Abstract
Powdery mildew, caused by Blumeria graminis f. sp. tritici (Bgt), is one of the most destructive foliar diseases of wheat. In this study, we combined the bulked segregant RNA sequencing (BSR-seq) and comparative genomics analysis to localize the powdery mildew resistance gene in Chinese landrace Xiaomaomai. Genetic analysis of F1 plants from a crossing of Xiaomaomai × Lumai23 and the derived F2 population suggests that a single recessive gene, designated as pmXMM, confers the resistance in this germplasm. A genetic linkage map was constructed using the newly developed SNP markers and pmXMM was mapped to the distal end of chromosome 2AL. The two flanking markers 2AL15 and 2AL34 were closely linked to pmXMM at the genetic distance of 3.9 cM and 1.4 cM, respectively. Using the diagnostic primers of Pm4, we confirmed that Xiaomaomai carries a Pm4 allele and the gene function was further validated by the virus-induced gene silencing (VIGS). In addition, we systematically analyzed pmXMM in comparison with the other Pm4 alleles. The results suggest that pmXMM is identical to Pm4d and Pm4e at sequence level. Pm4b is also not different from Pm4c according to their genome/amino acid sequences. Only a few nucleotide variances were detected between pmXMM and Pm4a/b, which indicate the haplotype variation of the Pm4 gene.
1. Introduction
Wheat (Triticum aestivum L.) is one of the most widely grown food crops worldwide. Its stable yield plays an important role in food security. Powdery mildew, caused by Blumeria graminis f. sp. tritici (Bgt), is one of the most devastating foliar diseases of wheat. At present, the most efficient and environmentally safe approach to counteract epidemics of powdery mildew is to breed disease-resistant cultivars in production. However, most of powdery mildew resistance (R) genes are race-specific which confer strong immunity to some but not all of the pathogen races. Therefore, molecular identification and utilization of new R genes to produce a wheat cultivar with improved disease resistance is challenging for wheat breeders.
To date, more than 60 loci for resistance to powdery mildew (Pm1–Pm66) have been documented [1] and over ten genes have been cloned in wheat landraces [2,3,4,5,6,7]. Most of the identified R genes in crops encode proteins with predicted coiled-coil-nucleotide-binding-site (CC-NBS) and leucine-rich repeat (LRR) domains [8,9]. The exception was Pm24, which encodes a tandem kinase [10]. Stpk-V, a key member of Pm21, was demonstrated to be a serine/threonine kinase [11]. Pm38/Lr34/Yr18/Sr57 and Pm26/Yr46/Lr67/Sr55, which, respectively, provides broad-spectrum resistance to multiple pathogens and encodes an ABC transporter and hexose transporter [12,13]. In addition, Pm4, a resistance gene valuable for the breeding, was recently cloned and illustrated to be a putative serine/threonine kinase [14,15].
Multiple Pm4 alleles located on chromosome 2AL have been reported. Pm4a and Pm4b originated from T. dicoccum and T. carthlicum, respectively, and can be found in many commercial cultivars in China [14,16,17]. Schmolke et al. (2012) identified a dominant gene Pm4d, which was introduced from T. monococcum. In addition, the two alleles Pm4c and Pm4e were detected in common wheat cultivars [18,19]. Besides the Pm4 locus, few other genes were also mapped to chromosome 2AL and demonstrated to be allelic or closely linked to Pm4, such as PmPS5A [20], pmLK906 [21], PmHNK54 [22], pmX [23], and Pm50 [24]. Therefore, they have been considered to be members of the Pm4 locus.
Next-generation sequencing offers a new genetic mapping strategy that combines bulked segregant analysis (BSA) with RNA-sequencing analysis for quick identification of wheat powdery mildew resistance genes [25]. Molecular markers developed from single nucleotide polymorphisms (SNP) enable the establishment of a high-resolution genetic linkage map and detection of the target genes. In this study, we have identified a recessive powdery mildew resistance gene from Chinese wheat landrace Xiaomaomai employing this technique. The constructed linkage map localized the resistance gene, designated as pmXMM, to chromosome 2AL, flanked by two newly designed SNP markers 2AL15 and 2AL34. We further confirmed that pmXMM was a haplotype of Pm4 showing an identical genome sequence to Pm4d and Pm4e. The functionality of pmXMM was further verified by the virus-induced gene silencing. This study suggests that Pm4 is a powdery mildew resistance gene valuable for developing future disease-resistant wheat cultivars.
2. Materials and Method
2.1. Plant Materials
Bread wheat cultivar Xiaomaomai is a Chinese landrace and shows resistance to powdery mildew. The susceptible recurrent parent Lumai23 is a Chinese winter wheat cultivar widely grown in the Shandong province. F1, F2, and F2:3 resistance segregation populations were created by crossing Xiaomaomai with Lumai23 and subsequently used for the genetic analysis and molecular mapping. Common wheat lines Khapli/8*Cc (carrying gene Pm4a), Armada (carrying gene Pm4b), 81-7241 (carrying gene Pm4c), and D29 (carrying gene Pm4c) were used in resistance spectrum and genetic analyses. Winter wheat cultivar Zhongzuo9504 was used as the susceptible control to assess the powdery mildew resistance.
2.2. Resistance Evaluation
The conidiospores of isolate Bgt1 were freshly increased on the susceptible cultivar Zhongzuo9504. When abundant sporulation was visible on the leaf area of Zhongzuo9504, the inoculation was conducted using the dusting technique: one-leaf-stage seedlings were sprayed with water evenly on the leaf area and then dusted with conidiospores of Bgt1. One pot of Zhongzuo9504 was used to inoculate at most five pots of tested seedlings. After inoculation, plants were grown in a growth chamber maintained at 16 °C, 16-h light, 8-h dark, and 50% humidity. After 7–10 days of inoculation, when the susceptible control Zhongzuo9504 plants were heavily diseased, the disease symptom of each plant was visually rated on a 0–4 scale as described by Liu et al. (1999). At least 15 plants were tested for each F2:3 family. Plants were considered as resistant when the infection types (IT) were 0–2, while considered susceptible when the scores were 3–4. The effectiveness of pmXMM in wheat line Xiaomaomai against powdery mildew was tested using 40 Bgt isolates derived from single-spore isolation collected from different wheat fields located in Hebei, Beijing, Tianjin, Shandong, Henan, Sichuan, and Shanxi of China (Supplementary Table S1).
2.3. Genotyping of F2:3 Lines Using BSR-Seq Analysis
The phenotypically contrasting F2:3 families against isolate Bgt1 were used to construct the resistant and susceptible RNA pools (respectively made of six homozygous resistant and homozygous susceptible F2:3 families) for RNA-seq analysis. Total RNA of the two bulks of leaf samples were separately extracted using the RNAsimple Total RNA Kit (Tiangen). RNA-Seq was done by the platform of Illumina HiSeq4000 (Beijing Southern Genome Research Technology Co., Ltd., Beijing, China). The raw sequencing reads were quality controlled using software Trimmomatic v0.36 [26] with the default parameters. Using software STAR v2.5.1b [27], the clean reads were aligned to the wheat reference genome assembly IWGSC RefSeq v1.0 [28] with the mismatch rate of less than 5%. The uniquely mapped read pairs were used in further analysis. The read alignments were masked for PCR duplications before they were used to call SNPs and InDels using small variant caller Strelka v2 [29]. The resulting SNPs and InDels with sequencing depth less than 6 were discarded, and the remaining ones were applied to bulked segregant analysis. Only variants with allele frequency difference (AFD) > 0.8 and p-value of Fisher’s exact test on read count data < 1 × 10−10 were classified as trait-associated variants and used as templates for marker development.
2.4. Marker Analysis
Genomic DNA was extracted from the leaf tissue of wheat seedlings using a DNAquik Plant System (Tiangen). The single nucleotide polymorphisms (SNPs) associated with the powdery mildew resistance were identified by BSR-Seq analysis and selected for genome-specific Kompetitive Allele Specific PCR (KASP) markers development using the Polymarker website (http://www.polymarker.info/ accessed on September 2020). PCR was performed in a 10 μL reaction mixture using 2 × KASP Master Mix (Std Rox, LGC) following the protocol. Polymorphic KASP markers between the parents and the contrasting DNA bulks were then used to construct the genetic linkage map of pmXMM. Polymorphism survey was also conducted with SSR markers linked to Pm4 locus on chromosome 2AL [18,19,23,30,31,32] (Supplementary Table S2). PCR was performed with a C1000 TouchTM Thermal Cycler (BIORAD) in a 10 μL reaction mixture using the 2 × M5 PAGE Taq PCR Mix (Mei5bio) according to the manufacturer’s instructions. The program condition was 95 °C for 3 min, 35 cycles of 94 °C for 25 s, 55 °C for 25 s, and 72 °C for 20 s, and a final extension at 72 °C for 5 min. PCR products were separated in 8% non-denaturing polyacrylamide gels with a 19:1 or 39:1 acrylamide/bisacrylamide ratio, and then silver-stained as described by Santos et al. (1993). The dCAP markers was developed to determine the SNPs of Pm4 alleles. A total of 443 bp fragments were first amplified using the primer pair Pm4.3 listed in Supplementary Table S3 and subjected to one-hour digestion at 37 °C using NeaI enzyme. Products were subsequently investigated on the 2% agarose gel.
2.5. Data Analysis and Linkage Map Construction
The Chi-squared test (χ2) was employed to examine whether the observed separation data in F2 and F2:3 population fit for Mendelian segregation ratio. The genetic distance between the polymorphic markers and the target gene was calculated by the Kosambi function and the genetic linkage map was constructed using software Mapdraw v2.1 [33]. A logarithm of the odd ratio (LOD) of 3.0 was used as the threshold for declaration of linkage and the maximum genetic distance allowed between markers was set at 50.0 cM.
2.6. BSMV Virus-Induced Gene Silencing
To prepare the recombinant BSMV:Pm4-V2 constructs, two different fragments (200–500 bp) amplified from Pm4 exons 6 and 7 were respectively subcloned into the pBS-BSMV-γ vector using a One Step Seamless Cloning kit (GeneBetter) according to the manufacturer’s instructions (Primer sequences were listed in Supplementary Table S3). For in vivo synthesis of viral RNA, an equimolar amount of pBS-BSMV-α, pBS-BSMV-β, and pBS-BSMV-γ vectors were first transformed into the one-month-old tobacco plants mediated by Agrobacterium. Ten days after injection, tobacco leaves with virus symptom were ground and subsequently used to rubbing the second leaf of Xiaomaomai plants at the two-leaf stage. Seedlings of the same age were inoculated with the wild-type (γ0) virus as a control. Fourteen days after virus infection, the third leaves of Xiaomaomai were collected for qRT-PCR analysis and the fourth leaves were infected with the isolate Bgt1. Powdery mildew phenotype was documented 10 days post inoculation.
2.7. RNA Isolation and RT-PCR
The third leaf of wheat seedling was harvested 14 days post inoculation of virus and ground in liquid nitrogen. RNA was extracted using the TRIzol® reagent (Biorigin) according to the manufacturer’s instructions. The FastKing gDNA Dispelling RT SuperMix kit (Tiangen) was employed to remove residual DNA and synthesize the corresponding cDNA using the following PCR program: 42 °C for 15 min and 95 °C for 3 min.
2.8. Real-Time Quantitative PCR
Real-time PCR was used to determine the transcript abundance of Pm4 gene. Samples were run in triplicate with PerfectStart Green qPCR SuperMix (TransGen Biotech) on a Bio-Rad CFX96 Touch Real-Time PCR Detection System. The wheat actin gene was used as a housekeeping control and qRT-PCR primers used for the target and reference genes are shown in Supplementary Table S3. The thermocycling conditions were 94 °C for the 30 s, followed by 44 cycles of 94 °C for 5 s, 58 °C for 15 s, and 72 °C for 10 s. Relative quantities were calculated and normalized to the reference genes using 2−ΔΔCt method [34].
3. Results
3.1. Resistance of Xiaomaomai to Different Bgt Isolates
Forty Bgt isolates collected from the northern part of China were used to examine the virulence spectrum of Xiaomaomai together with the wheat cultivars carrying Pm4a, Pm4b, and Pm4c (Supplementary Table S1). Xiaomaomai exhibited resistance to 12 isolates (30%, IT 0 to 2) and susceptibility to 28 isolates (IT 3 or 4) shown in Supplementary Table S1. Xiaomaomai and the wheat lines carrying Pm4 alleles revealed similar reaction patterns: line Khapli/8*Cc containing Pm4a was resistant to 30% of the isolates tested and 81-7241 carrying Pm4c was resistant to 32.5% of them. Line Armada containing Pm4b was effectively against to 37.5% isolates and exhibited different reaction to three isolates compared to Xiaomaomai. Zhongzuo9504 was employed as a susceptible control.
3.2. Inheritance of the Powdery Mildew Resistance in Xiaomaomai
Xiaomaomai and Lumai23 reacted differently when the seedlings were inoculated with Bgt1 isolate from Hebei province (Figure 1). Therefore, this isolate was used for phenotypic analysis on F1, F2, and F2:3 populations derived from Xiaomaomai × Lumai23 cross. All the F1 plants showed susceptible to Bgt1, while F2 plants exhibited a segregation ratio of 1:3 for resistant and susceptible plants (χ21:3 = 0.581, p = 0.446, Table 1). In the progeny test, a segregation ratio of 1:2:1 (homozygous resistant lines: heterozygous lines: homozygous susceptible lines) was observed in the F2:3 population, consisting of 177 families (χ2 1:2:1 = 0.831, p = 0.660). This result indicated that a single recessive gene, tentatively designated as pmXMM, confers the resistance to isolate Bgt1 in Xiaomaomai.

Figure 1.
The phenotypic reactions of resistant parent Xiaomaomai, susceptible parent Lumai23, and their F1 progenies to Bgt1 isolate. Zhongzuo9504 served as the susceptible control.

Table 1.
Progeny test of Xiaomaomai × Lumai23 cross in term of response to Blumeria graminis f. sp. tritici isolate Bgt1.
3.3. RNA-Seq Analysis of the RNA Bulks with Distinct Reactions to Bgt1
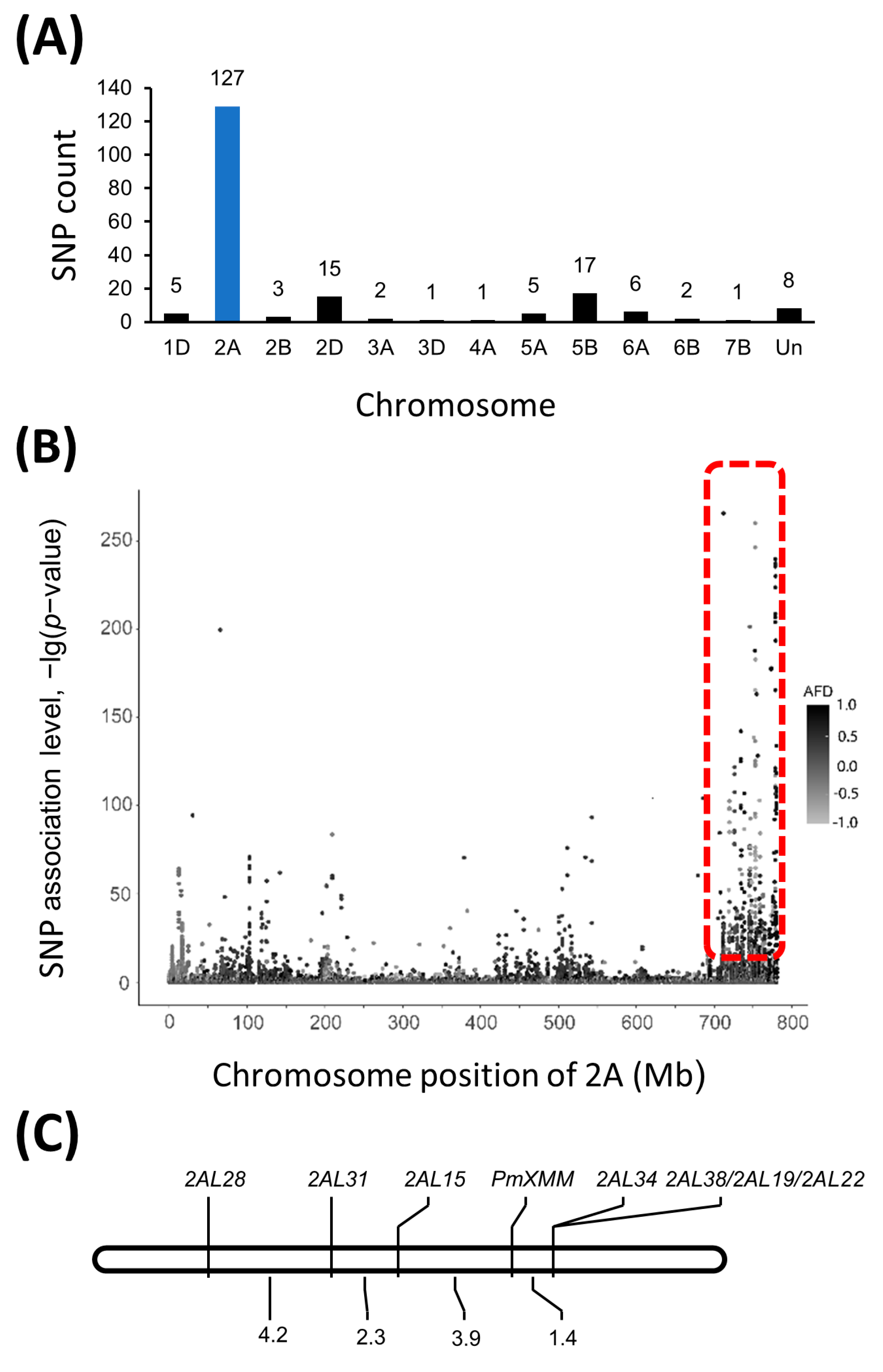
The RNA samples pooled from F2:3 families with known Bgt1 resistance and susceptibility were designated as Bulk-R and Bulk-S, respectively. Through RNA-Seq analysis performed on an Illumina HiSeq 4000 platform, 39,574,081 and 40,826,755 raw read pairs were created for Bulk-R and Bulk-S. After quality control, 36,902,452 read pairs of Bulk-R were selected and 31,793,499 (86.16%) of them were uniquely mapped to the wheat reference genome assembly IWGSC RefSeq v1.0 [28]. In addition, there were 39,313,629 high-quality reads and 35,409,901 (90.07%) uniquely mapped reads for the Bulk-S sample. A total of 328,746 variants were identified from those mapped reads by the Strelka software [29] with default parameters, of which 195 variants were found to be trait-associated (p < 1 × 10−10 and AFD > 0.8). We found that 127 trait-associated variants were enriched in a 54 Mb genomic interval (727,191,545–780,717,753) on chromosome arm 2AL in the Chinese Spring reference genome (Figure 2A,B, Supplementary Table S4), suggesting that the target gene was potentially localized in this region.
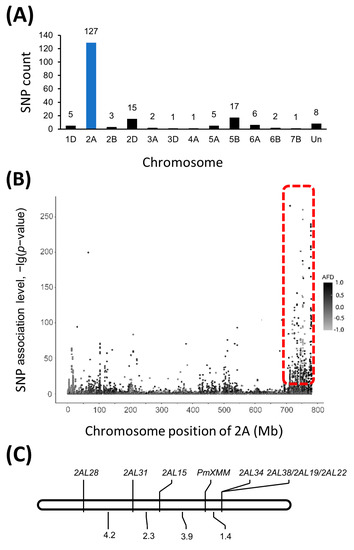
Figure 2.
BSR-Seq analysis and genetic linkage map of pmXMM. The number of single nucleotide polymorphisms (SNPs) distributed on different wheat chromosomes (A) and the SNP variants on chromosome 2A (B). Linkage map of pmXMM on chromosome 2AL (C).
3.4. Polymorphic Analysis of SNP Markers and Construction of Genetic Linkage Map
To further map the resistance gene, polymorphism survey was initially conducted with previously reported markers linked to Pm4 locus, which was also located on chromosome 2AL. The assay was carried out between the crossing parents as well as the two contrasting DNA bulks made of F2:3-resistant and -susceptible progenies. The surveyed markers were shown in Supplementary Table S2 and only Pm4b-associated maker STS470 was polymorphic between the two parents and contrasting DNA bulks. We subsequently developed the SNP markers flanking sequences of the SNPs potentially associated with the target gene on chromosome 2AL. Seven pairs of primers were shown to be able to differentiate the crossing parents (Supplementary Table S5). Consistent polymorphism was also detected between the DNA bulks containing resistant and susceptible progenies, indicating that they were likely linked to pmXMM. Therefore, these SNP markers were employed for the construction of a genetic linkage map using 355 F2:3 families derived from Xiaomaomai × Lumai23 cross. Results suggested that 2AL15, 2AL28, and 2AL31 were potentially mapped on the proximal side, while 2AL22, 2AL19, 2AL38, and 2AL34 were localized on the distal side of the target gene (Table 2, Figure 2C). pmXMM was localized in 3.7 Mb physical region (755,705,200–759,456,729) on chromosome 2AL flanked by markers 2AL15 and 2AL34 with genetic distances of 1.4 and 3.9 cM, respectively (Figure 2C).

Table 2.
Newly developed single nucleotide polymorphism (SNP) markers linked to pmXMM.
3.5. Identification of the pmXMM Candidate Gene
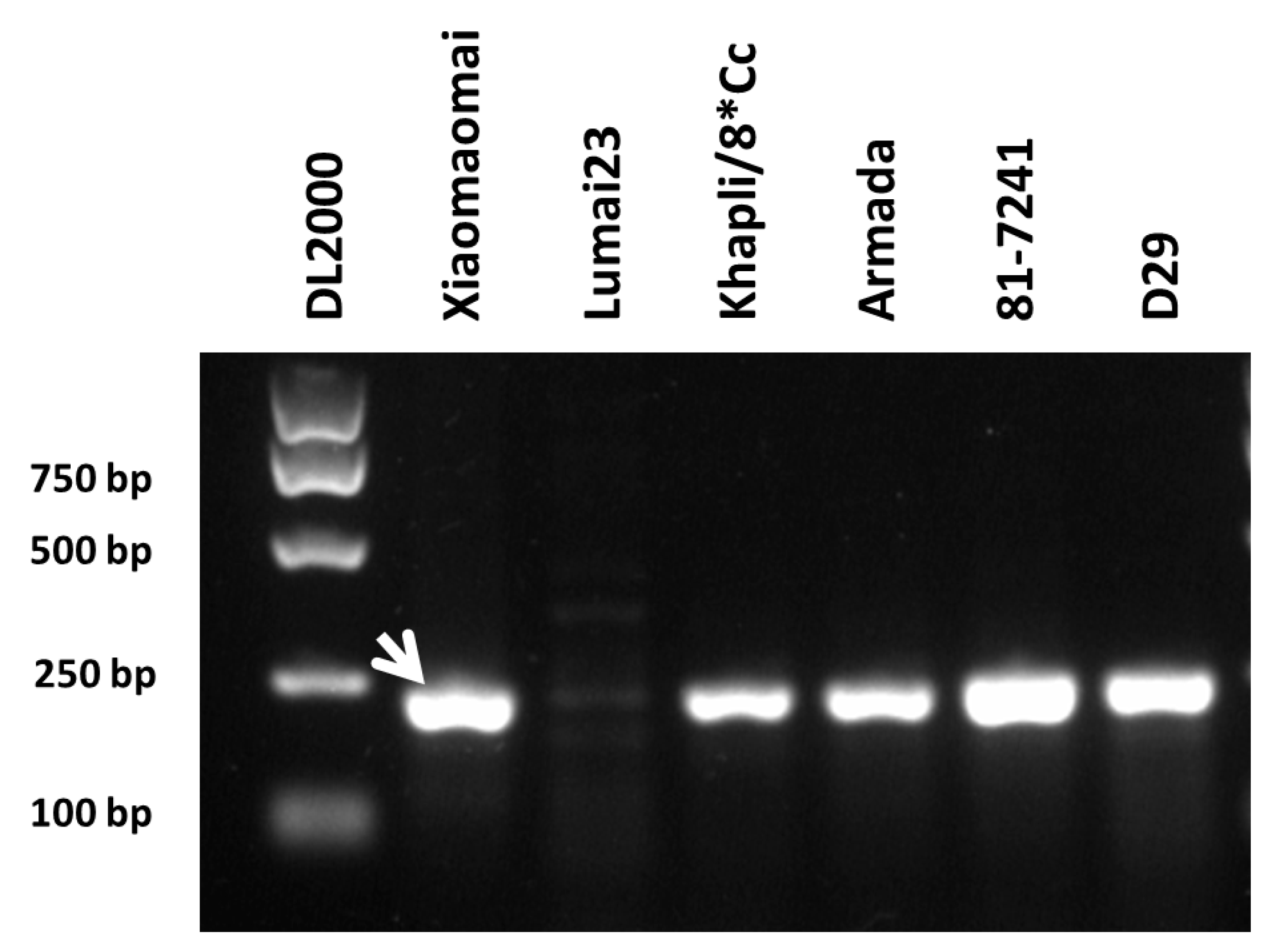
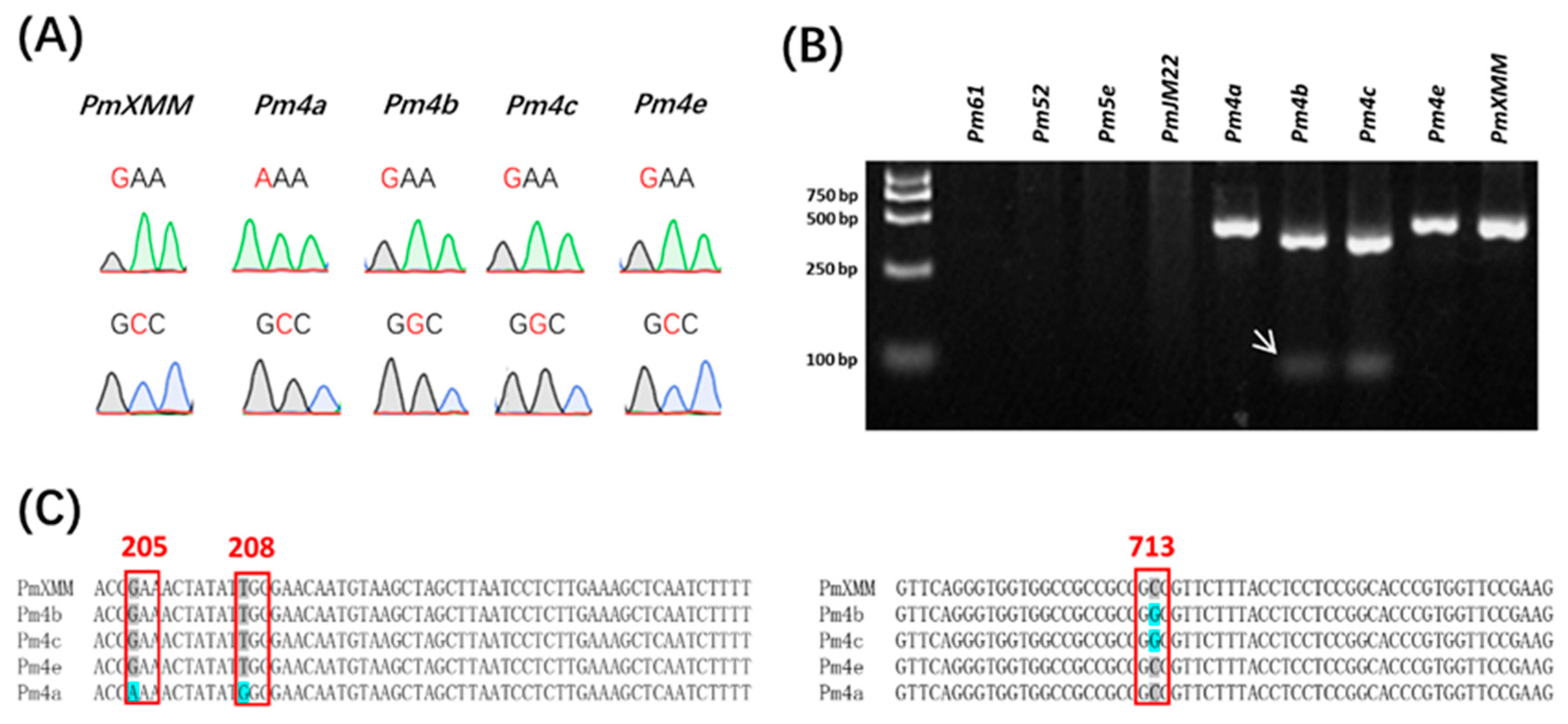
Pm4 gene mapped to the chromosomal region similar to pmXMM has been identified recently (Sánchez-Martín et al., 2021). It encodes a putative chimeric protein of a serine/threonine kinase and has two splicing variants (Pm4-V1 and Pm4-V2). By sequencing the exons of Pm4, Sánchez-Martín et al. (2021) have shown that the four amino acid polymorphisms listed in Table 3 represents the variation of Pm4-resistant proteins they have checked. To determine the allelic relationship of pmXMM to Pm4, we employed a molecular marker Pm4.1 designed from the Pm4 coding sequence to investigate the genetic background of the Xiaomaomai and Lumai23 (Supplementary Table S3). The presence of Pm4 alleles was detected in Xiaomaomai and the other Pm4 carrying lines, but not in susceptible parent line Lumai23 (Figure 3). In addition, linkage analysis illustrated that the Pm4-specific marker was co-segregating with the target gene (Table 2), suggesting that the resistance gene was likely to be an allele of Pm4. We further amplify the exon fragments of Pm4 alleles, covering the amino acid variances from the genomic DNA of wheat lines carrying pmXMM, Pm4a, Pm4b, Pm4c, and Pm4e using the primers listed in Supplementary Table S3 and their protein sequences were identified based on the two alternatively spliced transcripts of Pm4b (GenBank accession numbers of Pm4b_V1 CDS and Pm4b_V2 CDS were MT783929 and MT783930). By Sanger sequencing, we could differentiate pmXMM from the other Pm4 alleles by checking the SNPs at the 205 and 713 amino acid sites (Figure 4A,C). In addition, we have developed a dCAP marker that could be used for the genetic analysis of SNP at the 713 amino acid site of Pm4 (Figure 4B). However, wheat lines carrying other Pm genes and showing resistance to Bgt 1 isolate were detected blank on the 2% agarose gel using the same dCAP marker (Figure 4B). These results revealed that the investigated Pm4 genes were divided into three haplotypes: pmXMM exhibited the identical genome/amino acid sequence to Pm4e and Pm4d (Table 3, Sánchez-Martín et al., 2021). Pm4b seems to be as same as Pm4c according to their genome/amino acid sequence. Only a few nucleotide differences exist between pmXMM with Pm4a/b (Table 3).

Table 3.
Protein sequence comparison of pmXMM with the known Pm4-resistant alleles. Amino acid sequences of Pm4d and Pm4h were identified from the study of Sánchez-Martín et al. (2021).
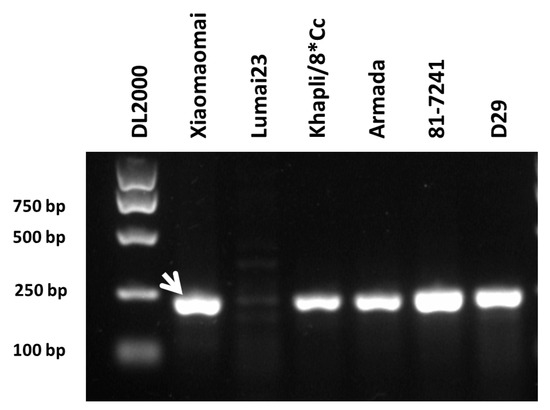
Figure 3.
Amplification pattern of Pm4-specific marker Pm4.1 amplified from the genomic DNA of Xiaomaomai, Lumai23, and wheat lines carrying Pm4a (Khapli/8*Cc), Pm4b (Armada), Pm4c (81-7241), and Pm4e (D29) in 1% agarose gel. The first lane was loaded with DL2000 DNA ladder. The white arrow indicates the amplicons specific for the Pm4 gene.
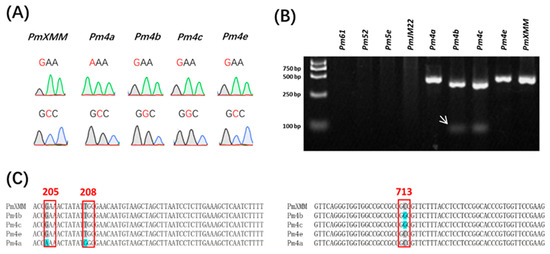
Figure 4.
Profiles of designed SNP markers differentiating pmXMM, Pm4a, Pm4b, Pm4c, and Pm4e. Sanger sequencing profiles of SNP markers targeting the 205 (Up) and 713 (Bottom) amino acid sites of Pm4 alleles (A). Polymorphic profile of wheat lines showing resistance to Bgt 1 isolate using the dCAP marker (B). White arrows indicate the polymorphic bands specific for Pm4b and Pm4c. No fragment was detected in wheat lines carrying Pm61, Pm52, Pm5e, and PmJM22. The first lane was loaded with a DL2000 DNA ladder. Sequence alignments of investigated Pm4 alleles at the 205, 208, and 713 amino acid variant sites (C). Conserved nucleotides are indicated by a grey color and the variant sites causing amino acid changes are highlighted by a blue color.
3.6. Silencing of pmXMM Using BSMV-VIGS
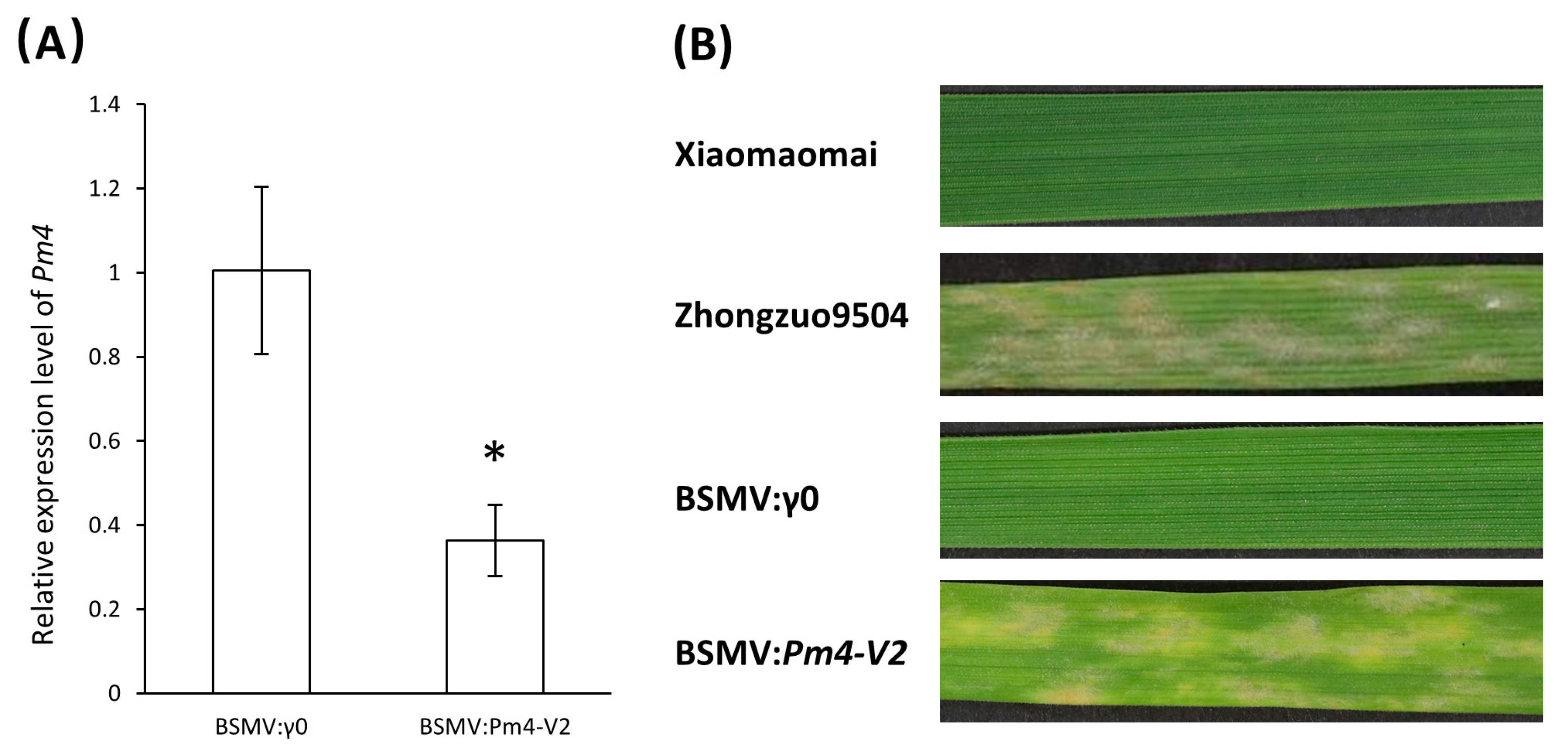
To verify the function of pmXMM in resistance to Bgt1 infection in Xiaomaomai, we performed the virus-induced gene silencing (VIGS) as developed with barley stripe mosaic virus (BSMV). Two constructs targeting Pm4-V2 variance were used to suppress the pmXMM expression in Xiaomaomai plants (Supplementary Table S3). Results of real-time quantitative PCR showed that BSMV:Pm4-V2 virus led to the downregulation of Pm4 transcript 14 days post inoculation (Figure 5A). Sporulating mildew colonies were observed covering large leaf areas of Xiaomaomai infected with BMSV:Pm4-V2 virus, suggesting that silencing of pmXMM resulted in the susceptibility of Xiaomaomai to Bgt1 isolate (Figure 5B). Wheat plants infected with wild-type virus BSMV:γ0 served as the control for all the analyses.
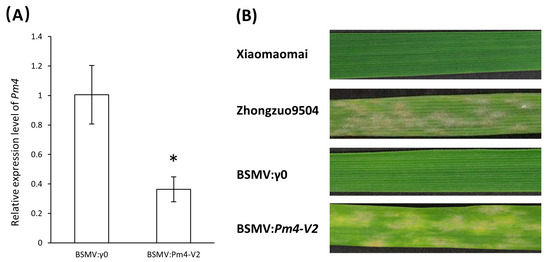
Figure 5.
Validation of pmXMM functionality by BSMV-VIGS. qRT-PCR analysis of plants infected with BSMV:γ0 and BSMV:Pm4-V2 virus (A). Data are mean ± SE calculated from three biological replicates and normalized to the actin expression level. Asterisk indicates a significant difference (p-value < 0.05) using Student’s t-test. Symptoms of the fourth leaves pre-inoculated with BSMV and then challenged with Bgt1 isolate (B).
4. Discussion
This study was designed to locate the candidate gene, conferring the resistance of powdery mildew in Chinese landrace Xiaomaomai. BSR-Seq analysis demonstrated that pmXMM, a recessive powdery mildew resistance gene, was located on the distal end of chromosome arm 2AL. Newly developed SNP markers mapped pmXMM gene in a 5.3 cM genetic interval corresponding to 3.7 Mb genomic region. Diagnostic primers and Sanger sequencing suggested that Xiaomaomai carries a Pm4 allele identical to Pm4d and Pm4e in genome sequences. In addition, the functionality of pmXMM in powdery mildew resistance was ultimately confirmed by the virus-induced gene silencing. Thus, we conclude that pmXMM is a recessive allele of the Pm4 locus.
Pm4 is used in disease resistance breeding because of its effectiveness in the resistance of Bgt isolates in certain regions of China and the United States [35,36]. In this study, the frequency of pmXMM in 90 wheat cultivars was evaluated (Supplementary Table S6). The results showed that pmXMM was present in eight wheat lines, including Huacheng 3366, Zhengyumai 518, Xinong 16, Zhongluo 08-1, Zhengmai 101, Jingnong 3668, 10BMY12, and Lankao 815. In the most recent study, Pm4 has been cloned by MutChromSeq and was demonstrated to encode a putative serine/threonine kinase [15]. Up to now, over ten wheat powdery mildew resistance genes have been cloned in wheat, including Pm2 [2], Pm3b [3], Pm5 [4], Pm8/Pm17 [5,37], Pm21 [7,38], Pm24 [10], Pm38/Lr34/Yr18/Sr57 [12], Pm41 [39], Pm60 [6], and Pm46/Lr67/Yr46/Sr55 [13]. Stpk-V, located on the Pm21 locus, was also a serine/threonine kinase [11]. Several disease resistance genes have been identified to contain a serine/threonine kinase domain, such as the stem rust resistance gene Rpg5 in barley and stripe rust resistance gene Yr36 in wheat [40,41].
Pm4 locus including multiple resistance alleles was previously reported to be located on chromosome 2AL (McIntosh and Bennett, 1979). Several Pm4 allelic genes have been identified to be dominant, including Pm4a (Ma et al., 2004), Pm4b [32,42], Pm4c/Pm23 (Hao et al., 2008), Pm4d (Schmolke et al., 2012), Pm4e (Li et al., 2017), and PmHNK54 (Xu et al., 2011). pmXMM showed the same genome/amino acid sequence as Pm4d and Pm4e (Table 3), suggesting that these three alleles were likely to be the same haplotype. However, we confirmed that pmXMM was a recessive gene by progeny analysis using F1 plants developed from the crossing of Xiaomaomai × Lumai23 and the derived F2 and F2:3 populations (Table 1). One explanation could be that the genomic background of Xiaomaomai was very different from the plant lines carrying Pm4d and Pm4e. The lack of polymorphism between Xiaomaomai and Lumai23 using common markers linked to Pm4 supports our speculation. Therefore, only two copies of the gene could lead to the resistance of Bgt isolates in Xiaomaomai. Consistent with our results, pmX (Fu et al., 2013) and pmLK906 [43] have been reported to be a recessive gene close to the chromosomal position of Pm4 alleles. Further research could be carried out to verify whether these two genes were new alleles of Pm4 using the diagnostic primers and Sanger sequencing.
In the present study, based on the BSR-Seq analysis, SNP markers were designed and used for the construction of the pmXMM linkage map (Figure 2C). pmXMM was located in chromosome 2AL and spanned a physical interval of about 3.7 Mb (755,705,200–759,456,729) on the Chinese Spring. However, a previous study demonstrated that Pm4e was placed in a physical interval of 762.5–768.0 Mb on chromosome 2AL through a fine mapping [44]. Consistently, Sánchez-Martín et al. (2021) reported that Pm4a/b was absent in Chinese Spring and the closest homology is located approximately at position 761 Mb. It seems that the chromosomal location of Pm4 genes in different wheat lines is close but still a little bit different. Similar phenomenon was also observed in the study of Fu et al. (2013), who showed that the common markers exhibited different genetic distances to the same Pm4 haplotype. For instance, it has been shown that STS marker XresPm4 was 6.5 cM from Pm4e but was cosegregating with Pm4d in previous studies [19,31]. In our work, most of the tested Pm4 associated markers failed to produce the polymorphic banding pattern between the crossing parents as well as the contrasting progeny bulks. Therefore, we speculated that an interstitial missing or inversion might have occurred in wheat plants, which could cause the variation of gene location referring to the Chinese spring genome. Other factors may be the map population and the different genetic background of the crossing parents.
Sánchez-Martín et al. (2021) have shown that Pm4 was a relatively widespread gene and the polymorphism in a single amino acid could determine the functionality of the protein. For instance, they have discovered three new Pm4 alleles (Pm4f, Pm4g, and Pm4h) by screening the genetic background of 512 wheat collections. Although there was a single nucleotide change, Pm4f carrying lines were susceptible to the tested isolate, while plants containing Pm4h showed powdery mildew resistance. In support of this claim, Xiaomaomai showed differential patterns of response to Bgt isolates compared to plant lines carrying Pm4a, Pm4b, and Pm4c (Supplementary Table S1). This may be due to the fact that few amino acid variances on the kinase domain or transmembrane domain are crucial for affecting the kinase activity. Consistently, it has been reported that the deletion of the specific two amino acids in the kinase I domain of Pm24 determined its resistance function [10]. In addition, Lr67, a multi-pathogen resistant gene that encodes a predicted hexose transporter, is different from its susceptible form by two amino acids [13]. Other factors, including the genetic background of a plant line, gene-gene interaction, host-pathogen interaction, and environmental conditions, may also contribute to the infection type of a wheat line.
In this study, we designed a new pair of primers that could specifically amplify 213 bp fragment of the Pm4 gene and produce the diagnostic banding pattern more efficiently (Figure 3). This was beneficial for marker-assisted breeding and stacking Pm4 with other Pm genes to improve the powdery mildew resistance of wheat.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms23031194/s1.
Author Contributions
G.S., S.X. and D.Y. designed the project. W.I., Y.L., J.H., W.P., B.Z., X.W., D.Q. and J.W. conducted the experiments. D.Y. and H.L. analyzed the results. D.Y. wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by China Agriculture Research System of MOF and MARA (CARS-03) and the Agricultural Science and Technology Innovation Program (CAAS-ZDRW202002).
Data Availability Statement
No applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Li, H.; Dong, Z.; Ma, C.; Xia, Q.; Tian, X.; Sehgal, S.; Koo, D.-H.; Friebe, B.; Ma, P.; Liu, W. A spontaneous wheat-aegilops longissima translocation carrying Pm66 confers resistance to powdery mildew. Theor. Appl. Genet. 2020, 133, 1149–1159. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Martín, J.; Steuernagel, B.; Ghosh, S.; Herren, G.; Hurni, S.; Adamski, N.; Vrána, J.; Kubaláková, M.; Krattinger, S.G.; Wicker, T. Rapid gene isolation in barley and wheat by mutant chromosome sequencing. Genome Biol. 2016, 17, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Yahiaoui, N.; Srichumpa, P.; Dudler, R.; Keller, B. Genome analysis at different ploidy levels allows cloning of the powdery mildew resistance gene Pm3b from hexaploid wheat. Plant J. 2004, 37, 528–538. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Guo, G.; Wang, Y.; Hu, T.; Wang, L.; Li, J.; Qiu, D.; Li, Y.; Wu, Q.; Lu, P. A rare single nucleotide variant in Pm5e confers powdery mildew resistance in common wheat. New Phytol. 2020, 228, 1011–1026. [Google Scholar] [CrossRef] [PubMed]
- Hurni, S.; Brunner, S.; Buchmann, G.; Herren, G.; Jordan, T.; Krukowski, P.; Wicker, T.; Yahiaoui, N.; Mago, R.; Keller, B. Rye Pm8 and wheat Pm3 are orthologous genes and show evolutionary conservation of resistance function against powdery mildew. Plant J. 2013, 76, 957–969. [Google Scholar] [CrossRef]
- Zou, S.; Wang, H.; Li, Y.; Kong, Z.; Tang, D. The NB-LRR gene Pm60 confers powdery mildew resistance in wheat. New Phytol. 2018, 218, 298–309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, H.; Zhu, S.; Zhao, R.; Jiang, Z.; Ji, Y.; Ji, J.; Qiu, D.; Li, H.; Bie, T. Pm21, encoding a typical CC-NBS-LRR protein, confers broad-spectrum resistance to wheat powdery mildew disease. Mol. Plant 2018, 11, 879–882. [Google Scholar] [CrossRef] [Green Version]
- Dodds, P.N.; Rathjen, J.P. Plant immunity: Towards an integrated view of plant-pathogen interactions. Nat. Rev. Genet. 2010, 11, 539–548. [Google Scholar] [CrossRef]
- Kourelis, J.; Van Der Hoorn, R.A. Defended to the nines: 25 years of resistance gene cloning identifies nine mechanisms for R protein function. Plant Cell 2018, 30, 285–299. [Google Scholar] [CrossRef] [Green Version]
- Lu, P.; Guo, L.; Wang, Z.; Li, B.; Li, J.; Li, Y.; Qiu, D.; Shi, W.; Yang, L.; Wang, N. A rare gain of function mutation in a wheat tandem kinase confers resistance to powdery mildew. Nat. Commun. 2020, 11, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Cao, A.; Xing, L.; Wang, X.; Yang, X.; Wang, W.; Sun, Y.; Qian, C.; Ni, J.; Chen, Y.; Liu, D. Serine/threonine kinase gene stpk-v, a key member of powdery mildew resistance gene Pm21, confers powdery mildew resistance in wheat. Proc. Natl. Acad. Sci. USA 2011, 108, 7727–7732. [Google Scholar] [CrossRef] [Green Version]
- Krattinger, S.G.; Lagudah, E.S.; Spielmeyer, W.; Singh, R.P.; Huerta-Espino, J.; McFadden, H.; Bossolini, E.; Selter, L.L.; Keller, B. A putative ABC transporter confers durable resistance to multiple fungal pathogens in wheat. Science 2009, 323, 1360–1363. [Google Scholar] [CrossRef] [Green Version]
- Moore, J.W.; Herrera-Foessel, S.; Lan, C.; Schnippenkoetter, W.; Ayliffe, M.; Huerta-Espino, J.; Lillemo, M.; Viccars, L.; Milne, R.; Periyannan, S. A recently evolved hexose transporter variant confers resistance to multiple pathogens in wheat. Nat. Genet. 2015, 47, 1494–1498. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Hsam, S.; Zeller, F. Identification of powdery mildew resistance genes in common wheat (Triticum aestivum L. em Thell.). Ix. Cultivars, land races and breeding lines grown in china. Plant Breed. 1997, 116, 233–238. [Google Scholar] [CrossRef]
- Sánchez-Martín, J.; Widrig, V.; Herren, G.; Wicker, T.; Zbinden, H.; Gronnier, J.; Spörri, L.; Praz, C.R.; Heuberger, M.; Kolodziej, M.C. Wheat Pm4 resistance to powdery mildew is controlled by alternative splice variants encoding chimeric proteins. Nat. Plants 2021, 7, 327–341. [Google Scholar] [CrossRef] [PubMed]
- Briggle, L. Transfer of resistance to Erysiphe graminis f. sp. tritici from Khapli Emmer and Yuma Durum to Hexaploid Wheat 1. Crop Sci. 1966, 6, 459–461. [Google Scholar] [CrossRef]
- McIntosh, R.; Bennett, F.G. Cytogenetical studies in wheat. IX. Monosomic analyses, telocentric mapping and linkage relationships of genes Sr21, Pm4 and Mle. Aust. J. Biol. Sci. 1979, 32, 115–126. [Google Scholar]
- Hao, Y.; Liu, A.; Wang, Y.; Feng, D.; Gao, J.; Li, X.; Liu, S.; Wang, H. Pm23: A new allele of Pm4 located on chromosome 2AL in wheat. Theor. Appl. Genet. 2008, 117, 1205–1212. [Google Scholar] [CrossRef]
- Li, N.; Jia, H.; Kong, Z.; Tang, W.; Ding, Y.; Liang, J.; Ma, H.; Ma, Z. Identification and marker-assisted transfer of a new powdery mildew resistance gene at the Pm4 locus in common wheat. Mol. Breed. 2017, 37, 1–9. [Google Scholar] [CrossRef]
- Zhu, Z.; Zhou, R.; Kong, X.; Dong, Y.; Jia, J. Microsatellite markers linked to 2 powdery mildew resistance genes introgressed from Triticum carthlicum accession PS5 into common wheat. Genome 2005, 48, 585–590. [Google Scholar] [CrossRef]
- Niu, J.; Wang, B.; Wang, Y.; Cao, A.; Qi, Z.; Shen, T. Chromosome location and microsatellite markers linked to a powdery mildew resistance gene in wheat line ‘lankao 90 (6)’. Plant Breed. 2008, 127, 346–349. [Google Scholar] [CrossRef]
- Xu, W.; Li, C.; Hu, L.; Wang, H.; Dong, H.; Zhang, J.; Zan, X. Identification and molecular mapping of PmHNK54: A novel powdery mildew resistance gene in common wheat. Plant Breed. 2011, 130, 603–607. [Google Scholar] [CrossRef]
- Fu, B.; Chen, Y.; Li, N.; Ma, H.; Kong, Z.; Zhang, L.; Jia, H.; Ma, Z. pmX: A recessive powdery mildew resistance gene at the Pm4 locus identified in wheat landrace Xiaohongpi. Theor. Appl. Genet. 2013, 126, 913–921. [Google Scholar] [CrossRef] [PubMed]
- Mohler, V.; Bauer, C.; Schweizer, G.; Kempf, H.; Hartl, L. Pm50: A new powdery mildew resistance gene in common wheat derived from cultivated emmer. J. Appl. Genet. 2013, 54, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Michelmore, R.W.; Paran, I.; Kesseli, R. Identification of markers linked to disease-resistance genes by bulked segregant analysis: A rapid method to detect markers in specific genomic regions by using segregating populations. Proc. Natl. Acad. Sci. USA 1991, 88, 9828–9832. [Google Scholar] [CrossRef] [Green Version]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [Green Version]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef]
- Appels, R.; Eversole, K.; Stein, N.; Feuillet, C.; Keller, B.; Rogers, J.; Pozniak, C.J.; Choulet, F.; Distelfeld, A.; Poland, J. Shifting the limits in wheat research and breeding using a fully annotated reference genome. Science 2018, 361. [Google Scholar]
- Kim, S.; Scheffler, K.; Halpern, A.L.; Bekritsky, M.A.; Noh, E.; Källberg, M.; Chen, X.; Kim, Y.; Beyter, D.; Krusche, P. Strelka2: Fast and accurate calling of germline and somatic variants. Nat. Methods 2018, 15, 591–594. [Google Scholar] [CrossRef]
- Ma, Z.-Q.; Wei, J.-B.; Cheng, S.-H. PCR-based markers for the powdery mildew resistance gene Pm4a in wheat. Theor. Appl. Genet. 2004, 109, 140–145. [Google Scholar] [CrossRef]
- Schmolke, M.; Mohler, V.; Hartl, L.; Zeller, F.J.; Hsam, S.L. A new powdery mildew resistance allele at the Pm4 wheat locus transferred from einkorn (Triticum monococcum). Mol. Breed. 2012, 29, 449–456. [Google Scholar] [CrossRef]
- Wu, P.; Xie, J.; Hu, J.; Qiu, D.; Liu, Z.; Li, J.; Li, M.; Zhang, H.; Yang, L.; Liu, H. Development of molecular markers linked to powdery mildew resistance gene Pm4b by combining SNP discovery from transcriptome sequencing data with bulked segregant analysis (BSR-Seq) in wheat. Front. Plant Sci. 2018, 9, 95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, R.-H.; Meng, J.L. Mapdraw: A microsoft excel macro for drawing genetic linkage maps based on given genetic linkage data. Yi Chuan Hered. 2003, 25, 317–321. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delta delta c(t)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Wang, Z.; Li, L.; He, Z.; Duan, X.; Zhou, Y.; Chen, X.; Lillemo, M.; Singh, R.; Wang, H.; Xia, X. Seedling and adult plant resistance to powdery mildew in chinese bread wheat cultivars and lines. Plant Dis. 2005, 89, 457–463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parks, R.; Carbone, I.; Murphy, J.P.; Marshall, D.; Cowger, C. Virulence structure of the eastern US wheat powdery mildew population. Plant Dis. 2008, 92, 1074–1082. [Google Scholar] [CrossRef]
- Singh, S.P.; Hurni, S.; Ruinelli, M.; Brunner, S.; Sanchez-Martin, J.; Krukowski, P.; Peditto, D.; Buchmann, G.; Zbinden, H.; Keller, B. Evolutionary divergence of the rye Pm17 and Pm8 resistance genes reveals ancient diversity. Plant Mol. Biol. 2018, 98, 249–260. [Google Scholar] [CrossRef]
- Xing, L.; Hu, P.; Liu, J.; Witek, K.; Zhou, S.; Xu, J.; Zhou, W.; Gao, L.; Huang, Z.; Zhang, R. Pm21 from Haynaldia villosa encodes a CC-NBS-LRR protein conferring powdery mildew resistance in wheat. Mol. Plant 2018, 11, 874–878. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Dong, L.; Li, B.; Wang, Z.; Xie, J.; Qiu, D.; Li, Y.; Shi, W.; Yang, L.; Wu, Q. A CNL protein in wild emmer wheat confers powdery mildew resistance. New Phytol. 2020, 228, 1027–1037. [Google Scholar] [CrossRef] [PubMed]
- Fu, D.; Uauy, C.; Distelfeld, A.; Blechl, A.; Epstein, L.; Chen, X.; Sela, H.; Fahima, T.; Dubcovsky, J. A kinase-start gene confers temperature-dependent resistance to wheat stripe rust. Science 2009, 323, 1357–1360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brueggeman, R.; Druka, A.; Nirmala, J.; Cavileer, T.; Drader, T.; Rostoks, N.; Mirlohi, A.; Bennypaul, H.; Gill, U.; Kudrna, D. The stem rust resistance gene Rpg5 encodes a protein with nucleotide-binding-site, leucine-rich, and protein kinase domains. Proc. Natl. Acad. Sci. USA 2008, 105, 14970–14975. [Google Scholar] [CrossRef] [Green Version]
- Yi, Y.; Liu, H.; Huang, X.; An, L.; Wang, F.; Wang, X. Development of molecular markers linked to the wheat powdery mildew resistance gene Pm4b and marker validation for molecular breeding. Plant Breed. 2008, 127, 116–120. [Google Scholar] [CrossRef]
- Niu, J.-S.; Jia, H.-Y.; Jun, Y.; Wang, B.-Q.; Shen, T.-M. Development of an STS marker linked to powdery mildew resistance genes PmLK906 and Pm4a by gene chip hybridization. Agric. Sci. China 2010, 9, 331–336. [Google Scholar] [CrossRef]
- Ullah, K.N.; Li, N.; Shen, T.; Wang, P.; Tang, W.; Ma, S.; Zhang, Z.; Jia, H.; Kong, Z.; Ma, Z. Fine mapping of powdery mildew resistance gene Pm4e in bread wheat (Triticum aestivum L.). Planta 2018, 248, 1319–1328. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).