Attenuation of Myocardial Dysfunction in Hypertensive Cardiomyopathy Using Non-R-Wave-Synchronized Cardiac Shock Wave Therapy
Abstract
:1. Introduction
2. Results
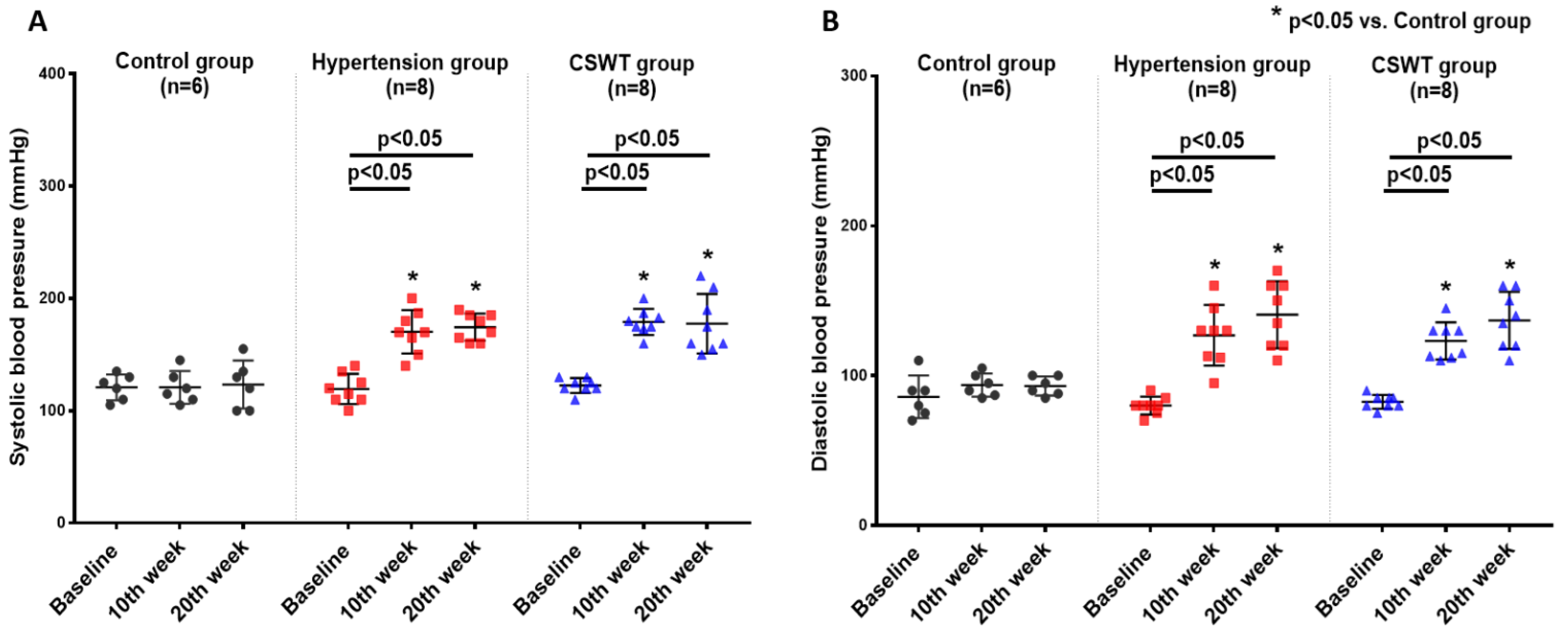
2.1. CSWT Has No Effect on Blood Pressure in Hypertensive Cardiomyopathy
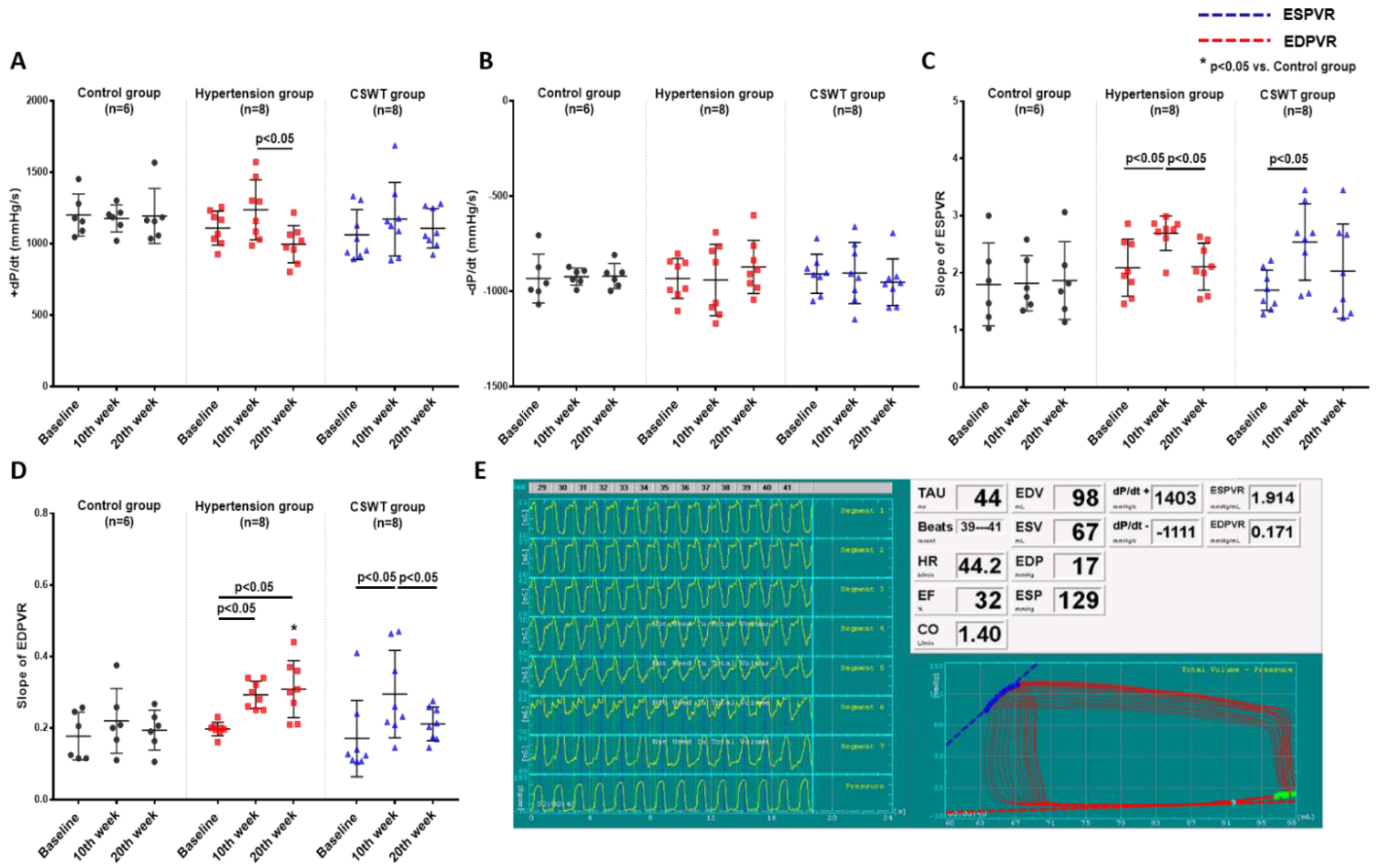
2.2. Hemodynamic Data
2.3. Echocardiographic Data
2.4. No Incidence of Arrhythmias during CSWT
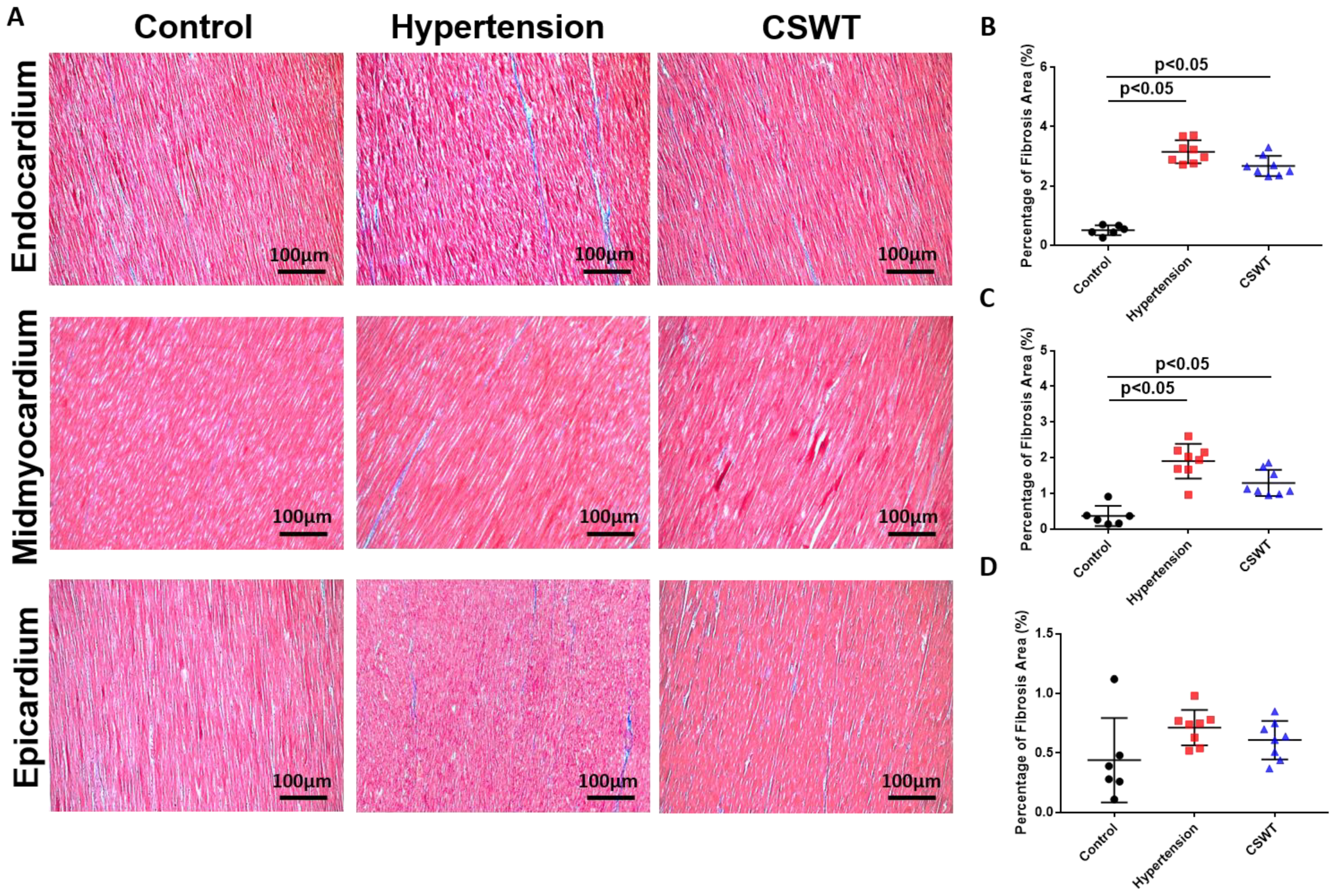
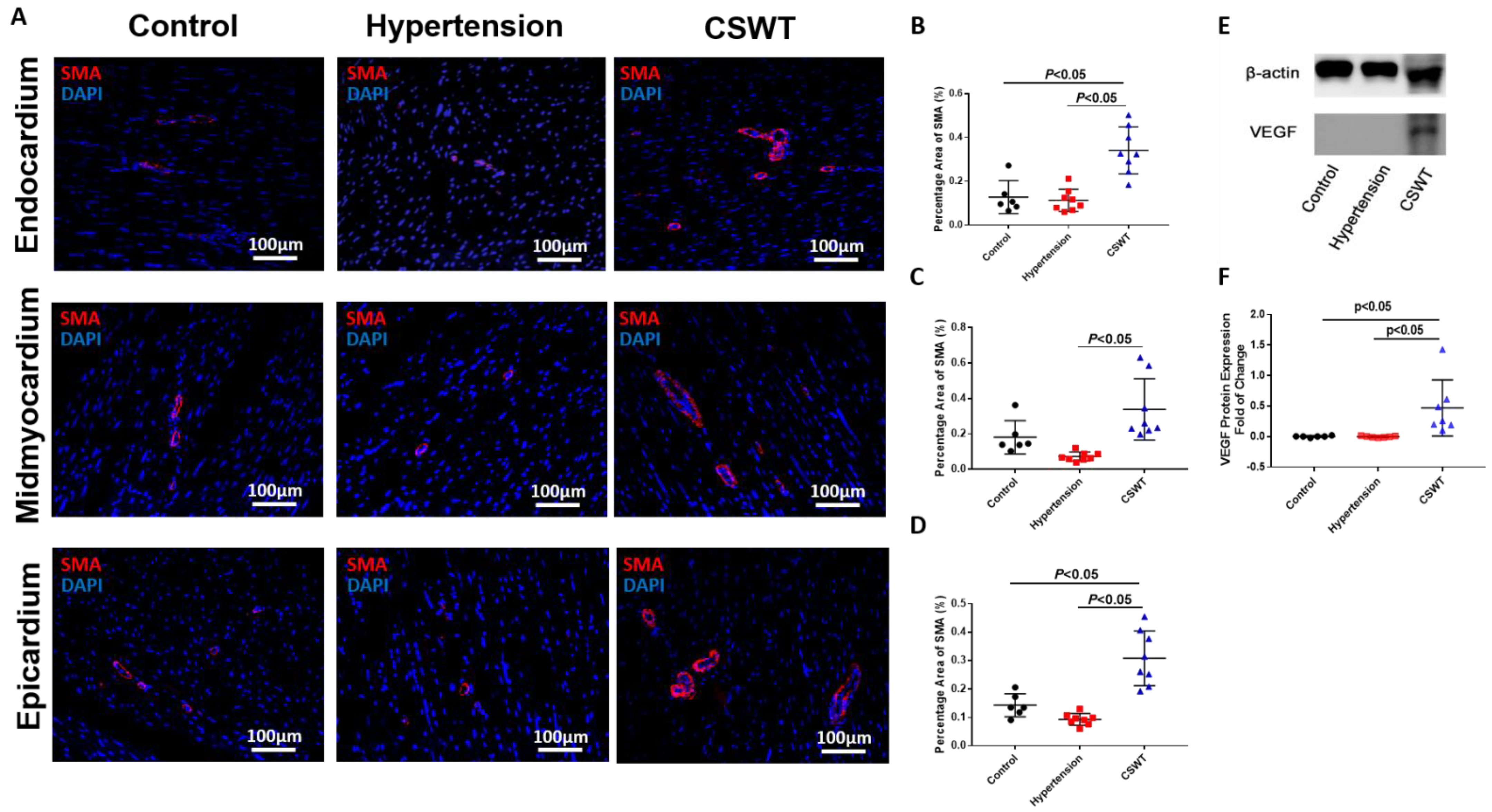
2.5. Histological and Immunohistochemical Data
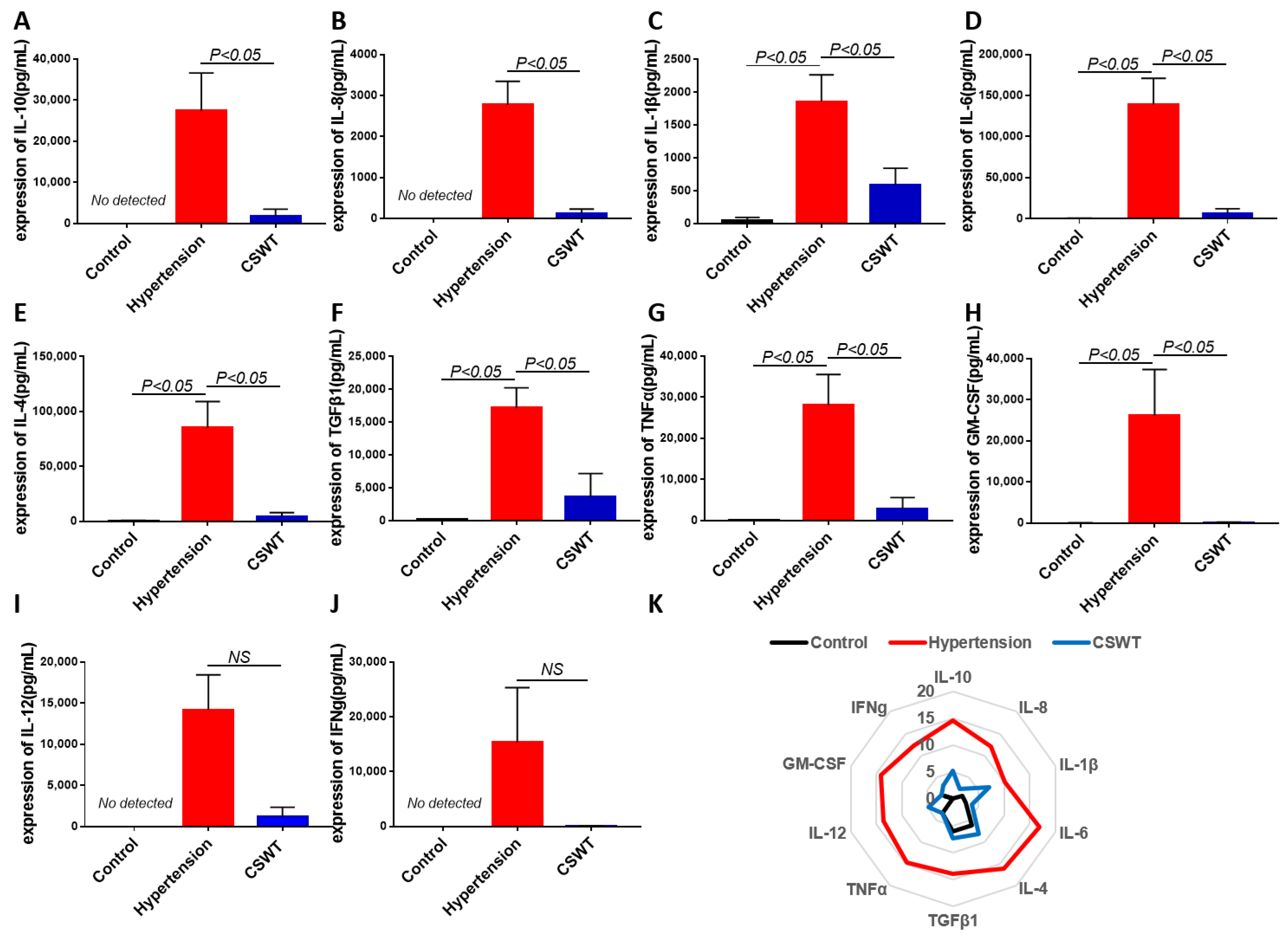
2.6. Cytokines Array Data
3. Discussion
3.1. Perspectives
3.2. Limitations
4. Methods and Materials
4.1. Animal Model of Hypertensive Cardiomyopathy
4.2. Cardiac Shock Wave Therapy
4.3. Echocardiographic Examination
4.4. Invasive Hemodynamic Assessment
4.5. Histological Assessment
4.6. Western Blot Analysis
4.7. Cytokine Array
4.8. Biomarker Assessment
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BP | blood pressure |
| CMD | coronary microvascular dysfunction |
| CSWT | cardiac shock wave therapy |
| EDPVR | end-diastolic pressure–volume relationship |
| ESPVR | end-systolic pressure–volume relationship |
| GM-CSF | granulocyte–macrophage colony-stimulation factor |
| HF | heart failure |
| HFpEF | heart failure with preserved ejection fraction |
| IL- | interleukin- |
| INFg | interferon gamma |
| LV | left ventricle |
| LVH | left ventricular hypertrophy |
| SMA | smooth muscle actin |
| TNFα | tumor necrosis factor α |
| TGFβ1 | transforming growth factor β1 |
| VEGF | vascular endothelial growth factor |
| vWF | von Willebrand factor |
References
- Roger, V.L. Epidemiology of Heart Failure: A Contemporary Perspective. Circ. Res. 2021, 128, 1421–1434. [Google Scholar] [CrossRef] [PubMed]
- Borlaug, B.A. Evaluation and management of heart failure with preserved ejection fraction. Nat. Rev. Cardiol. 2020, 17, 559–573. [Google Scholar] [CrossRef] [PubMed]
- Bayes-Genis, A.; Diez, J. Transition to heart failure in hypertension: Going to the heart of the matter. Eur. Heart J. 2022, 43, 3332–3334. [Google Scholar] [CrossRef] [PubMed]
- Paulus, W.J.; Tschope, C. A novel paradigm for heart failure with preserved ejection fraction: Comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. J. Am. Coll. Cardiol. 2013, 62, 263–271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berry, C.; Sykes, R. Microvascular Dysfunction in Heart Failure with Preserved Ejection Fraction: A Modifiable Therapeutic Target? JACC Cardiovasc. Imaging 2022, 15, 1012–1014. [Google Scholar] [CrossRef]
- Qiu, Q.; Chen, S.; Qiu, Y.; Mao, W. Cardiac Shock Wave Therapy in Coronary Artery Disease: A Systematic Review and Meta-Analysis. Front. Cardiovasc. Med. 2022, 9, 932193. [Google Scholar] [CrossRef]
- Kikuchi, Y.; Ito, K.; Shindo, T.; Hao, K.; Shiroto, T.; Matsumoto, Y.; Takahashi, J.; Matsubara, T.; Yamada, A.; Ozaki, Y.; et al. A multicenter trial of extracorporeal cardiac shock wave therapy for refractory angina pectoris: Report of the highly advanced medical treatment in Japan. Heart Vessel. 2019, 34, 104–113. [Google Scholar] [CrossRef]
- Ito, Y.; Ito, K.; Shiroto, T.; Tsuburaya, R.; Yi, G.J.; Takeda, M.; Fukumoto, Y.; Yasuda, S.; Shimokawa, H. Cardiac shock wave therapy ameliorates left ventricular remodeling after myocardial ischemia-reperfusion injury in pigs in vivo. Coron. Artery Dis. 2010, 21, 304–311. [Google Scholar] [CrossRef]
- Gotte, G.; Amelio, E.; Russo, S.; Marlinghaus, E.; Musci, G.; Suzuki, H. Short-time non-enzymatic nitric oxide synthesis from L-arginine and hydrogen peroxide induced by shock waves treatment. FEBS Lett. 2002, 520, 153–155. [Google Scholar] [CrossRef] [Green Version]
- Holfeld, J.; Lobenwein, D.; Tepekoylu, C.; Grimm, M. Shockwave therapy of the heart. Int. J. Surg. 2015, 24, 218–222. [Google Scholar] [CrossRef]
- Abe, Y.; Ito, K.; Hao, K.; Shindo, T.; Ogata, T.; Kagaya, Y.; Kurosawa, R.; Nishimiya, K.; Satoh, K.; Miyata, S.; et al. Extracorporeal low-energy shock-wave therapy exerts anti-inflammatory effects in a rat model of acute myocardial infarction. Circ. J. 2014, 78, 2915–2925. [Google Scholar] [CrossRef] [Green Version]
- Mariotto, S.; de Prati, A.; Cavalieri, E.; Amelio, E.M.E.; Suzuki, H. Extracorporeal shock wave therapy in inflammatory diseases: Molecular mechanism that triggers anti-inflammatory action. Curr. Med. Chem. 2009, 16, 2366–2372. [Google Scholar] [CrossRef]
- Gollmann-Tepekoylu, C.; Lobenwein, D.; Theurl, M.; Primessnig, U.; Lener, D.; Kirchmair, E.; Mathes, W.; Graber, M.; Polzl, L.; An, A.; et al. Shock Wave Therapy Improves Cardiac Function in a Model of Chronic Ischemic Heart Failure: Evidence for a Mechanism Involving VEGF Signaling and the Extracellular Matrix. J. Am. Heart Assoc. 2018, 7, e010025. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Liu, M. Cardiac shock wave therapy: An alternative non-invasive therapy for refractory angina. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 5402–5410. [Google Scholar]
- Ventura, H.O.; Lavie, C.J.; Mehra, M.R. Heart Failure with Preserved Ejection Fraction: The Quest for a Blood Pressure Goal. J. Am. Coll. Cardiol. 2020, 75, 1657–1658. [Google Scholar] [CrossRef]
- Anker, S.D.; Butler, J.; Filippatos, G.; Ferreira, J.P.; Bocchi, E.; Bohm, M.; Brunner-La Rocca, H.P.; Choi, D.J.; Chopra, V.; Chuquiure-Valenzuela, E.; et al. Empagliflozin in Heart Failure with a Preserved Ejection Fraction. N. Engl. J. Med. 2021, 385, 1451–1461. [Google Scholar] [CrossRef]
- Lam, C.S.P.; Voors, A.A.; de Boer, R.A.; Solomon, S.D.; van Veldhuisen, D.J. Heart failure with preserved ejection fraction: From mechanisms to therapies. Eur. Heart J. 2018, 39, 2780–2792. [Google Scholar] [CrossRef]
- Wintrich, J.; Kindermann, I.; Ukena, C.; Selejan, S.; Werner, C.; Maack, C.; Laufs, U.; Tschope, C.; Anker, S.D.; Lam, C.S.P.; et al. Therapeutic approaches in heart failure with preserved ejection fraction: Past, present, and future. Clin. Res. Cardiol. 2020, 109, 1079–1098. [Google Scholar] [CrossRef] [Green Version]
- Zhen, Z.; Liao, S.Y.; Zhu, Z.Y.; Sijia, S.; Au, K.W.; Lai, W.H.; Tsang, A.; Hai, J.S.H.; Tse, H.F. Catheter-Based Splanchnic Denervation for Treatment of Hypertensive Cardiomyopathy. Hypertension 2019, 74, 47–55. [Google Scholar] [CrossRef]
- Graber, M.; Nagele, F.; Hirsch, J.; Polzl, L.; Schweiger, V.; Lechner, S.; Grimm, M.; Cooke, J.P.; Gollmann-Tepekoylu, C.; Holfeld, J. Cardiac Shockwave Therapy—A Novel Therapy for Ischemic Cardiomyopathy? Front. Cardiovasc. Med. 2022, 9, 875965. [Google Scholar] [CrossRef]
- Holsapple, J.S.; Cooper, B.; Berry, S.H.; Staniszewska, A.; Dickson, B.M.; Taylor, J.A.; Bachoo, P.; Wilson, H.M. Low Intensity Shockwave Treatment Modulates Macrophage Functions Beneficial to Healing Chronic Wounds. Int. J. Mol. Sci. 2021, 22, 7844. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Sanchez, C.; Azar-Manzur, F.; Gonzalez-Pacheco, H.; Amezcua-Guerra, L.M.; Masso, F.; Marquez-Velasco, R.; Bojalil, R.; Carvajal-Juarez, I.; Alexanderson-Rosas, E.; Hernandez, S.; et al. Effectiveness and Safety of Extracorporeal Shockwave Myocardial Revascularization in Patients with Refractory Angina Pectoris and Heart Failure. Am. J. Cardiol. 2021, 144, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Di Meglio, F.; Nurzynska, D.; Castaldo, C.; Miraglia, R.; Romano, V.; De Angelis, A.; Piegari, E.; Russo, S.; Montagnani, S. Cardiac shock wave therapy: Assessment of safety and new insights into mechanisms of tissue regeneration. J. Cell. Mol. Med. 2012, 16, 936–942. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, Q.; Shen, T.; Yu, X.; Jia, N.; Zhu, K.; Wang, Q.; Liu, B.; He, Q. Cardiac Shock Wave Therapy Alleviates Hypoxia/Reoxygenation-Induced Myocardial Necroptosis by Modulating Autophagy. Biomed. Res. Int. 2021, 2021, 8880179. [Google Scholar] [CrossRef]
- Liu, B.; Zhang, Y.; Jia, N.; Lan, M.; Du, L.; Zhao, D.; He, Q. Study of the Safety of Extracorporeal Cardiac Shock Wave Therapy: Observation of the Ultrastructures in Myocardial Cells by Transmission Electron Microscopy. J. Cardiovasc. Pharmacol. Ther. 2018, 23, 79–88. [Google Scholar] [CrossRef] [Green Version]
- Liao, S.; Zhang, Y.; Ting, S.; Zhen, Z.; Luo, F.; Zhu, Z.; Jiang, Y.; Sun, S.; Lai, W.H.; Lian, Q.; et al. Potent immunomodulation and angiogenic effects of mesenchymal stem cells versus cardiomyocytes derived from pluripotent stem cells for treatment of heart failure. Stem. Cell Res. Ther. 2019, 10, 78. [Google Scholar] [CrossRef] [Green Version]
- Liao, S.Y.; Zhen, Z.; Liu, Y.; Au, K.W.; Lai, W.H.; Tsang, A.; Tse, H.F. Improvement of Myocardial Function Following Catheter-Based Renal Denervation in Heart Failure. JACC Basic Transl. Sci. 2017, 2, 270–281. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, F.; Zhen, Z.; Sun, S.-J.; Jiang, Y.; Liang, W.-H.; Belau, M.; Storz, R.; Liao, S.-Y.; Tse, H.-F. Attenuation of Myocardial Dysfunction in Hypertensive Cardiomyopathy Using Non-R-Wave-Synchronized Cardiac Shock Wave Therapy. Int. J. Mol. Sci. 2022, 23, 13274. https://doi.org/10.3390/ijms232113274
Li F, Zhen Z, Sun S-J, Jiang Y, Liang W-H, Belau M, Storz R, Liao S-Y, Tse H-F. Attenuation of Myocardial Dysfunction in Hypertensive Cardiomyopathy Using Non-R-Wave-Synchronized Cardiac Shock Wave Therapy. International Journal of Molecular Sciences. 2022; 23(21):13274. https://doi.org/10.3390/ijms232113274
Chicago/Turabian StyleLi, Fei, Zhe Zhen, Si-Jia Sun, Yu Jiang, Wei-Hao Liang, Markus Belau, Rafael Storz, Song-Yan Liao, and Hung-Fat Tse. 2022. "Attenuation of Myocardial Dysfunction in Hypertensive Cardiomyopathy Using Non-R-Wave-Synchronized Cardiac Shock Wave Therapy" International Journal of Molecular Sciences 23, no. 21: 13274. https://doi.org/10.3390/ijms232113274






