WNK1–OSR1 Signaling Regulates Angiogenesis-Mediated Metastasis towards Developing a Combinatorial Anti-Cancer Strategy
Abstract
:1. Introduction
2. Results
2.1. The Expression of wnk1a–osr1a/osr1b Is Upregulated during Embryonic Angiogenesis
2.2. The Expression of wnk1a and osr1b Is Upregulated in Endothelial Cells, and WNK1 and OSR1 Are Upregulated in Hepatoma Cells during Tumor-Induced Angiogenesis
2.3. [tert] and [tert x p53-/-] Transgenic Fish Develops HCC at 15 and 30 dpf
2.4. Expression of wnk1a, osr1b and stk39 Is Increased, and ppp2r1ba and ppp2r1bb Are Decreased in HCC Formation in the [tert] and [tert x p53−/−] Transgenic Fish
2.5. Expression of wnk1a and osr1b Is Increased in HCC Formation in the [HBx,src,p53−/−,RPIA] Transgenic Fish
2.6. Endothelial Cells Promote Hepatoma Cell Migration via WNK1–OSR1 axis
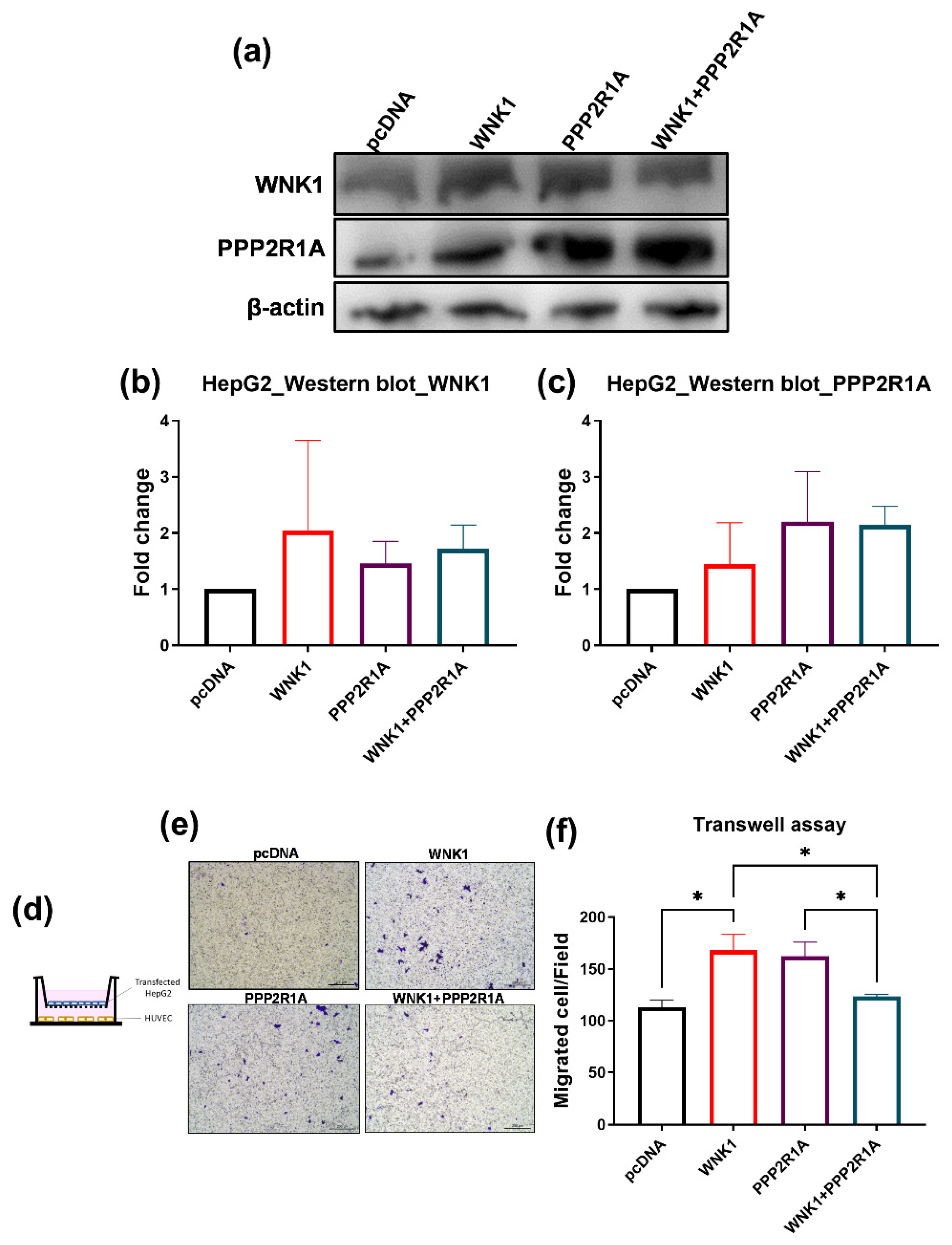
2.7. PPP2R1A Acts as a Repressor of WNK1 in Stimulating Hepatoma Cell Migration
2.8. Combinational Therapy Targeting WNK1–OSR1 with Oligo-Fucoidan Attenuates HCC Formation in [HBx,src,p53−/−,RPIA] Transgenic Fish
2.9. Oligo-Fucoidan Decreases the Elevated Cell Senescence-Associated β-Galactosidase Activity in Tert Transgenic Fish Treated with WNK1–OSR1 Inhibitors
3. Discussion
4. Materials and Methods
4.1. Zebrafish Husbandry
4.2. Embryo Collection
4.3. Cell Culture
4.4. Xenotransplantation
4.5. Embryonic Angiogenesis
4.6. RNA Extraction
4.7. Reverse-Transcription and Quantitative Polymerase Chain Reaction (QPCR)
4.8. Transfection
4.9. Transwell Migration Assay
4.10. Western Blot Analysis
4.11. Oral Gavage
4.12. Hematoxylin and Eosin Stain
4.13. Detection of Senescence-Associated β-galactosidase (SA-β-gal) Accumulation
4.14. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kocarnik, J.M.; Compton, K.; Dean, F.E.; Fu, W.; Gaw, B.L.; Harvey, J.D.; Henrikson, H.J.; Lu, D.; Pennini, A.; Xu, R.; et al. Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life Years for 29 Cancer Groups From 2010 to 2019: A Systematic Analysis for the Global Burden of Disease Study 2019. JAMA Oncol. 2022, 8, 420–444. [Google Scholar] [PubMed]
- Amini, M.; Looha, M.A.; Zarean, E.; Pourhoseingholi, M.A. Global pattern of trends in incidence, mortality, and mortality-to-incidence ratio rates related to liver cancer, 1990–2019: A longitudinal analysis based on the global burden of disease study. BMC Public Health 2022, 22, 604. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, W.; Jiang, L.; Chen, Y. Recent advances in systemic therapy for hepatocellular carcinoma. Biomark Res. 2022, 10, 3. [Google Scholar] [CrossRef]
- Ishii, Y.; Sakamoto, T.; Ito, R.; Yanaga, K. Anti-angiogenic therapy on hepatocellular carcinoma development and progression. J. Surg. Res. 2010, 158, 69–76. [Google Scholar] [CrossRef]
- El-Kenawi, A.E.; El-Remessy, A.B. Angiogenesis inhibitors in cancer therapy: Mechanistic perspective on classification and treatment rationales. Br. J. Pharmacol. 2013, 170, 712–729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, B.; English, J.M.; Wilsbacher, J.L.; Stippec, S.; Goldsmith, E.J.; Cobb, M.H. WNK1, a novel mammalian serine/threonine protein kinase lacking the catalytic lysine in subdomain II. J. Biol. Chem. 2000, 275, 16795–16801. [Google Scholar] [CrossRef] [Green Version]
- Veríssimo, F.; Jordan, P. WNK kinases, a novel protein kinase subfamily in multi-cellular organisms. Oncogene 2001, 20, 5562–5569. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Min, X.; Lee, B.H.; Cobb, M.H.; Goldsmith, E.J. Crystal structure of the kinase domain of WNK1, a kinase that causes a hereditary form of hypertension. Structure 2004, 12, 1303–1311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shekarabi, M.; Zhang, J.; Khanna, A.R.; Ellison, D.H.; Delpire, E.; Kahle, K.T. WNK Kinase Signaling in Ion Homeostasis and Human Disease. Cell Metab. 2017, 25, 285–299. [Google Scholar] [CrossRef] [Green Version]
- Conti, A.; Luchini, A.; Benassi, M.S.; Magagnoli, G.; Pierini, M.; Piccinni-Leopardi, M.; Quattrini, I.; Pollino, S.; Picci, P.; Liotta, L.A.; et al. Circulating Candidate Biomarkers in Giant Cell Tumors of Bone. Proteom. Clin. Appl. 2018, 12, e1800041. [Google Scholar] [CrossRef]
- Ho, Y.J.; Chang, J.; Yeh, K.T.; Gong, Z.; Lin, Y.M.; Lu, J.W. Prognostic and Clinical Implications of WNK Lysine Deficient Protein Kinase 1 Expression in Patients With Hepatocellular Carcinoma. In Vivo 2020, 34, 2631–2640. [Google Scholar] [CrossRef] [PubMed]
- Jaykumar, A.B.; Jung, J.U.; Parida, P.K.; Dang, T.T.; Wichaidit, C.; Kannangara, A.R.; Earnest, S.; Goldsmith, E.J.; Pearson, G.W.; Malladi, S.; et al. WNK1 Enhances Migration and Invasion in Breast Cancer Models. Mol. Cancer Ther. 2021, 20, 1800–1808. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, L.; Qin, J.; Wu, J.; Dai, X.; Xu, J. OSR1 phosphorylates the Smad2/3 linker region and induces TGF-β1 autocrine to promote EMT and metastasis in breast cancer. Oncogene 2021, 40, 68–84. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, T.; Li, N.; Wang, T.; Pu, Y.; Lin, R. Identification of a novel WNK1-ROS1 fusion in a lung adenocarcinoma sensitive to crizotinib. Lung Cancer 2019, 129, 92–94. [Google Scholar] [CrossRef] [PubMed]
- Sie, Z.L.; Li, R.Y.; Sampurna, B.P.; Hsu, P.J.; Liu, S.C.; Wang, H.D.; Huang, C.L.; Yuh, C.H. WNK1 Kinase Stimulates Angiogenesis to Promote Tumor Growth and Metastasis. Cancers 2020, 12, 575. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Su, Y.; Yin, C. Long non-coding RNA (lncRNA) five prime to Xist (FTX) promotes retinoblastoma progression by regulating the microRNA-320a/with-no-lysine kinases 1 (WNK1) axis. Bioengineered 2021, 12, 11622–11633. [Google Scholar] [CrossRef]
- Zhu, W.; Begum, G.; Pointer, K.; Clark, P.A.; Yang, S.S.; Lin, S.H.; Kahle, K.T.; Kuo, J.S.; Sun, D. WNK1-OSR1 kinase-mediated phospho-activation of Na+-K+-2Cl- cotransporter facilitates glioma migration. Mol. Cancer 2014, 13, 31. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Sun, W.; Chen, N.; Xu, C.; Wang, X.; Dong, K.; Zhang, B.; Zhang, J.; Hao, N.; Sun, A.; et al. Discovery of NKCC1 as a potential therapeutic target to inhibit hepatocellular carcinoma cell growth and metastasis. Oncotarget 2017, 8, 66328–66342. [Google Scholar] [CrossRef] [Green Version]
- Qiu, Z.; Dong, B.; Guo, W.; Piotr, R.; Longmore, G.; Yang, X.; Yu, Z.; Deng, J.; Evers, B.M.; Wu, Y. STK39 promotes breast cancer invasion and metastasis by increasing SNAI1 activity upon phosphorylation. Theranostics 2021, 11, 7658–7670. [Google Scholar] [CrossRef]
- Delaloy, C.; Hadchouel, J.; Imbert-Teboul, M.; Clemessy, M.; Houot, A.M.; Jeunemaitre, X. Cardiovascular expression of the mouse WNK1 gene during development and adulthood revealed by a BAC reporter assay. Am. J. Pathol. 2006, 169, 105–118. [Google Scholar] [CrossRef]
- Xie, J.; Wu, T.; Xu, K.; Huang, I.K.; Cleaver, O.; Huang, C.L. Endothelial-specific expression of WNK1 kinase is essential for angiogenesis and heart development in mice. Am. J. Pathol. 2009, 175, 1315–1327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xie, J.; Yoon, J.; Yang, S.S.; Lin, S.H.; Huang, C.L. WNK1 protein kinase regulates embryonic cardiovascular development through the OSR1 signaling cascade. J. Biol. Chem. 2013, 288, 8566–8574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chi, R.A.; Wang, T.; Huang, C.L.; Wu, S.P.; Young, S.L.; Lydon, J.P.; DeMayo, F.J. WNK1 regulates uterine homeostasis and its ability to support pregnancy. JCI Insight 2020, 5, e141832. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.G.; Tsai, S.M.; Tu, H.C.; Chen, W.C.; Kou, F.J.; Lu, J.W.; Wang, H.D.; Huang, C.L.; Yuh, C.H. Zebrafish WNK lysine deficient protein kinase 1 (wnk1) affects angiogenesis associated with VEGF signaling. PLoS ONE 2014, 9, e106129. [Google Scholar]
- Schwab, A.; Fabian, A.; Hanley, P.J.; Stock, C. Role of ion channels and transporters in cell migration. Physiol. Rev. 2012, 92, 1865–1913. [Google Scholar] [CrossRef]
- Hou, C.Y.; Ma, C.Y.; Yuh, C.H. WNK1 kinase signaling in metastasis and angiogenesis. Cell Signal 2022, 96, 110371. [Google Scholar] [CrossRef]
- Haesen, D.; Abbasi Asbagh, L.; Derua, R.; Hubert, A.; Schrauwen, S.; Hoorne, Y.; Amant, F.; Waelkens, E.; Sablina, A.; Janssens, V. Recurrent PPP2R1A Mutations in Uterine Cancer Act through a Dominant-Negative Mechanism to Promote Malignant Cell Growth. Cancer Res. 2016, 76, 5719–5731. [Google Scholar] [CrossRef] [Green Version]
- Yamada, K.; Park, H.M.; Rigel, D.F.; DiPetrillo, K.; Whalen, E.J.; Anisowicz, A.; Beil, M.; Berstler, J.; Brocklehurst, C.E.; Burdick, D.A.; et al. Small-molecule WNK inhibition regulates cardiovascular and renal function. Nat. Chem. Biol. 2016, 12, 896–898. [Google Scholar] [CrossRef]
- AlAmri, M.A.; Kadri, H.; Alderwick, L.J.; Simpkins, N.S.; Mehellou, Y. Rafoxanide and Closantel Inhibit SPAK and OSR1 Kinases by Binding to a Highly Conserved Allosteric Site on Their C-terminal Domains. ChemMedChem 2017, 12, 639–645. [Google Scholar] [CrossRef]
- Mayes-Hopfinger, L.; Enache, A.; Xie, J.; Huang, C.L.; Kochl, R.; Tybulewicz, V.L.J.; Fernandes-Alnemri, T.; Alnemri, E.S. Chloride sensing by WNK1 regulates NLRP3 inflammasome activation and pyroptosis. Nat. Commun. 2021, 12, 4546. [Google Scholar] [CrossRef]
- Lin, Y.; Qi, X.; Liu, H.; Xue, K.; Xu, S.; Tian, Z. The anti-cancer effects of fucoidan: A review of both in vivo and in vitro investigations. Cancer Cell Int. 2020, 20, 154. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.Y.; Yang, W.Y.; Cheng, C.C.; Lin, K.H.; Sampurna, B.P.; Chan, S.M.; Yuh, C.H. Low molecular weight fucoidan inhibits hepatocarcinogenesis and nonalcoholic fatty liver disease in zebrafish via ASGR/STAT3/HNF4A signaling. Clin. Transl. Med. 2020, 10, e252. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gorski, J.W.; Ueland, F.R.; Kolesar, J.M. CCNE1 Amplification as a Predictive Biomarker of Chemotherapy Resistance in Epithelial Ovarian Cancer. Diagnostics 2020, 10, 279. [Google Scholar] [CrossRef]
- Zhao, Z.M.; Yost, S.E.; Hutchinson, K.E.; Li, S.M.; Yuan, Y.C.; Noorbakhsh, J.; Liu, Z.; Warden, C.; Johnson, R.M.; Wu, X.; et al. CCNE1 amplification is associated with poor prognosis in patients with triple negative breast cancer. BMC Cancer 2019, 19, 96. [Google Scholar] [CrossRef]
- Aziz, K.; Limzerwala, J.F.; Sturmlechner, I.; Hurley, E.; Zhang, C.; Jeganathan, K.B.; Nelson, G.; Bronk, S.; Fierro Velasco, R.O.; van Deursen, E.J.; et al. Ccne1 Overexpression Causes Chromosome Instability in Liver Cells and Liver Tumor Development in Mice. Gastroenterology 2019, 157, 210–226.e12. [Google Scholar] [CrossRef]
- Diril, M.K.; Ratnacaram, C.K.; Padmakumar, V.C.; Du, T.; Wasser, M.; Coppola, V.; Tessarollo, L.; Kaldis, P. Cyclin-dependent kinase 1 (Cdk1) is essential for cell division and suppression of DNA re-replication but not for liver regeneration. Proc. Natl. Acad. Sci. USA 2012, 109, 3826–3831. [Google Scholar] [CrossRef] [Green Version]
- Sonntag, R.; Giebeler, N.; Nevzorova, Y.A.; Bangen, J.M.; Fahrenkamp, D.; Lambertz, D.; Haas, U.; Hu, W.; Gassler, N.; Cubero, F.J.; et al. Cyclin E1 and cyclin-dependent kinase 2 are critical for initiation, but not for progression of hepatocellular carcinoma. Proc. Natl. Acad. Sci. USA 2018, 115, 9282–9287. [Google Scholar] [CrossRef] [Green Version]
- Jin, X.; Beck, S.; Sohn, Y.W.; Kim, J.K.; Kim, S.H.; Yin, J.; Pian, X.; Kim, S.C.; Choi, Y.J.; Kim, H. Human telomerase catalytic subunit (hTERT) suppresses p53-mediated anti-apoptotic response via induction of basic fibroblast growth factor. Exp. Mol. Med. 2010, 42, 574–582. [Google Scholar] [CrossRef] [Green Version]
- Baba, A.I.; Câtoi, C. Tumor Cell Morphology; The Publishing House of the Romanian Academy: Bucharest, Romania, 2007; Chapter 3. [Google Scholar]
- Toda-Ishii, M.; Akaike, K.; Suehara, Y.; Mukaihara, K.; Kubota, D.; Kohsaka, S.; Okubo, T.; Mitani, K.; Mogushi, K.; Takagi, T.; et al. Clinicopathological effects of protein phosphatase 2, regulatory subunit A, alpha mutations in gastrointestinal stromal tumors. Mod. Pathol. 2016, 29, 1424–1432. [Google Scholar] [CrossRef] [Green Version]
- Cheng, C.C.; Yang, W.Y.; Hsiao, M.C.; Lin, K.H.; Lee, H.W.; Yuh, C.H. Transcriptomically Revealed Oligo-Fucoidan Enhances the Immune System and Protects Hepatocytes via the ASGPR/STAT3/HNF4A Axis. Biomolecules 2020, 10, 898. [Google Scholar] [CrossRef]
- Rui, X.; Pan, H.F.; Shao, S.L.; Xu, X.M. Anti-tumor and anti-angiogenic effects of Fucoidan on prostate cancer: Possible JAK-STAT3 pathway. BMC Complement. Altern. Med. 2017, 17, 378. [Google Scholar] [CrossRef] [Green Version]
- Coppé, J.-P.; Patil, C.K.; Rodier, F.; Sun, Y.; Muñoz, D.P.; Goldstein, J.; Nelson, P.S.; Desprez, P.-Y.; Campisi, J. Senescence-Associated Secretory Phenotypes Reveal Cell-Nonautonomous Functions of Oncogenic RAS and the p53 Tumor Suppressor. PLoS Biol. 2008, 6, e301. [Google Scholar] [CrossRef] [PubMed]
- Veikkola, T.; Alitalo, K. VEGFs, receptors and angiogenesis. Semin. Cancer Biol. 1999, 9, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Lohela, M.; Bry, M.; Tammela, T.; Alitalo, K. VEGFs and receptors involved in angiogenesis versus lymphangiogenesis. Curr. Opin. Cell Biol. 2009, 21, 154–165. [Google Scholar] [CrossRef] [PubMed]
- McMahon, G. VEGF receptor signaling in tumor angiogenesis. Oncologist 2000, 5 (Suppl. S1), 3–10. [Google Scholar] [CrossRef]
- Rosen, L.S. Clinical experience with angiogenesis signaling inhibitors: Focus on vascular endothelial growth factor (VEGF) blockers. Cancer Control 2002, 9 (Suppl. S2), 36–44. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, F.; Onwumeh-Okwundu, J.; Yukselen, Z.; Endaya Coronel, M.K.; Zaidi, M.; Guntipalli, P.; Garimella, V.; Gudapati, S.; Mezidor, M.D.; Andrews, K.; et al. Atezolizumab plus bevacizumab versus sorafenib or atezolizumab alone for unresectable hepatocellular carcinoma: A systematic review. World J. Gastrointest Oncol. 2021, 13, 1813–1832. [Google Scholar] [CrossRef] [PubMed]
- Jászai, J.; Schmidt, M.H.H. Trends and Challenges in Tumor Anti-Angiogenic Therapies. Cells 2019, 8, 1102. [Google Scholar] [CrossRef] [Green Version]
- Everaerts, W.; Nilius, B.; Owsianik, G. The vanilloid transient receptor potential channel TRPV4: From structure to disease. Prog. Biophys. Mol. Biol. 2010, 103, 2–17. [Google Scholar] [CrossRef]
- Thodeti, C.K.; Matthews, B.; Ravi, A.; Mammoto, A.; Ghosh, K.; Bracha, A.L.; Ingber, D.E. TRPV4 channels mediate cyclic strain-induced endothelial cell reorientation through integrin-to-integrin signaling. Circ. Res. 2009, 104, 1123–1130. [Google Scholar] [CrossRef]
- Thoppil, R.J.; Cappelli, H.C.; Adapala, R.K.; Kanugula, A.K.; Paruchuri, S.; Thodeti, C.K. TRPV4 channels regulate tumor angiogenesis via modulation of Rho/Rho kinase pathway. Oncotarget 2016, 7, 25849–25861. [Google Scholar] [CrossRef] [Green Version]
- Tomilin, V.N.; Pyrshev, K.; Khayyat, N.H.; Zaika, O.; Pochynyuk, O. With-No-Lysine Kinase 1 (WNK1) Augments TRPV4 Function in the Aldosterone-Sensitive Distal Nephron. Cells 2021, 10, 1482. [Google Scholar] [CrossRef]
- Spaderna, S.; Schmalhofer, O.; Hlubek, F.; Jung, A.; Kirchner, T.; Brabletz, T. Epithelial-mesenchymal and mesenchymal-epithelial transitions during cancer progression. Verh. Der Dtsch. Ges. Fur Pathol. 2007, 91, 21–28. [Google Scholar]
- Savagner, P.; Yamada, K.M.; Thiery, J.P. The zinc-finger protein slug causes desmosome dissociation, an initial and necessary step for growth factor-induced epithelial-mesenchymal transition. J. Cell Biol. 1997, 137, 1403–1419. [Google Scholar] [CrossRef]
- Dbouk, H.A.; Weil, L.M.; Perera, G.K.; Dellinger, M.T.; Pearson, G.; Brekken, R.A.; Cobb, M.H. Actions of the protein kinase WNK1 on endothelial cells are differentially mediated by its substrate kinases OSR1 and SPAK. Proc. Natl. Acad. Sci. USA 2014, 111, 15999–16004. [Google Scholar] [CrossRef] [Green Version]
- Rawal, P.; Siddiqui, H.; Hassan, M.; Choudhary, M.C.; Tripathi, D.M.; Nain, V.; Trehanpati, N.; Kaur, S. Endothelial Cell-Derived TGF-β Promotes Epithelial-Mesenchymal Transition via CD133 in HBx-Infected Hepatoma Cells. Front. Oncol. 2019, 9, 308. [Google Scholar] [CrossRef]
- Xu, J.; Lamouille, S.; Derynck, R. TGF-beta-induced epithelial to mesenchymal transition. Cell Res. 2009, 19, 156–172. [Google Scholar] [CrossRef]
- Yukawa, H.; Suzuki, K.; Aoki, K.; Arimoto, T.; Yasui, T.; Kaji, N.; Ishikawa, T.; Ochiya, T.; Baba, Y. Imaging of angiogenesis of human umbilical vein endothelial cells by uptake of exosomes secreted from hepatocellular carcinoma cells. Sci. Rep. 2018, 8, 6765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lam, E.W.; Brosens, J.J.; Gomes, A.R.; Koo, C.Y. Forkhead box proteins: Tuning forks for transcriptional harmony. Nat. Rev. Cancer 2013, 13, 482–495. [Google Scholar] [CrossRef]
- Nagendra, D.C.; Burke, J., 3rd; Maxwell, G.L.; Risinger, J.I. PPP2R1A mutations are common in the serous type of endometrial cancer. Mol. Carcinog. 2012, 51, 826–831. [Google Scholar] [CrossRef]
- Shih Ie, M.; Panuganti, P.K.; Kuo, K.T.; Mao, T.L.; Kuhn, E.; Jones, S.; Velculescu, V.E.; Kurman, R.J.; Wang, T.L. Somatic mutations of PPP2R1A in ovarian and uterine carcinomas. Am. J. Pathol. 2011, 178, 1442–1447. [Google Scholar] [CrossRef]
- Haraoka, Y.; Akieda, Y.; Nagai, Y.; Mogi, C.; Ishitani, T. Zebrafish imaging reveals TP53 mutation switching oncogene-induced senescence from suppressor to driver in primary tumorigenesis. Nat. Commun. 2022, 13, 1417. [Google Scholar] [CrossRef] [PubMed]











| Fish Lines | Promoter | Expressed Gene | Phenotype |
|---|---|---|---|
| AB wild-type | - | - | control |
| Tg(fli1:EGFP) | fli1 | EGFP | Green fluorescence in the vessels. |
| Tg(fabp10a:HBx,src,RPIA;myl7:EGFP)xtp53zdf1/zdf | fabp10a (liver-specific promoter) | HBx, src, RPIA tp53zdf1/zdf | Green fluorescence in heart. Develops HCC at 5 months of age. |
| Tg(fli1:wnk1a;myl7:EGFP) | fli1 | wnk1a | Green fluorescence in heart. Overexpressed wnk1a in the vessels. |
| Tg(fli1:EGFP)xTg(fli1:CreERT2;myl7:EGFP)xTg(loxP-wnk1a-DsRed-loxP) | fli1 | wnk1a CreERT2 loxP-wnk1a-DsRed-loxP | Vessels-specific knockout the wnk1a by adding the RU486 activates CreERT2. |
| Tg(fli1:EGFP)xTg(fli1:wnk1a) | fli1 | wnk1a, EGFP | |
| Tg(fabp10a:tert;myl7:EGFP)xtp53zdf1/zdf | fabp10a (liver-specific promoter) | tert tp53zdf1/zdf | Green fluorescence in heart. Develops HCC at 15, 30 days of age. |
| Tg(fabp10a:tert;myl7:EGFP) | fabp10a (liver-specific promoter) | tert | Green fluorescence in heart. Develops HCC at 15 days of age. |
| Gene Name | Primer Name | Sequencing |
|---|---|---|
| STK39 | h-Q-stk39_F | 5′-CATGAGTCAGTGCAGCCATC-3′ |
| h-Q-stk39_R | 5′-TGTGTTCTCCTCGGTTGACA-3′ | |
| OXSR1 | h-Q-oxsr1_F | 5′-AAAGACCTTTGTTGGCACCC-3′ |
| h-Q-oxsr1_R | 5′-AGGATCGTTCTGCAGTGTCA-3′ | |
| PPP2R1B | h-Q-ppp2r1b_F | 5′-GTCCTGACTTTGCCCACTGT-3′ |
| h-Q-ppp2r1b_R | 5′-GAACCAATCCCCACTTGCTA-3′ | |
| PPP2R1A | h-Q-PPP2R1A_F | 5′-TGACTGTCGGGAGAATGTGA-3′ |
| h-Q-PPP2R1A_R | 5′-GGGAGAGAGACCCATGATGA-3′ | |
| stk39 | z-Q-stk39_F | 5′-TGGACACCTGCACAAAACTG-3′ |
| z-Q-stk39_R | 5′-TCGTTTTCTTTGACCCTGCG-3′ | |
| oxsr1a | z-Q-oxsr1a_F | 5′-AGGTGGCCATTAAACGCATC-3′ |
| z-Q-oxsr1a_R | 5′-GCAACTTCATGACCAGCCAA-3′ | |
| oxsr1b | z-Q-oxsr1b_F | 5′-CATCAAACGCATCAATCTGG-3′ |
| z-Q-oxsr1b_R | 5′-CGGTCTTGTGTTCACCCTTT-3′ | |
| ppp2r1ba | z-Q-ppp2r1baF | 5′-TGGCAACAGTTGAAGAGACG-3′ |
| z-Q-ppp2r1baR | 5′-AGAGCCCACAAGCAGAGGTA-3′ | |
| ppp2r1bb | z-Q-ppp2r1bbF | 5′-AGACTTGGAGGCTCTGGTCA-3′ |
| z-Q-ppp2r1bbR | 5′-GGTCTCCCTGCTGTCTTCAG-3′ | |
| ccne1 | z-ccne1_F | 5′-CATGCCAAGCAAGAAAGTGCTA-3′ |
| z-ccne1_R | 5′-GTGCTGGGAACACCTTCAGT-3′ | |
| cdk1 | z-cdk1_F | 5′-CTCTGGGGACCCCTAACAAT-3′ |
| z-cdk1_R | 5′-CGGATGTGTCATTGCTTGTC-3′ | |
| cdk2 | z-cdk2_F | 5′-GGGCACTTTTGACATGGAGT-3′ |
| z-cdk2_R | 5′-GTGCTGGGAACACCTTCAGT-3′ | |
| actin | z-actin_F | 5′-CTCCATCATGAAGTGCGACGT-3′ |
| z-actin_R | 5′-CAGACGGAGTATTTGCGCTCA-3′ | |
| il1β | z-Q-il1b_ | 5′-CGCTCCACATCTCGTACTCA-3′ |
| z-Q-il1b_R | 5′-ATACGCGGTGCTGATAAACC-3′ |
| Component | Volume per Reaction (μL) |
|---|---|
| 5× iScript Reaction Mix | 4 |
| iScript Reverse Transcriptase | 1 |
| RNase-free water | variable |
| RNA template(1 μg) | variable |
| Total volume | 20 |
| Component | Volume per Reaction (μL) |
|---|---|
| cDNA (dilute with RNase-free water) | 3.8 |
| Primer (2.5 μΜ of forward and reverse primer) | 1.2 |
| 2X SYBR Green (Catalog #: 4385618, Thermo Scientific, Waltham, MA, USA) | 5 |
| Temperature | Time | Cycle | |
|---|---|---|---|
| Hold stage | 95 °C | 3 min | 1 |
| PCR stage | 95 °C | 1 s | 40 |
| 60 °C | 20 s | ||
| Melt curve stage | 95 °C | 15 s | 1 |
| 60 °C | 1 min | ||
| 95 °C | 15 s |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hou, C.-Y.; Ma, C.-Y.; Lin, Y.-J.; Huang, C.-L.; Wang, H.-D.; Yuh, C.-H. WNK1–OSR1 Signaling Regulates Angiogenesis-Mediated Metastasis towards Developing a Combinatorial Anti-Cancer Strategy. Int. J. Mol. Sci. 2022, 23, 12100. https://doi.org/10.3390/ijms232012100
Hou C-Y, Ma C-Y, Lin Y-J, Huang C-L, Wang H-D, Yuh C-H. WNK1–OSR1 Signaling Regulates Angiogenesis-Mediated Metastasis towards Developing a Combinatorial Anti-Cancer Strategy. International Journal of Molecular Sciences. 2022; 23(20):12100. https://doi.org/10.3390/ijms232012100
Chicago/Turabian StyleHou, Chia-Ying, Chung-Yung Ma, Yu-Ju Lin, Chou-Long Huang, Horng-Dar Wang, and Chiou-Hwa Yuh. 2022. "WNK1–OSR1 Signaling Regulates Angiogenesis-Mediated Metastasis towards Developing a Combinatorial Anti-Cancer Strategy" International Journal of Molecular Sciences 23, no. 20: 12100. https://doi.org/10.3390/ijms232012100





