Elsholtzia bodinieri Vaniot Ameliorated Acute Lung Injury by NQO1, BCL2 and PTGS2 In Silico and In Vitro Analyses
Abstract
:1. Introduction
2. Results
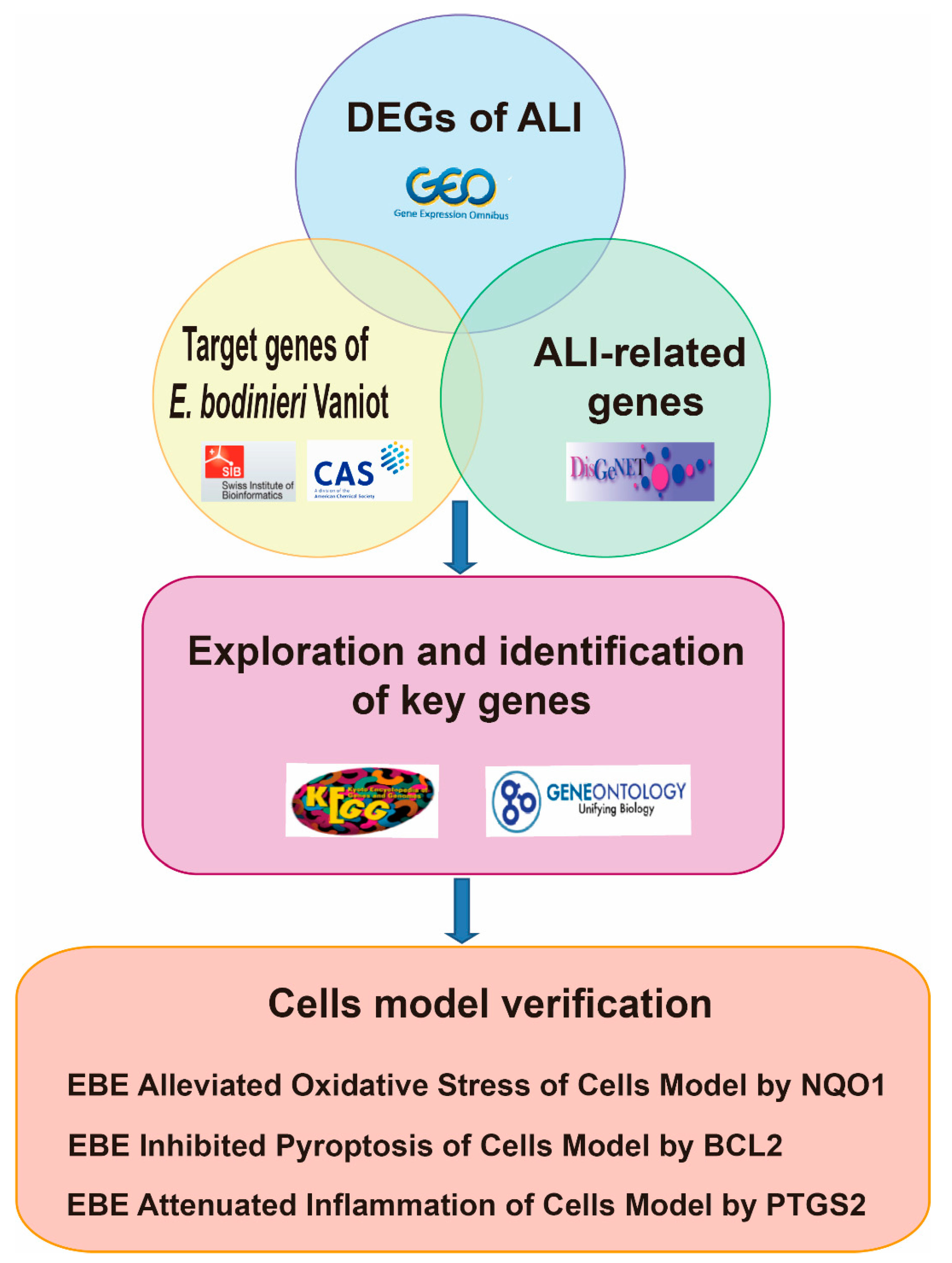
2.1. Screening Differentially Expressed Genes and Enrichment Analysis in ALI
2.2. Exploration and Identification of Key Genes in ALI Regulated by E. bodinieri Vaniot
2.3. EBE Alleviated Oxidative Stress of Cells Model by NQO1
2.4. EBE Inhibited Pyroptosis of Cells Model by BCL2
2.5. EBE Attenuated Inflammation of Cells Model by PTGS2
3. Discussion
4. Materials and Methods
4.1. ALI Gene Expression Data Acquisition and Processing
4.2. Exploration and Identification of Key Genes
4.3. Cell Experiments
4.4. Detection of Inflammatory Cytokines and Oxidative Stress Related Factors
4.5. Transmission Electron Microscopy
4.6. Western Blotting
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rubin, E.J.; Baden, L.R.; Morrissey, S. Audio Interview: Acute Lung Injury in COVID-19. N. Engl. J. Med. 2020, 383, e32. [Google Scholar] [CrossRef] [PubMed]
- Christiani, D.C. Vaping-Induced Acute Lung Injury. N. Engl. J. Med. 2019, 382, 960–962. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Zhang, C.; Lv, N.; Liang, Z.; Ma, T.; Cheng, H.; Xia, Y.; Shi, L. AdMSC-derived exosomes alleviate acute lung injury via transferring mitochondrial component to improve homeostasis of alveolar macrophages. Theranostics 2022, 12, 2928–2947. [Google Scholar] [CrossRef] [PubMed]
- Matthay, M.A.; Zemans, R.L.; Zimmerman, G.A.; Arabi, Y.M.; Beitler, J.R.; Mercat, A.; Herridge, M.; Randolph, A.G.; Calfee, C.S. Acute respiratory distress syndrome. Nat. Rev. Dis. Primers 2019, 5, 18. [Google Scholar] [CrossRef]
- Liu, H.; He, Y.; Lu, C.; Zhang, P.; Zhou, C.; Ni, Y.; Niu, W.; Yuan, X.; Li, P.; Zheng, J.; et al. Efficacy of pulmonary transplantation of engineered macrophages secreting IL-4 on acute lung injury in C57BL/6J mice. Cell Death Dis. 2019, 10, 664. [Google Scholar] [CrossRef] [Green Version]
- Tong, Y.; Yu, Z.; Chen, Z.; Zhang, R.; Ding, X.; Yang, X.; Niu, X.; Li, M.; Zhang, L.; Billiar, T.R.; et al. The HIV protease inhibitor Saquinavir attenuates sepsis-induced acute lung injury and promotes M2 macrophage polarization via targeting matrix metalloproteinase-9. Cell Death Dis. 2021, 12, 67. [Google Scholar] [CrossRef]
- Kong, L.; Deng, J.; Zhou, X.; Cai, B.; Zhang, B.; Chen, X.; Chen, Z.; Wang, W. Sitagliptin activates the p62-Keap1-Nrf2 signalling pathway to alleviate oxidative stress and excessive autophagy in severe acute pancreatitis-related acute lung injury. Cell Death Dis. 2021, 12, 928. [Google Scholar] [CrossRef]
- Yin, J.; Bai, C.-X. Pharmacotherapy for Adult Patients with Acute Respiratory Distress Syndrome. Chin. Med. J. 2018, 131, 1138–1141. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, L.; Du, J.; Shao, L.; Yu, F.; Li, R.; Zhong, J. Two new oleanane triterpenoid saponins from Elsholtzia bodinieri. Nat. Prod. Res. 2021, 35, 3658–3666. [Google Scholar] [CrossRef]
- Xiang, L.; Zhang, L.; Chen, X.; Xia, X.; Li, R.; Zhong, J. Ursane-type triterpenoid saponins from Elsholtzia bodinieri. Nat. Prod. Res. 2019, 33, 1349–1356. [Google Scholar] [CrossRef]
- Zhong, J.D.; Feng, Y.; Li, H.M.; Xia, X.S.; Li, R.T. A new flavonoid glycoside from Elsholtzia bodinieri. Nat. Prod. Res. 2016, 30, 2278–2284. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-Z.; Fu, L.-Z.; Li, H.-M.; Li, R.-T.; Deng, X.-L. Two new oleanane triterpenoid saponins from Elsholtzia bodinieri. Phytochem. Lett. 2012, 5, 572–575. [Google Scholar] [CrossRef]
- Zhao, X.-W.; Zhong, J.-D.; Li, H.-M.; Li, R.-T. Three new 18,19-seco-ursane glycosides from Elsholtzia bodinieri. Phytochem. Lett. 2015, 12, 308–312. [Google Scholar] [CrossRef]
- Zhong, J.-D.; Zhao, X.-W.; Li, H.-M.; Gao, L.-H.; Li, R.-T. Five New Oleanane Triterpenoid Saponins from the Aerial Parts of Elsholtzia bodinieri. Helv. Chim. Acta 2016, 99, 204–209. [Google Scholar] [CrossRef]
- Zhong, J.D.; Zhao, X.W.; Chen, X.Q.; Li, H.M.; Chen, C.H.; Xia, X.S.; Li, R.T. Two new ursane-type triterpenoid saponins from Elsholtzia bodinieri. Arch. Pharm. Res. 2016, 39, 771–777. [Google Scholar] [CrossRef]
- Hu, H.-B.; Jian, Y.-F.; Cao, H.; Zheng, X.-D. Phenolic Compounds from Elsholtzia Bodinieri Van’t. J. Chin. Chem. Soc. 2007, 54, 1189–1194. [Google Scholar] [CrossRef]
- McKenzie, B.A.; Fernandes, J.P.; Doan, M.A.L.; Schmitt, L.M.; Branton, W.G.; Power, C. Activation of the executioner caspases-3 and -7 promotes microglial pyroptosis in models of multiple sclerosis. J. Neuroinflamm. 2020, 17, 253. [Google Scholar] [CrossRef]
- Deng, Y.; Fu, Y.; Sheng, L.; Hu, Y.; Su, L.; Luo, J.; Yan, C.; Chi, W. The Regulatory NOD-Like Receptor NLRC5 Promotes Ganglion Cell Death in Ischemic Retinopathy by Inducing Microglial Pyroptosis. Front. Cell Dev. Biol. 2021, 9, 669696. [Google Scholar] [CrossRef]
- Totten, S.P.; Im, Y.K.; Cepeda Canedo, E.; Najyb, O.; Nguyen, A.; Hebert, S.; Ahn, R.; Lewis, K.; Lebeau, B.; La Selva, R.; et al. STAT1 potentiates oxidative stress revealing a targetable vulnerability that increases phenformin efficacy in breast cancer. Nat. Commun. 2021, 12, 3299. [Google Scholar] [CrossRef]
- Li, X.; Liu, Z.; Zhang, A.; Han, C.; Shen, A.; Jiang, L.; Boothman, D.A.; Qiao, J.; Wang, Y.; Huang, X.; et al. NQO1 targeting prodrug triggers innate sensing to overcome checkpoint blockade resistance. Nat. Commun. 2019, 10, 3251. [Google Scholar] [CrossRef]
- Wang, P.; Geng, J.; Gao, J.; Zhao, H.; Li, J.; Shi, Y.; Yang, B.; Xiao, C.; Linghu, Y.; Sun, X.; et al. Macrophage achieves self-protection against oxidative stress-induced ageing through the Mst-Nrf2 axis. Nat. Commun. 2019, 10, 755. [Google Scholar] [CrossRef] [PubMed]
- Hecker, L. Mechanisms and consequences of oxidative stress in lung disease: Therapeutic implications for an aging populace. Am. J. Physiol. Lung Cell Mol. Physiol. 2018, 314, L642–L653. [Google Scholar] [CrossRef] [PubMed]
- Nishida, T.; Naguro, I.; Ichijo, H. NAMPT-dependent NAD(+) salvage is crucial for the decision between apoptotic and necrotic cell death under oxidative stress. Cell Death Discov. 2022, 8, 195. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, J.; Yu, S.; Li, Y.; Zhu, J.; Zhang, K.; Zhang, R. Cell pyroptosis in health and inflammatory diseases. Cell Death Discov. 2022, 8, 191. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.; Bai, Y.; Deng, F.; Pan, Y.; Mei, S.; Zheng, Z.; Min, R.; Wu, Z.; Li, W.; Miao, R.; et al. Streptococcal pyrogenic exotoxin B cleaves GSDMA and triggers pyroptosis. Nature 2022, 602, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.G.; Chávez, C.L.; Zhang, C.; Sowden, M.; Yan, C.; Berk, B.C. The lipid peroxidation product 4-hydroxynonenal inhibits NLRP3 inflammasome activation and macrophage pyroptosis. Cell Death Differ. 2022, 29, 1790–1803. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.S.; Tan, L.; Jiang, T.; Zhu, X.C.; Wang, H.F.; Jia, C.D.; Yu, J.T. Amyloid-β induces NLRP1-dependent neuronal pyroptosis in models of Alzheimer’s disease. Cell Death Dis. 2014, 5, e1382. [Google Scholar] [CrossRef] [Green Version]
- Hou, J.; Zhao, R.; Xia, W.; Chang, C.-W.; You, Y.; Hsu, J.-M.; Nie, L.; Chen, Y.; Wang, Y.-C.; Liu, C.; et al. PD-L1-mediated gasdermin C expression switches apoptosis to pyroptosis in cancer cells and facilitates tumour necrosis. Nat. Cell Biol. 2020, 22, 1264–1275. [Google Scholar] [CrossRef]
- Shi, C.S.; Kehrl, J.H. Bcl-2 regulates pyroptosis and necroptosis by targeting BH3-like domains in GSDMD and MLKL. Cell Death Discov. 2019, 5, 151. [Google Scholar] [CrossRef] [Green Version]
- de Vasconcelos, N.M.; Van Opdenbosch, N.; Van Gorp, H.; Parthoens, E.; Lamkanfi, M. Single-cell analysis of pyroptosis dynamics reveals conserved GSDMD-mediated subcellular events that precede plasma membrane rupture. Cell Death Differ. 2019, 26, 146–161. [Google Scholar] [CrossRef]
- Kang, J.Y.; Xu, M.M.; Sun, Y.; Ding, Z.X.; Wei, Y.Y.; Zhang, D.W.; Wang, Y.G.; Shen, J.L.; Wu, H.M.; Fei, G.H. Melatonin attenuates LPS-induced pyroptosis in acute lung injury by inhibiting NLRP3-GSDMD pathway via activating Nrf2/HO-1 signaling axis. Int. Immunopharmacol. 2022, 109, 108782. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.H.; Duan, J.X.; Liu, S.K.; Xiong, J.B.; Guan, X.X.; Zhong, W.J.; Sun, C.C.; Zhang, C.Y.; Luo, X.Q.; Zhang, Y.F.; et al. A COX-2/sEH dual inhibitor PTUPB alleviates lipopolysaccharide-induced acute lung injury in mice by inhibiting NLRP3 inflammasome activation. Theranostics 2020, 10, 4749–4761. [Google Scholar] [CrossRef] [PubMed]
- Nelin, L.D.; Jin, Y.; Chen, B.; Liu, Y.; Rogers, L.K.; Reese, J. Cyclooxygenase-2 deficiency attenuates lipopolysaccharide-induced inflammation, apoptosis, and acute lung injury in adult mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2022, 322, R126–R135. [Google Scholar] [CrossRef] [PubMed]
- Robinson, E.K.; Worthington, A.; Poscablo, D.; Shapleigh, B.; Salih, M.M.; Halasz, H.; Seninge, L.; Mosqueira, B.; Smaliy, V.; Forsberg, E.C.; et al. lincRNA-Cox2 Functions to Regulate Inflammation in Alveolar Macrophages during Acute Lung Injury. J. Immunol. 2022, 208, 1886–1900. [Google Scholar] [CrossRef]
- Yao, W.; Shi, L.; Zhang, Y.; Dong, H.; Zhang, Y. Mesenchymal stem/stromal cell therapy for COVID-19 pneumonia: Potential mechanisms, current clinical evidence, and future perspectives. Stem Cell Res. Ther. 2022, 13, 124. [Google Scholar] [CrossRef]
- Abedi, F.; Hayes, A.W.; Reiter, R.; Karimi, G. Acute lung injury: The therapeutic role of Rho kinase inhibitors. Pharmacol. Res. 2020, 155, 104736. [Google Scholar] [CrossRef]
- Aggarwal, N.R.; D’Alessio, F.R.; Tsushima, K.; Sidhaye, V.K.; Cheadle, C.; Grigoryev, D.N.; Barnes, K.C.; King, L.S. Regulatory T cell-mediated resolution of lung injury: Identification of potential target genes via expression profiling. Physiol. Genom. 2010, 41, 109–119. [Google Scholar] [CrossRef] [Green Version]
- Altemeier, W.A.; Matute-Bello, G.; Gharib, S.A.; Glenny, R.W.; Martin, T.R.; Liles, W.C. Modulation of Lipopolysaccharide-Induced Gene Transcription and Promotion of Lung Injury by Mechanical Ventilation. J. Immunol. 2005, 175, 3369–3376. [Google Scholar] [CrossRef] [Green Version]
- Jacobson, J.R.; Barnard, J.W.; Grigoryev, D.N.; Ma, S.-F.; Tuder, R.M.; Garcia, J.G.N. Simvastatin attenuates vascular leak and inflammation in murine inflammatory lung injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 2005, 288, L1026–L1032. [Google Scholar] [CrossRef]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef]
- Wagner, A.B. SciFinder Scholar 2006: An Empirical Analysis of Research Topic Query Processing. J. Chem. Inf. Model. 2006, 46, 767–774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.; Chen, J.; Cheng, T.; Gindulyte, A.; He, J.; He, S.; Li, Q.; Shoemaker, B.A.; Thiessen, P.A.; Yu, B.; et al. PubChem in 2021: New data content and improved web interfaces. Nucleic Acids Res. 2020, 49, D1388–D1395. [Google Scholar] [CrossRef] [PubMed]
- Daina, A.; Michielin, O.; Zoete, V. SwissTargetPrediction: Updated data and new features for efficient prediction of protein targets of small molecules. Nucleic Acids Res. 2019, 47, W357–W364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piñero, J.; Ramírez-Anguita, J.M.; Saüch-Pitarch, J.; Ronzano, F.; Centeno, E.; Sanz, F.; Furlong, L.I. The DisGeNET knowledge platform for disease genomics: 2019 update. Nucleic Acids Res. 2019, 48, D845–D855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Y.; Zhou, B.; Pache, L.; Chang, M.; Khodabakhshi, A.H.; Tanaseichuk, O.; Benner, C.; Chanda, S.K. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 2019, 10, 1523. [Google Scholar] [CrossRef]





| No. | Compounds | Molecular | Classification |
|---|---|---|---|
| 1 | Quinic acid | C7H12O6 | Phenolic acid |
| 2 | Chlorogenic acid | C16H18O9 | Phenolic acid |
| 3 | Apigenin 6-C-glucoside 8-C-arabinoside | C26H28O14 | Flavanone glycosides |
| 4 | Luteolin-7-O-rutinoside | C27H30O15 | Flavanone glycosides |
| 5 | Eriodictyol-7-O-glucoside | C21H22O11 | Flavanone glycosides |
| 6 | Luteolin-7-O-glucoside | C21H20O11 | Flavanone glycosides |
| 7 | Apigenin-7-O-glucoside | C21H20O10 | Flavanone glycosides |
| 8 | Rosmarinic acid | C18H16O8 | Phenolic acid |
| 9 | (2R)Eriodictyol 7-O-(6″-3,4-dihydroxycinnamoyl)-β-D-glucopyranoside | C30H28O14 | Flavanone glycosides |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, J.; Jiang, X.; Chen, Y.; Guo, S.; Zhao, Z.; Cao, J.; Liu, Y.; Cheng, G.; Li, Y.; Tian, L. Elsholtzia bodinieri Vaniot Ameliorated Acute Lung Injury by NQO1, BCL2 and PTGS2 In Silico and In Vitro Analyses. Int. J. Mol. Sci. 2022, 23, 15651. https://doi.org/10.3390/ijms232415651
Sun J, Jiang X, Chen Y, Guo S, Zhao Z, Cao J, Liu Y, Cheng G, Li Y, Tian L. Elsholtzia bodinieri Vaniot Ameliorated Acute Lung Injury by NQO1, BCL2 and PTGS2 In Silico and In Vitro Analyses. International Journal of Molecular Sciences. 2022; 23(24):15651. https://doi.org/10.3390/ijms232415651
Chicago/Turabian StyleSun, Jin, Xiaoqian Jiang, Yuxu Chen, Shancheng Guo, Zhiye Zhao, Jianxin Cao, Yaping Liu, Guiguang Cheng, Ye Li, and Lei Tian. 2022. "Elsholtzia bodinieri Vaniot Ameliorated Acute Lung Injury by NQO1, BCL2 and PTGS2 In Silico and In Vitro Analyses" International Journal of Molecular Sciences 23, no. 24: 15651. https://doi.org/10.3390/ijms232415651








