Effect of Silica Microparticles on Interactions in Mono- and Multicomponent Membranes
Abstract
:1. Introduction
2. Results and Discussion
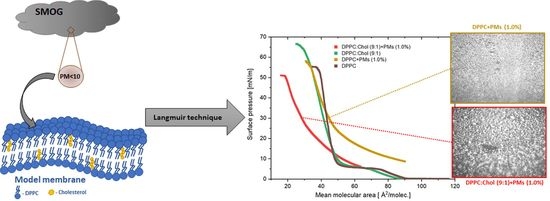
2.1. Impact of Cholesterol on the Model Lipid Monolayer with/without Silica Microparticles
| Alift-off [Å2/molec.] | Acollapse [Å2/molec.] | πcollapse [mN/m] | [mN/m] | |
|---|---|---|---|---|
| DPPC | 88 | 35 | 55.3 | 254.1 |
| Cholesterol * | 52 | 38 | 41.0 | 693.0 |
| DPPC:Chol (9:1) | 84 | 25 | 67.1 | 144.1 |
| DPPC + 0.033% MPs | 61 | 18 | 60.9 | 162.6 |
| DPPC:Chol(9:1) + 0.033% MPs | 63 | 19 | 53.8 | 125.2 |
| DPPC + 0.33% MPs | <90 | 31 | 58.2 | 104.7 |
| DPPC:Chol (9:1) + 0.33% MPs | <64 | 18 | 50.8 | 60.2 |
| DPPC + 0.83% MPs | <90 | 37 | 62.4 | 88.7 |
| DPPC:Chol (9:1) + 0.83% MPs | <64 | 29 | 49.5 | 46.1 |
2.2. Relaxation Experiment
2.2.1. Effect of Cholesterol Addition on the Stability of DPPC + MPs Monolayers
2.2.2. Influence of MPs Distribution on the Stability of the Lipid Monolayer
3. Materials and Methods
3.1. Materials
3.2. Isotherm π–A Experiments
3.3. Brewster Angle Microscopy (BAM)
3.4. Relaxation Experiments
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, J.; Mou, L.; Jiang, X. Surface chemistry of gold nanoparticles for health-related applications. Chem. Sci. 2020, 11, 923–936. [Google Scholar] [CrossRef] [Green Version]
- Bansal, S.A.; Kumar, V.; Karimi, J.; Singh, A.P.; Kumar, S. Role of gold nanoparticles in advanced biomedical applications. Nanoscale Adv. 2020, 2, 3764–3787. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.U.; Khan, M.; Malik, N.; Cho, M.H.; Khan, M.M. Recent progress of algae and blue–green algae-assisted synthesis of gold nanoparticles for various applications. Bioprocess Biosyst. Eng. 2019, 42, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Tim, B.; Błaszkiewicz, P.; Kotkowiak, M. Recent advances in metallic nanoparticle assemblies for surface-enhanced spectroscopy. Int. J. Mol. Sci. 2022, 23, 291. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.T.; Liao, C.Y.; Kuo, C.Y.; Kuo, H.W. The effects of PM2.5 from asian dust storms on emergency room visits for cardiovascular and respiratory diseases. Int. J. Environ. Res. Public Health 2017, 14, 428. [Google Scholar] [CrossRef] [Green Version]
- Prata, J.C. Airborne microplastics: Consequences to human health? Environ. Pollut. 2018, 234, 115–126. [Google Scholar] [CrossRef]
- Schmid, O.; Stoeger, T. Surface area is the biologically most effective dose metric for acute nanoparticle toxicity in the lung. J. Aerosol Sci. 2016, 99, 133–143. [Google Scholar] [CrossRef] [Green Version]
- Dwivedi, M.V.; Harishchandra, R.K.; Koshkina, O.; Maskos, M.; Galla, H.-J. Size Influences the Effect of Hydrophobic Nanoparticles on Lung Surfactant Model Systems. Biophys. J. 2014, 106, 289–298. [Google Scholar] [CrossRef] [Green Version]
- Allegri, M.; Bianchi, M.G.; Chiu, M.; Varet, J.; Costa, A.L.; Ortelli, S.; Blosi, M.; Bussolati, O.; Poland, C.A.; Bergamaschi, E. Shape-related toxicity of titanium dioxide nanofibers. PLoS ONE 2016, 11, e0151365. [Google Scholar] [CrossRef] [Green Version]
- Wieland, S.; Balmes, A.; Bender, J.; Kitzinger, J.; Meyer, F.; Ramsperger, A.F.R.M.; Roeder, F.; Tengelmann, C.; Wimmer, B.H.; Laforsch, C.; et al. From properties to toxicity: Comparing microplastics to other airborne microparticles. J. Hazard. Mater. 2022, 428, 128151. [Google Scholar] [CrossRef]
- Chubb, L.G.; Cauda, E.G. Characterizing particle size distributions of crystalline silica in gold mine dust. Aerosol Air Qual. Res. 2017, 17, 24–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barnes, H.; Goh, N.S.L.; Leong, T.L.; Hoy, R. Silica-associated lung disease: An old-world exposure in modern industries. Respirology 2019, 24, 1165–1175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Islam, M.Z.; Krajewska, M.; Hossain, S.I.; Prochaska, K.; Anwar, A.; Deplazes, E.; Saha, S.C. Concentration-dependent effect of the steroid drug prednisolone on a lung surfactant monolayer. Langmuir 2022, 38, 4188–4199. [Google Scholar] [CrossRef]
- Sheridan, A.J.; Slater, J.M.; Arnold, T.; Campbell, R.A.; Thompson, K.C. Changes to DPPC domain structure in the presence of carbon nanoparticles. Langmuir 2017, 33, 10374–10384. [Google Scholar] [CrossRef] [Green Version]
- Davies, M.J.; Birkett, J.W.; Bolton, H.; Moore, A. The influence of cannabis smoke and cannabis vapour on simulated lung surfactant function under physiologically relevant conditions. Surf. Interface Anal. 2019, 51, 558–565. [Google Scholar] [CrossRef]
- Tim, B.; Błaszkiewicz, P.; Kotkowiak, M. Altering model cell membranes by means of photoactivated organic functionalized gold nanorods. J. Mol. Liq. 2022, 349, 118179. [Google Scholar] [CrossRef]
- Jurak, M.; Miñones, J. Interactions of lauryl gallate with phospholipid components of biological membranes. Biochim. Biophys. Acta Biomembr. 2016, 1858, 1821–1832. [Google Scholar] [CrossRef]
- Dopierała, K.; Krajewska, M.; Weiss, M. Physicochemical characterization of oleanolic acid-human serum albumin complexes for pharmaceutical and biosensing applications. Langmuir 2020, 36, 3611–3623. [Google Scholar] [CrossRef]
- Dana, R.M. The monolayer technique: A potent tool for studying the interfacial properties of antimicrobial and membrane 860 lytic peptides and their interactions with lipid membranes. Biochim. Biophys. Acta 1999, 1462, 109–140. [Google Scholar] [CrossRef] [Green Version]
- Rojewska, M.; Smułek, W.; Kaczorek, E.; Prochaska, K. Langmuir monolayer techniques for the investigation of model bacterial membranes and antibiotic biodegradation mechanisms. Membranes 2021, 11, 707. [Google Scholar] [CrossRef]
- Hu, J.; Li, X.; Li, M.; Shang, Y.; He, Y.; Liu, H. Real-time monitoring of the effect of carbon nanoparticles on the surface behavior of DPPC/DPPG Langmuir monolayer. Colloid. Surf. B Biointerfaces 2020, 190, 110922–110932. [Google Scholar] [CrossRef]
- Zuo, Y.Y.; Veldhuizen, R.A.W.; Neumann, A.W.; Petersen, N.O.; Possmayer, F. Current perspectives in pulmonary surfactant—Inhibition, enhancement and evaluation. Biochim. Biophys. Acta Biomembr. 2008, 1778, 1947–1977. [Google Scholar] [CrossRef] [Green Version]
- Piosik, E.; Ziegler-Borowska, M.; Chełminiak-Dudkiewicz, D.; Martyński, T. Effect of aminated chitosan-coated Fe3O4 nanoparticles with applicational potential in nanomedicine on DPPG, DSPC, and POPC Langmuir monolayers as cell membrane models. Int. J. Mol. Sci. 2021, 22, 2467. [Google Scholar] [CrossRef]
- Guzmán, E.; Liggieri, L.; Santini, E.; Ferrari, M.; Ravera, F. Influence of silica nanoparticles on phase behavior and structural properties of DPPC-palmitic acid Langmuir monolayers. Colloid. Surf. A Physicochem. Eng. Asp. 2012, 413, 280–287. [Google Scholar] [CrossRef]
- Elbagory, A.; Meyer, M.; Cupido, C.; Hussein, A.A. Inhibition of bacteria associated with wound infection by biocompatible green synthesized gold nanoparticles from South African plant extracts. Nanomaterials 2017, 7, 417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baghdan, E.; Duse, L.; Schüer, J.J.; Pinnapireddy, S.R.; Pourasghar, M.; Schäfer, J.; Schneider, M.; Bakowsky, U. Development of inhalable curcumin loaded nano-in-microparticles for bronchoscopic photodynamic therapy. Eur. J. Pharm. Sci. 2019, 132, 63–71. [Google Scholar] [CrossRef]
- Rojewska, M.; Tim, B.; Prochaska, K. Interactions between silica particles and model phospholipid monolayers. J. Mol. Liq. 2022, 345, 116999. [Google Scholar] [CrossRef]
- Lin, J.; Miao, L.; Zhong, G.; Lin, C.H.; Dargazangy, R.; Alexander-Katz, A. Understanding the synergistic effect of physicochemical properties of nanoparticles and their cellular entry pathways. Commun. Biol. 2020, 3, 205. [Google Scholar] [CrossRef]
- Dopierała, K.; Maciejewski, H.; Prochaska, K. Interaction of polyhedral oligomeric silsesquioxane containing epoxycyclohexyl groups with cholesterol at the air/water interface. Colloid. Surf. B Biointerfaces 2016, 140, 135–141. [Google Scholar] [CrossRef]
- Kim, K.; Kim, C.; Byun, Y. Preparation of a dipalmitoylphosphatidylcholine/cholesterol Langmuir-Blodgett monolayer that suppresses protein adsorption. Langmuir 2001, 17, 5066–5070. [Google Scholar] [CrossRef]
- Guzmán, E.; Liggieri, L.; Santini, E.; Ferrari, M.; Ravera, F. Mixed DPPC–cholesterol Langmuir monolayers in presence of hydrophilic silica nanoparticles. Colloid. Surf. B Biointerfaces 2013, 105, 284–293. [Google Scholar] [CrossRef]
- Chou, T.H.; Chang, C.H. Thermodynamic behavior and relaxation processes of mixed DPPC/cholesterol monolayers at the air/water interface. Colloid. Surf. B Biointerfaces 2000, 17, 71–79. [Google Scholar] [CrossRef]
- Stottrup, B.L.; Keller, S.L. Phase behavior of lipid monolayers containing DPPC and cholesterol analogs. Biophys. J. 2006, 90, 3176–3183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miñones, J.; Pais, S.; Conde, O.; Dynarowicz-Łątka, P. Interactions between membrane sterols and phospholipids in model mammalian and fungi cellular membranes—A Langmuir monolayer stud. Biophys. Chem. 2009, 140, 69–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sabatini, K.; Mattila, J.P.; Kinnunen, P.K.J. Interfacial behavior of cholesterol, ergosterol, and lanosterol in mixtures with DPPC and DMPC. Biophys. J. 2008, 95, 2340–2355. [Google Scholar] [CrossRef] [Green Version]
- Giri, R.P.; Chakrabarti, A.; Mukhopadhyay, M.K. Cholesterol-induced structural changes in saturated phospholipid model membranes revealed through X ray scattering technique. J. Phys. Chem. B 2017, 121, 4081–4090. [Google Scholar] [CrossRef]
- Botet-Carrerasa, A.; Monteroa, M.T.; Domènecha, O.; Borrell, J.H. Effect of cholesterol on monolayer structure of different acyl chained phospholipids. Colloid. Surf. B Biointerfaces 2019, 174, 374–383. [Google Scholar] [CrossRef]
- Przykaza, K.; Woźniak, K.; Jurak, M.; Wiącek, A.E.; Mroczka, R. Properties of the Langmuir and Langmuir–Blodgett monolayers of cholesterol-cyclosporine A on water and polymer support. Adsorption 2019, 25, 923–936. [Google Scholar] [CrossRef] [Green Version]
- Anfinogenova, Y.; Shvedova, M.; Popov, S.V. Nanoparticles as agents targeting cholesterol crystallization in atherosclerosis. Med. Hypotheses 2017, 102, 19–22. [Google Scholar] [CrossRef]
- Alavizargar, A.; Keller, F.; Wedlich-Söldner, R.; Heuer, A. Effect of cholesterol vs. ergosterol on DPPC bilayer properties: Insights from atomistic simulations. J. Phys. Chem. B 2021, 125, 7679–7690. [Google Scholar] [CrossRef]
- Lin, H.; Guo, Y.; Ruan, Z.; Yang, Y.; Chen, Y.; Zheng, Y.; Cummings-Vaughn, L.A.; Rigdon, S.E.; Vaughn, M.G.; Sun, S.; et al. Ambient PM2.5 and O3 and their combined effects on prevalence of presbyopia among the elderly: A cross-sectional study in six low- and middle-income countries. Sci Total Environ. 2019, 655, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Mimura, T.; Ichinose, T.; Yamagami, S.; Fujishima, H.; Kamei, Y.; Goto, M.; Takada, S.; Matsubara, M. Airborne particulate matter (PM2.5) and the prevalence of allergic conjunctivitis in Japan. Sci Total Environ. 2014, 487, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Yue, K.; Sun, X.; Tang, J.; Wei, Y.; Zhang, X. A simulation study on the interaction between pollutant nanoparticles and the pulmonary surfactant monolayer. Int. J. Mol. Sci. 2019, 20, 3281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bañuelos-Frias, A.; Castañeda-Montiel, V.M.; Alvizo-Paez, E.R.; Vazquez-Martinez, E.A.; Gomez, E.; Ruiz-Garcia, J. Thermodynamic and mechanical properties of DMPC/cholesterol mixed monolayers at physiological conditions. Front. Phys. 2021, 9, 636149. [Google Scholar] [CrossRef]
- Miguel Diez, M.; Buckley, A.; Tetley, T.D.; Smith, R. The method of depositing CeO2 nanoparticles onto a DPPC monolayer affects surface tension behaviour. NanoImpact 2019, 16, 100186. [Google Scholar] [CrossRef]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tim, B.; Rojewska, M.; Prochaska, K. Effect of Silica Microparticles on Interactions in Mono- and Multicomponent Membranes. Int. J. Mol. Sci. 2022, 23, 12822. https://doi.org/10.3390/ijms232112822
Tim B, Rojewska M, Prochaska K. Effect of Silica Microparticles on Interactions in Mono- and Multicomponent Membranes. International Journal of Molecular Sciences. 2022; 23(21):12822. https://doi.org/10.3390/ijms232112822
Chicago/Turabian StyleTim, Beata, Monika Rojewska, and Krystyna Prochaska. 2022. "Effect of Silica Microparticles on Interactions in Mono- and Multicomponent Membranes" International Journal of Molecular Sciences 23, no. 21: 12822. https://doi.org/10.3390/ijms232112822