Biomechanical Forces Determine Fibroid Stem Cell Transformation and the Receptivity Status of the Endometrium: A Critical Appraisal
Abstract
1. Introduction
2. Results
2.1. Shape and Mechano-Adaptive Properties of the Myometrial Cells
2.2. Stimulation of Fibroid Stem Cells by Micro-Mechanical Forces
2.3. Biomechanical Features of Uterine Fibroids
2.4. Generation and Transport of Fibroid-Related Biomechanical Signals to the Endometrium
2.5. Impact of Fibroid Related Mechanotransduction on Receptivity
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Celik, O.; Acet, M.; Kucuk, T.; Haberal, E.T.; Acet, T.; Bozkurt, M.; Sahin, L.; Verit, F.F.; Caliskan, E. Surgery for benign gynecological disorders improve endometrium receptivity: A systematic review of the literature. Reprod. Sci. 2017, 24, 174–192. [Google Scholar] [CrossRef] [PubMed]
- Munro, M.G. Uterine polyps, adenomyosis, leiomyomas, and endometrial receptivity. Fertil. Steril. 2019, 111, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Celik, O.; Koc, O.; Yurci, A.; Ersahin, A.; Celik, N.; Tanilir, F.; Gurkan, N.; Ozyurt, R.; Ozcil, M.D.; Duran, B. Receptivity-based uterine fibroid surgery: An updated systematic review of the evidence. Clin. Exp. Obstet. Gynecol. 2022, 49, 114. [Google Scholar] [CrossRef]
- Fischer, C.P.; Kayisili, U.; Taylor, H.S. HOXA10 expression is decreased in endometrium of women with adenomyosis. Fertil. Steril. 2011, 95, 1133–1136. [Google Scholar] [CrossRef]
- Xiao, Y.; Li, T.; Xia, E.; Yang, X.; Sun, X.; Zhou, Y. Expression of integrin β3 and osteopontin in the eutopic endometrium of adenomyosis during the implantation window. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 170, 419–422. [Google Scholar] [CrossRef] [PubMed]
- Tremellen, K.P.; Russell, P. The distribution of immune cells and macrophages in the endometrium of women with recurrent reproductive failure. II: Adenomyosis and macrophages. J. Reprod. Immunol. 2012, 93, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Celik, O.; Unlu, C.; Otlu, B.; Celik, N.; Caliskan, E. Laparoscopic endometrioma resection increases peri-implantation endometrial HOXA-10 and HOXA-11 mRNA expression. Fertil. Steril. 2015, 104, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Celik, O.; Celik, E.; Turkcuoglu, I.; Yilmaz, E.; Ulas, M.; Simsek, Y.; Karaer, A.; Celik, N.; Aydin, N.E.; Ozerol, I. Surgical removal of endometrioma decreases the NF-kB1 (p50/105) and NF-kB p65 (Rel A) expression in the eutopic endometrium during the implantation window. Reprod. Sci. 2013, 20, 762–770. [Google Scholar] [CrossRef]
- Daftary, G.S.; Kayisli, U.; Seli, E.; Bukulmez, O.; Arici, A.; Taylor, H.S. Salpingectomy increases peri-implantation endometrial HOXA10 expression in women with hydrosalpinx. Fertil. Steril. 2007, 87, 367–372. [Google Scholar] [CrossRef]
- Ersahin, A.A.; Ersahin, S.; Gungor, N.D. Surgical removal of hydrosalpinx improves endometrium receptivity by decreasing nuclear factor-kappa B expression. Reprod. Sci. 2020, 27, 787–792. [Google Scholar] [CrossRef]
- Yoshino, O.; Nishii, O.; Osuga, Y.; Asada, H.; Okuda, S.; Orisaka, M.; Hori, M.; Fujiwara, T.; Hayashi, T. Myomectomy decreases abnormal uterine peristalsis and increases pregnancy rate. J. Minim. Invasive Gynecol. 2012, 19, 63–67. [Google Scholar] [CrossRef]
- Rogers, R.; Norian, J.; Malik, M.; Christman, G.; Abu-Asab, M.; Chen, F.; Korecki, C.; Iatridis, J.; Catherino, W.H.; Tuan, R.S. Mechanical homeostasis is altered in uterine leiomyoma. Am. J. Obstet. Gynecol. 2008, 198, 474.e1–474.e11. [Google Scholar] [CrossRef] [PubMed]
- Navarro, A.; Bariani, M.V.; Yang, Q.; Al-Hendy, A. Understanding the impact of uterine fibroids on human endometrium function. Front. Cell Dev. Biol. 2021, 9, 633180. [Google Scholar]
- Huang, S.; Ingber, D.E. Cell tension, matrix mechanics, and cancer development. Cancer Cell 2005, 8, 175–176. [Google Scholar] [CrossRef]
- Puech, P.-H.; Bongrand, P. Mechanotransduction as a major driver of cell behaviour: Mechanisms, and relevance to cell organization and future research. Open Biol. J. 2021, 11, 210256. [Google Scholar] [CrossRef]
- Alberts, B.; Johnson, A.; Lewis, J.; Morgan, D.; Raff, M.C.; Roberts, K.; Walter, P.; Wilson, J.H.; Hunt, T.; Alberts, B. Molecular Biology of the Cell, 6th ed.; Garland Science, Taylor and Francis Group: New York, NY, USA, 2015. [Google Scholar]
- Patterson, A.L.; George, J.W.; Chatterjee, A.; Carpenter, T.J.; Wolfrum, E.; Chesla, D.W.; Teixeira, J.M. Putative human myometrial and fibroid stem-like cells have mesenchymal stem cell and endometrial stromal cell properties. Hum. Reprod. 2020, 35, 44. [Google Scholar] [CrossRef] [PubMed]
- Laughlin, S.K.; Stewart, E.A. Uterine leiomyomas: Individualizing the approach to a heterogeneous condition. J. Obstet. Gynaecol. 2011, 117 Pt 1, 396–403. [Google Scholar] [CrossRef] [PubMed]
- Folkman, J.; Moscona, A. Role of cell shape in growth control. Nature 1978, 273, 345–349. [Google Scholar] [CrossRef]
- Calizo, R.C.; Bell, M.K.; Ron, A.; Hu, M.; Bhattacharya, S.; Wong, N.J.; Janssen, W.G.M.; Perumal, G.; Pederson, P.; Scarlata, S.; et al. Cell shape regulates subcellular organelle location to control early Ca2+ signal dynamics in vascular smooth muscle cells. Sci. Rep. 2020, 10, 17866. [Google Scholar] [CrossRef]
- Jorge, S.; Chang, S.; Barzilai, J.J.; Leppert, P.; Segars, J.H. Mechanical signaling in reproductive tissues: Mechanisms and importance. Reprod. Sci. 2014, 21, 1093–1107. [Google Scholar] [CrossRef]
- Schiffhauer, E.S.; Robinson, D.N. Mechanochemical signaling directs cell-shape change. Biophys. J. 2017, 112, 207–214. [Google Scholar] [CrossRef]
- Donnez, J.; Jadoul, P. What are the implications of myomas on fertility? A need for a debate? Hum. Reprod. 2002, 17, 1424–1430. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.C.; Segars, J.H. The impact and management of fibroids for fertility: An evidence-based approach. Obstet. Gynecol. Clin. N. Am. 2012, 39, 521–533. [Google Scholar] [CrossRef]
- Townsend, D.E.; Sparkes, R.; Baluda, M.C.; McClelland, G. Unicellular histogenesis of uterine leiomyomas as determined by electrophoresis of glucose-6-phosphate dehydrogenase. Am. J. Obstet. Gynecol. 1970, 107, 1168–1173. [Google Scholar] [CrossRef]
- Ligon, A.H.; Morton, C.C. Leiomyomata: Heritability and cytogenetic studies. Hum. Reprod. Update 2001, 7, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Isomursu, A.; Lerche, M.; Taskinen, M.E.; Ivaska, J.; Peuhu, E. Integrin signaling and mechanotransduction in regulation of somatic stem cells. Exp. Cell Res. 2019, 378, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Ikhena, D.E.; Bulun, S.E. Literature review on the role of uterine fibroids in endometrial function. Reprod. Sci. 2018, 25, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E. Uterine fibroids. N. Engl. J. Med. 2013, 369, 1344–1355. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Mas, A.; Diamond, M.P.; Al-Hendy, A. The mechanism and function of epigenetics in uterine leiomyoma development. Reprod. Sci. 2016, 23, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.; Ono, M.; Moravek, M.B.; Coon, J.S.; Navarro, A.; Monsivais, D.; Dyson, M.T.; Druschitz, S.A.; Malpani, S.S.; Serna, V.A. Human uterine leiomyoma stem/progenitor cells expressing CD34 and CD49b initiate tumors in vivo. J. Clin. Endocrinol. Metab. 2015, 100, E601–E606. [Google Scholar] [CrossRef]
- Prusinski Fernung, L.E.; Yang, Q.; Sakamuro, D.; Kumari, A.; Mas, A.; Al-Hendy, A. Endocrine disruptor exposure during development increases incidence of uterine fibroids by altering DNA repair in myometrial stem cells. Biol. Reprod. 2018, 99, 735–748. [Google Scholar] [CrossRef] [PubMed]
- Fernung, L.E.P.; Jones, K.; Mas, A.; Kleven, D.; Waller, J.L.; Al-Hendy, A. Expanding upon the human myometrial stem cell hypothesis and the role of race, hormones, age, and parity in a profibroid environment. Am. J. Pathol. 2018, 188, 2293–2306. [Google Scholar] [CrossRef] [PubMed]
- Andersen, J.; DyReyes, V.M.; Barbieri, R.L.; Coachman, D.M.; Miksicek, R.J. Leiomyoma primary cultures have elevated transcriptional response to estrogen compared with autologous myometrial cultures. J. Soc. Gynecol. Investig. 1995, 2, 542–551. [Google Scholar] [CrossRef]
- Ono, M.; Qiang, W.; Serna, V.; Yin, P.; Coon, J. Role of stem cells in human uterine leiomyoma growth. PLoS ONE 2012, 7, e36935. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Yin, P.; Navarro, A.; Moravek, M.B.; Coon, J.S.; Druschitz, S.A.; Serna, V.A.; Qiang, W.; Brooks, D.C.; Malpani, S.S. Paracrine activation of WNT/β-catenin pathway in uterine leiomyoma stem cells promotes tumor growth. Proc. Natl. Acad. Sci. USA 2013, 110, 17053–17058. [Google Scholar] [CrossRef]
- Hermon, T.L.; Moore, A.B.; Yu, L.; Kissling, G.E.; Castora, F.J.; Dixon, D. Estrogen receptor alpha (ERα) phospho-serine-118 is highly expressed in human uterine leiomyomas compared to matched myometrium. Virchows Arch. 2008, 453, 557–569. [Google Scholar] [CrossRef][Green Version]
- Rackow, B.W.; Taylor, H.S. Submucosal uterine leiomyomas have a global effect on molecular determinants of endometrial receptivity. Fertil. Steril. 2010, 93, 2027–2034. [Google Scholar] [CrossRef]
- Unlu, C.; Celik, O.; Celik, N.; Otlu, B. Expression of endometrial receptivity genes increase after myomectomy of intramural leiomyomas not distorting the endometrial cavity. Reprod. Sci. 2016, 23, 31–41. [Google Scholar] [CrossRef]
- Norian, J.M.; Owen, C.M.; Taboas, J.; Korecki, C.; Tuan, R.; Malik, M.; Catherino, W.H.; Segars, J.H. Characterization of tissue biomechanics and mechanical signaling in uterine leiomyoma. Matrix Biol. 2012, 31, 57–65. [Google Scholar] [CrossRef]
- Jayes, F.L.; Liu, B.; Moutos, F.T.; Kuchibhatla, M.; Guilak, F.; Leppert, P.C. Loss of stiffness in collagen-rich uterine fibroids after digestion with purified collagenase Clostridium histolyticum. Am. J. Obstet. Gynecol. 2016, 215, 596.e1–596.e8. [Google Scholar] [CrossRef]
- Holdsworth-Carson, S.J.; Zaitseva, M.; Vollenhoven, B.J.; Rogers, P.A. Clonality of smooth muscle and fibroblast cell populations isolated from human fibroid and myometrial tissues. Mol. Hum. Reprod. 2014, 20, 250–259. [Google Scholar] [CrossRef] [PubMed]
- Melica, M.E.; La Regina, G.; Parri, M.; Peired, A.J.; Romagnani, P.; Lasagni, L. Substrate stiffness modulates renal progenitor cell properties via a ROCK-mediated mechanotransduction mechanism. Cell 2019, 8, 1561. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Luo, J.-W.; Liang, T.; Lin, L.-X.; Luo, Z.-P.; Zhuang, Y.-Q.; Sun, Y.-L. Matrix stiffness regulates the differentiation of tendon-derived stem cells through FAK-ERK1/2 activation. Exp. Cell Res. 2018, 373, 62–70. [Google Scholar] [CrossRef]
- Geiger, B.; Spatz, J.; Bershadsky, A. Environmental sensing by cells through focal adhesions. Nat. Rev. Mol. Cell Biol. 2009, 23. [Google Scholar]
- Ghosh, K.; Thodeti, C.K.; Dudley, A.C.; Mammoto, A.; Klagsbrun, M.; Ingber, D.E. Tumor-derived endothelial cells exhibit aberrant Rho-mediated mechanosensing and abnormal angiogenesis in vitro. Proc. Natl. Acad. Sci. USA 2008, 105, 11305–11310. [Google Scholar] [CrossRef] [PubMed]
- Finn, C. Endocrine control of endometrial sensitivity during the induction of the decidual cell reaction in the mouse. J. Endocrinol. 1966, 36, 239–248. [Google Scholar] [CrossRef]
- Chan, L. Distribution of epithelial sodium channels (ENaC) subunits and cystic fibrosis transmembrane conductance regulator (CFTR) in murine reproductive tract. J. Membr. Biol. 2002, 185, 165–176. [Google Scholar] [CrossRef]
- Hama, K.; Aoki, J.; Inoue, A.; Endo, T.; Amano, T.; Motoki, R.; Kanai, M.; Ye, X.; Chun, J.; Matsuki, N. Embryo spacing and implantation timing are differentially regulated by LPA3-mediated lysophosphatidic acid signaling in mice. Biol. Reprod. 2007, 77, 954–959. [Google Scholar] [CrossRef]
- Payson, M.; Malik, M.; Morris, S.S.-n.; Segars, J.H.; Chason, R.; Catherino, W.H. Activating transcription factor 3 gene expression suggests that tissue stress plays a role in leiomyoma development. Fertil. Steril. 2009, 92, 748–755. [Google Scholar] [CrossRef][Green Version]
- Baranov, V.S.; Osinovskaya, N.S.; Yarmolinskaya, M.I. Pathogenomics of uterine fibroids development. Int. J. Mol. Sci. 2019, 20, 6151. [Google Scholar] [CrossRef]
- Machado-Lopez, A.; Simón, C.; Mas, A. Molecular and cellular insights into the development of uterine fibroids. Int. J. Mol. Sci. 2021, 22, 8483. [Google Scholar] [CrossRef] [PubMed]
- Mäkinen, N.; Mehine, M.; Tolvanen, J.; Kaasinen, E.; Li, Y.; Lehtonen, H.J.; Gentile, M.; Yan, J.; Enge, M.; Taipale, M. MED12, the mediator complex subunit 12 gene, is mutated at high frequency in uterine leiomyomas. Science 2011, 334, 252–255. [Google Scholar] [CrossRef]
- Ptacek, T.; Song, C.; Walker, C.; Sell, S. Physical mapping of distinct 7q22 deletions in uterine leiomyoma and analysis of a recently annotated 7q22 candidate gene. Cancer Genet. Cytogenet. 2007, 174, 116–120. [Google Scholar] [CrossRef] [PubMed]
- Jayes, F.L.; Liu, B.; Feng, L.; Aviles-Espinoza, N.; Leikin, S.; Leppert, P.C. Evidence of biomechanical and collagen heterogeneity in uterine fibroids. PLoS ONE 2019, 14, e0215646. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, S.; Miyamoto, S.; Brown, J.H. Initiation and Transduction of Stretch-induced RhoA and Rac1 Activation through Caveolae. cytosikeletal regulation of erk translocation. J. Biol. Chem. 2003, 278, 31111–31117. [Google Scholar] [CrossRef] [PubMed]
- Taglieri, D.M.; Johnson, K.R.; Burmeister, B.T.; Monasky, M.M.; Spindler, M.J.; DeSantiago, J.; Banach, K.; Conklin, B.R.; Carnegie, G.K. The C-terminus of the long AKAP13 isoform (AKAP-Lbc) is critical for development of compensatory cardiac hypertrophy. J. Mol. Cell. Cardiol. 2014, 66, 27–40. [Google Scholar] [CrossRef]
- Wettschureck, N.; Offermanns, S. Rho/Rho-kinase mediated signaling in physiology and pathophysiology. J. Mol. Med. 2002, 80, 629–638. [Google Scholar] [CrossRef]
- Schmidt, A.; Durgan, J.; Magalhaes, A.; Hall, A. Rho GTPases regulate PRK2/PKN2 to control entry into mitosis and exit from cytokinesis. EMBO J. 2007, 26, 1624–1636. [Google Scholar] [CrossRef]
- Deligdish, L.; Loewenthal, M. Endometrial changes associated with myomata of the uterus. J. Clin. Pathol. 1970, 23, 676–680. [Google Scholar] [CrossRef]
- Graff-Radford, J.; Jones, D.T.; Pruthi, R.K.; Flemming, K.D. A neurological complication of a uterine fibroid. Neurocrit. Care 2013, 18, 93–95. [Google Scholar] [CrossRef]
- Hynes, R.O. Integrins: Bidirectional, allosteric signaling machines. Cell 2002, 110, 673–687. [Google Scholar] [CrossRef]
- Del Rio, A.; Perez-Jimenez, R.; Liu, R.; Roca-Cusachs, P.; Fernandez, J.M.; Sheetz, M.P. Stretching single talin rod molecules activates vinculin binding. Science 2009, 323, 638–641. [Google Scholar] [CrossRef] [PubMed]
- Kulp, J.L.; Mamillapalli, R.; Taylor, H.S. Aberrant HOXA10 methylation in patients with common gynecologic disorders: Implications for reproductive outcomes. Reprod. Sci. 2016, 23, 455–463. [Google Scholar] [CrossRef]
- Makker, A.; Goel, M.M.; Nigam, D.; Bhatia, V.; Mahdi, A.A.; Das, V.; Pandey, A. Endometrial expression of homeobox genes and cell adhesion molecules in infertile women with intramural fibroids during window of implantation. Reprod. Sci. 2017, 24, 435–444. [Google Scholar] [CrossRef] [PubMed]
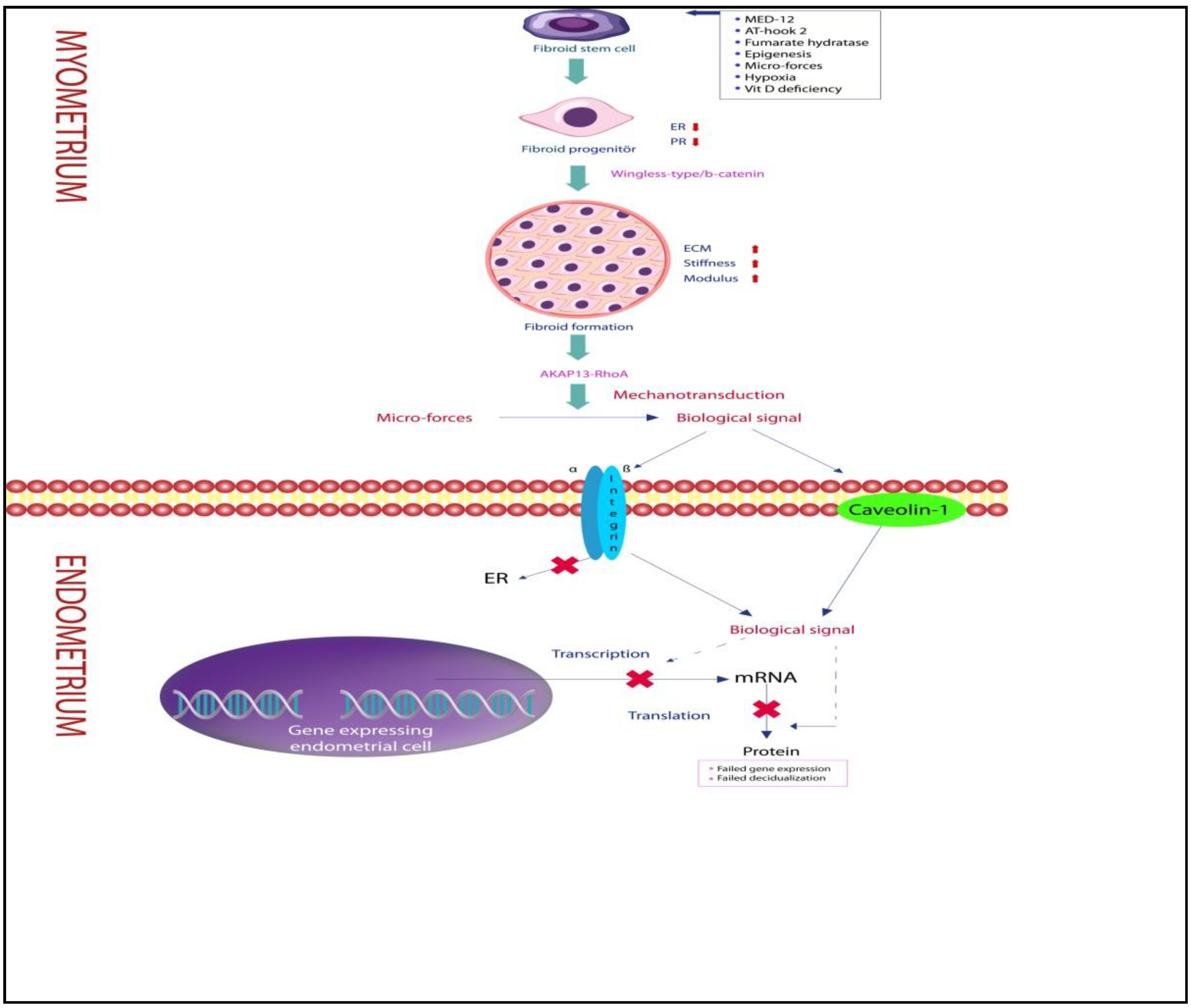

| Signal Molecules/Mechanotransducers | Main Mechanisms |
|---|---|
| ECM | Changes in ECM structure play a role in fibroid formation and growth. The ECM regulates the fibroid cell’s ability to sense and respond to changing microforces. Mechanical signals are transmitted as biological signals from the ECM scaffold via transmembrane receptors to the internal cytoskeleton via integrins, caveolins, and cadherins. Mechanical signals originating from the ECM are converted into biological signals via receptors sensitive to stretching, shear stress, and hydrostatic and osmotic pressures. |
| Collagen and glycosaminoglycan | Both generate peak stress mediated by the pseudo-dynamic modulus, enabling the mechanical stimulus to be converted into a biological signal stimulus. |
| AKAP13 | AKAP13 is involved in signal generation and transmission through direct communication with the actin cytoskeleton by regulating actin filament nucleation. AKAP13 is extensively expressed in fibroid cells and is involved in the transmission of osmotic signals. RhoA and MAPK are the main targets of AKAP13. |
| RhoA | RhoA is a molecule belonging to the Rho family of GTPases that switch between non-functional GDP and active GTP. Rho GTPases provide signal transduction by increasing the tension in the scaffold filaments of neighboring cells. Basal levels of active RhoA are equally expressed in myometrial and leiomyoma cells. |
| AKAP13-RhoA complex | The AKAP13-RhoA complex first stimulates stress-activated kinases followed by actin fibers. Contraction of myofilaments stimulates signal transmission in the endo-myometrial region. The AKAP13-RhoA complex induces the transmission of mechanical signals to the endometrium via estrogen receptors. |
| Mitogen-activated protein kinases, phosphatidylinositol-3 kinase, Janus kinase, and small GTPases. | They are pathway molecules responsible for possible mechanotransduction between fibroids and endometrium. |
| Stiffness and Modulus | Myometrium exhibits elastic modulus, whereas fibroids have cartilage-like modulus due to their dense matrix content. The stiffness of the myometrium is about 5 kPa, while the stiffness of the myometrium reaches about 15 kPa. A small fibroid with a large amount of ECM may have a higher stiffness than a large fibroid with a low amount of ECM. Therefore, signals due to an intramural myoma that is not in direct contact with the endometrium but with a stiffness above 15 kPa may spread to the endometrium and impair receptivity. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Celik, O.; Celik, N.; Gungor, N.D.; Celik, S.; Arslan, L.; Morciano, A.; Tinelli, A. Biomechanical Forces Determine Fibroid Stem Cell Transformation and the Receptivity Status of the Endometrium: A Critical Appraisal. Int. J. Mol. Sci. 2022, 23, 14201. https://doi.org/10.3390/ijms232214201
Celik O, Celik N, Gungor ND, Celik S, Arslan L, Morciano A, Tinelli A. Biomechanical Forces Determine Fibroid Stem Cell Transformation and the Receptivity Status of the Endometrium: A Critical Appraisal. International Journal of Molecular Sciences. 2022; 23(22):14201. https://doi.org/10.3390/ijms232214201
Chicago/Turabian StyleCelik, Onder, Nilufer Celik, Nur Dokuzeylul Gungor, Sudenaz Celik, Liya Arslan, Andrea Morciano, and Andrea Tinelli. 2022. "Biomechanical Forces Determine Fibroid Stem Cell Transformation and the Receptivity Status of the Endometrium: A Critical Appraisal" International Journal of Molecular Sciences 23, no. 22: 14201. https://doi.org/10.3390/ijms232214201
APA StyleCelik, O., Celik, N., Gungor, N. D., Celik, S., Arslan, L., Morciano, A., & Tinelli, A. (2022). Biomechanical Forces Determine Fibroid Stem Cell Transformation and the Receptivity Status of the Endometrium: A Critical Appraisal. International Journal of Molecular Sciences, 23(22), 14201. https://doi.org/10.3390/ijms232214201








