CCL5 Levels Predict Stroke Volume Growth in Acute Ischemic Stroke and Significantly Diminish in Hemorrhagic Stroke Patients
Abstract
:1. Introduction
2. Results
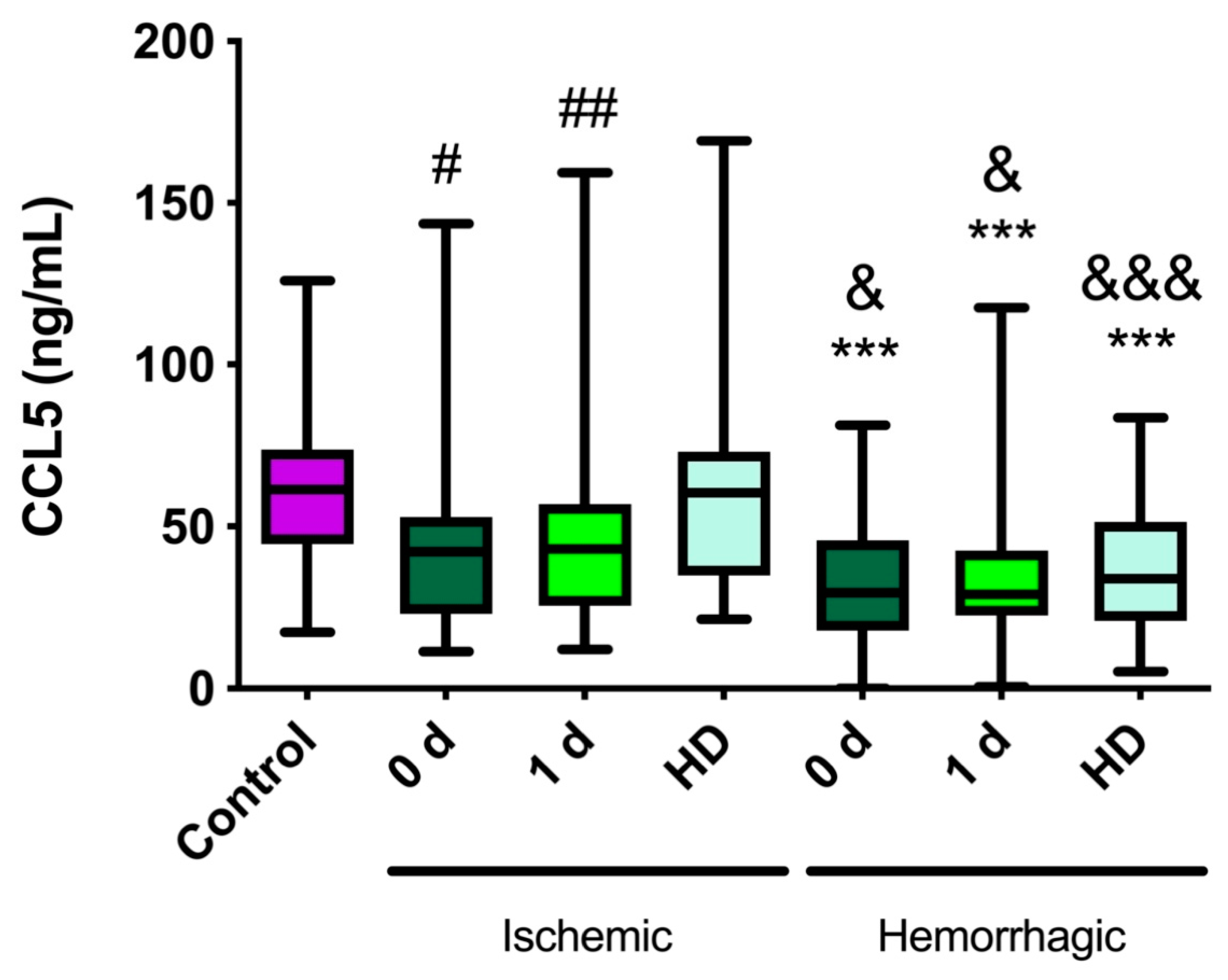
2.1. CCL5 Levels Are Lower in Stroke Patients Than in Healthy Controls
2.2. CCL5 Levels Are Lower in Hemorrhagic Stroke Patients Than in Ischemic Stroke Patients
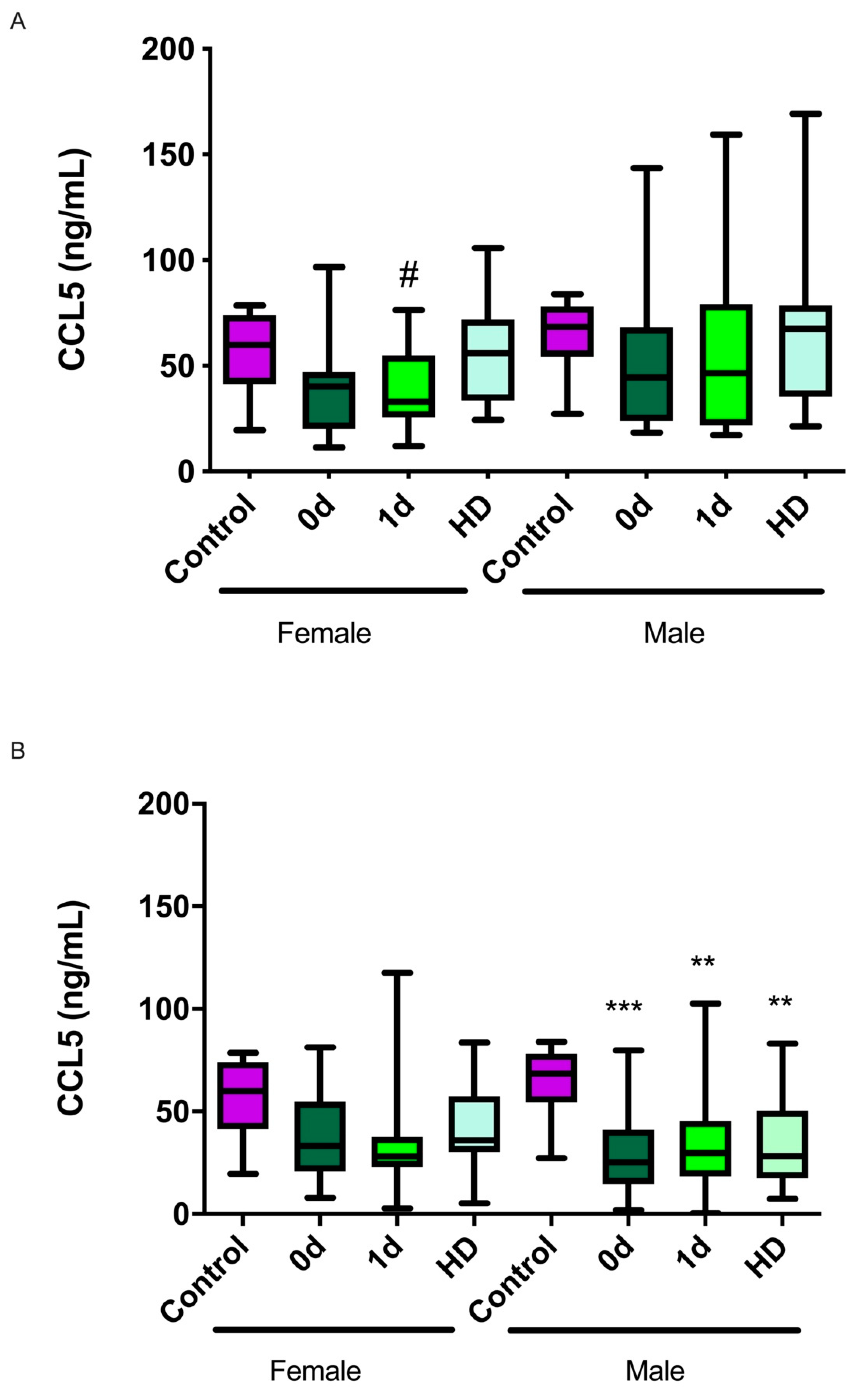
2.3. CCL5 Levels and Their Relationship with Sex
2.4. CCL5 Levels Are Lower in Cardioembolic and Lacunar Subtypes of Ischemic Stroke
2.5. CCL5 Levels and Their Association with Infarct Volume or Hematoma Size Growth
2.6. CCL5 Levels and Their Relationship with NIHSS at Admission
2.7. CCL5 Levels and Their Relationship with Neurological Severity at 3 Months
3. Discussion
4. Material and Methods
4.1. Patients
4.2. Variables of the Study
4.3. Determination of CCL5 Levels
4.4. Statistical Analysis
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Gorelick, P.B. The global burden of stroke: Persistent and disabling. Lancet Neurol. 2019, 18, 417–418. [Google Scholar] [CrossRef]
- Feigin, V.L.; Norrving, B.; Mensah, G.A. Global Burden of Stroke. Circ. Res. 2017, 120, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Donkor, E.S. Stroke in the 21(st) Century: A Snapshot of the Burden, Epidemiology, and Quality of Life. Stroke Res. Treat. 2018, 2018, 3238165. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, E.J.; Virani, S.S.; Callaway, C.W.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Chiuve, S.E.; Cushman, M.; Delling, F.N.; Deo, R.; et al. Heart Disease and Stroke Statistics-2018 Update: A Report From the American Heart Association. Circulation 2018, 137, e67–e492. [Google Scholar] [CrossRef] [PubMed]
- Lackland, D.T.; Roccella, E.J.; Deutsch, A.F.; Fornage, M.; George, M.G.; Howard, G.; Kissela, B.M.; Kittner, S.J.; Lichtman, J.H.; Lisabeth, L.D.; et al. Factors influencing the decline in stroke mortality: A statement from the American Heart Association/American Stroke Association. Stroke 2014, 45, 315–353. [Google Scholar] [CrossRef] [PubMed]
- Wein, T.; Lindsay, M.P.; Cote, R.; Foley, N.; Berlingieri, J.; Bhogal, S.; Bourgoin, A.; Buck, B.H.; Cox, J.; Davidson, D.; et al. Canadian stroke best practice recommendations: Secondary prevention of stroke, sixth edition practice guidelines, update 2017. Int. J. Stroke 2018, 13, 420–443. [Google Scholar] [CrossRef]
- Jickling, G.C.; Sharp, F.R. Biomarker panels in ischemic stroke. Stroke 2015, 46, 915–920. [Google Scholar] [CrossRef]
- Wang, Z.; Lin, Y.; Liu, Y.; Chen, Y.; Wang, B.; Li, C.; Yan, S.; Wang, Y.; Zhao, W. Serum Uric Acid Levels and Outcomes After Acute Ischemic Stroke. Mol. Neurobiol. 2016, 53, 1753–1759. [Google Scholar] [CrossRef]
- Sarikaya, H.; Ferro, J.; Arnold, M. Stroke prevention--medical and lifestyle measures. Eur. Neurol. 2015, 73, 150–157. [Google Scholar] [CrossRef]
- Boyd, L.A.; Hayward, K.S.; Ward, N.S.; Stinear, C.M.; Rosso, C.; Fisher, R.J.; Carter, A.R.; Leff, A.P.; Copland, D.A.; Carey, L.M.; et al. Biomarkers of stroke recovery: Consensus-based core recommendations from the Stroke Recovery and Rehabilitation Roundtable. Int. J. Stroke 2017, 12, 480–493. [Google Scholar] [CrossRef] [Green Version]
- Karin, M.; Clevers, H. Reparative inflammation takes charge of tissue regeneration. Nature 2016, 529, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Gelderblom, M.; Leypoldt, F.; Steinbach, K.; Behrens, D.; Choe, C.U.; Siler, D.A.; Arumugam, T.V.; Orthey, E.; Gerloff, C.; Tolosa, E.; et al. Temporal and spatial dynamics of cerebral immune cell accumulation in stroke. Stroke 2009, 40, 1849–1857. [Google Scholar] [CrossRef] [PubMed]
- Planas, A.M. Role of Immune Cells Migrating to the Ischemic Brain. Stroke 2018, 49, 2261–2267. [Google Scholar] [CrossRef] [PubMed]
- García-Berrocoso, T.; Giralt, D.; Llombart, V.; Bustamante, A.; Penalba, A.; Flores, A.; Ribó, M.; Molina, C.A.; Rosell, A.; Montaner, J. Chemokines after human ischemic stroke: From neurovascular unit to blood using protein arrays. Transl. Proteom. 2014, 3, 1–9. [Google Scholar] [CrossRef]
- Rostene, W.; Dansereau, M.A.; Godefroy, D.; Van Steenwinckel, J.; Reaux-Le Goazigo, A.; Melik-Parsadaniantz, S.; Apartis, E.; Hunot, S.; Beaudet, N.; Sarret, P. Neurochemokines: A menage a trois providing new insights on the functions of chemokines in the central nervous system. J. Neurochem. 2011, 118, 680–694. [Google Scholar] [CrossRef]
- Rostene, W.; Guyon, A.; Kular, L.; Godefroy, D.; Barbieri, F.; Bajetto, A.; Banisadr, G.; Callewaere, C.; Conductier, G.; Rovere, C.; et al. Chemokines and chemokine receptors: New actors in neuroendocrine regulations. Front. Neuroendocrinol. 2011, 32, 10–24. [Google Scholar] [CrossRef]
- Ceulemans, A.G.; Zgavc, T.; Kooijman, R.; Hachimi-Idrissi, S.; Sarre, S.; Michotte, Y. The dual role of the neuroinflammatory response after ischemic stroke: Modulatory effects of hypothermia. J. Neuroinflamm. 2010, 7, 74. [Google Scholar] [CrossRef]
- Badacz, R.; Podolec, J.; Przewlocki, T.; Siedlinski, M.; Jozefczuk, E.; Oleksy, H.; Baran, J.; Pieniazek, P.; Zmudka, K.; Kablak-Ziembicka, A. The role of chemokine CCL5/RANTES and metalloproteinase-9 as inflammatory modulators in symptomatic internal carotid artery stenosis. J. Physiol. Pharmacol. 2019, 70, 929. [Google Scholar] [CrossRef]
- Kong, Q.; Ma, X.; Lyu, J.X.; Wang, C.; Du, X.Y.; Guan, Y.Q. Plasma RANTES level is correlated with cardio-cerebral atherosclerosis burden in patients with ischemic cerebrovascular disease. Chronic. Dis. Transl. Med. 2020, 6, 46–54. [Google Scholar] [CrossRef]
- Montecucco, F.; Lenglet, S.; Gayet-Ageron, A.; Bertolotto, M.; Pelli, G.; Palombo, D.; Pane, B.; Spinella, G.; Steffens, S.; Raffaghello, L.; et al. Systemic and intraplaque mediators of inflammation are increased in patients symptomatic for ischemic stroke. Stroke 2010, 41, 1394–1404. [Google Scholar] [CrossRef] [Green Version]
- Tokami, H.; Ago, T.; Sugimori, H.; Kuroda, J.; Awano, H.; Suzuki, K.; Kiyohara, Y.; Kamouchi, M.; Kitazono, T. RANTES has a potential to play a neuroprotective role in an autocrine/paracrine manner after ischemic stroke. Brain Res. 2013, 1517, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Zaremba, J.; Ilkowski, J.; Losy, J. Serial measurements of levels of the chemokines CCL2, CCL3 and CCL5 in serum of patients with acute ischaemic stroke. Folia Neuropathol. 2006, 44, 282–289. [Google Scholar] [PubMed]
- Liesz, A.; Suri-Payer, E.; Veltkamp, C.; Doerr, H.; Sommer, C.; Rivest, S.; Giese, T.; Veltkamp, R. Regulatory T cells are key cerebroprotective immunomodulators in acute experimental stroke. Nat. Med. 2009, 15, 192–199. [Google Scholar] [CrossRef]
- Qin, X.; He, Z.; Zhao, D.; Li, L.; Yuan, L. The RANTES gene promoter polymorphisms are associated with the risk of atherothrombotic cerebral infarction in Northern Han Chinese. Clin. Chim. Acta 2011, 412, 1112–1115. [Google Scholar] [CrossRef]
- Bartholomaus, I.; Kawakami, N.; Odoardi, F.; Schlager, C.; Miljkovic, D.; Ellwart, J.W.; Klinkert, W.E.; Flugel-Koch, C.; Issekutz, T.B.; Wekerle, H.; et al. Effector T cell interactions with meningeal vascular structures in nascent autoimmune CNS lesions. Nature 2009, 462, 94–98. [Google Scholar] [CrossRef]
- Shichita, T.; Sugiyama, Y.; Ooboshi, H.; Sugimori, H.; Nakagawa, R.; Takada, I.; Iwaki, T.; Okada, Y.; Iida, M.; Cua, D.J.; et al. Pivotal role of cerebral interleukin-17-producing gammadeltaT cells in the delayed phase of ischemic brain injury. Nat. Med. 2009, 15, 946–950. [Google Scholar] [CrossRef]
- Podolec, J.; Trabka-Zawicki, A.; Badacz, R.; Siedlinski, M.; Tomala, M.; Bartus, K.; Legutko, J.; Przewlocki, T.; Zmudka, K.; Kablak-Ziembicka, A. Chemokine RANTES and IL-1beta in mild therapeutic hypothermia-treated patients after out-of-hospital sudden cardiac arrest. Postepy Kardiol. Interwencyjnej 2019, 15, 98–106. [Google Scholar] [CrossRef]
- Zimmermann, H.W.; Tacke, F. Modification of chemokine pathways and immune cell infiltration as a novel therapeutic approach in liver inflammation and fibrosis. Inflamm. Allergy Drug Targets 2011, 10, 509–536. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, Q.; Yao, J.; Zhou, X.; Zhao, J.; Zhang, X.; Dong, J.; Liao, L. Chemokine Receptor 5, a Double-Edged Sword in Metabolic Syndrome and Cardiovascular Disease. Front. Pharmacol. 2020, 11, 146. [Google Scholar] [CrossRef]
- Kohlmeier, J.E.; Reiley, W.W.; Perona-Wright, G.; Freeman, M.L.; Yager, E.J.; Connor, L.M.; Brincks, E.L.; Cookenham, T.; Roberts, A.D.; Burkum, C.E.; et al. Inflammatory chemokine receptors regulate CD8(+) T cell contraction and memory generation following infection. J. Exp. Med. 2011, 208, 1621–1634. [Google Scholar] [CrossRef] [Green Version]
- Sorce, S.; Bonnefont, J.; Julien, S.; Marq-Lin, N.; Rodriguez, I.; Dubois-Dauphin, M.; Krause, K.H. Increased brain damage after ischaemic stroke in mice lacking the chemokine receptor CCR5. Br. J. Pharmacol. 2010, 160, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Ishida, Y.; Kimura, A.; Kuninaka, Y.; Inui, M.; Matsushima, K.; Mukaida, N.; Kondo, T. Pivotal role of the CCL5/CCR5 interaction for recruitment of endothelial progenitor cells in mouse wound healing. J. Clin. Investig. 2012, 122, 711–721. [Google Scholar] [CrossRef] [PubMed]
- Suffee, N.; Hlawaty, H.; Meddahi-Pelle, A.; Maillard, L.; Louedec, L.; Haddad, O.; Martin, L.; Laguillier, C.; Richard, B.; Oudar, O.; et al. RANTES/CCL5-induced pro-angiogenic effects depend on CCR1, CCR5 and glycosaminoglycans. Angiogenesis 2012, 15, 727–744. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Dong, J.; Lobe, C.G.; Gong, P.; Liu, J.; Liao, L. CCR5 facilitates endothelial progenitor cell recruitment and promotes the stabilization of atherosclerotic plaques in ApoE-/- mice. Stem Cell Res. Ther. 2015, 6, 36. [Google Scholar] [CrossRef]
- Reape, T.J.; Groot, P.H. Chemokines and atherosclerosis. Atherosclerosis 1999, 147, 213–225. [Google Scholar] [CrossRef]
- Baird, A.E.; Donnan, G.A.; Austin, M.C.; Fitt, G.J.; Davis, S.M.; McKay, W.J. Reperfusion after thrombolytic therapy in ischemic stroke measured by single-photon emission computed tomography. Stroke 1994, 25, 79–85. [Google Scholar] [CrossRef]
- Jorgensen, H.S.; Sperling, B.; Nakayama, H.; Raaschou, H.O.; Olsen, T.S. Spontaneous reperfusion of cerebral infarcts in patients with acute stroke. Incidence, time course, and clinical outcome in the Copenhagen Stroke Study. Arch. Neurol. 1994, 51, 865–873. [Google Scholar] [CrossRef]
- Emsley, H.C.; Tyrrell, P.J. Inflammation and infection in clinical stroke. J. Cereb. Blood Flow Metab. 2002, 22, 1399–1419. [Google Scholar] [CrossRef]
- Ritter, L.S.; Orozco, J.A.; Coull, B.M.; McDonagh, P.F.; Rosenblum, W.I. Leukocyte accumulation and hemodynamic changes in the cerebral microcirculation during early reperfusion after stroke. Stroke 2000, 31, 1153–1161. [Google Scholar] [CrossRef]
- Fahey, T.J., 3rd; Tracey, K.J.; Tekamp-Olson, P.; Cousens, L.S.; Jones, W.G.; Shires, G.T.; Cerami, A.; Sherry, B. Macrophage inflammatory protein 1 modulates macrophage function. J. Immunol. 1992, 148, 2764–2769. [Google Scholar]
- Eming, S.A.; Martin, P.; Tomic-Canic, M. Wound repair and regeneration: Mechanisms, signaling, and translation. Sci. Transl. Med. 2014, 6, 265sr266. [Google Scholar] [CrossRef] [Green Version]
- Chamorro, A.; Dirnagl, U.; Urra, X.; Planas, A.M. Neuroprotection in acute stroke: Targeting excitotoxicity, oxidative and nitrosative stress, and inflammation. Lancet Neurol. 2016, 15, 869–881. [Google Scholar] [CrossRef]
- Terao, S.; Yilmaz, G.; Stokes, K.Y.; Russell, J.; Ishikawa, M.; Kawase, T.; Granger, D.N. Blood cell-derived RANTES mediates cerebral microvascular dysfunction, inflammation, and tissue injury after focal ischemia-reperfusion. Stroke 2008, 39, 2560–2570. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Xiong, X.; Zhang, Y.; Yan, D.; Jian, Z.; Xu, B.; Zhao, H. MKEY, a Peptide Inhibitor of CXCL4-CCL5 Heterodimer Formation, Protects Against Stroke in Mice. J. Am. Heart Assoc. 2016, 5, e003615. [Google Scholar] [CrossRef] [PubMed]
- Ping, S.; Qiu, X.; Kyle, M.; Zhao, L.R. Brain-derived CCR5 Contributes to Neuroprotection and Brain Repair after Experimental Stroke. Aging Dis. 2021, 12, 72–92. [Google Scholar] [CrossRef]
- Joy, M.T.; Ben Assayag, E.; Shabashov-Stone, D.; Liraz-Zaltsman, S.; Mazzitelli, J.; Arenas, M.; Abduljawad, N.; Kliper, E.; Korczyn, A.D.; Thareja, N.S.; et al. CCR5 Is a Therapeutic Target for Recovery after Stroke and Traumatic Brain Injury. Cell 2019, 176, 1143–1157 e1113. [Google Scholar]
- Victoria, E.C.G.; de Brito Toscano, E.C.; de Sousa Cardoso, A.C.; da Silva, D.G.; de Miranda, A.S.; da Silva Barcelos, L.; Sugimoto, M.A.; Sousa, L.P.; de Assis Lima, I.V.; de Oliveira, A.C.P.; et al. Knockdown of C-C Chemokine Receptor 5 (CCR5) is Protective Against Cerebral Ischemia and Reperfusion Injury. Curr. Neurovasc. Res. 2017, 14, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Jones, D.; Adams, R.J.; Brown, T.M.; Carnethon, M.; Dai, S.; De Simone, G.; Ferguson, T.B.; Ford, E.; Furie, K.; Gillespie, C.; et al. Executive summary: Heart disease and stroke statistics--2010 update: A report from the American Heart Association. Circulation 2010, 121, 948–954. [Google Scholar] [CrossRef]
- Manwani, B.; McCullough, L.D. Sexual dimorphism in ischemic stroke: Lessons from the laboratory. Womens Health 2011, 7, 319–339. [Google Scholar] [CrossRef]
- Wagner, I.; Volbers, B.; Kloska, S.; Doerfler, A.; Schwab, S.; Staykov, D. Sex differences in perihemorrhagic edema evolution after spontaneous intracerebral hemorrhage. Eur. J. Neurol. 2012, 19, 1477–1481. [Google Scholar] [CrossRef]
- Hsieh, J.T.; Ang, B.T.; Ng, Y.P.; Allen, J.C.; King, N.K. Comparison of Gender Differences in Intracerebral Hemorrhage in a Multi-Ethnic Asian Population. PLoS ONE 2016, 11, e0152945. [Google Scholar] [CrossRef]
- Umeano, O.; Phillips-Bute, B.; Hailey, C.E.; Sun, W.; Gray, M.C.; Roulhac-Wilson, B.; McDonagh, D.L.; Kranz, P.G.; Laskowitz, D.T.; James, M.L. Gender and age interact to affect early outcome after intracerebral hemorrhage. PLoS ONE 2013, 8, e81664. [Google Scholar] [CrossRef]
- Gokhale, S.; Caplan, L.R.; James, M.L. Sex differences in incidence, pathophysiology, and outcome of primary intracerebral hemorrhage. Stroke 2015, 46, 886–892. [Google Scholar] [CrossRef]
- Appelros, P.; Stegmayr, B.; Terent, A. Sex differences in stroke epidemiology: A systematic review. Stroke 2009, 40, 1082–1090. [Google Scholar] [CrossRef]
- Bustamante, A.; Penalba, A.; Orset, C.; Azurmendi, L.; Llombart, V.; Simats, A.; Pecharroman, E.; Ventura, O.; Ribo, M.; Vivien, D.; et al. Blood Biomarkers to Differentiate Ischemic and Hemorrhagic Strokes Might Allow Prehospital Thrombolysis. Neurology 2021, 96, e1928–e1939. [Google Scholar] [CrossRef]
- Julian-Villaverde, F.J.; Ochoa-Callejero, L.; Siles, E.; Martinez-Lara, E.; Martinez, A. Adrenomedullin Is a Diagnostic and Prognostic Biomarker for Acute Intracerebral Hemorrhage. Curr. Issues Mol. Biol. 2021, 43, 324–334. [Google Scholar] [CrossRef]
- Takeshita, Y.; Ransohoff, R.M. Inflammatory cell trafficking across the blood-brain barrier: Chemokine regulation and in vitro models. Immunol. Rev. 2012, 248, 228–239. [Google Scholar] [CrossRef]
- Sablot, D.; Belahsen, F.; Vuillier, F.; Cassarini, J.F.; Decavel, P.; Tatu, L.; Moulin, T.; Medeiros de Bustos, E. Predicting acute ischaemic stroke outcome using clinical and temporal thresholds. ISRN Neurol. 2011, 2011, 354642. [Google Scholar] [CrossRef]
- Banks, J.L.; Marotta, C.A. Outcomes validity and reliability of the modified Rankin scale: Implications for stroke clinical trials: A literature review and synthesis. Stroke 2007, 38, 1091–1096. [Google Scholar] [CrossRef]
- Herder, C.; Peeters, W.; Illig, T.; Baumert, J.; de Kleijn, D.P.; Moll, F.L.; Poschen, U.; Klopp, N.; Muller-Nurasyid, M.; Roden, M.; et al. RANTES/CCL5 and risk for coronary events: Results from the MONICA/KORA Augsburg case-cohort, Athero-Express and CARDIoGRAM studies. PLoS ONE 2011, 6, e25734. [Google Scholar] [CrossRef]
- Virani, S.S.; Nambi, V.; Hoogeveen, R.; Wasserman, B.A.; Coresh, J.; Gonzalez, F., 2nd; Chambless, L.E.; Mosley, T.H.; Boerwinkle, E.; Ballantyne, C.M. Relationship between circulating levels of RANTES (regulated on activation, normal T-cell expressed, and secreted) and carotid plaque characteristics: The Atherosclerosis Risk in Communities (ARIC) Carotid MRI Study. Eur. Heart J. 2011, 32, 459–468. [Google Scholar] [CrossRef] [PubMed]

| Age (Years), Median (Q1–Q3) | 75 (63.5–79) | |
| Sex (M) | 20 (55.5%) | |
| Risk factors | ||
| Arterial hypertension | 25 (69.4%) | |
| Diabetes mellitus | 12 (33.3%) | |
| Dyslipidemia | 20 (55.5%) | |
| Atrial fibrillation | 6 (16.6%) | |
| Previous stroke | 7 (19.4%) | |
| Previous treatment | ||
| Antihypertensives | 22 (61.1%) | |
| Statins | 14 (38.8%) | |
| Antiaggregants | 14 (38.8%) | |
| Anticoagulants | 9 (25.0%) | |
| TOAST | ||
| Atherothrombotic | 11 (30.5%) | |
| Cardioembolic | 16 (44.4%) | |
| Lacunar | 2 (5.5%) | |
| Cryptogenic | 5 (13.8%) | |
| Undetermined etiology | 2 (5.5%) | |
| mRankin | ||
| Basal | 0–1–2 | 32 (88.8%) |
| 3–4 | 4 (11.1%) | |
| 5–6 | 0 (0.0%) | |
| 3 months | 0–1–2 | 21 (58.3%) |
| 3–4 | 10 (27.7%) | |
| 5–6 | 5 (13.8%) | |
| NIHSS, median (Q1–Q3) | ||
| Basal | 6 (2–13.2) | |
| Hospital discharge | 1 (0–8) | |
| 3 months | 0 (0–5.2) | |
| Infarct volume at d0 (cm3), median (Q1–Q3) | 4.9 (1.3–22.6) | |
| Infarct volume at d7 (cm3), median (Q1–Q3) | 3.2 (1.4–20.8) | |
| CCL5 at d0 (ng/mL), median (Q1–Q3) | 42.3 (24.3–50.6) | |
| CCL5 at d1 (ng/mL), median (Q1–Q3) | 43.1 (25.6–56.4) | |
| CCL5 at HD (ng/mL), median (Q1–Q3) | 60.4 (35.4–71.9) | |
| Age (Years), Median (Q1–Q3) | 81 (72.7–87) | |
| Sex (M) | 38 (59.4%) | |
| Risk factors | ||
| Arterial hypertension | 49 (76.6%) | |
| Diabetes mellitus | 15 (23.4%) | |
| Dyslipidemia | 20 (31.2%) | |
| Atrial fibrillation | 18 (28.1%) | |
| Previous treatment | ||
| Antihypertensives | 45 (70.3%) | |
| Statins | 21 (32.8%) | |
| Antiaggregants | 19 (29.7%) | |
| Anticoagulants | 20 (31.5%) | |
| TOAST | ||
| Supratentorial | 41 (64.1%) | |
| Infratentorial | 6 (9.4%) | |
| Lobar | 14 (21.9%) | |
| Mixed | 3 (4.7%) | |
| mRankin | ||
| Basal | 0–1–2 | 54 (84.4%) |
| 3–4 | 10 (15.6%) | |
| 5–6 | 0 (0.0%) | |
| 3 months | 0–1–2 | 28 (43.7%) |
| 3–4 | 12 (18.7%) | |
| 5–6 | 24 (37.5%) | |
| NIHSS, median (Q1–Q3) | ||
| Basal | 7 (2–16) | |
| Hospital discharge | 2.5 (1–6.2) | |
| 3 months | 1.5 (0–3) | |
| Hematoma at d0 (cm3), median (Q1–Q3) | 4.5 (1–13.9) | |
| Hematoma at d1 (cm3), median (Q1–Q3) | 4.1 (1–10.9) | |
| CCL5 at d0 (ng/mL), median (Q1–Q3) | 29.7 (18.2–45.6) | |
| CCL5 at d1 (ng/mL), median (Q1–Q3) | 28.9 (22.5–40.5) | |
| CCL5 at HD (ng/mL), median (Q1–Q3) | 33.9 (22.0–51.1) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Julián-Villaverde, F.J.; Serrano-Ponz, M.; Ramalle-Gómara, E.; Martínez, A.; Ochoa-Callejero, L. CCL5 Levels Predict Stroke Volume Growth in Acute Ischemic Stroke and Significantly Diminish in Hemorrhagic Stroke Patients. Int. J. Mol. Sci. 2022, 23, 9967. https://doi.org/10.3390/ijms23179967
Julián-Villaverde FJ, Serrano-Ponz M, Ramalle-Gómara E, Martínez A, Ochoa-Callejero L. CCL5 Levels Predict Stroke Volume Growth in Acute Ischemic Stroke and Significantly Diminish in Hemorrhagic Stroke Patients. International Journal of Molecular Sciences. 2022; 23(17):9967. https://doi.org/10.3390/ijms23179967
Chicago/Turabian StyleJulián-Villaverde, Francisco José, Marta Serrano-Ponz, Enrique Ramalle-Gómara, Alfredo Martínez, and Laura Ochoa-Callejero. 2022. "CCL5 Levels Predict Stroke Volume Growth in Acute Ischemic Stroke and Significantly Diminish in Hemorrhagic Stroke Patients" International Journal of Molecular Sciences 23, no. 17: 9967. https://doi.org/10.3390/ijms23179967
APA StyleJulián-Villaverde, F. J., Serrano-Ponz, M., Ramalle-Gómara, E., Martínez, A., & Ochoa-Callejero, L. (2022). CCL5 Levels Predict Stroke Volume Growth in Acute Ischemic Stroke and Significantly Diminish in Hemorrhagic Stroke Patients. International Journal of Molecular Sciences, 23(17), 9967. https://doi.org/10.3390/ijms23179967







