AICAR Improves Outcomes of Metabolic Syndrome and Type 2 Diabetes Induced by High-Fat Diet in C57Bl/6 Male Mice
Abstract
:1. Introduction
2. Results
2.1. Body Weight and Body Weight Gain
2.2. Food Consumption
2.3. Insulin Resistance Test
2.4. Oral Glucose Tolerance Test
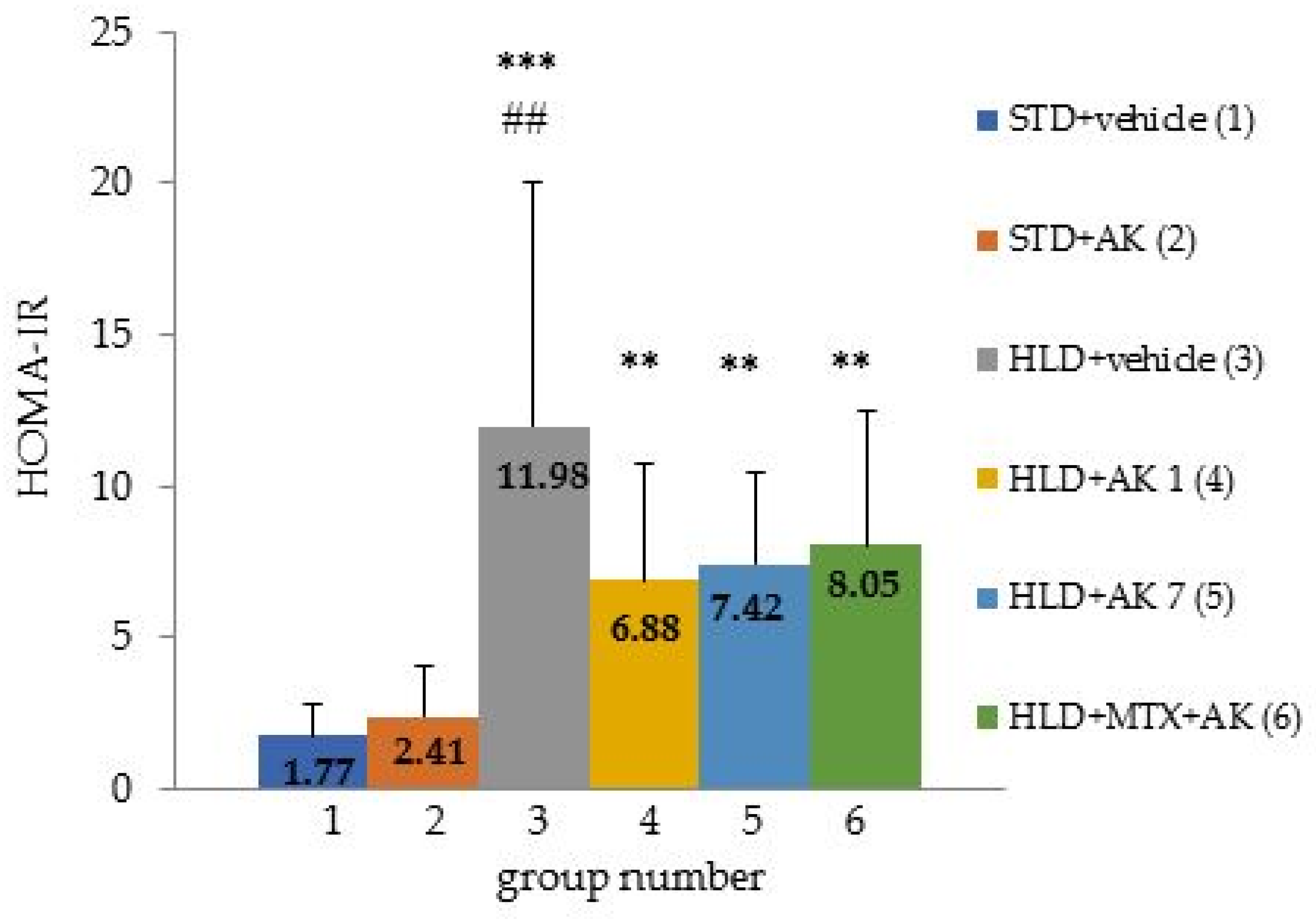
2.5. Insulin Resistance Assessment—HOMA-IR
2.6. Assessment of Locomotor Activity
2.7. Blood Chemistry
2.8. Necropsy Results
2.9. Organ Weight
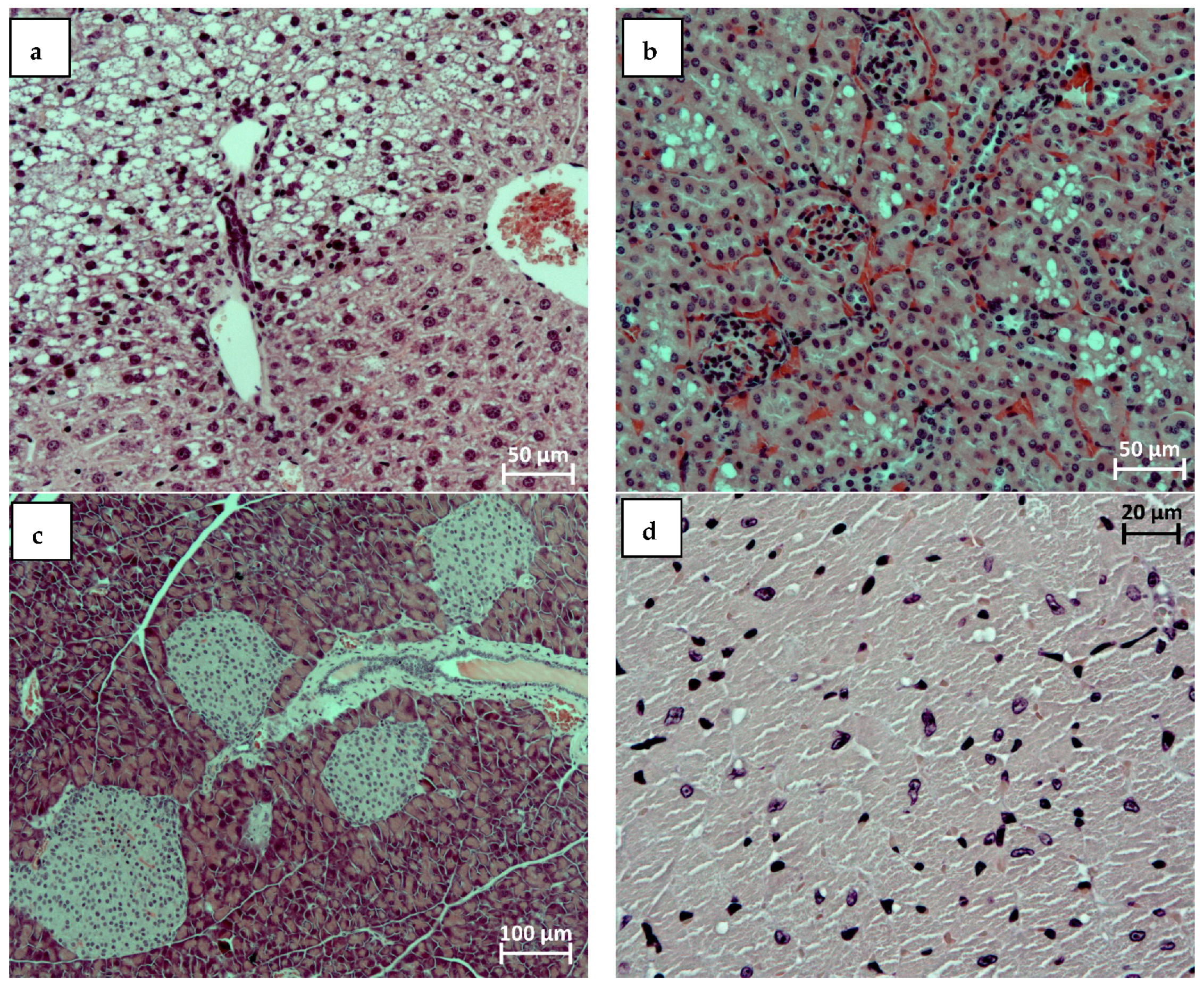
2.10. Histological Analysis Results
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Groups and Doses
4.3. Test Articles
4.4. Modeling Obesity, Metabolic Syndrome, and Diabetes Mellitus
4.5. Diet
4.5.1. Standard Diet (STD)
4.5.2. High Fat Diet (HFD)
4.6. Study Design
4.6.1. Food Deprivation
4.6.2. Glucometry
4.6.3. Insulin Resistance Test
4.6.4. Oral Glucose Tolerance Test
4.6.5. Assessment of Motor Activity
4.6.6. Blood Samples
4.6.7. Blood Chemistry
4.6.8. Enzyme Immunoassay for the Determination of Insulin
4.6.9. Organ Weight
4.6.10. Histopathology
4.6.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Favero, C.B.; Mandell, J.W. A pharmacological activator of AMP-activated protein kinase (AMPK) induces astrocyte stellation. Brain Res. 2007, 1168, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leung, J.M.; Stanley, T.; Mathew, J.; Curling, P.; Barash, P.; Salmenpera, M.; Reves, J.G.; Hollenberg, M.; Mangano, D.T. An initial multicenter, randomized controlled trial on the safety and efficacy of acadesine in patients undergoing coronary artery bypass graft surgery. Anesth. Analg. 1994, 78, 420–434. [Google Scholar] [CrossRef] [PubMed]
- AICAR. DB04944. Logo White. 2020. Available online: https://www.drugbank.ca/drugs/DB04944 (accessed on 30 September 2022).
- Cronstein, B.N.; Kamen, B.A. 5-Aminoimidazole-4-Carboxamide-1-β-4-Ribofuranoside (AICA-riboside) as a Targeting Agent for Therapy of Patients with Acute Lymphoblastic Leukemia: Are We There and Are There Pitfalls? J. Pediatr. Hematol. Oncol. 2007, 29, 805–807. [Google Scholar] [CrossRef] [PubMed]
- Cuthbertson, D.J.; Babraj, J.A.; Mustard, K.J.W.; Towler, M.C.; Green, K.A.; Wackerhage, H.; Leese, G.P.; Baar, K.; Thomason-Hughes, M.; Sutherland, C.; et al. 5-Aminoimidazole-4-Carboxamide 1-β-D-Ribofuranoside Acutely Stimulates Skeletal Muscle 2-Deoxyglucose Uptake in Healthy Men. Diabetes 2007, 56, 2078–2084. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viglino, C.; Foglia, B.; Montessuit, C. Chronic AICAR treatment prevents metabolic changes in cardiomyocytes exposed to free fatty acids. Pflüg. Arch. Eur. J. Physiol. 2019, 471, 1219–1234. [Google Scholar] [CrossRef]
- Henin, N.; Vincent, M.-F.; Gruber, H.E.; Van Den Berghe, G. Inhibition of fatty acid and cholesterol synthesis by stimulation of AMP-activated protein kinase. FASEB J. 1995, 9, 541–546. [Google Scholar] [CrossRef]
- Velasco, G.; Geelen, M.J.H.; Guzmán, M. Control of Hepatic Fatty Acid Oxidation by 5′-AMP-Activated Protein Kinase Involves a Malonyl-CoA-Dependent and a Malonyl-CoA-Independent Mechanism. Arch. Biochem. Biophys. 1997, 337, 169–175. [Google Scholar] [CrossRef]
- Lochhead, P.A.; Salt, I.P.; Walker, K.S.; Hardie, D.G.; Sutherland, C. 5-aminoimidazole-4-carboxamide riboside mimics the effects of insulin on the expression of the 2 key gluconeogenic genes PEPCK and glucose-6-phosphatase. Diabetes 2000, 49, 896–903. [Google Scholar] [CrossRef] [Green Version]
- Young, M.E.; Radda, G.K.; Leighton, B. Activation of glycogen phosphorylase and glycogenolysis in rat skeletal muscle by AICAR—An activator of AMP-activated protein kinase. FEBS Lett. 1996, 382, 43–47. [Google Scholar] [CrossRef] [Green Version]
- Merrill, G.F.; Kurth, E.J.; Hardie, D.G.; Winder, W.W. AICA riboside increases AMP-activated protein kinase, fatty acid oxidation, and glucose uptake in rat muscle. Am. J. Physiol. Metab. 1997, 273, E1107–E1112. [Google Scholar] [CrossRef]
- Bolster, D.R.; Crozier, S.J.; Kimball, S.R.; Jefferson, L.S. AMP-activated Protein Kinase Suppresses Protein Synthesis in Rat Skeletal Muscle through Down-regulated Mammalian Target of Rapamycin (mTOR) Signaling. J. Biol. Chem. 2002, 277, 23977–23980. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Russell, R.R.; Bergeron, R.; Shulman, G.I.; Young, L.H. Translocation of myocardial GLUT-4 and increased glucose uptake through activation of AMPK by AICAR. Am. J. Physiol. Heart Circ. Physiol. 1999, 277, H643–H649. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.R.; Li, J.; Coven, D.L.; Pypaert, M.; Zechner, C.; Palmeri, M.; Giordano, F.J.; Mu, J.; Birnbaum, M.J.; Young, L.H. AMP-activated protein kinase mediates ischemic glucose uptake and prevents postischemic cardiac dysfunction, apoptosis, and injury. J. Clin. Investig. 2004, 114, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Salvadó, L.; Coll, T.; Gómez-Foix, A.M.; Salmerón, E.; Barroso, E.; Palomer, X.; Vázquez-Carrera, M. Oleate prevents saturatedfatty-acid-induced ER stress, inflammation and insulin resistance in skeletal muscle cells through an AMPK-dependent mechanism. Diabetologia 2013, 56, 1372–1382. [Google Scholar] [CrossRef] [PubMed]
- Choi, R.H.; McConahay, A.; Johnson, M.B.; Jeong, H.-W.; Koh, H.-J. Adipose tissue-specific knockout of AMPKα1/α2 results in normal AICAR tolerance and glucose metabolism. Biochem. Biophys. Res. Commun. 2019, 519, 633–638. [Google Scholar] [CrossRef] [PubMed]
- Song, X.M.; Fiedler, M.; Galuska, D.; Ryder, J.W.; Fernström, M.; Chibalin, A.V.; Wallberg-Henriksson, H.; Zierath, J.R. 5-Aminoimidazole-4-carboxamide ribonucleoside treatment improves glucose homeostasis in insulin-resistant diabetic (ob/ob) mice. Diabetologia 2002, 45, 56–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buhl, E.S.; Jessen, N.; Pold, R.; Ledet, T.; Flyvbjerg, A.; Pedersen, S.B.; Pedersen, O.; Schmitz, O.; Lund, S. Long-Term AICAR Administration Reduces Metabolic Disturbances and Lowers Blood Pressure in Rats Displaying Features of the Insulin Resistance Syndrome. Diabetes 2002, 51, 2199–2206. [Google Scholar] [CrossRef] [Green Version]
- Iglesias, M.A.; Ye, J.-M.; Frangioudakis, G.; Saha, A.K.; Tomas, E.; Ruderman, N.B.; Cooney, G.J.; Kraegen, E.W. AICAR Administration Causes an Apparent Enhancement of Muscle and Liver Insulin Action in Insulin-Resistant High-Fat-Fed Rats. Diabetes 2002, 51, 2886–2894. [Google Scholar] [CrossRef] [Green Version]
- Pold, R.; Jensen, L.S.; Jessen, N.; Buhl, E.S.; Schmitz, O.; Flyvbjerg, A.; Fujii, N.; Goodyear, L.J.; Gotfredsen, C.F.; Brand, C.L.; et al. Long-Term AICAR Administration and Exercise Prevents Diabetes in ZDF Rats. Diabetes 2005, 54, 928–934. [Google Scholar] [CrossRef] [Green Version]
- Boon, H.; Bosselaar, M.; Praet, S.F.E.; Blaak, E.E.; Saris, W.H.M.; Wagenmakers, A.J.M.; McGee, S.L.; Tack, C.J.; Smits, P.; Hargreaves, M.; et al. Intravenous AICAR administration reduces hepatic glucose output and inhibits whole body lipolysis in type 2 diabetic patients. Diabetologia 2008, 51, 1893–1900. [Google Scholar] [CrossRef]
- Borgeson, E.; Sharma, K. Obesity, immunomodulation and chronic kidney disease. Curr. Opin. Pharmacol. 2013, 13, 618–624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mathew, A.V.; Okada, S.; Sharma, K. Obesity related kidney disease. Curr. Diabetes 2011, 7, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Samson Susan, L.; Garber Alan, J. Metabolic Syndrome. Endocrinol. Metab. Clin. N. Am. 2014, 43, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Börgeson, E.; Wallenius, V.; Syed, G.H.; Darshi, M.; Rodriguez, J.L.; Biörserud, C.; Ek, M.R.; Björklund, P.; Quiding-Järbrink, M.; Fändriks, L.; et al. AICAR ameliorates high-fat diet-associated pathophysiology in mouse and ex vivo models, independent of adiponectin. Diabetologia 2017, 60, 729–739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Halseth, A.E.; Ensor, N.J.; White, T.A.; Ross, S.A.; Gulve, E.A. Acute and chronic treatment of ob/ob and db/db mice with AICAR decreases blood glucose concentrations. Biochem. Biophys. Res. Commun. 2002, 294, 798–805. [Google Scholar] [CrossRef]
- Dixon, R.; Gourzis, J.; McDermott, D.; Fujitaki, J.; Dewland, P.; Gruber, H. AICA-Riboside: Safety, Tolerance, and Pharmacokinetics of a Novel Adenosine-Regulating Agent. J. Clin. Pharmacol. 1991, 31, 342–347. [Google Scholar] [CrossRef]
- Pirkmajer, S.; Kulkarni, S.S.; Tom, R.Z.; Ross, F.A.; Hawley, S.A.; Hardie, D.G.; Zierath, J.R.; Chibalin, A.V. Methotrexate promotes glucose uptake and lipid oxidation in skeletal muscle via AMPK activation. Diabetes 2015, 64, 360–369. [Google Scholar] [CrossRef] [Green Version]
- Decleves, A.-E.; Mathew, A.V.; Cunard, R.; Sharma, K. AMPK Mediates the Initiation of Kidney Disease Induced by a High-Fat Diet. J. Am. Soc. Nephrol. 2011, 22, 1846–1855. [Google Scholar] [CrossRef] [Green Version]
- Winzell, M.S.; Ahren, B. The High-Fat Diet-Fed Mouse: A Model for Studying Mechanisms and Treatment of Impaired Glucose Tolerance and Type 2 Diabetes. Diabetes 2004, 53 (Suppl. S3), S215–S219. [Google Scholar] [CrossRef] [Green Version]
- Sato, M.; Goto, T.; Inoue, E.; Miyaguchi, Y.; Toyoda, A. Dietary Intake of Immature Citrus tumida Hort. ex Tanaka Peels Suppressed Body Weight Gain and Fat Accumulation in a Mouse Model of Acute Obesity. J. Nutr. Sci. Vitaminol. 2019, 65, 19–23. [Google Scholar] [CrossRef]
- Nagy, C.; Einwallner, E. Study of In Vivo Glucose Metabolism in High-fat Diet-fed Mice Using Oral Glucose Tolerance Test (OGTT) and Insulin Tolerance Test (ITT). J. Vis. Exp. 2018, 131, e56672. Available online: https://www.jove.com/video/56672 (accessed on 19 October 2022). [CrossRef] [PubMed]
- Heydemann, A. An Overview of Murine High Fat Diet as a Model for Type 2 Diabetes Mellitus. J. Diabetes Res. 2016, 2016, 2902351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, J.; Quan, J.; Tsai, J.; Hobensack, C.K.; Sullivan, C.; Hector, R.; Reaven, G.M. Nongenetic mouse models of non-insulin-dependent diabetes mellitus. Metabolism 1998, 47, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Collins, S.; Martin, T.L.; Surwit, R.S.; Robidoux, J. Genetic vulnerability to diet-induced obesity in the C57BL/6J mouse: Physiological and molecular characteristics. Physiol. Behav. 2004, 81, 243–248. [Google Scholar] [CrossRef]
- Freeman, H.C.; Hugill, A.; Dear, N.T.; Ashcroft, F.M.; Cox, R.D. Deletion of nicotinamide nucleotide transhydrogenase: A new quantitive trait locus accounting for glucose intolerance in C57BL/6J mice. Diabetes 2006, 55, 2153–2156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, A.; Terauchi, Y. Lessons from Mouse Models of High-Fat Diet-Induced NAFLD. Int. J. Mol. Sci. 2013, 14, 21240. [Google Scholar] [CrossRef] [Green Version]
- Deji, N.; Kume, S.; Araki, S.; Soumura, M.; Sugimoto, T.; Isshiki, K.; Chin-Kanasaki, M.; Sakaguchi, M.; Koya, D.; Haneda, M.; et al. Structural and functional changes in the kidneys of high-fat diet-induced obese mice. Am. J. Physiol. Ren. Physiol. 2009, 296, F118–F126. [Google Scholar] [CrossRef] [Green Version]
- Huang, P.L. A comprehensive definition for metabolic syndrome. Dis. Model. Mech. 2009, 2, 231–237. [Google Scholar] [CrossRef] [Green Version]
- Glastras, S.J.; Chen, H.; Teh, R.; McGrath, R.T.; Chen, J.; Pollock, C.A.; Wong, M.G.; Saad, S. Mouse Models of Diabetes, Obesity and Related Kidney Disease. PLoS ONE 2016, 11, e0162131. [Google Scholar] [CrossRef] [Green Version]
- Giri, S.; Rattan, R.; Haq, E.; Khan, M.; Yasmin, R.; Won, J.S.; Key, L.; Singh, A.K.; Singh, I. AICAR inhibits adipocyte differentiation in 3T3L1 and restores metabolic alterations in diet-induced obesity mice model. Nutr. Metab. 2006, 3, 31. [Google Scholar] [CrossRef]
- Sun, Z.; Li, L.; Yan, Z.; Zhang, L.; Zang, G.; Qian, Y.; Wang, Z. Circadian rhythm disorders elevate macrophages cytokines release and promote multiple tissues/organs dysfunction in mice. Physiol. Behav. 2022, 249, 113772. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Loots, D.T. Experimental rodent models of type 2 diabetes: A review. Methods Find. Exp. Clin. Pharmacol. 2009, 31, 249–261. [Google Scholar] [CrossRef] [PubMed]
- Samsu, N. Diabetic Nephropathy: Challenges in Pathogenesis, Diagnosis, and Treatment. BioMed Res. Int. 2021, 2021, 1497449. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, S.; Sawa, N.; Mizuno, H.; Yamanouchi, M.; Suwabe, T.; Hoshino, J.; Kinowaki, K.; Ohashi, K.; Fujii, T.; Yamaguchi, Y.; et al. Development of osmotic vacuolization of proximal tubular epithelial cells following treatment with sodium-glucose transport protein 2 inhibitors in type II diabetes mellitus patients-3 case reports. CEN Case Rep. 2021, 10, 563–569. [Google Scholar] [CrossRef]
- Alicic, R.Z.; Cox, E.J.; Neumiller, J.J.; Tuttle, K.R. Incretin drugs in diabetic kidney disease: Biological mechanisms and clinical evidence. Nat. Rev. Nephrol. 2020, 17, 227–244. [Google Scholar] [CrossRef]
- Efstathiou, N.E.; Moustafa, G.A.; Maidana, D.E.; Konstantinou, E.K.; Notomi, S.; Barbisan, P.R.T.; Georgakopoulos, C.D.; Miller, J.W.; Vavvas, D.G. Acadesine suppresses TNF-α induced complement component 3 (C3), in retinal pigment epithelial (RPE) cells. PLoS ONE 2020, 15, e0244307. [Google Scholar] [CrossRef]
- Kwo, P.Y.; Cohen, S.M.; Lim, J.K. ACG clinical guideline: Evaluation of abnormal liver chemistries. Am. J. Gastroenterol. 2017, 112, 18–35. [Google Scholar] [CrossRef]
- Burtis, C.A.; Ashwood, E.R.; Bruns, D.E. Tietz Textbook of Clinical Chemistry and Molecular Diagnostics; Elsevier Health Sciences: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Ross, R.; Neeland, I.J.; Yamashita, S.; Shai, I.; Seidell, J.; Magni, P.; Santos, R.D.; Arsenault, B.; Cuevas, A.; Hu, F.B.; et al. Waist circumference as a vital sign in clinical practice: A Consensus Statement from the IAS and ICCR Working Group on Visceral Obesity. Nat. Rev. Endocrinol. 2020, 16, 177–189. [Google Scholar] [CrossRef] [Green Version]
- Ross, R.; Aru, J.; Freeman, J.; Hudson, R.; Janssen, I. Abdominal adiposity and insulin resistance in obese men. Am. J. Physiol. Endocrinol. Metab. 2002, 282, E657–E663. [Google Scholar] [CrossRef] [Green Version]
- Després, J.-P.; Lemieux, I. Abdominal obesity and metabolic syndrome. Nature 2006, 444, 881–887. [Google Scholar] [CrossRef]
- Lee, B.-C.; Lee, J. Cellular and molecular players in adipose tissue inflammation in the development of obesity-induced insulin resistance. Biochim. Biophys. Acta Mol. Basis Dis. 2014, 1842, 446–462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Puigserver, P.; Spiegelman, B.M. Peroxisome proliferator-activated receptor-gamma coactivator 1 alpha (PGC-1 alpha): Transcriptional coactivator and metabolic regulator. Endocr. Rev. 2003, 24, 78–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lehman, J.J.; Barger, P.M.; Kovacs, A.; Saffitz, J.E.; Medeiros, D.M.; Kelly, D.P. Peroxisome proliferator-activated receptor gamma coactivator-1 promotes cardiac mitochondrial biogenesis. J. Clin. Investig. 2000, 106, 847–856. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kale, M.; Nimje, N.; Aglawe, M.M.; Umekar, M.; Taksande, B.; Kotagale, N. Agmatine Modulates Anxiety and Depression-like Behaviour in Diabetic Insulin-Resistant Rats. Brain Res. 2020, 1747, 147045. [Google Scholar] [CrossRef] [PubMed]
- Mironov, A.S.; Ehrrais Lopes, L.; Korol’kova, N.V.; Muratova, V.A.; Tjaglov, B.V.; Nudler, E.A.; Shakulov, R.S.; Debabov, V.G.; Beburov, M.J. Method for Microbiologial Synthesis of Purine Nucleoside of 5′-Aminoimidazole-4-Carboxamide Riboside (Aicar) and Bacillus Subtilis Strain—Producer of Aicar. Patent RU2405833C2, 10 December 2020. Available online: https://patents.google.com/patent/RU2405833C2/en (accessed on 30 September 2022).
- Shi, S.-Q.; Ansari, T.S.; McGuinness, O.P.; Wasserman, D.H.; Johnson, C.H. Circadian disruption leads to insulin resistance and obesity. Curr. Biol. 2013, 23, 372–381. [Google Scholar] [CrossRef] [Green Version]
- Russo, G.T.; Minutoli, L.; Bitto, A.; Altavilla, D.; Alessi, E.; Giandalia, A.; Romeo, E.L.; Stagno, M.F.; Squadrito, F.; Cucinotta, D.; et al. Methotrexate Increases Skeletal Muscle GLUT4 Expression and Improves Metabolic Control in Experimental Diabetes. J. Nutr. Metab. 2012, 2012, 132056. [Google Scholar] [CrossRef]
| Week of the Study | Mean Body Weight, g | |||||
|---|---|---|---|---|---|---|
| Group 1 STD + Vehicle | Group 2 STD + AC 1 | Group 3 HFD + Vehicle | Group 4 HFD + AC 1 | Group 5 HFD + AC 7 | Group 6 HFD + MTX + AC 7 | |
| n = 10 | n = 10 | n = 10 | n = 10 | n = 10 & | n = 10 $ | |
| 0 | 21.4 ± 1.5 | 21.3 ± 1.7 | 21.7 ± 1.3 | 21.6 ± 1.2 | 21.7 ± 1.4 | 21.2 ± 2.1 |
| 1 | 23.2 ± 1.2 | 23.8 ± 1.1 | 23.4 ± 1.4 | 23.3 ± 1.6 | 22.8 ± 1.0 | 23.1 ± 1.3 |
| 2 | 24.1 ± 0.9 | 25.2 ± 1.5 | 24.2 ± 1.4 | 24.2 ± 1.9 | 23.6 ± 1.2 | 24.0 ± 1.4 |
| 3 | 24.1 ± 0.9 | 25.4 ± 1.7 | 24.6 ± 1.5 | 24.9 ± 1.9 | 24.1 ± 1.4 | 24.5 ± 1.3 |
| 4 | 24.7 ± 1.1 | 26.1 ± 1.5 | 26.8 ± 2.0 *** | 25.9 ± 1.9 | 26.4 ± 1.7 * | 26.7 ± 1.9 * |
| 5 | 25.6 ± 1.1 | 27.0 ± 1.6 | 28.5 ± 2.2 *** | 27.6 ± 2.2 * | 28.5 ± 2.2 ** | 28.6 ± 2.4 ** |
| 6 | 25.7 ± 1.2 | 27.1 ± 1.6 @@ | 30.4 ± 2.7 *** | 28.7 ± 2.0 | 30.3 ± 2.4 # | 30.3 ± 3.0 # |
| 7 | 26.1 ± 1.0 | 27.6 ± 1.6 @@ | 30.6 ± 2.6 *** | 29.2 ± 2.1 ** | 29.2 ± 2.6 ** | 29.5 ± 2.6 *** |
| 8 | 26.5 ± 1.1 | 27.9 ± 1.6 @@ | 30.5 ± 2.6 *** | 29.0 ± 2.2 ** | 28.8 ± 2.1 * | 29.2 ± 2.7 ** |
| 9 | 27.0 ± 1.0 | 28.2 ± 1.6 @@ | 31.7 ± 2.5 *** | 29.9 ± 2.8 ** | 29.3 ± 2.4 * | 29.2 ± 2.7 *@ |
| 10 | 27.0 ± 1.2 | 28.5 ± 1.5 @@@ | 32.2 ± 2.7 *** | 30.8 ± 2.7 ***# | 29.4 ± 2.4 **@ | 29.4 ± 2.8 *@@ |
| 11 | 27.5 ± 1.2 | 28.6 ± 1.5 @@@ | 32.9 ± 3.0 *** | 30.9 ± 2.9 **# | 29.9 ± 2.2 *@ | 29.6 ± 3.0 @ |
| 12 | 27.3 ± 1.3 | 29.0 ± 1.3 @@@ | 33.6 ± 3.3 *** | 31.7 ± 3.0 ***# | 30.4 ± 2.4 **@ | 30.3 ± 3.3 **@@ |
| Week of the Study | Average Weight Gain Relative to the First Day of the Study, % | |||||
|---|---|---|---|---|---|---|
| Group 1 STD + vehicle | Group 2 STD + AC 1 | Group 3 HFD + vehicle | Group 4 HFD + AC 1 | Group 5 HFD + AC 7 | Group 6 HFD + MTX + AC 7 | |
| n = 10 | n = 10 | n = 10 | n = 10 | n = 10 & | n = 10 $ | |
| 1 | 8.8 ± 5.0 | 12.2 ± 7.0 | 8.0 ± 5.7 | 7.8 ± 7.6 | 5.1 ± 4.5 | 9.2 ± 6.2 |
| 2 | 13.1 ± 6.2 | 19.0 ± 12.1 | 11.7 ± 5.5 | 12.2 ± 10.9 | 8.9 ± 5.0 | 13.5 ± 7.0 |
| 3 | 13.5 ± 7.7 | 19.8 ± 12.8 | 13.9 ± 6.3 | 15.3 ± 11.2 | 11.6 ± 7.3 | 15.7 ± 6.7 |
| 4 | 16.2 ± 7.7 | 23.0 ± 12.5 | 24.0 ± 8.0 | 20.1 ± 11. | 22.0 ± 7.8 | 26.1 ± 5.9 * |
| 5 | 20.1 ± 8.4 | 27.3 ± 13.7 | 31.6 ± 9.2 * | 28.0 ± 13.9 | 31.7 ± 10.5 * | 35.2 ± 8.8 ** |
| 6 | 20.7 ± 8.3 | 28.1 ± 13.6 @ | 40.5 ± 11.2 *** | 33.1 ± 12.2 * | 40.1 ± 11.2 ***# | 42.9 ± 10.5 ***# |
| 7 | 22.6 ± 6.9 | 30.4 ± 14.0 @ | 41.7 ± 12.4 *** | 35.3 ± 13.6 * | 35.2 ± 14.2 * | 39.6 ± 11.1 ** |
| 8 | 24.3 ± 8.7 | 31.7 ± 14.4 | 41.3 ± 12.8 ** | 34.3 ± 13.5 | 33.1 ± 12.1 | 38.6 ± 10.5 ** |
| 9 | 26.7 ± 9.0 | 32.9 ± 13.6 @ | 46.6 ± 12.6 *** | 38.7 ± 16.7 * | 35.2 ± 11.9 | 38.6 ± 13.0 |
| 10 | 26.6 ± 8.5 | 34.4 ± 14.3 @@ | 49.0 ± 13.2 *** | 42.8 ± 16.4 ** | 36.0 ± 12.8 | 39.8 ± 14.0 * |
| 11 | 29.4 ± 8.4 | 34.9 ± 14.1 @@ | 52.0 ± 14.9 *** | 43.1 ± 16.6 * | 38.6 ± 12.7 @ | 40.6 ± 13.0 |
| 12 | 28.0 ± 6.0 | 36.8 ± 13.0 @@ | 55.6 ± 15.7 *** | 47.2 ± 17.8 ** | 40.8 ± 13.8 * | 43.4 ± 11.8 * |
| Group | Group 1 STD + Vehicle | Group 2 STD + AC 1 | Group 3 HFD + Vehicle | Group 4 HFD + AC 1 | Group 5 HFD + AC 7 | Group 6 HFD + MTX + AC 7 |
|---|---|---|---|---|---|---|
| n = 5 | n = 5 | n = 5 | n = 5 | n = 4 & | n = 4 $ | |
| Initial values, mmol/L | 5.9 ± 1.5 | 6.9 ± 1.1 | 7.8 ± 2.2 | 8.9 ± 1.5 **## | 8.5 ± 1.3 ** | 9.1 ± 0.7 **## |
| After 20 min, mmol/L | 4.1 ± 0.3 ^ | 3.5 ± 0.5 ^^^ | 5.2 ± 1.0 *##^^ | 6.1 ± 1.1 ***###^^^ | 4.9 ± 0.6 #^^^ | 6.2 ± 0.7 ***###^^^ |
| After 40 min, mmol/L | 3.1 ± 07 ^^^ | 3.1 ± 0.4 ^^^ | 4.6 ± 0.4 **##^^ | 3.7 ± 0.6 ^^^ | 3.4 ± 0.4 @^^^ | 4.1 ± 1.5^^^ |
| After 60 min, mmol/L | 2.7 ± 1.0 ^^^ | 2.7 ± 0.7 ^^^ | 3.6 ± 04 *#^^^ | 3.5 ± 0.7 ^^^ | 3.0 ± 0.3 ^^^ | 2.8 ± 0.4 ^^^ |
| After 120 min, mmol/L | 3.2 ± 1.6 ^^^ | 2.1 ± 1.0 ^^^ | 3.6 ± 1.4 ^^^ | 3.3 ± 0.5 ^^^ | 2.9 ± 0.4 ^^^ | 3.5 ± 0.5 ^^^ |
| Week of the Study | Mean Food Consumption, % | |||||
|---|---|---|---|---|---|---|
| Group 1 STD + Vehicle | Group 2 STD + AC 1 | Group 3 HFD + Vehicle | Group 4 HFD + AC 1 | Group 5 HFD + AC 7 | Group 6 HFD + MTX + AC 7 | |
| n = 10 | n = 10 | n = 10 | n = 10 | n = 10 & | n = 10 $ | |
| 1 | 171.1 ± 14.8 | 161.3 ± 16.8 | 104.9 ± 21.1 ***### | 103.1 ± 31.1 ***### | 69.5 ± 31.2 ***### | 91.8 ± 25.4 ***### |
| 3 | 191.0 ± 41.2 | 194.9 ± 27.6 | 130.0 ± 20.0 ***### | 118.7 ± 30.4 ***### | 120.9 ± 9.3 ***### | 135.1 ± 16.9 ***### |
| 5 | 156.6 ± 14.5 ^^%%% | 162.6 ± 12.0 ^^%% | 104.8 ± 11.2 ***###^^^%%%ββ | 124.2 ± 27.2 ***###@@ | 110.8 ± 18.5 ***###%®®® | 112.1 ± 11.6 ***###^% |
| 7 | 196.3 ± 19.3 % | 206.8 ± 31.2 ^^%% | 128.9 ± 7.1 ***###^^^%%% | 121.4 ± 29.0 ***### | 128.2 ± 12.9 ***###§^^^%%%®® | 132.3 ± 10.1 ***###^^^%%% |
| 9 | 143.6 ± 13.1 ^^^%®® | 160.2 ± 29.6 ^^^%%% | 82.8 ± 10.1 ***###^^^® | 102.1 ± 21.1 ***### | 98.5 ± 20.1 ***###^%%% | 106.0 ± 29.8 ***###@^^% |
| 11 | 138.7 ± 33.2 ^^^%® | 143.8 ± 20.5 ^^^%%% | 118.4 ± 16.2 #^^^βββ | 122.1 ± 17.8 # | 115.3 ± 16.2 *#®®® | 97.8 ± 31.8 ***###§^^^%% |
| Group | Group 1 STD + Vehicle | Group 2 STD + AC 1 | Group 3 HFD + Vehicle | Group 4 HFD + AC 1 | Group 5 HFD + AC 7 | Group 6 HFD + MTX + AC 7 |
|---|---|---|---|---|---|---|
| n = 10 | n = 10 | n = 10 | n = 10 | n = 9 & | n = 9 $ | |
| Insulin, mIU/L | 6.23 ± 5.20 | 7.82 ± 4.10 | 32.92 ± 15.09 * | 18.31 ± 9.50 @ | 19.58 ± 6.74 *@ | 21.81 ± 11.36 * |
| Glucose, mmol/L | 6.05 ± 1.18 | 6.52 ± 1.03 | 7.63 ± 1.78 € | 8.36 ± 1.65 * | 8.44 ± 0.88 * | 8.7 ± 11.36 * |
| Group | Group 1 STD + Vehicle | Group 2 STD + AC 1 | Group 3 HFD + Vehicle | Group 4 HFD + AC 1 | Group 5 HFD + AC 7 | Group 6 HFD + MTX + AC 7 |
|---|---|---|---|---|---|---|
| n = 5 | n = 5 | n = 5 | n = 5 | n = 5 | n = 5 | |
| Initial values, mmol/L | 5.2 ± 0.4 | 6.2 ± 0.9 | 7.4 ± 1.5 ** | 7.9 ± 1.8 ***## | 8.4 ± 0.6 ***## | 7.4 ± 0.9 ** |
| After 30 min, mmol/L | 12.0 ± 1.0 βββ | 12.1 ± 1.6 βββ | 13.0 ± 1.7 βββ | 13.7 ± 1.0 βββ | 14.9 ± 2.7 **##βββ | 14.7 ± 1.0 *#βββ |
| After 60 min, mmol/L | 8.2 ± 1.7 βββψψψ | 7.9 ± 1.2 βψψψ | 10.1 ± 1.8 ##ββψψψ | 10.7 ± 1.5 *##ββψψ | 11.5 ± 1.0 **##ββψψ | 11.0 ± 1.8 **##βββψψψ |
| After 90 min, mmol/L | 6.9 ± 0.5 ββψψψ | 7.3 ± 0.8 ψψψ | 8.4 ± 1.2 *ψψ | 9.3 ± 0.8 ***##ψψψ | 10.2 ± 0.3 ***###@@ψψψ | 10.1 ± 1.4 ***###@@ββψψψ |
| After 120 min, mmol/L | 6.3 ± 0.7 ψψψ | 8.5 ± 1.3 *ββψψψ | 10.2 ± 1.4 ***ββψψ | 9.8 ± 1.8 ***ψψψ | 11.5 ± 1.9 ***##ββψψ | 10.0 ± 1.1 ***ββψψψ |
| Group | Group 1 STD + Vehicle | Group 2 STD + AC 1 | Group 3 HFD + Vehicle | Group 4 HFD + AC 1 | Group 5 HFD + AC 7 | Group 6 HFD + MTX + AC 7 |
|---|---|---|---|---|---|---|
| n = 10 | n = 10 | n = 10 | n = 10 | n = 9 & | n = 9 $ | |
| Urea, mmol/L | 10.7 ± 0.8 | 9.5 ± 1.0 | 6.9 ± 0.7 * | 6.7 ± 1.0 ** | 6.0 ± 0.5 ***## | 6.2 ± 0.9 ***## |
| Cholesterol, mmol/L | 2.68 ± 0.34 | 2.28 ± 0.42 | 5.50 ± 0.69 ***### | 4.67 ± 0.52 *##@@ | 4.71 ± 0.38 *#@ | 4.53 ± 0.43 #@@ |
| Triglycerides, mmol/L | 1.33 ± 0.25 | 1.04 ± 0.24 | 1.62 ± 0.64 | 1.02 ± 0.25 | 1.20 ± 0.15 | 0.97 ± 0.15 *@ |
| ALT, U/L | 63 ± 8 | 54 ± 13 | 37 ± 7 ** | 35 ± 10 ***# | 35 ± 7 ***# | 31 ± 4 ***## |
| AST, U/L | 67 ± 6 | 61 ± 13 | 57 ± 4 | 57 ± 8 | 58 ± 6 | 53 ± 4 ** |
| Total bilirubin, mmol/L | 7.6 ± 1.6 | 8.2 ± 1.1 | 6.4 ± 2.6 | 6.5 ± 1.8 | 5.1 ± 1.8 ## | 6.6 ± 1.6 |
| Creatinine, mmol/L | 48 ± 7 | 48 ± 4 | 47 ± 4 | 46 ± 4 | 46 ± 8 | 41 ± 4 |
| Alkaline phosphatase, U/L | 98 ± 9 | 88 ± 11 | 54 ± 7 ***## | 54 ± 8 **# | 54 ± 8 **# | 50 ± 5 ***### |
| Albumen, g/L | 41.9 ± 2.2 | 39.0 ± 2.1 | 40.6 ± 1.7 | 38.8 ± 1.4 * | 39.8 ± 2.0 | 38.4 ± 2.0 * |
| Calcium, mmol/L | 2.86 ± 0.19 | 2.72 ± 0.13 | 2.84 ± 0.14 | 2.75 ± 0.08 | 2.81 ± 0.15 | 2.66 ± 0.11 |
| Phosphates, mmol/L | 3.94 ± 0.53 | 3.63 ± 0.34 | 3.59 ± 0.50 | 3.62 ± 0.34 | 3.60 ± 0.39 | 3.28 ± 0.17 * |
| Total protein, g/L | 65.1 ± 3.9 | 61.7 ± 2.9 | 65.6 ± 2.7 | 63.1 ± 3.3 | 63.2 ± 4,0 | 61.7 ± 3.2 |
| Chlorides, mmol/L | 127.1 ± 3.5 | 126.0 ± 2.1 | 130.4 ± 4.1 | 128.6 ± 7.8 | 126 ± 3 | 124 ± 2 @ |
| Globulin, g/L | 23.1 ± 2.1 | 22.6 ± 1.0 | 25.0 ± 1.5 | 24.3 ± 2.3 | 23.4 ± 2.4 | 23.3 ± 1.5 |
| Albumin/globulin | 1.8 ± 0.1 | 1.7 ± 0.1 | 1.6 ± 0.1 ** | 1.6 ± 0.1 ** | 1.7 ± 0.1 | 1.7 ± 0.1 * |
| Parameter | Group 1 STD + Vehicle | Group 2 STD + AC 1 | Group 3 HFD + Vehicle | Group 4 HFD + AC 1 | Group 5 HFD + AC 7 | Group 6 HFD + MTX + AC 7 |
|---|---|---|---|---|---|---|
| n = 10 | n = 10 | n = 10 | n = 10 | n = 9 & | n = 9 $ | |
| Resting time (s) | 105 ± 19 | 105 ± 23 | 109 ± 21 | 99 ± 11 | 101 ± 29 | 101 ± 12 |
| Moving time (s) | 75 ± 19 | 75 ± 23 | 71 ± 21 | 81 ± 11 | 79 ± 29 | 79 ± 12 |
| Distance (m) | 16 ± 4 | 17 ± 5 | 14 ± 5 | 16 ± 2 | 16 ± 6 | 16 ± 3 |
| Time in center (s) | 53 ± 54 | 61 ± 52 | 32 ± 28 | 39 ± 28 | 32 ± 43 | 39 ± 41 |
| Distance in center (m) | 4 ± 3 | 5 ± 1 | 2 ± 1 ## | 3 ± 2 | 2 ± 1 # | 2 ± 2 # |
| Periphery time (s) | 127 ± 54 | 119 ± 52 | 148 ± 28 | 141 ± 38 | 148 ± 43 | 141 ± 41 |
| Periphery distance (m) | 12 ± 5 | 12 ± 6 | 12 ± 5 | 13 ±3 | 14 ± 6 | 14 ± 4 |
| Visits to center | 12 ± 7 | 15 ± 8 | 10 ± 10 | 12 ± 12 | 9 ± 6 | 6 ± 5 |
| Rearings | 5 ± 5 | 5 ± 7 | 6 ± 8 | 6 ± 6 | 5 ± 5 | 7 ± 7 |
| Deviation from the Norm | The Number of Animals with a Sign of Deviation from the Norm | |||||
|---|---|---|---|---|---|---|
| Group 1 STD + Vehicle | Group 2 STD + AC 1 | Group 3 HFD + Vehicle | Group 4 HFD + AC 1 | Group 5 HFD + AC 7 | Group 6 HFD + MTX + AC 7 | |
| n = 10 | n = 10 | n = 10 | n = 10 | n = 9 & | n = 9 $ | |
| No macroscopic abnormalities were found | 10 | 10 | 0 *** | 3 *** | 4 **@ | 1 *** |
| Visually in the abdominal cavity a large amount of fat deposits | 0 | 0 | 10 *** | 4 *@ | 4 *@@ | 5 **@ |
| Hydronephrosis of the kidney | 0 | 0 | 5 ** | 3 | 1 | 4 * |
| Enlargement of the spleen | 0 | 0 | 1 | 1 | 0 | 0 |
| Organ | Group 1 STD + Vehicle | Group 2 STD + AC 1 | Group 3 HFD + Vehicle | Group 4 HFD + AC 1 | Group 5 HFD + AC 7 | Group 6 HFD + MTX + AC 7 |
|---|---|---|---|---|---|---|
| n = 10 | n = 10 | n = 10 | n = 10 | n = 9 & | n = 9 $ | |
| Absolute mass of organs, g | ||||||
| Testes, g. | 0.187 ± 0.023 | 0.196 ± 0.015 | 0.188 ± 0.014 | 0.192 ± 0.019 | 0.188 ± 0.019 | 0.180 ± 0.017 |
| Spleen, g | 0.084 ± 0.037 | 0.089 ± 0.020 | 0.105 ± 0.043 | 0.201 ± 0.279 | 0.103 ± 0.039 | 0.081 ± 0.016 |
| Kidneys, g | 0.484 ± 0.390 | 0.433 ± 0.060 | 0.575 ± 0.186 | 0.465 ± 0.128 | 0.428 ± 0.157 | 0.440 ± 0.121 |
| Adrenal gland, g. | 0.0079 ± 0.0011 | 0.0075 ± 0.0025 | 0.0088 ± 0.0020 | 0.0144 ± 0.0243 | 0.0038 ± 0.0010 *#@@@ | 0.0048 ± 0.0021 @@ |
| Liver, g. | 1.59 ± 0.43 | 1.73 ± 0.19 | 1.37 ± 0.18 | 1.28 ± 0.19 *## | 1.27 ± 0.10 *## | 1.28 ± 0.14 *## |
| Heart, g. | 0.146 ± 0.020 | 0.146 ± 0.019 | 0.159 ± 0.015 | 0.158 ± 0.028 | 0.151 ± 0.014 | 0.148 ± 0.012 |
| Pancreas, g. | 0.158 ± 0.017 | 0.220 ± 0.075 | 0.160 ± 0.027 | 0.236 ± 0.272 | 0.118 ± 0.017 *###@@ | 0.148 ± 0.047 ## |
| Adipose tissue surrounding the epididymis, g. | 0.4556 ± 0.0708 | 0.4259 ± 0.0816 | 1.7047 ± 0.4698 ***### | 1.0991 ± 0.2389 #∞∞∞ | 1.2541 ± 0.3390 **#∞ | 1.1489 ± 0.4382 *##∞ |
| Relative mass of organs (relative to body mass), % | ||||||
| Testes, % | 0.679 ± 0.077 | 0.693 ± 0.061 | 0.571 ± 0.068 *## | 0.622 ± 0.074 | 0.626 ± 0.051 | 0.597 ± 0.040 |
| Spleen, % | 0.304 ± 0.131 | 0.315 ± 0.064 | 0.325 ± 0.169 | 0.682 ± 1.039 | 0.351 ± 0.159 | 0.269 ± 0.065 |
| Kidneys, % | 1.783 ± 1.495 | 1.526 ± 0.142 | 1.749 ± 0.591 | 1.504 ± 0.419 | 1.428 ± 0.517 | 1.453 ± 0.430 |
| Adrenal gland, %. | 0.0288 ± 0.0041 | 0.0264 ± 0.0075 | 0.0272 ± 0.0056 | 0.0438 ± 0.0693 | 0.0126 ± 0.0030 ***##@@@ | 0.0157 ± 0.0072 **@@ |
| Liver, %. | 5.752 ± 1.526 | 6.124 ± 0.681 | 4.141 ± 0.458 **# | 4.148 ± 0.502 *## | 4.253 ± 0.485 *## | 4.199 ± 0.333 ## |
| Heart, %. | 0.528 ± 0.059 | 0.516 ± 0.058 | 0.483 ± 0.062 | 0.508 ± 0.056 | 0.505 ± 0.073 | 0.486 ± 0.038 |
| Pancreas, g. | 0.575 ± 0.061 | 0.771 ± 0.234 | 0.484 ± 0.077 | 0.782 ± 0.954 | 0.393 ± 0.041 ***### | 0.499 ± 0.211 ## |
| Adipose tissue surrounding the epididymis, %. | 1.6562 ± 0.2532 | 1.5005 ± 0.2371 | 5.0842 ± 0.9595 ***### | 3.5412 ± 0.7005 #∞∞∞ | 4.1193 ± 0.8408 ***##∞ | 3.6893 ± 1.0977 *##∞ |
| Group 1 STD + Vehicle | Group 2 STD + AC 1 | Group 3 HFD + Vehicle | Group 4 HFD + AC 1 | Group 5 HFD + AC 7 | Group 6 HFD + MTX + AC 7 | |
|---|---|---|---|---|---|---|
| Animals Number | n = 10 | n = 10 | n = 10 | n = 10 | n = 9 | n = 9 |
| Description of changes in organs | The number and severity of changes in the organs | |||||
| LIVER: | ||||||
| Hypertrophy of hepatocytes | 0 | 3SL | 1SL | 1SL | 0 | 0 |
| TOTAL SCORE | 0 | 6 | 2 | 1 | 0 | 0 |
| Foci of mononuclear infiltration | 4MI 1SL | 4MI 5SL | 4MI 6SL | 1MI 9SL | 2MI 6SL | 2MI 4SL |
| TOTAL SCORE | 6 | 14 | 16 | 19 | 14 | 10 |
| Segmented neutrophils in foci of mononuclear infiltration | 0 | 3MI 2SL | 4SL | 4SL | 1MI 2SL | 2SL |
| TOTAL SCORE | 0 | 7 | 8 | 8 | 5 | 4 |
| Focal angiectasia and intrahepatocellular erythrocytes | 0 | 1SL | 0 | 0 | 1SL | 0 |
| TOTAL SCORE | 0 | 2 | 0 | 0 | 2 | 0 |
| Accumulation of glycogen in hepatocytes | 0 | 0 | 2SL 3MO | 3SL 1MO | 4SL | 2SL 1MO |
| TOTAL SCORE | 0 | 0 | 13 | 9 | 8 | 7 |
| Lipid inclusions in the cytoplasm of hepatocytes | 0 | 0 | 1SL 1MO | 0 | 1MI 2SL 1MO 1MA | 1MI 1MO |
| TOTAL SCORE | 0 | 0 | 5 | 0 | 12 | 4 |
| Description of changes in organs | The number and severity of changes in the organs | |||||
| PANCREAS: | ||||||
| Cell hyperplasia of the islets of Langerhans | 0 | 1SL | 4SL | 1SL 1MO | 3SL 1MO | 2SL 2MO |
| TOTAL SCORE | 0 | 2 | 8 | 5 | 9 | 10 |
| Foci of mononuclear infiltration | 0 | 1MI 2SL 1MO | 1MI 1SL | 1SL | 1SL | 1MI 1SL |
| TOTAL SCORE | 0 | 8 | 3 | 2 | 2 | 3 |
| Lipomatosis | 3MI | 2MI | 1MI 1SL | 3MI | 3MI | 1MI 1SL |
| TOTAL SCORE | 3 | 2 | 3 | 3 | 3 | 3 |
| Exocrinocytes with vacuolar changes in the cytoplasm | 0 | 1MI | 1MI | 0 | 0 | 0 |
| TOTAL SCORE | 0 | 1 | 1 | 0 | 0 | 0 |
| TOTAL SCORE | 0 | 1 | 1 | 0 | 0 | 0 |
| ADRENALS: | ||||||
| Hypertrophy of epithelial cells of the fascicular zone of the cortical substance | 1SL | 0 | 1MI | 1SL | 2MO | 0 |
| TOTAL SCORE | 2 | 0 | 1 | 2 | 6 | 0 |
| KIDNEYS: | ||||||
| Chronic progressive nephropathy | 2MI | 1MI 1SL | 2SL | 1MI 1SL | 1MI 1SL 1MO | 1MI 1SL |
| TOTAL SCORE | 2 | 3 | 4 | 3 | 6 | 3 |
| Foci of mononuclear infiltration | 0 | 1MI | 1MI 1SL 1MO 2MA | 2MO | 1MO | 1MI 2MO |
| TOTAL SCORE | 0 | 1 | 14 | 6 | 3 | 7 |
| Foci of accumulation of siderophages | 0 | 0 | 1MI | 1MI | 0 | 1MI |
| TOTAL SCORE | 0 | 0 | 1 | 1 | 0 | 1 |
| Group 1 STD + vehicle | Group 2 STD + AC 1 | Group 3 HFD + vehicle | Group 4 HFD + AC 1 | Group 5 HFD + AC 7 | Group 6 HFD + MTX + AC 7 | |
| Animals number | n = 10 | n = 10 | n = 10 | n = 10 | n = 9 | n = 9 |
| Description of changes in organs | The number and severity of changes in the organs | |||||
| Foci of neutrophilic infiltration | 0 | 0 | 1SL 1MO | 2SL 1MO | 1SL | 2SL 1MO |
| TOTAL SCORE | 0 | 0 | 5 | 7 | 2 | 7 |
| Accumulation of neutrophils in the lumen of the ureter | 0 | 0 | 0 | 1MO | 0 | 0 |
| TOTAL SCORE | 0 | 0 | 0 | 3 | 0 | 0 |
| Vacuolization of the cytoplasm of renal tubular epithelial cells | 0 | 1MI | 2MI 1SL 6MO 1MA | 3MI 3SL 2MO 1MA | 1MI 5SL 3MO | 1MI 3SL 4MO 1MA |
| TOTAL SCORE | 0 | 1 | 24 | 19 | 20 | 23 |
| Dilatation of the renal pelvis | 0 | 0 | 5SE | 1SL 3SE | 4SL 2MO 1SE | 1SL 2MA 2SE |
| TOTAL SCORE | 0 | 0 | 25 | 17 | 19 | 20 |
| Atrophy of the renal parenchyma | 0 | 0 | 2MA 3SE | 1SL 3MA | 4SL 2MO 1SE | 1SL 2MO 1MA 1SE |
| TOTAL SCORE | 0 | 0 | 23 | 14 | 19 | 17 |
| Expansion of the lumen of the renal tubules | 0 | 0 | 1SL 3MO | 3MO | 2SL 1MO | 1SL 2MO 1MA |
| TOTAL SCORE | 0 | 0 | 11 | 9 | 7 | 12 |
| HEART: | ||||||
| Mineral deposits in myocardial interstitium | 0 | 0 | 1MI | 1MI 1SL | 1MI 2SL | 1MI |
| TOTAL SCORE | 0 | 0 | 1 | 3 | 5 | 1 |
| Lipid inclusions in the sarcoplasm of cardiomyocytes | 0 | 0 | 3SL | 3MI 1SL | 4MI 1SL | 3MI 1SL |
| TOTAL SCORE | 0 | 0 | 6 | 5 | 6 | 5 |
| Group # | Diet | Group | Articles, Doses, Scheme | Animal Numbers |
|---|---|---|---|---|
| 1 | STD | STD + vehicle | Saline | 1–10 |
| 2 | STD | STD + AC 1 | AICAR (500 mg/kg/three times a week from study week 1, 10 mL/kg, ip) | 11–20 |
| 3 | HFD | HFD + vehicle | Saline (three times a week from study week 1, 10 mL/kg, ip) | 21–30 |
| 4 | HFD | HFD + AC 1 | AICAR (500 mg/kg/three times a week from the 1st week of the study, 10 mL/kg, ip) | 31–40 |
| 5 | HFD | HFD + AC 7 | AICAR (500 mg/kg/three times a week from the 7th week of the study, 10 mL/kg, ip) | 41–50 |
| 6 | HFD | HFD + MTX + AC 7 | Metotrexate + AICAR (1 mg/kg/weekly 10 mL/kg, ip + 500 mg/kg/three times a week from the 7th week of the study, 10 mL/kg, ip) | 51–60 |
| Alanine transaminase (alt) | Calcium | Urea |
| Aspartate aminotransferase (ast) | Chloride | Total bilirubin |
| Alkaline phosphatase (ap) | Phosphate | Albumin |
| Creatinine | Cholesterine | Globulin |
| Total protein | Triglyceride | Albumin/globulin * |
| Adrenal gland | Kidneys |
| Heart | Spleen |
| Pancreas | Testes |
| Liver | Adipose tissue surrounding the epididymis |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tukhovskaya, E.A.; Shaykhutdinova, E.R.; Pakhomova, I.A.; Slashcheva, G.A.; Goryacheva, N.A.; Sadovnikova, E.S.; Rasskazova, E.A.; Kazakov, V.A.; Dyachenko, I.A.; Frolova, A.A.; et al. AICAR Improves Outcomes of Metabolic Syndrome and Type 2 Diabetes Induced by High-Fat Diet in C57Bl/6 Male Mice. Int. J. Mol. Sci. 2022, 23, 15719. https://doi.org/10.3390/ijms232415719
Tukhovskaya EA, Shaykhutdinova ER, Pakhomova IA, Slashcheva GA, Goryacheva NA, Sadovnikova ES, Rasskazova EA, Kazakov VA, Dyachenko IA, Frolova AA, et al. AICAR Improves Outcomes of Metabolic Syndrome and Type 2 Diabetes Induced by High-Fat Diet in C57Bl/6 Male Mice. International Journal of Molecular Sciences. 2022; 23(24):15719. https://doi.org/10.3390/ijms232415719
Chicago/Turabian StyleTukhovskaya, Elena A., Elvira R. Shaykhutdinova, Irina A. Pakhomova, Gulsara A. Slashcheva, Natalya A. Goryacheva, Elena S. Sadovnikova, Ekaterina A. Rasskazova, Vitaly A. Kazakov, Igor A. Dyachenko, Alina A. Frolova, and et al. 2022. "AICAR Improves Outcomes of Metabolic Syndrome and Type 2 Diabetes Induced by High-Fat Diet in C57Bl/6 Male Mice" International Journal of Molecular Sciences 23, no. 24: 15719. https://doi.org/10.3390/ijms232415719






