Identification of Nifurtimox and Chrysin as Anti-Influenza Virus Agents by Clinical Transcriptome Signature Reversion
Abstract
:1. Introduction
2. Results
2.1. Identification of IAV-Infection-Related Informative Genes
2.2. Compound Screening by TSR with CMap
2.3. Identification of Nifurtimox and Chrysin with Activities against IAV Infection
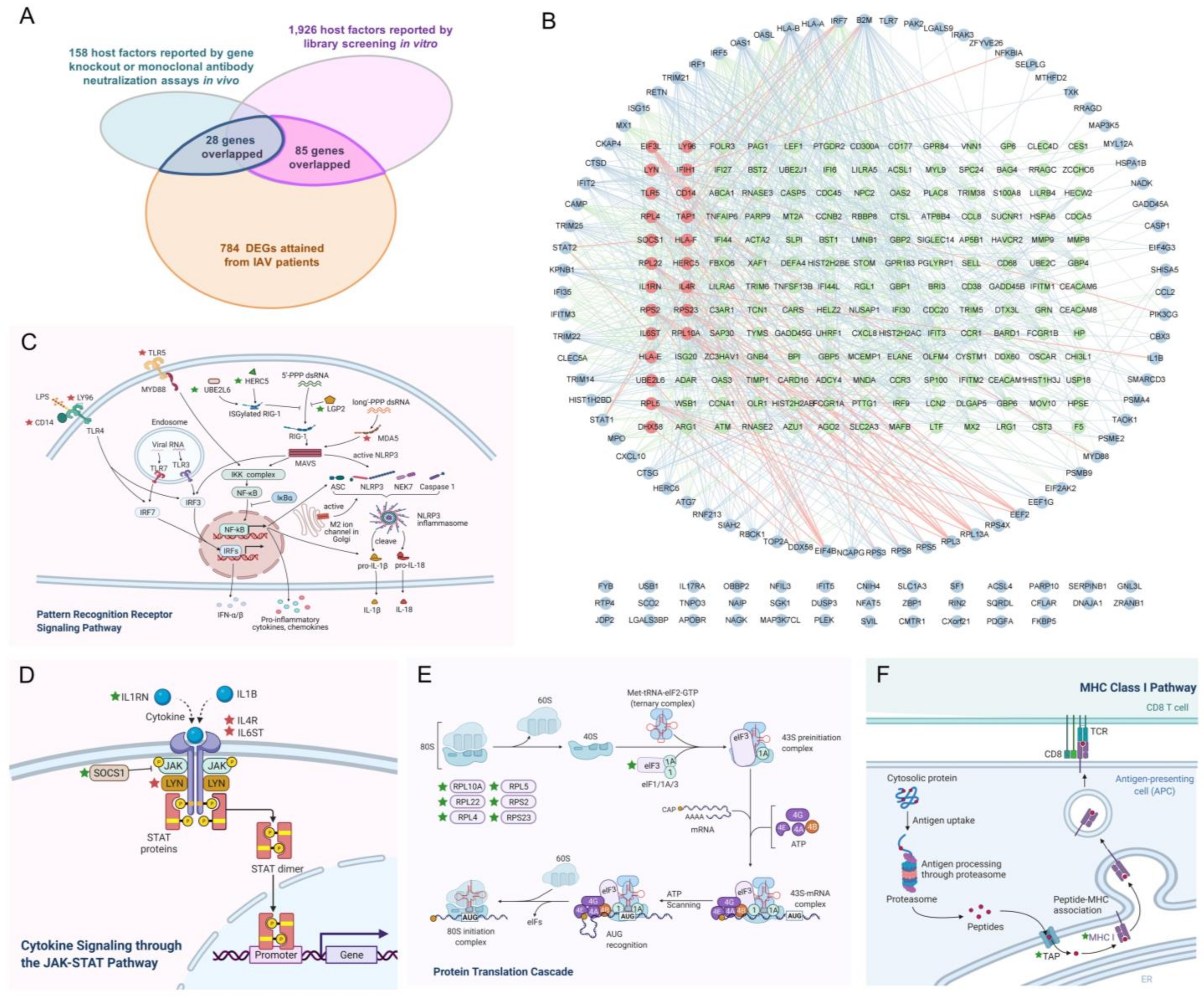
2.4. Multiple Informative Genes Reversed by Chrysin and Nifurtimox Played Crucial Roles in the Two Compounds’ Anti-IAV Activities
2.5. Chrysin and Nifurtimox Exerted Their Anti-IAV Effects through Regulating Multiple Pathways
2.6. Combination of Nifurtimox and Chrysin Exhibited a Synergistic Effect against IAV Infection
3. Discussion
4. Materials and Methods
4.1. Acquirement of Clinical Database
4.2. Identification of Informative Genes
4.3. Compound Screening by Transcriptome Reversal Paradigm with CMap
4.4. Cells, Viruses, and Reagents
4.5. Cell Viability Assay
4.6. Cytopathic Effect Assay
4.7. Virion RNA Detection Assay
4.8. Hemagglutinin Detection Assay
4.9. Total RNA Extraction, RNA Sequencing, and Differentially Expressed Gene Analysis
4.10. Network-Based Analysis of Drug Combinations
4.11. Drug Combination Assay
4.12. Bioinformatics Analysis
4.13. Statistical Analysis
4.14. Code Availability
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Influenza (Seasonal). Available online: https://www.who.int/en/news-room/fact-sheets/detail/influenza-(seasonal) (accessed on 28 January 2021).
- Sasaki, E.; Hamaguchi, I.; Mizukami, T. Pharmacodynamic and Safety Considerations for Influenza Vaccine and Adjuvant Design. Expert Opin. Drug Metab. Toxicol. 2020, 16, 1051–1061. [Google Scholar] [CrossRef] [PubMed]
- Center for Drug Evaluation and Research. Influenza (Flu) Antiviral Drugs and Related Information; FDA: Silver Spring, MD, USA, 2020.
- CDC. Influenza Antiviral Drug Resistance. Available online: https://www.cdc.gov/flu/treatment/antiviralresistance.htm (accessed on 28 January 2021).
- Bright, R.A.; Shay, D.K.; Shu, B.; Cox, N.J.; Klimov, A.I. Adamantane Resistance among Influenza A Viruses Isolated Early during the 2005-2006 Influenza Season in the United States. JAMA 2006, 295, 891–894. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Samson, M.; Pizzorno, A.; Abed, Y.; Boivin, G. Influenza Virus Resistance to Neuraminidase Inhibitors. Antivir. Res. 2013, 98, 174–185. [Google Scholar] [CrossRef]
- Chen, B.; Garmire, L.; Calvisi, D.F.; Chua, M.-S.; Kelley, R.K.; Chen, X. Harnessing Big “omics” Data and AI for Drug Discovery in Hepatocellular Carcinoma. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 238–251. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.; Domrachev, M.; Lash, A.E. Gene Expression Omnibus: NCBI Gene Expression and Hybridization Array Data Repository. Nucleic Acids Res. 2002, 30, 207–210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brazma, A.; Parkinson, H.; Sarkans, U.; Shojatalab, M.; Vilo, J.; Abeygunawardena, N.; Holloway, E.; Kapushesky, M.; Kemmeren, P.; Lara, G.G.; et al. ArrayExpress—A Public Repository for Microarray Gene Expression Data at the EBI. Nucleic Acids Res. 2003, 31, 68–71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lamb, J.; Crawford, E.D.; Peck, D.; Modell, J.W.; Blat, I.C.; Wrobel, M.J.; Lerner, J.; Brunet, J.-P.; Subramanian, A.; Ross, K.N.; et al. The Connectivity Map: Using Gene-Expression Signatures to Connect Small Molecules, Genes, and Disease. Science 2006, 313, 1929–1935. [Google Scholar] [CrossRef] [Green Version]
- Karczewski, K.J.; Snyder, M.P. Integrative Omics for Health and Disease. Nat. Rev. Genet. 2018, 19, 299–310. [Google Scholar] [CrossRef]
- Siavelis, J.C.; Bourdakou, M.M.; Athanasiadis, E.I.; Spyrou, G.M.; Nikita, K.S. Bioinformatics Methods in Drug Repurposing for Alzheimer’s Disease. Brief. Bioinform. 2016, 17, 322–335. [Google Scholar] [CrossRef]
- Le, B.L.; Iwatani, S.; Wong, R.J.; Stevenson, D.K.; Sirota, M. Computational Discovery of Therapeutic Candidates for Preventing Preterm Birth. JCI Insight 2020, 5, e133761. [Google Scholar] [CrossRef]
- Kim, I.-W.; Jang, H.; Kim, J.H.; Kim, M.G.; Kim, S.; Oh, J.M. Computational Drug Repositioning for Gastric Cancer Using Reversal Gene Expression Profiles. Sci. Rep. 2019, 9, 2660. [Google Scholar] [CrossRef] [Green Version]
- Dönertaş, H.M.; Fuentealba Valenzuela, M.; Partridge, L.; Thornton, J.M. Gene Expression-Based Drug Repurposing to Target Aging. Aging Cell 2018, 17, e12819. [Google Scholar] [CrossRef] [Green Version]
- GEO. Accession Viewer. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE111368 (accessed on 15 February 2021).
- Dunning, J.; Blankley, S.; Hoang, L.T.; Cox, M.; Graham, C.M.; James, P.L.; Bloom, C.I.; Chaussabel, D.; Banchereau, J.; Brett, S.J.; et al. Progression of Whole-Blood Transcriptional Signatures from Interferon-Induced to Neutrophil-Associated Patterns in Severe Influenza. Nat. Immunol. 2018, 19, 625–635. [Google Scholar] [CrossRef] [Green Version]
- GEO. Accession Viewer. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE68310 (accessed on 16 February 2021).
- Zhai, Y.; Franco, L.M.; Atmar, R.L.; Quarles, J.M.; Arden, N.; Bucasas, K.L.; Wells, J.M.; Niño, D.; Wang, X.; Zapata, G.E.; et al. Host Transcriptional Response to Influenza and Other Acute Respiratory Viral Infections—A Prospective Cohort Study. PLoS Pathog. 2015, 11, e1004869. [Google Scholar] [CrossRef] [Green Version]
- Shinde, A.; Luo, J.; Bharathi, S.S.; Shi, H.; Beck, M.E.; McHugh, K.J.; Alcorn, J.F.; Wang, J.; Goetzman, E.S. Increased Mortality from Influenza Infection in Long-Chain Acyl-CoA Dehydrogenase Knockout Mice. Biochem. Biophys. Res. Commun. 2018, 497, 700–704. [Google Scholar] [CrossRef]
- Davis, I.C. Cardiopulmonary Dysfunction And Lung Injury Are Attenuated In Influenza-Infected A1-Adenosine Receptor-Knockout Mice. In Dinfluences and Outcomes of Respiratory Viral Infections; American Thoracic Society International Conference Abstracts; American Thoracic Society: New York, NY, USA, 2012; p. A5714. [Google Scholar]
- Aeffner, F.; Woods, P.S.; Davis, I.C. Activation of A1-Adenosine Receptors Promotes Leukocyte Recruitment to the Lung and Attenuates Acute Lung Injury in Mice Infected with Influenza A/WSN/33 (H1N1) Virus. J. Virol. 2014, 88, 10214–10227. [Google Scholar] [CrossRef] [Green Version]
- Schloer, S.; Hübel, N.; Masemann, D.; Pajonczyk, D.; Brunotte, L.; Ehrhardt, C.; Brandenburg, L.-O.; Ludwig, S.; Gerke, V.; Rescher, U. The Annexin A1/FPR2 Signaling Axis Expands Alveolar Macrophages, Limits Viral Replication, and Attenuates Pathogenesis in the Murine Influenza A Virus Infection Model. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2019, 33, 12188–12199. [Google Scholar] [CrossRef] [Green Version]
- Herbert, J.; Hutchinson, W.L.; Carr, J.; Ives, J.; Jakob-Roetne, R.; Yamamura, K.-I.; Suzuki, M.; Pepys, M.B. Influenza Virus Infection Is Not Affected by Serum Amyloid P Component. Mol. Med. 2002, 8, 9–15. [Google Scholar] [CrossRef] [Green Version]
- Rodrigue-Gervais, I.G.; Labbé, K.; Dagenais, M.; Dupaul-Chicoine, J.; Champagne, C.; Morizot, A.; Skeldon, A.; Brincks, E.L.; Vidal, S.M.; Griffith, T.S.; et al. Cellular Inhibitor of Apoptosis Protein CIAP2 Protects against Pulmonary Tissue Necrosis during Influenza Virus Infection to Promote Host Survival. Cell Host Microbe 2014, 15, 23–35. [Google Scholar] [CrossRef] [Green Version]
- Sundar, I.K.; Ahmad, T.; Yao, H.; Hwang, J.; Gerloff, J.; Lawrence, B.P.; Sellix, M.T.; Rahman, I. Influenza A Virus-Dependent Remodeling of Pulmonary Clock Function in a Mouse Model of COPD. Sci. Rep. 2015, 4, 9927. [Google Scholar] [CrossRef] [Green Version]
- Schmid, E.T.; Pang, I.K.; Carrera Silva, E.A.; Bosurgi, L.; Miner, J.J.; Diamond, M.S.; Iwasaki, A.; Rothlin, C.V. AXL Receptor Tyrosine Kinase Is Required for T Cell Priming and Antiviral Immunity. eLife 2016, 5, e12414. [Google Scholar] [CrossRef] [PubMed]
- Tuzlak, S.; Schenk, R.L.; Vasanthakumar, A.; Preston, S.P.; Haschka, M.D.; Zotos, D.; Kallies, A.; Strasser, A.; Villunger, A.; Herold, M.J. The BCL-2 pro-Survival Protein A1 Is Dispensable for T Cell Homeostasis on Viral Infection. Cell Death Differ. 2017, 24, 523–533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rattan, A.; Pawar, S.D.; Nawadkar, R.; Kulkarni, N.; Lal, G.; Mullick, J.; Sahu, A. Synergy between the Classical and Alternative Pathways of Complement Is Essential for Conferring Effective Protection against the Pandemic Influenza A(H1N1) 2009 Virus Infection. PLoS Pathog. 2017, 13, e1006248. [Google Scholar] [CrossRef] [Green Version]
- Barlow, P.G.; Svoboda, P.; Mackellar, A.; Nash, A.A.; York, I.A.; Pohl, J.; Davidson, D.J.; Donis, R.O. Antiviral Activity and Increased Host Defense against Influenza Infection Elicited by the Human Cathelicidin LL-37. PLoS ONE 2011, 6, e25333. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.G.; Dash, P.; Aldridge, J.R.; Ellebedy, A.H.; Reynolds, C.; Funk, A.J.; Martin, W.J.; Lamkanfi, M.; Webby, R.J.; Boyd, K.L.; et al. The Intracellular Sensor NLRP3 Mediates Key Innate and Healing Responses to Influenza A Virus via the Regulation of Caspase-1. Immunity 2009, 30, 566–575. [Google Scholar] [CrossRef] [Green Version]
- Zheng, M.; Karki, R.; Vogel, P.; Kanneganti, T.-D. Caspase-6 Is a Key Regulator of Innate Immunity, Inflammasome Activation, and Host Defense. Cell 2020, 181, 674–687.e13. [Google Scholar] [CrossRef]
- Mbawuike, I.N.; Pacheco, S.; Acuna, C.L.; Switzer, K.C.; Zhang, Y.; Harriman, G.R. Mucosal Immunity to Influenza without IgA: An IgA Knockout Mouse Model. J. Immunol. 1999, 162, 2530–2537. [Google Scholar]
- Herold, S.; Steinmueller, M.; von Wulffen, W.; Cakarova, L.; Pinto, R.; Pleschka, S.; Mack, M.; Kuziel, W.A.; Corazza, N.; Brunner, T.; et al. Lung Epithelial Apoptosis in Influenza Virus Pneumonia: The Role of Macrophage-Expressed TNF-Related Apoptosis-Inducing Ligand. J. Exp. Med. 2008, 205, 3065–3077. [Google Scholar] [CrossRef]
- Dawson, T.C.; Beck, M.A.; Kuziel, W.A.; Henderson, F.; Maeda, N. Contrasting Effects of CCR5 and CCR2 Deficiency in the Pulmonary Inflammatory Response to Influenza A Virus. Am. J. Pathol. 2000, 156, 1951–1959. [Google Scholar] [CrossRef] [Green Version]
- Kuriakose, T.; Tripp, R.A.; Watford, W.T. Tumor Progression Locus 2 Promotes Induction of IFNλ, Interferon Stimulated Genes and Antigen-Specific CD8+ T Cell Responses and Protects against Influenza Virus. PLoS Pathog. 2015, 11, e1005038. [Google Scholar] [CrossRef]
- Gao, R.; Wang, L.; Bai, T.; Zhang, Y.; Bo, H.; Shu, Y. C-Reactive Protein Mediating Immunopathological Lesions: A Potential Treatment Option for Severe Influenza A Diseases. EBioMedicine 2017, 22, 133–142. [Google Scholar] [CrossRef] [Green Version]
- Schneider, C.; Nobs, S.P.; Heer, A.K.; Kurrer, M.; Klinke, G.; van Rooijen, N.; Vogel, J.; Kopf, M. Alveolar Macrophages Are Essential for Protection from Respiratory Failure and Associated Morbidity Following Influenza Virus Infection. PLoS Pathog. 2014, 10, e1004053. [Google Scholar] [CrossRef] [Green Version]
- Li, F.; Okreglicka, K.M.; Pohlmeier, L.M.; Schneider, C.; Kopf, M. Long-Term Culture of Fetal Monocyte Precursors In Vitro Allowing the Generation of Bona Fide Alveolar Macrophages In Vivo. biorXiv 2021. Available online: https://www.biorxiv.org/content/10.1101/2021.06.04.447115v2.full (accessed on 13 January 2022).
- Snelgrove, R.J.; Edwards, L.; Rae, A.J.; Hussell, T. An Absence of Reactive Oxygen Species Improves the Resolution of Lung Influenza Infection. Eur. J. Immunol. 2006, 36, 1364–1373. [Google Scholar] [CrossRef]
- Teijaro, J.R.; Walsh, K.B.; Cahalan, S.; Fremgen, D.M.; Roberts, E.; Scott, F.; Martinborough, E.; Peach, R.; Oldstone, M.B.A.; Rosen, H. Endothelial Cells Are Central Orchestrators of Cytokine Amplification during Influenza Virus Infection. Cell 2011, 146, 980–991. [Google Scholar] [CrossRef] [Green Version]
- Farley, K.; Stolley, J.M.; Zhao, P.; Cooley, J.; Remold-O’Donnell, E. A SerpinB1 Regulatory Mechanism Is Essential for Restricting Neutrophil Extracellular Trap Generation. J. Immunol. 2012, 189, 4574–4581. [Google Scholar] [CrossRef] [Green Version]
- Tan, S.-Y.; Chowdhury, S.; Polak, N.; Gorrell, M.D.; Weninger, W. Fibroblast Activation Protein Is Dispensable in the Anti-Influenza Immune Response in Mice. PLoS ONE 2017, 12, e0171194. [Google Scholar] [CrossRef] [Green Version]
- DiLillo, D.J.; Tan, G.S.; Palese, P.; Ravetch, J.V. Broadly Neutralizing Hemagglutinin Stalk-Specific Antibodies Require FcγR Interactions for Protection against Influenza Virus in Vivo. Nat. Med. 2014, 20, 143–151. [Google Scholar] [CrossRef]
- Wang, K.; Lai, C.; Li, T.; Wang, C.; Wang, W.; Ni, B.; Bai, C.; Zhang, S.; Han, L.; Gu, H.; et al. Basic Fibroblast Growth Factor Protects against Influenza A Virus-Induced Acute Lung Injury by Recruiting Neutrophils. J. Mol. Cell Biol. 2018, 10, 573–585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Masemann, D.; Leite Dantas, R.; Sitnik, S.; Schied, T.; Nordhoff, C.; Ludwig, S.; Wixler, V. The Four-and-a-Half LIM Domain Protein 2 Supports Influenza A Virus-Induced Lung Inflammation by Restricting the Host Adaptive Immune Response. Am. J. Pathol. 2018, 188, 1236–1245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Z.; Cao, M.; Guo, Y.; Zhao, L.; Wang, J.; Jia, X.; Li, J.; Wang, C.; Gabriel, G.; Xue, Q.; et al. Fragile X Mental Retardation Protein Stimulates Ribonucleoprotein Assembly of Influenza A Virus. Nat. Commun. 2014, 5, 3259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, C.; Jiao, S.; Wang, G.; Gao, Y.; Liu, C.; He, X.; Zhang, C.; Xiao, J.; Li, W.; Zhang, G.; et al. The Immune Adaptor ADAP Regulates Reciprocal TGF-Β1-Integrin Crosstalk to Protect from Influenza Virus Infection. PLoS Pathog. 2015, 11, e1004824. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, S.; Horie, M.; Daidoji, T.; Honda, T.; Yasugi, M.; Kuno, A.; Komori, T.; Okuzaki, D.; Narimatsu, H.; Nakaya, T.; et al. Influenza A Virus-Induced Expression of a GalNAc Transferase, GALNT3, via MicroRNAs Is Required for Enhanced Viral Replication. J. Virol. 2016, 90, 1788–1801. [Google Scholar] [CrossRef] [Green Version]
- Ichikawa, A.; Kuba, K.; Morita, M.; Chida, S.; Tezuka, H.; Hara, H.; Sasaki, T.; Ohteki, T.; Ranieri, V.M.; dos Santos, C.C.; et al. CXCL10-CXCR3 Enhances the Development of Neutrophil-Mediated Fulminant Lung Injury of Viral and Nonviral Origin. Am. J. Respir. Crit. Care Med. 2013, 187, 65–77. [Google Scholar] [CrossRef]
- Sakai, S.; Kawamata, H.; Mantani, N.; Kogure, T.; Shimada, Y.; Terasawa, K.; Sakai, T.; Imanishi, N.; Ochiai, H. Therapeutic Effect of Anti-Macrophage Inflammatory Protein 2 Antibody on Influenza Virus-Induced Pneumonia in Mice. J. Virol. 2000, 74, 2472–2476. [Google Scholar] [CrossRef] [Green Version]
- Gounder, A.P.; Yokoyama, C.C.; Jarjour, N.N.; Bricker, T.L.; Edelson, B.T.; Boon, A.C.M. Interferon Induced Protein 35 Exacerbates H5N1 Influenza Disease through the Expression of IL-12p40 Homodimer. PLoS Pathog. 2018, 14, e1007001. [Google Scholar] [CrossRef]
- Pinto, A.K.; Williams, G.D.; Szretter, K.J.; White, J.P.; Proença-Módena, J.L.; Liu, G.; Olejnik, J.; Brien, J.D.; Ebihara, H.; Mühlberger, E.; et al. Human and Murine IFIT1 Proteins Do Not Restrict Infection of Negative-Sense RNA Viruses of the Orthomyxoviridae, Bunyaviridae, and Filoviridae Families. J. Virol. 2015, 89, 9465–9476. [Google Scholar] [CrossRef] [Green Version]
- Szretter, K.J.; Gangappa, S.; Belser, J.A.; Zeng, H.; Chen, H.; Matsuoka, Y.; Sambhara, S.; Swayne, D.E.; Tumpey, T.M.; Katz, J.M. Early Control of H5N1 Influenza Virus Replication by the Type I Interferon Response in Mice. J. Virol. 2009, 83, 5825–5834. [Google Scholar] [CrossRef] [Green Version]
- Shepardson, K.M.; Larson, K.; Johns, L.L.; Stanek, K.; Cho, H.; Wellham, J.; Henderson, H.; Rynda-Apple, A. IFNAR2 Is Required for Anti-Influenza Immunity and Alters Susceptibility to Post-Influenza Bacterial Superinfections. Front. Immunol. 2018, 9, 2589. [Google Scholar] [CrossRef]
- Liu, B.; Bao, L.; Wang, L.; Li, F.; Wen, M.; Li, H.; Deng, W.; Zhang, X.; Cao, B. Anti-IFN-γ Therapy Alleviates Acute Lung Injury Induced by Severe Influenza A (H1N1) Pdm09 Infection in Mice. J. Microbiol. Immunol. Infect. 2021, 54, 396–403. [Google Scholar] [CrossRef]
- Weiss, I.D.; Wald, O.; Wald, H.; Beider, K.; Abraham, M.; Galun, E.; Nagler, A.; Peled, A. IFN-Gamma Treatment at Early Stages of Influenza Virus Infection Protects Mice from Death in a NK Cell-Dependent Manner. J. Interferon Cytokine Res. Off. J. Int. Soc. Interferon Cytokine Res. 2010, 30, 439–449. [Google Scholar] [CrossRef]
- Kopf, M.; Brombacher, F.; Bachmann, M.F. Role of IgM Antibodies versus B Cells in Influenza Virus-Specific Immunity. Eur. J. Immunol. 2002, 32, 2229–2236. [Google Scholar] [CrossRef]
- Schmitz, N.; Kurrer, M.; Bachmann, M.F.; Kopf, M. Interleukin-1 Is Responsible for Acute Lung Immunopathology but Increases Survival of Respiratory Influenza Virus Infection. J. Virol. 2005, 79, 6441–6448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lauder, S.N.; Jones, E.; Smart, K.; Bloom, A.; Williams, A.S.; Hindley, J.P.; Ondondo, B.; Taylor, P.R.; Clement, M.; Fielding, C.; et al. Interleukin-6 Limits Influenza-Induced Inflammation and Protects against Fatal Lung Pathology. Eur. J. Immunol. 2013, 43, 2613–2625. [Google Scholar] [CrossRef] [Green Version]
- Kang, M.C.; Choi, D.-H.; Choi, Y.W.; Park, S.J.; Namkoong, H.; Park, K.S.; Ahn, S.-S.; Surh, C.D.; Yoon, S.-W.; Kim, D.-J.; et al. Intranasal Introduction of Fc-Fused Interleukin-7 Provides Long-Lasting Prophylaxis against Lethal Influenza Virus Infection. J. Virol. 2015, 90, 2273–2284. [Google Scholar] [CrossRef] [Green Version]
- Sun, K.; Torres, L.; Metzger, D.W. A Detrimental Effect of Interleukin-10 on Protective Pulmonary Humoral Immunity during Primary Influenza A Virus Infection. J. Virol. 2010, 84, 5007–5014. [Google Scholar] [CrossRef] [Green Version]
- Monteiro, J.M.; Harvey, C.; Trinchieri, G. Role of Interleukin-12 in Primary Influenza Virus Infection. J. Virol. 1998, 72, 4825–4831. [Google Scholar] [CrossRef] [Green Version]
- Abdul-Careem, M.F.; Mian, M.F.; Yue, G.; Gillgrass, A.; Chenoweth, M.J.; Barra, N.G.; Chew, M.V.; Chan, T.; Al-Garawi, A.A.; Jordana, M.; et al. Critical Role of Natural Killer Cells in Lung Immunopathology during Influenza Infection in Mice. J. Infect. Dis. 2012, 206, 167–177. [Google Scholar] [CrossRef]
- Crowe, C.R.; Chen, K.; Pociask, D.A.; Alcorn, J.F.; Krivich, C.; Enelow, R.I.; Ross, T.M.; Witztum, J.L.; Kolls, J.K. Critical Role of IL-17RA in Immunopathology of Influenza Infection. J. Immunol. 2009, 183, 5301–5310. [Google Scholar] [CrossRef] [Green Version]
- Denton, A.E.; Doherty, P.C.; Turner, S.J.; La Gruta, N.L. IL-18, but Not IL-12, Is Required for Optimal Cytokine Production by Influenza Virus-Specific CD8+ T Cells. Eur. J. Immunol. 2007, 37, 368–375. [Google Scholar] [CrossRef]
- Hatesuer, B.; Hoang, H.T.T.; Riese, P.; Trittel, S.; Gerhauser, I.; Elbahesh, H.; Geffers, R.; Wilk, E.; Schughart, K. Deletion of Irf3 and Irf7 Genes in Mice Results in Altered Interferon Pathway Activation and Granulocyte-Dominated Inflammatory Responses to Influenza A Infection. J. Innate Immun. 2017, 9, 145–161. [Google Scholar] [CrossRef]
- Forbester, J.L.; Clement, M.; Wellington, D.; Yeung, A.; Dimonte, S.; Marsden, M.; Chapman, L.; Coomber, E.L.; Tolley, C.; Lees, E.; et al. IRF5 Promotes Influenza Virus-Induced Inflammatory Responses in Human Induced Pluripotent Stem Cell-Derived Myeloid Cells and Murine Models. J. Virol. 2020, 94, e00121-20. [Google Scholar] [CrossRef] [Green Version]
- Okazaki, T.; Higuchi, M.; Takeda, K.; Iwatsuki-Horimoto, K.; Kiso, M.; Miyagishi, M.; Yanai, H.; Kato, A.; Yoneyama, M.; Fujita, T.; et al. The ASK Family Kinases Differentially Mediate Induction of Type I Interferon and Apoptosis during the Antiviral Response. Sci. Signal. 2015, 8, ra78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Desmet, E.A.; Hollenbaugh, J.A.; Sime, P.J.; Wright, T.W.; Topham, D.J.; Sant, A.J.; Takimoto, T.; Dewhurst, S.; Maggirwar, S.B. Mixed Lineage Kinase 3 Deficiency Delays Viral Clearance in the Lung and Is Associated with Diminished Influenza-Induced Cytopathic Effect in Infected Cells. Virology 2010, 400, 224–232. [Google Scholar] [CrossRef] [Green Version]
- Snelgrove, R.J.; Goulding, J.; Didierlaurent, A.M.; Lyonga, D.; Vekaria, S.; Edwards, L.; Gwyer, E.; Sedgwick, J.D.; Barclay, A.N.; Hussell, T. A Critical Function for CD200 in Lung Immune Homeostasis and the Severity of Influenza Infection. Nat. Immunol. 2008, 9, 1074–1083. [Google Scholar] [CrossRef]
- Sugamata, R.; Dobashi, H.; Nagao, T.; Yamamoto, K.-I.; Nakajima, N.; Sato, Y.; Aratani, Y.; Oshima, M.; Sata, T.; Kobayashi, K.; et al. Contribution of Neutrophil-Derived Myeloperoxidase in the Early Phase of Fulminant Acute Respiratory Distress Syndrome Induced by Influenza Virus Infection. Microbiol. Immunol. 2012, 56, 171–182. [Google Scholar] [CrossRef]
- Pavlovic, J.; Arzet, H.A.; Hefti, H.P.; Frese, M.; Rost, D.; Ernst, B.; Kolb, E.; Staeheli, P.; Haller, O. Enhanced Virus Resistance of Transgenic Mice Expressing the Human MxA Protein. J. Virol. 1995, 69, 4506–4510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ito, T.; Allen, R.M.; Carson, W.F.; Schaller, M.; Cavassani, K.A.; Hogaboam, C.M.; Lukacs, N.W.; Matsukawa, A.; Kunkel, S.L. The Critical Role of Notch Ligand Delta-like 1 in the Pathogenesis of Influenza A Virus (H1N1) Infection. PLoS Pathog. 2011, 7, e1002341. [Google Scholar] [CrossRef] [Green Version]
- Yageta, Y.; Ishii, Y.; Morishima, Y.; Masuko, H.; Ano, S.; Yamadori, T.; Itoh, K.; Takeuchi, K.; Yamamoto, M.; Hizawa, N. Role of Nrf2 in Host Defense against Influenza Virus in Cigarette Smoke-Exposed Mice. J. Virol. 2011, 85, 4679–4690. [Google Scholar] [CrossRef] [Green Version]
- Karupiah, G.; Chen, J.H.; Mahalingam, S.; Nathan, C.F.; MacMicking, J.D. Rapid Interferon Gamma-Dependent Clearance of Influenza A Virus and Protection from Consolidating Pneumonitis in Nitric Oxide Synthase 2-Deficient Mice. J. Exp. Med. 1998, 188, 1541–1546. [Google Scholar] [CrossRef] [Green Version]
- Pérez de Castro, I.; Diaz, R.; Malumbres, M.; Hernández, M.-I.; Jagirdar, J.; Jiménez, M.; Ahn, D.; Pellicer, A. Mice Deficient for N-Ras: Impaired Antiviral Immune Response and T-Cell Function. Cancer Res. 2003, 63, 1615–1622. [Google Scholar] [PubMed]
- Leyva-Grado, V.H.; Ermler, M.E.; Schotsaert, M.; Gonzalez, M.G.; Gillespie, V.; Lim, J.K.; García-Sastre, A. Contribution of the Purinergic Receptor P2X7 to Development of Lung Immunopathology during Influenza Virus Infection. mBio 2017, 8, e00229-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dittmann, M.; Hoffmann, H.-H.; Scull, M.A.; Gilmore, R.H.; Bell, K.L.; Ciancanelli, M.; Wilson, S.J.; Crotta, S.; Yu, Y.; Flatley, B.; et al. A Serpin Shapes the Extracellular Environment to Prevent Influenza A Virus Maturation. Cell 2015, 160, 631–643. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Fu, Z.; Liang, H.; Wang, Y.; Qi, X.; Ding, M.; Sun, X.; Zhou, Z.; Huang, Y.; Gu, H.; et al. H5N1 Influenza Virus-Specific MiRNA-like Small RNA Increases Cytokine Production and Mouse Mortality via Targeting Poly(RC)-Binding Protein 2. Cell Res. 2018, 28, 157–171. [Google Scholar] [CrossRef] [Green Version]
- Guo, L.; Feng, K.; Wang, Y.C.; Mei, J.J.; Ning, R.T.; Zheng, H.W.; Wang, J.J.; Worthen, G.S.; Wang, X.; Song, J.; et al. Critical Role of CXCL4 in the Lung Pathogenesis of Influenza (H1N1) Respiratory Infection. Mucosal Immunol. 2017, 10, 1529–1541. [Google Scholar] [CrossRef]
- Nobs, S.P.; Schneider, C.; Heer, A.K.; Huotari, J.; Helenius, A.; Kopf, M. PI3Kγ Is Critical for Dendritic Cell-Mediated CD8+ T Cell Priming and Viral Clearance during Influenza Virus Infection. PLoS Pathog. 2016, 12, e1005508. [Google Scholar] [CrossRef] [Green Version]
- Berri, F.; Rimmelzwaan, G.F.; Hanss, M.; Albina, E.; Foucault-Grunenwald, M.-L.; Lê, V.B.; Vogelzang-van Trierum, S.E.; Gil, P.; Camerer, E.; Martinez, D.; et al. Plasminogen Controls Inflammation and Pathogenesis of Influenza Virus Infections via Fibrinolysis. PLoS Pathog. 2013, 9, e1003229. [Google Scholar] [CrossRef] [Green Version]
- Balachandran, S.; Roberts, P.C.; Brown, L.E.; Truong, H.; Pattnaik, A.K.; Archer, D.R.; Barber, G.N. Essential Role for the DsRNA-Dependent Protein Kinase PKR in Innate Immunity to Viral Infection. Immunity 2000, 13, 129–141. [Google Scholar] [CrossRef] [Green Version]
- Chida, J.; Hara, H.; Yano, M.; Uchiyama, K.; Das, N.R.; Takahashi, E.; Miyata, H.; Tomioka, Y.; Ito, T.; Kido, H.; et al. Prion Protein Protects Mice from Lethal Infection with Influenza A Viruses. PLoS Pathog. 2018, 14, e1007049. [Google Scholar] [CrossRef] [Green Version]
- Garcia, C.C.; Russo, R.C.; Guabiraba, R.; Fagundes, C.T.; Polidoro, R.B.; Tavares, L.P.; Salgado, A.P.C.; Cassali, G.D.; Sousa, L.P.; Machado, A.V.; et al. Platelet-Activating Factor Receptor Plays a Role in Lung Injury and Death Caused by Influenza A in Mice. PLoS Pathog. 2010, 6, e1001171. [Google Scholar] [CrossRef] [Green Version]
- Reading, P.C.; Bozza, S.; Gilbertson, B.; Tate, M.; Moretti, S.; Job, E.R.; Crouch, E.C.; Brooks, A.G.; Brown, L.E.; Bottazzi, B.; et al. Antiviral Activity of the Long Chain Pentraxin PTX3 against Influenza Viruses. J. Immunol. 2008, 180, 3391–3398. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.; Haist, V.; Baumgärtner, W.; Schughart, K. Sustained Viral Load and Late Death in Rag2−/− Mice after Influenza A Virus Infection. Virol. J. 2010, 7, 172. [Google Scholar] [CrossRef] [Green Version]
- Silverman, R.H. Viral Encounters with 2′,5′-Oligoadenylate Synthetase and RNase L during the Interferon Antiviral Response. J. Virol. 2007, 81, 12720–12729. [Google Scholar] [CrossRef] [Green Version]
- Lai, C.; Wang, K.; Zhao, Z.; Zhang, L.; Gu, H.; Yang, P.; Wang, X. C-C Motif Chemokine Ligand 2 (CCL2) Mediates Acute Lung Injury Induced by Lethal Influenza H7N9 Virus. Front. Microbiol. 2017, 8, 587. [Google Scholar] [CrossRef] [Green Version]
- LeVine, A.M.; Whitsett, J.A.; Hartshorn, K.L.; Crouch, E.C.; Korfhagen, T.R. Surfactant Protein D Enhances Clearance of Influenza A Virus from the Lung in Vivo. J. Immunol. 2001, 167, 5868–5873. [Google Scholar] [CrossRef]
- Glaser, L.; Conenello, G.; Paulson, J.; Palese, P. Effective Replication of Human Influenza Viruses in Mice Lacking a Major Alpha2,6 Sialyltransferase. Virus Res. 2007, 126, 9–18. [Google Scholar] [CrossRef]
- Oda, T.; Akaike, T.; Hamamoto, T.; Suzuki, F.; Hirano, T.; Maeda, H. Oxygen Radicals in Influenza-Induced Pathogenesis and Treatment with Pyran Polymer-Conjugated SOD. Science 1989, 244, 974–976. [Google Scholar] [CrossRef]
- Davidson, S.; Crotta, S.; McCabe, T.M.; Wack, A. Pathogenic Potential of Interferon Aβ in Acute Influenza Infection. Nat. Commun. 2014, 5, 3864. [Google Scholar] [CrossRef] [Green Version]
- Gopal, R.; Lee, B.; McHugh, K.J.; Rich, H.E.; Ramanan, K.; Mandalapu, S.; Clay, M.E.; Seger, P.J.; Enelow, R.I.; Manni, M.L.; et al. STAT2 Signaling Regulates Macrophage Phenotype During Influenza and Bacterial Super-Infection. Front. Immunol. 2018, 9, 2151. [Google Scholar] [CrossRef] [Green Version]
- Carlson, C.M.; Turpin, E.A.; Moser, L.A.; O’Brien, K.B.; Cline, T.D.; Jones, J.C.; Tumpey, T.M.; Katz, J.M.; Kelley, L.A.; Gauldie, J.; et al. Transforming Growth Factor-β: Activation by Neuraminidase and Role in Highly Pathogenic H5N1 Influenza Pathogenesis. PLoS Pathog. 2010, 6, e1001136. [Google Scholar] [CrossRef] [Green Version]
- Shirey, K.A.; Lai, W.; Patel, M.C.; Pletneva, L.M.; Pang, C.; Kurt-Jones, E.; Lipsky, M.; Roger, T.; Calandra, T.; Tracey, K.J.; et al. Novel Strategies for Targeting Innate Immune Responses to Influenza. Mucosal Immunol. 2016, 9, 1173–1182. [Google Scholar] [CrossRef] [Green Version]
- Shinya, K.; Okamura, T.; Sueta, S.; Kasai, N.; Tanaka, M.; Ginting, T.E.; Makino, A.; Eisfeld, A.J.; Kawaoka, Y. Toll-like Receptor Pre-Stimulation Protects Mice against Lethal Infection with Highly Pathogenic Influenza Viruses. Virol. J. 2011, 8, 97. [Google Scholar] [CrossRef] [Green Version]
- Le Goffic, R.; Balloy, V.; Lagranderie, M.; Alexopoulou, L.; Escriou, N.; Flavell, R.; Chignard, M.; Si-Tahar, M. Detrimental Contribution of the Toll-like Receptor (TLR)3 to Influenza A Virus-Induced Acute Pneumonia. PLoS Pathog. 2006, 2, e53. [Google Scholar] [CrossRef]
- Hatesuer, B.; Bertram, S.; Mehnert, N.; Bahgat, M.M.; Nelson, P.S.; Pöhlmann, S.; Pöhlman, S.; Schughart, K. Tmprss2 Is Essential for Influenza H1N1 Virus Pathogenesis in Mice. PLoS Pathog. 2013, 9, e1003774. [Google Scholar] [CrossRef] [Green Version]
- DeBerge, M.P.; Ely, K.H.; Enelow, R.I. Soluble, but Not Transmembrane, TNF-α Is Required during Influenza Infection to Limit the Magnitude of Immune Responses and the Extent of Immunopathology. J. Immunol. 2014, 192, 5839–5851. [Google Scholar] [CrossRef] [Green Version]
- Maelfait, J.; Roose, K.; Bogaert, P.; Sze, M.; Saelens, X.; Pasparakis, M.; Carpentier, I.; van Loo, G.; Beyaert, R. A20 (Tnfaip3) Deficiency in Myeloid Cells Protects against Influenza A Virus Infection. PLoS Pathog. 2012, 8, e1002570. [Google Scholar] [CrossRef]
- Yan, W.; Wei, J.; Deng, X.; Shi, Z.; Zhu, Z.; Shao, D.; Li, B.; Wang, S.; Tong, G.; Ma, Z. Transcriptional Analysis of Immune-Related Gene Expression in P53-Deficient Mice with Increased Susceptibility to Influenza A Virus Infection. BMC Med. Genom. 2015, 8, 52. [Google Scholar] [CrossRef] [Green Version]
- Lai, C.; Struckhoff, J.J.; Schneider, J.; Martinez-Sobrido, L.; Wolff, T.; García-Sastre, A.; Zhang, D.-E.; Lenschow, D.J. Mice Lacking the ISG15 E1 Enzyme UbE1L Demonstrate Increased Susceptibility to Both Mouse-Adapted and Non-Mouse-Adapted Influenza B Virus Infection. J. Virol. 2009, 83, 1147–1151. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, S.; Gregory, D.; Smith, A.; Kobzik, L. MARCO Regulates Early Inflammatory Responses against Influenza: A Useful Macrophage Function with Adverse Outcome. Am. J. Respir. Cell Mol. Biol. 2011, 45, 1036–1044. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brincks, E.L.; Katewa, A.; Kucaba, T.A.; Griffith, T.S.; Legge, K.L. CD8 T Cells Utilize TRAIL to Control Influenza Virus Infection. J. Immunol. 2008, 181, 4918–4925. [Google Scholar] [CrossRef]
- Högner, K.; Wolff, T.; Pleschka, S.; Plog, S.; Gruber, A.D.; Kalinke, U.; Walmrath, H.-D.; Bodner, J.; Gattenlöhner, S.; Lewe-Schlosser, P.; et al. Correction: Macrophage-Expressed IFN-β Contributes to Apoptotic Alveolar Epithelial Cell Injury in Severe Influenza Virus Pneumonia. PLoS Pathog. 2016, 12, e1005716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lupfer, C.; Thomas, P.G.; Anand, P.K.; Vogel, P.; Milasta, S.; Martinez, J.; Huang, G.; Green, M.; Kundu, M.; Chi, H.; et al. Receptor Interacting Protein Kinase 2-Mediated Mitophagy Regulates Inflammasome Activation during Virus Infection. Nat. Immunol. 2013, 14, 480–488. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Snell, L.M.; McPherson, A.J.; Lin, G.H.Y.; Sakaguchi, S.; Pandolfi, P.P.; Riccardi, C.; Watts, T.H. CD8 T Cell-Intrinsic GITR Is Required for T Cell Clonal Expansion and Mouse Survival Following Severe Influenza Infection. J. Immunol. 2010, 185, 7223–7234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gazit, R.; Gruda, R.; Elboim, M.; Arnon, T.I.; Katz, G.; Achdout, H.; Hanna, J.; Qimron, U.; Landau, G.; Greenbaum, E.; et al. Lethal Influenza Infection in the Absence of the Natural Killer Cell Receptor Gene Ncr1. Nat. Immunol. 2006, 7, 517–523. [Google Scholar] [CrossRef]
- Zhang, H.; Luo, J.; Alcorn, J.F.; Chen, K.; Fan, S.; Pilewski, J.; Liu, A.; Chen, W.; Kolls, J.K.; Wang, J. AIM2 Inflammasome Is Critical for Influenza-Induced Lung Injury and Mortality. J. Immunol. 2017, 198, 4383–4393. [Google Scholar] [CrossRef] [Green Version]
- Schattgen, S.A.; Gao, G.; Kurt-Jones, E.A.; Fitzgerald, K.A. Cutting Edge: DNA in the Lung Microenvironment during Influenza Virus Infection Tempers Inflammation by Engaging the DNA Sensor AIM. J. Immunol. 2016, 196, 29–33. [Google Scholar] [CrossRef] [Green Version]
- Hartweger, H.; Schweighoffer, E.; Davidson, S.; Peirce, M.J.; Wack, A.; Tybulewicz, V.L.J. Themis2 Is Not Required for B Cell Development, Activation, and Antibody Responses. J. Immunol. 2014, 193, 700–707. [Google Scholar] [CrossRef] [Green Version]
- Coulombe, F.; Jaworska, J.; Verway, M.; Tzelepis, F.; Massoud, A.; Gillard, J.; Wong, G.; Kobinger, G.; Xing, Z.; Couture, C.; et al. Targeted Prostaglandin E2 Inhibition Enhances Antiviral Immunity through Induction of Type I Interferon and Apoptosis in Macrophages. Immunity 2014, 40, 554–568. [Google Scholar] [CrossRef] [Green Version]
- Sidahmed, A.M.; Leon, A.J.; Banner, D.; Kelvin, A.A.; Rowe, T.; Boudakov, I.; Degousse, N.; Rubin, B.B.; Kelvin, D.J. CXCL14 Deficiency Does Not Impact the Outcome of Influenza or Escherichia Coli Infections in Mice. J. Infect. Dev. Ctries. 2014, 8, 1301–1306. [Google Scholar] [CrossRef] [Green Version]
- Kedzierski, L.; Tate, M.D.; Hsu, A.C.; Kolesnik, T.B.; Linossi, E.M.; Dagley, L.; Dong, Z.; Freeman, S.; Infusini, G.; Starkey, M.R.; et al. Suppressor of Cytokine Signaling (SOCS)5 Ameliorates Influenza Infection via Inhibition of EGFR Signaling. eLife 2017, 6, e20444. [Google Scholar] [CrossRef]
- Peng, N.; Liu, S.; Xia, Z.; Ren, S.; Feng, J.; Jing, M.; Gao, X.; Wiemer, E.A.C.; Zhu, Y. Inducible Major Vault Protein Plays a Pivotal Role in Double-Stranded RNA- or Virus-Induced Proinflammatory Response. J. Immunol. 2016, 196, 2753–2766. [Google Scholar] [CrossRef] [Green Version]
- Fu, B.; Wang, L.; Li, S.; Dorf, M.E. ZMPSTE24 Defends against Influenza and Other Pathogenic Viruses. J. Exp. Med. 2017, 214, 919–929. [Google Scholar] [CrossRef]
- Kenney, A.D.; McMichael, T.M.; Imas, A.; Chesarino, N.M.; Zhang, L.; Dorn, L.E.; Wu, Q.; Alfaour, O.; Amari, F.; Chen, M.; et al. IFITM3 Protects the Heart during Influenza Virus Infection. Proc. Natl. Acad. Sci. USA 2019, 116, 18607–18612. [Google Scholar] [CrossRef] [Green Version]
- Jia, W.; Li, H.; He, Y.-W. Pattern Recognition Molecule Mindin Promotes Intranasal Clearance of Influenza Viruses. J. Immunol. 2008, 180, 6255–6261. [Google Scholar] [CrossRef] [Green Version]
- Lu, Q.; Yokoyama, C.C.; Williams, J.W.; Baldridge, M.T.; Jin, X.; DesRochers, B.; Bricker, T.; Wilen, C.B.; Bagaitkar, J.; Loginicheva, E.; et al. Homeostatic Control of Innate Lung Inflammation by Vici Syndrome Gene Epg5 and Additional Autophagy Genes Promotes Influenza Pathogenesis. Cell Host Microbe 2016, 19, 102–113. [Google Scholar] [CrossRef] [Green Version]
- Downey, J.; Pernet, E.; Coulombe, F.; Allard, B.; Meunier, I.; Jaworska, J.; Qureshi, S.; Vinh, D.C.; Martin, J.G.; Joubert, P.; et al. RIPK3 Interacts with MAVS to Regulate Type I IFN-Mediated Immunity to Influenza A Virus Infection. PLoS Pathog. 2017, 13, e1006326. [Google Scholar] [CrossRef]
- McMahon, M.; Ye, S.; Izzard, L.; Dlugolenski, D.; Tripp, R.A.; Bean, A.G.D.; McCulloch, D.R.; Stambas, J. Correction: ADAMTS5 Is a Critical Regulator of Virus-Specific T Cell Immunity. PLoS Biol. 2019, 17, e3000558. [Google Scholar] [CrossRef]
- Seki, M.; Kohno, S.; Newstead, M.W.; Zeng, X.; Bhan, U.; Lukacs, N.W.; Kunkel, S.L.; Standiford, T.J. Critical Role of IL-1 Receptor-Associated Kinase-M in Regulating Chemokine-Dependent Deleterious Inflammation in Murine Influenza Pneumonia. J. Immunol. 2010, 184, 1410–1418. [Google Scholar] [CrossRef] [Green Version]
- Hemmers, S.; Teijaro, J.R.; Arandjelovic, S.; Mowen, K.A. PAD4-Mediated Neutrophil Extracellular Trap Formation Is Not Required for Immunity against Influenza Infection. PLoS ONE 2011, 6, e22043. [Google Scholar] [CrossRef]
- Kandasamy, M.; Suryawanshi, A.; Tundup, S.; Perez, J.T.; Schmolke, M.; Manicassamy, S.; Manicassamy, B. RIG-I Signaling Is Critical for Efficient Polyfunctional T Cell Responses during Influenza Virus Infection. PLoS Pathog. 2016, 12, e1005754. [Google Scholar] [CrossRef]
- Teng, O.; Chen, S.-T.; Hsu, T.-L.; Sia, S.F.; Cole, S.; Valkenburg, S.A.; Hsu, T.-Y.; Zheng, J.T.; Tu, W.; Bruzzone, R.; et al. CLEC5A-Mediated Enhancement of the Inflammatory Response in Myeloid Cells Contributes to Influenza Virus Pathogenicity In Vivo. J. Virol. 2017, 91, e01813-16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Q.; Lin, L.; Tong, Y.; Liu, Y.; Mou, J.; Wang, X.; Wang, X.; Gong, Y.; Zhao, Y.; Liu, Y.; et al. TRIM29 Negatively Controls Antiviral Immune Response through Targeting STING for Degradation. Cell Discov. 2018, 4, 13. [Google Scholar] [CrossRef] [Green Version]
- Allen, I.C.; Scull, M.A.; Moore, C.B.; Holl, E.K.; McElvania-TeKippe, E.; Taxman, D.J.; Guthrie, E.H.; Pickles, R.J.; Ting, J.P.-Y. The NLRP3 Inflammasome Mediates in Vivo Innate Immunity to Influenza A Virus through Recognition of Viral RNA. Immunity 2009, 30, 556–565. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, B.J.; Cho, S.W.; Jeon, Y.J.; An, S.; Jo, A.; Lim, J.H.; Kim, D.-Y.; Won, T.-B.; Han, D.H.; Rhee, C.-S.; et al. Intranasal Delivery of Duox2 DNA Using Cationic Polymer Can Prevent Acute Influenza A Viral Infection in Vivo Lung. Appl. Microbiol. Biotechnol. 2018, 102, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Moser, E.K.; Sun, J.; Kim, T.S.; Braciale, T.J. IL-21R Signaling Suppresses IL-17+ Gamma Delta T Cell Responses and Production of IL-17 Related Cytokines in the Lung at Steady State and after Influenza A Virus Infection. PLoS ONE 2015, 10, e0120169. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Sharma, P.; Jefferson, M.; Zhang, W.; Bone, B.; Kipar, A.; Bitto, D.; Coombes, J.L.; Pearson, T.; Man, A.; et al. Non-Canonical Autophagy Functions of ATG16L1 in Epithelial Cells Limit Lethal Infection by Influenza A Virus. EMBO J. 2021, 40, e105543. [Google Scholar] [CrossRef] [PubMed]
- Wein, A.N.; Dunbar, P.R.; McMaster, S.R.; Li, Z.-R.T.; Denning, T.L.; Kohlmeier, J.E. IL-36γ Protects against Severe Influenza Infection by Promoting Lung Alveolar Macrophage Survival and Limiting Viral Replication. J. Immunol. 2018, 201, 573–582. [Google Scholar] [CrossRef] [Green Version]
- Zou, Z.; Yan, Y.; Shu, Y.; Gao, R.; Sun, Y.; Li, X.; Ju, X.; Liang, Z.; Liu, Q.; Zhao, Y.; et al. Angiotensin-Converting Enzyme 2 Protects from Lethal Avian Influenza A H5N1 Infections. Nat. Commun. 2014, 5, 3594. [Google Scholar] [CrossRef]
- Le Bel, M.; Gosselin, J. Leukotriene B4 Enhances NOD2-Dependent Innate Response against Influenza Virus Infection. PLoS ONE 2015, 10, e0139856. [Google Scholar] [CrossRef] [Green Version]
- Uematsu, T.; Iizasa, E.; Kobayashi, N.; Yoshida, H.; Hara, H. Loss of CARD9-Mediated Innate Activation Attenuates Severe Influenza Pneumonia without Compromising Host Viral Immunity. Sci. Rep. 2015, 5, 17577. [Google Scholar] [CrossRef] [Green Version]
- Allen, I.C.; Moore, C.B.; Schneider, M.; Lei, Y.; Davis, B.K.; Scull, M.A.; Gris, D.; Roney, K.E.; Zimmermann, A.G.; Bowzard, J.B.; et al. NLRX1 Protein Attenuates Inflammatory Responses to Infection by Interfering with the RIG-I-MAVS and TRAF6-NF-ΚB Signaling Pathways. Immunity 2011, 34, 854–865. [Google Scholar] [CrossRef] [Green Version]
- Thapa, R.J.; Ingram, J.P.; Ragan, K.B.; Nogusa, S.; Boyd, D.F.; Benitez, A.A.; Sridharan, H.; Kosoff, R.; Shubina, M.; Landsteiner, V.J.; et al. DAI Senses Influenza A Virus Genomic RNA and Activates RIPK3-Dependent Cell Death. Cell Host Microbe 2016, 20, 674–681. [Google Scholar] [CrossRef] [Green Version]
- Lafferty, E.I.; Flaczyk, A.; Angers, I.; Homer, R.; d’Hennezel, E.; Malo, D.; Piccirillo, C.A.; Vidal, S.M.; Qureshi, S.T. An ENU-Induced Splicing Mutation Reveals a Role for Unc93b1 in Early Immune Cell Activation Following Influenza A H1N1 Infection. Genes Immun. 2014, 15, 320–332. [Google Scholar] [CrossRef]
- Seo, G.J.; Kim, C.; Shin, W.-J.; Sklan, E.H.; Eoh, H.; Jung, J.U. TRIM56-Mediated Monoubiquitination of CGAS for Cytosolic DNA Sensing. Nat. Commun. 2018, 9, 613. [Google Scholar] [CrossRef] [Green Version]
- Lupfer, C.R.; Stokes, K.L.; Kuriakose, T.; Kanneganti, T.-D. Deficiency of the NOD-Like Receptor NLRC5 Results in Decreased CD8+ T Cell Function and Impaired Viral Clearance. J. Virol. 2017, 91, e00377-17. [Google Scholar] [CrossRef] [Green Version]
- Tan, K.S.; Olfat, F.; Phoon, M.C.; Hsu, J.P.; Howe, J.L.C.; Seet, J.E.; Chin, K.C.; Chow, V.T.K. In Vivo and in Vitro Studies on the Antiviral Activities of Viperin against Influenza H1N1 Virus Infection. J. Gen. Virol. 2012, 93, 1269–1277. [Google Scholar] [CrossRef]
- Khoufache, K.; Berri, F.; Nacken, W.; Vogel, A.B.; Delenne, M.; Camerer, E.; Coughlin, S.R.; Carmeliet, P.; Lina, B.; Rimmelzwaan, G.F.; et al. PAR1 Contributes to Influenza A Virus Pathogenicity in Mice. J. Clin. Investig. 2013, 123, 206–214. [Google Scholar] [CrossRef]
- Major, J.; Crotta, S.; Llorian, M.; McCabe, T.M.; Gad, H.H.; Priestnall, S.L.; Hartmann, R.; Wack, A. Type I and III Interferons Disrupt Lung Epithelial Repair during Recovery from Viral Infection. Science 2020, 369, 712–717. [Google Scholar] [CrossRef]
- Oltean, T.; Van San, E.; Divert, T.; Vanden Berghe, T.; Saelens, X.; Maelfait, J.; Takahashi, N.; Vandenabeele, P. Viral Dosing of Influenza A Infection Reveals Involvement of RIPK3 and FADD, but Not MLKL. Cell Death Dis. 2021, 12, 471. [Google Scholar] [CrossRef]
- Ito, T.; Itakura, J.; Takahashi, S.; Sato, M.; Mino, M.; Fushimi, S.; Yamada, M.; Morishima, T.; Kunkel, S.L.; Matsukawa, A. Sprouty-Related Ena/Vasodilator-Stimulated Phosphoprotein Homology 1-Domain-Containing Protein-2 Critically Regulates Influenza A Virus-Induced Pneumonia. Crit. Care Med. 2016, 44, e530–e543. [Google Scholar] [CrossRef] [Green Version]
- Ramirez-Ortiz, Z.G.; Prasad, A.; Griffith, J.W.; Pendergraft, W.F.; Cowley, G.S.; Root, D.E.; Tai, M.; Luster, A.D.; El Khoury, J.; Hacohen, N.; et al. The Receptor TREML4 Amplifies TLR7-Mediated Signaling during Antiviral Responses and Autoimmunity. Nat. Immunol. 2015, 16, 495–504. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- LeVine, A.M.; Hartshorn, K.; Elliott, J.; Whitsett, J.; Korfhagen, T. Absence of SP-A Modulates Innate and Adaptive Defense Responses to Pulmonary Influenza Infection. Am. J. Physiol. Lung Cell. Mol. Physiol. 2002, 282, L563–L572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nair, S.; Huynh, J.P.; Lampropoulou, V.; Loginicheva, E.; Esaulova, E.; Gounder, A.P.; Boon, A.C.M.; Schwarzkopf, E.A.; Bradstreet, T.R.; Edelson, B.T.; et al. Irg1 Expression in Myeloid Cells Prevents Immunopathology during M. Tuberculosis Infection. J. Exp. Med. 2018, 215, 1035–1045. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hao, L.; Sakurai, A.; Watanabe, T.; Sorensen, E.; Nidom, C.A.; Newton, M.A.; Ahlquist, P.; Kawaoka, Y. Drosophila RNAi Screen Identifies Host Genes Important for Influenza Virus Replication. Nature 2008, 454, 890–893. [Google Scholar] [CrossRef] [Green Version]
- Shapira, S.D.; Gat-Viks, I.; Shum, B.O.V.; Dricot, A.; de Grace, M.M.; Wu, L.; Gupta, P.B.; Hao, T.; Silver, S.J.; Root, D.E.; et al. A Physical and Regulatory Map of Host-Influenza Interactions Reveals Pathways in H1N1 Infection. Cell 2009, 139, 1255–1267. [Google Scholar] [CrossRef] [Green Version]
- Brass, A.L.; Huang, I.-C.; Benita, Y.; John, S.P.; Krishnan, M.N.; Feeley, E.M.; Ryan, B.J.; Weyer, J.L.; van der Weyden, L.; Fikrig, E.; et al. The IFITM Proteins Mediate Cellular Resistance to Influenza A H1N1 Virus, West Nile Virus, and Dengue Virus. Cell 2009, 139, 1243–1254. [Google Scholar] [CrossRef] [Green Version]
- Ward, S.E.; Kim, H.S.; Komurov, K.; Mendiratta, S.; Tsai, P.-L.; Schmolke, M.; Satterly, N.; Manicassamy, B.; Forst, C.V.; Roth, M.G.; et al. Host Modulators of H1N1 Cytopathogenicity. PLoS ONE 2012, 7, e39284. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, T.; Kawakami, E.; Shoemaker, J.E.; Lopes, T.J.S.; Matsuoka, Y.; Tomita, Y.; Kozuka-Hata, H.; Gorai, T.; Kuwahara, T.; Takeda, E.; et al. Influenza Virus-Host Interactome Screen as a Platform for Antiviral Drug Development. Cell Host Microbe 2014, 16, 795–805. [Google Scholar] [CrossRef] [Green Version]
- Estrin, M.A.; Hussein, I.T.M.; Puryear, W.B.; Kuan, A.C.; Artim, S.C.; Runstadler, J.A. Host-Directed Combinatorial RNAi Improves Inhibition of Diverse Strains of Influenza A Virus in Human Respiratory Epithelial Cells. PLoS ONE 2018, 13, e0197246. [Google Scholar] [CrossRef]
- Heaton, B.E.; Kennedy, E.M.; Dumm, R.E.; Harding, A.T.; Sacco, M.T.; Sachs, D.; Heaton, N.S. A CRISPR Activation Screen Identifies a Pan-Avian Influenza Virus Inhibitory Host Factor. Cell Rep. 2017, 20, 1503–1512. [Google Scholar] [CrossRef] [Green Version]
- Han, J.; Perez, J.T.; Chen, C.; Li, Y.; Benitez, A.; Kandasamy, M.; Lee, Y.; Andrade, J.; tenOever, B.; Manicassamy, B. Genome-Wide CRISPR/Cas9 Screen Identifies Host Factors Essential for Influenza Virus Replication. Cell Rep. 2018, 23, 596–607. [Google Scholar] [CrossRef] [Green Version]
- Li, B.; Clohisey, S.M.; Chia, B.S.; Wang, B.; Cui, A.; Eisenhaure, T.; Schweitzer, L.D.; Hoover, P.; Parkinson, N.J.; Nachshon, A.; et al. Genome-Wide CRISPR Screen Identifies Host Dependency Factors for Influenza A Virus Infection. Nat. Commun. 2020, 11, 164. [Google Scholar] [CrossRef] [Green Version]
- Sui, B.; Bamba, D.; Weng, K.; Ung, H.; Chang, S.; Van Dyke, J.; Goldblatt, M.; Duan, R.; Kinch, M.S.; Li, W.-B. The Use of Random Homozygous Gene Perturbation to Identify Novel Host-Oriented Targets for Influenza. Virology 2009, 387, 473–481. [Google Scholar] [CrossRef] [Green Version]
- Karlas, A.; Machuy, N.; Shin, Y.; Pleissner, K.-P.; Artarini, A.; Heuer, D.; Becker, D.; Khalil, H.; Ogilvie, L.A.; Hess, S.; et al. Genome-Wide RNAi Screen Identifies Human Host Factors Crucial for Influenza Virus Replication. Nature 2010, 463, 818–822. [Google Scholar] [CrossRef]
- König, R.; Stertz, S.; Zhou, Y.; Inoue, A.; Hoffmann, H.-H.; Bhattacharyya, S.; Alamares, J.G.; Tscherne, D.M.; Ortigoza, M.B.; Liang, Y.; et al. Human Host Factors Required for Influenza Virus Replication. Nature 2010, 463, 813–817. [Google Scholar] [CrossRef]
- Pizzorno, A.; Terrier, O.; Nicolas de Lamballerie, C.; Julien, T.; Padey, B.; Traversier, A.; Roche, M.; Hamelin, M.-E.; Rhéaume, C.; Croze, S.; et al. Repurposing of Drugs as Novel Influenza Inhibitors From Clinical Gene Expression Infection Signatures. Front. Immunol. 2019, 10, 60. [Google Scholar] [CrossRef] [Green Version]
- Quispe-Laime, A.M.; Bracco, J.D.; Barberio, P.A.; Campagne, C.G.; Rolfo, V.E.; Umberger, R.; Meduri, G.U. H1N1 Influenza A Virus-Associated Acute Lung Injury: Response to Combination Oseltamivir and Prolonged Corticosteroid Treatment. Intensive Care Med. 2010, 36, 33–41. [Google Scholar] [CrossRef]
- Kim, H.; Lee, M.-K.; Ko, J.; Park, C.-J.; Kim, M.; Jeong, Y.; Hong, S.; Varani, G.; Choi, B.-S. Aminoglycoside Antibiotics Bind to the Influenza A Virus RNA Promoter. Mol. Biosyst. 2012, 8, 2857–2859. [Google Scholar] [CrossRef]
- Long, J.; Wright, E.; Molesti, E.; Temperton, N.; Barclay, W. Antiviral Therapies against Ebola and Other Emerging Viral Diseases Using Existing Medicines That Block Virus Entry. F1000Research 2015, 4, 30. [Google Scholar] [CrossRef] [Green Version]
- Martin, P.B. A Trial of Sulphamethoxypyridazine (S.M.P.) in the Management of Influenza. J. Coll. Gen. Pract. 1961, 4, 426–430. [Google Scholar]
- Enkirch, T.; Sauber, S.; Anderson, D.E.; Gan, E.S.; Kenanov, D.; Maurer-Stroh, S.; von Messling, V. Identification and in Vivo Efficacy Assessment of Approved Orally Bioavailable Human Host Protein-Targeting Drugs With Broad Anti-Influenza A Activity. Front. Immunol. 2019, 10, 1097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, X.; An, L. Application of Liothyronine in Preparation of Medicine for Treatment or Prevention of Influenza Virus Infection. Chinese Patent CN103705497B, 1 April 2015. [Google Scholar]
- Chen, X.; Liao, Q.; An, L. Application of Doxylamine Succinate in Preparing Drug for Treating or Preventing Influenza Virus. Chinese Patent CN103251590B, 17 December 2014. [Google Scholar]
- Love, M.I.; Huber, W.; Anders, S. Moderated Estimation of Fold Change and Dispersion for RNA-Seq Data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tripathi, S.; Tecle, T.; Verma, A.; Crouch, E.; White, M.; Hartshorn, K.L. The Human Cathelicidin LL-37 Inhibits Influenza A Viruses through a Mechanism Distinct from That of Surfactant Protein D or Defensins. J. Gen. Virol. 2013, 94, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Xue, Q.; Wan, Y.; Yang, Y.; Wang, J.; Hung, T. Lysosome-Associated Membrane Glycoprotein 3 Is Involved in Influenza A Virus Replication in Human Lung Epithelial (A549) Cells. Virol. J. 2011, 8, 384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanyal, S.; Ashour, J.; Maruyama, T.; Altenburg, A.F.; Cragnolini, J.J.; Bilate, A.; Avalos, A.M.; Kundrat, L.; García-Sastre, A.; Ploegh, H.L. Type-I Interferon Imposes a TSG101/ISG15 Checkpoint at the Golgi for Glycoprotein Trafficking during Influenza Virus Infection. Cell Host Microbe 2013, 14, 510–521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diebold, S.S.; Kaisho, T.; Hemmi, H.; Akira, S.; Reis e Sousa, C. Innate Antiviral Responses by Means of TLR7-Mediated Recognition of Single-Stranded RNA. Science 2004, 303, 1529–1531. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Hernandez, L.D.; Galán, J.E.; Janeway, C.A.; Medzhitov, R.; Flavell, R.A. IRAK-M Is a Negative Regulator of Toll-like Receptor Signaling. Cell 2002, 110, 191–202. [Google Scholar] [CrossRef] [Green Version]
- Ehrhardt, C.; Marjuki, H.; Wolff, T.; Nürnberg, B.; Planz, O.; Pleschka, S.; Ludwig, S. Bivalent Role of the Phosphatidylinositol-3-Kinase (PI3K) during Influenza Virus Infection and Host Cell Defence. Cell. Microbiol. 2006, 8, 1336–1348. [Google Scholar] [CrossRef]
- Garcia, C.C.; Weston-Davies, W.; Russo, R.C.; Tavares, L.P.; Rachid, M.A.; Alves-Filho, J.C.; Machado, A.V.; Ryffel, B.; Nunn, M.A.; Teixeira, M.M. Complement C5 Activation during Influenza A Infection in Mice Contributes to Neutrophil Recruitment and Lung Injury. PLoS ONE 2013, 8, e0064443. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.G.; Hartl, D.; Lee, G.R.; Koller, B.; Matsuura, H.; Da Silva, C.A.; Sohn, M.H.; Cohn, L.; Homer, R.J.; Kozhich, A.A.; et al. Role of Breast Regression Protein 39 (BRP-39)/Chitinase 3-like-1 in Th2 and IL-13–Induced Tissue Responses and Apoptosis. J. Exp. Med. 2009, 206, 1149–1166. [Google Scholar] [CrossRef] [Green Version]
- Han, X.; Liu, H.; Huang, H.; Liu, X.; Jia, B.; Gao, G.F.; Zhang, F. ID2 and ID3 Are Indispensable for Th1 Cell Differentiation during Influenza Virus Infection in Mice. Eur. J. Immunol. 2019, 49, 476–489. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Quintero, J.; Wang, X.; Tipper, J.; Burkett, P.R.; Zuñiga, J.; Ashtekar, A.R.; Polverino, F.; Rout, A.; Yambayev, I.; Hernández, C.; et al. Matrix Metalloproteinase-9 Deficiency Protects Mice from Severe Influenza A Viral Infection. JCI Insight 2018, 3, 99022. [Google Scholar] [CrossRef]
- Chen, X.; Liu, S.; Goraya, M.U.; Maarouf, M.; Huang, S.; Chen, J.-L. Host Immune Response to Influenza A Virus Infection. Front. Immunol. 2018, 9, 320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rehwinkel, J.; Gack, M.U. RIG-I-like Receptors: Their Regulation and Roles in RNA Sensing. Nat. Rev. Immunol. 2020, 20, 537–551. [Google Scholar] [CrossRef]
- Buss, C.; Opitz, B.; Hocke, A.C.; Lippmann, J.; van Laak, V.; Hippenstiel, S.; Krüll, M.; Suttorp, N.; Eitel, J. Essential Role of Mitochondrial Antiviral Signaling, IFN Regulatory Factor (IRF)3, and IRF7 in Chlamydophila Pneumoniae-Mediated IFN-Beta Response and Control of Bacterial Replication in Human Endothelial Cells. J. Immunol. 2010, 184, 3072–3078. [Google Scholar] [CrossRef] [Green Version]
- Ning, S.; Pagano, J.S.; Barber, G.N. IRF7: Activation, Regulation, Modification and Function. Genes Immun. 2011, 12, 399–414. [Google Scholar] [CrossRef] [Green Version]
- Hrincius, E.R.; Dierkes, R.; Anhlan, D.; Wixler, V.; Ludwig, S.; Ehrhardt, C. Phosphatidylinositol-3-Kinase (PI3K) Is Activated by Influenza Virus VRNA via the Pathogen Pattern Receptor Rig-I to Promote Efficient Type I Interferon Production. Cell. Microbiol. 2011, 13, 1907–1919. [Google Scholar] [CrossRef]
- Zhang, M.; Chen, W.; Zhou, W.; Bai, Y.; Gao, J. Critical Role of IRAK-M in Regulating Antigen-Induced Airway Inflammation. Am. J. Respir. Cell Mol. Biol. 2017, 57, 547–559. [Google Scholar] [CrossRef]
- Song, N.; Li, P.; Jiang, Y.; Sun, H.; Cui, J.; Zhao, G.; Li, D.; Guo, Y.; Chen, Y.; Gao, J.; et al. C5a Receptor1 Inhibition Alleviates Influenza Virus-Induced Acute Lung Injury. Int. Immunopharmacol. 2018, 59, 12–20. [Google Scholar] [CrossRef]
- Lamichhane, P.P.; Samarasinghe, A.E. The Role of Innate Leukocytes during Influenza Virus Infection. J. Immunol. Res. 2019, 2019, 8028725. [Google Scholar] [CrossRef] [Green Version]
- Ma, Y.; Zhang, Y.; Zhu, L. Role of Neutrophils in Acute Viral Infection. Immun. Inflamm. Dis. 2021, 9, 1186–1196. [Google Scholar] [CrossRef] [PubMed]
- Camp, J.V.; Jonsson, C.B. A Role for Neutrophils in Viral Respiratory Disease. Front. Immunol. 2017, 8, 550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamamoto, K.; Miyoshi-Koshio, T.; Utsuki, Y.; Mizuno, S.; Suzuki, K. Virucidal Activity and Viral Protein Modification by Myeloperoxidase: A Candidate for Defense Factor of Human Polymorphonuclear Leukocytes against Influenza Virus Infection. J. Infect. Dis. 1991, 164, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Sun, X.; Goie, J.Y.G.; Zhang, Y. Regulation of Host Immune Responses against Influenza A Virus Infection by Mitogen-Activated Protein Kinases (MAPKs). Microorganisms 2020, 8, 1067. [Google Scholar] [CrossRef]
- Dai, J.-P.; Wang, Q.-W.; Su, Y.; Gu, L.-M.; Deng, H.-X.; Chen, X.-X.; Li, W.-Z.; Li, K.-S. Oxymatrine Inhibits Influenza A Virus Replication and Inflammation via TLR4, P38 MAPK and NF-ΚB Pathways. Int. J. Mol. Sci. 2018, 19, 965. [Google Scholar] [CrossRef] [Green Version]
- Ampomah, P.B.; Lim, L.H.K. Influenza A Virus-Induced Apoptosis and Virus Propagation. Apoptosis 2020, 25, 1–11. [Google Scholar] [CrossRef]
- Kim, S.-R.; Jeong, M.-S.; Mun, S.-H.; Cho, J.; Seo, M.-D.; Kim, H.; Lee, J.; Song, J.-H.; Ko, H.-J. Antiviral Activity of Chrysin against Influenza Virus Replication via Inhibition of Autophagy. Viruses 2021, 13, 1350. [Google Scholar] [CrossRef]
- Zhou, Y.; Pu, J.; Wu, Y. The Role of Lipid Metabolism in Influenza A Virus Infection. Pathogens 2021, 10, 303. [Google Scholar] [CrossRef]
- Domanska, M.K.; Wrona, D.; Kasson, P.M. Multiphasic Effects of Cholesterol on Influenza Fusion Kinetics Reflect Multiple Mechanistic Roles. Biophys. J. 2013, 105, 1383–1387. [Google Scholar] [CrossRef] [Green Version]
- Oguin, T.; Lindsley, C.; Thomas, P.; Brown, H. Effects of Lipid Signaling on Innate Immune Networks during Influenza Infection. (INM3P.360). J. Immunol. 2015, 194, 127.1. [Google Scholar]
- Jamieson, A.M.; Yu, S.; Annicelli, C.H.; Medzhitov, R. Influenza Virus-Induced Glucocorticoids Compromise Innate Host Defense against a Secondary Bacterial Infection. Cell Host Microbe 2010, 7, 103–114. [Google Scholar] [CrossRef] [Green Version]
- Graham, A.C.; Temple, R.M.; Obar, J.J. Mast Cells and Influenza a Virus: Association with Allergic Responses and Beyond. Front. Immunol. 2015, 6, 238. [Google Scholar] [CrossRef] [Green Version]
- Tavares, L.P.; Teixeira, M.M.; Garcia, C.C. The Inflammatory Response Triggered by Influenza Virus: A Two Edged Sword. Inflamm. Res. 2017, 66, 283–302. [Google Scholar] [CrossRef]
- Turpin, E.; Luke, K.; Jones, J.; Tumpey, T.; Konan, K.; Schultz-Cherry, S. Influenza Virus Infection Increases P53 Activity: Role of P53 in Cell Death and Viral Replication. J. Virol. 2005, 79, 8802–8811. [Google Scholar] [CrossRef] [Green Version]
- Menche, J.; Sharma, A.; Kitsak, M.; Ghiassian, S.; Vidal, M.; Loscalzo, J.; Barabási, A.-L. Uncovering Disease-Disease Relationships through the Incomplete Human Interactome. Science 2015, 347, 1257601. [Google Scholar] [CrossRef] [Green Version]
- Cheng, F.; Kovács, I.A.; Barabási, A.-L. Network-Based Prediction of Drug Combinations. Nat. Commun. 2019, 10, 1197. [Google Scholar] [CrossRef]
- Chou, T.-C. Theoretical Basis, Experimental Design, and Computerized Simulation of Synergism and Antagonism in Drug Combination Studies. Pharmacol. Rev. 2006, 58, 621–681. [Google Scholar] [CrossRef]
- Guo, J.; Guo, Y. The in vitro HAART pharmacodynamics study with dolutegravir as the “anchor”. Yao Xue Xue Bao 2015, 50, 50–58. [Google Scholar]
- Zhu, T.; Cao, S.; Su, P.-C.; Patel, R.; Shah, D.; Chokshi, H.B.; Szukala, R.; Johnson, M.E.; Hevener, K.E. Hit Identification and Optimization in Virtual Screening: Practical Recommendations Based Upon a Critical Literature Analysis. J. Med. Chem. 2013, 56, 6560–6572. [Google Scholar] [CrossRef] [Green Version]
- WHO Model List of Essential Medicines. Available online: https://www.who.int/publications-detail-redirect/WHOMVPEMPIAU2019.06 (accessed on 23 July 2021).
- Forsyth, C.J.; Hernandez, S.; Olmedo, W.; Abuhamidah, A.; Traina, M.I.; Sanchez, D.R.; Soverow, J.; Meymandi, S.K. Safety Profile of Nifurtimox for Treatment of Chagas Disease in the United States. Clin. Infect. Dis. 2016, 63, 1056–1062. [Google Scholar] [CrossRef] [Green Version]
- Stass, H.; Just, S.; Weimann, B.; Ince, I.; Willmann, S.; Feleder, E.; Freitas, C.; Yerino, G.; Münster, U. Clinical Investigation of the Biopharmaceutical Characteristics of Nifurtimox Tablets—Implications for Quality Control and Application. Eur. J. Pharm. Sci. 2021, 166, 105940. [Google Scholar] [CrossRef]
- Zhang, X. Direct Anti-HCV Agents. Acta Pharm. Sin. B 2016, 6, 26–31. [Google Scholar] [CrossRef] [Green Version]
- Kitchen, D.B.; Decornez, H.; Furr, J.R.; Bajorath, J. Docking and Scoring in Virtual Screening for Drug Discovery: Methods and Applications. Nat. Rev. Drug Discov. 2004, 3, 935–949. [Google Scholar] [CrossRef]
- Sotriffer, C. Virtual Screening: Principles, Challenges, and Practical Guidelines; Wiley-VCH Verlag: Weinheim, Germany, 2011. [Google Scholar]
- Smith, S.B.; Dampier, W.; Tozeren, A.; Brown, J.R.; Magid-Slav, M. Identification of Common Biological Pathways and Drug Targets across Multiple Respiratory Viruses Based on Human Host Gene Expression Analysis. PLoS ONE 2012, 7, e33174. [Google Scholar] [CrossRef]
- Iorio, F.; Isacchi, A.; di Bernardo, D.; Brunetti-Pierri, N. Identification of Small Molecules Enhancing Autophagic Function from Drug Network Analysis. Autophagy 2010, 6, 1204–1205. [Google Scholar] [CrossRef] [Green Version]
- Kumar, A.; Zhang, K.Y.J. Advances in the Development of Shape Similarity Methods and Their Application in Drug Discovery. Front. Chem. 2018, 6, 315. [Google Scholar] [CrossRef]
- Hall, B.S.; Wilkinson, S.R. Activation of Benznidazole by Trypanosomal Type I Nitroreductases Results in Glyoxal Formation. Antimicrob. Agents Chemother. 2012, 56, 115–123. [Google Scholar] [CrossRef] [Green Version]
- Sherif, I.O.; Al-Mutabagani, L.A.; Sabry, D.; Elsherbiny, N.M. Antineoplastic Activity of Chrysin against Human Hepatocellular Carcinoma: New Insight on GPC3/SULF2 Axis and LncRNA-AF085935 Expression. Int. J. Mol. Sci. 2020, 21, 7642. [Google Scholar] [CrossRef]
- Yeo, H.; Lee, Y.H.; Koh, D.; Lim, Y.; Shin, S.Y. Chrysin Inhibits NF-ΚB-Dependent CCL5 Transcription by Targeting IκB Kinase in the Atopic Dermatitis-Like Inflammatory Microenvironment. Int. J. Mol. Sci. 2020, 21, 7348. [Google Scholar] [CrossRef]
- Angelopoulou, E.; Pyrgelis, E.-S.; Piperi, C. Neuroprotective Potential of Chrysin in Parkinson’s Disease: Molecular Mechanisms and Clinical Implications. Neurochem. Int. 2020, 132, 104612. [Google Scholar] [CrossRef]
- Song, J.H.; Moon, K.Y.; Lee, S.C.; Kim, S.S. Inhibition of Hypoxia-Inducible Factor-1α and Vascular Endothelial Growth Factor by Chrysin in a Rat Model of Choroidal Neovascularization. Int. J. Mol. Sci. 2020, 21, 2842. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van der Worp, H.B.; Howells, D.W.; Sena, E.S.; Porritt, M.J.; Rewell, S.; O’Collins, V.; Macleod, M.R. Can Animal Models of Disease Reliably Inform Human Studies? PLoS Med. 2010, 7, e1000245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Justice, M.J.; Dhillon, P. Using the Mouse to Model Human Disease: Increasing Validity and Reproducibility. Dis. Model. Mech. 2016, 9, 101–103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Home—GEO—NCBI. Available online: https://www.ncbi.nlm.nih.gov/geo/ (accessed on 24 February 2021).
- GEO Accession Viewer. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GPL10558 (accessed on 2 March 2021).
- R: The R Project for Statistical Computing. Available online: https://www.r-project.org/ (accessed on 8 August 2021).
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. Limma Powers Differential Expression Analyses for RNA-Sequencing and Microarray Studies. Nucleic Acid Res. 2015, 43, e47. [Google Scholar] [CrossRef] [PubMed]
- GEO Accession Viewer. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GPL96 (accessed on 24 February 2021).
- Xu, C.; Xin, Y.; Chen, M.; Ba, M.; Guo, Q.; Zhu, C.; Guo, Y.; Shi, J. Discovery, Synthesis, and Optimization of an N-Alkoxy Indolylacetamide against HIV-1 Carrying NNRTI-Resistant Mutations from the Isatis Indigotica Root. Eur. J. Med. Chem. 2020, 189, 112071. [Google Scholar] [CrossRef]
- Tang, K.; Zhang, X.; Guo, Y. Identification of the Dietary Supplement Capsaicin as an Inhibitor of Lassa Virus Entry. Acta Pharm. Sin. B 2020, 10, 789–798. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A Flexible Trimmer for Illumina Sequence Data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.; Langmead, B.; Salzberg, S.L. HISAT: A Fast Spliced Aligner with Low Memory Requirements. Nat. Methods 2015, 12, 357–360. [Google Scholar] [CrossRef] [Green Version]
- Tang, K.; Wu, Y.; Chen, S.; Xin, Y.; Guo, Y. Discovery of the Anti-Influenza A Virus Activity of SB216763 and Cyclosporine A by Mining Infected Cells and Compound Cellular Signatures. Chin. Chem. Lett. 2021, in press. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, B.; Pache, L.; Chang, M.; Khodabakhshi, A.H.; Tanaseichuk, O.; Benner, C.; Chanda, S.K. Metascape Provides a Biologist-Oriented Resource for the Analysis of Systems-Level Datasets. Nat. Commun. 2019, 10, 1523. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A Software Environment for Integrated Models of Biomolecular Interaction Networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef]
- Kolde, R. Pheatmap: Pretty Heatmaps; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Neuwirth, E. RColorBrewer: ColorBrewer Palettes; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Wickham, H.; Chang, W.; Henry, L.; Pedersen, T.L.; Takahashi, K.; Wilke, C.; Woo, K.; Yutani, H.; Dunnington, D.; RStudio. Ggplot2: Create Elegant Data Visualisations Using the Grammar of Graphics; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]






| GEO Accession No. | Number of Patients | Number of Controls | Time Points | Number of Patients Used in This Study | Number of Controls Used in This Study | Time Point of Patients’ Samples Used in This Study |
|---|---|---|---|---|---|---|
| GSE111368 [16,17] | 109 | 130 | T1 (recruitment); T2 (approximately 48 h after T1); T3 (at least 4 weeks after T1) [16,17] | 40 | 130 | T1 (recruitment) |
| GSE68310 [18,19] | 133 | 128 | Before illness; 0, 2, 4, 6, and 21 days later after illness onset (winter); next spring [18,19] | 41 | 41 | 0 days later after illness onset |
| Compounds | Enrichment Score | p Value | Infectivity (%) ± SD |
|---|---|---|---|
| Nipecotic Acid | −0.833 | 0.00141 | 99.2 ± 1.9 |
| Difenidol | −0.783 | 0.02097 | 101.0 ± 0.3 |
| Metaraminol | −0.781 | 0.00477 | 99.5 ± 1.4 |
| Cetirizine | −0.751 | 0.00774 | 96.6 ± 2.6 |
| Aztreonam | −0.747 | 0.00198 | 99.2 ± 1.3 |
| Chrysin | −0.739 | 0.03656 | 28.0 ± 6.0 |
| Sulfinpyrazone | −0.737 | 0.00957 | 93.2 ± 4.1 |
| Propylthiouracil | −0.724 | 0.01176 | 97.2 ± 2.5 |
| Pentoxyverine | −0.721 | 0.01239 | 100.5 ± 0.2 |
| Betahistine | −0.718 | 0.01281 | 98.0 ± 0.7 |
| Ketorolac | −0.716 | 0.01323 | 57.5 ± 11.4 |
| Proxyphylline | −0.710 | 0.01466 | 98.6 ± 0.8 |
| Pyrazinamide | −0.706 | 0.01546 | 99.2 ± 1.0 |
| Levonorgestrel | −0.693 | 0.00209 | 100.9 ± 6.5 |
| Glibenclamide | −0.683 | 0.02216 | 84.0 ± 6.2 |
| Delsoline | −0.669 | 0.02707 | 98.9 ± 0.8 |
| Pyrantel | −0.656 | 0.01164 | 83.1 ± 20.7 |
| Pronetalol | −0.653 | 0.03364 | 97.8 ± 0.7 |
| Nifurtimox | −0.646 | 0.03728 | 0.0 ± 12.7 |
| Gene Symbol | Gene Name | Main Events Involved in IAV Infection | References | |
|---|---|---|---|---|
| Genes reversed by chrysin | CAMP | Cathelicidin antimicrobial peptide | Having a direct effect on virus particles | [171] |
| LAMP3 | Lysosomal-associated membrane protein 3 | Having a direct effect on virus particles | [172] | |
| ISG15 | ISG15 ubiquitin-like modifier | Disturbing life cycle of IAV infection; Participating in innate immune response | [173] | |
| TLR7 | Toll-like receptor 7 | Participating in innate immune response | [174] | |
| IRF7 | Interferon regulatory factor 7 | Participating in innate immune response | [67] | |
| IL1B | Interleukin 1 beta | Participating in innate immune response | [59] | |
| IRAK3 | Interleukin 1 receptor associated kinase 3 | Participating in innate immune response | [175] | |
| PIK3CG | Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit gamma | Participating in innate immune response | [176] | |
| C5 | Complement C5 | Participating in innate immune response | [177] | |
| CHI3L1 | Chitinase 3 like 1 | Participating in adapted immune response | [178] | |
| ID3 | Inhibitor of DNA binding 3 | Participating in adapted immune response | [179] | |
| Genes reversed by nifurtimox | MMP9 | Matrix metallopeptidase 9 | Participating in adapted immune response | [180] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xin, Y.; Chen, S.; Tang, K.; Wu, Y.; Guo, Y. Identification of Nifurtimox and Chrysin as Anti-Influenza Virus Agents by Clinical Transcriptome Signature Reversion. Int. J. Mol. Sci. 2022, 23, 2372. https://doi.org/10.3390/ijms23042372
Xin Y, Chen S, Tang K, Wu Y, Guo Y. Identification of Nifurtimox and Chrysin as Anti-Influenza Virus Agents by Clinical Transcriptome Signature Reversion. International Journal of Molecular Sciences. 2022; 23(4):2372. https://doi.org/10.3390/ijms23042372
Chicago/Turabian StyleXin, Yijing, Shubing Chen, Ke Tang, You Wu, and Ying Guo. 2022. "Identification of Nifurtimox and Chrysin as Anti-Influenza Virus Agents by Clinical Transcriptome Signature Reversion" International Journal of Molecular Sciences 23, no. 4: 2372. https://doi.org/10.3390/ijms23042372






