RhFGF21 Protects Epidermal Cells against UVB-Induced Apoptosis through Activating AMPK-Mediated Autophagy
Abstract
1. Introduction
2. Results
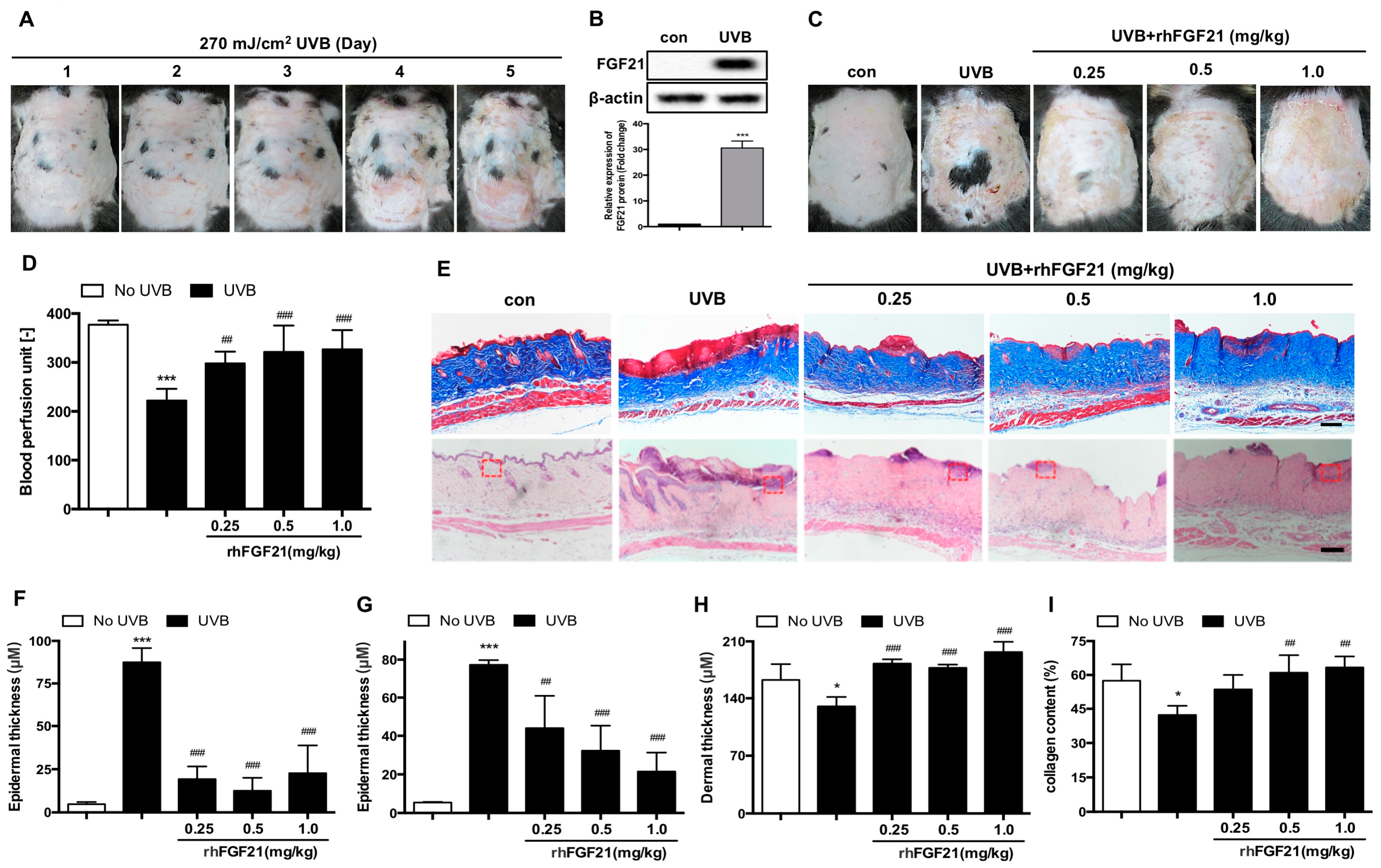
2.1. Treatment with RhFGF21 Protected Mouse Skin Tissue against UVB-Induced Injury
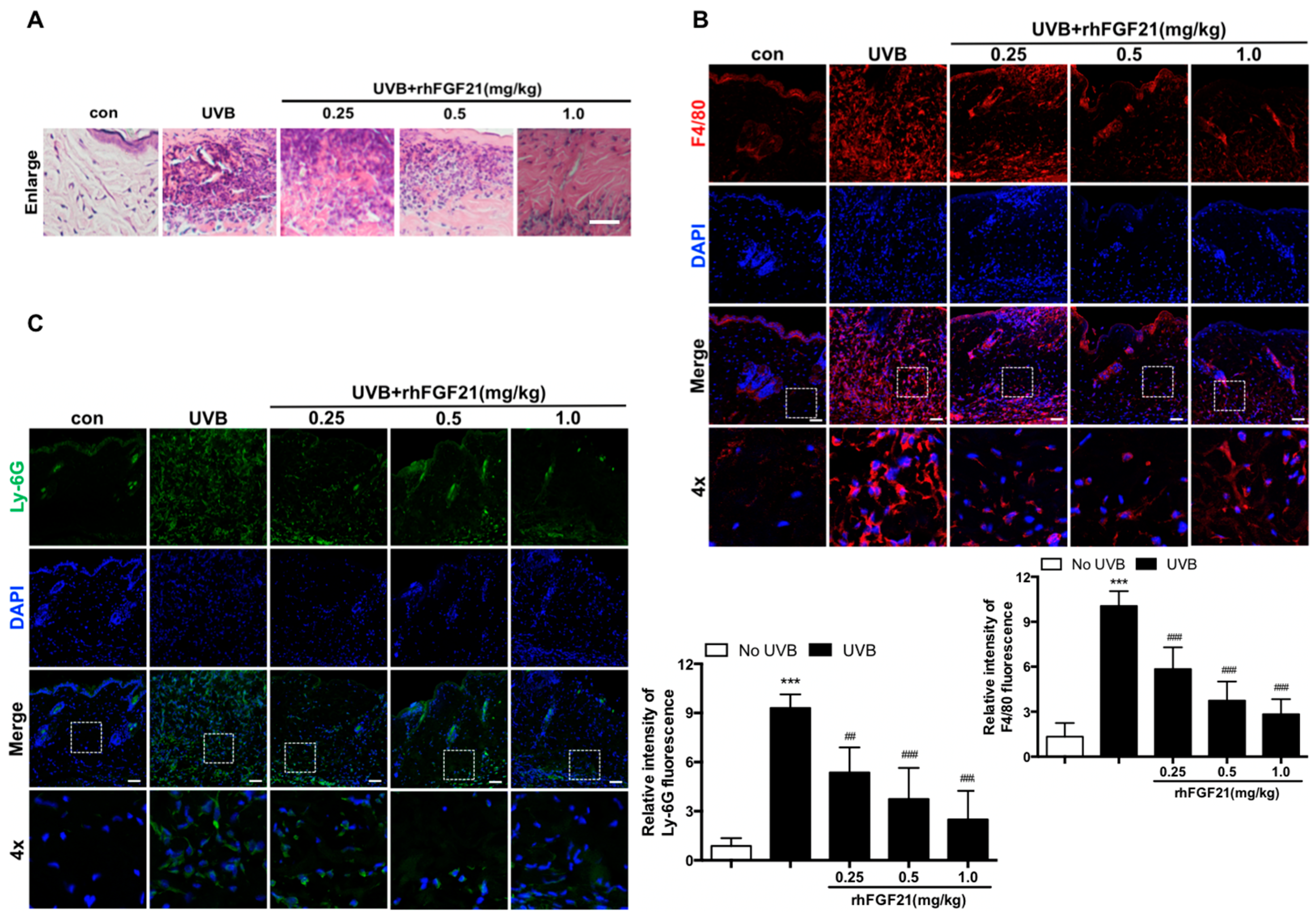
2.2. RhFGF21 Inhibited the Inflammatory Cell Infiltration in UVB-Irradiated Mouse Skin
2.3. RhFGF21 Inhibited the Inflammatory Response and Epidermal Cell Apoptosis in UVB-Irradiated Mouse Skin
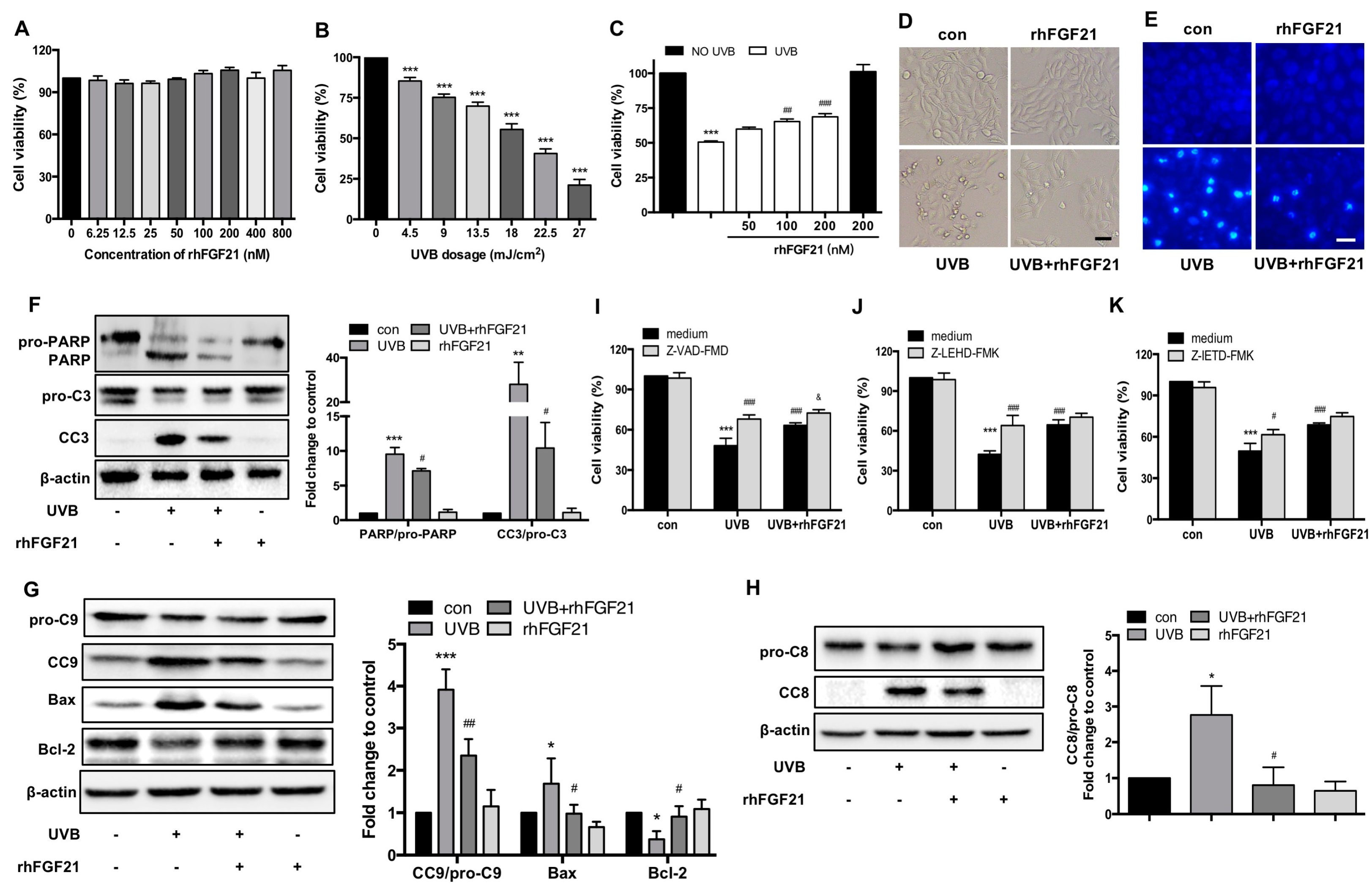
2.4. Treatment with RhFGF21 Protected HaCaT Cells from UVB-Induced Apoptosis
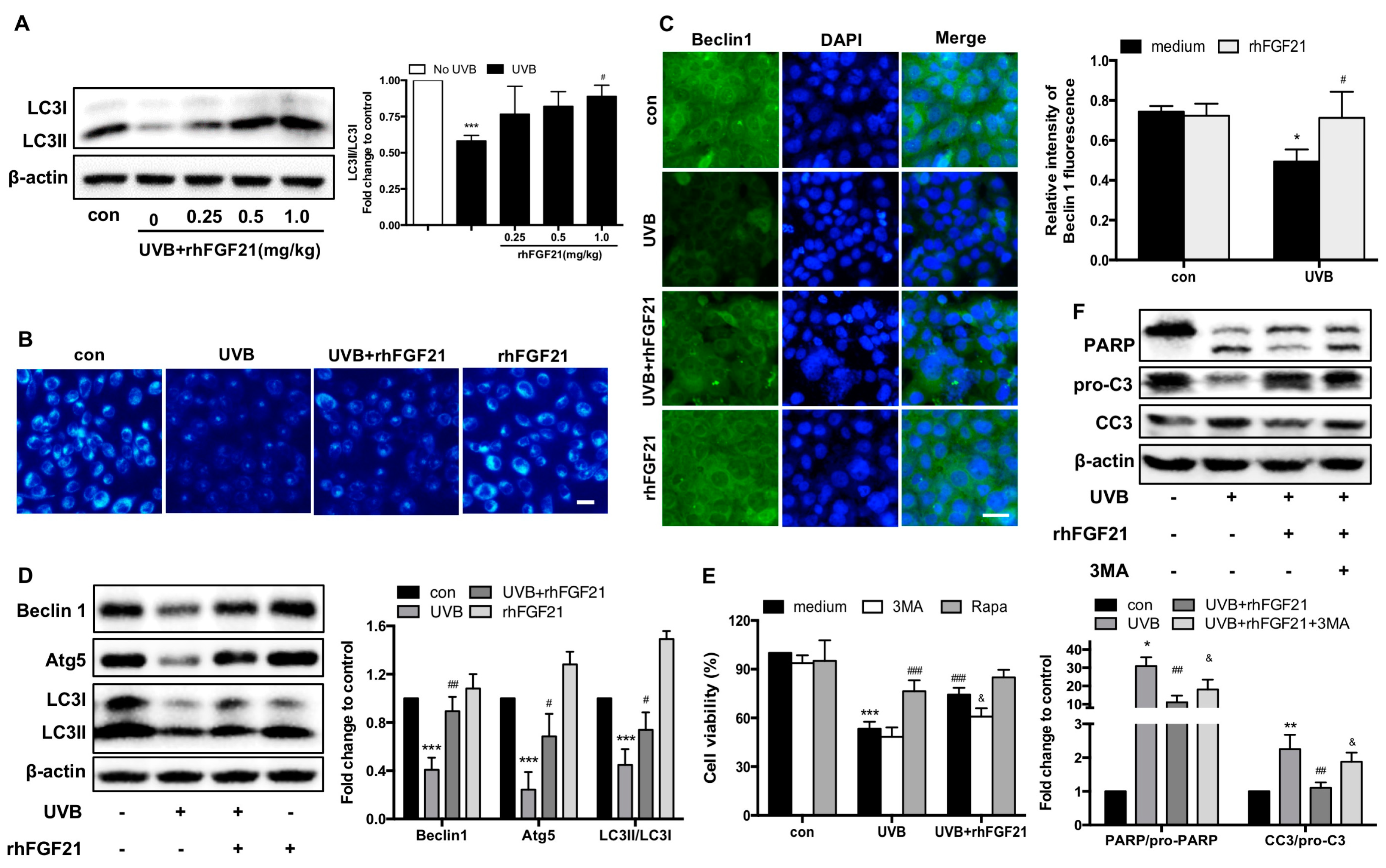
2.5. RhFGF21 Rescued UVB-Irradiated HaCaT Cells by Inducing Autophagy
2.6. RhFGF21 Protected HaCaT Cells from UVB-Induced Apoptosis by Activating AMPK-Mediated Autophagy
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Experimental Protocol In Vivo
4.3. Laser Doppler Flowmetry
4.4. Histopathological Analysis
4.5. TUNEL Assay
4.6. Measurement of IL-1β, TNFα, IL-6 and PGE2 Levels
4.7. The Epidermal Cell Extraction
4.8. Cell Culture
4.9. UVB Exposure In Vitro
4.10. Cell Viability
4.11. MDC Staining
4.12. Hoechst 33258 Staining
4.13. Reverse Transcription Polymerase Chain Reaction (RT-PCR)
4.14. Western Blot Analysis
4.15. Immunofluorescence Staining
4.16. SiRNA Transfection
4.17. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Li, X.Q.; Cai, L.M.; Liu, J.; Ma, Y.L.; Kong, Y.H.; Li, H.; Jiang, M. Liquiritin suppresses UVBinduced skin injury through prevention of inflammation, oxidative stress and apoptosis through the TLR4/MyD88/NFkappaB and MAPK/caspase signaling pathways. Int. J. Mol. Med. 2018, 42, 1445–1459. [Google Scholar] [PubMed]
- Liu, W.; Otkur, W.; Li, L.; Wang, Q.; He, H.; Ye, Y.; Zhang, Y.; Hayashi, T.; Tashiro, S.; Onodera, S.; et al. Autophagy induced by silibinin protects human epidermoid carcinoma A431 cells from UVB-induced apoptosis. J. Photochem. Photobiol. B 2013, 123, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Ivan, A.L.; Campanini, M.Z.; Martinez, R.M.; Ferreira, V.S.; Steffen, V.S.; Vicentini, F.T.; Vilela, F.M.; Martins, F.S.; Zarpelon, A.C.; Cunha, T.M.; et al. Pyrrolidine dithiocarbamate inhibits UVB-induced skin inflammation and oxidative stress in hairless mice and exhibits antioxidant activity in vitro. J. Photochem. Photobiol. B 2014, 138, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.H.; Do, M.H.; Oh, Y.L.; Cho, D.W.; Kim, S.H.; Kim, S.Y. Dietary fermented soybean suppresses UVB-induced skin inflammation in hairless mice via regulation of the MAPK signaling pathway. J. Agric. Food Chem. 2014, 62, 8962–8972. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Zhang, M.; Luo, H.; Li, H. Isoorientin alleviates UVB-induced skin injury by regulating mitochondrial ROS and cellular autophagy. Biochem. Biophys. Res. Commun. 2019, 514, 1133–1139. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Otkur, W.; Zhang, Y.; Li, Q.; Ye, Y.; Zang, L.; He, H.; Hayashi, T.; Tashiro, S.; Onodera, S.; et al. Silibinin protects murine fibroblast L929 cells from UVB-induced apoptosis through the simultaneous inhibition of ATM-p53 pathway and autophagy. FEBS J. 2013, 280, 4572–4584. [Google Scholar] [CrossRef] [PubMed]
- Faurschou, A.; Beyer, D.M.; Schmedes, A.; Bogh, M.K.; Philipsen, P.A.; Wulf, H.C. The relation between sunscreen layer thickness and vitamin D production after ultraviolet B exposure: A randomized clinical trial. Br. J. Dermatol. 2012, 167, 391–395. [Google Scholar] [CrossRef]
- Zhu, S.; Ma, L.; Wu, Y.; Ye, X.; Zhang, T.; Zhang, Q.; Rasoul, L.M.; Liu, Y.; Guo, M.; Zhou, B.; et al. FGF21 treatment ameliorates alcoholic fatty liver through activation of AMPK-SIRT1 pathway. Acta Biochim. Biophys. Sin. 2014, 46, 1041–1048. [Google Scholar] [CrossRef]
- Song, Y.; Ding, J.; Jin, R.; Jung, J.; Li, S.; Yang, J.; Wang, A.; Li, Z. Expression and purification of FGF21 in Pichia pastoris and its effect on fibroblast-cell migration. Mol. Med. Rep. 2016, 13, 3619–3626. [Google Scholar] [CrossRef] [PubMed]
- Itoh, N.; Ornitz, D.M. Fibroblast growth factors: From molecular evolution to roles in development, metabolism and disease. J. Biochem. 2011, 149, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yang, R.; Chen, M.; Zhou, Q.; Zheng, Y.; Lu, C.; Bi, J.; Sun, W.; Huang, T.; Li, L.; et al. KGF-2 and FGF-21 poloxamer 407 hydrogel coordinates inflammation and proliferation homeostasis to enhance wound repair of scalded skin in diabetic rats. BMJ Open Diabetes Res. Care 2020, 8, e001009. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.M.; Song, S.S.; Xiao, H.; Gao, P.; Li, X.J.; Si, L.Y. Fibroblast growth factor 21 protects against high glucose induced cellular damage and dysfunction of endothelial nitric-oxide synthase in endothelial cells. Cell Physiol. Biochem. 2014, 34, 658–671. [Google Scholar] [CrossRef] [PubMed]
- Taliyan, R.; Chandran, S.K.; Kakoty, V. Therapeutic Approaches to Alzheimer’s Type of Dementia: A Focus on FGF21 Mediated Neuroprotection. Curr. Pharm. Des. 2019, 25, 2555–2568. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhao, Y.; Zou, Y.; Huang, W.; Zhu, L.; Liu, F.; Wang, D.; Guo, K.; Hu, J.; Chen, J.; et al. Heparin-poloxamer hydrogel-encapsulated rhFGF21 enhances wound healing in diabetic mice. FASEB J. 2019, 33, 9858–9870. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Hu, J.; Liu, H.; Xiong, Y.; Zou, Y.; Huang, W.; Shao, M.; Wu, J.; Yu, L.; Wang, X.; et al. FGF21 Protects the Blood-Brain Barrier by Upregulating PPARgamma via FGFR1/beta-klotho after Traumatic Brain Injury. J. Neurotrauma. 2018, 35, 2091–2103. [Google Scholar] [CrossRef] [PubMed]
- Levine, B.; Kroemer, G. Autophagy in the pathogenesis of disease. Cell 2008, 132, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Velentzas, A.D.; Nezis, I.P.; Stravopodis, D.J.; Papassideri, I.S.; Margaritis, L.H. Apoptosis and autophagy function cooperatively for the efficacious execution of programmed nurse cell death during Drosophila virilis oogenesis. Autophagy 2007, 3, 130–132. [Google Scholar] [CrossRef]
- Li, Y.; Chen, Y. AMPK and Autophagy. Adv. Exp. Med. Biol. 2019, 1206, 85–108. [Google Scholar]
- Villanueva-Paz, M.; Cotan, D.; Garrido-Maraver, J.; Oropesa-Avila, M.; de la Mata, M.; Delgado-Pavon, A.; de Lavera, I.; Alcocer-Gomez, E.; Alvarez-Cordoba, M.; Sanchez-Alcazar, J.A. AMPK Regulation of Cell Growth, Apoptosis, Autophagy, and Bioenergetics. Exp. Suppl. 2016, 107, 45–71. [Google Scholar]
- Liu, X.; Xu, Q.; Long, X.; Liu, W.; Zhao, Y.; Hayashi, T.; Hattori, S.; Fujisaki, H.; Ogura, T.; Tashiro, S.I.; et al. Silibinin-induced autophagy mediated by PPARalpha-sirt1-AMPK pathway participated in the regulation of type I collagen-enhanced migration in murine 3T3-L1 preadipocytes. Mol. Cell. Biochem. 2019, 450, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Fan, X.; Meng, X.; Sun, S.; Su, Y.; Ling, X.; Chen, X.; Wang, K.; Dai, X.; Zhang, C.; et al. FGF21 promotes ischaemic angiogenesis and endothelial progenitor cells function under diabetic conditions in an AMPK/NAD+-dependent manner. J. Cell. Mol. Med. 2021, 25, 3091–3102. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.; Chen, H.; Lin, J.; Xu, H.; Wu, H.; Bao, G.; Li, J.; Deng, X.; Shui, X.; Gao, W.; et al. FGF21 augments autophagy in random-pattern skin flaps via AMPK signaling pathways and improves tissue survival. Cell Death Dis. 2019, 10, 872. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.M.; Im, A.R.; Park, S.K.; Shin, H.S.; Chae, S.W. Protective Effects of Timosaponin AIII against UVB-Radiation Induced Inflammation and DNA Injury in Human Epidermal Keratinocytes. Biol. Pharm. Bull. 2019, 42, 1524–1531. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.; Hwang, E.; Ngo, H.T.T.; Seo, S.A.; Yi, T.H. Sambucus nigra L. ameliorates UVB-induced photoaging and inflammatory response in human skin keratinocytes. Cytotechnology 2019, 71, 1003–1017. [Google Scholar] [CrossRef] [PubMed]
- Yeo, I.J.; Park, J.H.; Jang, J.S.; Lee, D.Y.; Park, J.E.; Choi, Y.E.; Joo, J.H.; Song, J.K.; Jeon, H.O.; Hong, J.T. Inhibitory effect of Carnosol on UVB-induced inflammation via inhibition of STAT3. Arch. Pharm. Res. 2019, 42, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Shirakata, Y.; Kimura, R.; Nanba, D.; Iwamoto, R.; Tokumaru, S.; Morimoto, C.; Yokota, K.; Nakamura, M.; Sayama, K.; Mekada, E.; et al. Heparin-binding EGF-like growth factor accelerates keratinocyte migration and skin wound healing. J. Cell Sci. 2005, 118, 2363–2370. [Google Scholar] [CrossRef] [PubMed]
- Ha, J.H.; Kim, H.N.; Moon, K.B.; Jeon, J.H.; Jung, D.H.; Kim, S.J.; Mason, H.S.; Shin, S.Y.; Kim, H.S.; Park, K.M. Recombinant Human Acidic Fibroblast Growth Factor (aFGF) Expressed in Nicotiana benthamiana Potentially Inhibits Skin Photoaging. Planta Med. 2017, 83, 862–869. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Ye, J.; Zhu, J.; Xiao, Z.; He, C.; Shi, H.; Wang, Y.; Lin, C.; Zhang, H.; Zhao, Y.; et al. Heparin-Based Coacervate of FGF2 Improves Dermal Regeneration by Asserting a Synergistic Role with Cell Proliferation and Endogenous Facilitated VEGF for Cutaneous Wound Healing. Biomacromolecules 2016, 17, 2168–2177. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhu, J.; He, C.; Xiao, Z.; Ye, J.; Li, Y.; Chen, A.; Zhang, H.; Li, X.; Lin, L.; et al. Comparative Study of Heparin-Poloxamer Hydrogel Modified bFGF and aFGF for in Vivo Wound Healing Efficiency. ACS Appl. Mater. Interfaces 2016, 8, 18710–18721. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Li, Y.; Wu, Y.; Zhao, Y.; Chen, H.; Yuan, Y.; Xu, K.; Zhang, H.; Lu, Y.; Wang, J.; et al. Heparin-Poloxamer Thermosensitive Hydrogel Loaded with bFGF and NGF Enhances Peripheral Nerve Regeneration in Diabetic Rats. Biomaterials 2018, 168, 24–37. [Google Scholar] [CrossRef]
- Zhao, Y.; Ye, S.; Lin, J.; Liang, F.; Chen, J.; Hu, J.; Chen, K.; Fang, Y.; Chen, X.; Xiong, Y.; et al. NmFGF1-Regulated Glucolipid Metabolism and Angiogenesis Improves Functional Recovery in a Mouse Model of Diabetic Stroke and Acts via the AMPK Signaling Pathway. Front. Pharmacol. 2021, 12, 680351. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Ma, J.; Mu, D.; Li, B.; Lian, B.; Sun, C. FGF21 Protects Dopaminergic Neurons in Parkinson’s Disease Models Via Repression of Neuroinflammation. Neurotox. Res. 2020, 37, 616–627. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, Y.; Zhang, Z.; Liu, Q.; Gu, J. Cardioprotective effects of fibroblast growth factor 21 against doxorubicin-induced toxicity via the SIRT1/LKB1/AMPK pathway. Cell Death Dis. 2017, 8, e3018. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Uchi, H.; Furue, M. Antioxidant cinnamaldehyde attenuates UVB-induced photoaging. J. Dermatol. Sci. 2019, 96, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Chae, M.; Son, E.D.; Bae, I.H.; Cho, E.G.; Kim, H.J.; Jung, J.Y. UVB-dependent inhibition of lipin-1 protects against proinflammatory responses in human keratinocytes. Exp. Mol. Med. 2020, 52, 293–307. [Google Scholar] [CrossRef] [PubMed]
- Choi, A.M.; Ryter, S.W.; Levine, B. Autophagy in human health and disease. N. Engl. J. Med. 2013, 368, 651–662. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Pan, Y.; Xiong, R.; Zheng, J.; Li, Q.; Zhang, S.; Li, X.; Pan, X.; Yang, S. FGF21 mediates the protective effect of fenofibrate against acetaminophen -induced hepatotoxicity via activating autophagy in mice. Biochem. Biophys. Res. Commun. 2018, 503, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.T.W.; Li, S.Y.T.; Leung, P.S. Fibroblast Growth Factor 21 Stimulates Pancreatic Islet Autophagy via Inhibition of AMPK-mTOR Signaling. Int. J. Mol. Sci. 2019, 20, 2517. [Google Scholar] [CrossRef] [PubMed]
- Salminen, A.; Kaarniranta, K.; Kauppinen, A. Regulation of longevity by FGF21: Interaction between energy metabolism and stress responses. Ageing Res. Rev. 2017, 37, 79–93. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.L.; Qiang, L.; Han, W.; Ming, M.; Viollet, B.; He, Y.Y. Role of AMPK in UVB-induced DNA damage repair and growth control. Oncogene 2013, 32, 2682–2689. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Xiao, W.; Zeng, Y.; Liu, M.H.; Li, G.H.; Tang, Z.H.; Qu, S.L.; Hao, Y.M.; Yuan, H.Q.; Jiang, Z.S. Fibroblast growth factor-21 alleviates hypoxia/reoxygenation injury in H9c2 cardiomyocytes by promoting autophagic flux. Int. J. Mol. Med. 2019, 43, 1321–1330. [Google Scholar] [CrossRef] [PubMed]
- Racanelli, A.C.; Kikkers, S.A.; Choi, A.M.K.; Cloonan, S.M. Autophagy and inflammation in chronic respiratory disease. Autophagy 2018, 14, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.L.; Lu, Z.Y.; Zhang, X.; Liu, W.W.; Yao, G.D.; Liu, X.L.; Liu, W.; Wu, Q.J.; Hayashi, T.; Yamato, M.; et al. Gelatin promotes cell aggregation and pro-inflammatory cytokine production in PMA-stimulated U937 cells by augmenting endocytosis-autophagy pathway. Int. J. Biochem. Cell Biol. 2018, 95, 132–142. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xiao, Y.; Fu, L.; Zhao, H.; Zhang, Y.; Wan, X.; Qin, Y.; Huang, Y.; Gao, H.; Li, X. High-level expression and purification of soluble recombinant FGF21 protein by SUMO fusion in Escherichia coli. BMC Biotechnol. 2010, 10, 14. [Google Scholar] [CrossRef]


| Gene | Primer Sequences |
|---|---|
| ACTB | 5′-GTGCTATGTTGCTCTAGACTTCG-3′ 5′-ATGCCACAGGATTCCATACC-3′ |
| Il1b | 5′-AAGCCTCGTGCTGTCGGACC-3′ 5′-TGAGGCCCAAGGCCACAGGT-3′ |
| Tnf | 5′-CAAGGGACAAGGCTGCCCCG-3′ 5′-GCAGGGGCTCTTGACGGCAG-3′ |
| IL6 | 5′-AGAAGGAGTGGCTAAGGACCAA-3′ 5′-AACGCACTAGGTTTGCCGAGTA-3′ |
| COX2 | 5′-CCCTTGGGTGTCAAAGGTAA-3′ 5′-GCCCTCGCTTATGATCTGTC-3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Y.; Lin, J.; Li, J.; Bwalya, C.; Xu, Y.; Niu, Y.; Zhang, Y.; Wu, J.; Xu, Y.; Chen, J.; et al. RhFGF21 Protects Epidermal Cells against UVB-Induced Apoptosis through Activating AMPK-Mediated Autophagy. Int. J. Mol. Sci. 2022, 23, 12466. https://doi.org/10.3390/ijms232012466
Zhao Y, Lin J, Li J, Bwalya C, Xu Y, Niu Y, Zhang Y, Wu J, Xu Y, Chen J, et al. RhFGF21 Protects Epidermal Cells against UVB-Induced Apoptosis through Activating AMPK-Mediated Autophagy. International Journal of Molecular Sciences. 2022; 23(20):12466. https://doi.org/10.3390/ijms232012466
Chicago/Turabian StyleZhao, Yeli, Jingjing Lin, Jiana Li, Canol Bwalya, Yuyun Xu, Yue Niu, Yujie Zhang, Junyi Wu, Yifan Xu, Jun Chen, and et al. 2022. "RhFGF21 Protects Epidermal Cells against UVB-Induced Apoptosis through Activating AMPK-Mediated Autophagy" International Journal of Molecular Sciences 23, no. 20: 12466. https://doi.org/10.3390/ijms232012466
APA StyleZhao, Y., Lin, J., Li, J., Bwalya, C., Xu, Y., Niu, Y., Zhang, Y., Wu, J., Xu, Y., Chen, J., Ye, S., & Lin, L. (2022). RhFGF21 Protects Epidermal Cells against UVB-Induced Apoptosis through Activating AMPK-Mediated Autophagy. International Journal of Molecular Sciences, 23(20), 12466. https://doi.org/10.3390/ijms232012466





