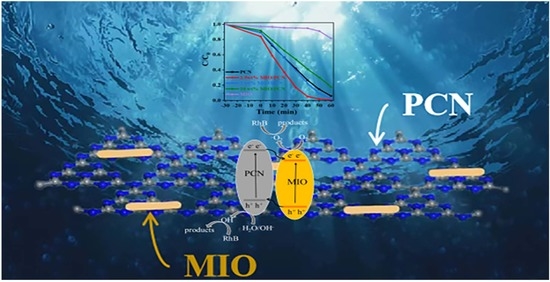
Rhombohedral/Cubic In2O3 Phase Junction Hybridized with Polymeric Carbon Nitride for Photodegradation of Organic Pollutants
Abstract
1. Introduction
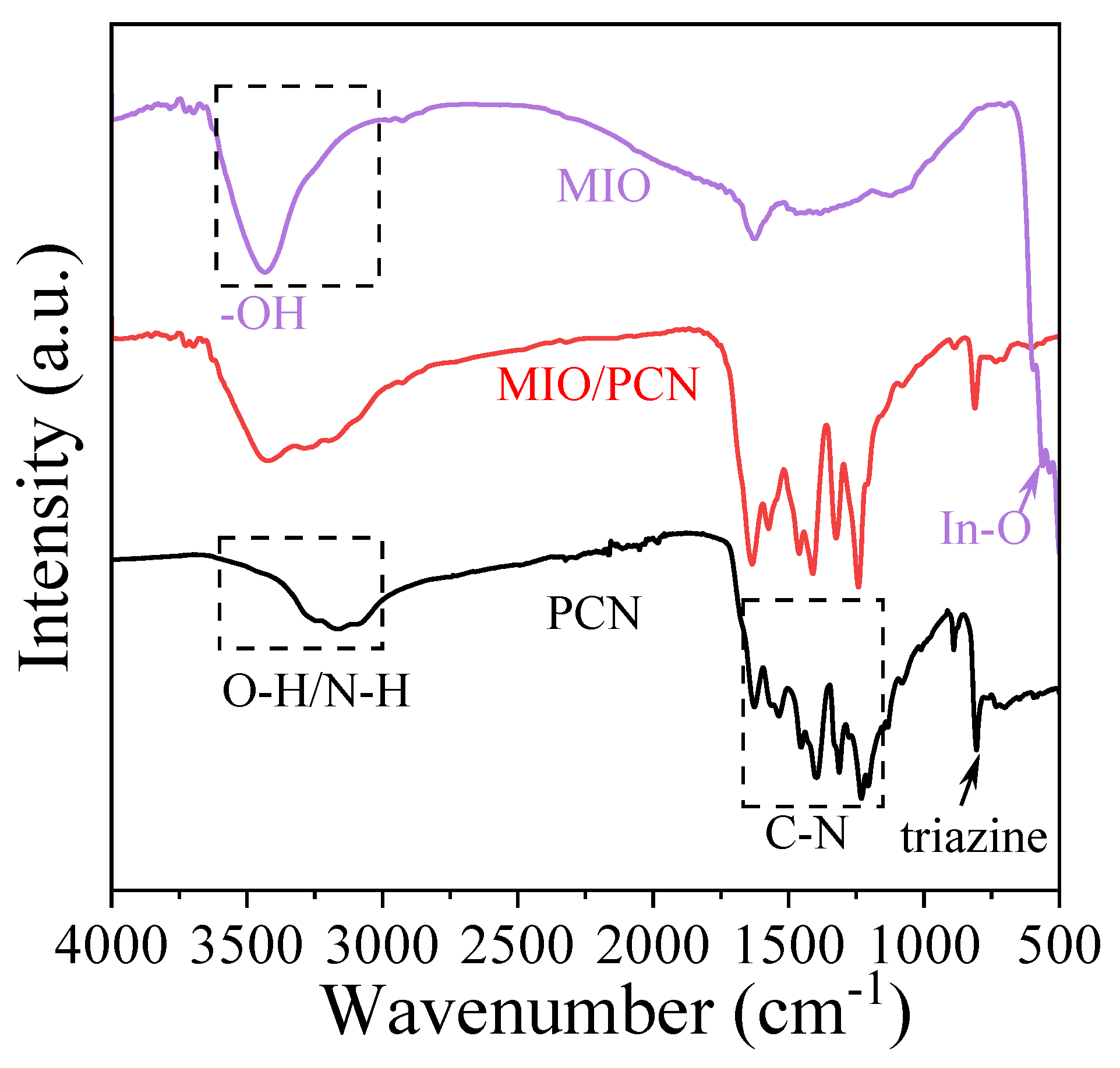
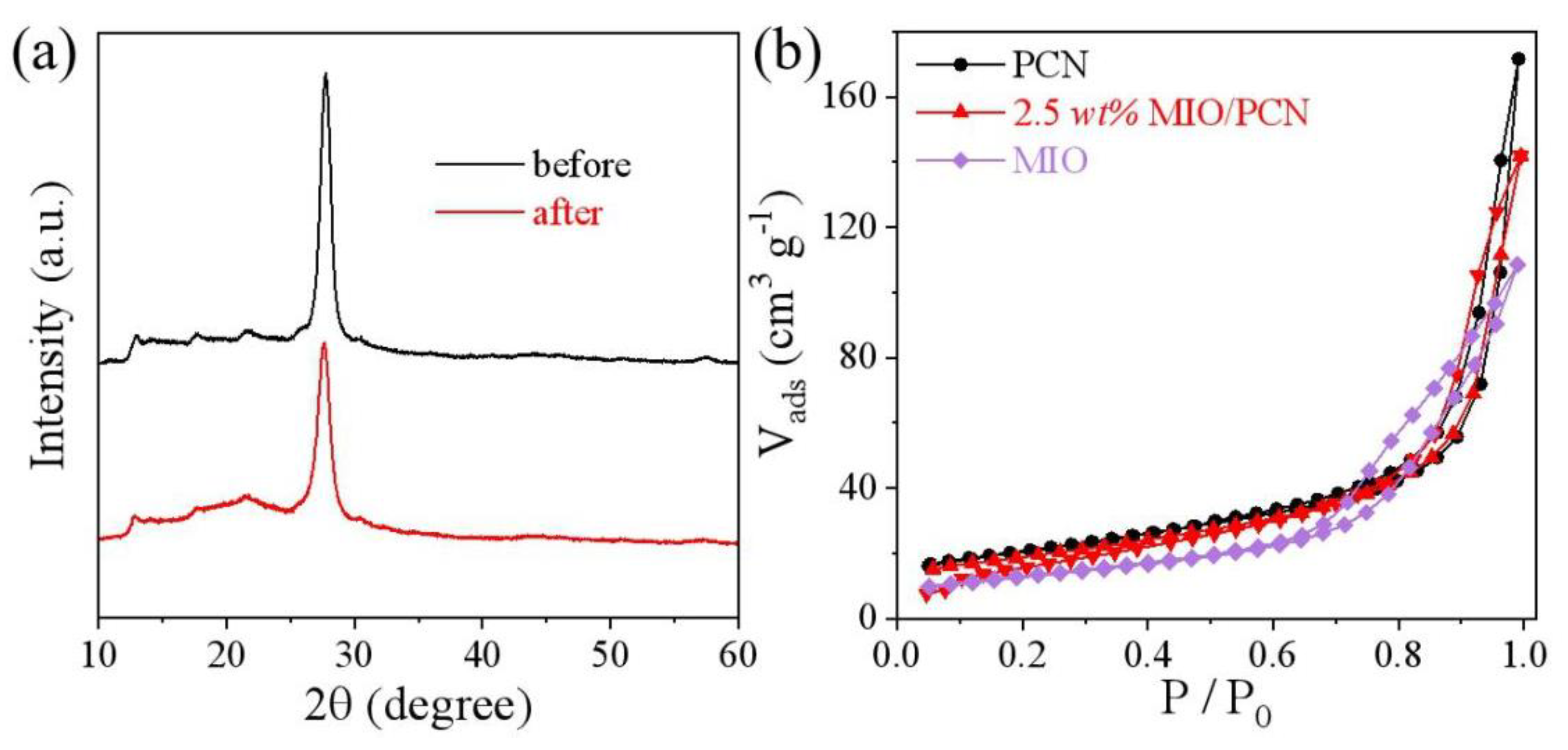
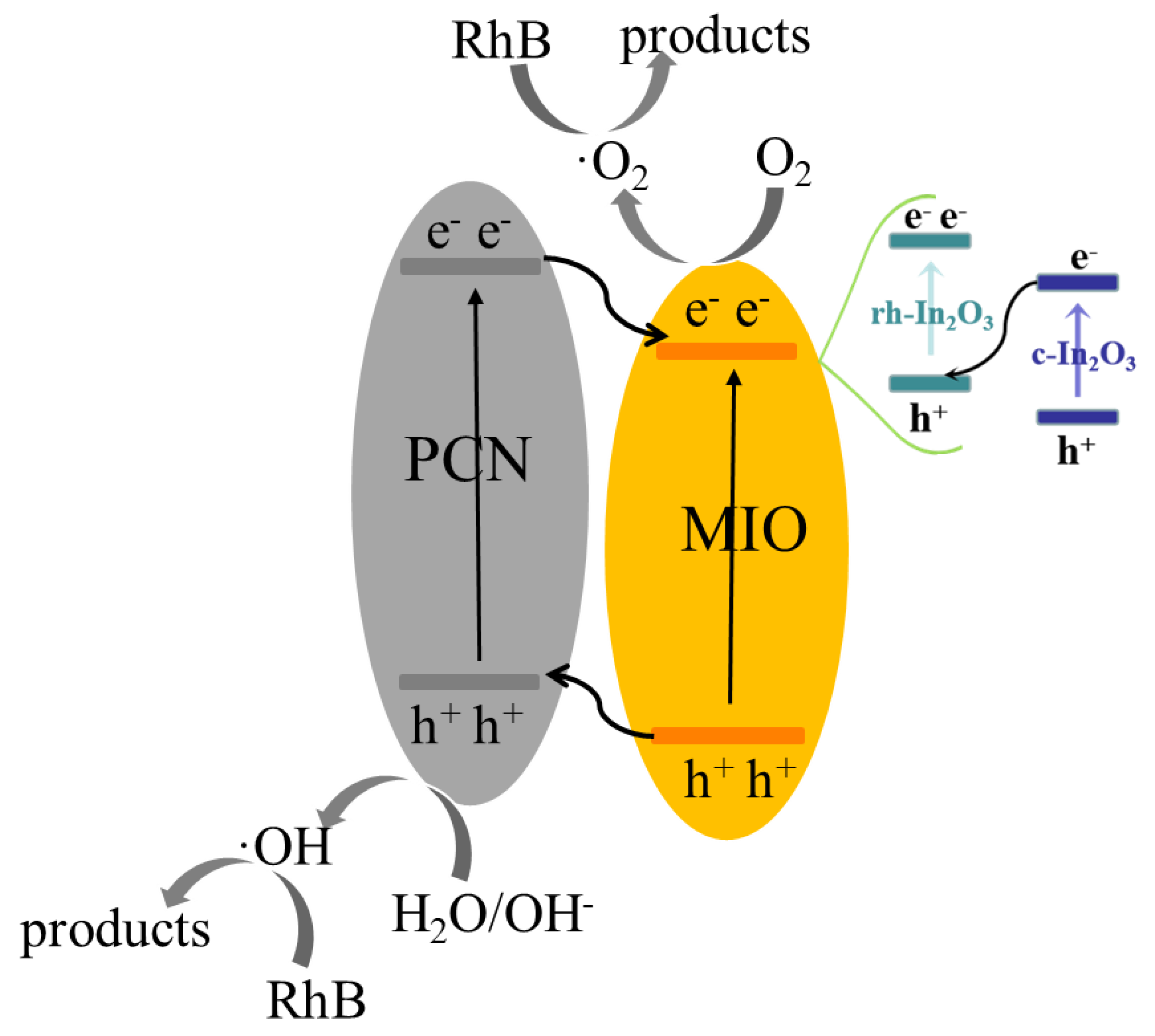
2. Results and Discussion
3. Materials and Methods
3.1. Preparation of MIL-68(In)-NH2
3.2. Preparation of MIO/PCN and PCN
3.3. Characterization
3.4. Photocatalytic Rhodamine b Degradation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, L.; Song, Y.; Li, Y.; Yin, Y.; Cai, Y. Role of light in methylmercury photodegradation: From irradiation to absorption in the presence of organic ligands. Sci. Total Environ. 2022, 848, 157550. [Google Scholar] [CrossRef] [PubMed]
- Smulek, W.; Bielan, Z.; Pacholak, A.; Zdarta, A.; Zgola-Grzeskowiak, A.; Zielinska-Jurek, A.; Kaczorek, E. Nitrofurazone Removal from Water Enhanced by Coupling Photocatalysis and Biodegradation. Int. J. Mol. Sci. 2021, 22, 2186. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Huang, W.-Y.; Zhou, Q.; Chen, X.-R.; Yang, K.; Li, D.; Dionysiou, D.D. Ag-decorated 3D flower-like Bi2MoO6/rGO with boosted photocatalytic performance for removal of organic pollutants. Rare Met. 2020, 40, 1086–1098. [Google Scholar] [CrossRef]
- Vinothkumar, K.; Jyothi, M.; Lavanya, C.; Sakar, M.; Valiyaveettil, S.; Balakrishna, R.G. Strongly co-ordinated MOF-PSF matrix for selective adsorption, separation and photodegradation of dyes. Chem. Eng. J. 2022, 428, 132561. [Google Scholar] [CrossRef]
- Zeng, R.; Lian, K.; Su, B.; Lu, L.; Lin, J.; Tang, D.; Lin, S.; Wang, X. Versatile Synthesis of Hollow Metal Sulfides via Reverse Cation Exchange Reactions for Photocatalytic CO2 Reduction. Angew. Chem. Int. Ed. Engl. 2021, 60, 25055–25062. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhang, Y.; Wang, X.; Wang, Z.; Huang, L.; Wu, H.; Ren, J.; Gu, C.; Chen, Z. Enhanced photodegradation of tylosin in the presence of natural montmorillonite: Synergistic effects of adsorption and surface hydroxyl radicals. Sci. Total Environ. 2022, 855, 158750. [Google Scholar] [CrossRef]
- Yu, Z.; Yang, K.; Yu, C.; Lu, K.; Huang, W.; Xu, L.; Zou, L.; Wang, S.; Chen, Z.; Hu, J.; et al. Steering Unit Cell Dipole and Internal Electric Field by Highly Dispersed Er atoms Embedded into NiO for Efficient CO2 Photoreduction. Adv. Funct. Mater. 2022, 32, 2111999. [Google Scholar] [CrossRef]
- Divakaran, K.; Baishnisha, A.; Balakumar, V.; Perumal, K.N.; Meenakshi, C.; Kannan, R.S. Photocatalytic degradation of tetracycline under visible light using TiO2@sulfur doped carbon nitride nanocomposite synthesized via in-situ method. J. Environ. Chem. Eng. 2021, 9, 105560. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, J.; Xia, Y.; Xun, M.; Chen, H.; Liu, X.; Yin, X. Metal-Free Carbon Quantum Dots Implant Graphitic Carbon Nitride: Enhanced Photocatalytic Dye Wastewater Purification with Simultaneous Hydrogen Production. Int. J. Mol. Sci. 2020, 21, 1054. [Google Scholar] [CrossRef]
- Li, S.; Cai, M.; Liu, Y.; Zhang, J.; Wang, C.; Zang, S.; Li, Y.; Zhang, P.; Li, X. In-situ constructing C3N5 nanosheets/Bi2WO6 nanodots S-scheme heterojunction with enhanced structural defects for efficiently photocatalytic removal of tetracycline and Cr(VI). Inorg. Chem. Front. 2022, 9, 2479–2497. [Google Scholar] [CrossRef]
- Lin, L.; Lin, Z.; Zhang, J.; Cai, X.; Wang, X. Molecular-level insights on the reactive facet of carbon nitride single crystals photocatalysing overall water splitting. Nat. Catal. 2020, 3, 649–655. [Google Scholar] [CrossRef]
- Pei, Z.; Gu, J.; Wang, Y.; Tang, Z.; Liu, Z.; Huang, Y.; Zhao, J.; Chen, Z.; Zhi, C. Component matters: Paving the roadmap toward enhanced electrocatalytic performance of graphitic C3N4-based catalysts via atomic tuning. ACS Nano 2017, 11, 6004–6014. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Hou, Y.; Fu, X.; Wang, X. Semiconducting Polymers for Oxygen Evolution Reaction under Light Illumination. Chem. Rev. 2022, 122, 4204–4256. [Google Scholar] [CrossRef]
- Kumar, A.; Raizada, P.; Thakur, V.; Khan, A.A.P.; Singh, N.; Singh, P. An overview on polymeric carbon nitride assisted photocatalytic CO2 reduction: Strategically manoeuvring solar to fuel conversion efficiency. Chem. Eng. Sci. 2021, 230, 116219. [Google Scholar] [CrossRef]
- Zhan, H.; Zhou, Q.; Li, M.; Zhou, R.; Mao, Y.; Wang, P. Photocatalytic O2 activation and reactive oxygen species evolution by surface B-N bond for organic pollutants degradation. Appl. Catal. B Environ. 2022, 310, 121329. [Google Scholar] [CrossRef]
- Di, G.; Zhu, Z.; Zhang, H.; Zhu, J.; Qiu, Y.; Yin, D.; Kuppers, S. Visible-light degradation of sulfonamides by Z-scheme ZnO/g-C3N4 heterojunctions with amorphous Fe2O3 as electron mediator. J. Colloid. Interface Sci. 2019, 538, 256–266. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Maeda, K.; Thomas, A.; Takanabe, K.; Antonietti, M. A metal-free polymeric photocatalyst for hydrogen production from water under visible light. Nat. Mater. 2009, 8, 76–80. [Google Scholar] [CrossRef]
- Gao, S.; Liu, Y.; Zhu, J.; Wang, Y.; Han, X.; Xia, X.; Zhao, X. The synthesis of novel FeS2/g-C3N4 nanocomposites for the removal of tetracycline under visible-light irradiation. Environ. Sci. Water Res. Technol. 2021, 7, 1430–1442. [Google Scholar] [CrossRef]
- Wei, X.; Wang, X.; Pu, Y.; Liu, A.; Chen, C.; Zou, W.; Zheng, Y.; Huang, J.; Zhang, Y.; Yang, Y.; et al. Facile ball-milling synthesis of CeO2/g-C3N4 Z-scheme heterojunction for synergistic adsorption and photodegradation of methylene blue: Characteristics, kinetics, models, and mechanisms. Chem. Eng. J. 2021, 420, 127719. [Google Scholar] [CrossRef]
- Zhao, H.; Jian, L.; Gong, M.; Jing, M.; Li, H.; Mao, Q.; Lu, T.; Guo, Y.; Ji, R.; Chi, W.; et al. Transition-Metal-Based Cocatalysts for Photocatalytic Water Splitting. Small Struct. 2022, 3, 2100229. [Google Scholar] [CrossRef]
- Shao, B.; Wang, J.; Liu, Z.; Zeng, G.; Tang, L.; Liang, Q.; He, Q.; Wu, T.; Liu, Y.; Yuan, X. Ti3C2Tx MXene decorated black phosphorus nanosheets with improved visible-light photocatalytic activity: Experimental and theoretical studies. J. Mater. Chem. A 2020, 8, 5171–5185. [Google Scholar] [CrossRef]
- Shahiduzzaman, M.; Visal, S.; Kuniyoshi, M.; Kaneko, T.; Umezu, S.; Katsumata, T.; Iwamori, S.; Kakihana, M.; Taima, T.; Isomura, M.; et al. Low-temperature-processed brookite-based TiO2 heterophase junction enhances performance of planar perovskite solar cells. Nano Lett. 2018, 19, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, X.; Bai, Y.; Li, C.; Gao, Y.; Li, R.; Li, C. Boosting photocatalytic water splitting by tuning built-in electric field at phase junction. J. Mater. Chem. A 2019, 7, 10264–10272. [Google Scholar] [CrossRef]
- Gao, Y.; Zhu, J.; An, H.; Yan, P.; Huang, B.; Chen, R.; Fan, F.; Li, C. Directly Probing Charge Separation at Interface of TiO2 Phase Junction. J. Phys. Chem. Lett. 2017, 8, 1419–1423. [Google Scholar] [CrossRef]
- Zhang, J.; Xu, Q.; Feng, Z.; Li, M.; Li, C. Importance of the relationship between surface phases and photocatalytic activity of TiO2. Angew. Chem. 2008, 120, 1790–1793. [Google Scholar] [CrossRef]
- Ai, Z.; Zhao, G.; Zhong, Y.; Shao, Y.; Huang, B.; Wu, Y.; Hao, X. Phase junction CdS: High efficient and stable photocatalyst for hydrogen generation. Appl. Catal. B Environ. 2018, 221, 179–186. [Google Scholar] [CrossRef]
- Liu, F.; Shi, R.; Wang, Z.; Weng, Y.; Che, C.M.; Chen, Y. Direct Z-Scheme Hetero-phase Junction of Black/Red Phosphorus for Photocatalytic Water Splitting. Angew. Chem. Int. Ed. Engl. 2019, 58, 11791–11795. [Google Scholar] [CrossRef]
- Han, L.; Jing, F.; Luo, X.; Zhong, Y.-L.; Wang, K.; Zang, S.-H.; Teng, D.-H.; Liu, Y.; Chen, J.; Yang, C.; et al. Environment friendly and remarkably efficient photocatalytic hydrogen evolution based on metal organic framework derived hexagonal/cubic In2O3 phase-junction. Appl. Catal. B Environ. 2021, 282, 119602. [Google Scholar] [CrossRef]
- Friedmann, D.; Caruso, R. Indium Oxides and Related Indium-based Photocatalysts for Water Treatment: Materials Studied, Photocatalytic Performance, and Special Highlights. Sol. RRL 2021, 5, 2100086. [Google Scholar] [CrossRef]
- Feng, Y.; Yan, T.; Wu, T.; Zhang, N.; Yang, Q.; Sun, M.; Yan, L.; Du, B.; Wei, Q. A label-free photoelectrochemical aptasensing platform base on plasmon Au coupling with MOF-derived In2O3@g-C3N4 nanoarchitectures for tetracycline detection. Sens. Actuators B Chem. 2019, 298, 12817. [Google Scholar] [CrossRef]
- Jin, L.-N.; Liu, Q.; Sun, W.-Y. Size-controlled indium(iii)-benzenedicarboxylate hexagonal rods and their transformation to In2O3 hollow structures. Cryst. Eng. Comm. 2013, 15, 4775–4784. [Google Scholar] [CrossRef]
- Wang, S.; Guan, B.Y.; Lou, X.W.D. Construction of ZnIn2S4-In2O3 Hierarchical Tubular Heterostructures for Efficient CO2 Photoreduction. J. Am. Chem. Soc. 2018, 140, 5037–5040. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Sun, L.; Zhan, W.; Li, Y.; Wang, X.; Han, X. Engineering an effective noble-metal-free photocatalyst for hydrogen evolution: Hollow hexagonal porous micro-rods assembled from In2O3@carbon core-shell nanoparticles. J. Mater. Chem. A 2018, 6, 15747–15754. [Google Scholar] [CrossRef]
- Sun, D.; Wang, W.; Zhang, N.; Liu, C.; Li, X.; Zhou, J.; Ruan, S. G-C3N4/In2O3 composite for effective formaldehyde detection. Sens. Actuators B Chem. 2022, 358, 131414. [Google Scholar] [CrossRef]
- Xu, M.; Zhao, X.; Jiang, H.; Chen, S.; Huo, P. MOFs-derived C-In2O3/g-C3N4 heterojunction for enhanced photoreduction CO2. J. Environ. Chem. Eng. 2021, 9, 106469. [Google Scholar] [CrossRef]
- Jin, X.; Guan, Q.; Tian, T.; Li, H.; Han, Y.; Hao, F.; Cui, Y.; Li, W.; Zhu, Y.; Zhang, Y. In2O3/boron doped g-C3N4 heterojunction catalysts with remarkably enhanced visible-light photocatalytic efficiencies. Appl. Surf. Sci. 2020, 504, 144241. [Google Scholar] [CrossRef]
- Uddin, A.; Rauf, A.; Wu, T.; Khan, R.; Yu, Y.; Tan, L.; Jiang, F.; Chen, H. In2O3/oxygen doped g-C3N4 towards photocatalytic BPA degradation: Balance of oxygen between metal oxides and doped g-C3N4. J. Colloid. Interface Sci. 2021, 602, 261–273. [Google Scholar] [CrossRef]
- Pan, Z.; Zhao, M.; Zhuzhang, H.; Zhang, G.; Anpo, M.; Wang, X. Gradient Zn-Doped Poly Heptazine Imides Integrated with a van der Waals Homojunction Boosting Visible Light-Driven Water Oxidation Activities. ACS Catal. 2021, 11, 13463–13471. [Google Scholar] [CrossRef]
- Vinoth, S.; Rajaitha, P.; Venkadesh, A.; Devi, K.S.S.; Radhakrishnan, S.; Pandikumar, A. Nickel sulfide-incorporated sulfur-doped graphitic carbon nitride nanohybrid interface for non-enzymatic electrochemical sensing of glucose. Nanoscale Adv. 2020, 2, 4242–4250. [Google Scholar] [CrossRef]
- Zang, S.; Cai, X.; Chen, M.; Teng, D.; Jing, F.; Leng, Z.; Zhou, Y.; Lin, F. Tunable Carrier Transfer of Polymeric Carbon Nitride with Charge-Conducting CoV2O6·2H2O for Photocatalytic O2 Evolution. Nanomaterials 2022, 12, 1931. [Google Scholar] [CrossRef]
- Cai, M.; Li, R.; Wang, F.; Guo, X.; Bai, Q.; Sun, L.; Han, X. Architecture of designed hollow indium oxide microspheres assembled by porous nanosheets with high gas sensing capacity. J. Alloys Compd. 2017, 729, 222–230. [Google Scholar] [CrossRef]
- Zhang, K.; Zhou, M.; Yang, K.; Yu, C.; Mu, P.; Yu, Z.; Lu, K.; Huang, W.; Dai, W. Photocatalytic H2O2 production and removal of Cr (VI) via a novel Lu3NbO7: Yb, Ho/CQDs/AgInS2/In2S3 heterostructure with broad spectral response. J. Hazard. Mater. 2022, 423, 127172. [Google Scholar] [CrossRef] [PubMed]
- Sprynskyy, M.; Szczyglewska, P.; Wojtczak, I.; Nowak, I.; Witkowski, A.; Buszewski, B.; Feliczak-Guzik, A. Diatom Biosilica Doped with Palladium(II) Chloride Nanoparticles as New Efficient Photocatalysts for Methyl Orange Degradation. Int. J. Mol. Sci. 2021, 22, 6734. [Google Scholar] [CrossRef]
- Chen, M.; Xu, J.; Tang, R.; Yuan, S.; Min, Y.; Xu, Q.; Shi, P. Roles of microplastic-derived dissolved organic matter on the photodegradation of organic micropollutants. J. Hazard. Mater. 2022, 440, 129784. [Google Scholar] [CrossRef] [PubMed]
- Adeleke, J.; Theivasanthi, T.; Thiruppathi, M.; Akomolafe, T.; Alabi, A. Photocatalytic degradation of methylene blue by ZnO/NiFe2O4 nanoparticles. Appl. Surf. Sci. 2018, 455, 195–200. [Google Scholar] [CrossRef]
- Yadav, R.; Kumar, R.; Aliyan, A.; Dobal, P.S.; Biradar, S.; Vajtai, R.; Singh, D.P.; Martí, A.A.; Ajayan, P.M. Facile synthesis of highly fluorescent free-standing films comprising graphitic carbon nitride (g-C3N4) nanolayers. New J. Chem. 2020, 44, 2644–2651. [Google Scholar] [CrossRef]
- Li, S.; Cai, M.; Liu, Y.; Wang, C.; Yan, R.; Chen, X. Constructing Cd0.5Zn0.5S/Bi2WO6 S-scheme heterojunction for boosted photocatalytic antibiotic oxidation and Cr (VI) reduction. Adv. Powder Mater. 2023, 2, 100073. [Google Scholar] [CrossRef]
- Fan, G.; Luo, J.; Guo, L.; Lin, R.; Zheng, X.; Snyder, S.A. Doping Ag/AgCl in zeolitic imidazolate framework-8 (ZIF-8) to enhance the performance of photodegradation of methylene blue. Chemosphere 2018, 209, 44–52. [Google Scholar] [CrossRef]
- Lin, B.; Chen, H.; Zhou, Y.; Luo, X.; Tian, D.; Yan, X.; Duan, R.; Di, J.; Kang, L.; Zhou, A.; et al. 2D/2D atomic double-layer WS2/Nb2O5 shell/core nanosheets with ultrafast interfacial charge transfer for boosting photocatalytic H2 evolution. Chin. Chem. Lett. 2021, 32, 3128–3132. [Google Scholar] [CrossRef]
- Yan, L.; Gao, H.; Chen, Y. Na-Doped Graphitic Carbon Nitride for Removal of Aqueous Contaminants via Adsorption and Photodegradation. ACS Appl. Nano Mater. 2021, 4, 7746–7757. [Google Scholar] [CrossRef]
- Mu, P.; Zhou, M.; Yang, K.; Chen, X.; Yu, Z.; Lu, K.; Huang, W.; Yu, C.; Dai, W. Cd0.5Zn0.5S/CoWO4 Nanohybrids with a Twinning Homojunction and an Interfacial S-Scheme Heterojunction for Efficient Visible-Light-Induced Photocatalytic CO2 Reduction. Inorg. Chem. 2021, 60, 14854–14865. [Google Scholar] [CrossRef] [PubMed]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, X.; Wang, Y.; Tang, S.; Mo, L.; Leng, Z.; Zang, Y.; Jing, F.; Zang, S. Rhombohedral/Cubic In2O3 Phase Junction Hybridized with Polymeric Carbon Nitride for Photodegradation of Organic Pollutants. Int. J. Mol. Sci. 2022, 23, 14293. https://doi.org/10.3390/ijms232214293
Cai X, Wang Y, Tang S, Mo L, Leng Z, Zang Y, Jing F, Zang S. Rhombohedral/Cubic In2O3 Phase Junction Hybridized with Polymeric Carbon Nitride for Photodegradation of Organic Pollutants. International Journal of Molecular Sciences. 2022; 23(22):14293. https://doi.org/10.3390/ijms232214293
Chicago/Turabian StyleCai, Xiaorong, Yaning Wang, Shuting Tang, Liuye Mo, Zhe Leng, Yixian Zang, Fei Jing, and Shaohong Zang. 2022. "Rhombohedral/Cubic In2O3 Phase Junction Hybridized with Polymeric Carbon Nitride for Photodegradation of Organic Pollutants" International Journal of Molecular Sciences 23, no. 22: 14293. https://doi.org/10.3390/ijms232214293
APA StyleCai, X., Wang, Y., Tang, S., Mo, L., Leng, Z., Zang, Y., Jing, F., & Zang, S. (2022). Rhombohedral/Cubic In2O3 Phase Junction Hybridized with Polymeric Carbon Nitride for Photodegradation of Organic Pollutants. International Journal of Molecular Sciences, 23(22), 14293. https://doi.org/10.3390/ijms232214293