Added Value of Biophysics to Study Lipid-Driven Biological Processes: The Case of Surfactins, a Class of Natural Amphiphile Molecules
Abstract
1. Introduction
2. Global Mechanism of Surfactins–Lipid Membrane Interaction
2.1. Spontaneous Insertion of Surfactins at the Interface of the Lipid Membrane
2.2. Surfactins Progressively Disorganize Lipids Constituting Membranes with Increasing Surfactins-to-Lipid Ratio
3. The Interaction of Surfactins with Lipid Membranes Is Influenced by the Lipid Composition Complexity
3.1. Influence of the Hydrophobic Core Structure
3.2. Influence of the Membrane Surface
4. Surfactin’s Structure Influences Their Interactions with Biomimetic Membranes
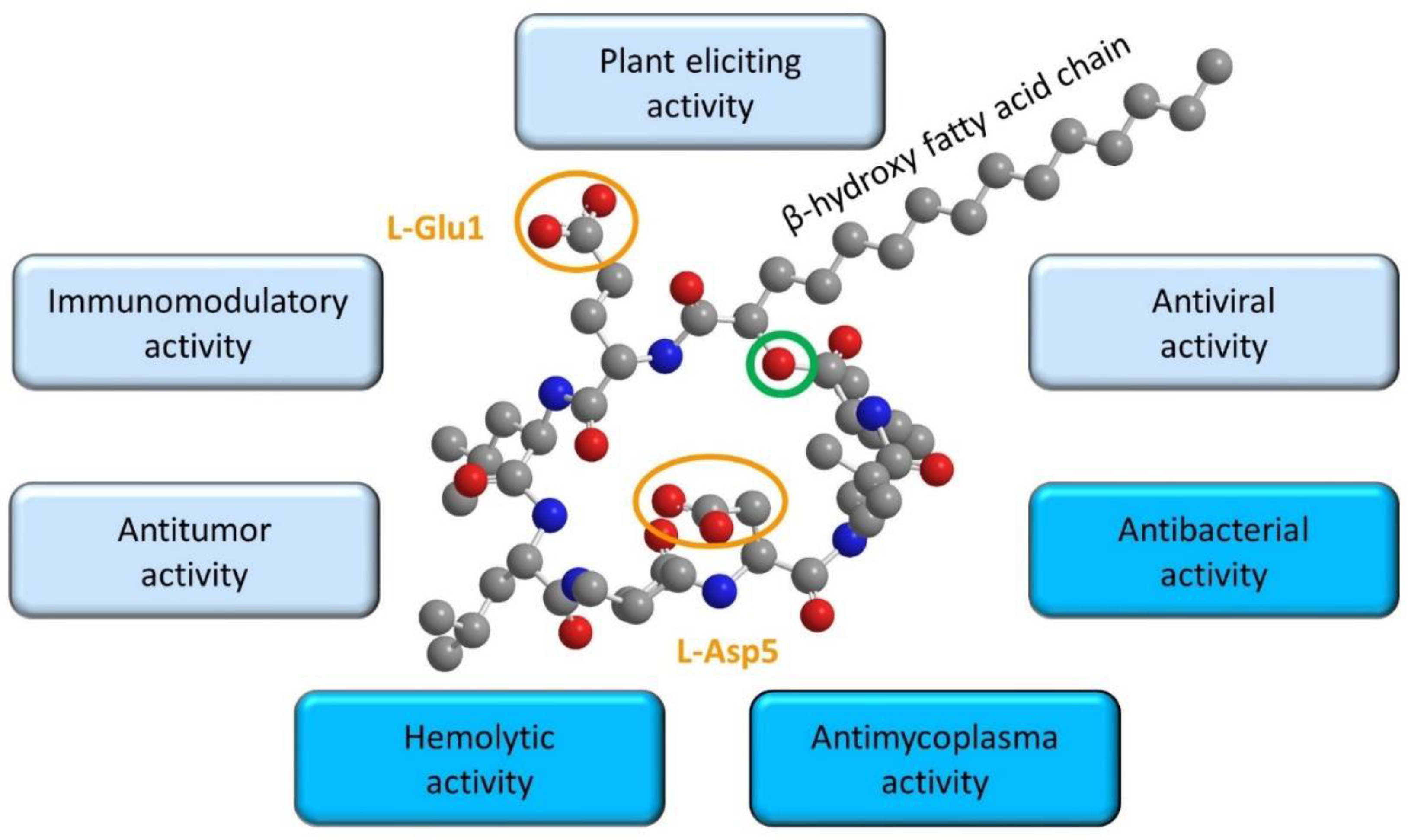
5. Relationships between Surfactin Biophysical Properties and Biological Activities
6. Biomimetic Models to Solve Challenges in Biology
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AFM | Atomic force microscopy |
| CL | cardiolipin |
| CMC | Critical micellar concentration |
| DEPE | 1,2-dielaidoyl-sn-glycero-3-phosphoethanolamine (18:1 (Δ9-trans) PE) |
| DMPC | 1,2-dimyristoyl-sn-glycero-3-phosphocholine (14:0 PC) |
| DMPG | 1,2-dimyristoyl-sn-glycero-3-phosphoglycerol (14:0 PG) |
| DMPS | 1,2-dimyristoyl-sn-glycero-3-phospho-L-serine (14:0 PS) |
| DOPC | 1,2-dioleoyl-sn-glycero-3-phosphocholine (18:1 (Δ9-cis) PC) |
| DOPG | 1,2-dioleoyl-sn-glycero-3-phosphoglycerol (18:1 (Δ9-cis) PG) |
| DPPA | 1,2-dipalmitoyl-sn-glycero-3-phosphate (16:0 PA) |
| DPPC | 1,2-dipalmitoyl-sn-glycero-3-phosphocholine (16:0 PC) |
| DPPE | 1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine (16:0 PE) |
| DPPS | 1,2-dipalmitoyl-sn-glycero-3-phospho-L-serine (16:0 PS) |
| DSC | Differential scanning calorimetry |
| DSPC | 1,2-distearoyl-sn-glycero-3-phosphocholine (18:0 PC) |
| ITC | Isothermal titration calorimetry |
| K | Membrane–water partition coefficient |
| NMR | Nuclear magnetic resonance |
| NR | Neutron reflectometry |
| PA | phosphatidic acid |
| PC | phosphatidylcholine |
| PE | phosphatidylethanolamine |
| PG | phosphatidylglycerol |
| POPC | 1-palmitoyl-2-oleoyl-glycero-3-phosphocholine (16:0–18:1 PC) |
| PS | phosphatidylserine |
| Rb | Surfactin-to-lipid ratio |
| Rbsat | Surfactin-to-lipid ratio at the onset of solubilization |
| Rbsol | Surfactin-to-lipid ratio at completion of solubilization |
| SANS | Small angle neutron scattering |
| soy PC | soy phosphatidylcholine |
| SRFs | Surfactins |
References
- Cordelier, S.; Crouzet, J.; Gilliard, G.; Dorey, S.; Deleu, M.; Dhondt-Cordelier, S. Deciphering the Role of Plant Plasma Membrane Lipids in Response to Invasion Patterns: How Could Biology and Biophysics Help? J. Exp. Bot. 2022, 73, 2765–2784. [Google Scholar] [CrossRef] [PubMed]
- Balleza, D.; Alessandrini, A.; Beltrán García, M.J. Role of Lipid Composition, Physicochemical Interactions, and Membrane Mechanics in the Molecular Actions of Microbial Cyclic Lipopeptides. J. Membrane Biol. 2019, 252, 131–157. [Google Scholar] [CrossRef] [PubMed]
- Otzen, D.E. Biosurfactants and Surfactants Interacting with Membranes and Proteins: Same but Different? Biochim. Biophys. Acta (BBA) Biomembr. 2017, 1859, 639–649. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Zhang, S.; Wang, Y.; Li, Y.; Wang, X.; Yang, Q. Surfactin Inhibits Membrane Fusion during Invasion of Epithelial Cells by Enveloped Viruses. J. Virol. 2018, 92, e00809–e00818. [Google Scholar] [CrossRef] [PubMed]
- Vollenbroich, D.; Özel, M.; Vater, J.; Kamp, R.M.; Pauli, G. Mechanism of Inactivation of Enveloped Viruses by the Biosurfactant Surfactin from Bacillus Subtilis. Biologicals 1997, 25, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Monnier, N.; Furlan, A.L.; Buchoux, S.; Deleu, M.; Dauchez, M.; Rippa, S.; Sarazin, C. Exploring the Dual Interaction of Natural Rhamnolipids with Plant and Fungal Biomimetic Plasma Membranes through Biophysical Studies. Int. J. Mol. Sci. 2019, 20, 1009. [Google Scholar] [CrossRef]
- Carravilla, P.; Nieva, J.L. HIV Antivirals: Targeting the Functional Organization of the Lipid Envelope. Future Virol. 2018, 13, 129–140. [Google Scholar] [CrossRef]
- Silverman, J.A.; Perlmutter, N.G.; Shapiro, H.M. Correlation of Daptomycin Bactericidal Activity and Membrane Depolarization in Staphylococcus Aureus. Antimicrob. Agents Chemother. 2003, 47, 2538–2544. [Google Scholar] [CrossRef]
- González-Jaramillo, L.M.; Aranda, F.J.; Teruel, J.A.; Villegas-Escobar, V.; Ortiz, A. Antimycotic Activity of Fengycin C Biosurfactant and Its Interaction with Phosphatidylcholine Model Membranes. Colloids Surf. B Biointerfaces 2017, 156, 114–122. [Google Scholar] [CrossRef]
- Huang, H.W. Daptomycin, Its Membrane-Active Mechanism vs. That of Other Antimicrobial Peptides. Biochim. Biophys. Acta (BBA) Biomembr. 2020, 1862, 183395. [Google Scholar] [CrossRef]
- Gray, D.A.; Wenzel, M. More Than a Pore: A Current Perspective on the In Vivo Mode of Action of the Lipopeptide Antibiotic Daptomycin. Antibiotics 2020, 9, 17. [Google Scholar] [CrossRef] [PubMed]
- Schellenberger, R.; Crouzet, J.; Nickzad, A.; Shu, L.-J.; Kutschera, A.; Gerster, T.; Borie, N.; Dawid, C.; Cloutier, M.; Villaume, S.; et al. Bacterial Rhamnolipids and Their 3-Hydroxyalkanoate Precursors Activate Arabidopsis Innate Immunity through Two Independent Mechanisms. Proc. Natl. Acad. Sci. USA 2021, 118, e2101366118. [Google Scholar] [CrossRef] [PubMed]
- Henry, G.; Deleu, M.; Jourdan, E.; Thonart, P.; Ongena, M. The Bacterial Lipopeptide Surfactin Targets the Lipid Fraction of the Plant Plasma Membrane to Trigger Immune-Related Defence Responses. Cell. Microbiol. 2011, 13, 1824–1837. [Google Scholar] [CrossRef]
- Hoff, G.; Arguelles Arias, A.; Boubsi, F.; Pršić, J.; Meyer, T.; Ibrahim, H.M.M.; Steels, S.; Luzuriaga, P.; Legras, A.; Franzil, L.; et al. Surfactin Stimulated by Pectin Molecular Patterns and Root Exudates Acts as a Key Driver of the Bacillus-Plant Mutualistic Interaction. mBio 2021, 12, e01774-21. [Google Scholar] [CrossRef]
- Lam, V.B.; Meyer, T.; Arias, A.A.; Ongena, M.; Oni, F.E.; Höfte, M. Bacillus Cyclic Lipopeptides Iturin and Fengycin Control Rice Blast Caused by Pyricularia oryzae in Potting and Acid Sulfate Soils by Direct Antagonism and Induced Systemic Resistance. Microorganisms 2021, 9, 1441. [Google Scholar] [CrossRef] [PubMed]
- Desmyttere, H.; Deweer, C.; Muchembled, J.; Sahmer, K.; Jacquin, J.; Coutte, F.; Jacques, P. Antifungal Activities of Bacillus subtilis Lipopeptides to Two Venturia inaequalis Strains Possessing Different Tebuconazole Sensitivity. Front. Microbiol. 2019, 10, 2327. [Google Scholar] [CrossRef]
- Das, P.; Mukherjee, S.; Sen, R. Antimicrobial Potential of a Lipopeptide Biosurfactant Derived from a Marine Bacillus circulans. J. Appl. Microbiol. 2008, 104, 1675–1684. [Google Scholar] [CrossRef]
- Falardeau, J.; Wise, C.; Novitsky, L.; Avis, T.J. Ecological and Mechanistic Insights Into the Direct and Indirect Antimicrobial Properties of Bacillus subtilis Lipopeptides on Plant Pathogens. J. Chem. Ecol. 2013, 39, 869–878. [Google Scholar] [CrossRef]
- Cao, X.; Wang, A.; Wang, C.; Mao, D.; Lu, M.; Cui, Y.; Jiao, R. Surfactin Induces Apoptosis in Human Breast Cancer MCF-7 Cells through a ROS/JNK-Mediated Mitochondrial/Caspase Pathway. Chem. Biol. Interact. 2010, 183, 357–362. [Google Scholar] [CrossRef]
- Walvekar, S.; Yasaswi, S.; Shetty, K.; Yadav, K.S. Chapter 11—Applications of Surfactin and Other Biosurfactants in Anticancer Activity. In Green Sustainable Process for Chemical and Environmental Engineering and Science; Inamuddin, Adetunji, C.O., Ahamed, M.I., Eds.; Academic Press: Cambridge, MA, USA, 2022; pp. 223–234. ISBN 978-0-323-85146-6. [Google Scholar]
- Kracht, M.; Rokos, H.; Özel, M.; Kowall, M.; Pauli, G.; Vater, J. Antiviral and Hemolytic Activities of Surfactin Isoforms and Their Methyl Ester Derivatives. J. Antibiot. 1999, 52, 613–619. [Google Scholar] [CrossRef]
- Johnson, B.A.; Hage, A.; Kalveram, B.; Mears, M.; Plante, J.A.; Rodriguez, S.E.; Ding, Z.; Luo, X.; Bente, D.; Bradrick, S.S.; et al. Peptidoglycan-Associated Cyclic Lipopeptide Disrupts Viral Infectivity. J. Virol. 2019, 93, e0128219. [Google Scholar] [CrossRef] [PubMed]
- Meena, K.R.; Kanwar, S.S. Lipopeptides as the Antifungal and Antibacterial Agents: Applications in Food Safety and Therapeutics. BioMed Res. Int. 2015, 2015, e473050. [Google Scholar] [CrossRef] [PubMed]
- Ongena, M.; Jourdan, E.; Adam, A.; Paquot, M.; Brans, A.; Joris, B.; Arpigny, J.-L.; Thonart, P. Surfactin and Fengycin Lipopeptides of Bacillus subtilis as Elicitors of Induced Systemic Resistance in Plants. Environ. Microbiol. 2007, 9, 1084–1090. [Google Scholar] [CrossRef]
- Ongena, M.; Jacques, P. Bacillus lipopeptides: Versatile Weapons for Plant Disease Biocontrol. Trends Microbiol. 2008, 16, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Pérez-García, A.; Romero, D.; de Vicente, A. Plant Protection and Growth Stimulation by Microorganisms: Biotechnological Applications of Bacilli in Agriculture. Curr. Opin. Biotechnol. 2011, 22, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Pršić, J.; Ongena, M. Elicitors of Plant Immunity Triggered by Beneficial Bacteria. Front. Plant Sci. 2020, 11, 594530. [Google Scholar] [CrossRef] [PubMed]
- Théatre, A.; Cano-Prieto, C.; Bartolini, M.; Laurin, Y.; Deleu, M.; Niehren, J.; Fida, T.; Gerbinet, S.; Alanjary, M.; Medema, M.H.; et al. The Surfactin-Like Lipopeptides from Bacillus spp.: Natural Biodiversity and Synthetic Biology for a Broader Application Range. Front. Bioeng. Biotechnol. 2021, 9, 623701. [Google Scholar] [CrossRef]
- Chen, X.; Lu, Y.; Shan, M.; Zhao, H.; Lu, Z.; Lu, Y. A Mini-Review: Mechanism of Antimicrobial Action and Application of Surfactin. World J. Microbiol. Biotechnol. 2022, 38, 143. [Google Scholar] [CrossRef]
- Pinkas, D.; Fišer, R.; Kozlík, P.; Dolejšová, T.; Hryzáková, K.; Konopásek, I.; Mikušová, G. Bacillus subtilis Cardiolipin Protects Its Own Membrane against Surfactin-Induced Permeabilization. Biochim. Biophys. Acta (BBA) Biomembr. 2020, 1862, 183405. [Google Scholar] [CrossRef]
- Oftedal, L.; Myhren, L.; Jokela, J.; Gausdal, G.; Sivonen, K.; Døskeland, S.O.; Herfindal, L. The Lipopeptide Toxins Anabaenolysin A and B Target Biological Membranes in a Cholesterol-Dependent Manner. Biochim. Biophys. Acta (BBA) Biomembr. 2012, 1818, 3000–3009. [Google Scholar] [CrossRef]
- Gerbeau-Pissot, P.; Der, C.; Thomas, D.; Anca, I.-A.; Grosjean, K.; Roche, Y.; Perrier-Cornet, J.-M.; Mongrand, S.; Simon-Plas, F. Modification of Plasma Membrane Organization in Tobacco Cells Elicited by Cryptogein. Plant Physiol. 2014, 164, 273–286. [Google Scholar] [CrossRef] [PubMed]
- Deleu, M.; Crowet, J.-M.; Nasir, M.N.; Lins, L. Complementary Biophysical Tools to Investigate Lipid Specificity in the Interaction between Bioactive Molecules and the Plasma Membrane: A Review. Biochim. Biophys. Acta (BBA) Biomembr. 2014, 1838, 3171–3190. [Google Scholar] [CrossRef] [PubMed]
- Peetla, C.; Stine, A.; Labhasetwar, V. Biophysical Interactions with Model Lipid Membranes: Applications in Drug Discovery and Drug Delivery. Mol. Pharm. 2009, 6, 1264–1276. [Google Scholar] [CrossRef]
- Yeagle, P.L. Chapter 6—Laboratory Membrane Systems. In The Membranes of Cells, 3rd ed.; Yeagle, P.L., Ed.; Academic Press: Boston, MA, USA, 2016; pp. 95–114. ISBN 978-0-12-800047-2. [Google Scholar]
- Munusamy, S.; Conde, R.; Bertrand, B.; Munoz-Garay, C. Biophysical Approaches for Exploring Lipopeptide-Lipid Interactions. Biochimie 2020, 170, 173–202. [Google Scholar] [CrossRef]
- Heerklotz, H.; Seelig, J. Detergent-Like Action of the Antibiotic Peptide Surfactin on Lipid Membranes. Biophys. J. 2001, 81, 1547–1554. [Google Scholar] [CrossRef]
- Razafindralambo, H.; Dufour, S.; Paquot, M.; Deleu, M. Thermodynamic Studies of the Binding Interactions of Surfactin Analogues to Lipid Vesicles: Application of Isothermal Titration Calorimetry. J. Therm. Anal. Calorim. 2009, 95, 817–821. [Google Scholar] [CrossRef]
- Deleu, M.; Bouffioux, O.; Razafindralambo, H.; Paquot, M.; Hbid, C.; Thonart, P.; Jacques, P.; Brasseur, R. Interaction of Surfactin with Membranes: A Computational Approach. Langmuir 2003, 19, 3377–3385. [Google Scholar] [CrossRef]
- Heerklotz, H.; Wieprecht, T.; Seelig, J. Membrane Perturbation by the Lipopeptide Surfactin and Detergents as Studied by Deuterium NMR. J. Phys. Chem. B 2004, 108, 4909–4915. [Google Scholar] [CrossRef]
- Shen, H.-H.; Thomas, R.K.; Taylor, P. The Location of the Biosurfactant Surfactin in Phospholipid Bilayers Supported on Silica Using Neutron Reflectometry. Langmuir 2010, 26, 320–327. [Google Scholar] [CrossRef]
- Fan, H.Y.; Nazari, M.; Raval, G.; Khan, Z.; Patel, H.; Heerklotz, H. Utilizing Zeta Potential Measurements to Study the Effective Charge, Membrane Partitioning, and Membrane Permeation of the Lipopeptide Surfactin. Biochim. Biophys. Acta (BBA) Biomembr. 2014, 1838, 2306–2312. [Google Scholar] [CrossRef]
- Fiedler, S.; Heerklotz, H. Vesicle Leakage Reflects the Target Selectivity of Antimicrobial Lipopeptides from Bacillus subtilis. Biophys. J. 2015, 109, 2079–2089. [Google Scholar] [CrossRef] [PubMed]
- Patel, H.; Huynh, Q.; Bärlehner, D.; Heerklotz, H. Additive and Synergistic Membrane Permeabilization by Antimicrobial (Lipo)Peptides and Detergents. Biophys. J. 2014, 106, 2115–2125. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, C.; Teruel, J.A.; Aranda, F.J.; Ortiz, A. Molecular Mechanism of Membrane Permeabilization by the Peptide Antibiotic Surfactin. Biochim. Biophys. Acta (BBA) Biomembr. 2003, 1611, 91–97. [Google Scholar] [CrossRef]
- Liu, J.; Zou, A.; Mu, B. Surfactin Effect on the Physicochemical Property of PC Liposome. Colloids Surf. A Physicochem. Eng. Asp. 2010, 361, 90–95. [Google Scholar] [CrossRef]
- Kell, H.; Holzwarth, J.F.; Boettcher, C.; Heenan, R.K.; Vater, J. Physicochemical Studies of the Interaction of the Lipoheptapeptide Surfactin with Lipid Bilayers of L-α-Dimyristoyl Phosphatidylcholine. Biophys. Chem. 2007, 128, 114–124. [Google Scholar] [CrossRef]
- Deleu, M.; Lorent, J.; Lins, L.; Brasseur, R.; Braun, N.; El Kirat, K.; Nylander, T.; Dufrêne, Y.F.; Mingeot-Leclercq, M.-P. Effects of Surfactin on Membrane Models Displaying Lipid Phase Separation. Biochim. Biophys. Acta (BBA) Biomembr. 2013, 1828, 801–815. [Google Scholar] [CrossRef]
- Francius, G.; Dufour, S.; Deleu, M.; Paquot, M.; Mingeot-Leclercq, M.-P.; Dufrêne, Y.F. Nanoscale Membrane Activity of Surfactins: Influence of Geometry, Charge and Hydrophobicity. Biochim. Biophys. Acta (BBA) Biomembr. 2008, 1778, 2058–2068. [Google Scholar] [CrossRef]
- Brasseur, R.; Braun, N.; El Kirat, K.; Deleu, M.; Mingeot-Leclercq, M.-P.; Dufrêne, Y.F. The Biologically Important Surfactin Lipopeptide Induces Nanoripples in Supported Lipid Bilayers. Langmuir 2007, 23, 9769–9772. [Google Scholar] [CrossRef]
- Buchoux, S.; Lai-Kee-Him, J.; Garnier, M.; Tsan, P.; Besson, F.; Brisson, A.; Dufourc, E.J. Surfactin-Triggered Small Vesicle Formation of Negatively Charged Membranes: A Novel Membrane-Lysis Mechanism. Biophys. J. 2008, 95, 3840–3849. [Google Scholar] [CrossRef]
- Heerklotz, H.; Seelig, J. Leakage and Lysis of Lipid Membranes Induced by the Lipopeptide Surfactin. Eur. Biophys. J. 2007, 36, 305–314. [Google Scholar] [CrossRef]
- Ostroumova, O.S.; Malev, V.V.; Ilin, M.G.; Schagina, L.V. Surfactin Activity Depends on the Membrane Dipole Potential. Langmuir 2010, 26, 15092–15097. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Huang, W.; Wang, E. An Electrochemical Study on the Interaction of Surfactin with a Supported Bilayer Lipid Membrane on a Glassy Carbon Electrode. J. Electroanal. Chem. 2005, 577, 349–354. [Google Scholar] [CrossRef]
- Grau, A.; Gómez Fernández, J.C.; Peypoux, F.; Ortiz, A. A Study on the Interactions of Surfactin with Phospholipid Vesicles. Biochim. Biophys. Acta (BBA) Biomembr. 1999, 1418, 307–319. [Google Scholar] [CrossRef]
- Bouffioux, O.; Berquand, A.; Eeman, M.; Paquot, M.; Dufrêne, Y.F.; Brasseur, R.; Deleu, M. Molecular Organization of Surfactin–Phospholipid Monolayers: Effect of Phospholipid Chain Length and Polar Head. Biochim. Biophys. Acta (BBA) Biomembr. 2007, 1768, 1758–1768. [Google Scholar] [CrossRef]
- Eeman, M.; Berquand, A.; Dufrêne, Y.F.; Paquot, M.; Dufour, S.; Deleu, M. Penetration of Surfactin into Phospholipid Monolayers: Nanoscale Interfacial Organization. Langmuir 2006, 22, 11337–11345. [Google Scholar] [CrossRef]
- Shen, H.-H.; Thomas, R.K.; Penfold, J.; Fragneto, G. Destruction and Solubilization of Supported Phospholipid Bilayers on Silica by the Biosurfactant Surfactin. Langmuir 2010, 26, 7334–7342. [Google Scholar] [CrossRef]
- Watts, A. NMR of Lipids. In Encyclopedia of Biophysics; Roberts, G.C.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1727–1738. ISBN 978-3-642-16712-6. [Google Scholar]
- Maget-Dana, R.; Ptak, M. Interactions of Surfactin with Membrane Models. Biophys. J. 1995, 68, 1937–1943. [Google Scholar] [CrossRef]
- Hovakeemian, S.G.; Liu, R.; H. Gellman, S.; Heerklotz, H. Correlating Antimicrobial Activity and Model Membrane Leakage Induced by Nylon-3 Polymers and Detergents. Soft Matter 2015, 11, 6840–6851. [Google Scholar] [CrossRef]
- Uttlová, P.; Pinkas, D.; Bechyňková, O.; Fišer, R.; Svobodová, J.; Seydlová, G. Bacillus subtilis Alters the Proportion of Major Membrane Phospholipids in Response to Surfactin Exposure. Biochim. Biophys. Acta (BBA) Biomembr. 2016, 1858, 2965–2971. [Google Scholar] [CrossRef]
- Dufour, S.; Deleu, M.; Nott, K.; Wathelet, B.; Thonart, P.; Paquot, M. Hemolytic Activity of New Linear Surfactin Analogs in Relation to Their Physico-Chemical Properties. Biochim. Biophys. Acta (BBA) Gen. Subj. 2005, 1726, 87–95. [Google Scholar] [CrossRef]
- Coronel, J.R.; Aranda, F.J.; Teruel, J.A.; Marqués, A.; Manresa, Á.; Ortiz, A. Kinetic and Structural Aspects of the Permeabilization of Biological and Model Membranes by Lichenysin. Langmuir 2016, 32, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Coronel, J.R.; Marqués, A.; Manresa, Á.; Aranda, F.J.; Teruel, J.A.; Ortiz, A. Interaction of the Lipopeptide Biosurfactant Lichenysin with Phosphatidylcholine Model Membranes. Langmuir 2017, 33, 9997–10005. [Google Scholar] [CrossRef] [PubMed]
- Grangemard, I.; Wallach, J.; Maget-Dana, R.; Peypoux, F. Lichenysin. Appl. Biochem. Biotechnol. 2001, 90, 199–210. [Google Scholar] [CrossRef]
- Maget-Dana, R.; Ptak, M. Interfacial Properties of Surfactin. J. Colloid Interface Sci. 1992, 153, 285–291. [Google Scholar] [CrossRef]
- Gao, L.; Han, J.; Liu, H.; Qu, X.; Lu, Z.; Bie, X. Plipastatin and Surfactin Coproduction by Bacillus subtilis PB2-L and Their Effects on Microorganisms. Antonie Van Leeuwenhoek 2017, 110, 1007–1018. [Google Scholar] [CrossRef]
- D’Auria, L.; Deleu, M.; Dufour, S.; Mingeot-Leclercq, M.-P.; Tyteca, D. Surfactins Modulate the Lateral Organization of Fluorescent Membrane Polar Lipids: A New Tool to Study Drug:Membrane Interaction and Assessment of the Role of Cholesterol and Drug Acyl Chain Length. Biochim. Biophys. Acta (BBA) Biomembr. 2013, 1828, 2064–2073. [Google Scholar] [CrossRef]
- Wójtowicz, K.; Czogalla, A.; Trombik, T.; Łukaszewicz, M. Surfactin Cyclic Lipopeptides Change the Plasma Membrane Composition and Lateral Organization in Mammalian Cells. Biochim. Biophys. Acta (BBA) Biomembr. 2021, 1863, 183730. [Google Scholar] [CrossRef]
- Seydlová, G.; Svobodová, J. Review of Surfactin Chemical Properties and the Potential Biomedical Applications. Cent. Eur. J. Med. 2008, 3, 123–133. [Google Scholar] [CrossRef]
- Fei, D.; Liu, F.-F.; Gang, H.-Z.; Liu, J.-F.; Yang, S.-Z.; Ye, R.-Q.; Mu, B.-Z. A New Member of the Surfactin Family Produced by Bacillus subtilis with Low Toxicity on Erythrocyte. Process Biochem. 2020, 94, 164–171. [Google Scholar] [CrossRef]
- Seydlová, G.; Fišer, R.; Cabala, R.; Kozlík, P.; Svobodová, J.; Pátek, M. Surfactin Production Enhances the Level of Cardiolipin in the Cytoplasmic Membrane of Bacillus subtilis. Biochim. Biophys. Acta 2013, 1828, 2370–2378. [Google Scholar] [CrossRef]
- Jourdan, E.; Henry, G.; Duby, F.; Dommes, J.; Barthélemy, J.P.; Thonart, P.; Ongena, M. Insights into the Defense-Related Events Occurring in Plant Cells Following Perception of Surfactin-Type Lipopeptide from Bacillus subtilis. MPMI 2009, 22, 456–468. [Google Scholar] [CrossRef] [PubMed]
- Mohd Isa, M.H.; Shamsudin, N.H.; Al-Shorgani, N.K.N.; Alsharjabi, F.A.; Kalil, M.S. Evaluation of Antibacterial Potential of Biosurfactant Produced by Surfactin-Producing Bacillus Isolated from Selected Malaysian Fermented Foods. Food Biotechnol. 2020, 34, 1–24. [Google Scholar] [CrossRef]
- Bahadur, S.; Pathak, K.; Pattnaik, S.; Swain, K. Chapter 29—Biosurfactants for Optimal Delivery of Poorly Soluble Therapeutic Agents. In Green Sustainable Process for Chemical and Environmental Engineering and Science; Inamuddin, Adetunji, C.O., Ahamed, M.I., Eds.; Academic Press: Cambridge, MA, USA, 2022; pp. 543–558. ISBN 978-0-323-85146-6. [Google Scholar]
- Sarkar, S.; Saha, A.; Biswas, A.; Islam, S.K.M. Chapter 12—Inhibitory Activity of Biosurfactants against H+-K+ ATPases and Defense against Gastric Ulcers. In Green Sustainable Process for Chemical and Environmental Engineering and Science; Inamuddin, Adetunji, C.O., Ahamed, M.I., Eds.; Academic Press: Cambridge, MA, USA, 2022; pp. 235–242. ISBN 978-0-323-85146-6. [Google Scholar]
- Colpitts, C.C.; Ustinov, A.V.; Epand, R.F.; Epand, R.M.; Korshun, V.A.; Schang, L.M. 5-(Perylen-3-Yl)Ethynyl-Arabino-Uridine (AUY11), an Arabino-Based Rigid Amphipathic Fusion Inhibitor, Targets Virion Envelope Lipids To Inhibit Fusion of Influenza Virus, Hepatitis C Virus, and Other Enveloped Viruses. J. Virol. 2013, 87, 3640–3654. [Google Scholar] [CrossRef] [PubMed]
- Herzog, M.; Tiso, T.; Blank, L.M.; Winter, R. Interaction of Rhamnolipids with Model Biomembranes of Varying Complexity. Biochim. Biophys. Acta (BBA)—Biomembr. 2020, 1862, 183431. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gilliard, G.; Furlan, A.L.; Smeralda, W.; Pršić, J.; Deleu, M. Added Value of Biophysics to Study Lipid-Driven Biological Processes: The Case of Surfactins, a Class of Natural Amphiphile Molecules. Int. J. Mol. Sci. 2022, 23, 13831. https://doi.org/10.3390/ijms232213831
Gilliard G, Furlan AL, Smeralda W, Pršić J, Deleu M. Added Value of Biophysics to Study Lipid-Driven Biological Processes: The Case of Surfactins, a Class of Natural Amphiphile Molecules. International Journal of Molecular Sciences. 2022; 23(22):13831. https://doi.org/10.3390/ijms232213831
Chicago/Turabian StyleGilliard, Guillaume, Aurélien L. Furlan, Willy Smeralda, Jelena Pršić, and Magali Deleu. 2022. "Added Value of Biophysics to Study Lipid-Driven Biological Processes: The Case of Surfactins, a Class of Natural Amphiphile Molecules" International Journal of Molecular Sciences 23, no. 22: 13831. https://doi.org/10.3390/ijms232213831
APA StyleGilliard, G., Furlan, A. L., Smeralda, W., Pršić, J., & Deleu, M. (2022). Added Value of Biophysics to Study Lipid-Driven Biological Processes: The Case of Surfactins, a Class of Natural Amphiphile Molecules. International Journal of Molecular Sciences, 23(22), 13831. https://doi.org/10.3390/ijms232213831




