Abstract
In mammals, female fertility is determined by the outcome of follicular development (ovulation or atresia). The TGF-β/SMAD signaling pathway is an important regulator of this outcome. However, the molecular mechanism by which the TGF-β/SMAD signaling pathway regulates porcine follicular atresia has not been fully elucidated. Microrchidia family CW-type zinc finger 2 (MORC2) is anovel epigenetic regulatory protein widely expressed in plants, nematodes, and mammals. Our previous studies showed that MORC2 is a potential downstream target gene of the TGF-β/SMAD signaling pathway. However, the role of MORC2 in porcine follicular atresia is unknown. To investigate this, qRT-PCR, western blotting, and TdT-mediated dUTP nick-end labeling were performed. Additionally, the luciferase activity assay was conductedto confirm that the TGF-β/SMAD signaling pathway regulates MORC2. Our results demonstrate that MORC2 is animportant anti-apoptotic molecule that prevents porcine follicular atresia via a pathway involving mitochondrial apoptosis, not DNA repair. Notably, this studyrevealsthat the TGF-β/SMAD signaling pathway inhibits porcine granulosa cell apoptosis by up-regulating MORC2. The transcription factor SMAD4 regulated the expression of MORC2 by binding to its promoter. Our results will help to reveal the mechanism underlying porcine follicular atresia and improve the reproductive efficiency of sows.
1. Introduction
The reproductive traits of sows seriously affect the economic benefits of the pig industry. Ovarian follicular development and ovulation are the main factors that affect female reproductive performance. Fewer than 1% of follicles undergo ovulation, and the remainder undergoes atresia and degradation, leading to a huge waste of reproductive resources. The reproductive potential of female livestock could be enhanced if follicular atresia could be prevented. The causes of follicular atresia are complex. Most research has focused on cell apoptosis, autophagy, oxidative stress, hypoxia, non-coding RNA, and other aspects [1,2,3,4,5]. However, the essential factors that cause follicular atresia remain unclear. Follicular atresia limits the fertility of sows, and granulosa cell apoptosis is a hallmark of this process. The TGF-β/SMAD signaling pathway is closely related to female fertility. Core factors involved in this pathway, such as TGF-β1, TGFBR2, SMAD2/3, SMAD1/5, and SMAD4, are key candidates to affect female reproduction [6,7,8,9,10,11]. Moreover, the TGF-β/SMAD signaling pathway affects follicular atresia in pigs [11,12,13]. Activation of the TGF-β/SMAD signaling pathway inhibits apoptosis of follicular granulosa cells, while blockade of this pathway has the opposite effect [14,15]. However, the molecular mechanism by which the TGF-β/SMAD signaling pathway regulates granulosa cell apoptosis has not been fully elucidated.
Microrchidia (MORC) family proteins, which were first reported and characterized in germ cells, are members of the ATPase family and are critical for life. Research into the functions of MORC proteins is just beginning. MORCs, including MORC1, MORC2, MORC3, and MORC4, are linked to immunological disorders, neurogenic disorders, and cancer [16,17,18,19]. In addition, recent research showed that MORC3 has a primary anti-viral function [17,19]. Generally speaking, MORCs contain a conserved GHKL-ATPase domain at the N-terminus, a conserved CW-type zinc finger domain in the middle, and several coiled-coil domains. At present, people pay more attention to MORC2 and MORC3 proteins. MORC2, also known as ZCWCC1, is widely expressed, especially in the ovary, testis, brain, uterus, and stomach [20]. As an epigenetic regulatory protein, the function of MORC2 has not been fully elucidated but is usually associated with gene silencing, chromatin remodeling, and DNA repair, and it affects cell survival and participates in the regulation of cell apoptosis [21,22,23,24,25]. In addition, compared with other MORC proteins, MORC2 has unique structural characteristics. The nuclear localization signal and proline-rich domain of human MORC2 play a role in transcriptional inhibition in gastric and colon cancer cells. However, research on MORC2 is in its infancy. Mutation of MORC2 is associated with Charcot-Marie-tooth disease and neuro-developmental disorders [16]. In addition, MORC2 is highly expressed in many cancers [20]. The function of MORC2 in normal somatic and germ cells remains poorly understood. In 2019, Liu et al. found that MORC2 inhibits differentiation and promotes the proliferation of C2C12 mouse skeletal muscle cells [26]. Moreover, MORC2 is highly expressed in mouse ovaries and testes, and the inactivation of its homologous gene (MORC2b) blocks ovarian development and prevents the formation of mature follicles [27]. This suggests that MORC2 plays an important role in the regulation of mammalian ovarian development and reproduction. Our previous studies showed that TGF-β1 inhibits apoptosis of porcine follicular granulosa cells [11]. Meantime, transcriptomic studies identified 1025 differentially expressed genes in porcine follicular granulosa cells upon SMAD4 interference, of which 530 were up-regulated and 495 were down-regulated [28]. Among which, MORC2 was significantly down-regulated, indicating there is an association between the TGF-β/SMAD signaling pathway and MORC2, which needs to be studied further. While there is no report about the function of MORC2 in large domestic animals such as pigs.
This study aimed to explore the effect of MORC2 on porcine granulosa cell apoptosis and the role in the process of porcine follicular atresia regulated by the TGF-β/SMAD4 signaling pathway. In this study, we discovered that MORC2 is an anti-apoptotic factor inporcine granulosa cells. Functional assays demonstrated that MORC2 inhibits cell apoptosis via the mitochondrial apoptosis pathway, not the DNA repair pathway. Moreover, we revealed that the TGF-β/SMAD signaling pathway up-regulates MORC2. The current research intended to explore the effect of the MORC2 gene on porcine granulosa cell apoptosis and elaborated on the physiological role of the TGF-β/SMAD4 signaling pathway in porcine follicular atresia.
2. Results
2.1. MORC2 Inhibits Apoptosis of Porcine Follicular Granulosa Cells
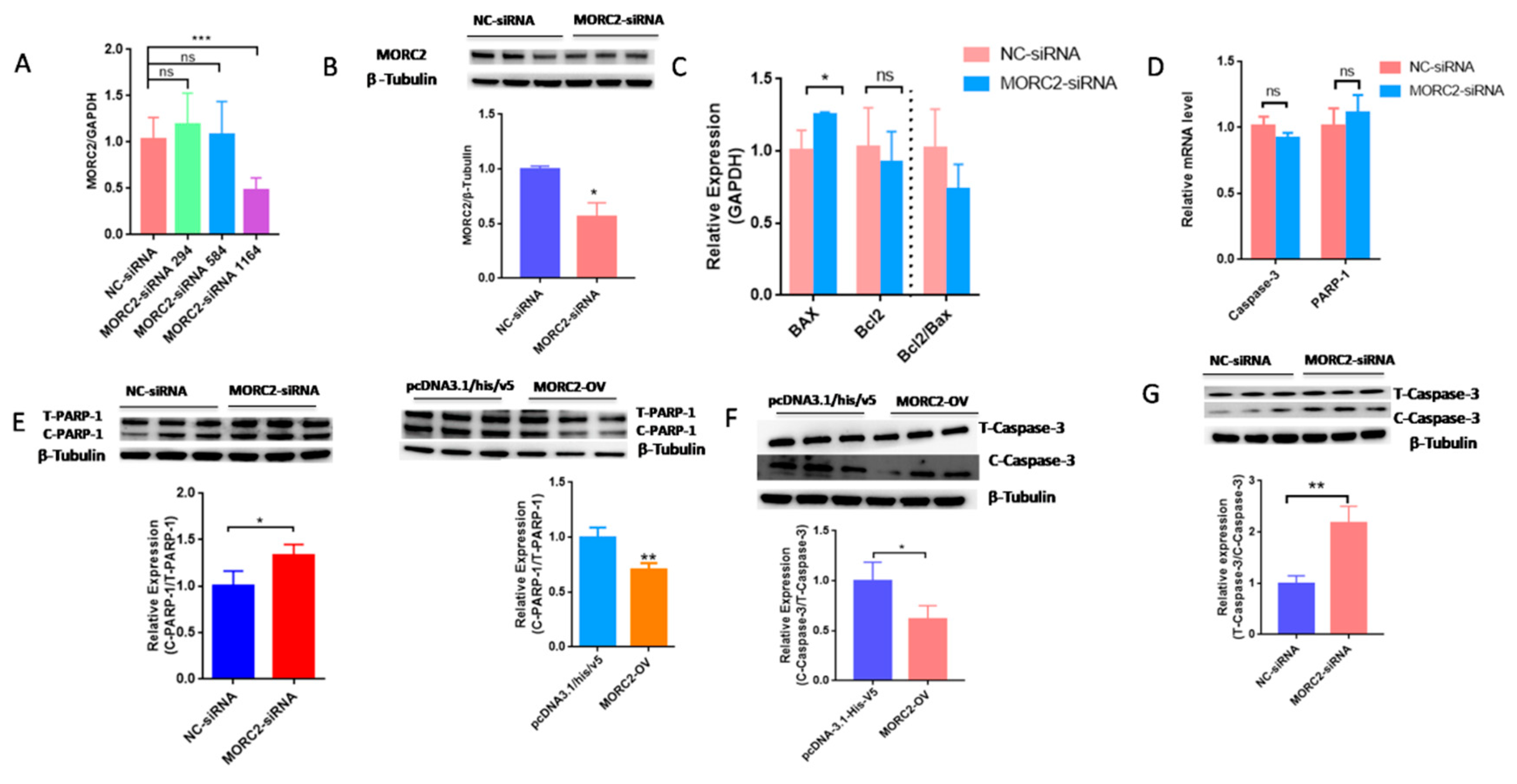
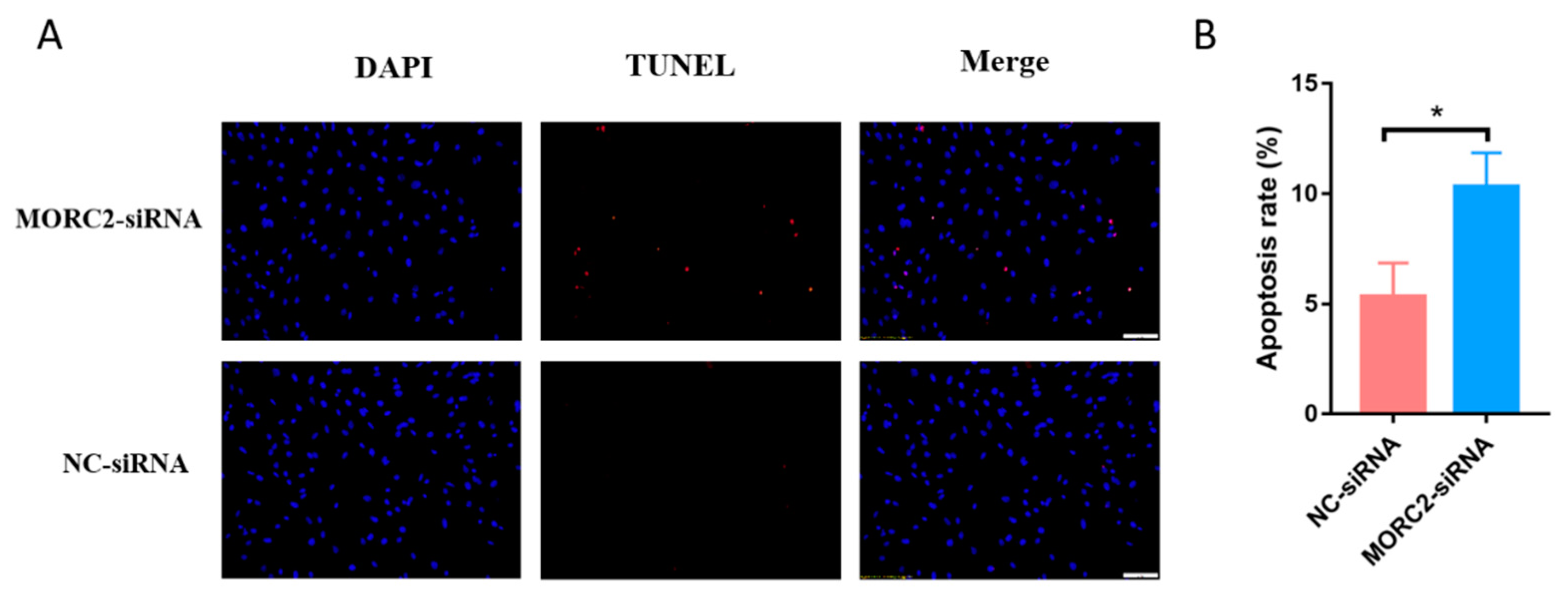
MORC2 plays an important role in the regulation of cell fate. We aimed to knock down or over-express MORC2in order to elucidate its function in the apoptosis of porcine follicular granulosa cells. An effective MORC2-targeting siRNA (MORC2-siRNA) was screened by qRT-PCR (Figure 1A) and western blotting (WB, Figure 1B). That indicated MORC2-siRNA-1164 wasthe effective siRNA. qRT-PCR showed that MORC2 knockdown significantly reduced the Bcl2/Bax ratio (Figure 1C), but did not affect the transcription levels of Caspase-3 and PARP-1 (Figure 1D). WB showed that cleavage of Caspase-3 and PARP-1 was promoted by MORC2 knockdown but inhibited by MORC2 over-expression (Figure 1E,F). In addition, TdT-mediated dUTP nick-end labeling (TUNEL) staining showed that MORC2 knockdown significantly promoted apoptosis of porcine granulosa cells (Figure 2A,B). These results suggest that MORC2 is an anti-apoptotic protein in granulosa cells.
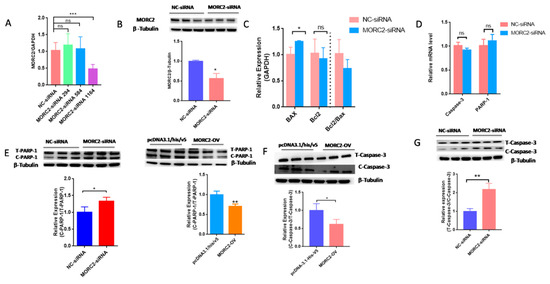
Figure 1.
Knockdown of MORC2 induces apoptosis-related gene expression in porcine granulosa cells. (A) qRT-PCR analyse of MORC2 mRNA relative expression in porcine granulosa cells transfected with different MORC2-targeting siRNAs. (B) The relative protein expression of MORC2 in porcine granulosa cell transfected with MORC2-siRNA. (C) qRT-PCR analysis of Bax, Bcl2, and Bax/Bcl2 upon depletion of MORC2 by siRNA. (D,E) qRT-PCR analysis of Caspase-3 and PARP-1 in porcine granulosa cells transfected with MORC2-siRNA. GAPDH was used as a loading control. (F,G) Representative WB and quantification of the cleaved Caspase-3 and cleaved PARP-1 levels in porcine granulosa cells depleted of MORC2.NC-siRNA indicates small interfering RNA of the negative control. MORC2-siRNA means small interfering RNA of MORC2 gene. MORC2-siRNA 294, 584, and 1164 indicate different small interfering RNA target different sites in the MORC2 gene. MORC2-OV indicates the MORC2 overexpression vector. T-PARP-1: total protein of PARP-1; C-PARP-1 cleaved protein of PARP-1. T-Caspase-3: total protein of Caspase-3; C-Caspase-3: cleaved protein of Caspase-3. Results are shown as mean ± SEM (n = 3). ns means no significance (p > 0.05); * p < 0.05, ** p < 0.01, *** p < 0.001.
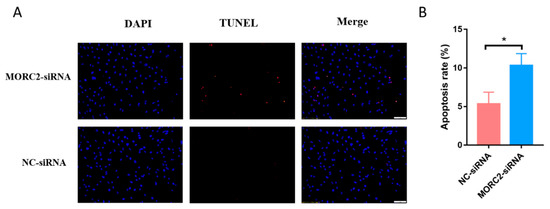
Figure 2.
MORC2 attenuates porcine granulosa cell apoptosis. (A) TUNEL assay to detect apoptotic porcine granulosa cells transfected with MORC2 siRNA. The Scale bar = 50 μm. (B) Quantification of apoptotic porcine granulosa cells upon treatment with MORC2-siRNA. DAPI was used to label nuclei and TUNEL was used to label apoptotic cells. * p < 0.05.
2.2. MORC2 Inhibits Apoptosis of Porcine Granulosa Cells Independently of the DNA Repair Pathway
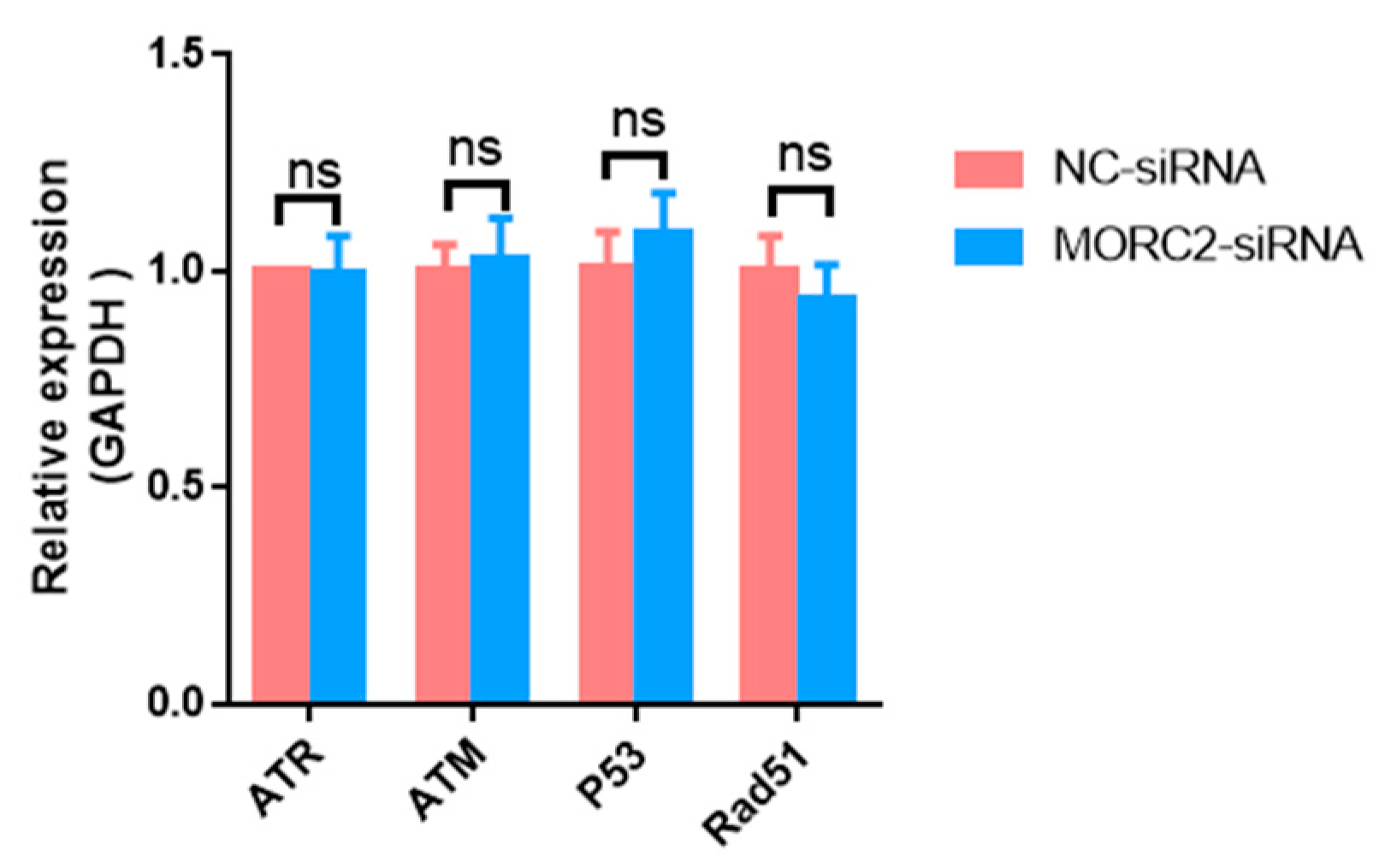
The DNA damage response is essential for the proper functions of all cells and organisms. MORC2, which contains an ATPase module, has been mechanistically linked to the DNA damage response. To explore the function of MORC2, we examined whether it affects the DNA damage response. Ataxia-telangiectasia mutated (ATM) protein kinase, Ataxia-telangiectasia and Rad3-related (ATR), p53, and Rad51 are the main factors involved in the DNA damage response. qRT-PCR showed that knockdown of MORC2 did not significantly alter the expression of these factors (Figure 3). These results indicate that MORC2 inhibits granulosa cell apoptosis independently of the DNA repair pathway.
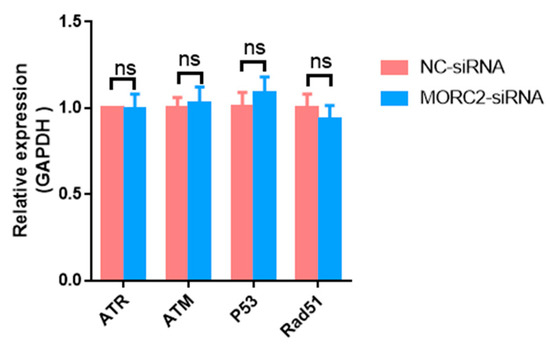
Figure 3.
Expression of genes related to the DNA repair pathway. qRT-PCR analysis of mRNA expression of ATR, ATM, p53, and Rad51 upon knockdown of MORC2. GAPDH was used as a loading control. ns means no significance (p > 0.05).
2.3. MORC2 Is a Downstream Target of the TGF-β/SMAD Signaling Pathway
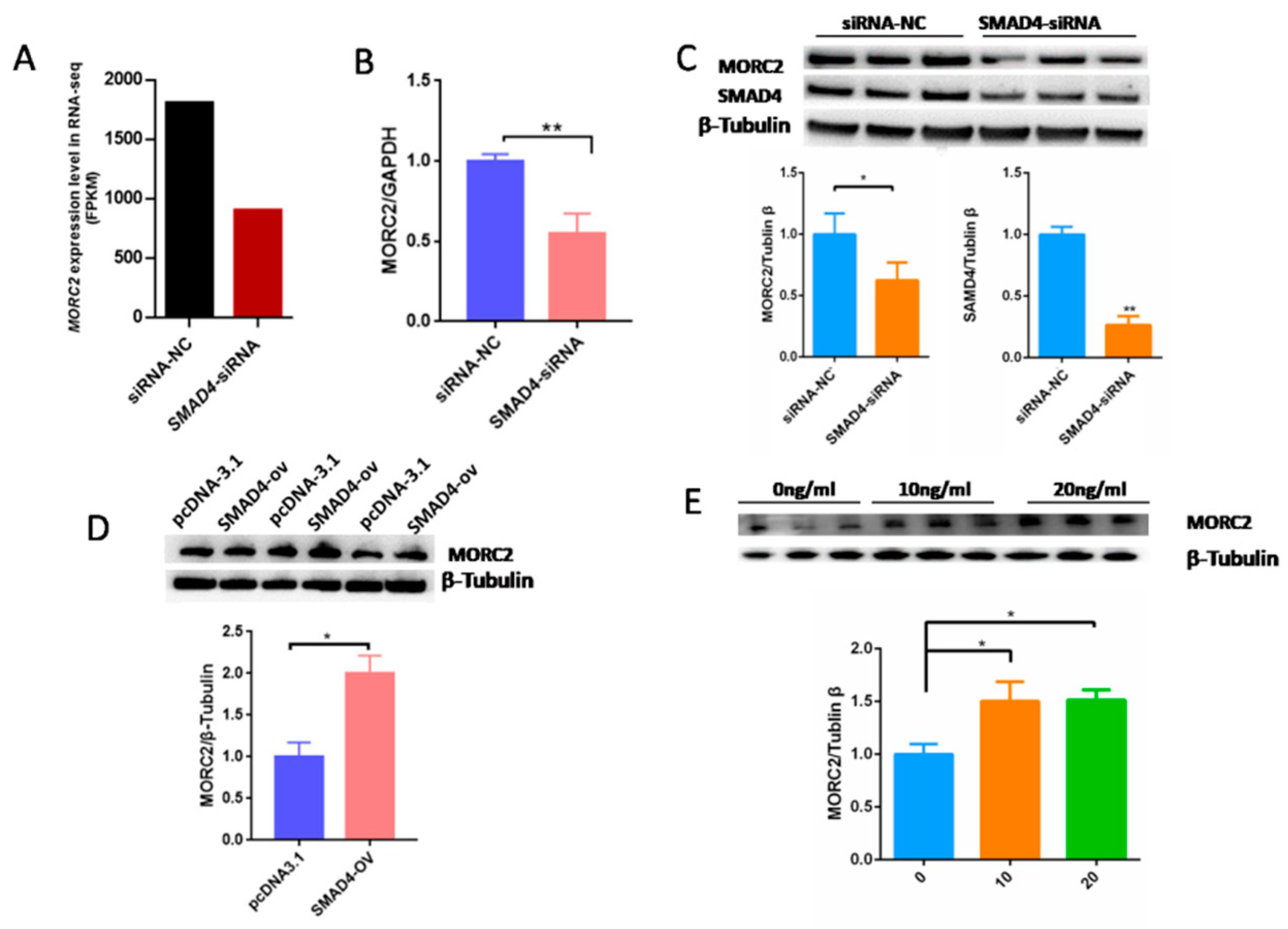
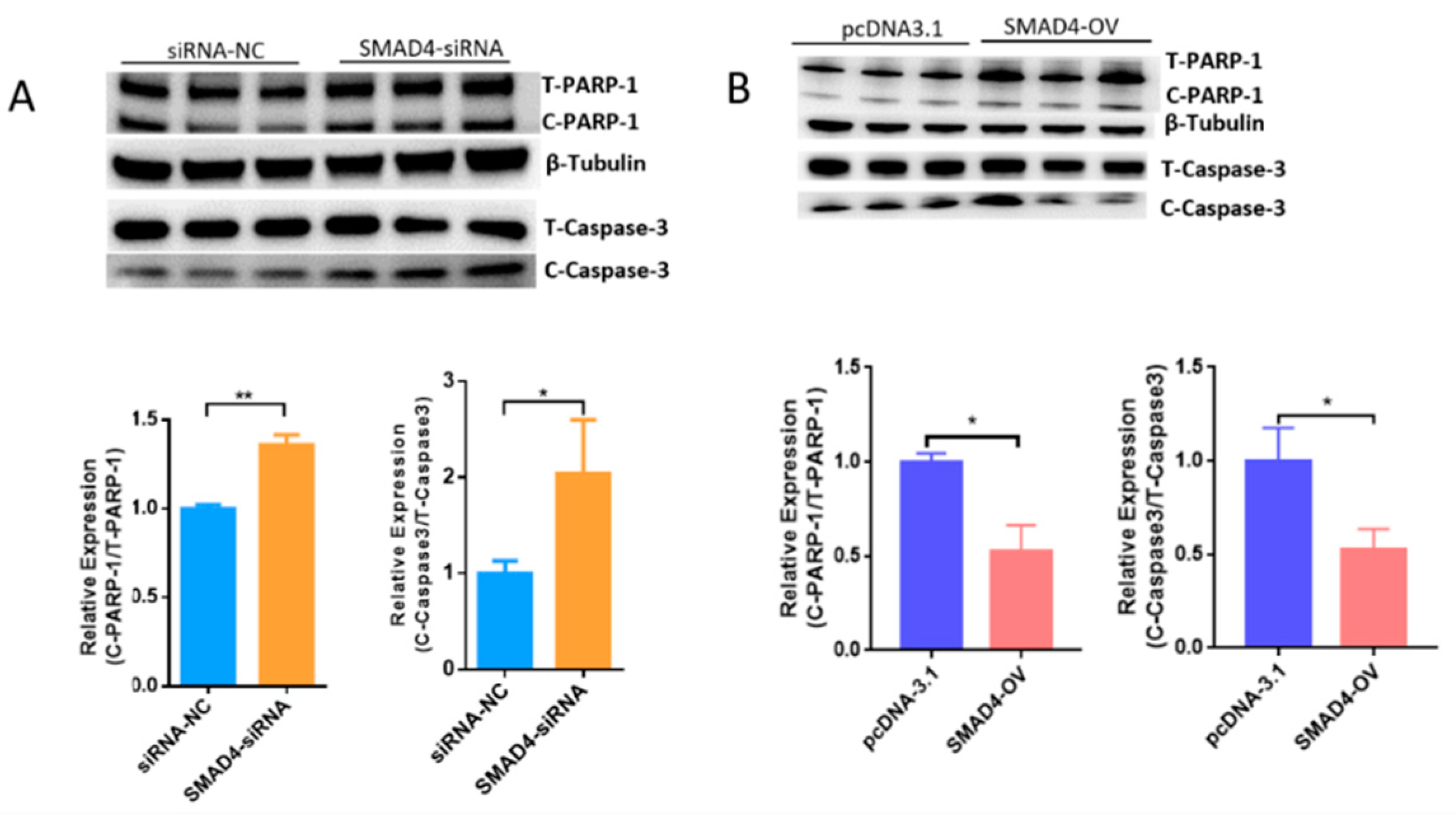
Our previous results demonstrated that the TGF-β/SMAD signaling pathway is critical for the determination of ovarian fate and SMAD4 is a core component of this pathway. Based on RNA-seq data (GSE65696) obtained after the knockdown of SMAD4 in porcine granulosa cells [28], we found that SMAD4 knockdown significantly down-regulated MORC2 (Figure 4A). This was confirmed by qRT-PCR (Figure 4B) and WB (Figure 4C). On the contrary, SMAD4 over-expression dramatically increased the MORC2 protein level (Figure 4D). Similarly, TGF-β1 treatment increased MORC2 protein expression (Figure 4E). These results suggest that the TGF-β/SMAD pathway induces MORC2 expression in porcine granulosa cells. So, does TGF-β/SMAD signaling pathway regulates porcine granulosa cell apoptosis through MORC2? The expression levels of Caspase-3 and PARP-1 were detected after knockdown and over-expression of SMAD4. The results showed that the knockdown of SMAD4 promoted the cleavage of Caspase-3 and PARP-1 (Figure 5A), but over-expression of SMAD4 had the opposite effect (Figure 5B), which further supported our hypothesis.
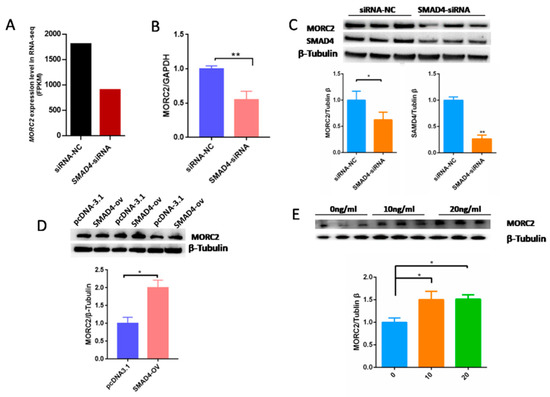
Figure 4.
The TGF-β/SMAD signaling pathway significantly up-regulates MORC2 in porcine granulosa cells. (A) RNA-seq analysis of the MORC2 mRNA expression level (fragments per kb of exon per million fragments mapped; FPKM) in porcine granulosa cells treated with negative control siRNA (NC-siRNA) or SMAD4 knockdown RNA (SMAD4-siRNA). (B) Confirmation of the RNA-seq result by qRT-PCR after treatment with SMAD4 siRNA. GAPDH was used as a loading control. (C,D) WB analysis of theMORC2 protein level in porcine granulosa cells upon knockdown or over-expression of SMAD4 (SMAD4-OV). (E) The MORC2 protein level in porcine granulosa cells treated with TGF-β1. β-Tubulin served as a loading control. Results are shown as mean ± SEM (n = 3). * p < 0.05, ** p < 0.01.
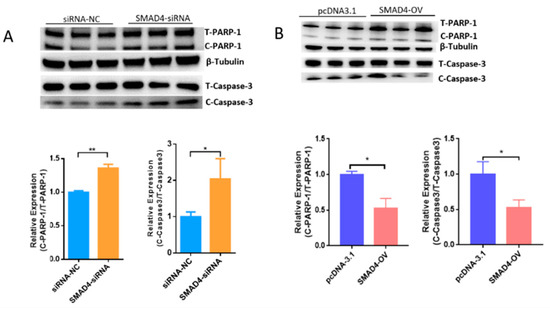
Figure 5.
WB analysis of the Caspase-3 and PARP-1 protein level in porcine granulosa cellsupon knockdown (A) or over-expression of SMAD4 (B). SMAD4-OV: SMAD4 overexpression vector. Results are shown as mean ± SEM (n = 3). * p < 0.05, ** p < 0.01.
2.4. The Transcription Factor SMAD4 Directly Binds to the MORC2Promoter
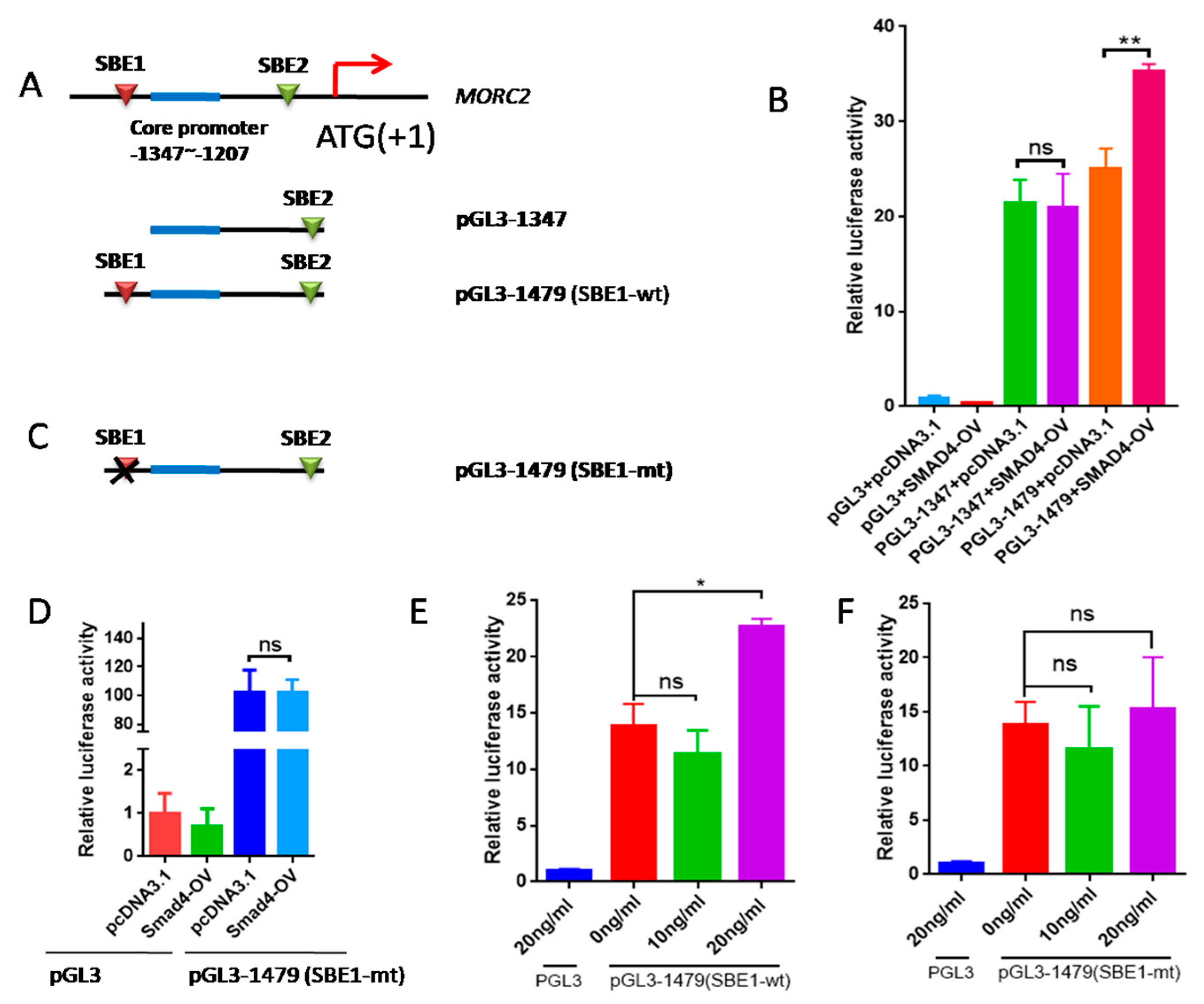
SMAD4 is an important transcription factor that regulates the expression of many genes. We previously reported that the core promoter region of the porcine MORC2 gene is located between -1207 and -1347 in the 5′ regulatory region (ATG is +1) [29]. To study the effect of the TGF-β/SMAD signaling pathway on MORC2 expression, smad binding elements (SBE) sites were predicted using PROMO and JASPAR online software. Two SBE sites in the promoter of MORC2 were identified (Figure 6A). SBE1 was contained in the pGL3-1479 vector and SBE2 was contained in the pGL3-1347 vector. SMAD4 over-expression increased relative luciferase activity of pGL3-1479, but not of pGL3-1347 (Figure 6B). This indicates that SMAD4 binds to SBE1. To verify this, the binding sites were deleted (Figure 6C). SMAD4 over-expression did not affect the luciferase activity of the MORC2 mutant promoter (Figure 6D). Relative luciferase activity of pGL3-1479 was significantly increased by treatment with 10 and 20 ng/mL TGF-β1, but not by treatment with 5 ng/mL TGF-β1 (Figure 6E), whereas relative luciferase activity of the MORC2 mutant promoter was not affected (Figure 6F). These results indicate that the transcription factor SMAD4 binds to SBE1 of the MORC2 promoter.
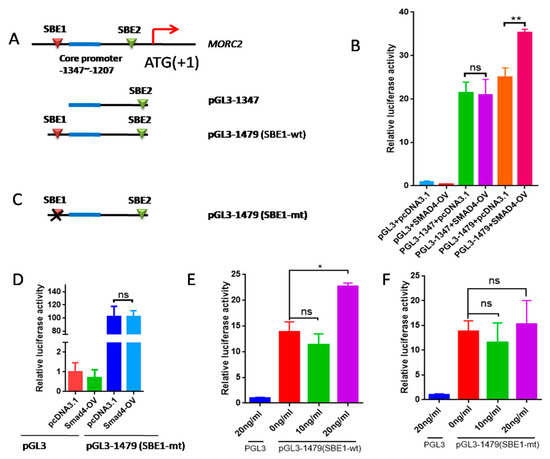
Figure 6.
The transcription factor SMAD4 induces transcription of MORC2. (A) Schematic showing insertion of the MORC2 promoter into the pGL3-basic vector. Potential SBE sites are indicated by red boxes. ATG was +1. (B) Changes of relative luciferase activity with or without SMAD4 over-expression. (C) The schematic of SBE1-mt in MORC2 promoter. mt indicates SBE site mutation. (D) Changes of relative luciferase activity of pGL3-1479 (SBE1-mt) with or without SMAD4 overexpression. (E,F) Changes in relative luciferase activity of the SEB1-wt or SBE1-mut MORC2 promoter treated with TGF-β1. Data are shown as mean ± SEM. (n = 3). * p < 0.05; ** p < 0.01; ns, no significance.
3. Discussion
In 1996, the first member of the MORC family was identified in testes and named MORC1. MORC1 is mainly expressed in embryonic stem cells and thymocytes, MORC2 and MORC3 are expressed ubiquitously, and MORC4 is mainly expressed in the placenta and pituitary gland. Decades of research have revealed that not only MORC1, but also MORC2 and MORC3 are closely related to the regulation of animal reproduction [27,30,31]. However, most studies of MORC proteins mainly concentrated on male reproduction. MORC1 is specifically expressed in the male germline and its ablation results in male sterility with meiotic arrest [32,33]. In addition, loss of MORC3 reduces the pregnancy rate and leads to subfertility in male mice but does not affect the litter size of female mice [34]. However, MORC2 is not only related to male reproduction but also to female reproduction. Both male and female Morc2b−/− mice are sterile. Ovaries of adult Morc2b−/− female mice are much smaller than those of heterozygous littermates and lack oocytes [27]. This indicates that MORC2 is important for female reproduction. MORC1 induces cell apoptosis in mouse testes [31] and MORC2 mediates cell apoptosis in neurons and oocytes [16,27]. However, it is unknown whether MORC2 affects the apoptosis of porcine granulosa cells. Here, we reported that MORC2 is an anti-apoptotic protein in porcine granulosa cells. Apoptosis of granulosa cells increased when MORC2 was knocked down. This indicates that MORC2 is an anti-apoptotic factor in porcine granulosa cells. The DNA damage response is a process by which DNA is repaired via the actions of various enzymes. ATM, ART, p53, and Rad51 are markers of DNA repair. MORC2 was reported to be a DNA damage response gene [16,22,35]. Therefore, we investigated whether MORC2 inhibits porcine granulosa cell apoptosis via the DNA repair response. MORC2 knockdown did not affect the expression of DNA repair response genes. Meanwhile, knockdown of MORC2 significantly increased expression of the pro-apoptotic gene Bax as well as the levels of cleaved Caspase-3 and PARP-1, indicating that MORC2 inhibits apoptosis via the mitochondrial apoptosis pathway. MORC2 may be a candidate marker of reproductive performance because it decreases porcine granulosa cell apoptosis.
Atresia occurs in follicles at all stages. Many studies have revealed the factors that cause follicular atresia. However, these are just the tip of the iceberg, and many unknown factors await further investigation. In porcine ovaries, the ovulation rate is an important parameter for the reproductive efficiency of sows. In vivo and in vitro experiments revealed that gonadal steroids, growth factors, and cytokines are essential for follicular development. Among various intra-ovarian paracrine and autocrine factors, members of the TGF-β superfamily are best studied in terms of their effects on follicular atresia. TGF-β1 is an important member of this superfamily and is crucial for granulosa determination of granulose cell fate and female fertility [14,36,37]. The TGF-β1 gene is significantly associated with the reproductive performance of Large White sows [37] and is involved in porcine granulosa cell apoptosis [14].The results of TGF-β1 regulating granulosa cell apoptosis are inconsistent. In porcine ovarian granulosa cells, TGF-β1 inhibits granulosa cell apoptosis [14]. However, TGF-β1 promotes granulosa cell apoptosis in polycystic ovary syndrome [38] and bovine granulosa cells [39]. Therefore, TGF-β1 not only inhibits but also promotes granulosa cell apoptosis. This indicates that the functions of TGF-β1 differ according to the physiological conditions. SMAD4 is the only common mediator of Smad (Co-SMAD), and accumulating evidence indicates that it is associated with ovarian development, especially granulosa cell apoptosis [11,40,41]. In general, SMAD4 and regulatory SMADs (R-SMADs) form transcriptional complexes and regulate gene expression by binding to promoters of genes in the nucleus. Here, we identified a binding site for SMAD4 in the MORC2 promoter; therefore, MORC2 is a novel gene regulated by the TGF-β signaling pathway.
Research on MORC2 has mainly focused on its function and structure. MORC2 mutations cause Charcot-Marie-Tooth neuropathy type 2Z disease [42], and recent research showed that MORC2 is aberrantly highly expressed in many cancers, including breast cancer [25,35], hepatocellular carcinoma cells [18], and gastric cancer cells [43]. Although the present study revealed the role of MORC2, further studies are needed. The function of MORC2 in porcine granulose cell apoptosis of large domestic animals (pigs) was preliminarily investigated in this study, and the effect of MORC2 on the reproductive performance of pigs will be further explored. MORC2 contains a highly conserved GHKL-ATPase domain, a CC domain, and a CW zinc finger domain. Thus, MORC2 may function in transcription regulation, chromatin remodeling, and DNA repair. Therefore, it is necessary to study the regulation of MORC2 expression. However, research has mainly focused on its regulation at the post-transcriptional level. For instance, GPER1 phosphorylates threonine 582 of MORC2 [23]. MORC2 767 lysine could be acetylated by the acetyltransferase NAT10 and deacetylase SIRT2 [35]. In addition, threonine 556 of MORC2 is O-GlcNAcylated by TGF-β1 [25]. However, there are few studies of the upstream regulatory signals of MORC2 expression. For glucose-induced MORC2 expression, c-Myc binds to the promoter of MORC2 and activates its transcription [44]. Although the promoter of MORC2 contains two ESR1-binding sites, E2 does not affect its mRNA expression [23]. To reveal the function of MORC2, it is necessary to study its transcriptional regulation. Although a previous study showed that TGF-β1 induces O-GlcNAcylation of MORC2 by regulating GFAT, it was unclear whether TGF-β1 directly regulates MORC2 expression. One of the highlights of this study is that it shows that MORC2 is a direct target of the TGF-β/SMAD signaling pathway.
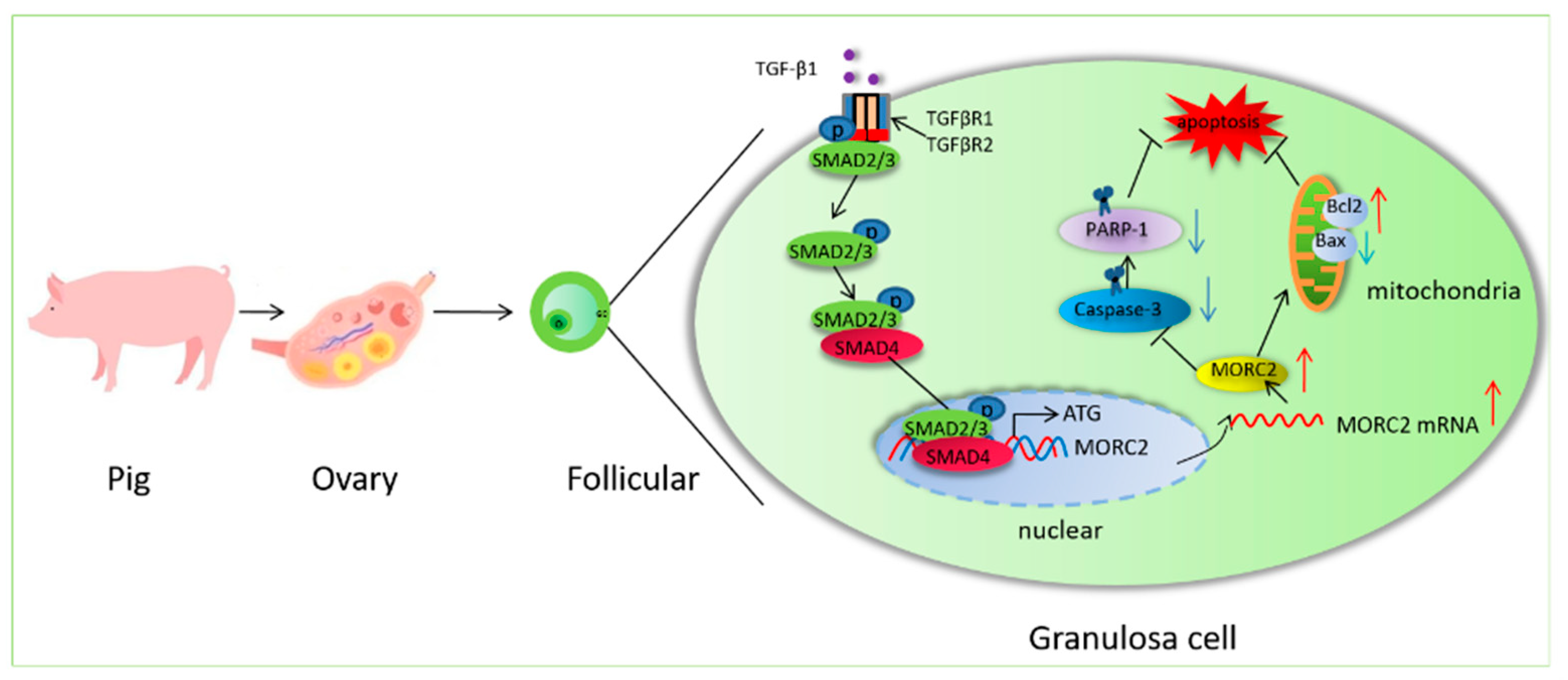
In summary, the present study provides evidence that MORC2 is an anti-apoptotic factor in porcine granulosa cells. It also shows that the transcription factor SMAD4 binds to the MORC2 promoter. The findings presented here reveal a previously unrecognized functional and mechanistic role of the TGF-β/SMAD signaling pathway in follicular atresia. Based on our results, we propose a molecular mechanism (Figure 7).
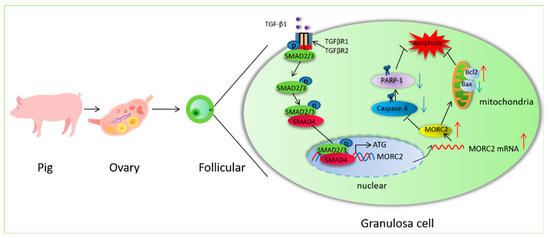
Figure 7.
Schematic showing how the TGF-β/SMAD signaling pathway regulates porcine granulosa cell apoptosis by targeting MORC2. O:Oocyte; GC: granulosa cell. Red arrows indicate provementand the blue arrows indicate inhibition.
4. Materials and Methods
4.1. Reagents
DMEM/F12 and penicillin/streptomycin (Life Technologies Co., Carlsbad, CA, USA), HighGene Transfection reagent (ABclonal, Wuhan, China), RNAiso Plus, and a PrimeScript™ RT reagent Kit with gDNA Eraser (Perfect Real Time) (Taraka, Dalian, China), a One Step TUNEL Apoptosis Assay Kit and a BCA Protein Assay Kit (Beyotime Biotechnology, Shanghai, China), a Dual-Luciferase Reporter Assay Kit (Vazyme, Nanjing, China), TGF-β1 (NovoProtein, Suzhou, China), an anti-MORC2 rabbit polyclonal antibody (Sangon Biotech, Shanghai, China), an anti-SMAD4 rabbit polyclonal antibody (Santa Cruz Biotechnology, Shanghai, China), and PVDF membrane (Merck Millipore, Darmstadt, Germany) were purchased from the indicated suppliers.Primers were synthesized by Shangya (Hangzhou, China). siRNAs were synthesized by GenePharma (Shanghai, China). NovoStart® SYBR qPCR SuperMix Plus was purchased from Novoprotein (Nanjing, China).
4.2. Isolation and Culture of Porcine Granulosa Cells
The animal protocols were approved by the Institutional Animal Care and Use Committee (IACUC) of Jiangsu University of Science and Technology (G2022SJ12, Zhenjiang, China). Animal care and handling followed the IACUC guidelines.
Fresh ovaries were collected from 6-month-old commercial sows from the local slaughterhouse. A total of 72 sows were used. Healthy follicles of 3~5 mm were selected to collect the porcine ovarian granulosa cell. The follicles were immediately immersed in saline containing penicillin/streptomycin and transported to the laboratory within 2 h. Porcine granulosa cells were collected from healthy follicles (3~5 mm diameter) using 22-gauge needles. The cells were washed twice with phosphate-buffered saline (PBS) and centrifugation at 800× g for 5 min. Then the cells were cultured in DMEM/F12 containing 10% fetal bovine serum, and 1% penicillin-streptomycin at 37 °C in an incubator containing 5% CO2.
4.3. Plasmid Construction
The MORC2 promoter was cloned into the pGL3-basic vector between HindIII and KpnI. The SEB sites were predicted using PROMO (http://alggen.lsi.upc.es/cgi-bin/promo_v3/promo/promoinit.cgi?dirDB=TF_8.3 (accessed on 15 June 2021)) and JASPAR (https://jaspar.genereg.net (accessed on 2 July 2021)). The SEB-mut vector was generated using a Mut Express® II Fast Mutagenesis Kit V2 (Vazyme, Nangjing, China) and verified by Sanger sequencing. The coding domain sequence (CDS) region of MORC2 was inserted into the pcDNA3.1-His-v5 over-expression vector. The CDS of SMAD4 was inserted into the pcDNA3.1 over-expression vector. The primers used for plasmid construction are listed in Table 1.

Table 1.
The siRNAs used in this study.
4.4. Cell Transfection
Porcine granulosa cells were seeded into 12-well plates and transfected when their density reached 70%. The siRNAs are listed in Table 1. Briefly, 50 pmol siRNA and 3 μL High Gene Transfection reagent were mixed in 100 μL serum-free DMEM/F12 and added to the cell culture. The plate was gently shaken. After 4–6 h of transfection, half the medium was replaced by a fresh complete medium. At 24 or 48 h after transfection, cells were analyzed by various methods, such as RT-PCR, WB, and luciferase assays.
4.5. Total RNA Extraction and Quantitative PCR
Total RNA was extracted from porcine granulosa cells using RNAiso Plus. In total, 1 µg total RNA was reverse-transcribed using a PrimeScript™ RT reagent Kit with gDNAEraser according to the manufacturer’s instructions. qRT-PCR was performed using the following conditions: 95 °C for 1 min, followed by 35 cycles of 95 °C for 20 s, 60 °C for 20 s, and 72 °C for 30 s. The primers are listed in Table 2. The relative expression of each gene was calculated using the 2−ΔΔCt method. GAPDH was used as a reference gene.

Table 2.
Primers used for qRT-PCR and plasmids construction.
4.6. WB Analysis
Whole cells were lysed on ice for 30 min with RIPA buffer containing protease inhibitors. The lysate was centrifuged for 10 min at 4 °C at 12,000× g. The total protein concentration was determined using a BCA Protein Assay Kit. In total, 10 µg total protein was loaded per lane, separated by 4–20% SDS-PAGE, and transferred to a PVDF membrane. The membrane was blocked with 5% non-fat milk for 2 hat room temperature, incubated with primary antibodies overnight at 4 °C, washed thrice with TBST, incubated with secondary HRP-conjugated antibodies, washed thrice with TBST, and incubated with ECL reagent. WB images were captured using a chemiluminescence imaging system (Qinxiang, Shanghai, China). β-Tubulin was used as an internal control.
4.7. Tunel
Porcine granulosa cells were washed once with phosphate-buffered saline (PBS), fixed with 4% paraformaldehyde for 30 min, washed with PBS, incubated with PBS containing 0.3% Triton X-100 at room temperature for 5 min, washed twice with PBS, incubated with TUNEL solution for 60 min at 37 °C, and washed twice with PBS. Slides were sealed with anti-fluorescence quenching DAPI solution. Cy3 was detected using an excitation wavelength of 550 nm and an emission wavelength of 570 nm (red fluorescence).
4.8. Luciferase Reporter Gene Assay
Porcine granulosa cells were collected after transfection for 24 h, lysed using 1× cell lysis buffer, and centrifuged for 10 min at 12,000× g. Analysis was performed using a Dual-luciferase Reporter Assay Kit (Vazyme, Nanjing, China). Firefly and Renilla luciferase activities were evaluated and normalized for each sample.
4.9. Statistical Analysis
GraphPad Prism 6 (GraphPad Software, San Diego, CA, USA) was used to perform statistical analysis. The two-tailed Student’s t-test was used to compare two groups. All data are presented as the mean ± SEM of at least three independent experiments. p < 0.05 was considered statistically significant (*) and p < 0.01 was considered highly statistically significant (**).
5. Conclusions
We demonstrated that MORC2 regulates porcine granulosa cell apoptosis. The TGF-β/SMAD signaling pathway inhibits porcine granulosa cell apoptosis via binding of SMAD4 to the promoter of MORC2 and activation of its expression. Our findings provide insight into the mechanism by which the TGF-β/SMAD signaling pathway functions in reproductive regulation. The results provide new ideas and a theoretical basis for reducing follicular atresia and improving the reproductive efficiency of domestic animals and reproductive health.
Author Contributions
Conceptualization, J.L. and S.Y.; methodology, N.Q., W.X., M.L. and Y.Q.; software, G.L.; formal analysis, J.L. and S.Y.; data curation, J.L. and N.Q.; writing—original draft preparation, J.L. and N.Q.; writing—review and editing, J.L. and S.Y.; project administration, J.L.; funding acquisition, J.L., S.Y. and G.L. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Natural Science Foundation of Jiangsu Province (BK20200994 to J.L. (Jiying Liu)), Postgraduate Research & Practice Innovation Program of Jiangsu Province National Natural Science (SJCX22_1980 to N.Q. (Nanna Qi)), Foundation of China for Youth (32102542 to G.L. (Gang Luo)), 2020_Basic Science Research of Nantong University (JC2020042 to S.Y. (Shali Yu)).
Institutional Review Board Statement
The study was conducted in accordance with the Jiangsu university of science and technology (G2022SJ12, Zhenjiang, China), and approved by the Ethics Committee.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original data in the article can be obtained directly from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Bao, T.; Yao, J.; Zhou, S.; Ma, Y.; Dong, J.; Zhang, C.; Mi, Y. Naringin prevents follicular atresia by inhibiting oxidative stress in the aging chicken. Poult. Sci. 2022, 101, 101891. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, Y.; Zhang, J.; Du, X.; Li, Q.; Pan, Z. circSLC41A1 Resists Porcine Granulosa Cell Apoptosis and Follicular Atresia by Promoting SRSF1 through miR-9820-5p Sponging. Int. J. Mol. Sci. 2022, 23, 1509. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Cao, Y.; Jiang, Y.; Wei, Y.; Liu, H. Melatonin protects mouse granulosa cells against oxidative damage by inhibiting FOXO1-mediated autophagy: Implication of an antioxidation-independent mechanism. Redox Biol. 2018, 18, 138–157. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.; Pan, Z.; Du, X.; Zhang, J.; Liu, H.; Li, Q. NORHA, a novel follicular atresia-related lncRNA, promotes porcine granulosa cell apoptosis via the miR-183-96-182 cluster and FoxO1 axis. J. Anim. Sci. Biotechnol. 2021, 12, 103. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, Y.; Li, H.; Chen, B.; Liu, Z.; Wu, G.; Li, C.; Li, R.; Cao, Y.; Zhou, J.; et al. Sulforaphane Acts Through NFE2L2 to Prevent Hypoxia-Induced Apoptosis in Porcine Granulosa Cells via Activating Antioxidant Defenses and Mitophagy. J. Agric. Food Chem. 2022, 70, 8097–8110. [Google Scholar] [CrossRef]
- Monsivais, D.; Matzuk, M.M.; Pangas, S.A. The TGF-beta Family in the Reproductive Tract. Cold Spring Harb. Perspect. Biol. 2017, 9, a022251. [Google Scholar] [CrossRef]
- Monsivais, D.; Nagashima, T.; Prunskaite-Hyyrylainen, R.; Nozawa, K.; Shimada, K.; Tang, S.; Hamor, C.; Agno, J.E.; Chen, F.; Masand, R.P.; et al. Endometrial receptivity and implantation require uterine BMP signaling through an ACVR2A-SMAD1/SMAD5 axis. Nat. Commun. 2021, 12, 3386. [Google Scholar] [CrossRef]
- Zhang, K.; Rajput, S.K.; Lee, K.B.; Wang, D.; Huang, J.; Folger, J.K.; Knott, J.G.; Zhang, J.; Smith, G.W. Evidence supporting a role for SMAD2/3 in bovine early embryonic development: Potential implications for embryotropic actions of follistatin. Biol. Reprod. 2015, 93, 86. [Google Scholar] [CrossRef]
- Cheng, J.C.; Fang, L.; Yan, Y.; He, J.; Guo, Y.; Jia, Q.; Gao, Y.; Han, X.; Sun, Y.P. TGF-beta1 stimulates aromatase expression and estradiol production through SMAD2 and ERK1/2 signaling pathways in human granulosa-lutein cells. J. Cell Physiol. 2021, 236, 6619–6629. [Google Scholar] [CrossRef]
- Li, Q.; Agno, J.E.; Edson, M.A.; Nagaraja, A.K.; Nagashima, T.; Matzuk, M.M. Transforming growth factor beta receptor type 1 is essential for female reproductive tract integrity and function. PLoS Genet. 2011, 7, e1002320. [Google Scholar] [CrossRef]
- Liu, J.; Du, X.; Zhou, J.; Pan, Z.; Liu, H.; Li, Q. MicroRNA-26b functions as a proapoptotic factor in porcine follicular Granulosa cells by targeting Sma-and Mad-related protein 4. Biol. Reprod. 2014, 91, 146. [Google Scholar] [CrossRef] [PubMed]
- Terenina, E.; Fabre, S.; Bonnet, A.; Monniaux, D.; Robert-Granie, C.; SanCristobal, M.; Sarry, J.; Vignoles, F.; Gondret, F.; Monget, P.; et al. Differentially expressed genes and gene networks involved in pig ovarian follicular atresia. Physiol. Genom. 2017, 49, 67–80. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.; Du, X.; Zhang, J.; Wang, Y.; Wang, M.; Pan, Z.; Li, Q. SMAD4-induced knockdown of the antisense long noncoding RNA BRE-AS contributes to granulosa cell apoptosis. Mol. Ther. Nucleic Acids 2021, 25, 251–263. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Liu, J.; Pan, Z.; Du, X.; Li, X.; Ma, B.; Yao, W.; Li, Q.; Liu, H. The let-7g microRNA promotes follicular granulosa cell apoptosis by targeting transforming growth factor-beta type 1 receptor. Mol. Cell. Endocrinol. 2015, 409, 103–112. [Google Scholar] [CrossRef]
- Granados-Aparici, S.; Hardy, K.; Franks, S.; Sharum, I.B.; Waite, S.L.; Fenwick, M.A. SMAD3 directly regulates cell cycle genes to maintain arrest in granulosa cells of mouse primordial follicles. Sci. Rep. 2019, 9, 6513. [Google Scholar] [CrossRef]
- Lee, G.S.; Kwak, G.; Bae, J.H.; Han, J.P.; Nam, S.H.; Lee, J.H.; Song, S.; Kim, G.D.; Park, T.S.; Choi, Y.K.; et al. Morc2a p.S87L mutant mice develop peripheral and central neuropathies associated with neuronal DNA damage and apoptosis. Dis. Model. Mech. 2021, 14, dmm049123. [Google Scholar] [CrossRef]
- Gaidt, M.M.; Morrow, A.; Fairgrieve, M.R.; Karr, J.P.; Yosef, N.; Vance, R.E. Self-guarding of MORC3 enables virulence factor-triggered immunity. Nature 2021, 600, 138–142. [Google Scholar] [CrossRef]
- Wang, T.; Qin, Z.Y.; Wen, L.Z.; Guo, Y.; Liu, Q.; Lei, Z.J.; Pan, W.; Liu, K.J.; Wang, X.W.; Lai, S.J.; et al. Epigenetic restriction of Hippo signaling by MORC2 underlies stemness of hepatocellular carcinoma cells. Cell Death Differ. 2018, 25, 2086–2100. [Google Scholar] [CrossRef]
- Groh, S.; Milton, A.V.; Marinelli, L.K.; Sickinger, C.V.; Russo, A.; Bollig, H.; de Almeida, G.P.; Schmidt, A.; Forne, I.; Imhof, A.; et al. Morc3 silences endogenous retroviruses by enabling Daxx-mediated histone H3.3 incorporation. Nat. Commun. 2021, 12, 5996. [Google Scholar] [CrossRef]
- Ding, Q.S.; Zhang, L.; Wang, B.C.; Zeng, Z.; Zou, X.Q.; Cao, P.B.; Zhou, G.M.; Tang, M.; Wu, L.; Wu, L.L.; et al. Aberrant high expression level of MORC2 is a common character in multiple cancers. Hum. Pathol. 2018, 76, 58–67. [Google Scholar] [CrossRef]
- Douse, C.H.; Bloor, S.; Liu, Y.; Shamin, M.; Tchasovnikarova, I.A.; Timms, R.T.; Lehner, P.J.; Modis, Y. Neuropathic MORC2 mutations perturb GHKL ATPase dimerization dynamics and epigenetic silencing by multiple structural mechanisms. Nat. Commun. 2018, 9, 651. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, D.Q. MORC2 regulates DNA damage response through a PARP1-dependent pathway. Nucleic Acids Res. 2019, 47, 8502–8520. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Xie, H.Y.; Yang, L.F.; Zhang, L.; Zhang, F.L.; Liu, H.Y.; Li, D.Q.; Shao, Z.M. Stabilization of MORC2 by estrogen and antiestrogens through GPER1- PRKACA-CMA pathway contributes to estrogen-induced proliferation and endocrine resistance of breast cancer cells. Autophagy 2020, 16, 1061–1076. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Liu, H.Y.; Yu, T.J.; Lu, Q.; Zhang, F.L.; Liu, G.Y.; Shao, Z.M.; Li, D.Q. O-GlcNAcylation of MORC2 at threonine 556 by OGT couples TGF-beta signaling to breast cancer progression. Cell Death Differ. 2022, 29, 861–873. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.L.; Wang, C.Y.; Cai, X.Z.; Chen, W.; Wang, X.H.; Li, F. Identification and expression analysis of a novel CW-type zinc finger protein MORC2 in cancer cells. Anat. Rec. 2010, 293, 1002–1009. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, Q.; Ruan, B.; Chen, W.; Zheng, J.; Xu, B.; Jiang, P.; Miao, Z.; Li, F.; Guo, J.Y.; et al. MORC2 regulates C/EBPalpha-mediated cell differentiation via sumoylation. Cell Death Differ. 2019, 26, 1905–1917. [Google Scholar] [CrossRef]
- Shi, B.; Xue, J.; Zhou, J.; Kasowitz, S.D.; Zhang, Y.; Liang, G.; Guan, Y.; Shi, Q.; Liu, M.; Sha, J.; et al. MORC2B is essential for meiotic progression and fertility. PLoS Genet. 2018, 14, e1007175. [Google Scholar] [CrossRef]
- Zhang, L.; Du, X.; Wei, S.; Li, D.; Li, Q. A comprehensive transcriptomic view on the role of SMAD4 gene by RNAi-mediated knockdown in porcine follicular granulosa cells. Reproduction 2016, 152, 81–89. [Google Scholar] [CrossRef]
- Liu, L.; Luo, G.; Situ, X.; Li, M. Cloning and Identification of Promoter Region of Porcine MORC2 Gene. Chin. J. Anim. Sci. 2022, 1–12. [Google Scholar] [CrossRef]
- Kojima-Kita, K.; Kuramochi-Miyagawa, S.; Nakayama, M.; Miyata, H.; Jacobsen, S.E.; Ikawa, M.; Koseki, H.; Nakano, T. MORC3, a novel MIWI2 association partner, as an epigenetic regulator of piRNA dependent transposon silencing in male germ cells. Sci. Rep. 2021, 11, 20472. [Google Scholar] [CrossRef]
- Inoue, N.; Hess, K.D.; Moreadith, R.W.; Richardson, L.L.; Handel, M.A.; Watson, M.L.; Zinn, A.R. New gene family defined by MORC, a nuclear protein required for mouse spermatogenesis. Hum. Mol. Genet. 1999, 8, 1201–1207. [Google Scholar] [CrossRef] [PubMed]
- Pastor, W.A.; Stroud, H.; Nee, K.; Liu, W.; Pezic, D.; Manakov, S.; Lee, S.A.; Moissiard, G.; Zamudio, N.; Bourc’His, D.; et al. MORC1 represses transposable elements in the mouse male germline. Nat. Commun. 2014, 5, 5795. [Google Scholar] [CrossRef] [PubMed]
- Cheung, S.; Parrella, A.; Rosenwaks, Z.; Palermo, G.D. Genetic and epigenetic profiling of the infertile male. PLoS ONE 2019, 14, e214275. [Google Scholar] [CrossRef]
- Desai, V.P.; Chouaref, J.; Wu, H.; Pastor, W.A.; Kan, R.L.; Oey, H.M.; Li, Z.; Ho, J.; Vonk, K.; San, L.G.D.; et al. The role of MORC3 in silencing transposable elements in mouse embryonic stem cells. Epigenet. Chromatin 2021, 14, 49. [Google Scholar] [CrossRef]
- Liu, H.Y.; Liu, Y.Y.; Yang, F.; Zhang, L.; Zhang, F.L.; Hu, X.; Shao, Z.M.; Li, D.Q. Acetylation of MORC2 by NAT10 regulates cell-cycle checkpoint control and resistance to DNA-damaging chemotherapy and radiotherapy in breast cancer. Nucleic Acids Res. 2020, 48, 3638–3656. [Google Scholar] [CrossRef]
- Li, Q.; Du, X.; Wang, L.; Shi, K.; Li, Q. TGF-beta1 controls porcine granulosa cell states: A miRNA-mRNA network view. Theriogenology 2021, 160, 50–60. [Google Scholar] [CrossRef]
- Du, X.; Wang, L.; Li, Q.; Wu, W.; Shang, P.; Chamba, Y.; Pan, Z.; Li, Q. miR-130a/TGF-beta1 axis is involved in sow fertility by controlling granulosa cell apoptosis. Theriogenology 2020, 157, 407–417. [Google Scholar] [CrossRef]
- Shen, H.; Wang, Y. Activation of TGF-beta1/Smad3 signaling pathway inhibits the development of ovarian follicle in polycystic ovary syndrome by promoting apoptosis of granulosa cells. J. Cell. Physiol. 2019, 234, 11976–11985. [Google Scholar] [CrossRef]
- Zheng, X.; Boerboom, D.; Carriere, P.D. Transforming growth factor-beta1 inhibits luteinization and promotes apoptosis in bovine granulosa cells. Reproduction 2009, 137, 969–977. [Google Scholar] [CrossRef]
- Du, X.; Li, Q.; Yang, L.; Liu, L.; Cao, Q.; Li, Q. SMAD4 activates Wnt signaling pathway to inhibit granulosa cell apoptosis. Cell Death Dis. 2020, 11, 373. [Google Scholar] [CrossRef]
- Liu, L.; Li, Q.; Yang, L.; Li, Q.; Du, X. SMAD4 Feedback Activates the Canonical TGF-beta Family Signaling Pathways. Int. J. Mol. Sci. 2021, 22, 24. [Google Scholar] [CrossRef]
- Sivera, R.; Lupo, V.; Frasquet, M.; Argente-Escrig, H.; Alonso-Perez, J.; Diaz-Manera, J.; Querol, L.; Del, M.G.M.; Ignacio, P.S.; Garcia-Sobrino, T.; et al. Charcot-Marie-Tooth disease due to MORC2 mutations in Spain. Eur. J. Neurol. 2021, 28, 3001–3011. [Google Scholar] [CrossRef]
- Tong, Y.; Li, Y.; Gu, H.; Wang, C.; Liu, F.; Shao, Y.; Li, F. HSF1, in association with MORC2, downregulates ArgBP2 via the PRC2 family in gastric cancer cells. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 1104–1114. [Google Scholar] [CrossRef]
- Guddeti, R.K.; Thomas, L.; Kannan, A.; Karyala, P.; Pakala, S.B. The chromatin modifier MORC2 affects glucose metabolism by regulating the expression of lactate dehydrogenase A through a feed forward loop with c-Myc. FEBS Lett. 2021, 595, 1289–1302. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).