Research Progress on the Protective Effect of Brown Algae-Derived Polysaccharides on Metabolic Diseases and Intestinal Barrier Injury
Abstract
:1. Introduction
2. Brown Algae Polysaccharides
2.1. Classification of Brown Algae Polysaccharides
2.1.1. Alginate
2.1.2. Fucoidan
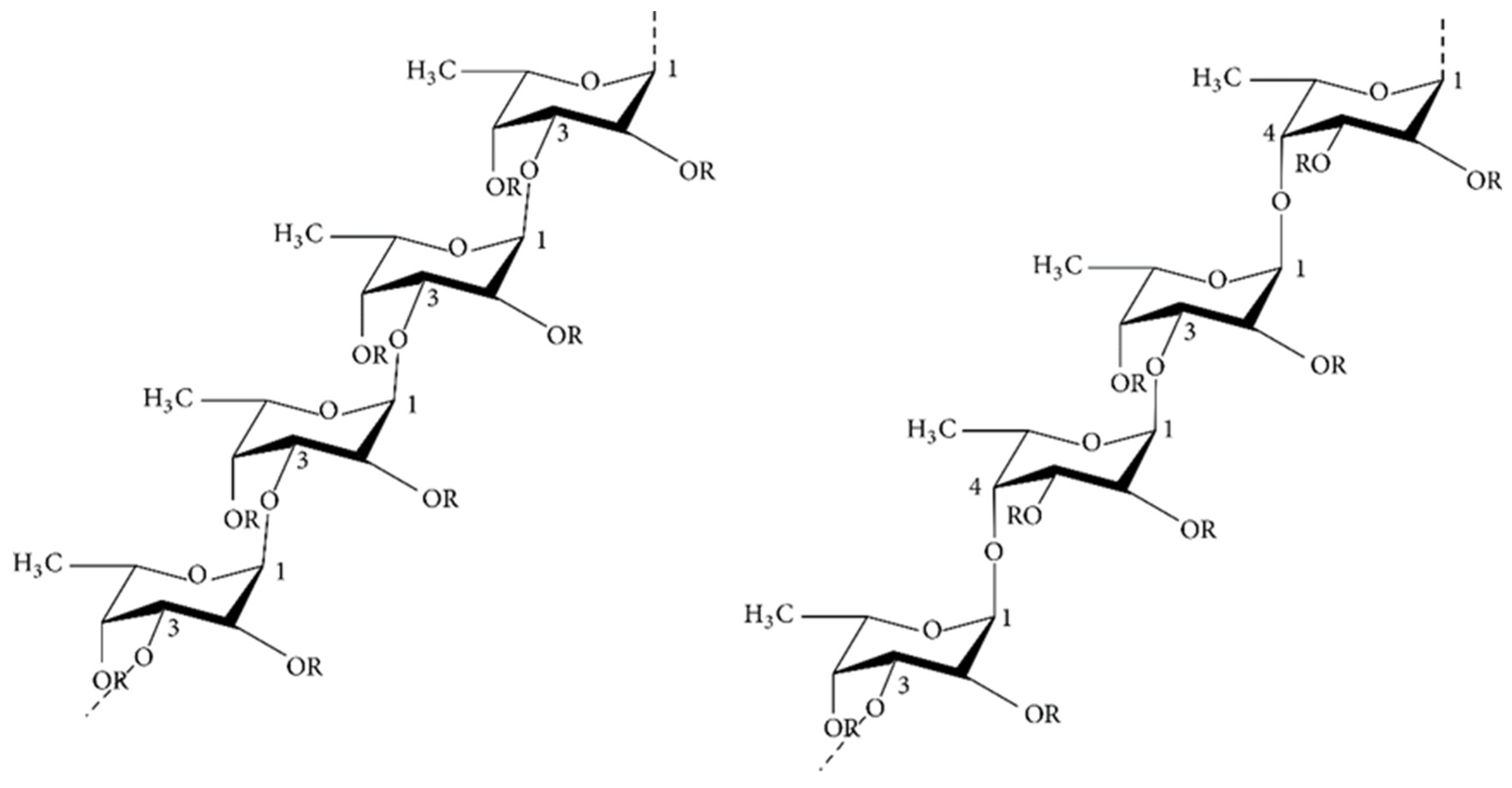
2.1.3. Laminaran
2.2. Extraction of Brown Algae Polysaccharides
3. Protective Effect of Brown Algae-Derived Polysaccharides on Metabolic Diseases and Intestinal Barrier Injury
3.1. Brown Algae-Derived Polysaccharides Maintain Intestinal Barrier Integrity
3.1.1. Maintaining the Integrity of the Physical Barrier
3.1.2. Enhancement of Intestinal Chemical Barrier Function
3.1.3. Improving Intestinal Immune Barrier Protection
3.1.4. Maintenance of Intestinal Microbial Barrier by the Intestinal Microbiota Balance
3.2. Inhibition of Lipid Peroxidation Damage by Brown Algae-Derived Polysaccharides
3.3. Inhibition of Inflammatory Cytokines by Brown Algae-Derived Polysaccharides
4. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, S.; Yu, M. Role of Goblet Cells in Intestinal Barrier and Mucosal Immunity. J. Inflamm. Res. 2021, 14, 3171–3183. [Google Scholar] [CrossRef] [PubMed]
- Bain, C.C.; Schridde, A. Origin, Differentiation, and Function of Intestinal Macrophages. Front. Immunol. 2018, 9, 2733. [Google Scholar] [CrossRef] [PubMed]
- Huo, J.; Wu, Z.; Sun, W.; Wang, Z.; Wu, J.; Huang, M.; Wang, B.; Sun, B. Protective Effects of Natural Polysaccharides on Intestinal Barrier Injury: A Review. J. Agric. Food Chem. 2022, 70, 711–735. [Google Scholar] [CrossRef] [PubMed]
- Sauruk da Silva, K.; Carla da Silveira, B.; Bueno, L.R.; Malaquias da Silva, L.C.; da Silva Fonseca, L.; Fernandes, E.S.; Maria-Ferreira, D. Beneficial Effects of Polysaccharides on the Epithelial Barrier Function in Intestinal Mucositis. Front. Physiol. 2021, 12, 714846. [Google Scholar] [CrossRef]
- Bao, Y.F.; Dong, C.; Ji, J.; Gu, Z.F. Dysregulation of gut microbiome is linked to disease activity of rheumatic diseases. Clin. Rheumatol. 2020, 39, 2523–2528. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wu, S.Y.; Cheng, Y.; Liu, Q.H.; Su, L.J.; Yang, Y.; Zhang, X.; Wu, M.J.; Choi, J.I.; Tong, H.B. Sargassum fusiforme Alginate Relieves Hyperglycemia and Modulates Intestinal Microbiota and Metabolites in Type 2 Diabetic Mice. Nutrients 2021, 13, 2887. [Google Scholar] [CrossRef]
- Ma, Q.T.; Li, Y.Q.; Li, P.F.; Wang, M.; Wang, J.K.; Tang, Z.Y.; Wang, T.; Luo, L.L.; Wang, C.G.; Zhao, B.S. Research progress in the relationship between type 2 diabetes mellitus and intestinal flora. Biomed. Pharm. 2019, 117, 109138. [Google Scholar] [CrossRef]
- Tang, Q.; Cao, L. Intestinal flora and neurological disorders. Sheng Wu Gong Cheng Xue Bao = Chin. J. Biotechnol. 2021, 37, 3757–3780. [Google Scholar] [CrossRef]
- Hills, R.D., Jr.; Pontefract, B.A.; Mishcon, H.R.; Black, C.A.; Sutton, S.C.; Theberge, C.R. Gut Microbiome: Profound Implications for Diet and Disease. Nutrients 2019, 11, 1613. [Google Scholar] [CrossRef]
- Xu, S.Y.; Huang, X.; Cheong, K.L. Recent Advances in Marine Algae Polysaccharides: Isolation, Structure, and Activities. Mar. Drugs 2017, 15, 388. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.P.; Zheng, Y.T.; Zhang, Y.; Yang, Y.Y.; Wang, P.Y.; Imre, B.; Wong, A.C.Y.; Hsieh, Y.S.Y.; Wang, D.M. Brown Algae Carbohydrates: Structures, Pharmaceutical Properties, and Research Challenges. Mar. Drugs 2021, 19, 620. [Google Scholar] [CrossRef] [PubMed]
- Chi, S.; Liu, T.; Wang, X.; Wang, R.; Wang, S.; Wang, G.; Shan, G.; Liu, C. Functional genomics analysis reveals the biosynthesis pathways of important cellular components (alginate and fucoidan) of Saccharina. Curr. Genet. 2018, 64, 259–273. [Google Scholar] [CrossRef] [PubMed]
- Dobrincic, A.; Balbino, S.; Zoric, Z.; Pedisic, S.; Bursac Kovacevic, D.; Elez Garofulic, I.; Dragovic-Uzelac, V. Advanced Technologies for the Extraction of Marine Brown Algal Polysaccharides. Mar. Drugs 2020, 18, 168. [Google Scholar] [CrossRef]
- Hwang, P.A.; Phan, N.N.; Lu, W.J.; Hieu, B.T.N.; Lin, Y.C. Low-molecular-weight fucoidan and high-stability fucoxanthin from brown seaweed exert prebiotics and anti-inflammatory activities in Caco-2 cells. Food Nutr. Res. 2016, 60, 32033. [Google Scholar] [CrossRef] [PubMed]
- Iraha, A.; Chinen, H.; Hokama, A.; Yonashiro, T.; Kinjo, T.; Kishimoto, K.; Nakamoto, M.; Hirata, T.; Kinjo, N.; Higa, F.; et al. Fucoidan enhances intestinal barrier function by upregulating the expression of claudin-1. World J. Gastroenterol. 2013, 19, 5500–5507. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Luthuli, S.; Yang, Y.; Cheng, Y.; Zhang, Y.; Wu, M.J.; Choi, J.I.; Tong, H.B. Therapeutic and nutraceutical potentials of a brown seaweed Sargassum Fusiforme. Food Sci. Nutr. 2020, 8, 5195–5205. [Google Scholar] [CrossRef]
- Xue, M.L.; Ji, X.Q.; Liang, H.; Liu, Y.; Wang, B.; Sun, L.L.; Li, W.W. The effect of fucoidan on intestinal flora and intestinal barrier function in rats with breast cancer. Food Funct. 2018, 9, 1214–1223. [Google Scholar] [CrossRef]
- Holdt, S.L.; Kraan, S. Bioactive compounds in seaweed: Functional food applications and legislation. J. Appl. Phycol. 2011, 23, 543–597. [Google Scholar] [CrossRef]
- Charoensiddhi, S.; Conlon, M.A.; Vuaran, M.S.; Franco, C.M.M.; Zhang, W. Impact of extraction processes on prebiotic potential of the brown seaweed Ecklonia radiata by in vitro human gut bacteria fermentation. J. Funct. Foods 2016, 24, 221–230. [Google Scholar] [CrossRef]
- Lee, S.H.; Ko, C.I.; Jee, Y.; Jeong, Y.; Kim, M.; Kim, J.S.; Jeon, Y.J. Anti-inflammatory effect of fucoidan extracted from Ecklonia cava in zebrafish model. Carbohydr. Polym. 2013, 92, 84–89. [Google Scholar] [CrossRef]
- Oh, S.; Shim, M.; Son, M.; Jang, J.T.; Son, K.H.; Byun, K. Attenuating Effects of Dieckol on Endothelial Cell Dysfunction via Modulation of Th17/Treg Balance in the Intestine and Aorta of Spontaneously Hypertensive Rats. Antioxidants 2021, 10, 298. [Google Scholar] [CrossRef]
- Khuituan, P.; Huipao, N.; Jeanmard, N.; Thantongsakul, S.; Promjun, W.; Chuthong, S.; Tipbunjong, C.; Peerakietkhajorn, S. Sargassum plagiophyllum Extract Enhances Colonic Functions and Modulates Gut Microbiota in Constipated Mice. Nutrients 2022, 14, 496. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Yan, J.; Bak, J.; Park, J.; Lee, H.; Kim, H. Sargassum thunbergii Extract Attenuates High-Fat Diet-Induced Obesity in Mice by Modulating AMPK Activation and the Gut Microbiota. Foods 2022, 11, 2529. [Google Scholar] [CrossRef]
- Wu, Q.; Wu, S.; Cheng, Y.; Zhang, Z.; Mao, G.; Li, S.; Yang, Y.; Zhang, X.; Wu, M.; Tong, H. Sargassum fusiforme fucoidan modifies gut microbiota and intestinal metabolites during alleviation of hyperglycemia in type 2 diabetic mice. Food Funct. 2021, 12, 3572–3585. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zuo, J.; Yan, L.; Cheng, Y.; Li, Q.; Wu, S.; Chen, L.; Thring, R.W.; Yang, Y.; Gao, Y.; et al. Sargassum fusiforme Fucoidan Alleviates High-Fat Diet-Induced Obesity and Insulin Resistance Associated with the Improvement of Hepatic Oxidative Stress and Gut Microbiota Profile. J. Agric. Food Chem. 2020, 68, 10626–10638. [Google Scholar] [CrossRef]
- Fu, X.; Zhan, Y.; Li, N.; Yu, D.; Gao, W.; Gu, Z.; Zhu, L.; Li, R.; Zhu, C. Enzymatic Preparation of Low-Molecular-Weight Laminaria japonica Polysaccharides and Evaluation of Its Effect on Modulating Intestinal Microbiota in High-Fat-Diet-Fed Mice. Front. Bioeng. Biotechnol. 2021, 9, 820892. [Google Scholar] [CrossRef]
- Bolton, J.J. The biogeography of kelps (Laminariales, Phaeophyceae): A global analysis with new insights from recent advances in molecular phylogenetics. Helgol. Mar. Res. 2010, 64, 263–279. [Google Scholar] [CrossRef]
- Wang, L.; Ai, C.; Wen, C.; Qin, Y.; Liu, Z.; Wang, L.; Gong, Y.; Su, C.; Wang, Z.; Song, S. Fucoidan isolated from Ascophyllum nodosum alleviates gut microbiota dysbiosis and colonic inflammation in antibiotic-treated mice. Food Funct. 2020, 11, 5595–5606. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L.; Morrison, L.; Shukla, P.S.; Critchley, A.T. A concise review of the brown macroalga Ascophyllum nodosum (Linnaeus) Le Jolis. J. Appl. Phycol. 2020, 32, 3561–3584. [Google Scholar] [CrossRef]
- Vodouhe, M.; Marois, J.; Guay, V.; Leblanc, N.; Weisnagel, S.J.; Bilodeau, J.F.; Jacques, H. Marginal Impact of Brown Seaweed Ascophyllum nodosum and Fucus vesiculosus Extract on Metabolic and Inflammatory Response in Overweight and Obese Prediabetic Subjects. Mar. Drugs 2022, 20, 174. [Google Scholar] [CrossRef]
- Wang, X.; Shan, X.; Dun, Y.; Cai, C.; Hao, J.; Li, G.; Cui, K.; Yu, G. Anti-Metabolic Syndrome Effects of Fucoidan from Fucus vesiculosus via Reactive Oxygen Species-Mediated Regulation of JNK, Akt, and AMPK Signaling. Molecules 2019, 24, 3319. [Google Scholar] [CrossRef]
- Jiang, P.; Zheng, W.; Sun, X.; Jiang, G.; Wu, S.; Xu, Y.; Song, S.; Ai, C. Sulfated polysaccharides from Undaria pinnatifida improved high fat diet-induced metabolic syndrome, gut microbiota dysbiosis and inflammation in BALB/c mice. Int. J. Biol. Macromol 2021, 167, 1587–1597. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.P.; Jia, J.H.; Jiang, P.R.; Zheng, W.Y.; Li, X.F.; Song, S.; Ai, C.Q. Polysaccharides from edible brown seaweed Undaria pinnatifida are effective against high-fat diet-induced obesity in mice through the modulation of intestinal microecology. Food Funct. 2022, 13, 2581–2593. [Google Scholar] [CrossRef] [PubMed]
- Jia, R.B.; Li, Z.R.; Lin, L.Z.; Luo, D.H.; Chen, C.; Zhao, M.M. The potential mechanisms of Macrocystis pyrifera polysaccharides mitigating type 2 diabetes in rats. Food Funct. 2022, 13, 7918–7929. [Google Scholar] [CrossRef] [PubMed]
- Purcell-Meyerink, D.; Packer, M.A.; Wheeler, T.T.; Hayes, M. Aquaculture Production of the Brown Seaweeds Laminaria digitata and Macrocystis pyrifera: Applications in Food and Pharmaceuticals. Molecules 2021, 26, 1306. [Google Scholar] [CrossRef]
- Brownlee, I.A.; Allen, A.; Pearson, J.P.; Dettmar, P.W.; Havler, M.E.; Atherton, M.R.; Onsoyen, E. Alginate as a source of dietary fiber. Crit. Rev. Food Sci. Nutr. 2005, 45, 497–510. [Google Scholar] [CrossRef]
- Fawzy, M.A.; Gomaa, M.; Hifney, A.F.; Abdel-Gawad, K.M. Optimization of alginate alkaline extraction technology from Sargassum latifolium and its potential antioxidant and emulsifying properties. Carbohydr. Polym. 2017, 157, 1903–1912. [Google Scholar] [CrossRef]
- Augst, A.D.; Kong, H.J.; Mooney, D.J. Alginate hydrogels as biomaterials. Macromol. Biosci. 2006, 6, 623–633. [Google Scholar] [CrossRef]
- Cong, Q.F.; Xiao, F.; Liao, W.F.; Dong, Q.; Ding, K. Structure and biological activities of an alginate from Sargassum fusiforme, and its sulfated derivative. Int. J. Biol. Macromol. 2014, 69, 252–259. [Google Scholar] [CrossRef]
- Sen, M. Effects of molecular weight and ratio of guluronic acid to mannuronic acid on the antioxidant properties of sodium alginate fractions prepared by radiation-induced degradation. Appl. Radiat. Isot. 2011, 69, 126–129. [Google Scholar] [CrossRef]
- den Besten, G.; van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 2013, 54, 2325–2340. [Google Scholar] [CrossRef]
- Gupta, S.; Lokesh, J.; Abdelhafiz, Y.; Siriyappagouder, P.; Pierre, R.; Sorensen, M.; Fernandes, J.M.O.; Kiron, V. Macroalga-Derived Alginate Oligosaccharide Alters Intestinal Bacteria of Atlantic Salmon. Front. Microbiol. 2019, 10, 2037. [Google Scholar] [CrossRef] [PubMed]
- Miao, B.C.; Geng, M.Y.; Li, J.; Li, F.C.; Chen, H.X.; Guan, H.S.; Ding, J. Sulfated polymannuroguluronate, a novel anti-acquired immune deficiency syndrome (AIDS) drug candidate, targeting CD4 in lymphocytes. Biochem. Pharm. 2004, 68, 641–649. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.Q.; Han, W.W.; Zhang, M.F.; Zang, R.C.; Du, K.X.; Li, L.; Ma, X.M.; Li, C.X.; Wang, S.X.; Qiu, P.J.; et al. Sulfated polymannuroguluronate TGC161 ameliorates leukopenia by inhibiting CD4(+) T cell apoptosis. Carbohydr. Polym. 2020, 247, 116728. [Google Scholar] [CrossRef] [PubMed]
- Salih, A.E.M.; Thissera, B.; Yaseen, M.; Hassane, A.S.I.; El-Seedi, H.R.; Sayed, A.M.; Rateb, M.E. Marine Sulfated Polysaccharides as Promising Antiviral Agents: A Comprehensive Report and Modeling Study Focusing on SARS CoV-2. Mar. Drugs 2021, 19, 406. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Aroca, A.; Ferrandis-Montesinos, M.; Wang, R.B. Antiviral Properties of Alginate-Based Biomaterials: Promising Antiviral Agents against SARS-CoV-2. ACS Appl. Bio Mater. 2021, 4, 5897–5907. [Google Scholar] [CrossRef]
- Liu, F.; Wang, X.; Shi, H.J.; Wang, Y.M.; Xue, C.H.; Tang, Q.J. Polymannuronic acid ameliorated obesity and inflammation associated with a high-fat and high-sucrose diet by modulating the gut microbiome in a murine model. Brit. J. Nutr. 2017, 117, 1332–1342. [Google Scholar] [CrossRef]
- Vo, T.S.; Kim, S.K. Fucoidans as a natural bioactive ingredient for functional foods. J. Funct. Foods 2013, 5, 16–27. [Google Scholar] [CrossRef]
- Ale, M.T.; Mikkelsen, J.D.; Meyer, A.S. Important Determinants for Fucoidan Bioactivity: A Critical Review of Structure-Function Relations and Extraction Methods for Fucose-Containing Sulfated Polysaccharides from Brown Seaweeds. Mar. Drugs 2011, 9, 2106–2130. [Google Scholar] [CrossRef]
- Berteau, O.; Mulloy, B. Sulfated fucans, fresh perspectives: Structures, functions, and biological properties of sulfated fucans and an overview of enzymes active toward this class of polysaccharide. Glycobiology 2003, 13, 29R–40R. [Google Scholar] [CrossRef] [Green Version]
- Cumashi, A.; Ushakova, N.A.; Preobrazhenskaya, M.E.; D’Incecco, A.; Piccoli, A.; Totani, L.; Tinari, N.; Morozevich, G.E.; Berman, A.E.; Bilan, M.I.; et al. A comparative study of the anti-inflammatory, anticoagulant, antiangiogenic, and antiadhesive activities of nine different fucoidans from brown seaweeds. Glycobiology 2007, 17, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Pomin, V.H.; Mourao, P.A.S. Structure, biology, evolution, and medical importance of sulfated fucans and galactans. Glycobiology 2008, 18, 1016–1027. [Google Scholar] [CrossRef]
- Luo, J.M.; Wang, Z.; Fan, B.; Wang, L.; Liu, M.Y.; An, Z.Z.; Zhao, X. A comparative study of the effects of different fucoidans on cefoperazone-induced gut microbiota disturbance and intestinal inflammation. Food Funct. 2021, 12, 9087–9097. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.J.; Chang, Y.G.; Gao, Y.; Wang, X.; Chen, X.; Wang, Y.M.; Xue, C.H.; Tang, Q.J. Dietary fucoidan of Acaudina molpadioides alters gut microbiota and mitigates intestinal mucosal injury induced by cyclophosphamide. Food Funct. 2017, 8, 3383–3393. [Google Scholar] [CrossRef]
- Ahmadi, A.; Moghadamtousi, S.Z.; Abubakar, S.; Zandi, K. Antiviral Potential of Algae Polysaccharides Isolated from Marine Sources: A Review. BioMed Res. Int. 2015, 2015, 825203. [Google Scholar] [CrossRef] [PubMed]
- Imbs, T.I.; Ermakova, S.P.; Malyarenko, O.S.; Isakov, V.V.; Zvyagintseva, T.N. Structural elucidation of polysaccharide fractions from the brown alga Coccophora langsdorfii and in vitro investigation of their anticancer activity. Carbohydr. Polym. 2016, 135, 162–168. [Google Scholar] [CrossRef]
- Rioux, L.E.; Turgeon, S.L.; Beaulieu, M. Structural characterization of laminaran and galactofucan extracted from the brown seaweed Saccharina longicruris. Phytochemistry 2010, 71, 1586–1595. [Google Scholar] [CrossRef]
- Pereira, M.S.; Mulloy, B.; Mourao, P.A.S. Structure and anticoagulant activity of sulfated fucans: Comparison between the regular, repetitive, and linear fucans from echinoderms with the more heterogeneous and branched polymers from brown algae. J. Biol. Chem. 1999, 274, 7656–7667. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, S.G.; Kim, J.; Guevarra, R.B.; Lee, J.H.; Kim, E.; Kim, S.I.; Unno, T. Laminarin favorably modulates gut microbiota in mice fed a high-fat diet. Food Funct. 2016, 7, 4193–4201. [Google Scholar] [CrossRef]
- An, C.; Kuda, T.; Yazaki, T.; Takahashi, H.; Kimura, B. FLX Pyrosequencing Analysis of the Effects of the Brown-Algal Fermentable Polysaccharides Alginate and Laminaran on Rat Cecal Microbiotas. Appl. Environ. Microb. 2013, 79, 860–866. [Google Scholar] [CrossRef] [Green Version]
- Zhang, R.; Zhang, X.X.; Tang, Y.X.; Mao, J.L. Composition, isolation, purification and biological activities of Sargassum fusiforme polysaccharides: A review. Carbohydr. Polym. 2020, 228, 115381. [Google Scholar] [CrossRef]
- Zhao, S.F.; He, Y.; Wang, C.G.; Assani, I.; Hou, P.L.; Feng, Y.; Yang, J.J.; Wang, Y.H.; Liao, Z.X.; Shen, S.D. Isolation, Characterization and Bioactive Properties of Alkali-Extracted Polysaccharides from Enteromorpha prolifera. Mar. Drugs 2020, 18, 552. [Google Scholar] [CrossRef] [PubMed]
- Hahn, T.; Lang, S.; Ulber, R.; Muffler, K. Novel procedures for the extraction of fucoidan from brown algae. Process. Biochem. 2012, 47, 1691–1698. [Google Scholar] [CrossRef]
- Jia, R.B.; Li, Z.R.; Wu, J.; Ou, Z.R.; Zhu, Q.Y.; Sun, B.G.; Lin, L.Z.; Zhao, M.M. Physicochemical properties of polysaccharide fractions from Sargassum fusiforme and their hypoglycemic and hypolipidemic activities in type 2 diabetic rats. Int. J. Biol. Macromol. 2020, 147, 428–438. [Google Scholar] [CrossRef]
- Jiang, H.; Yang, S.Q.; Chakka, V.P.; Qian, W.W.; Wei, X.Y.; Zhu, Q.; Zhou, T. Purification and Biological Activities of Enzymatically Degraded Sargassum fusiforme Polysaccharides. Chem. Biodivers. 2021, 18, e2000930. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xu, M.; Hu, C.; Liu, A.; Chen, J.; Gu, C.; Zhang, X.; You, C.; Tong, H.; Wu, M.; et al. Sargassum fusiforme Fucoidan SP2 Extends the Lifespan of Drosophila melanogaster by Upregulating the Nrf2-Mediated Antioxidant Signaling Pathway. Oxid. Med. Cell. Longev. 2019, 2019, 8918914. [Google Scholar] [CrossRef] [PubMed]
- Abraham, R.E.; Su, P.; Puri, M.; Raston, C.L.; Zhang, W. Optimisation of biorefinery production of alginate, fucoidan and laminarin from brown seaweed Durvillaea potatorum. Algal Res. 2019, 38, 101389. [Google Scholar] [CrossRef]
- January, G.G.; Naidoo, R.K.; Kirby-McCullough, B.; Bauer, R. Assessing methodologies for fucoidan extraction from South African brown algae. Algal Res. 2019, 40, 101517, Corrigendum in Algal Res. 2019, 41, 101571. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Chattopadhyay, R.; Mitra, S.; Crowe, S.E. Oxidative Stress: An Essential Factor in the Pathogenesis of Gastrointestinal Mucosal Diseases. Physiol. Rev. 2014, 94, 329–354. [Google Scholar] [CrossRef]
- Mall, J.P.G.; Fart, F.; Sabet, J.A.; Lindqvist, C.M.; Nestestog, R.; Hegge, F.T.; Keita, A.V.; Brummer, R.J.; Schoultz, I. Effects of Dietary Fibres on Acute Indomethacin-Induced Intestinal Hyperpermeability in the Elderly: A Randomised Placebo Controlled Parallel Clinical Trial. Nutrients 2020, 12, 1954. [Google Scholar] [CrossRef]
- Ikeda-Ohtsubo, W.; Nadal, A.L.; Zaccaria, E.; Iha, M.; Kitazawa, H.; Kleerebezem, M.; Brugman, S. Intestinal Microbiota and Immune Modulation in Zebrafish by Fucoidan From Okinawa Mozuku (Cladosiphon okamuranus). Front. Nutr. 2020, 7, 67. [Google Scholar] [CrossRef] [PubMed]
- Kong, Q.H.; Zhang, R.F.; You, L.J.; Ma, Y.X.; Liao, L.; Pedisic, S. In vitro fermentation characteristics of polysaccharide from Sargassum fusiforme and its modulation effects on gut microbiota. Food Chem. Toxicol. 2021, 151, 112145. [Google Scholar] [CrossRef]
- Huang, J.; Huang, J.L.; Li, Y.; Wang, Y.L.; Wang, F.H.; Qiu, X.; Liu, X.; Li, H.J. Sodium Alginate Modulates Immunity, Intestinal Mucosal Barrier Function, and Gut Microbiota in Cyclophosphamide-Induced Immunosuppressed BALB/c Mice. J. Agric Food Chem. 2021, 69, 7064–7073. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.W.; Hwang, S.J.; Han, M.H.; Lee, D.S.; Yoo, J.S.; Choi, I.W.; Cha, H.J.; Kim, S.; Kim, H.S.; Kim, G.Y.; et al. Fucoidan inhibits lipopolysaccharide-induced inflammatory responses in RAW 264.7 macrophages and zebrafish larvae. Mol. Cell Toxicol. 2017, 13, 405–417. [Google Scholar] [CrossRef]
- Ramnani, P.; Chitarrari, R.; Tuohy, K.; Grant, J.; Hotchkiss, S.; Philp, K.; Campbell, R.; Gill, C.; Rowland, I. In vitro fermentation and prebiotic potential of novel low molecular weight polysaccharides derived from agar and alginate seaweeds. Anaerobe 2012, 18, 1–6. [Google Scholar] [CrossRef]
- Ai, C.Q.; Jiang, P.R.; Liu, Y.L.; Duan, M.M.; Sun, X.N.; Luo, T.R.; Jiang, G.P.; Song, S. The specific use of alginate from Laminaria japonica by Bacteroides species determined its modulation of the Bacteroides community. Food Funct. 2019, 10, 4304–4314. [Google Scholar] [CrossRef] [PubMed]
- Nakata, T.; Kyoui, D.; Takahashi, H.; Kimura, B.; Kuda, T. Inhibitory effects of laminaran and alginate on production of putrefactive compounds from soy protein by intestinal microbiota in vitro and in rats. Carbohydr Polym. 2016, 143, 61–69. [Google Scholar] [CrossRef]
- Tang, Y.P.; Pu, Q.Y.; Zhao, Q.L.; Zhou, Y.F.; Jiang, X.X.; Han, T. Effects of Fucoidan Isolated From Laminaria japonica on Immune Response and Gut Microbiota in Cyclophosphamide-Treated Mice. Front. Immunol. 2022, 13, 916618. [Google Scholar] [CrossRef]
- Fu, X.; Cao, C.L.; Ren, B.B.; Zhang, B.; Huang, Q.; Li, C. Structural characterization and in vitro fermentation of a novel polysaccharide from Sargassum thunbergii and its impact on gut microbiota. Carbohydr. Polym. 2018, 183, 230–239. [Google Scholar] [CrossRef]
- Shang, Q.S.; Shan, X.D.; Cai, C.; Hao, J.J.; Li, G.Y.; Yu, G.L. Dietary fucoidan modulates the gut microbiota in mice by increasing the abundance of Lactobacillus and Ruminococcaceae. Food Funct. 2016, 7, 3224–3232. [Google Scholar] [CrossRef]
- Vancamelbeke, M.; Vermeire, S. The intestinal barrier: A fundamental role in health and disease. Expert Rev. Gastroent 2017, 11, 821–834. [Google Scholar] [CrossRef]
- Branca, J.J.V.; Gulisano, M.; Nicoletti, C. Intestinal epithelial barrier functions in ageing. Ageing Res. Rev. 2019, 54, 100938. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T. Regulation of the intestinal barrier by nutrients: The role of tight junctions. Anim. Sci. J. 2020, 91, e13357. [Google Scholar] [CrossRef] [PubMed]
- Bhat, A.A.; Uppada, S.; Achkar, I.W.; Hashem, S.; Yadav, S.K.; Shanmugakonar, M.; Al-Naemi, H.A.; Haris, M.; Uddin, S. Tight Junction Proteins and Signaling Pathways in Cancer and Inflammation: A Functional Crosstalk. Front. Physiol. 2018, 9, 1942. [Google Scholar] [CrossRef] [PubMed]
- Groschwitz, K.R.; Hogan, S.P. Intestinal barrier function: Molecular regulation and disease pathogenesis. J. Allergy Clin. Immunol. 2009, 124, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Matayoshi, M.; Teruya, J.; Yasumoto-Hirose, M.; Teruya, R.; Miura, N.; Takeda, R. Improvement of defecation in healthy individuals with infrequent bowel movements through the ingestion of dried Mozuku powder: A randomized, double-blind, parallel-group study. Funct. Foods Health Dis. 2017, 7, 735–742. [Google Scholar] [CrossRef]
- Sun, T.; Liang, H.; Xue, M.L.; Liu, Y.; Gong, A.J.; Jiang, Y.S.; Qin, Y.M.; Yang, J.; Meng, D.Y. Protective effect and mechanism of fucoidan on intestinal mucosal barrier function in NOD mice. Food Agric. Immunol. 2020, 31, 922–936. [Google Scholar] [CrossRef]
- Wan, J.; Zhang, J.; Yin, H.; Chen, D.; Yu, B.; He, J. Ameliorative effects of alginate oligosaccharide on tumour necrosis factor-alpha-induced intestinal epithelial cell injury. Int. Immunopharmacol. 2020, 89, 107084. [Google Scholar] [CrossRef]
- Ding, X.M.; Hu, X.Y.; Chen, Y.; Xie, J.H.; Ying, M.X.; Wang, Y.D.; Yu, Q. Differentiated Caco-2 cell models in food-intestine interaction study: Current applications and future trends. Trends Food Sci. Technol. 2021, 107, 455–465. [Google Scholar] [CrossRef]
- Zhang, M.M.; Wu, C.C. The relationship between intestinal goblet cells and the immune response. Biosci. Rep. 2020, 40, 10. [Google Scholar] [CrossRef]
- Sicard, J.F.; Le Bihan, G.; Vogeleer, P.; Jacques, M.; Harel, J. Interactions of Intestinal Bacteria with Components of the Intestinal Mucus. Front. Cell. Infect. Microbiol. 2017, 7, 387. [Google Scholar] [CrossRef] [PubMed]
- Paone, P.; Cani, P.D. Mucus barrier, mucins and gut microbiota: The expected slimy partners? Gut 2020, 69, 2232–2243. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.G.; Ryan, M.; O’Doherty, J.V.; Reilly, P.; Bahar, B.; Sweeney, T. Effects of dietary supplementation with laminarin derived from Laminaria hyperborea and Laminaria digitata on colonic mucin gene expression in pigs. Livest. Sci. 2010, 133, 204–206. [Google Scholar] [CrossRef]
- Gao, J.; Lin, L.Z.; Sun, B.G.; Zhao, M.M. Comparison Study on Polysaccharide Fractions from Laminaria japonica: Structural Characterization and Bile Acid Binding Capacity. J. Agric. Food Chem. 2017, 65, 9790–9798. [Google Scholar] [CrossRef] [PubMed]
- Ruder, B.; Becker, C. At the Forefront of the Mucosal Barrier: The Role of Macrophages in the Intestine. Cells 2020, 9, 2162. [Google Scholar] [CrossRef]
- Zhou, H.; Wang, L.; Liu, F. Immunological Impact of Intestinal T Cells on Metabolic Diseases. Front. Immunol. 2021, 12, 639902. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Ding, R.X.; Sun, J.; Liu, J.; Kan, J.; Jin, C.H. The impacts of natural polysaccharides on intestinal microbiota and immune responses-a review. Food Funct. 2019, 10, 2290–2312. [Google Scholar] [CrossRef]
- Xu, X.F.; Zhang, X.W. Effects of cyclophosphamide on immune system and gut microbiota in mice. Microbiol. Res. 2015, 171, 97–106. [Google Scholar] [CrossRef]
- Li, W.J.; Li, L.; Zhen, W.Y.; Wang, L.F.; Pan, M.; Lv, J.Q.; Wang, F.; Yao, Y.F.; Nie, S.P.; Xie, M.Y. Ganoderma atrum polysaccharide ameliorates ROS generation and apoptosis in spleen and thymus of immunosuppressed mice. Food Chem. Toxicol. 2017, 99, 199–208. [Google Scholar] [CrossRef]
- Feng, G.; Su, L.J.; Chen, S.; Teng, W.; Zhou, D.J.; Yin, C.; Guo, S.D. In vitro and in vivo immunoregulatory activity of sulfated fucan from the sea cucumber A. leucoprocta. Int. J. Biol. Macromol. 2021, 187, 931–938. [Google Scholar] [CrossRef]
- Yamamoto, M.; Yoshizaki, K.; Kishimoto, T.; Ito, H. IL-6 is required for the development of Th1 cell-mediated murine colitis. J. Immunol. 2000, 164, 4878–4882. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, S.; Nagaoka, M.; Hara, T.; Kimura-Takagi, I.; Mistuyama, K.; Ueyama, S. Fucoidan derived from Cladosiphon okamuranus tokida ameliorates murine chronic colitis through the down-regulation of interleukin-6 production on colonic epithelial cells. Clin. Exp. Immunol. 2004, 136, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Raghavendran, H.R.B.; Srinivasan, P.; Rekha, S. Immunomodulatory activity of fucoidan against aspirin-induced gastric mucosal damage in rats. Int. Immunopharmacol. 2011, 11, 157–163. [Google Scholar] [CrossRef]
- Artis, D. Epithelial-cell recognition of commensal bacteria and maintenance of immune homeostasis in the gut. Nat. Rev. Immunol. 2008, 8, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Li, S.Y.; Wang, L.N.; Liu, B.; He, N.N. Unsaturated alginate oligosaccharides attenuated obesity-related metabolic abnormalities by modulating gut microbiota in high-fat-diet mice. Food Funct. 2020, 11, 4773–4784. [Google Scholar] [CrossRef]
- Luthuli, S.; Wu, S.; Cheng, Y.; Zheng, X.; Wu, M.; Tong, H. Therapeutic Effects of Fucoidan: A Review on Recent Studies. Mar. Drugs 2019, 17, 487. [Google Scholar] [CrossRef]
- Vandana, U.K.; Barlaskar, N.H.; Gulzar, A.B.M.; Laskar, I.H.; Kumar, D.; Paul, P.; Pandey, P.; Mazumder, P.B. Linking gut microbiota with the human diseases. Bioinformation 2020, 16, 196–208. [Google Scholar] [CrossRef]
- Spiljar, M.; Merkler, D.; Trajkovski, M. The Immune System Bridges the Gut Microbiota with Systemic Energy Homeostasis: Focus on TLRs, Mucosal Barrier, and SCFAs. Front. Immunol. 2017, 8, 1353. [Google Scholar] [CrossRef]
- Sommer, F.; Ruhlemann, M.C.; Bang, C.; Hoppner, M.; Rehman, A.; Kaleta, C.; Schmitt-Kopplin, P.; Dempfle, A.; Weidinger, S.; Ellinghaus, E.; et al. Microbiomarkers in inflammatory bowel diseases: Caveats come with caviar. Gut 2017, 66, 1734–1738. [Google Scholar] [CrossRef]
- Walsh, A.M.; Sweeney, T.; O’Shea, C.J.; Doyle, D.N.; O’Doherty, J.V. Effect of dietary laminarin and fucoidan on selected microbiota, intestinal morphology and immune status of the newly weaned pig. Brit. J. Nutr. 2013, 110, 1630–1638. [Google Scholar] [CrossRef] [Green Version]
- Zuo, T.; Li, X.M.; Chang, Y.G.; Duan, G.F.; Yu, L.; Zheng, R.; Xue, C.H.; Tang, Q.J. Dietary fucoidan of Acaudina molpadioides and its enzymatically degraded fragments could prevent intestinal mucositis induced by chemotherapy in mice. Food Funct. 2015, 6, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.Z.; O’Toole, P.W. Impact of diet on the human intestinal microbiota. Curr. Opin. Food Sci. 2015, 2, 71–77. [Google Scholar] [CrossRef]
- Blaak, E.E.; Canfora, E.E.; Theis, S.; Frost, G.; Groen, A.K.; Mithieux, G.; Nauta, A.; Scott, K.; Stahl, B.; van Harsselaar, J.; et al. Short chain fatty acids in human gut and metabolic health. Benef. Microbes 2020, 11, 411–455. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Ma, L.; Chen, Q.Z.; Zhang, P.Y.; Chen, C.; Jia, L.L.; Li, H.J. Fucoidan alleviates dyslipidemia and modulates gut microbiota in high-fat diet-induced mice. J. Funct. Foods 2018, 48, 220–227. [Google Scholar] [CrossRef]
- Cheng, Y.; Sibusiso, L.; Hou, L.F.; Jiang, H.J.; Chen, P.C.; Zhang, X.; Wu, M.J.; Tong, H.B. Sargassum fusiforme fucoidan modifies the gut microbiota during alleviation of streptozotocin-induced hyperglycemia in mice. Int. J. Biol. Macromol. 2019, 131, 1162–1170. [Google Scholar] [CrossRef]
- He, N.N.; Yang, Y.; Wang, H.Y.; Liu, N.A.; Yang, Z.Z.; Li, S.Y. Unsaturated alginate oligosaccharides (UAOS) protects against dextran sulfate sodium-induced colitis associated with regulation of gut microbiota. J. Funct. Foods 2021, 83, 104536. [Google Scholar] [CrossRef]
- Liu, J.; Tan, Y.Z.; Cheng, H.; Zhang, D.D.; Feng, W.W.; Peng, C. Functions of Gut Microbiota Metabolites, Current Status and Future Perspectives. Aging Dis. 2022, 13, 1106–1126. [Google Scholar] [CrossRef]
- Schoeler, M.; Caesar, R. Dietary lipids, gut microbiota and lipid metabolism. Rev. Endocr. Metab. Disord. 2019, 20, 461–472. [Google Scholar] [CrossRef]
- Rooks, M.G.; Garrett, W.S. Gut microbiota, metabolites and host immunity. Nat. Rev. Immunol. 2016, 16, 341–352. [Google Scholar] [CrossRef]
- Martin-Gallausiaux, C.; Marinelli, L.; Blottiere, H.M.; Larraufie, P.; Lapaque, N. SCFA: Mechanisms and functional importance in the gut. Proc. Nutr. Soc. 2021, 80, 37–49. [Google Scholar] [CrossRef]
- Phull, A.R.; Kim, S.J. Fucoidan as bio-functional molecule: Insights into the anti-inflammatory potential and associated molecular mechanisms. J. Funct. Foods 2017, 38, 415–426. [Google Scholar] [CrossRef]
- Sanjeewa, K.K.A.; Fernando, I.P.S.; Kim, E.A.; Ahn, G.; Jee, Y.; Jeon, Y.J. Anti-inflammatory activity of a sulfated polysaccharide isolated from an enzymatic digest of brown seaweed Sargassum horneri in RAW 264.7 cells. Nutr. Res. Pract. 2017, 11, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Liu, H.J.; Zhang, R.F.; Dong, L.H.; Liu, L.; Ma, Y.X.; Jia, X.C.; Wang, G.J.; Zhang, M.W. Physicochemical properties and prebiotic activities of polysaccharides from longan pulp based on different extraction techniques. Carbohydr. Polym. 2019, 206, 344–351. [Google Scholar] [CrossRef] [PubMed]






| Genus | Species | Distribution | References |
|---|---|---|---|
| Ecklonia | Ecklonia radiata, Ecklonia cava | Distributed in Australia, Korea, China and other countries, in China mainly in Liaodong, Shandong, Zhejiang, Fujian Province. | [19,20,21] |
| Sargassum | Sargassum fusiforme, Sargassum plagiophyllum, Sargassum thunbergii | Asian countries are more widely distributed, widely distributed in Fujian Province, Zhejiang Province, China, Korea, Japan. | [22,23,24,25] |
| Laminaria | Laminaria japonica | It is mainly distributed in the northwestern Pacific Ocean. Widely distributed in Japan, Russia, China and other countries. | [26,27] |
| Ascophyllum | Ascophyllum nodosum | Distributed in the coastal waters of the North Atlantic Ocean, such as Canada, Norway, Ireland, the United Kingdom, France and other countries. | [28,29] |
| Fucus | Fucus vesiculosus | Mostly found in tropical and subtropical seas. | [30,31] |
| Undaria | Undaria pinnatifida | Mainly in the northwest coast of the North Pacific Ocean, native to China, Japan and the Korean Peninsula. | [32,33] |
| Macrocystis | Macrocystis pyrifera | Mainly distributed along the eastern Pacific coast. | [34,35] |
| Source | Type | Inducer | Models | Function | References |
|---|---|---|---|---|---|
| Cladosiphon okamuranus Tokida | Fucoidan | H2O2 | Caco-2 cells | Fucoidan remarkably reduced H2O2-induced paracellular permeability. Up-regulation of endogenous expression of claudin-1, claudin-2, and occludin in Caco-2 cells. | [15] |
| Acaudina molpadioides | Fucoidan | Cyclophosphamide (CPA) | Mice | Fucoidan intervention alleviates inflammation, increases tight junction protein expression, and increases the abundance of Coprococcus, Rikenella, and Butyricicoccus. | [54] |
| Laminaria japonica | Fucoidan | cefoperazone | Mice | Inhibiting the production of pro-inflammatory cytokines, restored the richness and diversity of intestinal microbiota, and improved the structural damage of intestinal mucosa. | [53] |
| Cladosiphon okamuranus | Fucoidan | - | Zebrafish | Down-regulation of the relative expression of the pro-inflammatory gene IL-1β. | [71] |
| Ascophyllum nodosum | Fucoidan | ciprofloxacin-metronidazole | Mice | Increased the abundance of Ruminococcaceae, and Akkermansia, and decreased the abundance of Proteus and Enterococcus; inhibited the overproduction of TNF-α, IL-1β, and IL-6, and promoted the expression of IL-10. | [28] |
| Sargassum fusiforme | Sulfated polysaccharide | - | - | In vitro fermentation increased the abundance of Faecalibacterium, Phascolarctobacterium, Bifidobacterium, and Lactobacillus. | [72] |
| Laminaria | Alginate | CPA | Mice | Up-regulation of tight junction protein expression reduced intestinal mucosal damage, decreased serum D-lactate and lipopolysaccharide concentrations, and downregulated toll-like receptor 4 (TLR4) and mitogen-activated protein kinase (MAPK) pathway expression to reduce intestinal inflammation. | [73] |
| Fucus vesiculosus | Fucoidan | LPS | RAW 264.7 Cell | Inhibited the secretion of NO, PGE 2 and TNF-α, IL-1β and reduced the production of intracellular reactive oxygen species. | [74] |
| Ascophyllum nodosum | Alginate | - | - | Promoted the growth of Bifidobacteria and Lactobacilli, increased levels of acetate and propionate. | [75] |
| Laminaria japonica | Alginate | - | - | Increased the abundance of Bacteroides. | [76] |
| Eisenia bicyclis | Laminaran | - | - | Inhibited ammonia, phenol, and indole production by human fecal microbiota and reduced indole levels in the cecum. | [77] |
| Ecklonia radiata | Polysaccharides | - | - | Increase in total bacteria, Bifidobacterium, Lactobacillus and increase in total SCFA, acetic and propionic acids. | [19] |
| Ecklonia cava | Fucoidan | LPS | Zebrafish | Inhibiting ROS and NO production induced by LPS treatment to alleviate inflammation. | [20] |
| Sargassum fusiforme | Fucoidan | Streptozotocin | Mice | Fucoidan significantly reduced fasting blood glucose, improved glucose tolerance, reduced oxidative stress in diabetic mice, and increased the abundance of beneficial intestinal microbes including Bacteroides, Faecalibacterium and Blautia. | [24] |
| Sargassum fusiforme | Fucoidan | High-fat diet | Mice | Fucoidan improves HFD-induced insulin resistance by activating the Nrf2 pathway, remodeling the intestinal microbiota, and reducing intestinal inflammation. | [25] |
| Laminaria japonica | Fucoidan | CPA | Mice | Fucoidan increased spleen and thymus indices, increased serum levels of IL-6, IL-1β, TNF-α, and IgG, and improved immunosuppression in mice. Increased the abundance of Lactobacillaceae and Alistipes, and decreased the abundance of Erysipelotrichia, Turicibacter, Romboutsia, Peptostreptococcaceae, and Faecalibaculum. | [78] |
| Ascophyllum nodosum | Fucoidan | ciprofloxacin-metronidazole | Mice | Dietary fucoidan prevented colon shortening and alleviated colon tissue damage by increasing the abundance of potentially beneficial bacteria (e.g., Ruminococcaceae_UCG_014 and Akkermansia) and decreasing the abundance of harmful bacteria (e.g., Aspergillus and Enterococcus), fucoidan also inhibited the overproduction of TNF-α, IL-1β, and IL-6 and promoted the expression of IL-10. | [28] |
| Fucus vesiculosus | Fucoidan | Sodium palmitate | HepG2 Cells Mice | Fucoidan significantly reduced the phosphorylation level of JNK and increased the phosphorylation of protein kinase B (pAkt). It improved hyperglycemia and serum insulin levels in mice with metabolic syndrome. | [31] |
| Undaria pinnatifida | Fucoidan | High-fat diet | Mice | Fucoidan reduces weight gain, fat accumulation and intestinal permeability in mice with metabolic syndrome. Intestinal Firmicutes and Bacteroidetes in fucoidan-treated high-fat diet mice were restored to normal levels and promoted the production of SCFAS and enhanced the expression level of IL-10. | [32] |
| Sargassum thunbergii | crude polysaccharide | - | - | Increased abundance of intestinal beneficial bacteria such as Bifidobacterium, Roseburia, Parasutterella, and Fusicatenibacter by in vitro fecal fermentation after 24 h of fermentation. | [79] |
| Laminaria japonica | Fucoidan | - | Mice | Increased in the abundance of Ruminococcaceae and Lactobacillu and decreased in the serum levels of lipopolysaccharide-binding protein. | [80] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Y.; Liang, M.; Ouyang, D.; Tong, H.; Wu, M.; Su, L. Research Progress on the Protective Effect of Brown Algae-Derived Polysaccharides on Metabolic Diseases and Intestinal Barrier Injury. Int. J. Mol. Sci. 2022, 23, 10784. https://doi.org/10.3390/ijms231810784
Yang Y, Liang M, Ouyang D, Tong H, Wu M, Su L. Research Progress on the Protective Effect of Brown Algae-Derived Polysaccharides on Metabolic Diseases and Intestinal Barrier Injury. International Journal of Molecular Sciences. 2022; 23(18):10784. https://doi.org/10.3390/ijms231810784
Chicago/Turabian StyleYang, Ying, Meina Liang, Dan Ouyang, Haibin Tong, Mingjiang Wu, and Laijin Su. 2022. "Research Progress on the Protective Effect of Brown Algae-Derived Polysaccharides on Metabolic Diseases and Intestinal Barrier Injury" International Journal of Molecular Sciences 23, no. 18: 10784. https://doi.org/10.3390/ijms231810784






