Clinical Application of a Rapid IgM-IgG Combined Antibody Test for SARS-CoV-2 Infection Diagnosis. J. Med. Virol. 2020, 92, 1518–1524. [Google Scholar] [CrossRef]
- Adeyinka, A.; Bailey, K.; Pierre, L.; Kondamudi, N. COVID 19 Infection: Pediatric Perspectives. J. Am. Coll. Emerg. Physicians Open 2021, 2. [Google Scholar] [CrossRef]
- Patel, A.; Jernigan, D.B.; Abdirizak, F.; Abedi, G.; Aggarwal, S.; Albina, D.; Allen, E.; Andersen, L.; Anderson, J.; Anderson, M. Initial Public Health Response and Interim Clinical Guidance for the 2019 Novel Coronavirus Outbreak—United States, December 31, 2019–February 4, 2020. Morb. Mortal. Wkly. Rep. 2020, 69, 140. [Google Scholar] [CrossRef] [PubMed]
- Bellocchio, L.; Inchingolo, A.D.; Inchingolo, A.M.; Lorusso, F.; Malcangi, G.; Santacroce, L.; Scarano, A.; Bordea, I.R.; Hazballa, D.; D’Oria, M.T.; et al. Cannabinoids Drugs and Oral Health-From Recreational Side-Effects to Medicinal Purposes: A Systematic Review. Int. J. Mol. Sci. 2021, 22, 8329. [Google Scholar] [CrossRef] [PubMed]
- Taleghani, N.; Taghipour, F. Diagnosis of COVID-19 for Controlling the Pandemic: A Review of the State-of-the-Art. Biosens. Bioelectron. 2021, 174, 112830. [Google Scholar] [CrossRef] [PubMed]
- Ejazi, S.A.; Ghosh, S.; Ali, N. Antibody Detection Assays for COVID-19 Diagnosis: An Early Overview. Immunol. Cell Biol. 2021, 99, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Higgins, V.; Fabros, A.; Kulasingam, V. Quantitative Measurement of Anti-SARS-CoV-2 Antibodies: Analytical and Clinical Evaluation. J. Clin. Microbiol. 2021, 59, e03149-20. [Google Scholar] [CrossRef] [PubMed]
- Rokni, M.; Ghasemi, V.; Tavakoli, Z. Immune Responses and Pathogenesis of SARS-CoV-2 during an Outbreak in Iran: Comparison with SARS and MERS. Rev. Med. Virol. 2020, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tantuoyir, M.M.; Rezaei, N. Serological Tests for COVID-19: Potential Opportunities. Cell Biol. Int. 2021, 45, 740–748. [Google Scholar] [CrossRef] [PubMed]
- Werner, M.; Pervan, P.; Glück, V.; Zeman, F.; Koller, M.; Burkhardt, R.; Glück, T.; Wenzel, J.J.; Schmidt, B.; Gessner, A. Evaluation of a Broad Panel of SARS-CoV-2 Serological Tests for Diagnostic Use. J. Clin. Med. 2021, 10, 1580. [Google Scholar] [CrossRef]
- Sun, B.; Feng, Y.; Mo, X.; Zheng, P.; Wang, Q.; Li, P.; Peng, P.; Liu, X.; Chen, Z.; Huang, H. Kinetics of SARS-CoV-2 Specific IgM and IgG Responses in COVID-19 Patients. Emerg. Microbes Infect. 2020, 9, 940–948. [Google Scholar] [CrossRef]
- Kyosei, Y.; Namba, M.; Makioka, D.; Kokubun, A.; Watabe, S.; Yoshimura, T.; Sasaki, T.; Shioda, T.; Ito, E. Ultrasensitive Detection of SARS-CoV-2 Spike Proteins Using the Thio-NAD Cycling Reaction: A Preliminary Study before Clinical Trials. Microorganisms 2021, 9, 2214. [Google Scholar] [CrossRef]
- Okba, N.M.; Müller, M.A.; Li, W.; Wang, C.; GeurtsvanKessel, C.H.; Corman, V.M.; Lamers, M.M.; Sikkema, R.S.; de Bruin, E.; Chandler, F.D. Severe Acute Respiratory Syndrome Coronavirus 2- Specific Antibody Responses in Coronavirus Disease Patients. Emerg. Infect. Dis. 2020, 26, 1478–1488. [Google Scholar] [CrossRef] [PubMed]
- Oguntuyo, K.Y.; Stevens, C.S.; Hung, C.-T.; Ikegame, S.; Acklin, J.A.; Kowdle, S.S.; Carmichael, J.C.; Chiu, H.-P.; Azarm, K.D.; Haas, G.D.; et al. Quantifying Absolute Neutralization Titers against SARS-CoV-2 by a Standardized Virus Neutralization Assay Allows for Cross-Cohort Comparisons of COVID-19 Sera. Infect. Dis. 2020, 12, e02492-20. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Fan, Y.; Lai, Y.; Han, T.; Li, Z.; Zhou, P.; Pan, P.; Wang, W.; Hu, D.; Liu, X.; et al. Coronavirus Infections and Immune Responses. J. Med. Virol. 2020, 92, 424–432. [Google Scholar] [CrossRef] [PubMed]
- Sarvaria, A.; Madrigal, J.A.; Saudemont, A. B Cell Regulation in Cancer and Anti-Tumor Immunity. Cell Mol. Immunol. 2017, 14, 662–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atri, C.; Guerfali, F.; Laouini, D. Role of Human Macrophage Polarization in Inflammation during Infectious Diseases. IJMS 2018, 19, 1801. [Google Scholar] [CrossRef] [Green Version]
- SeyedAlinaghi, S.; Mehrtak, M.; MohsseniPour, M.; Mirzapour, P.; Barzegary, A.; Habibi, P.; Moradmand-Badie, B.; Afsahi, A.M.; Karimi, A.; Heydari, M.; et al. Genetic Susceptibility of COVID-19: A Systematic Review of Current Evidence. Eur. J. Med. Res. 2021, 26, 46. [Google Scholar] [CrossRef]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider Cytokine Storm Syndromes and Immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Sang, E.R.; Tian, Y.; Miller, L.C.; Sang, Y. Epigenetic Evolution of ACE2 and IL-6 Genes: Non-Canonical Interferon-Stimulated Genes Correlate to COVID-19 Susceptibility in Vertebrates. Genes 2021, 12, 154. [Google Scholar] [CrossRef]
- Murakami, M.; Kamimura, D.; Hirano, T. Pleiotropy and Specificity: Insights from the Interleukin 6 Family of Cytokines. Immunity 2019, 50, 812–831. [Google Scholar] [CrossRef] [Green Version]
- Garbers, C.; Heink, S.; Korn, T.; Rose-John, S. Interleukin-6: Designing Specific Therapeutics for a Complex Cytokine. Nat. Rev. Drug Discov. 2018, 17, 395–412. [Google Scholar] [CrossRef]
- Morelli, C.; Francavilla, M.; Stabile Ianora, A.A.; Cozzolino, M.; Gualano, A.; Stellacci, G.; Sacco, A.; Lorusso, F.; Pedote, P.; De Ceglie, M. The Multifaceted COVID-19: CT Aspects of Its Atypical Pulmonary and Abdominal Manifestations and Complications in Adults and Children. A Pictorial Review. Microorganisms 2021, 9, 2037. [Google Scholar] [CrossRef] [PubMed]
- Bhimraj, A.; Morgan, R.L.; Shumaker, A.H.; Lavergne, V.; Baden, L.; Cheng, V.C.-C.; Edwards, K.M.; Gandhi, R.; Muller, W.J.; O’Horo, J.C.; et al. Infectious Diseases Society of America Guidelines on the Treatment and Management of Patients With Coronavirus Disease 2019 (COVID-19). Clin. Infect. Dis. 2020, ciaa478. [Google Scholar] [CrossRef] [PubMed]
- Balzanelli, G.M.; Distratis, P.; Aityan, S.K.; Amatulli, F.; Catucci, O.; Cefalo, A.; Dipalma, G.; Inchingolo, F.; Lazzaro, R.; Nguyen, K.C.D.; et al. COVID-19 and COVID-like Patients: A Brief Analysis and Findings of Two Deceased Cases. Open Access Maced. J. Med. Sci. 2020, 8, 490–495. [Google Scholar] [CrossRef]
- Nguyen, K.C. Could Immunity Boosting Therapy Help COVID-19 Patient? BJSTR 2020, 30, 23149–23150. [Google Scholar] [CrossRef]
- Asano, T.; Boisson, B.; Onodi, F.; Matuozzo, D.; Moncada-Velez, M.; Maglorius Renkilaraj, M.R.L.; Zhang, P.; Meertens, L.; Bolze, A.; Materna, M.; et al. X-Linked Recessive TLR7 Deficiency in ~1% of Men under 60 Years Old with Life-Threatening COVID-19. Sci. Immunol. 2021, 6, eabl4348. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Petrini, M.; Mastrangelo, F.; Noumbissi, S.; Lorusso, F. The Effects of Liquid Disinfection and Heat Sterilization Processes on Implant Drill Roughness: Energy Dispersion X-Ray Microanalysis and Infrared Thermography. J. Clin. Med. 2020, 9, 1019. [Google Scholar] [CrossRef] [Green Version]
- Inchingolo, A.D.; Malcangi, G.; Inchingolo, A.M.; Piras, F.; Settanni, V.; Garofoli, G.; Palmieri, G.; Ceci, S.; Patano, A.; De Leonardis, N.; et al. Benefits and Implications of Resveratrol Supplementation on Microbiota Modulations: A Systematic Review of the Literature. Int. J. Mol. Sci. 2022, 23, 4027. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Bastard, P.; Liu, Z.; Le Pen, J.; Moncada-Velez, M.; Chen, J.; Ogishi, M.; Sabli, I.K.D.; Hodeib, S.; Korol, C.; et al. Inborn Errors of Type I IFN Immunity in Patients with Life-Threatening COVID-19. Science 2020, 370, eabd4570. [Google Scholar] [CrossRef] [PubMed]
- Balzanelli, G.M.; Distratis, P.; Amatulli, F.; Catucci, O.; Cefalo, A.; Lazzaro, R.; Palazzo, D.; Aityan, K.S.; Dipalma, G.; Inchingolo, F. Clinical Features in Predicting COVID-19. Biomed. J. Sci. Tech. Res. 2020, 29, 22921–22926. [Google Scholar]
- Balzanelli, M.G.; Distratis, P.; Lazzaro, R.; Cefalo, A.; Dangela, G.; Catucci, O.; Palazzo, D.; Amatulli, F.; Tomassone, D.; Pham, V.; et al. Would the End Of COVID-19 Infection as a Chronic Disease? J. Stem Cells Res. Dev. Ther. 2020. [Google Scholar] [CrossRef]
- Balzanelli, M.G.; Distratis, P.; Aityan, S.K.; Amatulli, F.; Catucci, O.; Cefalo, A.; De Michele, A.; Dipalma, G.; Inchingolo, F.; Lazzaro, R.; et al. An Alternative “Trojan Horse” Hypothesis for COVID-19: Immune Deficiency of IL-10 and SARS-CoV-2 Biology. EMIDDT 2021, 22, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Bevilacqua, V.; Altini, N.; Prencipe, B.; Brunetti, A.; Villani, L.; Sacco, A.; Morelli, C.; Ciaccia, M.; Scardapane, A. Lung Segmentation and Characterization in COVID-19 Patients for Assessing Pulmonary Thromboembolism: An Approach Based on Deep Learning and Radiomics. Electronics 2021, 10, 2475. [Google Scholar] [CrossRef]
- Bavaro, D.F.; Poliseno, M.; Scardapane, A.; Belati, A.; De Gennaro, N.; Stabile Ianora, A.A.; Angarano, G.; Saracino, A. Occurrence of Acute Pulmonary Embolism in COVID-19-A Case Series. Int. J. Infect. Dis. 2020, 98, 225–226. [Google Scholar] [CrossRef] [PubMed]
- Scardapane, A.; Villani, L.; Bavaro, D.F.; Passerini, F.; Ianora, A.A.S.; Lucarelli, N.M.; Angarano, G.; Portincasa, P.; Palmieri, V.O.; Saracino, A. Pulmonary Artery Filling Defects in COVID-19 Patients Revealed Using CT Pulmonary Angiography: A Predictable Complication? Biomed. Res. Int. 2021, 2021, 8851736. [Google Scholar] [CrossRef]
- Spencer, J.P.; Trondsen Pawlowski, R.H.; Thomas, S. Vaccine Adverse Events: Separating Myth from Reality. Am. Fam. Physician 2017, 95, 786–794. [Google Scholar] [PubMed]
- Krammer, F. SARS-CoV-2 Vaccines in Development. Nature 2020, 586, 516–527. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. FDA Approves First COVID-19 Vaccine. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-first-covid-19-vaccine (accessed on 12 July 2022).
- EMA First COVID-19 Vaccine Approved for Children Aged 12 to 15 in EU. Available online: https://www.ema.europa.eu/en/news/first-covid-19-vaccine-approved-children-aged-12-15-eu (accessed on 12 July 2022).
- Dai, L.; Gao, G.F. Viral Targets for Vaccines against COVID-19. Nat. Rev. Immunol. 2021, 21, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Gobbi, F.; Buonfrate, D.; Moro, L.; Rodari, P.; Piubelli, C.; Caldrer, S.; Riccetti, S.; Sinigaglia, A.; Barzon, L. Antibody Response to the BNT162b2 MRNA COVID-19 Vaccine in Subjects with Prior SARS-CoV-2 Infection. Viruses 2021, 13, 422. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, F.P.; Germinario, C.A.; Migliore, G.; Vimercati, L.; Martinelli, A.; Lobifaro, A.; Tafuri, S.; Stefanizzi, P.; Control Room Working Group. BNT162b2 MRNA COVID-19 Vaccine Effectiveness in the Prevention of SARS-CoV-2 Infection: A Preliminary Report. J. Infect. Dis. 2021, 224, 431–434. [Google Scholar] [CrossRef]
- Greenwood, B. The Contribution of Vaccination to Global Health: Past, Present and Future. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130433. [Google Scholar] [CrossRef] [Green Version]
- Lurie, N.; Saville, M.; Hatchett, R.; Halton, J. Developing COVID-19 Vaccines at Pandemic Speed. N. Engl. J. Med. 2020, 382, 1969–1973. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.Y.; Thone, M.N.; Kwon, Y.J. COVID-19 Vaccines: The Status and Perspectives in Delivery Points of View. Adv. Drug Deliv. Rev. 2021, 170, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Mallajosyula, V.; Tato, C.M.; Tan, G.S.; Wang, T.T. SARS-CoV-2 Vaccines in Advanced Clinical Trials: Where Do We Stand? Adv. Drug Deliv. Rev. 2021, 172, 314–338. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, Y.; Huang, B.; Deng, W.; Quan, Y.; Wang, W.; Xu, W.; Zhao, Y.; Li, N.; Zhang, J. Development of an Inactivated Vaccine Candidate, BBIBP-CorV, with Potent Protection against SARS-CoV-2. Cell 2020, 182, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Duan, K.; Zhang, Y.; Zhao, D.; Zhang, H.; Xie, Z.; Li, X.; Peng, C.; Zhang, Y.; Zhang, W. Effect of an Inactivated Vaccine against SARS-CoV-2 on Safety and Immunogenicity Outcomes: Interim Analysis of 2 Randomized Clinical Trials. JAMA 2020, 324, 951–960. [Google Scholar] [CrossRef]
- Gao, Q.; Bao, L.; Mao, H.; Wang, L.; Xu, K.; Yang, M.; Li, Y.; Zhu, L.; Wang, N.; Lv, Z.; et al. Development of an Inactivated Vaccine Candidate for SARS-CoV-2. Science 2020, 369, 77–81. [Google Scholar] [CrossRef]
- Palacios, R.; Patiño, E.G.; de Oliveira Piorelli, R.; Conde, M.T.R.P.; Batista, A.P.; Zeng, G.; Xin, Q.; Kallas, E.G.; Flores, J.; Ockenhouse, C.F.; et al. Double-Blind, Randomized, Placebo-Controlled Phase III Clinical Trial to Evaluate the Efficacy and Safety of Treating Healthcare Professionals with the Adsorbed COVID-19 (Inactivated) Vaccine Manufactured by Sinovac—PROFISCOV: A Structured Summary of a Study Protocol for a Randomised Controlled Trial. Trials 2020, 21, 853. [Google Scholar] [CrossRef]
- Mallapaty, S. China COVID Vaccine Reports Mixed Results—What Does That Mean for the Pandemic? Nature 2021, 15. [Google Scholar] [CrossRef]
- Prevention, B.V. A Randomized, Double-Blind, Placebo-Controlled Phase 3 Study to Assess the Efficacy and Safety of Ad26. COV2. S for the Prevention of SARS-CoV-2-Mediated COVID-19 in Adults Aged 18 Years and Older. 2020; in press. [Google Scholar]
- Sheikh, A.B.; Pal, S.; Javed, N.; Shekhar, R. COVID-19 Vaccination in Developing Nations: Challenges and Opportunities for Innovation. Infect. Dis. Rep. 2021, 13, 429–436. [Google Scholar] [CrossRef]
- Logunov, D.Y.; Dolzhikova, I.V.; Shcheblyakov, D.V.; Tukhvatulin, A.I.; Zubkova, O.V.; Dzharullaeva, A.S.; Kovyrshina, A.V.; Lubenets, N.L.; Grousova, D.M.; Erokhova, A.S.; et al. Safety and Efficacy of an RAd26 and RAd5 Vector-Based Heterologous Prime-Boost COVID-19 Vaccine: An Interim Analysis of a Randomised Controlled Phase 3 Trial in Russia. Lancet 2021, 397, 671–681. [Google Scholar] [CrossRef]
- Bangaru, S.; Ozorowski, G.; Turner, H.L.; Antanasijevic, A.; Huang, D.; Wang, X.; Torres, J.L.; Diedrich, J.K.; Tian, J.-H.; Portnoff, A.D.; et al. Structural Analysis of Full-Length SARS-CoV-2 Spike Protein from an Advanced Vaccine Candidate. Science 2020, 370, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- BioNTech SE A PHASE 1/2/3, Placebo-Controlled, Randomized, Observer-Blind, Dose-Finding Study to Evaluate the Safety, Tolerability, Immunogenicity, and Efficacy of SARS-CoV-2 RNA Vaccine Candidates against COVID-19 in Healthy Indi-viduals. Available online: clinicaltrials.gov (accessed on 12 July 2022).
- Mahase, E. COVID-19: Moderna Vaccine Is Nearly 95% Effective, Trial Involving High Risk and Elderly People Shows. BMJ Br. Med. J. (Online) 2020, 371. [Google Scholar] [CrossRef]
- Mahase, E. COVID-19: UK Government Asks Regulator to Assess Oxford Vaccine as Questions Are Raised over Interim Data. BMJ Br. Med. J. (Online) 2020, 371, m4670. [Google Scholar] [CrossRef] [PubMed]
- EMA EMA and ECDC Update on COVID-19. Available online: https://www.ema.europa.eu/en/news/ema-ecdc-update-covid-19 (accessed on 12 July 2022).
- National Cohort Study of Effectiveness and Safety of SARS-CoV-2/COVID-19 Vaccines. Available online: clinicaltrials.gov (accessed on 12 July 2022).
- Fabiani, M.; Ramigni, M.; Gobbetto, V.; Mateo-Urdiales, A.; Pezzotti, P.; Piovesan, C. Effectiveness of the Comirnaty (BNT162b2, BioNTech/Pfizer) Vaccine in Preventing SARS-CoV-2 Infection among Healthcare Workers, Treviso Province, Veneto Region, Italy, 27 December 2020 to 24 March 2021. Eurosurveillance 2021, 26, 2100420. [Google Scholar] [CrossRef]
- BioNTech/Pfizer Vaccine Authorised Answers to Frequently Asked Questions on AIFA’s Website. Available online: https://www.aifa.gov.it/en/-/autorizzato-il-vaccino-biontech-pfizer (accessed on 12 July 2022).
- Abbasi, J. COVID-19 and MRNA Vaccines—First Large Test for a New Approach. JAMA 2020, 324, 1125. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.ema.europa.eu/en/medicines/human/epar/comirnaty (accessed on 12 July 2022).
- Development of Therapeutics and Vaccines. Available online: https://www.eu-patient.eu/COVID-19/covid-resource-point/therapeutics-and-vaccines/ (accessed on 12 July 2022).
- Hou, X.; Zaks, T.; Langer, R.; Dong, Y. Lipid Nanoparticles for MRNA Delivery. Nat. Rev. Mater. 2021, 6, 1078–1094. [Google Scholar] [CrossRef]
- Lamb, Y.N. BNT162b2 MRNA COVID-19 Vaccine: First Approval. Drugs 2021, 81, 495–501. [Google Scholar] [CrossRef]
- Frenck, R.W.; Klein, N.P.; Kitchin, N.; Gurtman, A.; Absalon, J.; Lockhart, S.; Perez, J.L.; Walter, E.B.; Senders, S.; Bailey, R.; et al. Safety, Immunogenicity, and Efficacy of the BNT162b2 COVID-19 Vaccine in Adolescents. N. Engl. J. Med. 2021, 385, 239–250. [Google Scholar] [CrossRef]
- Sahin, U.; Muik, A.; Vogler, I.; Derhovanessian, E.; Kranz, L.M.; Vormehr, M.; Quandt, J.; Bidmon, N.; Ulges, A.; Baum, A.; et al. BNT162b2 Induces SARS-CoV-2-Neutralising Antibodies and T Cells in Humans; Infectious Diseases (except HIV/AIDS). medRxiv 2020. [Google Scholar] [CrossRef]
- Dagan, N.; Barda, N.; Kepten, E.; Miron, O.; Perchik, S.; Katz, M.A.; Hernán, M.A.; Lipsitch, M.; Reis, B.; Balicer, R.D. BNT162b2 MRNA COVID-19 Vaccine in a Nationwide Mass Vaccination Setting. N. Engl. J. Med. 2021, 384, 1412–1423. [Google Scholar] [CrossRef]
- Thompson, M.G.; Burgess, J.L.; Naleway, A.L.; Tyner, H.L.; Yoon, S.K.; Meece, J.; Olsho, L.E.W.; Caban-Martinez, A.J.; Fowlkes, A.; Lutrick, K.; et al. Interim Estimates of Vaccine Effectiveness of BNT162b2 and MRNA-1273 COVID-19 Vaccines in Preventing SARS-CoV-2 Infection Among Health Care Personnel, First Responders, and Other Essential and Frontline Workers—Eight U.S. Locations, December 2020–March 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Patano, A.; Cirulli, N.; Beretta, M.; Plantamura, P.; Inchingolo, A.D.; Inchingolo, A.M.; Bordea, I.R.; Malcangi, G.; Marinelli, G.; Scarano, A.; et al. Education Technology in Orthodontics and Paediatric Dentistry during the COVID-19 Pandemic: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 6056. [Google Scholar] [CrossRef] [PubMed]
- Bordea, I.R.; Xhajanka, E.; Candrea, S.; Bran, S.; Onișor, F.; Inchingolo, A.D.; Malcangi, G.; Pham, V.H.; Inchingolo, A.M.; Scarano, A.; et al. Coronavirus (SARS-CoV-2) Pandemic: Future Challenges for Dental Practitioners. Microorganisms 2020, 8, 1704. [Google Scholar] [CrossRef] [PubMed]
- Swann, O.V.; Holden, K.A.; Turtle, L.; Pollock, L.; Fairfield, C.J.; Drake, T.M.; Seth, S.; Egan, C.; Hardwick, H.E.; Halpin, S.; et al. Clinical Characteristics of Children and Young People Admitted to Hospital with COVID-19 in United Kingdom: Prospective Multicentre Observational Cohort Study. BMJ 2020, 370, m3249. [Google Scholar] [CrossRef]
- Dohan Ehrenfest, D.M.; Del Corso, M.; Inchingolo, F.; Sammartino, G.; Charrier, J.-B. Platelet-Rich Plasma (PRP) and Platelet-Rich Fibrin (PRF) in Human Cell Cultures: Growth Factor Release and Contradictory Results. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2010, 110, 418–421, author reply 421–422. [Google Scholar] [CrossRef]
- King, H.; Deshpande, S.; Woodbridge, T.; Hilliard, T.; Standing, J.; Lewis, M.; Ward, L.; Finn, A.; Roderick, M. Initial Experience of the Safety and Tolerability of the BNT162b2 (Pfizer-Bio-N-Tech) Vaccine in Extremely Vulnerable Children Aged 12–15 Years. Arch. Dis. Child. 2022, 107, 205–207. [Google Scholar] [CrossRef]
- Callaway, E. What Pfizer’s Landmark COVID Vaccine Results Mean for the Pandemic. Nature 2020. [Google Scholar] [CrossRef]
- WHO. Vaccine Tracker and Landscape; WHO: Geneva, Switzerland, 2021. [Google Scholar]
- Draft Landscape of COVID-19 Candidate Vaccines. Available online: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines (accessed on 4 January 2021).
- Mulligan, M.J.; Lyke, K.E.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Raabe, V.; Bailey, R.; Swanson, K.A.; et al. Phase I/II Study of COVID-19 RNA Vaccine BNT162b1 in Adults. Nature 2020, 586, 589–593. [Google Scholar] [CrossRef]
- Pfizer, U.S. CDC Committee of Independent Health Experts Recommends Vaccination with Pfizer and Biontech COVID-19 Vaccine for Persons Ages 16 Years and Older 2020. Available online: https://www.pfizer.com/news/press-release/press-release-detail/us-cdc-committee-independent-health-experts-recommends (accessed on 4 January 2021).
- Liu, Y.; Liu, J.; Xia, H.; Zhang, X.; Fontes-Garfias, C.R.; Swanson, K.A.; Cai, H.; Sarkar, R.; Chen, W.; Cutler, M.; et al. Neutralizing Activity of BNT162b2-Elicited Serum. N. Engl. J. Med. 2021, 384, 1466–1468. [Google Scholar] [CrossRef]
- Barda, N.; Dagan, N.; Ben-Shlomo, Y.; Kepten, E.; Waxman, J.; Ohana, R.; Hernán, M.A.; Lipsitch, M.; Kohane, I.; Netzer, D.; et al. Safety of the BNT162b2 MRNA COVID-19 Vaccine in a Nationwide Setting. N. Engl. J. Med. 2021, 385, 1078–1090. [Google Scholar] [CrossRef]
- Oliver, S.E.; Gargano, J.W.; Marin, M.; Wallace, M.; Curran, K.G.; Chamberland, M.; McClung, N.; Campos-Outcalt, D.; Morgan, R.L.; Mbaeyi, S.; et al. The Advisory Committee on Immunization Practices’ Interim Recommendation for Use of Moderna COVID-19 Vaccine—United States, December 2020. MMWR Morb. Mortal. Wkly. Rep. 2021, 69, 1653–1656. [Google Scholar] [CrossRef] [PubMed]
- COVID-19 Vaccine Tracker and Landscape. Available online: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines (accessed on 12 July 2022).
- Fix, O.K.; Blumberg, E.A.; Chang, K.; Chu, J.; Chung, R.T.; Goacher, E.K.; Hameed, B.; Kaul, D.R.; Kulik, L.M.; Kwok, R.M.; et al. American Association for the Study of Liver Diseases Expert Panel Consensus Statement: Vaccines to Prevent Coronavirus Disease 2019 Infection in Patients With Liver Disease. Hepatology 2021, 74, 1049–1064. [Google Scholar] [CrossRef]
- COVID-19, Il Vaccino Astra Zeneca Efficace “per Errore” Univadis. Available online: https://www.univadis.it/viewarticle/covid-19-il-vaccino-astra-zeneca-efficace-per-errore-734126 (accessed on 4 January 2021).
- Emary, K.R.W.; Golubchik, T.; Aley, P.K.; Ariani, C.V.; Angus, B.; Bibi, S.; Blane, B.; Bonsall, D.; Cicconi, P.; Charlton, S.; et al. Efficacy of ChAdOx1 NCoV-19 (AZD1222) Vaccine against SARS-CoV-2 Variant of Concern 202012/01 (B.1.1.7): An Exploratory Analysis of a Randomised Controlled Trial. Lancet 2021, 397, 1351–1362. [Google Scholar] [CrossRef]
- PINHO, A.C. COVID-19 Vaccine AstraZeneca: PRAC Preliminary View Suggests No Specific Issue with Batch Used in Austria. Available online: https://www.ema.europa.eu/en/news/covid-19-vaccine-astrazeneca-prac-preliminary-view-suggests-no-specific-issue-batch-used-austria (accessed on 12 March 2021).
- AIFA Imposes Ban of Use of AstraZeneca Batch. Investigations in Progress in Coordination with EMA. Available online: https://www.aifa.gov.it/en/web/guest/-/aifa-dispone-divieto-di-utilizzo-di-un-lotto-astrazeneca-accertamenti-in-corso-in-coordinamento-con-ema (accessed on 12 July 2022).
- Bertelli, M.; Kiani, A.K.; Paolacci, S.; Manara, E.; Kurti, D.; Dhuli, K.; Bushati, V.; Miertus, J.; Pangallo, D.; Baglivo, M.; et al. Hydroxytyrosol: A Natural Compound with Promising Pharmacological Activities. J. Biotechnol. 2020, 309, 29–33. [Google Scholar] [CrossRef]
- Ergoren, M.C.; Paolacci, S.; Manara, E.; Dautaj, A.; Dhuli, K.; Anpilogov, K.; Camilleri, G.; Suer, H.K.; Sayan, M.; Tuncel, G.; et al. A Pilot Study on the Preventative Potential of Alpha-Cyclodextrin and Hydroxytyrosol against SARS-CoV-2 Transmission. Acta Bio Med. Atenei Parm. 2020, 91, e2020022. [Google Scholar] [CrossRef]
- Burgos, R.M.; Badowski, M.E.; Drwiega, E.; Ghassemi, S.; Griffith, N.; Herald, F.; Johnson, M.; Smith, R.O.; Michienzi, S.M. The Race to a COVID-19 Vaccine: Opportunities and Challenges in Development and Distribution. DIC 2021, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Haynes, B.F.; Corey, L.; Fernandes, P.; Gilbert, P.B.; Hotez, P.J.; Rao, S.; Santos, M.R.; Schuitemaker, H.; Watson, M.; Arvin, A. Prospects for a Safe COVID-19 Vaccine. Sci. Transl. Med. 2020, 12, eabe0948. [Google Scholar] [CrossRef]
- Commissioner, O. of the FDA Issues Emergency Use Authorization for Third COVID-19 Vaccine. Available online: https://www.fda.gov/news-events/press-announcements/fda-issues-emergency-use-authorization-third-covid-19-vaccine (accessed on 11 March 2021).
- Statement from NIH and BARDA on the FDA Emergency Use Authorization of the Janssen COVID-19 Vaccine|National Institutes of Health (NIH). Available online: https://www.nih.gov/news-events/news-releases/statement-nih-barda-fda-emergency-use-authorization-janssen-covid-19-vaccine (accessed on 2 March 2021).
- Ashraf, M.U.; Kim, Y.; Kumar, S.; Seo, D.; Ashraf, M.; Bae, Y.-S. COVID-19 Vaccines (Revisited) and Oral-Mucosal Vector System as a Potential Vaccine Platform. Vaccines 2021, 9, 171. [Google Scholar] [CrossRef]
- Zhang, H.; Kang, Z.; Gong, H.; Xu, D.; Wang, J.; Li, Z.; Li, Z.; Cui, X.; Xiao, J.; Zhan, J.; et al. Digestive System Is a Potential Route of COVID-19: An Analysis of Single-Cell Coexpression Pattern of Key Proteins in Viral Entry Process. Gut 2020, 69, 1010–1018. [Google Scholar] [CrossRef]
- Hou, Y.J.; Okuda, K.; Edwards, C.E.; Martinez, D.R.; Asakura, T.; Dinnon, K.H.; Kato, T.; Lee, R.E.; Yount, B.L.; Mascenik, T.M.; et al. SARS-CoV-2 Reverse Genetics Reveals a Variable Infection Gradient in the Respiratory Tract. Cell 2020, 182, 429–446.e14. [Google Scholar] [CrossRef]
- Sims, A.C.; Baric, R.S.; Yount, B.; Burkett, S.E.; Collins, P.L.; Pickles, R.J. Severe Acute Respiratory Syndrome Coronavirus Infection of Human Ciliated Airway Epithelia: Role of Ciliated Cells in Viral Spread in the Conducting Airways of the Lungs. J. Virol. 2005, 79, 15511–15524. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biopharma-Reporter.Com Vaxart’s Oral COVID-19 Tablet V—Cerca Con Google. Available online: https://www.google.it/search?q=biopharma-reporter.com+Vaxart%E2%80%99s+Oral+COVID-19+Tablet+V&ei=ribNYuvfOK2-xc8PsdSioAo&ved=0ahUKEwjr7orc7fL4AhUtX_EDHTGqCKQQ4dUDCA4&uact=5&oq=biopharma-reporter.com+Vaxart%E2%80%99s+Oral+COVID-19+Tablet+V&gs_lcp=Cgdnd3Mtd2l6EAMyBAghEBVKBAhBGAFKBAhGGABQzgdYzgdglgtoAXAAeACAAbcCiAG3ApIBAzMtMZgBAKABAqABAcABAQ&sclient=gws-wiz (accessed on 12 July 2022).
- Iwasaki, A.; Pillai, P.S. Innate Immunity to Influenza Virus Infection. Nat. Rev. Immunol. 2014, 14, 315–328. [Google Scholar] [CrossRef] [PubMed]
- iosBio Stabilitech Biopharma Announces Name Change to IosBio. Available online: https://www.globenewswire.com/news-release/2020/09/30/2101099/0/en/Stabilitech-Biopharma-announces-name-change-to-iosBio.html (accessed on 12 July 2022).
- Callaway, E. COVID Vaccines and Kids: Five Questions as Trials Begin. Nature 2021, 592, 670–671. [Google Scholar] [CrossRef] [PubMed]
- Montenegro, V.; Inchingolo, A.D.; Malcangi, G.; Limongelli, L.; Marinelli, G.; Coloccia, G.; Laudadio, C.; Patano, A.; Inchingolo, F.; Bordea, I.R.; et al. Compliance of Children with Removable Functional Appliance with Microchip Integrated during COVID-19 Pandemic: A Systematic Review. J. Biol. Regul. Homeost. Agents 2021, 35, 365–377. [Google Scholar] [CrossRef] [PubMed]
- Rose, A.; Stevo, C.; Alatovic, J.; Maas, S. Pfizer and BioNTech Announce Positive Topline Results from Pivotal Trial of COVID-19 Vaccine in Children 5 to 11 Years; Pfizer Inc.: New York, NY, USA, 2021. [Google Scholar]
- 240. Reuters CDC Backs Pfizer COVID-19 Vaccine for Young Children. The Guardian, 18 January 2021.
- CDC COVID-19 Vaccines for Children & Teens. Available online: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/recommendations/children-teens.html (accessed on 7 January 2022).
- Baang, J.H.; Smith, C.; Mirabelli, C.; Valesano, A.L.; Manthei, D.M.; Bachman, M.A.; Wobus, C.E.; Adams, M.; Washer, L.; Martin, E.T.; et al. Prolonged Severe Acute Respiratory Syndrome Coronavirus 2 Replication in an Immunocompromised Patient. J. Infect. Dis. 2021, 223, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Mullins, E.; Hudak, M.L.; Banerjee, J.; Getzlaff, T.; Townson, J.; Barnette, K.; Playle, R.; Perry, A.; Bourne, T.; Lees, C.C. Pregnancy and Neonatal Outcomes of COVID-19: Coreporting of Common Outcomes from PAN-COVID and AAP-SONPM Registries. Ultrasound Obstet. Gynecol. 2021, 57, 573–581. [Google Scholar] [CrossRef]
- Kadiwar, S.; Smith, J.J.; Ledot, S.; Johnson, M.; Bianchi, P.; Singh, N.; Montanaro, C.; Gatzoulis, M.; Shah, N.; Ukor, E.-F. Were Pregnant Women More Affected by COVID-19 in the Second Wave of the Pandemic? Lancet 2021, 397, 1539–1540. [Google Scholar] [CrossRef]
- Malcangi, G.; Inchingolo, A.D.; Inchingolo, A.M.; Santacroce, L.; Marinelli, G.; Mancini, A.; Vimercati, L.; Maggiore, M.E.; D’Oria, M.T.; Hazballa, D.; et al. COVID-19 Infection in Children, Infants and Pregnant Subjects: An Overview of Recent Insights and Therapies. Microorganisms 2021, 9, 1964. [Google Scholar] [CrossRef] [PubMed]
- Riley, L.E.; Jamieson, D.J. Inclusion of Pregnant and Lactating Persons in COVID-19 Vaccination Efforts. Ann. Intern. Med. 2021, 174, 701–702. [Google Scholar] [CrossRef] [PubMed]
- Mangat, C.; Milosavljevic, N. BNT162b2 Vaccination during Pregnancy Protects Both the Mother and Infant: Anti-SARS-CoV-2 S Antibodies Persistently Positive in an Infant at 6 Months of Age. Case Rep. Pediatr. 2021, 2021, 6901131. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, D.; Sridhar, A.; Lakshminrusimha, S.; Higgins, R.D.; Saade, G. COVID-19 Vaccine Considerations during Pregnancy and Lactation. Am. J. Perinatol. 2021, 38, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Pace, R.M.; Williams, J.E.; Järvinen, K.M.; Belfort, M.B.; Pace, C.D.; Lackey, K.A.; Gogel, A.C.; Nguyen-Contant, P.; Kanagaiah, P.; Fitzgerald, T. COVID-19 and Human Milk: SARS-CoV-2, Antibodies, and Neutralizing Capacity. medRxiv 2020. [Google Scholar] [CrossRef]
- Aygun, H. Vitamin D Can Prevent COVID-19 Infection-Induced Multiple Organ Damage. Naunyn Schmiedebergs Arch. Pharm. 2020, 393, 1157–1160. [Google Scholar] [CrossRef]
- Science, M.; Maguire, J.L.; Russell, M.L.; Smieja, M.; Walter, S.D.; Loeb, M. Low Serum 25-Hydroxyvitamin D Level and Risk of Upper Respiratory Tract Infection in Children and Adolescents. Clin. Infect. Dis. 2013, 57, 392–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gunasekar, P.; Swier, V.J.; Fleegel, J.P.; Boosani, C.S.; Radwan, M.M.; Agrawal, D.K. Vitamin D and Macrophage Polarization in Epicardial Adipose Tissue of Atherosclerotic Swine. PLoS ONE 2018, 13, e0199411. [Google Scholar] [CrossRef] [Green Version]
- Charitos, I.A.; Ballini, A.; Bottalico, L.; Cantore, S.; Passarelli, P.C.; Inchingolo, F.; D’Addona, A.; Santacroce, L. Special Features of SARS-CoV-2 in Daily Practice. WJCC 2020, 8, 3920–3933. [Google Scholar] [CrossRef] [PubMed]
- Vimercati, L.; De Maria, L.; Quarato, M.; Caputi, A.; Gesualdo, L.; Migliore, G.; Cavone, D.; Sponselli, S.; Pipoli, A.; Inchingolo, F.; et al. Association between Long COVID and Overweight/Obesity. JCM 2021, 10, 4143. [Google Scholar] [CrossRef]
- Santacroce, L.; Charitos, I.A.; Ballini, A.; Inchingolo, F.; Luperto, P.; De Nitto, E.; Topi, S. The Human Respiratory System and Its Microbiome at a Glimpse. Biology 2020, 9, 318. [Google Scholar] [CrossRef]
- Wang, Z.; Schmidt, F.; Weisblum, Y.; Muecksch, F.; Barnes, C.O.; Finkin, S.; Schaefer-Babajew, D.; Cipolla, M.; Gaebler, C.; Lieberman, J.A.; et al. MRNA Vaccine-Elicited Antibodies to SARS-CoV-2 and Circulating Variants. Immunology 2021, 592, 616–622. [Google Scholar] [CrossRef]
- Ai-ris, Y.C.; McMahan, K.; Yu, J.; Tostanoski, L.H.; Aguayo, R.; Ansel, J.; Chandrashekar, A.; Patel, S.; Bondzie, E.A.; Sellers, D. Immunogenicity of COVID-19 MRNA Vaccines in Pregnant and Lactating Women. JAMA 2021, 325, 2370–2380. [Google Scholar]
- Zeng, H.; Xu, C.; Fan, J.; Tang, Y.; Deng, Q.; Zhang, W.; Long, X. Antibodies in Infants Born to Mothers With COVID-19 Pneumonia. JAMA 2020, 323, 1848–1849. [Google Scholar] [CrossRef]
- Kohler, P.F.; Farr, R.S. Elevation of Cord over Maternal IgG Immunoglobulin: Evidence for an Active Placental IgG Transport. Nature 1966, 210, 1070–1071. [Google Scholar] [CrossRef]
- Ng, W.F.; Wong, S.F.; Lam, A.; Mak, Y.F.; Yao, H.; Lee, K.C.; Chow, K.M.; Yu, W.C.; Ho, L.C. The Placentas of Patients with Severe Acute Respiratory Syndrome: A Pathophysiological Evaluation. Pathology 2006, 38, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Perl, S.H.; Uzan-Yulzari, A.; Klainer, H.; Asiskovich, L.; Youngster, M.; Rinott, E.; Youngster, I. SARS-CoV-2–Specific Antibodies in Breast Milk after COVID-19 Vaccination of Breastfeeding Women. JAMA 2021, 325, 2013–2014. [Google Scholar] [CrossRef] [PubMed]
- Berry, S.D.; Johnson, K.S.; Myles, L.; Herndon, L.; Montoya, A.; Fashaw, S.; Gifford, D. Lessons Learned from Frontline Skilled Nursing Facility Staff Regarding COVID-19 Vaccine Hesitancy. J. Am. Geriatr. Soc. 2021, 69, 1140–1146. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, D.C.; Nassau, D.E.; Khodamoradi, K.; Ibrahim, E.; Blachman-Braun, R.; Ory, J.; Ramasamy, R. Sperm Parameters Before and After COVID-19 MRNA Vaccination. JAMA 2021, 326, 273. [Google Scholar] [CrossRef]
- Al-Ansari, R.Y.; Al-Sharari, M.; Al-Saadi, T. Palms and Soles Itchiness as a Side Effect of COVID-19 Vaccination. J. Infect Public Health 2021, 14, 1389–1391. [Google Scholar] [CrossRef] [PubMed]
- Larson, K.F.; Ammirati, E.; Adler, E.D.; Cooper, L.T.; Hong, K.N.; Saponara, G.; Couri, D.; Cereda, A.; Procopio, A.; Cavalotti, C.; et al. Myocarditis After BNT162b2 and MRNA-1273 Vaccination. Circulation 2021, 144, 506–508. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, B.; Kamat, I.; Hotez, P.J. Myocarditis With COVID-19 MRNA Vaccines. Circulation 2021, 144, 471–484. [Google Scholar] [CrossRef]
- Marshall, M.; Ferguson, I.D.; Lewis, P.; Jaggi, P.; Gagliardo, C.; Collins, J.S.; Shaughnessy, R.; Caron, R.; Fuss, C.; Corbin, K.J.E.; et al. Symptomatic Acute Myocarditis in 7 Adolescents after Pfizer-BioNTech COVID-19 Vaccination. Pediatrics 2021, 148, e2021052478. [Google Scholar] [CrossRef]
- Lu, L.; Xiong, W.; Mu, J.; Zhang, Q.; Zhang, H.; Zou, L.; Li, W.; He, L.; Sander, J.W.; Zhou, D. The Potential Neurological Effect of the COVID-19 Vaccines: A Review. Acta Neurol. Scand. 2021, 144, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Souders, C.; Carmel, M.; Anger, J.T. Low Rates of Urologic Side Effects Following Coronavirus Disease Vaccination: An Analysis of the Food and Drug Administration Vaccine Adverse Event Reporting System. Urology 2021, 153, 11–13. [Google Scholar] [CrossRef] [PubMed]
- Arora, P.; Sardana, K.; Mathachan, S.R.; Malhotra, P. Herpes Zoster after Inactivated COVID-19 Vaccine: A Cutaneous Adverse Effect of the Vaccine. J Cosmet Dermtol. 2021, 20, 3389–3390. [Google Scholar] [CrossRef] [PubMed]
- Kayser, F.; Fourneau, H.; Mazy, O.C.; Mazy, S. Breast Implant Seroma: A SARS-CoV-2 MRNA Vaccine Side Effect. J. Clin. Ultrasound. 2021, 49, 984–986. [Google Scholar] [CrossRef]
- Esba, L.C.A.; Al Jeraisy, M. Reported Adverse Effects Following COVID-19 Vaccination at a Tertiary Care Hospital, Focus on Cerebral Venous Sinus Thrombosis (CVST). Expert Rev. Vaccines 2021, 20, 1037–1042. [Google Scholar] [CrossRef]
- Lebedev, L.; Sapojnikov, M.; Wechsler, A.; Varadi-Levi, R.; Zamir, D.; Tobar, A.; Levin-Iaina, N.; Fytlovich, S.; Yagil, Y. Minimal Change Disease Following the Pfizer-BioNTech COVID-19 Vaccine. Am. J. Kidney Dis. 2021, 78, 142–145. [Google Scholar] [CrossRef]
- Fowler, N.; Mendez Martinez, N.R.; Pallares, B.V.; Maldonado, R.S. Acute-Onset Central Serous Retinopathy after Immunization with COVID-19 MRNA Vaccine. Am. J. Ophthalmol. Case Rep. 2021, 23, 101136. [Google Scholar] [CrossRef]
- Mahajan, S.; Zhang, F.; Mahajan, A.; Zimnowodzki, S. Parsonage Turner Syndrome after COVID-19 Vaccination. Muscle Nerve 2021, 64, E3–E4. [Google Scholar] [CrossRef]
- Cirillo, N. Reported Orofacial Adverse Effects of COVID-19 Vaccines: The Knowns and the Unknowns. J. Oral Pathol. Med. 2021, 50, 424–427. [Google Scholar] [CrossRef]
- Grupper, A.; Rabinowich, L.; Schwartz, D.; Schwartz, I.F.; Ben-Yehoyada, M.; Shashar, M.; Katchman, E.; Halperin, T.; Turner, D.; Goykhman, Y.; et al. Reduced Humoral Response to MRNA SARS-CoV-2 BNT162b2 Vaccine in Kidney Transplant Recipients without Prior Exposure to the Virus. Am. J. Transpl. 2021, 21, 2719–2726. [Google Scholar] [CrossRef] [PubMed]
- Mazzola, A.; Todesco, E.; Drouin, S.; Hazan, F.; Marot, S.; Thabut, D.; Varnous, S.; Soulié, C.; Barrou, B.; Marcelin, A.-G.; et al. Poor Antibody Response After Two Doses of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Vaccine in Transplant Recipients. Clin. Infect. Dis. 2022, 74, 1093–1096. [Google Scholar] [CrossRef] [PubMed]
- Hause, A.M.; Gee, J.; Baggs, J.; Abara, W.E.; Marquez, P.; Thompson, D.; Su, J.R.; Licata, C.; Rosenblum, H.G.; Myers, T.R.; et al. COVID-19 Vaccine Safety in Adolescents Aged 12–17 Years—United States, December 14, 2020–July 16, 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1053–1058. [Google Scholar] [CrossRef] [PubMed]
- Leone, F.; Cerasuolo, P.G.; Bosello, S.L.; Verardi, L.; Fiori, E.; Cocciolillo, F.; Merlino, B.; Zoli, A.; D’Agostino, M.A. Adult-Onset Still’s Disease Following COVID-19 Vaccination. Lancet Rheumatol. 2021, 3, e678–e680. [Google Scholar] [CrossRef]
- Rela, M.; Jothimani, D.; Vij, M.; Rajakumar, A.; Rammohan, A. Auto-Immune Hepatitis Following COVID Vaccination. J. Autoimmun. 2021, 123, 102688. [Google Scholar] [CrossRef] [PubMed]
- Sebode, M.; Hartl, J.; Vergani, D.; Lohse, A.W. The International Autoimmune Hepatitis Group (IAIHG) Autoimmune Hepatitis: From Current Knowledge and Clinical Practice to Future Research Agenda. Liver Int. 2018, 38, 15–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finsterer, J. Guillain-Barre Syndrome 15 Days after COVID-19 despite SARS-CoV-2 Vaccination. IDCases 2021, 25, e01226. [Google Scholar] [CrossRef]
- Thaler, J.; Ay, C.; Gleixner, K.V.; Hauswirth, A.W.; Cacioppo, F.; Grafeneder, J.; Quehenberger, P.; Pabinger, I.; Knöbl, P. Successful Treatment of Vaccine-induced Prothrombotic Immune Thrombocytopenia (VIPIT). J. Thromb. Haemost. 2021, 19, 1819–1822. [Google Scholar] [CrossRef]
- Cox, D. Targeting SARS-CoV-2-Platelet Interactions in COVID-19 and Vaccine-Related Thrombosis. Front. Pharmacol. 2021, 12, 708665. [Google Scholar] [CrossRef] [PubMed]
- Cocco, G.; Delli Pizzi, A.; Fabiani, S.; Cocco, N.; Boccatonda, A.; Frisone, A.; Scarano, A.; Schiavone, C. Lymphadenopathy after the Anti-COVID-19 Vaccine: Multiparametric Ultrasound Findings. Biology 2021, 10, 652. [Google Scholar] [CrossRef]
- Schiaffino, S.; Pinker, K.; Magni, V.; Cozzi, A.; Athanasiou, A.; Baltzer, P.A.T.; Camps Herrero, J.; Clauser, P.; Fallenberg, E.M.; Forrai, G.; et al. Axillary Lymphadenopathy at the Time of COVID-19 Vaccination: Ten Recommendations from the European Society of Breast Imaging (EUSOBI). Insights Imaging 2021, 12, 119. [Google Scholar] [CrossRef] [PubMed]
- Dotan, A.; Shoenfeld, Y. Perspectives on Vaccine Induced Thrombotic Thrombocytopenia. J. Autoimmun. 2021, 121, 102663. [Google Scholar] [CrossRef] [PubMed]
- Perumalswami, P.; Peng, L.; Odin, J.A. Vaccination as a Triggering Event for Autoimmune Hepatitis. Semin. Liver Dis. 2009, 29, 331–334. [Google Scholar] [CrossRef] [PubMed]
- van Gemeren, M.A.J.; van Wijngaarden, P.; Doukas, M.; de Man, R.A. Vaccine-Related Autoimmune Hepatitis: The Same Disease as Idiopathic Autoimmune Hepatitis? Two Clinical Reports and Review. Scand. J. Gastroenterol. 2017, 52, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Asplund Högelin, K.; Ruffin, N.; Pin, E.; Månberg, A.; Hober, S.; Gafvelin, G.; Grönlund, H.; Nilsson, P.; Khademi, M.; Olsson, T.; et al. Development of Humoral and Cellular Immunological Memory against SARS-CoV-2 despite B Cell Depleting Treatment in Multiple Sclerosis. iScience 2021, 24, 103078. [Google Scholar] [CrossRef] [PubMed]
- Marinelli, G.; Inchingolo, A.D.; Inchingolo, A.M.; Malcangi, G.; Limongelli, L.; Montenegro, V.; Coloccia, G.; Laudadio, C.; Patano, A.; Inchingolo, F.; et al. White Spot Lesions in Orthodontics: Prevention and Treatment. A Descriptive Review. J. Biol. Regul. Homeost. Agents 2021, 35, 227–240. [Google Scholar] [CrossRef] [PubMed]
- Adina, S. Orthopedic Joint Stability Influences Growth and Maxillary Development: Clinical Aspects. J. Biol. Regul. Homeost. Agents 2020, 34. [Google Scholar] [CrossRef]
- Patianna, A.G.; Ballini, A.; Meneghello, M.; Cantore, S.; Inchingolo, A.M.; Dipalma, G.; Inchingolo, A.D.; Inchingolo, F.; Malcangi, G.; Lucchese, A.; et al. Comparison of Conventional Orthognathic Surgery and “Surgery-First” Protocol: A New Weapon against Time. J. Biol. Regul. Homeost Agents 2019, 33, 59–67. [Google Scholar] [PubMed]
- Cantore, S.; Ballini, A.; De Vito, D.; Martelli, F.S.; Georgakopoulos, I.; Almasri, M.; Dibello, V.; Altini, V.; Farronato, G.; Dipalma, G.; et al. Characterization of Human Apical Papilla-Derived Stem Cells. J. Biol. Regul. Homeost. Agents 2017, 31, 901–910. [Google Scholar] [PubMed]
- Ballini, A.; Cantore, S.; Fotopoulou, E.A.; Georgakopoulos, I.P.; Athanasiou, E.; Bellos, D.; Paduanelli, G.; Saini, R.; Dipalma, G.; Inchingolo, F. Combined Sea Salt-Based Oral Rinse with Xylitol in Orthodontic Patients: Clinical and Microbiological Study. J. Biol. Regul. Homeost. Agents 2019, 33, 263–268. [Google Scholar]
- Cantore, S.; Ballini, A.; Farronato, D.; Malcangi, G.; Dipalma, G.; Assandri, F.; Garagiola, U.; Inchingolo, F.; De Vito, D.; Cirulli, N. Evaluation of an Oral Appliance in Patients with Mild to Moderate Obstructive Sleep Apnea Syndrome Intolerant to Continuous Positive Airway Pressure Use: Preliminary Results. Int. J. Immunopathol. Pharm. 2016, 29, 267–273. [Google Scholar] [CrossRef] [Green Version]
- Ballini, A.; Cantore, S.; Scacco, S.; Perillo, L.; Scarano, A.; Aityan, S.K.; Contaldo, M.; Cd Nguyen, K.; Santacroce, L.; Syed, J.; et al. A Comparative Study on Different Stemness Gene Expression between Dental Pulp Stem Cells vs. Dental Bud Stem Cells. Eur. Rev. Med. Pharm. Sci. 2019, 23, 1626–1633. [Google Scholar] [CrossRef]
- Iannetta, M.; Cesta, N.; Stingone, C.; Malagnino, V.; Teti, E.; Vitale, P.; De Simone, G.; Rossi, B.; Ansaldo, L.; Compagno, M.; et al. Mild Clinical Manifestations of SARS-CoV-2 Related Pneumonia in Two Patients with Multiple Sclerosis under Treatment with Ocrelizumab. Mult. Scler. Relat. Disord. 2020, 45, 102442. [Google Scholar] [CrossRef] [PubMed]
- Khayat-Khoei, M.; Conway, S.; Rubinson, D.A.; Jarolim, P.; Houtchens, M.K. Negative Anti-SARS-CoV-2 S Antibody Response Following Pfizer SARS-CoV-2 Vaccination in a Patient on Ocrelizumab. J. Neurol. 2021, 268, 3592–3594. [Google Scholar] [CrossRef] [PubMed]
- Smets, I.; Reyes, S.; Baker, D.; Giovannoni, G. Blunted Vaccines Responses after Ocrelizumab Highlight Need for Immunizations Prior to Treatment. Mult. Scler. Relat. Disord. 2021, 50, 102851. [Google Scholar] [CrossRef] [PubMed]
- Thornton, J.R.; Harel, A. Negative SARS-CoV-2 Antibody Testing Following COVID-19 Infection in Two MS Patients Treated with Ocrelizumab. Mult. Scler. Relat. Disord. 2020, 44, 102341. [Google Scholar] [CrossRef]
- Achiron, A.; Mandel, M.; Dreyer-Alster, S.; Harari, G.; Magalashvili, D.; Sonis, P.; Dolev, M.; Menascu, S.; Flechter, S.; Falb, R.; et al. Humoral Immune Response to COVID-19 MRNA Vaccine in Patients with Multiple Sclerosis Treated with High-Efficacy Disease-Modifying Therapies. Adv. Neurol. Disord. 2021, 14, 17562864211012836. [Google Scholar] [CrossRef]
- Murugesan, K.; Jagannathan, P.; Pham, T.D.; Pandey, S.; Bonilla, H.F.; Jacobson, K.; Parsonnet, J.; Andrews, J.R.; Weiskopf, D.; Sette, A.; et al. Interferon-γ Release Assay for Accurate Detection of Severe Acute Respiratory Syndrome Coronavirus 2 T-Cell Response. Clin. Infect. Dis. 2021, 73, e3130–e3132. [Google Scholar] [CrossRef]
- Iannetta, M.; Landi, D.; Cola, G.; Malagnino, V.; Teti, E.; Fraboni, D.; Buccisano, F.; Grelli, S.; Coppola, L.; Campogiani, L.; et al. T-Cell Responses to SARS-CoV-2 in Multiple Sclerosis Patients Treated with Ocrelizumab Healed from COVID-19 with Absent or Low Anti-Spike Antibody Titers. Mult. Scler. Relat. Disord. 2021, 55, 103157. [Google Scholar] [CrossRef]
- Bajaj, V.; Gadi, N.; Spihlman, A.P.; Wu, S.C.; Choi, C.H.; Moulton, V.R. Aging, Immunity, and COVID-19: How Age Influences the Host Immune Response to Coronavirus Infections? Front. Physiol. 2021, 11, 571416. [Google Scholar] [CrossRef]
- Wei, J.; Stoesser, N.; Matthews, P.C.; Ayoubkhani, D.; Studley, R.; Bell, I.; Bell, J.I.; Newton, J.N.; Farrar, J.; Diamond, I.; et al. Antibody Responses to SARS-CoV-2 Vaccines in 45,965 Adults from the General Population of the United Kingdom. Nat. Microbiol. 2021, 6, 1140–1149. [Google Scholar] [CrossRef]
- Yang, H.S.; Costa, V.; Racine-Brzostek, S.E.; Acker, K.P.; Yee, J.; Chen, Z.; Karbaschi, M.; Zuk, R.; Rand, S.; Sukhu, A.; et al. Association of Age With SARS-CoV-2 Antibody Response. JAMA Netw. Open 2021, 4, e214302. [Google Scholar] [CrossRef] [PubMed]
- García-Hernández, M.H.; Rodríguez-Varela, E.; García-Jacobo, R.E.; Hernández-De la Torre, M.; Uresti-Rivera, E.E.; González-Amaro, R.; Portales-Pérez, D.P. Frequency of Regulatory B Cells in Adipose Tissue and Peripheral Blood from Individuals with Overweight, Obesity and Normal-Weight. Obes. Res. Clin. Pract. 2018, 12, 513–519. [Google Scholar] [CrossRef]
- Sheridan, P.A.; Paich, H.A.; Handy, J.; Karlsson, E.A.; Hudgens, M.G.; Sammon, A.B.; Holland, L.A.; Weir, S.; Noah, T.L.; Beck, M.A. Obesity Is Associated with Impaired Immune Response to Influenza Vaccination in Humans. Int. J. Obes. 2012, 36, 1072–1077. [Google Scholar] [CrossRef] [Green Version]
- Eliakim, A.; Swindt, C.; Zaldivar, F.; Casali, P.; Cooper, D.M. Reduced Tetanus Antibody Titers in Overweight Children. Autoimmunity 2006, 39, 137–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lacson, E.; Argyropoulos, C.P.; Manley, H.J.; Aweh, G.; Chin, A.I.; Salman, L.H.; Hsu, C.M.; Johnson, D.S.; Weiner, D.E. Immunogenicity of SARS-CoV-2 Vaccine in Dialysis. J. Am. Soc. Nephrol. 2021, 32, 2735–2742. [Google Scholar] [CrossRef] [PubMed]
- Malard, F.; Gaugler, B.; Gozlan, J.; Bouquet, L.; Fofana, D.; Siblany, L.; Eshagh, D.; Adotevi, O.; Laheurte, C.; Ricard, L.; et al. Weak Immunogenicity of SARS-CoV-2 Vaccine in Patients with Hematologic Malignancies. Blood Cancer J. 2021, 11, 142. [Google Scholar] [CrossRef] [PubMed]
- Michiels, Y.; Houhou-Fidouh, N.; Collin, G.; Berger, J.; Kohli, E. Impact of Low-Dose Methotrexate–Adalimumab Combination Therapy on the Antibody Response Induced by the MRNA-1273 SARS-CoV-2 Vaccine: Case of an Elderly Patient with Rheumatoid Arthritis. Vaccines 2021, 9, 883. [Google Scholar] [CrossRef]
- Anand, S.P.; Prévost, J.; Nayrac, M.; Beaudoin-Bussières, G.; Benlarbi, M.; Gasser, R.; Brassard, N.; Laumaea, A.; Gong, S.Y.; Bourassa, C.; et al. Longitudinal Analysis of Humoral Immunity against SARS-CoV-2 Spike in Convalescent Individuals up to 8 Months Post-Symptom Onset. Cell Rep. Med. 2021, 2, 100290. [Google Scholar] [CrossRef]
- Tissot, N.; Brunel, A.-S.; Bozon, F.; Rosolen, B.; Chirouze, C.; Bouiller, K. Patients with History of COVID-19 Had More Side Effects after the First Dose of COVID-19 Vaccine. Vaccine 2021, 39, 5087–5090. [Google Scholar] [CrossRef]
- Modenese, A.; Paduano, S.; Bargellini, A.; Bellucci, R.; Marchetti, S.; Bruno, F.; Grazioli, P.; Vivoli, R.; Gobba, F. Neutralizing Anti-SARS-CoV-2 Antibody Titer and Reported Adverse Effects, in a Sample of Italian Nursing Home Personnel after Two Doses of the BNT162b2 Vaccine Administered Four Weeks Apart. Vaccines 2021, 9, 652. [Google Scholar] [CrossRef]
- Helmold Hait, S.; Vargas-Inchaustegui, D.A.; Musich, T.; Mohanram, V.; Tuero, I.; Venzon, D.J.; Bear, J.; Rosati, M.; Vaccari, M.; Franchini, G.; et al. Early T Follicular Helper Cell Responses and Germinal Center Reactions Are Associated with Viremia Control in Immunized Rhesus Macaques. J. Virol. 2019, 93, e01687-18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turner, J.S.; O’Halloran, J.A.; Kalaidina, E.; Kim, W.; Schmitz, A.J.; Zhou, J.Q.; Lei, T.; Thapa, M.; Chen, R.E.; Case, J.B.; et al. SARS-CoV-2 MRNA Vaccines Induce Persistent Human Germinal Centre Responses. Nature 2021, 596, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, N.; Kuo, H.-H.; Boucau, J.; Farmer, J.R.; Allard-Chamard, H.; Mahajan, V.S.; Piechocka-Trocha, A.; Lefteri, K.; Osborn, M.; Bals, J.; et al. Loss of Bcl-6-Expressing T Follicular Helper Cells and Germinal Centers in COVID-19. Cell 2020, 183, 143–157.e13. [Google Scholar] [CrossRef]
- Barrett, J.R.; Belij-Rammerstorfer, S.; Dold, C.; Ewer, K.J.; Folegatti, P.M.; Gilbride, C.; Halkerston, R.; Hill, J.; Jenkin, D.; Stockdale, L.; et al. Phase 1/2 Trial of SARS-CoV-2 Vaccine ChAdOx1 NCoV-19 with a Booster Dose Induces Multifunctional Antibody Responses. Nat. Med. 2021, 27, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Khoury, D.S.; Cromer, D.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Subbarao, K.; Kent, S.J.; Triccas, J.A.; Davenport, M.P. Neutralizing Antibody Levels Are Highly Predictive of Immune Protection from Symptomatic SARS-CoV-2 Infection. Nat. Med. 2021, 27, 1205–1211. [Google Scholar] [CrossRef]
- Brochot, E.; Demey, B.; Touzé, A.; Belouzard, S.; Dubuisson, J.; Schmit, J.-L.; Duverlie, G.; Francois, C.; Castelain, S.; Helle, F. Anti-Spike, Anti-Nucleocapsid and Neutralizing Antibodies in SARS-CoV-2 Inpatients and Asymptomatic Individuals. Front. Microbiol. 2020, 11, 584251. [Google Scholar] [CrossRef]
- Tauzin, A.; Nayrac, M.; Benlarbi, M.; Gong, S.Y.; Gasser, R.; Beaudoin-Bussières, G.; Brassard, N.; Laumaea, A.; Vézina, D.; Prévost, J.; et al. A Single BNT162b2 MRNA Dose Elicits Antibodies with Fc-Mediated Effector Functions and Boost Pre-Existing Humoral and T Cell Responses. Immunology 2021. preprint. [Google Scholar]
- Trougakos, I.P.; Terpos, E.; Zirou, C.; Sklirou, A.D.; Apostolakou, F.; Gumeni, S.; Charitaki, I.; Papanagnou, E.-D.; Bagratuni, T.; Liacos, C.-I.; et al. Comparative Kinetics of SARS-CoV-2 Anti-Spike Protein RBD IgGs and Neutralizing Antibodies in Convalescent and Naïve Recipients of the BNT162b2 MRNA Vaccine versus COVID-19 Patients. BMC Med. 2021, 19, 208. [Google Scholar] [CrossRef]
- Levi, R.; Azzolini, E.; Pozzi, C.; Ubaldi, L.; Lagioia, M.; Mantovani, A.; Rescigno, M. One Dose of SARS-CoV-2 Vaccine Exponentially Increases Antibodies in Individuals Who Have Recovered from Symptomatic COVID-19. J. Clin. Investig. 2021, 131, e149154. [Google Scholar] [CrossRef]
- Doria-Rose, N.; Suthar, M.S.; Makowski, M.; O’Connell, S.; McDermott, A.B.; Flach, B.; Ledgerwood, J.E.; Mascola, J.R.; Graham, B.S.; Lin, B.C.; et al. Antibody Persistence through 6 Months after the Second Dose of MRNA-1273 Vaccine for COVID-19. N. Engl. J. Med. 2021, 384, 2259–2261. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Noumbissi, S.; Gupta, S.; Inchingolo, F.; Stilla, P.; Lorusso, F. Scanning Electron Microscopy Analysis and Energy Dispersion X-Ray Microanalysis to Evaluate the Effects of Decontamination Chemicals and Heat Sterilization on Implant Surgical Drills: Zirconia vs. Steel. Appl. Sci. 2019, 9, 2837. [Google Scholar] [CrossRef] [Green Version]
- Scarano, A.; Piattelli, A.; Assenza, B.; Carinci, F.; Donato, L.D.; Romani GLMerla, A. Infrared Thermographic Evaluation of Temperature Modifications Induced during Implant Site Preparation with Steel vs. Zirconia Implant Drill. J. Clin. Med. 2020, 9, 148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scarano, A.; Murmura, G.; Vantaggiato, G.; Lauritano, D.; Silvestre-Rangil, J.; Di Cerbo, A.; Lorusso, F. Delayed Expansion of Atrophic Mandible (Deam): A Case Report. Oral Implantol. 2017, 10, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Balzanelli, M.G.; Distratis, P.; Dipalma, G.; Vimercati, L.; Inchingolo, A.D.; Lazzaro, R.; Aityan, S.K.; Maggiore, M.E.; Mancini, A.; Laforgia, R.; et al. SARS-CoV-2 Virus Infection May Interfere CD34+ Hematopoietic Stem Cells and Megakaryocyte–Erythroid Progenitors Differentiation Contributing to Platelet Defection towards Insurgence of Thrombocytopenia and Thrombophilia. Microorganisms 2021, 9, 1632. [Google Scholar] [CrossRef]
- Rapone, B.; Ferrara, E.; Corsalini, M.; Qorri, E.; Converti, I.; Lorusso, F.; Delvecchio, M.; Gnoni, A.; Scacco, S.; Scarano, A. Inflammatory Status and Glycemic Control Level of Patients with Type 2 Diabetes and Periodontitis: A Randomized Clinical Trial. Int. J. Environ. Res. Public Health 2021, 18, 3018. [Google Scholar] [CrossRef] [PubMed]
- Kalemaj, Z.; Scarano, A.; Valbonetti, L.; Rapone, B.; Grassi, F.R. Bone Response to Four Dental Implants with Different Surface Topographies: A Histologic and Histometric Study in Minipigs. Int. J. Periodontics Restor. Dent. 2016, 36, 745–754. [Google Scholar] [CrossRef] [Green Version]
- Grassi, F.R.; Grassi, R.; Rapone, B.; Alemanno, G.; Balena, A.; Kalemaj, Z. Dimensional Changes of Buccal Bone Plate in Immediate Implants Inserted through Open Flap, Open Flap and Bone Grafting and Flapless Techniques: A Cone-Beam Computed Tomography Randomized Controlled Clinical Trial. Clin. Oral Implants Res. 2019, 30, 1155–1164. [Google Scholar] [CrossRef]
- Lorusso, F.; Noumbissi, S.; Francesco, I.; Rapone, B.; Khater, A.G.A.; Scarano, A. Scientific Trends in Clinical Research on Zirconia Dental Implants: A Bibliometric Review. Materials 2020, 13, 5534. [Google Scholar] [CrossRef]
- Favresse, J.; Bayart, J.-L.; Mullier, F.; Elsen, M.; Eucher, C.; Van Eeckhoudt, S.; Roy, T.; Wieers, G.; Laurent, C.; Dogné, J.-M.; et al. Antibody Titres Decline 3-Month Post-Vaccination with BNT162b2. Emerg. Microbes Infect. 2021, 10, 1495–1498. [Google Scholar] [CrossRef]
- Lorusso, F.; Tartaglia, G.; Inchingolo, F.; Scarano, A. Early Response and Clinical Efficacy of a Mouthwash Containing Chlorhexidine, Anti Discoloration System, Polyvinylpyrrolidone/Vinyl Acetate and Sodium DNA in Periodontitis Model: A Triple-Blind Randomized Controlled Clinical Trial. Dent. J. 2022, 10, 101. [Google Scholar] [CrossRef]
- Müller, L.; Andrée, M.; Moskorz, W.; Drexler, I.; Walotka, L.; Grothmann, R.; Ptok, J.; Hillebrandt, J.; Ritchie, A.; Rabl, D.; et al. Age-Dependent Immune Response to the Biontech/Pfizer BNT162b2 Coronavirus Disease 2019 Vaccination. Clin. Infect. Dis. 2021, 73, 2065–2072. [Google Scholar] [CrossRef] [PubMed]
- Macedo-Ojeda, G.; Muñoz-Valle, J.; Yokogawa-Teraoka, P.; Machado-Sulbarán, A.; Loza-Rojas, M.; Arredondo, A.G.; Tejeda-Constantini, R.; Vega-Magaña, A.; González-Estevez, G.; García-Chagollán, M.; et al. COVID-19 Screening By Anti-SARS-CoV-2 Antibody Seropositivity: Clinical And Epidemiological Characteristics, Comorbidities, And Food Intake Quality. Int. J. Environ. Res. Public Health 2021, 18, 8995. [Google Scholar] [CrossRef] [PubMed]
- Pellini, R.; Venuti, A.; Pimpinelli, F.; Abril, E.; Blandino, G.; Campo, F.; Conti, L.; De Virgilio, A.; De Marco, F.; Di Domenico, E.G.; et al. Early Onset of SARS-CoV-2 Antibodies after First Dose of BNT162b2: Correlation with Age, Gender and BMI. Vaccines 2021, 9, 685. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; McCullough, P.A.; Tecson, K.M. Vitamin D Deficiency in Association with Endothelial Dysfunction: Implications for Patients WithCOVID-19. Rev. Cardiovasc. Med. 2020, 21, 339. [Google Scholar] [CrossRef]
- Bellocchio, L.; Bordea, I.R.; Ballini, A.; Lorusso, F.; Hazballa, D.; Isacco, C.G.; Malcangi, G.; Inchingolo, A.D.; Dipalma, G.; Inchingolo, F.; et al. Environmental Issues and Neurological Manifestations Associated with COVID-19 Pandemic: New Aspects of the Disease? Int. J. Environ. Res. Public Health 2020, 17, 8049. [Google Scholar] [CrossRef]
- Inchingolo, F.; Martelli, F.S.; Gargiulo Isacco, C.; Borsani, E.; Cantore, S.; Corcioli, F.; Boddi, A.; Nguyễn, K.C.D.; De Vito, D.; Aityan, S.K.; et al. Chronic Periodontitis and Immunity, Towards the Implementation of a Personalized Medicine: A Translational Research on Gene Single Nucleotide Polymorphisms (SNPs) Linked to Chronic Oral Dysbiosis in 96 Caucasian Patients. Biomedicines 2020, 8, 115. [Google Scholar] [CrossRef]
- Ballini, A.; Gnoni, A.; De Vito, D.; Dipalma, G.; Cantore, S.; Gargiulo Isacco, C.; Saini, R.; Santacroce, L.; Topi, S.; Scarano, A.; et al. Effect of Probiotics on the Occurrence of Nutrition Absorption Capacities in Healthy Children: A Randomized Double-Blinded Placebo-Controlled Pilot Study. Eur. Rev. Med. Pharm. Sci. 2019, 23, 8645–8657. [Google Scholar] [CrossRef]
- Santacroce, L.; Inchingolo, F.; Topi, S.; Del Prete, R.; Di Cosola, M.; Charitos, I.A.; Montagnani, M. Potential Beneficial Role of Probiotics on the Outcome of COVID-19 Patients: An Evolving Perspective. Diabetes Metab. Syndr. Clin. Res. Rev. 2021, 15, 295–301. [Google Scholar] [CrossRef]
- Bastard, P.; Rosen, L.B.; Zhang, Q.; Michailidis, E.; Hoffmann, H.-H.; Zhang, Y.; Dorgham, K.; Philippot, Q.; Rosain, J.; Béziat, V.; et al. Autoantibodies against Type I IFNs in Patients with Life-Threatening COVID-19. Science 2020, 370, eabd4585. [Google Scholar] [CrossRef]
- Lazear, H.M.; Schoggins, J.W.; Diamond, M.S. Shared and Distinct Functions of Type I and Type III Interferons. Immunity 2019, 50, 907–923. [Google Scholar] [CrossRef]
- Balzanelli, M.G.; Distratis, P.; Dipalma, G.; Vimercati, L.; Catucci, O.; Amatulli, F.; Cefalo, A.; Lazzaro, R.; Palazzo, D.; Aityan, S.K.; et al. Immunity Profiling of COVID-19 Infection, Dynamic Variations of Lymphocyte Subsets, a Comparative Analysis on Four Different Groups. Microorganisms 2021, 9, 2036. [Google Scholar] [CrossRef] [PubMed]
- Busnadiego, I.; Fernbach, S.; Pohl, M.O.; Karakus, U.; Huber, M.; Trkola, A.; Stertz, S.; Hale, B.G. Antiviral Activity of Type I, II, and III Interferons Counterbalances ACE2 Inducibility and Restricts SARS-CoV-2. mBio 2020, 11, e01928-20. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xiao, M.; Zhang, S.; Xia, P.; Cao, W.; Jiang, W.; Chen, H.; Ding, X.; Zhao, H.; Zhang, H.; et al. Coagulopathy and Antiphospholipid Antibodies in Patients with COVID-19. N. Engl. J. Med. 2020, 382, e38. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Han, T.; Chen, J.; Hou, C.; Hua, L.; He, S.; Guo, Y.; Zhang, S.; Wang, Y.; Yuan, J.; et al. Clinical and Autoimmune Characteristics of Severe and Critical Cases of COVID-19. Clin. Transl. Sci. 2020, 13, 1077–1086. [Google Scholar] [CrossRef] [PubMed]
- Zuniga, M.; Gomes, C.; Carsons, S.E.; Bender, M.T.; Cotzia, P.; Miao, Q.R.; Lee, D.C.; Rodriguez, A. Autoimmunity to Annexin A2 Predicts Mortality among Hospitalised COVID-19 Patients. Eur. Respir. J. 2021, 58, 2100918. [Google Scholar] [CrossRef]
- Gomes, C.; Zuniga, M.; Crotty, K.A.; Qian, K.; Tovar, N.C.; Lin, L.H.; Argyropoulos, K.V.; Clancy, R.; Izmirly, P.; Buyon, J.; et al. Autoimmune Anti-DNA and Anti-Phosphatidylserine Antibodies Predict Development of Severe COVID-19. Life Sci. Alliance 2021, 4, e202101180. [Google Scholar] [CrossRef]
- Schultze, J.L.; Aschenbrenner, A.C. COVID-19 and the Human Innate Immune System. Cell 2021, 184, 1671–1692. [Google Scholar] [CrossRef]
- Combadière, B. Immunité Adaptative Contre Le Virus SARS-CoV-2. Med. Sci. 2020, 36, 908–913. [Google Scholar] [CrossRef]
- Goel, R.R.; Painter, M.M.; Apostolidis, S.A.; Mathew, D.; Meng, W.; Rosenfeld, A.M.; Lundgreen, K.A.; Reynaldi, A.; Khoury, D.S.; Pattekar, A.; et al. MRNA Vaccination Induces Durable Immune Memory to SARS-CoV-2 with Continued Evolution to Variants of Concern. Science 2021, 374, abm0829. [Google Scholar] [CrossRef]
- Callow, K.A.; Parry, H.F.; Sergeant, M.; Tyrrell, D.A.J. The Time Course of the Immune Response to Experimental Coronavirus Infection of Man. Epidemiol. Infect. 1990, 105, 435–446. [Google Scholar] [CrossRef] [Green Version]
- Novelli, G.; Biancolella, M.; Mehrian-Shai, R.; Colona, V.L.; Brito, A.F.; Grubaugh, N.D.; Vasiliou, V.; Luzzatto, L.; Reichardt, J.K.V. COVID-19 One Year into the Pandemic: From Genetics and Genomics to Therapy, Vaccination, and Policy. Hum. Genom. 2021, 15, 27. [Google Scholar] [CrossRef] [PubMed]
- Zeberg, H.; Pääbo, S. A Genomic Region Associated with Protection against Severe COVID-19 Is Inherited from Neandertals. Proc. Natl. Acad. Sci. USA 2021, 118, e2026309118. [Google Scholar] [CrossRef] [PubMed]
- Thevarajan, I.; Nguyen, T.H.O.; Koutsakos, M.; Druce, J.; Caly, L.; van de Sandt, C.E.; Jia, X.; Nicholson, S.; Catton, M.; Cowie, B.; et al. Breadth of Concomitant Immune Responses Prior to Patient Recovery: A Case Report of Non-Severe COVID-19. Nat. Med. 2020, 26, 453–455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cunha, L.L.; Perazzio, S.F.; Azzi, J.; Cravedi, P.; Riella, L.V. Remodeling of the Immune Response With Aging: Immunosenescence and Its Potential Impact on COVID-19 Immune Response. Front. Immunol. 2020, 11, 1748. [Google Scholar] [CrossRef]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors Associated with COVID-19-Related Death Using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef]
- Koning, R.; Bastard, P.; Casanova, J.-L.; Brouwer, M.C.; van de Beek, D.; The Amsterdam U.M.C. COVID-19 Biobank Investigators. Autoantibodies against Type I Interferons Are Associated with Multi-Organ Failure in COVID-19 Patients. Intensive Care Med. 2021, 47, 704–706. [Google Scholar] [CrossRef]
- Zhou, W.; Wang, W. Auto-Antibodies against Type I IFNs Are Associated with Severe COVID-19 Pneumonia. Sig. Transduct. Target 2021, 6, 96. [Google Scholar] [CrossRef]
- Liao, M.; Liu, Y.; Yuan, J.; Wen, Y.; Xu, G.; Zhao, J.; Cheng, L.; Li, J.; Wang, X.; Wang, F.; et al. Single-Cell Landscape of Bronchoalveolar Immune Cells in Patients with COVID-19. Nat. Med. 2020, 26, 842–844. [Google Scholar] [CrossRef]
- Rodrigues, T.S.; de Sá, K.S.G.; Ishimoto, A.Y.; Becerra, A.; Oliveira, S.; Almeida, L.; Gonçalves, A.V.; Perucello, D.B.; Andrade, W.A.; Castro, R.; et al. Inflammasomes Are Activated in Response to SARS-CoV-2 Infection and Are Associated with COVID-19 Severity in Patients. J. Exp. Med. 2021, 218, e20201707. [Google Scholar] [CrossRef]
- Colona, V.L.; Vasiliou, V.; Watt, J.; Novelli, G.; Reichardt, J.K.V. Update on Human Genetic Susceptibility to COVID-19: Susceptibility to Virus and Response. Hum Genom. 2021, 15, 57. [Google Scholar] [CrossRef]
- Netea, M.G.; Domínguez-Andrés, J.; Barreiro, L.B.; Chavakis, T.; Divangahi, M.; Fuchs, E.; Joosten, L.A.B.; van der Meer, J.W.M.; Mhlanga, M.M.; Mulder, W.J.M.; et al. Defining Trained Immunity and Its Role in Health and Disease. Nat Rev Immunol 2020, 20, 375–388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Severe COVID-19 GWAS Group; Ellinghaus, D.; Degenhardt, F.; Bujanda, L.; Buti, M.; Albillos, A.; Invernizzi, P.; Fernández, J.; Prati, D.; Baselli, G.; et al. Genomewide Association Study of Severe COVID-19 with Respiratory Failure. N. Engl. J. Med. 2020, 383, 1522–1534. [Google Scholar] [CrossRef] [PubMed]
- Zeberg, H.; Pääbo, S. The Major Genetic Risk Factor for Severe COVID-19 Is Inherited from Neanderthals. Nature 2020, 587, 610–612. [Google Scholar] [CrossRef]
- Villaescusa, L.; Zaragozá, F.; Gayo-Abeleira, I.; Zaragozá, C. A New Approach to the Management of COVID-19. Antagonists of IL-6: Siltuximab. Adv. Ther. 2022, 39, 1126–1148. [Google Scholar] [CrossRef] [PubMed]
- Sagris, M.; Theofilis, P.; Antonopoulos, A.S.; Oikonomou, E.; Tsioufis, K.; Tousoulis, D. Genetic Predisposition and Inflammatory Inhibitors in COVID-19: Where Do We Stand? Biomedicines 2022, 10, 242. [Google Scholar] [CrossRef] [PubMed]

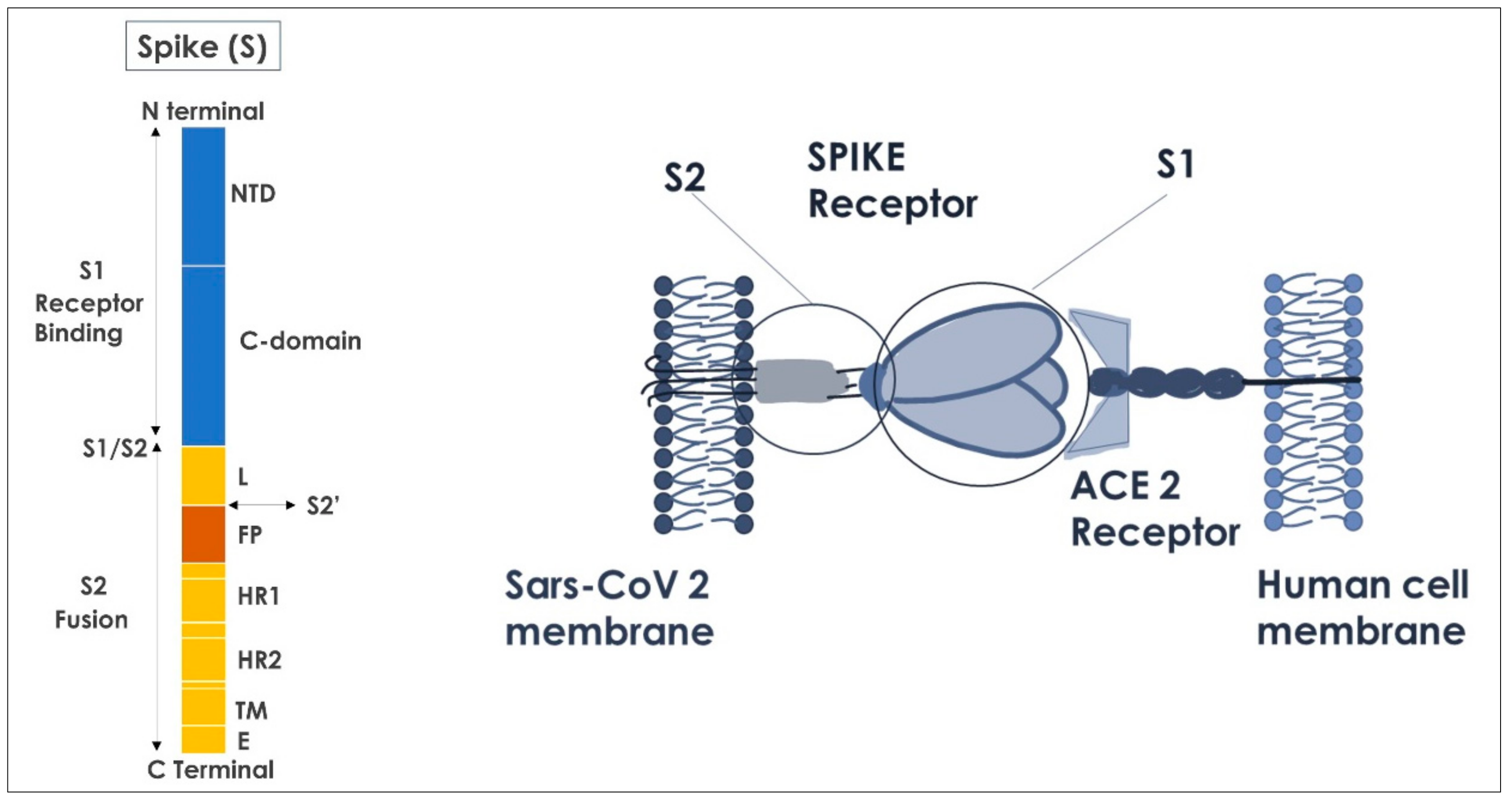
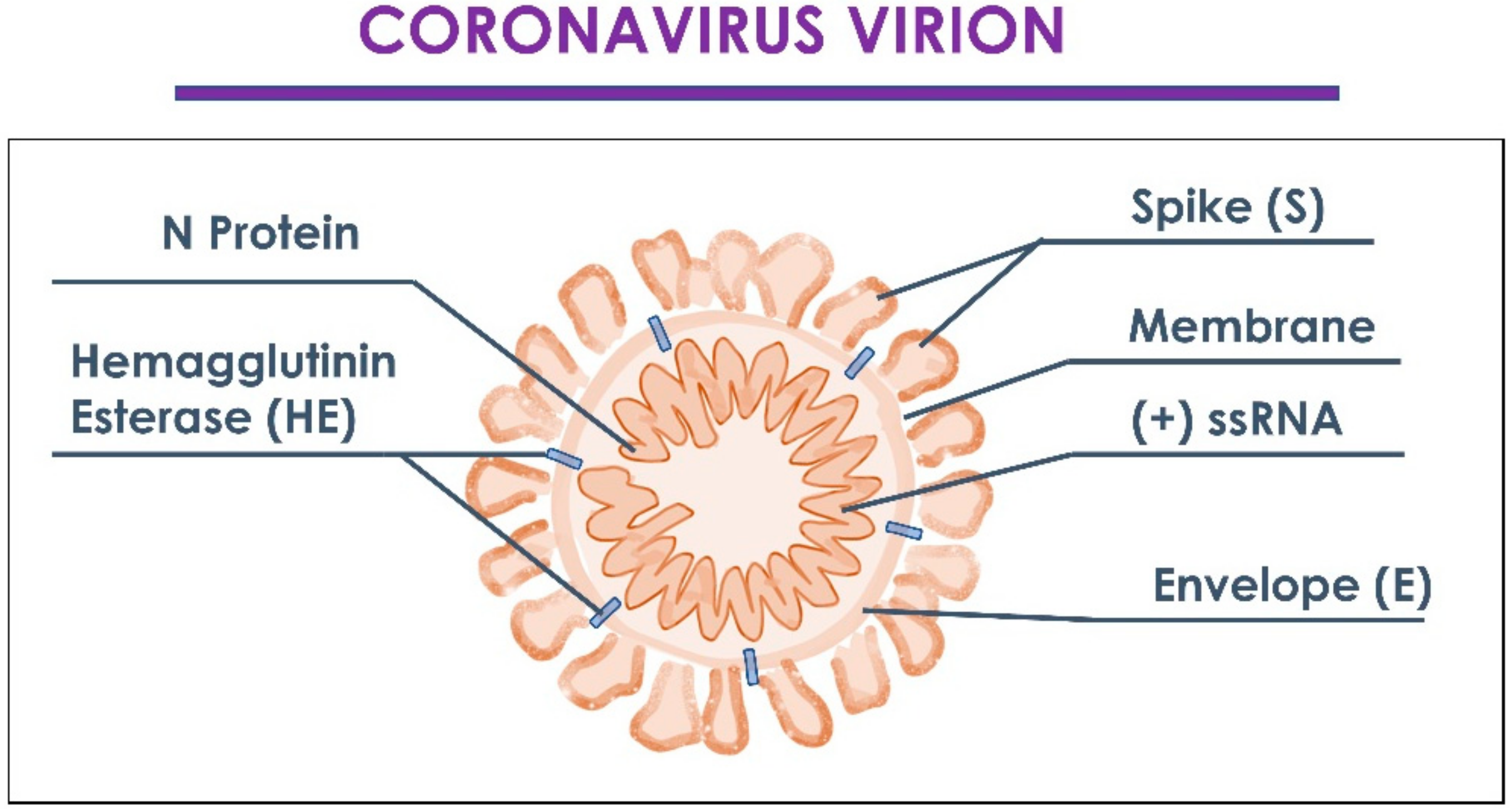

| Codification | Vector Lineages | Clades | Nextstrain Clades | Aminoacid Changes | Emerging Regions and Period of Strain Detection | Date of First Detection |
|---|---|---|---|---|---|---|
| Alpha | B.1.1.7 | GRY | 20I.V1 | +S: 484K +S: 452R | United Kingdom, Sep-2020 | 18-Dec-2020 |
| Beta | B.1.351 | GH/501Y.V2 | 20H.V2 | +S: L18F | South Africa, May-2020 | 18-Dec-2020 |
| Gamma | P.1 | GR/501Y.V3 | 20J.V3 | +S: 681H | Brazil, Nov-2020 | 11-Jan-2021 |
| Delta | B.1.617.2 | G/478K.V1 | 21; 21I; 21J | +S: 417N +S: 484K | India, Oct-2020 | VOI: 4-Apr-2021 VOC: 11-May-2021 |
| Omicron | B.1.1.529 (Subvariants: BA 1, BA 2, BA 3, BA 4 and BA 5) | GR/484A | 21K | -+S: L452R -+S: F486V -+S: R493Q | Multiple countries, Nov-2021 | VUM: 24-Nov-2021 VOC: 26-Nov-2021 Sub BA 4/5: January 2022 |
| Codification | Vector Lineages | Clades | Nextstrain Clades | Emerging Regions and Period of Strain Detection | Date of First Detection |
|---|---|---|---|---|---|
| Lambda | C.37 | GR/452Q.V1 | 21G | Peru, Dec-2020 | 14-Jun-2021 |
| Mu | B.1.621 | GH | 21H | Colombia, Jan-2021 | 30-Aug-2021 |
| Vector Lineages | Clades | Nextstrain Clades | Emerging Regions and Period of Strain Detection | Date of First Detection |
|---|---|---|---|---|
| R.1 | GR | - | Multiple countrie Jan-2021 | 07-Apr-2021 |
| B.1.466.2 | GH | - | Indonesia, Nov-2020 | 28-Apr-2021 |
| B.1.1.318 | GR | - | Multiple countries, Jan-2021 | 02-Jun-2021 |
| B.1.1.519 | GR | 20B/S.732A | Multiple countries, Nov-2020 | 02-Jun-2021 |
| C.36.3 | GR | - | Multiple countries, Jan-2021 | 16-Jun-2021 |
| B.1.214.2 | G | - | Multiple countries, Nov-2020 | 30-Jun-2021 |
| B.1.427 B.1.429 | GH/452R.V1 | 21C | United States of America, Mar-2020 | VOI: 5-Mar-2021 VUM: 6-Jul-2021 |
| B.1.1.523 | GR | - | Multiple countries, May-2020 | 14-Jul-2021 |
| B.1.619 | G | 20A/S.126A | Multiple countries May-2020 | 14-Jul-2021 |
| B.1.620 | G | - | Multiple countries, November 2020 | 14-Jul-2021 |
| C.1.2 | GR | - | South Africa, May 2021 | 01-Sep-2021 |
| B.1.617.1 | G/452R.V3 | 21B | India, Oct-2020 | VOI: 4-Apr-2021 VUM: 20-Sep-2021 |
| B.1.526 | GH/253G.V1 | 21F | United States of America, Nov-2020 | VOI: 24-Mar-2021 VUM: 20-Sep-2021 |
| B.1.525 | G/484K.V3 | 21D | Multiple countries, Dec-2020 | VOI:17-Mar-2021 VUM: 20-Sep-2021 |
| B.1.630 | GH | - | Dominican Republic, Mar-2021 | 12-Oct-2021 |
| Coronovac Sinovac Vaccine | |
|---|---|
| General Characteristics | |
| Type of Vaccine | Inactivated SARS-CoV-2 Virus Inactivating Agent: β-propiolactone |
| Storage temperature | 2–8 °C |
| Vaccine Administration and dosages | Two doses 0.5 mL (~14 days) |
| Phase III Efficacy rate | 50.4% |
| Variant Efficacy | B.1.1.7 B.1.351; P1 |
| Sputnik V Vaccine | |
|---|---|
| General Characteristics | |
| Type of Vaccine | dsDNA Vaccine Adenovirus Delivered AD26/AD5 |
| Storage temperature | Long-Term Storage (2 years): −18 °C Short-Term Storage (3 month): 2–8 °C |
| Vaccine Administration and dosages | Two doses 0.5 mL (~28 days) |
| Phase III Efficacy rate | 91% |
| Variant Efficacy | B.1.1.7 B.1.351; P1 |
| PLATFORM | VACCINE | DEVELOPER |
|---|---|---|
| RNA vaccine | BNT162 | Pfizer/BioNTech/Fosun |
| RNA vaccine | Spikevax (COVID-19 Vaccine Moderna) | Moderna/NIAID |
| Non-replicating viral vector | Vaxzevria ChAdOx1-S | Astrazeneca AB |
| Non-replicating viral vector | Ad26.COV2-S | Janssen-Cilag International NV |
| BNT162b2 Pfizer/BioNTech Vaccine | |
|---|---|
| General Characteristics | |
| Type of Vaccine | mRNA Vaccine Lipid Nanoparticles Delivered |
| Storage temperature | Long-term Storage (6 month): −70 °C Short-Term Storage (5 days): 2–8 °C |
| Vaccine Administration and dosages | Two doses 0.3 mL (~21 days) |
| Phase III Efficacy rate | 95% |
| Variant Efficacy | B.1.1.7; B.1.135; P1; B.1.429; B.1.617.2 |
| m-RNA-1273 Moderna/NIAID Vaccine | |
|---|---|
| General Characteristics | |
| Type of Vaccine | mRNA Vaccine Lipid Nanoparticles Delivered |
| Storage temperature | Long-Term Storage (6 month): −20 °C Short-Term Storage (30 days): 2–8 °C |
| Vaccine Administration and dosages | Two doses 0.5 mL (~28 days) |
| Phase III Efficacy rate | 94.5% |
| Variant Efficacy | B.1.1.7; B.1.135; P.1; B.1.429 B.1.617.2 |
| AZD1222 (ChAdOx1 nCoV-19) Astrazeneca Vaccine | |
|---|---|
| General Characteristics | |
| Type of Vaccine | dsDNA Vaccine Adenovirus Delivered |
| Storage temperature | about 2–8 °C (6 month) |
| Vaccine Administration and dosages | Two doses 0.5 mL (12 weeks) |
| Phase III Efficacy rate | 81.3% |
| Variant Efficacy | B.1.1.7 B.1.135 P.1B.1.429 B.1.617.2 |
| Ad26.COV-S Janssen Pharmaceutical Vaccine | |
|---|---|
| General Characteristics | |
| Type of Vaccine | dsDNA Vaccine Adenovirus Delivered |
| Storage temperature | Long-Term Storage (2 years): −20 °C Short-Term Storage (3 month): 2–8 °C |
| Vaccine Administration and dosages | Single dose 0.5 mL |
| Phase III Efficacy rate | 66% |
| Variant Efficacy | 72% B.1.1.7. 57% B.1.135 & P.1 B.1.617.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inchingolo, A.D.; Malcangi, G.; Ceci, S.; Patano, A.; Corriero, A.; Vimercati, L.; Azzollini, D.; Marinelli, G.; Coloccia, G.; Piras, F.; et al. Effectiveness of SARS-CoV-2 Vaccines for Short- and Long-Term Immunity: A General Overview for the Pandemic Contrast. Int. J. Mol. Sci. 2022, 23, 8485. https://doi.org/10.3390/ijms23158485
Inchingolo AD, Malcangi G, Ceci S, Patano A, Corriero A, Vimercati L, Azzollini D, Marinelli G, Coloccia G, Piras F, et al. Effectiveness of SARS-CoV-2 Vaccines for Short- and Long-Term Immunity: A General Overview for the Pandemic Contrast. International Journal of Molecular Sciences. 2022; 23(15):8485. https://doi.org/10.3390/ijms23158485
Chicago/Turabian StyleInchingolo, Alessio Danilo, Giuseppina Malcangi, Sabino Ceci, Assunta Patano, Alberto Corriero, Luigi Vimercati, Daniela Azzollini, Grazia Marinelli, Giovanni Coloccia, Fabio Piras, and et al. 2022. "Effectiveness of SARS-CoV-2 Vaccines for Short- and Long-Term Immunity: A General Overview for the Pandemic Contrast" International Journal of Molecular Sciences 23, no. 15: 8485. https://doi.org/10.3390/ijms23158485
APA StyleInchingolo, A. D., Malcangi, G., Ceci, S., Patano, A., Corriero, A., Vimercati, L., Azzollini, D., Marinelli, G., Coloccia, G., Piras, F., Barile, G., Settanni, V., Mancini, A., De Leonardis, N., Garofoli, G., Palmieri, G., Isacco, C. G., Rapone, B., Scardapane, A., ... Inchingolo, F. (2022). Effectiveness of SARS-CoV-2 Vaccines for Short- and Long-Term Immunity: A General Overview for the Pandemic Contrast. International Journal of Molecular Sciences, 23(15), 8485. https://doi.org/10.3390/ijms23158485
























