Excess BAFF Alters NR4As Expression Levels and Breg Function of Human Precursor-like Marginal Zone B-Cells in the Context of HIV-1 Infection
Abstract
:1. Introduction
2. Results
2.1. Socio-Demographic and Data Characteristics of the Cohorts Used in This Study
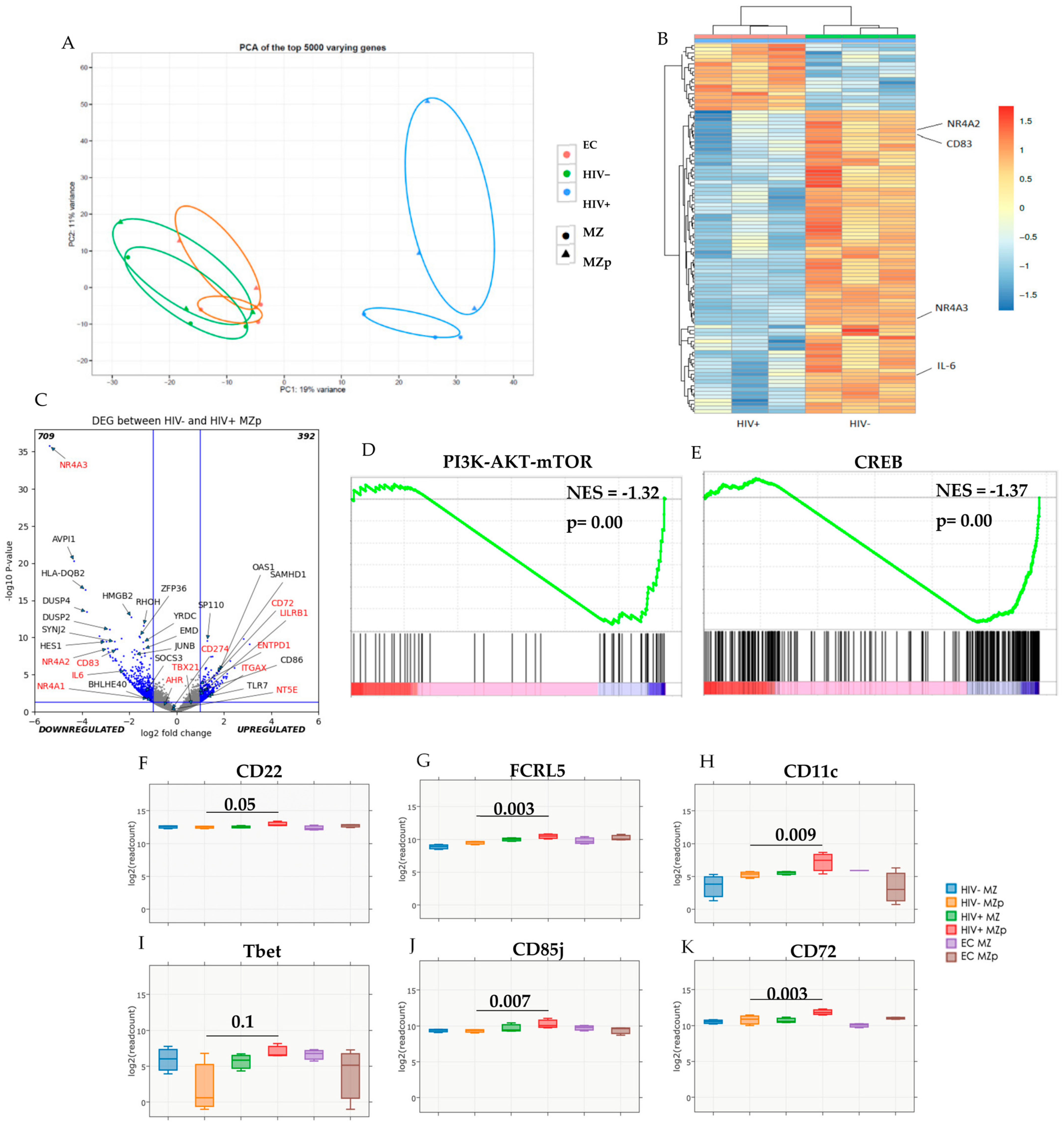
2.2. Blood MZp B-Cells from Progressors Show An Altered Transcriptomic Profile
2.3. Blood MZp B-Cells from Progressors Express Lower Levels of Regulatory Markers
2.4. The Breg Function of Blood MZp B-Cells from Progressors Is Altered, despite ART
2.5. Excess BAFF Directly Contributes to the Altered Breg Potential of MZp B-cells
3. Discussion
4. Materials and Methods
4.1. Specimen Collection and Clinical Data
4.2. Cell Sorting of Human Blood MZ and MZp B-Cells, RNA Isolation and Sequencing
4.3. Volcano Plot and Gene Set Enrichment Analyses (GSEAs)
4.4. Multicolor Flow-Cytometry
4.5. Human Tonsillar B-Cells
4.6. qPCR Characterisation of NR4A1, NR4A3 and CD83 mRNA Expression Levels by Blood MZp B-Cells
4.7. Breg Functional Assays
4.7.1. For Assays with Tonsillar Populations
4.7.2. For Assays with Blood Cell Populations
4.8. MoDC: B-Cell Co-Cultures
4.9. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- UNAIDS. Global HIV & AIDS Statistics—Fact Sheet. 2021. Available online: https://www.unaids.org/en/resources/fact-sheet (accessed on 8 November 2022).
- De Pablo-Bernal, R.S.; Ruiz-Mateos, E.; Rosado, I.; Dominguez-Molina, B.; Alvarez-Ríos, A.I.; Carrillo-Vico, A.; De La Rosa, R.; Delgado, J.; Muñoz-Fernández, M.A.; Leal, M.; et al. TNF-α levels in HIV-infected patients after long-term suppressive cART persist as high as in elderly, HIV-uninfected subjects. J. Antimicrob. Chemother. 2014, 69, 3041–3046. [Google Scholar] [CrossRef] [Green Version]
- Fontaine, J.; Chagnon-Choquet, J.; Valcke, H.S.; Poudrier, J.; Roger, M.; the Montreal Primary HIV Infection and Long-Term Non-Progressor Study Groups. High expression levels of B lymphocyte stimulator (BLyS) by dendritic cells correlate with HIV-related B-cell disease progression in humans. Blood 2011, 117, 145–155. [Google Scholar] [CrossRef]
- Aranguren, M.K.D.-L.; El-Far, M.; Chartrand-Lefebvre, C.; Routy, J.-P.; Tremblay, C.; Durand, M.; Poudrier, J.; Roger, R. Subclinical Atherosclerosis is Associated with Discrepancies in BAFF and APRIL Levels and Altered Breg Potential of Precursor-like Marginal Zone B-cells in HIVTreated Individuals. bioRxiv 2022. [Google Scholar] [CrossRef]
- Moir, S.; Fauci, A.S. B cells in HIV infection and disease. Nat. Rev. Immunol. 2009, 9, 235–245. [Google Scholar] [CrossRef] [Green Version]
- Poudrier, J.; Chagnon-Choquet, J.; Roger, M. Influence of Dendritic Cells on B-Cell Responses during HIV Infection. Clin. Dev. Immunol. 2012, 2012, 592187. [Google Scholar] [CrossRef] [Green Version]
- Chagnon-Choquet, J.; Gauvin, J.; Roger, J.; Fontaine, J.; Poudrier, J.; Vassal, A.; Legault, M.; Routy, J.P.; Tremblay, C.; Thomas, R.; et al. HIV Nef Promotes Expression of B-Lymphocyte Stimulator by Blood Dendritic Cells During HIV Infection in Humans. J. Infect. Dis. 2014, 211, 1229–1240. [Google Scholar] [CrossRef] [Green Version]
- Poudrier, J.; Weng, X.; Kay, D.G.; Paré, G.; Calvo, E.L.; Hanna, Z.; Kosco-Vilbois, M.H.; Jolicoeur, P. The AIDS Disease of CD4C/HIV Transgenic Mice Shows Impaired Germinal Centers and Autoantibodies and Develops in the Absence of IFN-γ and IL-6. Immunity 2001, 15, 173–185. [Google Scholar] [CrossRef] [Green Version]
- Poudrier, J.; Soulas, C.; Chagnon-Choquet, J.; Burdo, T.; Autissier, P.; Oskar, K.; Williams, K.C.; Roger, M. High Expression Levels of BLyS/BAFF by Blood Dendritic Cells and Granulocytes Are Associated with B-cell dysregulation in SIV-Infected Rhesus Macaques. PLoS ONE 2015, 10, e0131513. [Google Scholar] [CrossRef]
- Fourcade, L.; Sabourin-Poirier, C.; Perraud, V.; Faucher, M.-C.; Chagnon-Choquet, J.; Labbé, A.-C.; Alary, M.; Guédou, F.; Poudrier, J.; Roger, M. Natural Immunity to HIV is associated with Low BLyS/BAFF levels and low frequencies of innate marginal zone like CD1c+ B-cells in the genital tract. PLoS Pathog. 2019, 15, e1007840. [Google Scholar] [CrossRef] [Green Version]
- Sabourin-Poirier, C.; Fourcade, L.; Chagnon-Choquet, J.; Labbé, A.C.; Alary, M.; Guédou, F.; Poudrier, J.; Roger, M. Blood B Lymphocyte Stimulator (BLyS)/BAFF levels may reflect natural immunity to HIV in highly exposed uninfected Beninese Commercial Sex Workers. Sci. Rep. 2016, 6, 32318. [Google Scholar] [CrossRef]
- Cerutti, A.; Cols, M.; Puga, I. Marginal zone B cells: Virtues of innate-like antibody-producing lymphocytes. Nat. Rev. Immunol. 2013, 13, 118–132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, B.; Qiao, X.; Klasse, P.J.; Chiu, A.; Chadburn, A.; Knowles, D.M.; Moore, J.P.; Cerutti, A. HIV-1 Envelope Triggers Polyclonal Ig Class Switch Recombination through a CD40-Independent Mechanism Involving BAFF and C-Type Lectin Receptors. J. Immunol. 2006, 176, 3931–3941. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Henrick, B.M.; Yao, X.-D.; Zahoor, M.; Abimiku, A.; Osawe, S.; Rosenthal, K.L. TLR10 Senses HIV-1 Proteins and Significantly Enhances HIV-1 Infection. Front. Immunol. 2019, 10, 482. [Google Scholar] [CrossRef]
- Doyon-Laliberté, K.; Chagnon-Choquet, J.; Byrns, M.; Aranguren, M.; Memmi, M.; Chrobak, P.; Stagg, J.; Poudrier, J.; Roger, M. NR4A Expression by Human Marginal Zone B-Cells. Antibodies 2019, 8, 50. [Google Scholar] [CrossRef] [Green Version]
- Sekiya, T.; Kashiwagi, I.; Yoshida, R.; Fukaya, T.; Morita, R.; Kimura, A.; Ichinose, H.; Metzger, D.; Chambon, P.; Yoshimura, A. Nr4a receptors are essential for thymic regulatory T cell development and immune homeostasis. Nat. Immunol. 2013, 14, 230–237. [Google Scholar] [CrossRef]
- Bandukwala, H.S.; Rao, A. ’Nurr’ishing Treg cells: Nr4a transcription factors control Foxp3 expression. Nat. Immunol. 2013, 14, 201–203. [Google Scholar] [CrossRef]
- Duren, R.P.; Boudreaux, S.P.; Conneely, O.M. Genome Wide Mapping of NR4A Binding Reveals Cooperativity with ETS Factors to Promote Epigenetic Activation of Distal Enhancers in Acute Myeloid Leukemia Cells. PLoS ONE 2016, 11, e0150450. [Google Scholar] [CrossRef]
- Ashouri, J.; Weiss, A. Endogenous Nur77 Is a Specific Indicator of Antigen Receptor Signaling in Human T and B Cells. J. Immunol. 2016, 198, 657–668. [Google Scholar] [CrossRef] [Green Version]
- Odagiu, L.; May, J.; Boulet, S.; Baldwin, T.A.; Labrecque, N. Role of the Orphan Nuclear Receptor NR4A Family in T-Cell Biology. Front. Endocrinol. 2021, 11, 624122. [Google Scholar] [CrossRef]
- Tel-Karthaus, N.; Kers-Rebel, E.D.; Looman, M.W.; Ichinose, H.; De Vries, C.J.; Ansems, M. Nuclear Receptor Nur77 Deficiency Alters Dendritic Cell Function. Front. Immunol. 2018, 9, 1797. [Google Scholar] [CrossRef]
- Iizuka-Koga, M.; Nakatsukasa, H.; Ito, M.; Akanuma, T.; Lu, Q.; Yoshimura, A. Induction and maintenance of regulatory T cells by transcription factors and epigenetic modifications. J. Autoimmun. 2017, 83, 113–121. [Google Scholar] [CrossRef]
- Nus, M.; Sage, A.P.; Lu, Y.; Masters, L.; Lam, B.Y.H.; Newland, S.; Weller, S.; Tsiantoulas, D.; Raffort, J.; Marcus, D.; et al. Marginal zone B cells control the response of follicular helper T cells to a high-cholesterol diet. Nat. Med. 2017, 23, 601–610. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nus, M.; Basatemur, G.; Galan, M.; Cros-Brunsó, L.; Zhao, T.X.; Masters, L.; Harrison, J.; Figg, N.; Tsiantoulas, D.; Geissmann, F.; et al. NR4A1 Deletion in Marginal Zone B Cells Exacerbates Atherosclerosis in Mice—Brief Report. Arter. Thromb. Vasc. Biol. 2020, 40, 2598–2604. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.; Oberle, N.; Krammer, P.H. Molecular Mechanisms of Treg-Mediated T Cell Suppression. Front. Immunol. 2012, 3, 51. [Google Scholar] [CrossRef] [Green Version]
- Chagnon-Choquet, J.; Fontaine, J.; Poudrier, J.; Roger, M.; for the Montreal Primary HIV Infection and Slow Progressor Study Groups. IL-10 and Lymphotoxin-α Expression Profiles within Marginal Zone-Like B-Cell Populations Are Associated with Control of HIV-1 Disease Progression. PLoS ONE 2014, 9, e101949. [Google Scholar] [CrossRef]
- Saze, Z.; Schuler, P.J.; Hong, C.-S.; Cheng, D.; Jackson, E.K.; Whiteside, T.L. Adenosine production by human B cells and B cell–mediated suppression of activated T cells. Blood 2013, 122, 9–18. [Google Scholar] [CrossRef] [Green Version]
- Mackay, F.; Woodcock, S.A.; Lawton, P.; Ambrose, C.; Baetscher, M.; Schneider, P.; Tschopp, J.; Browning, J. Mice Transgenic for Baff Develop Lymphocytic Disorders along with Autoimmune Manifestations. J. Exp. Med. 1999, 190, 1697–1710. [Google Scholar] [CrossRef]
- Hanna, Z.; Kay, D.G.; Cool, M.; Jothy, S.; Rebai, N.; Jolicoeur, P. Transgenic Mice Expressing Human Immunodeficiency Virus Type 1 in Immune Cells Develop a Severe AIDS-Like Disease. J. Virol. 1998, 72, 121–132. [Google Scholar] [CrossRef] [Green Version]
- Hanna, Z.; Kay, D.G.; Rebai, N.; Guimond, A.; Jothy, S.; Jolicoeur, P. Nef Harbors a Major Determinant of Pathogenicity for an AIDS-like Disease Induced by HIV-1 in Transgenic Mice. Cell 1998, 95, 163–175. [Google Scholar] [CrossRef] [Green Version]
- Steri, M.; Orrù, V.; Idda, M.L.; Pitzalis, M.; Pala, M.; Zara, I.; Sidore, C.; Faà, V.; Floris, M.; Deiana, M.; et al. Overexpression of the Cytokine BAFF and Autoimmunity Risk. N. Engl. J. Med. 2017, 376, 1615–1626. [Google Scholar] [CrossRef]
- Nakayamada, S.; Tanaka, Y. BAFF- and APRIL-targeted therapy in systemic autoimmune diseases. Inflamm. Regen. 2016, 36, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Borhis, G.; Trovato, M.; Chaoul, N.; Ibrahim, H.; Richard, Y. B-Cell-Activating Factor and the B-Cell Compartment in HIV/SIV Infection. Front. Immunol. 2017, 8, 1338. [Google Scholar] [CrossRef] [Green Version]
- Sintes, J.; Gentile, M.; Zhang, S.; Garcia-Carmona, Y.; Magri, G.; Cassis, L.; Segura-Garzón, D.; Ciociola, A.; Grasset, E.K.; Bascones, S.; et al. mTOR intersects antibody-inducing signals from TACI in marginal zone B cells. Nat. Commun. 2017, 8, 1462. [Google Scholar] [CrossRef] [Green Version]
- Yi, Z.; Lin, W.W.; Stunz, L.L.; Bishop, G.A. Roles for TNF-receptor associated factor 3 (TRAF3) in lymphocyte functions. Cytokine Growth Factor Rev. 2013, 25, 147–156. [Google Scholar] [CrossRef] [Green Version]
- Mambetsariev, N.; Lin, W.W.; Stunz, L.L.; Hanson, B.M.; Hildebrand, J.M.; Bishop, G.A. Nuclear TRAF3 is a negative regulator of CREB in B cells. Proc. Natl. Acad. Sci. USA 2016, 113, 1032–1037. [Google Scholar] [CrossRef] [Green Version]
- Volakakis, N.; Kadkhodaei, B.; Joodmardi, E.; Wallis, K.; Panman, L.; Silvaggi, J.; Spiegelman, B.M.; Perlmann, T. NR4A orphan nuclear receptors as mediators of CREB-dependent neuroprotection. Proc. Natl. Acad. Sci. USA 2010, 107, 12317–12322. [Google Scholar] [CrossRef] [Green Version]
- Bridi, M.S.; Hawk, J.D.; Chatterjee, S.; Safe, S.; Abel, T. Pharmacological Activators of the NR4A Nuclear Receptors Enhance LTP in a CREB/CBP-Dependent Manner. Neuropsychopharmacology 2016, 42, 1243–1253. [Google Scholar] [CrossRef] [Green Version]
- Wen, A.Y.; Sakamoto, K.M.; Miller, L.S. The Role of the Transcription Factor CREB in Immune Function. J. Immunol. 2010, 185, 6413–6419. [Google Scholar] [CrossRef] [Green Version]
- Safe, S.; Jin, U.H.; Morpurgo, B.; Abudayyeh, A.; Singh, M.; Tjalkens, R.B. Nuclear receptor 4A (NR4A) family—orphans no more. J. Steroid Biochem. Mol. Biol. 2016, 157, 48–60. [Google Scholar] [CrossRef] [Green Version]
- Boudreaux, S.P.; Duren, R.P.; Call, S.G.; Nguyen, L.; Freire, P.R.; Narayanan, P.; Redell, M.S.; Conneely, O.M. Drug targeting of NR4A nuclear receptors for treatment of acute myeloid leukemia. Leukemia 2018, 33, 52–63. [Google Scholar] [CrossRef]
- Deutsch, A.J.A.; Angerer, H.; Fuchs, T.E.; Neumeister, P. The nuclear orphan receptors NR4A as therapeutic target in cancer therapy. Anti-Cancer Agents Med. Chem. 2012, 12, 1001–1014. [Google Scholar] [CrossRef] [PubMed]
- Antonioli, L.; Pacher, P.; Vizi, E.S.; Haskó, G. CD39 and CD73 in immunity and inflammation. Trends Mol. Med. 2013, 19, 355–367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, W.X.; Huang, H.H.; Huang, L.; Shi, J.J.; Jiao, Y.M.; Zhang, C.; Jin, L.; Yang, T.; Shi, M.; Tu, B.; et al. Skewed CD39/CD73/adenosine pathway in B cells is associated with innate immune hyperactivation in chronic HIV-1 infection. Transl. Med. Commun. 2019, 4, 4. [Google Scholar] [CrossRef]
- Kim, E.-S.; Ackermann, C.; Tóth, I.; Dierks, P.; Eberhard, J.M.; Wroblewski, R.; Scherg, F.; Geyer, M.; E Schmidt, R.; Beisel, C.; et al. Down-regulation of CD73 on B cells of patients with viremic HIV correlates with B cell activation and disease progression. J. Leukoc. Biol. 2017, 101, 1263–1271. [Google Scholar] [CrossRef] [Green Version]
- Rothan, C.; Yero, A.; Shi, T.; Farnos, O.; Chartrand-Lefebvre, C.; El-Far, M.; Costiniuk, C.T.; Tsoukas, C.; Tremblay, C.; Durand, M.; et al. Antiretroviral therapy-treated HIV-infected adults with coronary artery disease are characterized by a distinctive regulatory T-cell signature. AIDS 2021, 35, 1003–1014. [Google Scholar] [CrossRef]
- Zhou, S.N.; Zhang, N.; Liu, H.H.; Xia, P.; Zhang, C.; Song, J.W.; Fan, X.; Shi, M.; Jin, L.; Zhang, J.Y.; et al. Skewed CD39/CD73/adenosine pathway contributes to B-cell hyperactivation and disease progression in patients with chronic hepatitis B. Gastroenterol. Rep. 2020, 9, 49–58. [Google Scholar] [CrossRef]
- Crean, D.; Cummins, E.P.; Bahar, B.; Mohan, H.; McMorrow, J.P.; Murphy, E.P. Adenosine Modulates NR4A Orphan Nuclear Receptors to Attenuate Hyperinflammatory Responses in Monocytic Cells. J. Immunol. 2015, 195, 1436–1448. [Google Scholar] [CrossRef] [Green Version]
- Allard, B.; Allard, D.; Buisseret, L.; Stagg, J. The adenosine pathway in immuno-oncology. Nat. Rev. Clin. Oncol. 2020, 17, 611–629. [Google Scholar] [CrossRef]
- Piper, C.J.; Rosser, E.C.; Oleinika, K.; Nistala, K.; Krausgruber, T.; Rendeiro, A.; Banos, A.; Drozdov, I.; Villa, M.; Thomson, S.; et al. Aryl Hydrocarbon Receptor Contributes to the Transcriptional Program of IL-10-Producing Regulatory B Cells. Cell Rep. 2019, 29, 1878–1892.e7. [Google Scholar] [CrossRef] [Green Version]
- Poudrier, J.; Weng, X.; Kay, D.G.; Hanna, Z.; Jolicoeur, P. The AIDS-Like Disease of CD4C/Human Immunodeficiency Virus Transgenic Mice Is Associated with Accumulation of Immature CD11bHi Dendritic Cells. J. Virol. 2003, 77, 11733–11744. [Google Scholar] [CrossRef]
- Louis, K.; Bailly, E.; Macedo, C.; Lau, L.; Ramaswami, B.; Chang, A.; Chandran, U.; Landsittel, D.; Gu, X.; Chalasani, G.; et al. T-bet+CD27+CD21– B cells poised for plasma cell differentiation during antibody-mediated rejection of kidney transplants. JCI Insight 2021, 6, e148881. [Google Scholar] [CrossRef] [PubMed]
- Austin, J.W.; Buckner, C.M.; Kardava, L.; Wang, W.; Zhang, X.; Melson, V.A.; Swanson, R.G.; Martins, A.J.; Zhou, J.Q.; Hoehn, K.B.; et al. Overexpression of T-bet in HIV infection is associated with accumulation of B cells outside germinal centers and poor affinity maturation. Sci. Transl. Med. 2019, 11, eaax0904. [Google Scholar] [CrossRef]
- Knox, J.J.; Kaplan, D.E.; Betts, M.R. T-bet-expressing B cells during HIV and HCV infections. Cell. Immunol. 2017, 321, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, M.C.; Ramonell, R.P.; Nguyen, D.C.; Cashman, K.S.; Saini, A.S.; Haddad, N.S.; Ley, A.M.; Kyu, S.; Howell, J.C.; Ozturk, T.; et al. Extrafollicular B cell responses correlate with neutralizing antibodies and morbidity in COVID-19. Nat. Immunol. 2020, 21, 1506–1516. [Google Scholar] [CrossRef] [PubMed]
- Knox, J.J.; Buggert, M.; Kardava, L.; Seaton, K.E.; Eller, M.; Canaday, D.H.; Robb, M.L.; Ostrowski, M.A.; Deeks, S.G.; Slifka, M.K.; et al. T-bet+ B cells are induced by human viral infections and dominate the HIV gp140 response. JCI Insight 2017, 2, e92943. [Google Scholar] [CrossRef] [Green Version]
- Mauri, C. Novel Frontiers in Regulatory B cells. Immunol. Rev. 2021, 299, 5–9. [Google Scholar] [CrossRef]
- Attanavanich, K.; Kearney, J.F. Marginal Zone, but Not Follicular B Cells, Are Potent Activators of Naive CD4 T Cells. J. Immunol. 2004, 172, 803–811. [Google Scholar] [CrossRef] [Green Version]
- Cinamon, G.; Zachariah, M.A.; Lam, O.M.; Foss, F.W.; Cyster, J.G. Follicular shuttling of marginal zone B cells facilitates antigen transport. Nat. Immunol. 2008, 9, 54–62. [Google Scholar] [CrossRef] [Green Version]
- Bialecki, E.; Paget, C.; Fontaine, J.; Capron, M.; Trottein, F.; Faveeuw, C. Role of Marginal Zone B Lymphocytes in Invariant NKT Cell Activation. J. Immunol. 2009, 182, 6105–6113. [Google Scholar] [CrossRef] [Green Version]
- Doyon-Laliberté, K.; Aranguren, M.; Poudrier, J.; Roger, M. Marginal Zone B-Cell Populations and Their Regulatory Potential in the Context of HIV and Other Chronic Inflammatory Conditions. Int. J. Mol. Sci. 2022, 23, 3372. [Google Scholar] [CrossRef]
- Mercier, F.; Boulassel, M.R.; Yassine-Diab, B.; Tremblay, C.; Bernard, N.F.; Sekaly, R.P.; Routy, J.P. Persistent human immunodeficiency virus-1 antigenaemia affects the expression of interleukin-7Ralpha on central and effector memory CD4+ and CD8+ T cell subsets. Clin. Exp. Immunol. 2008, 152, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mootha, V.K.; Lindgren, C.M.; Eriksson, K.-F.; Subramanian, A.; Sihag, S.; Lehar, J.; Puigserver, P.; Carlsson, E.; Ridderstråle, M.; Laurila, E.; et al. PGC-1α-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat. Genet. 2003, 34, 267–273. [Google Scholar] [CrossRef] [PubMed]



| RNA-Seq Analysis | Flow-Cytometry Analysis | |||||
|---|---|---|---|---|---|---|
| Group | HIV+ (n = 3) | EC (n = 3) | p-Value | HIV+ ART− (n = 10) | HIV+ ART+ (n = 10) | p-Value |
| Age | 38.67 (28–44) | 38.67 (37–40) | 0.7 | NA | NA | NA |
| Viral load | 213,353 (51,045–418,465) | NA 1 | NA | 1,258,139 (374–6,193,724) | 41.4 (20–234) | 0.002 |
| CD4+ T-cell count (cells/mm3) | 540 (380–760) | 819 (737–920) | 0.2 | 447 (284–590) | 776.5 (565–1210) | 0.0006 |
| CD8+ T-cell count (cells/mm3) | 871–(520–1153) | 821 (303–1130) | >0.99 | 1128 (0–3190) | 634.3 (210–1080) | 0.06 |
| ART duration (weeks) | NA | NA | NA | NA | 92 (84–96) | NA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Doyon-Laliberté, K.; Aranguren, M.; Byrns, M.; Chagnon-Choquet, J.; Paniconi, M.; Routy, J.-P.; Tremblay, C.; Quintal, M.-C.; Brassard, N.; Kaufmann, D.E.; et al. Excess BAFF Alters NR4As Expression Levels and Breg Function of Human Precursor-like Marginal Zone B-Cells in the Context of HIV-1 Infection. Int. J. Mol. Sci. 2022, 23, 15142. https://doi.org/10.3390/ijms232315142
Doyon-Laliberté K, Aranguren M, Byrns M, Chagnon-Choquet J, Paniconi M, Routy J-P, Tremblay C, Quintal M-C, Brassard N, Kaufmann DE, et al. Excess BAFF Alters NR4As Expression Levels and Breg Function of Human Precursor-like Marginal Zone B-Cells in the Context of HIV-1 Infection. International Journal of Molecular Sciences. 2022; 23(23):15142. https://doi.org/10.3390/ijms232315142
Chicago/Turabian StyleDoyon-Laliberté, Kim, Matheus Aranguren, Michelle Byrns, Josiane Chagnon-Choquet, Matteo Paniconi, Jean-Pierre Routy, Cécile Tremblay, Marie-Claude Quintal, Nathalie Brassard, Daniel E. Kaufmann, and et al. 2022. "Excess BAFF Alters NR4As Expression Levels and Breg Function of Human Precursor-like Marginal Zone B-Cells in the Context of HIV-1 Infection" International Journal of Molecular Sciences 23, no. 23: 15142. https://doi.org/10.3390/ijms232315142





