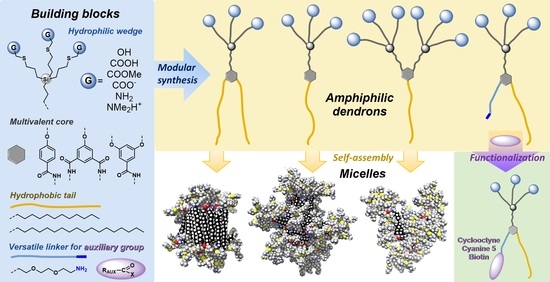
Adaptive Synthesis of Functional Amphiphilic Dendrons as a Novel Approach to Artificial Supramolecular Objects
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterization of Carbosilane Amphiphilic Dendrons
2.2. Dynamic Light Scattering and Zeta Potential
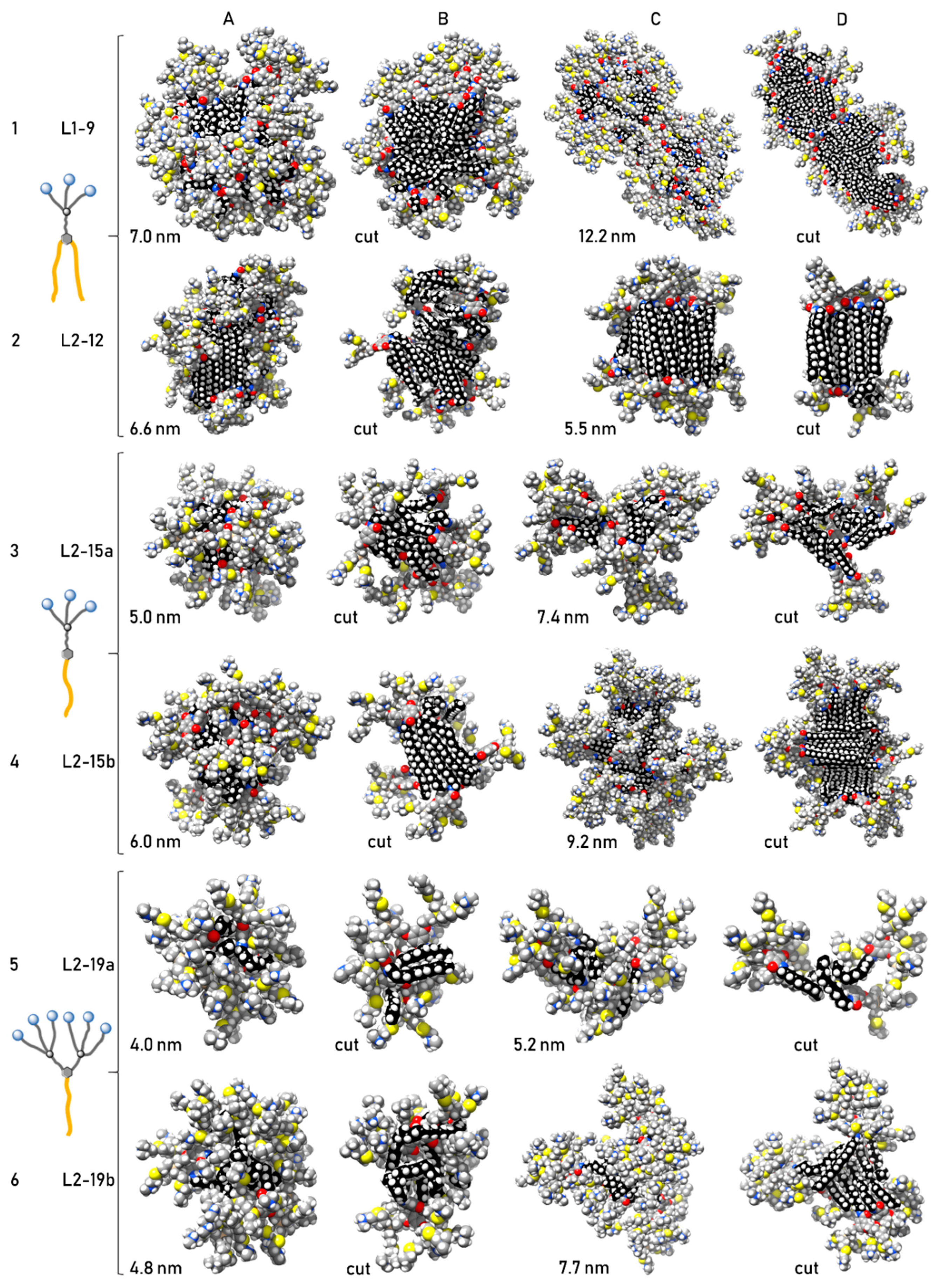
2.3. Computational Study
3. Materials and Methods
3.1. General Remarks
3.2. Dynamic Light Scattering (DLS)
3.3. Computational Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Filipczak, N.; Pan, J.; Yalamarty, S.S.K.; Torchilin, V.P. Recent advancements in liposome technology. Adv. Drug Deliv. Rev. 2020, 156, 4–22. [Google Scholar] [CrossRef] [PubMed]
- Soussan, E.; Cassel, S.; Blanzat, M.; Rico-Lattes, I. Drug delivery by soft matter: Matrix and vesicular carriers. Angew. Chem. Int. Ed. 2009, 48, 274–288. [Google Scholar] [CrossRef] [PubMed]
- Thota, B.N.S.; Urner, L.H.; Haag, R. Supramolecular architectures of dendritic amphiphiles in water. Chem. Rev. 2016, 116, 2079–2102. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, M.; Shrestha, L.K.; Mori, T.; Ji, Q.; Hill, J.P.; Ariga, K. Amphiphile nanoarchitectonics: From basic physical chemistry to advanced applications. Phys. Chem. Chem. Phys. 2013, 15, 10580–10611. [Google Scholar] [CrossRef]
- Eivazihollagh, A.; Svanedal, I.; Edlund, H.; Norgren, M. On chelating surfactants: Molecular perspectives and application prospects. J. Mol. Liq. 2019, 278, 688–705. [Google Scholar] [CrossRef]
- Polarz, S.; Landsmann, S.; Klaiber, A. Hybrid Surfactant Systems with Inorganic Constituents. Angew. Chem. Int. Ed. 2014, 53, 946–954. [Google Scholar] [CrossRef] [Green Version]
- Tevet, S.; Wagle, S.S.; Slor, G.; Amir, R.J. Tuning the Reactivity of Micellar Nanoreactors by Precise Adjustments of the Amphiphile and Substrate Hydrophobicity. Macromolecules 2021, 54, 11419–11426. [Google Scholar] [CrossRef]
- Puras, G.; Mashal, M.; Zárate, J.; Agirre, M.; Ojeda, E.; Grijalvo, S.; Eritja, R.; Diaz-Tahoces, A.; Martínez Navarrete, G.; Avilés-Trigueros, M.; et al. A novel cationic niosome formulation for gene delivery to the retina. J. Control. Release 2014, 174, 27–36. [Google Scholar] [CrossRef]
- Dailing, E.A.; Kilchrist, K.V.; Tierney, J.W.; Fletcher, R.B.; Evans, B.C.; Duvall, C.L. Modifying Cell Membranes with Anionic Polymer Amphiphiles Potentiates Intracellular Delivery of Cationic Peptides. ACS Appl. Mater. Interfaces 2020, 12, 50222–50235. [Google Scholar] [CrossRef]
- Kalhapure, R.S.; Jadhav, M.; Rambharose, S.; Mocktar, C.; Singh, S.; Renukuntla, J.; Govender, T. pH-responsive chitosan nanoparticles from a novel twin-chain anionic amphiphile for controlled and targeted delivery of vancomycin. Colloids Surf. B Biointerfaces 2017, 158, 650–657. [Google Scholar] [CrossRef]
- Astruc, D.; Boisselier, E.; Ornelas, C. Dendrimers designed for functions: From physical, photophysical, and supramolecular properties to applications in sensing, catalysis, molecular electronics, photonics, and nanomedicine. Chem. Rev. 2010, 110, 1857–1959. [Google Scholar] [CrossRef] [PubMed]
- Apartsin, E.; Caminade, A. Supramolecular Self-Associations of Amphiphilic Dendrons and Their Properties. Chem. A Eur. J. 2021, 27, 1–24. [Google Scholar] [CrossRef]
- Chen, J.; Ellert-Miklaszewska, A.; Garofalo, S.; Dey, A.K.; Tang, J.; Jiang, Y.; Clément, F.; Marche, P.N.; Liu, X.; Kaminska, B.; et al. Synthesis and use of an amphiphilic dendrimer for siRNA delivery into primary immune cells. Nat. Protoc. 2021, 16, 327–351. [Google Scholar] [CrossRef] [PubMed]
- Lyu, Z.; Ding, L.; Huang, A.Y.T.; Kao, C.L.; Peng, L. Poly(amidoamine)dendrimers: Covalent and supramolecular synthesis. Mater. Today Chem. 2019, 13, 34–48. [Google Scholar] [CrossRef]
- Nierengarten, J.-F.; Eckert, J.-F.; Rio, Y.; del Pilar Carreon, M.; Gallani, J.-L.; Guillon, D. Amphiphilic Diblock Dendrimers: Synthesis and Incorporation in Langmuir and Langmuir−Blodgett Films. J. Am. Chem. Soc. 2001, 123, 9743–9748. [Google Scholar] [CrossRef] [PubMed]
- Hawker, C.J.; Fréchet, J.M.J. Unusual Macromolecular Architectures: The Convergent Growth Approach to Dendritic Polyesters and Novel Block Copolymers. J. Am. Chem. Soc. 1992, 114, 8405–8413. [Google Scholar] [CrossRef]
- Zibarov, A.; Oukhrib, A.; Aujard Catot, J.; Turrin, C.-O.; Caminade, A. AB5 Derivatives of Cyclotriphosphazene for the Synthesis of Dendrons and Their Applications. Molecules 2021, 26, 4017. [Google Scholar] [CrossRef]
- Keller, M.; Collière, V.; Reiser, O.; Caminade, A.M.; Majoral, J.P.; Ouali, A. Pyrene-tagged dendritic catalysts noncovalently grafted onto magnetic Co/C nanoparticles: An efficient and recyclable system for drug synthesis. Angew. Chem. Int. Ed. 2013, 52, 3626–3629. [Google Scholar] [CrossRef]
- Qiu, J.; Chen, L.; Zhan, M.; Laurent, R.; Bignon, J.; Mignani, S.; Shi, X.; Caminade, A.M.; Majoral, J.P. Facile Synthesis of Amphiphilic Fluorescent Phosphorus Dendron-Based Micelles as Antiproliferative Agents: First Investigations. Bioconjug. Chem. 2021, 32, 339–349. [Google Scholar] [CrossRef]
- Krasheninina, O.A.; Apartsin, E.K.; Fuentes, E.; Szulc, A.; Ionov, M.; Venyaminova, A.G.; Shcharbin, D.; de la Mata, F.J.; Bryszewska, M.; Gómez, R. Complexes of pro-apoptotic sirnas and carbosilane dendrimers: Formation and effect on cancer cells. Pharmaceutics 2019, 11, 25. [Google Scholar] [CrossRef] [Green Version]
- Gutierrez-Ulloa, C.E.; Buyanova, M.Y.; Apartsin, E.K.; Venyaminova, A.G.; De La Mata, F.J.; Valiente, M.; Gómez, R. Amphiphilic carbosilane dendrons as a novel synthetic platform toward micelle formation. Org. Biomol. Chem. 2017, 15, 7352–7364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, S.K.; Zimmerman, S.C. Water-soluble polyglycerol dendrimers with two orthogonally reactive core functional groups for one-pot functionalization. Macromolecules 2015, 48, 2504–2508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, S.; Moussodia, R.O.; Sun, H.J.; Leowanawat, P.; Muncan, A.; Nusbaum, C.D.; Chelling, K.M.; Heiney, P.A.; Klein, M.L.; André, S.; et al. Mimicking biological membranes with programmable glycan ligands self-assembled from amphiphilic Janus glycodendrimers. Angew. Chem. Int. Ed. 2014, 53, 10899–10903. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Ludwig, A.K.; Romanò, C.; Buzzacchera, I.; Sherman, S.E.; Vetro, M.; Vértesy, S.; Kaltner, H.; Reed, E.H.; Möller, M.; et al. Exploring functional pairing between surface glycoconjugates and human galectins using programmable glycodendrimersomes. Proc. Natl. Acad. Sci. USA 2018, 115, E2509–E2518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiao, Q.; Sherman, S.E.; Wilner, S.E.; Zhou, X.; Dazen, C.; Baumgart, T.; Reed, E.H.; Hammer, D.A.; Shinoda, W.; Klein, M.L.; et al. Janus dendrimersomes coassembled from fluorinated, hydrogenated, and hybrid Janus dendrimers as models for cell fusion and fission. Proc. Natl. Acad. Sci. USA 2017, 114, E7045–E7053. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiao, Q.; Rubien, J.D.; Wang, Z.; Reed, E.H.; Hammer, D.A.; Sahoo, D.; Heiney, P.A.; Yadavalli, S.S.; Goulian, M.; Wilner, S.E.; et al. Self-Sorting and Coassembly of Fluorinated, Hydrogenated, and Hybrid Janus Dendrimers into Dendrimersomes. J. Am. Chem. Soc. 2016, 138, 12655–12663. [Google Scholar] [CrossRef] [Green Version]
- Krupková, A.; Kubátová, K.; Šťastná, L.Č.; Cuřínová, P.; Müllerová, M.; Karban, J.; Čermák, J.; Strašák, T. Poly(Imidazolium) carbosilane dendrimers: Synthesis, catalytic activity in redox esterification of α,β-unsaturated aldehydes and recycling via organic solvent nanofiltration. Catalysts 2021, 11, 1317. [Google Scholar] [CrossRef]
- Müllerová, M.; Maciel, D.; Nunes, N.; Wrobel, D.; Stofik, M.; Červenková Šťastná, L.; Krupková, A.; Cuřínová, P.; Nováková, K.; Božík, M.; et al. Carbosilane Glycodendrimers for Anticancer Drug Delivery: Synthetic Route, Characterization, and Biological Effect of Glycodendrimer–Doxorubicin Complexes. Biomacromolecules 2021, 23, 276–290. [Google Scholar] [CrossRef]
- Herma, R.; Wrobel, D.; Liegertová, M.; Müllerová, M.; Strašák, T.; Maly, M.; Semerádtová, A.; Štofik, M.; Appelhans, D.; Maly, J. Carbosilane dendrimers with phosphonium terminal groups are low toxic non-viral transfection vectors for siRNA cell delivery. Int. J. Pharm. 2019, 562, 51–65. [Google Scholar] [CrossRef]
- Strašák, T.; Malý, J.; Wróbel, D.; Malý, M.; Herma, R.; Čermák, J.; Müllerová, M.; Št′astná, L.Č.; Cuřínová, P. Phosphonium carbosilane dendrimers for biomedical applications-synthesis, characterization and cytotoxicity evaluation. RSC Adv. 2017, 7, 18724–18744. [Google Scholar] [CrossRef] [Green Version]
- Liegertová, M.; Wrobel, D.; Herma, R.; Müllerová, M.; Šťastná, L.Č.; Cuřínová, P.; Strašák, T.; Malý, M.; Čermák, J.; Smejkal, J.; et al. Evaluation of toxicological and teratogenic effects of carbosilane glucose glycodendrimers in zebrafish embryos and model rodent cell lines. Nanotoxicology 2018, 12, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Ordanini, S.; Cellesi, F. Complex Polymeric Architectures Self-Assembling in Unimolecular Micelles: Preparation, Characterization and Drug Nanoencapsulation. Pharmaceutics 2018, 10, 209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sherman, S.E.; Xiao, Q.; Percec, V. Mimicking complex biological membranes and their programmable glycan ligands with dendrimersomes and glycodendrimersomes. Chem. Rev. 2017, 117, 6538–6631. [Google Scholar] [CrossRef] [PubMed]
- Franc, G.; Kakkar, A.K. “Click” methodologies: Efficient, simple and greener routes to design dendrimers. Chem. Soc. Rev. 2010, 39, 1536–1544. [Google Scholar] [CrossRef]
- Marchetti, P.; Jimenez Solomon, M.F.; Szekely, G.; Livingston, A.G. Molecular separation with organic solvent nanofiltration: A critical review. Chem. Rev. 2014, 114, 10735–10806. [Google Scholar] [CrossRef]
- Shende, V.S.; Saptal, V.B.; Bhanage, B.M. Recent Advances Utilized in the Recycling of Homogeneous Catalysis. Chem. Rec. 2019, 1–23. [Google Scholar] [CrossRef]
- Vandezande, P.; Gevers, L.E.M.; Vankelecom, I.F.J. Solvent resistant nanofiltration: Separating on a molecular level. Chem. Soc. Rev. 2008, 37, 365–405. [Google Scholar] [CrossRef]
- Cuřínová, P.; Winkler, M.; Krupková, A.; Císařová, I.; Budka, J.; Wun, C.N.; Blechta, V.; Malý, M.; Červenková Št’astná, L.; Sýkora, J.; et al. Transport of Anions across the Dialytic Membrane Induced by Complexation toward Dendritic Receptors. ACS Omega 2021, 6, 15514–15522. [Google Scholar] [CrossRef]
- Červenková Št’astná, L.; Krupková, A.; Petrickovic, R.; Müllerová, M.; Matoušek, J.; Koštejn, M.; Cuřínová, P.; Jandová, V.; Šabata, S.; Strašák, T. Multivalent Bifunctional Carbosilane Dendrimer-Supported Ammonium and Phosphonium Organocatalysts for the Coupling of CO2 and Epoxides. ACS Sustain. Chem. Eng. 2020, 8, 11692–11703. [Google Scholar] [CrossRef]
- Gong, C.; Glass, T.E.; Gibson, H.W. Poly(urethane/crown ether rotaxane)s with Solvent Switchable Microstructures. Macromolecules 1998, 31, 308–313. [Google Scholar] [CrossRef]
- Uno, M.; Ban, H.S.; Nabeyama, W.; Nakamura, H. de novo Design and synthesis of N-benzylanilines as new candidates for VEGFR tyrosine kinase inhibitors. Org. Biomol. Chem. 2008, 6, 979. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, I.; Falck-Pedersen, M.L.; Undheim, K. Synthesis of silacycloalkenes and silaspirenes by Ru(II)-catalyzed ring-closing metathesis reactions. J. Organomet. Chem. 2001, 625, 160–172. [Google Scholar] [CrossRef]
- Favre, A.; Grugier, J.; Brans, A.; Joris, B.; Marchand-Brynaert, J. 6-Aminopenicillanic acid (6-APA) derivatives equipped with anchoring arms. Tetrahedron 2012, 68, 10818–10826. [Google Scholar] [CrossRef]
- Bayly, C.I.; Cieplak, P.; Cornell, W.; Kollman, P.A. A well-behaved electrostatic potential based method using charge restraints for deriving atomic charges: The RESP model. J. Phys. Chem. 1993, 97, 10269–10280. [Google Scholar] [CrossRef]
- Vanquelef, E.; Simon, S.; Marquant, G.; Garcia, E.; Klimerak, G.; Delepine, J.C.; Cieplak, P.; Dupradeau, F.-Y. R.E.D. Server: A web service for deriving RESP and ESP charges and building force field libraries for new molecules and molecular fragments. Nucleic Acids Res. 2011, 39, W511–W517. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, M.W.; Baldridge, K.K.; Boatz, J.A.; Elbert, S.T.; Gordon, M.S.; Jensen, J.H.; Koseki, S.; Matsunaga, N.; Nguyen, K.A.; Su, S.; et al. General atomic and molecular electronic structure system. J. Comput. Chem. 1993, 14, 1347–1363. [Google Scholar] [CrossRef]
- Gordon, M.S.; Schmidt, M.W. Advances in electronic structure theory. In Theory and Applications of Computational Chemistry; Elsevier: Amsterdam, The Netherlands, 2005; pp. 1167–1189. [Google Scholar]
- Wang, J.; Wolf, R.M.; Caldwell, J.W.; Kollman, P.A.; Case, D.A. Development and testing of a general amber force field. J. Comput. Chem. 2004, 25, 1157–1174. [Google Scholar] [CrossRef]
- Case, D.A.; Brozell, S.R.; Cerutti, D.S.; Cheatham, T.E., III; Cruzeiro, V.W.D.; Darden, T.A.; Duke, R.E.; Ghoreishi, D.; Gohlke, H.; Goetz, A.W.; et al. AMBER. 2018. Available online: https://ambermd.org/doc12/Amber18.pdf (accessed on 13 January 2022).
- Lii, J.-H.; Allinger, N.L. The MM3 force field for amides, polypeptides and proteins. J. Comput. Chem. 1991, 12, 186–199. [Google Scholar] [CrossRef]
- Martínez, L.; Andrade, R.; Birgin, E.G.; Martínez, J.M. PACKMOL: A package for building initial configurations for molecular dynamics simulations. J. Comput. Chem. 2009, 30, 2157–2164. [Google Scholar] [CrossRef]
- Jorgensen, W.L.; Chandrasekhar, J.; Madura, J.D.; Impey, R.W.; Klein, M.L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 1983, 79, 926–935. [Google Scholar] [CrossRef]
- Ryckaert, J.-P.; Ciccotti, G.; Berendsen, H.J. Numerical integration of the cartesian equations of motion of a system with constraints: Molecular dynamics of n-alkanes. J. Comput. Phys. 1977, 23, 327–341. [Google Scholar] [CrossRef] [Green Version]
- Wu, X.; Brooks, B.R.; Vanden-Eijnden, E. Self-guided Langevin dynamics via generalized Langevin equation. J. Comput. Chem. 2016, 37, 595–601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Götz, A.W.; Williamson, M.J.; Xu, D.; Poole, D.; Le Grand, S.; Walker, R.C. Routine Microsecond Molecular Dynamics Simulations with AMBER on GPUs. 1. Generalized Born. J. Chem. Theory Comput. 2012, 8, 1542–1555. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin Win, K.; Feng, S.S. Effects of particle size and surface coating on cellular uptake of polymeric nanoparticles for oral delivery of anticancer drugs. Biomaterials 2005, 26, 2713–2722. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Wei, X.; Chen, Z.; Zhang, X.; Yang, G.; Zhou, S. Multifunctional nanoplatforms for subcellular delivery of drugs in cancer therapy. Prog. Mater. Sci. 2020, 107, 100599. [Google Scholar] [CrossRef]
- Kheraldine, H.; Rachid, O.; Habib, A.M.; Al Moustafa, A.E.; Benter, I.F.; Akhtar, S. Emerging innate biological properties of nano-drug delivery systems: A focus on PAMAM dendrimers and their clinical potential. Adv. Drug Deliv. Rev. 2021, 178, 113908. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.G. How lipids affect the activities of integral membrane proteins. Biochim. Biophys. Acta Biomembr. 2004, 1666, 62–87. [Google Scholar] [CrossRef] [Green Version]
- Malik, N.; Wiwattanapatapee, R.; Klopsch, R.; Lorenz, K.; Frey, H.; Weener, J.W.; Meijer, E.W.; Paulus, W.; Duncan, R. Dendrimers: Relationship between structure and biocompatibility in vitro, and preliminary studies on the biodistribution of 125I-labelled polyamidoamine dendrimers in vivo. J. Control. Release 2000, 65, 133–148. [Google Scholar] [CrossRef]
- Shao, X.-R.; Wei, X.-Q.; Song, X.; Hao, L.-Y.; Cai, X.-X.; Zhang, Z.-R.; Peng, Q.; Lin, Y.-F. Independent effect of polymeric nanoparticle zeta potential/surface charge, on their cytotoxicity and affinity to cells. Cell Prolif. 2015, 48, 465–474. [Google Scholar] [CrossRef]
- Ziemba, B.; Janaszewska, A.; Ciepluch, K.; Krotewicz, M.; Fogel, W.A.; Appelhans, D.; Voit, B.; Bryszewska, M.; Klajnert, B. In vivo toxicity of poly(propyleneimine) dendrimers. J. Biomed. Mater. Res. Part A 2011, 99A, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Nagarajan, R. (Ed.) Self-Assembly: From Surfactants to Nanoparticles; John Wiley&Sons, Inc.: Hoboken, NJ, USA, 2018; ISBN 9781119001379. [Google Scholar]
- Xu, L.; Shao, L.; Chen, L.; Hu, M.; Bi, Y. Synthesis and Solution Properties of a Novel Thermosensitive Poly(benzyl ether) Dendron with Oligoethyleneoxy Chains at the Periphery. Chem. Lett. 2010, 39, 1177–1179. [Google Scholar] [CrossRef]
- Xiang, S.; Wagner, J.; Lückerath, T.; Müllen, K.; Ng, D.Y.W.; Hedrich, J.; Weil, T. Reversing A β Fibrillation and Inhibiting A β Primary Neuronal Cell Toxicity Using Amphiphilic Polyphenylene Dendrons. Adv. Healthc. Mater. 2021, 2101854. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Peng, F.; Kang, Y.; Gong, D.; Fan, J.; Zhang, W.; Qiu, F. High-Loading Self-Assembling Peptide Nanoparticles as a Lipid-Free Carrier for Hydrophobic General Anesthetics. Int. J. Nanomed. 2021, 16, 5317. [Google Scholar] [CrossRef] [PubMed]
- Simms, B.L.; Ji, N.; Chandrasiri, I.; Zia, M.F.; Udemgba, C.S.; Kaur, R.; Delcamp, J.H.; Flynt, A.; Tan, C.; Watkins, D.L. Physicochemical properties and bio-interfacial interactions of surface modified PDLLA-PAMAM linear dendritic block copolymers. J. Polym. Sci. 2021, 59, 2177–2192. [Google Scholar] [CrossRef]
- Chandrasiri, I.; Abebe, D.G.; Loku Yaddehige, M.; Williams, J.S.D.; Zia, M.F.; Dorris, A.; Barker, A.; Simms, B.L.; Parker, A.; Vinjamuri, B.P.; et al. Self-Assembling PCL-PAMAM Linear Dendritic Block Copolymers (LDBCs) for Bioimaging and Phototherapeutic Applications. ACS Appl. Bio Mater. 2020, 3, 5664–5677. [Google Scholar] [CrossRef] [PubMed]
- Manunta, M.; Tan, P.H.; Sagoo, P.; Kashe, K.; George, A.J.T. Gene delivery by dendrimers operates via a cholesterol dependent pathway. Nucleic Acids Res. 2004, 32, 2730–2739. [Google Scholar] [CrossRef] [Green Version]
- Tan, Q.Y.; Wang, N.; Yang, H.; Chen, L.; Xiong, H.R.; Zhang, L.K.; Liu, J.; Zhao, C.J.; Zhang, J.Q. Preparation and characterization of lipid vesicles containing uricase. Drug Deliv. 2010, 17, 28–37. [Google Scholar] [CrossRef]
- Sze, A.; Erickson, D.; Ren, L.; Li, D. Zeta-potential measurement using the Smoluchowski equation and the slope of the current time relationship in electroosmotic flow. J. Colloid Interface Sci. 2003, 261, 402–410. [Google Scholar] [CrossRef]




| Amphiphilic Structure | Size (nm) | Zeta Potential 1 (mV) | ||||||
|---|---|---|---|---|---|---|---|---|
| 0.1 (µM) | 0.5 (µM) | 1 (µM) | 5 (µM) | 10 (µM) | 50 (µM) | 100 (µM) | ||
| L1–9 | 69 ± 13 | 55 ± 5 | 52 ± 4 | 47 ± 18 | 28 ± 2 | 8 ± 1 | 8 ± 1 | 39.3 ± 3.0 |
| L2–12 | 124 ± 2 | 76 ± 34 | 25 ± 19 | 13 ± 2 | 36 ± 2 | 24 ± 18 | 7 ± 1 | 71.5 ± 3.5 |
| L2–15a | 78 ± 27 | 75 ± 5 | 104 ± 1 | 68 ± 8 | 73 ± 4 | 60 ± 7 | 70 ± 1 | 62.7 ± 7.0 |
| L2–15b | 20 ± 4 | 20 ± 8 | 20 ± 6 | 25 ± 4 | 23 ± 3 | 25 ± 4 | 28 ± 5 | 68.5 ± 0.5 |
| L2–19a | 17 ± 4 | 18 ± 6 | 31 ± 47 | 36 ± 29 | 67 ± 37 | 79 ± 9 | 63 ± 11 | 71.7 ± 1.6 |
| L2–19b | 6 ± 7 | 3 ± 1 | 7 ± 3 | 7 ± 4 | 6 ± 2 | 7 ± 2 | 7 ± 1 | 52.8 ± 8.6 |
| L1–8 | 6 ± 2 | 7 ± 1 | 5 ± 3 | 7 ± 1 | 7 ± 4 | 7 ± 2 | 6 ± 2 | −27.2 ± 3.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Edr, A.; Wrobel, D.; Krupková, A.; Šťastná, L.Č.; Cuřínová, P.; Novák, A.; Malý, J.; Kalasová, J.; Malý, J.; Malý, M.; et al. Adaptive Synthesis of Functional Amphiphilic Dendrons as a Novel Approach to Artificial Supramolecular Objects. Int. J. Mol. Sci. 2022, 23, 2114. https://doi.org/10.3390/ijms23042114
Edr A, Wrobel D, Krupková A, Šťastná LČ, Cuřínová P, Novák A, Malý J, Kalasová J, Malý J, Malý M, et al. Adaptive Synthesis of Functional Amphiphilic Dendrons as a Novel Approach to Artificial Supramolecular Objects. International Journal of Molecular Sciences. 2022; 23(4):2114. https://doi.org/10.3390/ijms23042114
Chicago/Turabian StyleEdr, Antonín, Dominika Wrobel, Alena Krupková, Lucie Červenková Šťastná, Petra Cuřínová, Aleš Novák, Jan Malý, Jitka Kalasová, Jan Malý, Marek Malý, and et al. 2022. "Adaptive Synthesis of Functional Amphiphilic Dendrons as a Novel Approach to Artificial Supramolecular Objects" International Journal of Molecular Sciences 23, no. 4: 2114. https://doi.org/10.3390/ijms23042114