Dermatopontin Influences the Development of Obesity-Associated Colon Cancer by Changes in the Expression of Extracellular Matrix Proteins
Abstract
:1. Introduction
2. Results
2.1. Colon Cancer Decreases DPT Circulating Levels and Its mRNA Expression in the Colon
2.2. Influence of Inflammation-Related Factors in the Expression of DPT in HT-29 Cells
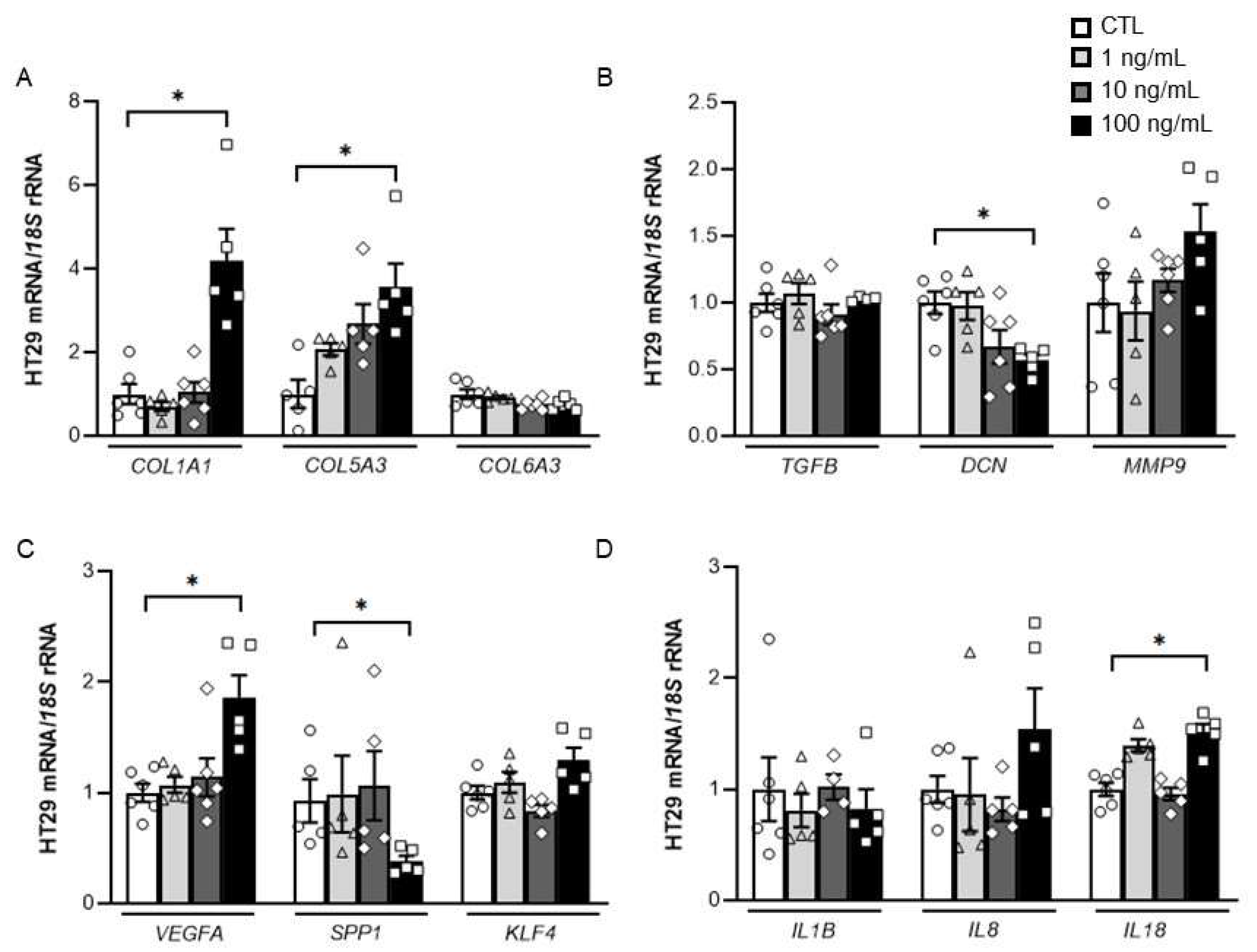
2.3. Impact of DPT on the Expression of Key ECM Remodelling-Related Genes in HT-29 Cells
2.4. Adipocyte-Conditioned Media Upregulate DPT Expression Levels in Tumour Cells
3. Discussion
4. Materials and Methods
4.1. Patient Selection
4.2. Analytical Measurements
4.3. RNA Isolation and Real-Time PCR
4.4. Cell Cultures
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Sauer, A.G.; Fedewa, S.A.; Butterly, L.F.; Anderson, J.C.; Cercek, A.; Smith, R.A.; Jemal, A. Colorectal cancer statistics. CA Cancer J. Clin. 2020, 70, 145–164. [Google Scholar] [CrossRef]
- Arnold, M.; Pandeya, N.; Byrnes, G.; Renehan, A.G.; Stevens, G.A.; Ezzati, M.; Ferlay, J.; Miranda, J.J.; Romieu, I.; Dikshit, R.; et al. Global burden of cancer attributable to high body-mass index in 2012: A population-based study. Lancet Oncol. 2015, 16, 36–46. [Google Scholar] [CrossRef]
- Collaborators, G.B.D.C.C. The global, regional, and national burden of colorectal cancer and its attributable risk factors in 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study. Lancet Gastroenterol. Hepatol. 2019, 4, 913–933. [Google Scholar]
- Song, M.; Giovannucci, E. Preventable Incidence and Mortality of Carcinoma Associated With Lifestyle Factors Among White Adults in the United States. JAMA Oncol. 2016, 2, 1154–1161. [Google Scholar] [CrossRef]
- Fleming, C.A.; O’Connell, E.P.; Kavanagh, R.G.; O’Leary, D.P.; Twomey, M.; Corrigan, M.A.; Wang, J.H.; Maher, M.M.; O’Connor, O.J.; Redmond, H.P. Body Composition, Inflammation, and 5-Year Outcomes in Colon Cancer. JAMA Netw. Open 2021, 4, e2115274. [Google Scholar] [CrossRef]
- Kim, M.; Lee, C.; Park, J. Extracellular matrix remodeling facilitates obesity-associated cancer progression. Trends Cell Biol. 2022. [Google Scholar] [CrossRef]
- Crewe, C.; An, Y.A.; Scherer, P.E. The ominous triad of adipose tissue dysfunction: Inflammation, fibrosis, and impaired angiogenesis. J. Clin. Investig. 2017, 127, 74–82. [Google Scholar] [CrossRef]
- Herrera, J.; Henke, C.A.; Bitterman, P. Extracellular matrix as a driver of progressive fibrosis. J. Clin. Investig. 2018, 128, 45–53. [Google Scholar] [CrossRef]
- Huang, J.; Zhang, L.; Wan, D.; Zhou, L.; Zheng, S.; Lin, S.; Qiao, Y. Extracellular matrix and its therapeutic potential for cancer treatment. Signal Transduct. Target. Ther. 2021, 6, 1–24. [Google Scholar] [CrossRef]
- Catalán, V.; Gomez-Ambrosi, J.; Rodríguez, A.; Frühbeck, G. Role of extracellular matrix remodelling in adipose tissue pathophysiology: Relevance in the development of obesity. Histol. Histopathol. 2012, 27, 1515–1528. [Google Scholar]
- Unamuno, X.; Gómez-Ambrosi, J.; Becerril, S.; Álvarez-Cienfuegos, F.J.; Ramírez, B.; Rodríguez, A.; Ezquerro, S.; Valentí, V.; Moncada, R.; Mentxaka, A.; et al. Changes in mechanical properties of adipose tissue after bariatric surgery driven by extracellular matrix remodelling and neovascularization are associated with metabolic improvements. Acta Biomater. 2022, 141, 264–279. [Google Scholar] [CrossRef] [PubMed]
- Han, C.Y.; Kang, I.; Harten, I.A.; Gebe, J.A.; Chan, C.K.; Omer, M.; Alonge, K.M.; Hartigh, L.J.D.; Kjerulf, D.G.; Goodspeed, L.; et al. Adipocyte-Derived Versican and Macrophage-Derived Biglycan Control Adipose Tissue Inflammation in Obesity. Cell Rep. 2020, 31, 107818. [Google Scholar] [CrossRef] [PubMed]
- Hepler, C.; Shan, B.; Zhang, Q.; Henry, G.H.; Shao, M.; Vishvanath, L.; Ghaben, A.L.; Mobley, A.B.; Strand, D.; Hon, G.C.; et al. Identification of functionally distinct fibro-inflammatory and adipogenic stromal subpopulations in visceral adipose tissue of adult mice. eLife 2018, 7, e39636. [Google Scholar] [CrossRef]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 2014, 15, 786–801. [Google Scholar] [CrossRef] [PubMed]
- Neame, P.J.; Choi, H.U.; Rosenberg, L.C. The Isolation and Primary Structure of a 22-kDa Extracellular Matrix Protein from Bovine Skin. J. Biol. Chem. 1989, 264, 5474–5479. [Google Scholar] [CrossRef]
- Unamuno, X.; Gómez-Ambrosi, J.; Ramírez, B.; Rodríguez, A.; Becerril, S.; Valentí, V.; Moncada, R.; Silva, C.; Salvador, J.; Frühbeck, G.; et al. Dermatopontin, A Novel Adipokine Promoting Adipose Tissue Extracellular Matrix Remodelling and Inflammation in Obesity. J. Clin. Med. 2020, 9, 1069. [Google Scholar] [CrossRef]
- Takeda, U.; Utani, A.; Wu, J.; Shinkai, H.; Adachi, E.; Koseki, H.; Taniguchi, M.; Matsumoto, T.; Ohashi, T.; Sato, M. Targeted Disruption of Dermatopontin Causes Abnormal Collagen Fibrillogenesis. J. Investig. Dermatol. 2002, 119, 678–683. [Google Scholar] [CrossRef]
- Lefebvre, P.; Lalloyer, F.; Bauge, E.; Pawlak, M.; Gheeraert, C.; Dehondt, H.; Vanhoutte, J.; Woitrain, E.; Hennuyer, N.; Mazuy, C.; et al. Interspecies NASH disease activity whole-genome profiling identifies a fibrogenic role of PPARa-regulated dermatopontin. JCI Insight 2017, 2, e92264. [Google Scholar] [CrossRef]
- Okamoto, O.; Fujiwara, S. Dermatopontin, a Novel Player in the Biology of the Extracellular Matrix. Connect. Tissue Res. 2006, 47, 177–189. [Google Scholar] [CrossRef]
- Okamoto, O.; Fujiwara, S.; Abe, M.; Sato, Y. Dermatopontin interacts with transforming growth factor b and enhances its biological activity. Biochem J. 1999, 337, 537–541. [Google Scholar] [CrossRef]
- Okamoto, O.; Hozumi, K.; Katagiri, F.; Takahashi, N.; Sumiyoshi, H.; Matsuo, N.; Yoshioka, H.; Nomizu, M.; Fujiwara, S. Dermatopontin promotes epidermal keratinocyte adhesion via a3b1 integrin and a proteoglycan receptor. Biochemistry 2010, 49, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Sadr-Nabavi, A.; Bouromand-Noughabi, S.; Tayebi-Meybodi, N.; Dadkhah, K.; Amini, N.; Meindl, A.; Abbaszadegan, M.R. Non-collagenous extracellular matrix protein dermatopontin may play a role as another component of trans-forming growth factor-b signaling pathway in colon carcinogenesis. Iran J. Basic Med. Sci. 2021, 24, 444–450. [Google Scholar] [PubMed]
- Guo, Y.; Li, H.; Guan, H.; Ke, W.; Liang, W.; Xiao, H.; Li, Y. Dermatopontin inhibits papillary thyroid cancer cell proliferation through MYC repression. Mol. Cell. Endocrinol. 2019, 480, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Qiu, J.; He, G.; Geng, C.; He, W.; Liu, C.; Cai, D.; Pan, H.; Tian, Q. Dermatopontin inhibits WNT signaling pathway via CXXC finger protein 4 in hepatocellular carcinoma. J. Cancer 2020, 11, 6288–6298. [Google Scholar] [CrossRef]
- Dahl, E.; Sadr-Nabavi, A.; Klopocki, E.; Betz, B.; Grube, S.; Kreutzfeld, R.; Himmelfarb, M.; An, H.-X.; Gelling, S.; Klaman, I.; et al. Systematic identification and molecular characterization of genes differentially expressed in breast and ovarian cancer. J. Pathol. 2005, 205, 21–28. [Google Scholar] [CrossRef]
- Yamatoji, M.; Kasamatsu, A.; Kouzu, Y.; Koike, H.; Sakamoto, Y.; Ogawara, K.; Shiiba, M.; Tanzawa, H.; Uzawa, K. Dermatopontin: A potential predictor for metastasis of human oral cancer. Int. J. Cancer 2012, 130, 2903–2911. [Google Scholar] [CrossRef]
- Avgerinos, K.I.; Spyrou, N.; Mantzoros, C.S.; Dalamaga, M. Obesity and cancer risk: Emerging biological mechanisms and perspectives. Metabolism 2019, 92, 121–135. [Google Scholar] [CrossRef]
- Catalán, V.; Aviles-Olmos, I.; Rodríguez, A.; Becerril, S.; Fernández-Formoso, J.A.; Kiortsis, D.; Portincasa, P.; Gómez-Ambrosi, J.; Frühbeck, G. Time to consider the “Exposome Hypothesis” in the development of the obesity pandemic. Nutrients 2022, 14, 1597. [Google Scholar] [CrossRef]
- Ligibel, J.A.; Alfano, C.M.; Courneya, K.S.; Demark-Wahnefried, W.; Burger, R.A.; Chlebowski, R.T.; Fabian, C.J.; Gucalp, A.; Hershman, D.L.; Hudson, M.M.; et al. American Society of Clinical Oncology Position Statement on Obesity and Cancer. J. Clin. Oncol. 2014, 32, 3568–3574. [Google Scholar] [CrossRef]
- Yarnoz-Esquiroz, P.; Olazaran, L.; Aguas-Ayesa, M.; Perdomo, C.M.; García-Goñi, M.; Silva, C.; Fernández-Formoso, J.A.; Escalada, J.; Montecucco, F.; Portincasa, P.; et al. ‘Obesities’: Position statement on a complex disease entity with multifaceted drivers. Eur. J. Clin. Invest. 2022, 52, e13811. [Google Scholar] [CrossRef]
- Calle, E.E.; Rodriguez, C.; Walker-Thurmond, K.; Thun, M.J. Overweight, Obesity, and Mortality from Cancer in a Prospectively Studied Cohort of U.S. Adults. N. Engl. J. Med. 2003, 348, 1625–1638. [Google Scholar] [CrossRef]
- Catalán, V.; Gómez-Ambrosi, J.; Rodríguez, A.; Frühbeck, G. Adipose tissue immunity and cancer. Front. Physiol. 2013, 4, 275. [Google Scholar] [CrossRef]
- Frühbeck, G.; Gómez-Ambrosi, J. Control of body weight: A physiologic and transgenic perspective. Diabetologia 2003, 46, 143–172. [Google Scholar] [CrossRef]
- Pérez-Hernandez, A.I.; Catalán, V.; Gómez-Ambrosi, J.; Rodríguez, A.; Frühbeck, G. Mechanisms linking excess adiposity and carcinogenesis promotion. Front Endocrinol. 2014, 5, 65. [Google Scholar]
- Quail, D.F.; Dannenberg, A.J. The obese adipose tissue microenvironment in cancer development and progression. Nat. Rev. Endocrinol. 2019, 15, 139–154. [Google Scholar] [CrossRef]
- Li, X.; Feng, P.; Ou, J.; Luo, Z.; Dai, P.; Wei, D.; Zhang, C. Dermatopontin is expressed in human liver and is downregulated in hepatocellular carcinoma. Biochemistry 2009, 74, 979–985. [Google Scholar] [CrossRef]
- Akhurst, R.J.; Derynck, R. TGF-b signaling in cancer-a double-edged sword. Trends Cell Biol. 2001, 11, S44–S51. [Google Scholar]
- Derynck, R.; Akhurst, R.J.; Balmain, A. TGF-b signaling in tumor suppression and cancer progression. Nat Genet. 2001, 29, 117–129. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Kramer, A.; Astuti, Y.; Elfstrum, A.; Lehrke, M.J.; Tolar, J.; Blazar, B.R.; Blake, A.; Taisto, M.; Furcich, J.W.; Nolan, E.E.; et al. An irradiated marrow niche reveals a small non-collagenous protein mediator of homing, dermatopontin. Blood Adv. 2021, 5, 3609–3622. [Google Scholar] [CrossRef]
- Kuroda, K.; Okamoto, O.; Shinkai, H. Dermatopontin expression is decreased in hypertrophic scar and systemic sclerosis skin fibroblasts and is regulated by transforming growth factor-b1, interleukin-4, and matrix collagen. J. Investig. Dermatol. 1999, 112, 706–710. [Google Scholar] [CrossRef]
- Xu, S.; Xu, H.; Wang, W.; Li, S.; Li, H.; Li, T.; Zhang, W.; Yu, X.; Liu, L. The role of collagen in cancer: From bench to bedside. J. Transl. Med. 2019, 17, 309. [Google Scholar] [CrossRef]
- Okamoto, O.; Suzuki, Y.; Kimura, S.; Shinkai, H. Extracellular Matrix 22-kDa Protein Interacts with Decorin Core Protein and Is Expressed in Cutaneous Fibrosis. J. Biochem. 1996, 119, 106–114. [Google Scholar] [CrossRef]
- MacBeath, J.; Shackleton, D.; Hulmes, D. Tyrosine-rich acidic matrix protein (TRAMP) accelerates collagen fibril formation in vitro. J. Biol. Chem. 1993, 268, 19826–19832. [Google Scholar] [CrossRef]
- Vogel, K.G.; Paulsson, M.; Heinegård, D. Specific inhibition of type I and type II collagen fibrillogenesis by the small proteoglycan of tendon. Biochem. J. 1984, 223, 587–597. [Google Scholar] [CrossRef]
- Mao, L.; Yang, J.; Yue, J.; Chen, Y.; Zhou, H.; Fan, D.; Zhang, Q.; Buraschi, S.; Iozzo, R.V.; Bi, X. Decorin deficiency promotes epithelial-mesenchymal transition and colon cancer metastasis. Matrix Biol. 2021, 95, 1–14. [Google Scholar] [CrossRef]
- Krishnaswamy, V.R.; Balaguru, U.M.; Chatterjee, S.; Korrapati, P.S. Dermatopontin augments angiogenesis and modulates the expression of transforming growth factor b1 and integrin a3b1 in endothelial cells. Eur. J. Cell Biol. 2017, 96, 266–275. [Google Scholar] [CrossRef]
- Amilca-Seba, K.; Sabbah, M.; Larsen, A.; Denis, J. Osteopontin as a Regulator of Colorectal Cancer Progression and Its Clinical Applications. Cancers 2021, 13, 3793. [Google Scholar] [CrossRef]
- Zhao, H.; Chen, Q.; Alam, A.; Cui, J.; Suen, K.C.; Soo, A.P.; Eguchi, S.; Gu, J.; Ma, D. The role of osteopontin in the progression of solid organ tumour. Cell Death Dis. 2018, 9, 356. [Google Scholar] [CrossRef]
- Catalán, V.; Gómez-Ambrosi, J.; Rodríguez, A.; Ramírez, B.; Izaguirre, M.; Hernández-Lizoain, J.L.; Baixauli, J.; Martí, P.; Valentí, V.; Moncada, R.; et al. Increased obesity-associated circulating levels of the extracellular matrix proteins os-teopontin, chitinase-3 like-1 and tenascin C are associated with colon cancer. PLoS ONE 2016, 11, e0162189. [Google Scholar]
- Johnston, N.I.F.; Gunasekharan, V.K.; Ravindranath, A.; O’Connell, C.; Johnston, P.G.; El-Tanani, M.K. Osteopontin as a target for cancer therapy. Front. Biosci. 2008, 13, 4361–4372. [Google Scholar] [CrossRef]
- Ferrara, N.; Gerber, H.-P.; LeCouter, J. The biology of VEGF and its receptors. Nat. Med. 2003, 9, 669–676. [Google Scholar] [CrossRef]
- Catalán, V.; Gómez-Ambrosi, J.; Rodríguez, A.; Ramírez, B.; Silva, C.; Rotellar, F.; Hernández-Lizoain, J.L.; Baixauli, J.; Valentí, V.; Pardo, F.; et al. Up-regulation of the novel proinflammatory adipokines lipocalin-2, chitinase-3 like-1 and osteopontin as well as angiogenic-related factors in visceral adipose tissue of patients with colon cancer. J. Nutr. Biochem. 2011, 22, 634–641. [Google Scholar] [CrossRef]
- Dirat, B.; Bochet, L.; Dabek, M.; Daviaud, D.; Dauvillier, S.; Majed, B.; Wang, Y.Y.; Meulle, A.; Salles, B.; Le Gonidec, S.; et al. Cancer-Associated Adipocytes Exhibit an Activated Phenotype and Contribute to Breast Cancer Invasion. Cancer Res. 2011, 71, 2455–2465. [Google Scholar] [CrossRef]
- Frühbeck, G.; Mentxaka, A.; Ahechu, P.; Gómez-Ambrosi, J.; Ramírez, B.; Becerril, S.; Rodríguez, A.; Unamuno, X.; Cienfuegos, J.A.; Casado, M.; et al. The Differential Expression of the Inflammasomes in Adipose Tissue and Colon Influences the Development of Colon Cancer in a Context of Obesity by Regulating Intestinal Inflammation. J. Inflamm. Res. 2021, 14, 6431–6446. [Google Scholar] [CrossRef]
- Pulido, M.R.; Diaz-Ruiz, A.; Jiménez-Gómez, Y.; Garcia-Navarro, S.; Gracia-Navarro, F.; Tinahones, F.; López-Miranda, J.; Frühbeck, G.; Vázquez-Martínez, R.; Malagón, M.M. Rab18 Dynamics in Adipocytes in Relation to Lipogenesis, Lipolysis and Obesity. PLoS ONE 2011, 6, e22931. [Google Scholar] [CrossRef]
- Gómez-Ambrosi, J.; Silva, C.; Catalán, V.; Rodríguez, A.; Galofré, J.C.; Escalada, J.; Valentí, V.; Rotellar, F.; Romero, S.; Ramírez, B.; et al. Clinical Usefulness of a New Equation for Estimating Body Fat. Diabetes Care 2012, 35, 383–388. [Google Scholar] [CrossRef]
- Fortuño, A.; Rodríguez, A.; Gómez-Ambrosi, J.; Muniz, P.; Salvador, J.; Diez, J.; Frühbeck, G. Leptin inhibits an-giotensin II-induced intracellular calcium increase and vasoconstriction in the rat aorta. Endocrinology 2002, 143, 3555–3560. [Google Scholar] [CrossRef]
- Catalán, V.; Gómez-Ambrosi, J.; Rodríguez, A.; Ramírez, B.; Ortega, V.A.; Hernández-Lizoain, J.L.; Baixauli, J.; Becerril, S.; Rotellar, F.; Valentí, V.; et al. IL-32α-induced inflammation constitutes a link between obesity and colon cancer. OncoImmunology 2017, 6, e1328338. [Google Scholar] [CrossRef]
- Rodríguez, A.; Gomez-Ambrosi, J.; Catalan, V.; Rotellar, F.; Valentí, V.; Silva, C.; Mugueta, M.D.C.; Pulido, M.R.; Vázquez, R.; Salvador, J.; et al. The ghrelin O-acyltransferase–ghrelin system reduces TNF-α-induced apoptosis and autophagy in human visceral adipocytes. Diabetologia 2012, 55, 3038–3050. [Google Scholar] [CrossRef]



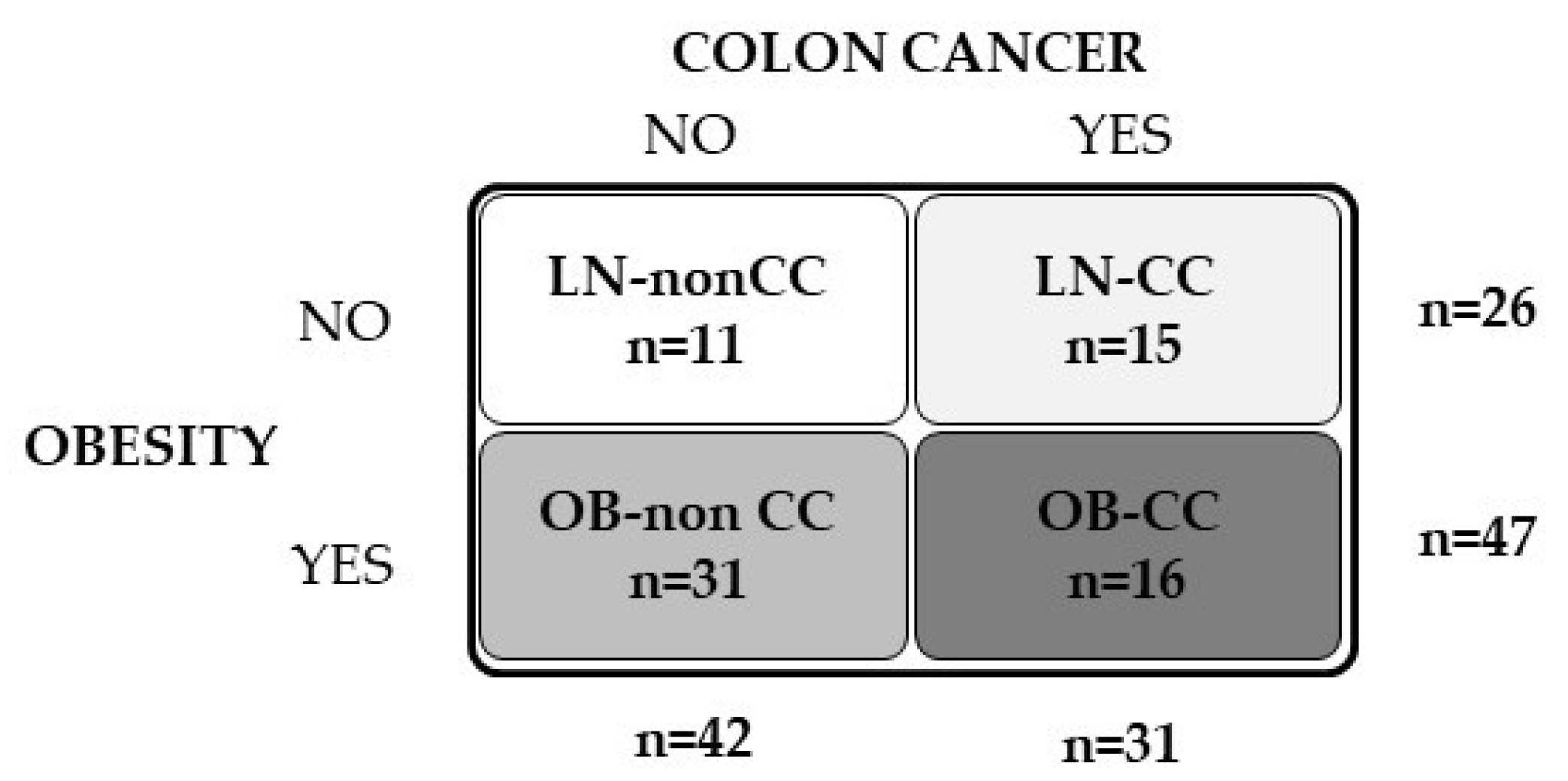
| Lean | OB | ||||||
|---|---|---|---|---|---|---|---|
| non-CC | CC | non-CC | CC | p OB | p CC | p OBxCC | |
| n (male, female) | 11 (7, 4) | 15 (8, 7) | 31 (19, 12) | 16 (9, 7) | |||
| Age (years) | 53 ± 3 | 63 ± 3 | 55 ± 1 | 62 ± 3 | 0.518 | <0.001 | 0.925 |
| Body weight (kg) | 65.4 ± 1.9 | 63.4 ± 1.8 | 84.9 ± 2.1 | 81.1 ± 2.9 | <0.001 | 0.303 | 0.753 |
| Body mass index (kg/m2) | 23.1 ± 0.2 | 22.5 ± 0.4 | 36.7 ± 0.7 | 34.2 ± 0.8 | <0.001 | 0.065 | 0.772 |
| Estimated body fat (%) | 27.8 ± 2.2 | 29.1 ± 1.4 | 36.7 ± 1.2 | 34.2 ± 1.8 | <0.001 | 0.268 | 0.369 |
| Waist circumference (cm) | 83 ± 2 | 80 ± 2 | 100 ± 2 | 115 ± 7 | <0.001 | 0.270 | 0.228 |
| Fasting glucose (mg/dL) | 103 ± 5 | 142 ± 12 | 113 ± 5 | 127 ± 11 | 0.775 | 0.020 | 0.221 |
| Free fatty acids (mg/dL) | 11.7 ± 1.5 | 26.5 ± 2.4 | 15.2 ±1.2 | 22.6 ± 2.1 | 0.893 | <0.001 | 0.086 |
| Triglycerides (mg/dL) | 93 ± 15 | 116 ± 11 | 118 ± 13 | 151 ± 28 | 0.079 | 0.398 | 0.324 |
| CRP (mg/L) | 0.90 ± 0.07 | 1.39 ± 0.82 | 1.63 ± 0.09 | 9.64 ± 1.49 *** | <0.001 | <0.001 | <0.001 |
| Fibrinogen (mg/dL) | 330 ± 18 | 273 ± 26 | 303 ± 20 | 447 ± 48 ‡ | 0.203 | 0.398 | 0.017 |
| CEA (ng/mL) | 1.58 ± 0.32 | 2.55 ± 0.44 | 1.68 ± 0.28 | 8.41 ± 2.60 | 0.267 | 0.021 | 0.401 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Catalán, V.; Domench, P.; Gómez-Ambrosi, J.; Ramírez, B.; Becerril, S.; Mentxaka, A.; Rodríguez, A.; Valentí, V.; Moncada, R.; Baixauli, J.; et al. Dermatopontin Influences the Development of Obesity-Associated Colon Cancer by Changes in the Expression of Extracellular Matrix Proteins. Int. J. Mol. Sci. 2022, 23, 9222. https://doi.org/10.3390/ijms23169222
Catalán V, Domench P, Gómez-Ambrosi J, Ramírez B, Becerril S, Mentxaka A, Rodríguez A, Valentí V, Moncada R, Baixauli J, et al. Dermatopontin Influences the Development of Obesity-Associated Colon Cancer by Changes in the Expression of Extracellular Matrix Proteins. International Journal of Molecular Sciences. 2022; 23(16):9222. https://doi.org/10.3390/ijms23169222
Chicago/Turabian StyleCatalán, Victoria, Paula Domench, Javier Gómez-Ambrosi, Beatriz Ramírez, Sara Becerril, Amaia Mentxaka, Amaia Rodríguez, Víctor Valentí, Rafael Moncada, Jorge Baixauli, and et al. 2022. "Dermatopontin Influences the Development of Obesity-Associated Colon Cancer by Changes in the Expression of Extracellular Matrix Proteins" International Journal of Molecular Sciences 23, no. 16: 9222. https://doi.org/10.3390/ijms23169222








