Photocatalytic Degradation of 4,4′-Isopropylidenebis(2,6-dibromophenol) on Magnetite Catalysts vs. Ozonolysis Method: Process Efficiency and Toxicity Assessment of Disinfection By-Products
Abstract
:1. Introduction
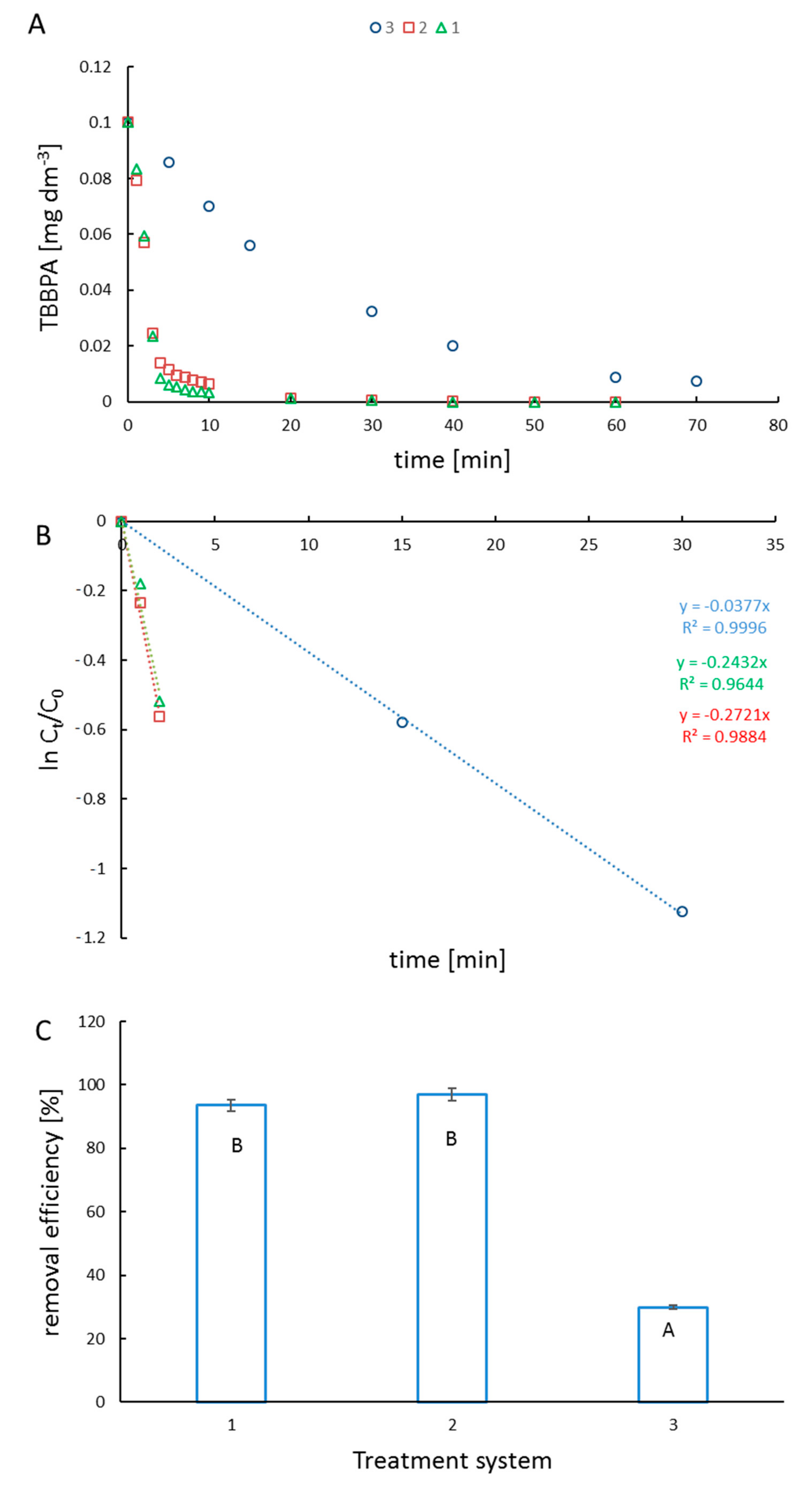
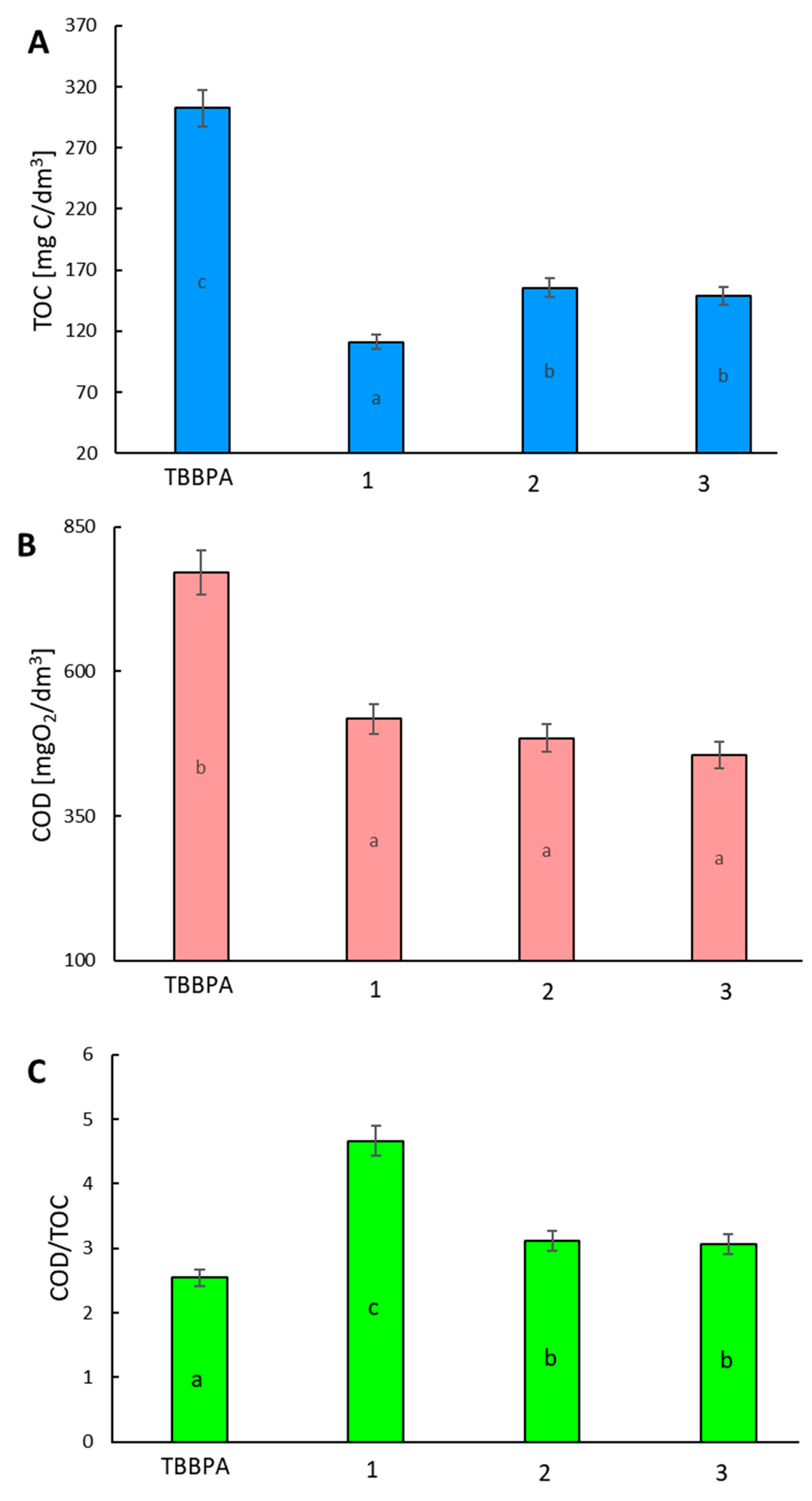
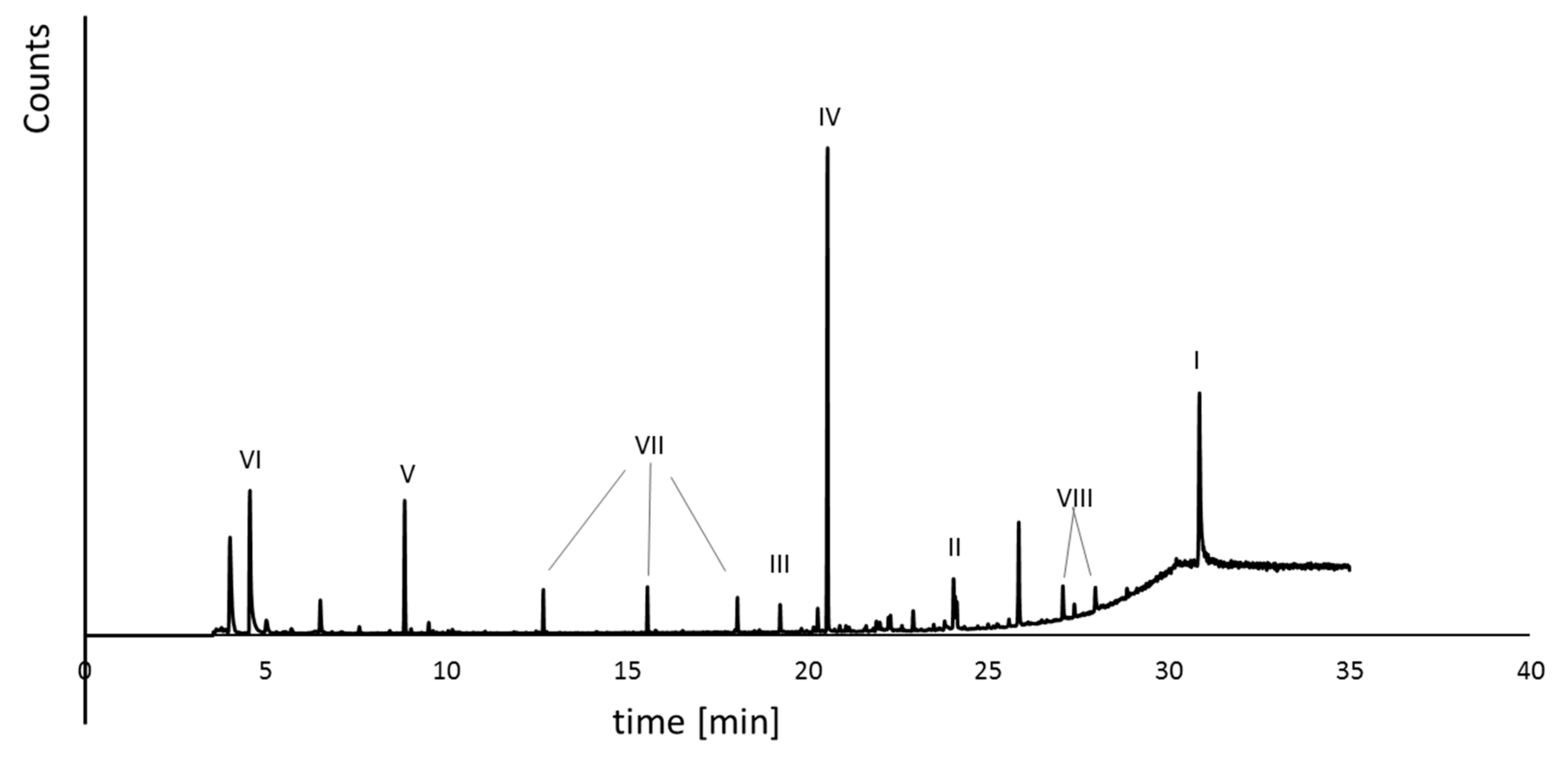
2. Results and Discussion
3. Materials and Methods
3.1. Chemical Reagents and Catalysts
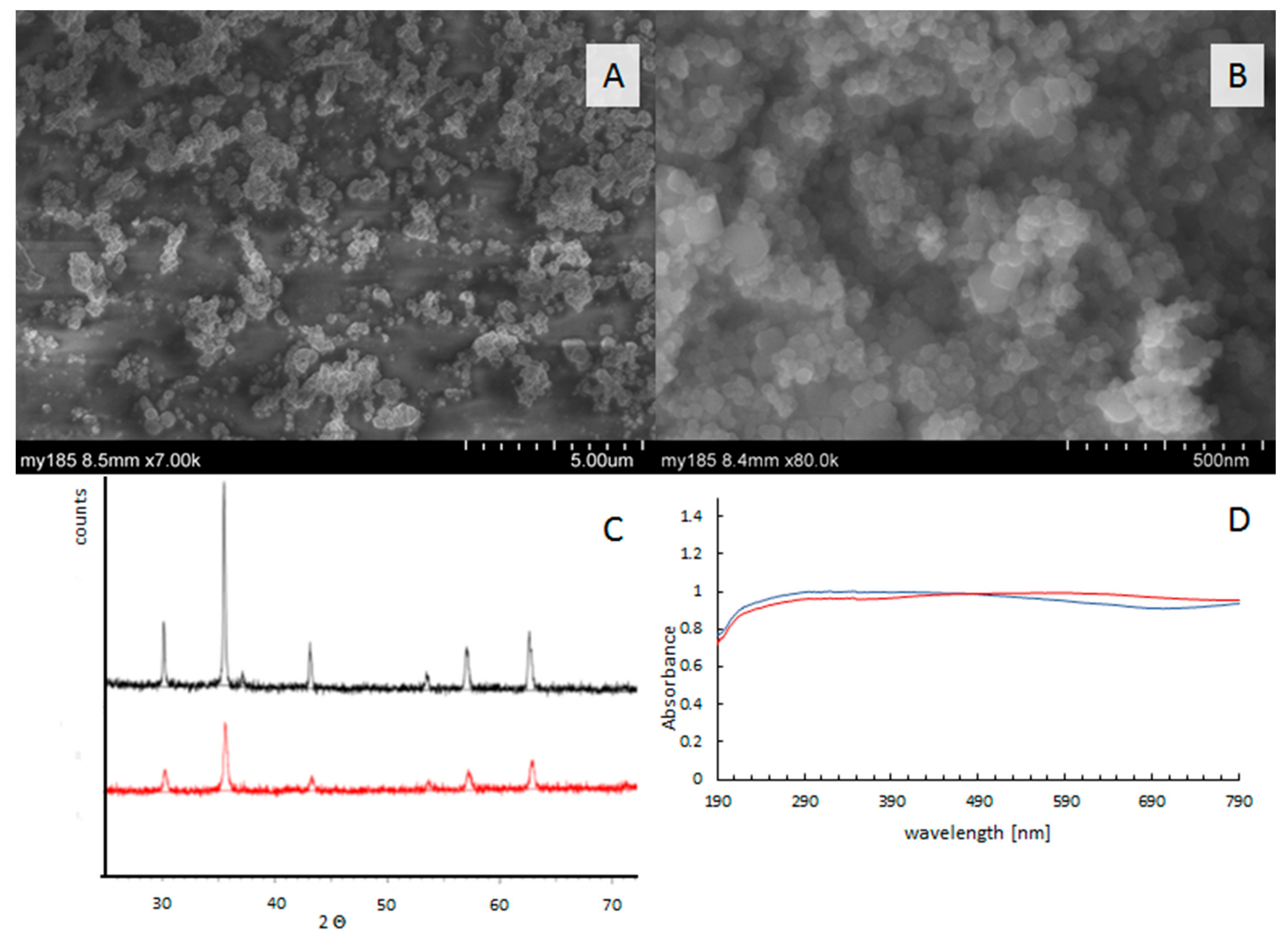
3.2. Catalyst Characterization
3.3. Degradation Procedure
3.3.1. Photocatalytic Degradation of TBBPA
3.3.2. Ozonolysis
3.3.3. Toxicity Tests
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, J.; Chan, K.M. Evaluation of the toxic effects of brominated compounds (BDE-47, 99, 209, TBBPA) and bisphenol A (BPA) using a zebrafish liver cell line, ZFL. Aquat. Toxicol. 2015, 159, 138–147. [Google Scholar] [CrossRef]
- Harrad, S.; Abdallah, M.A.; Rose, N.L.; Turner, S.D.; Davidson, T.A. Current-use brominated flame retardants in water, sediment, and fish from English lakes. Environ. Sci. Technol. 2009, 43, 9077–9083. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.Q.; Wong, C.K.C.; Zheng, J.S.; Bouwman, H.; Barra, R.; Wahlström, B.; Neretin, L.; Wong, M.H. Bisphenol A (BPA) in China: A review of sources, environmental levels, and potential human health impacts. Environ. Int. 2012, 42, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Tetrabromobisphenol A (TBBPA) Market-By Grade, By Application, By End Use and By Geography Analysis-Forecast 2020–2025. Available online: https://www.industryarc.com/Research/Tetrabromobisphenol-A-Market-Research-501611 (accessed on 17 August 2021).
- Barontini, F.; Cozzani, V.; Marsanich, K.; Raffa, V.; Petarca, L. An experimental investigation of tetrabromobisphenol A decomposition pathways. J. Anal. Appl. Pyrolysis 2004, 72, 41–53. [Google Scholar] [CrossRef]
- Söderström, G.; Marklund, S. PBCDD and PBCDF form incineration of waste containing brominated flame retardants. Environ. Sci. Technol. 2002, 36, 1959–1964. [Google Scholar] [CrossRef] [PubMed]
- Han, S.K.; Bilski, P.; Karriker, B.; Sik, R.H.; Chignell, C.F. Oxidation of Flame Retardant Tetrabromobisphenol A by Singlet Oxygen. Environ. Sci. Technol. 2008, 42, 166–172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moraes, B.S.; Loro, V.L.; Glusczak, L.; Pretto, A.; Menezes, C.; Marchezan, E.; de Oliveira Machado, S. Effects of four rice herbicides on some metabolic and toxicology parameters of teleost fish (Leporinus obtusidens). Chemosphere 2007, 68, 1597–1601. [Google Scholar] [CrossRef]
- Carballo, J.L.; Hernández-Inda, Z.L.; Pérez, P.; García-Grávalos, M.D. A comparison between two brine shrimp assays to detect in vitro cytotoxicity in marine natural products. BMC Biotechnol. 2002, 2, 17. [Google Scholar] [CrossRef]
- Veni, T.; Pushpanathan, T. Comparison of the Artemia salina and Artemia fransiscana bioassays for toxicity of Indian medicinal plants. J. Coastal Life Med. 2014, 2, 453–457. [Google Scholar]
- Mayorga, P.; Pérez, K.R.; Cruz, S.M.; Cáceres, A. Comparison of bioassays using the anostracan crustaceans Artemia salina and Thamnocephalus platyurus for plant extract toxicity screening. Bras. J. Pharm. 2010, 20, 897–903. [Google Scholar] [CrossRef] [Green Version]
- Kabekkodu, S. (Ed.) PDF-4+ 2015 (Database); International Centre for Diffraction Data: Newtown Square, PA, USA, 2015. [Google Scholar]
- Gutz, I.G.R. pH Calculation and Acid-Base Titration Curves—Freeware for Data Analysis and Simulation, (n.d.). Available online: http://www.iq.usp.br/gutz/Curtipot_.html (accessed on 6 April 2020).
- Regazzoni, A.E.; Blesa, M.A.; Maroto, A.J.G. Interfacial properties of zirconium dioxide and magnetite in water. J. Colloid Interface Sci. 1983, 91, 560–570. [Google Scholar] [CrossRef]
- White, A.F.; Peterson, M.L.; Hochella, M.F. Electrochemistry and dissolution kinetics of magnetite and ilmenite. Geochim. Cosmochim. Acta 1994, 58, 1859–1875. [Google Scholar] [CrossRef]
- Byun, J.D.; Kim, T.D.; Jung, B.; Shin, T.; Kim, H. TOC as a potential index for organic contents of wastewater treatment plant effluents. J. Korean Soc. Environ. Anal. 2010, 13, 99–103. [Google Scholar]
- Costa, J.A.; Farias, N.C.; Queirós, Y.G.C.; Mansur, C.R.E. Determination of oil-in-water using nanoemulsions as solvents and UV visible and total organic carbon detection methods. Talanta 2013, 107, 304–311. [Google Scholar] [CrossRef] [Green Version]
- He, W.; Chen, M.; Schlautman, M.A.; Hur, J. Dynamic exchanges between DOM and POM pools in coastal and inland aquatic ecosystems: A review. Sci. Total Environ. 2016, 551–552, 415–428. [Google Scholar] [CrossRef]
- Yang, L.; Hur, J. Critical evaluation of spectroscopic indices for organic matter source tracing via end member mixing analysis based on two contrasting sources. Water Res. 2014, 59, 80–89. [Google Scholar] [CrossRef]
- Bekiari, V.; Avramidis, P. Data quality in water analysis: Validation of combustion-infrared and combustion-chemiluminescence methods for the simultaneous determination of total organic carbon (TOC) and total nitrogen (TN). Intern. J. Environ. Anal. Chem. 2013, 94, 65–76. [Google Scholar] [CrossRef]
- Bisutti, I.; Hilke, I.; Schumacher, J.; Raessler, M. A novel single-run dual temperature combustion (SRDTC) method for the determination of organic, inorganic, and total carbon in soil samples. Talanta 2007, 71, 521–528. [Google Scholar] [CrossRef]
- Dubber, D.; Gray, N.F. Replacement of chemical oxygen demand (COD) with total organic carbon (TOC) for monitoring wastewater treatment performance to minimize the disposal of toxic analytical waste. J. Environ. Sci. Health 2010, 45, 1595–1600. [Google Scholar] [CrossRef]
- Park, D.S.M.; Lee, H.S.; Rhee, D.S.; Shin, H.S. Improvement in the analytical procedure for total organic carbon measurements in particle-containing water samples. J. Korean Soc. Environ. Anal. 2019, 22, 41–49. [Google Scholar]
- Eriksson, J.; Rahm, S.; Green, N.; Bergman, A.; Jakobsson, E. Photochemical transformations of tetrabromobisphenol A and related phenols in water. Chemosphere 2004, 54, 117–126. [Google Scholar] [CrossRef]
- Bao, Y.; Niu, J. Photochemical transformation of tetrabromobisphenol A under simulated sunlight irradiation: Kinetics, mechanism and influencing factors. Chemosphere 2015, 134, 550–556. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Weber, W.J. Transformation and removal of bisphenol A from aqueous phase via peroxidase-mediated oxidative coupling reactions: Efficacy, products, and pathways. Environ. Sci. Technol. 2005, 39, 6029–6036. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Liu, W.; Gan, J. Oxidative removal of bisphenol A by manganese dioxide: Efficacy, products, and pathways. Environ. Sci. Technol. 2009, 43, 3860–3864. [Google Scholar] [CrossRef]
- Wang, C.; Zhu, L.; Song, C.; Shan, G.; Chen, P. Characterization of photocatalyst Bi3.84W0.16O6.24 and its photodegradation on bisphenol A under simulated solar light irradiation. Appl. Catal. B Environ. 2011, 105, 229–236. [Google Scholar]
- Birnbaum, L.S.; Staskal, D.F. Brominated flame retardants: Cause for concern? Environ. Health Perspect. 2003, 112, 9–17. [Google Scholar] [CrossRef]
- Ahmed, A.; Labu, Z.K.; Dey, S.K.; Hira, A.; Howlader, M.S.I.; Mohamed Hemayet Hossain, M.H.; Roy, J. Phytochemical screening antibacterial and cytotoxic activity of different fractions of Xylocarpus mehongesis Bark. Ibnosina J. Med. Biomed. Sci. 2013, 5, 206–213. [Google Scholar] [CrossRef]
- Yang, S.; Wang, S.; Wu, F.; Yan, Z.; Liu, H. Tetrabromobisphenol A: Tissue distribution in fish, and seasonal variation in water and sediment of Lake Chaohu, China. Environ. Sci. Pollut. Res. 2012, 19, 4090–4096. [Google Scholar] [CrossRef]
- Quade, S.C.; Alaee, M.; Marvin, C.; Hale, R.; Solomon, K.R.; Bunce, N.J.; Fisk, A.T. Determination of tetrabromobisphenol-A in Detroit River sediment and sewage sludge. Organohalogen Compd. 2003, 62, 327–330. [Google Scholar]
- Morris, S.; Allchin, C.R.; Zegers, B.N.; Haftaka, J.J.H.; Boon, J.P.; Belpaire, C.; Leonards, P.E.G.; van Leeuwen, S.P.J.; de Boer, J. Distribution and fate of HBCD and TBBPA brominated flame retardants in North Sea estuaries and aquatic food webs. Environ. Sci. Technol. 2004, 38, 5497–5504. [Google Scholar] [CrossRef]
- Castritsi-Catharios, J.; Syriou, V.; Miliou, H.; Zouganelis, G.D. Toxicity effects of bisphenol A to the nauplii of the brine shrimp Artemia franciscana. J. Biological Res.-Tessaloniki 2013, 19, 1–8. [Google Scholar]
- Vindimian, E. REGTOX-EV6.xls. Available online: http://www.normalesup.org/~vindimian/en_index.html (accessed on 15 August 2020).
- European Commission. Technical Guidance Document in Support of Commission Directive 93/67/EEC on Risk Assessment for New Notified Substances. Part II, Environmental Risk Assessment; European Commission: Luxembourg, 1996; pp. 243–503. [Google Scholar]
- Ekonomou, G.; Lolas, A.; Castritsi-Catharios, J.; Neofitou, C.; Zouganelis, G.D.; Tsiropoulos, N.; Exadactylos, A. Mortality and Effect on Growth of Artemia Franciscana Exposed to Two Common Organic Pollutants. Water 2019, 11, 1614. [Google Scholar] [CrossRef] [Green Version]
- Staples, C.A.; Dorn, P.B.; Klecka, G.M.; O’Block, S.T.; Harris, L.R. A review of the environmental fate, effects and exposures of bisphenol A. Chemosphere 1998, 36, 2149–2173. [Google Scholar] [CrossRef]
- Ozlem, C.A.; Hatice, P. Effects of bisphenol A on the embryonic development of sea urchin (Paracentrotus lividus). Environ. Toxicol. 2008, 23, 387–392. [Google Scholar] [CrossRef]
- Mihaich, E.E.; Friederich, U.; Caspers, N.; Hall, A.T.; Klecka, G.M.; Dimond, S.S.; Staples, C.A.; Ortego, L.S.; Hentges, S.G. Acute and chronic toxicity testing of bisphenol A with aquatic invertebrates and plants. Ecotoxicol. Environ. Saf. 2009, 72, 1392–1399. [Google Scholar] [CrossRef] [PubMed]
- Debenest, T.; Gagne, F.; Petit, A.-N.; Andre, C.; Kohli, M.; Blaise, C. Ecotoxicology of a brominated flame retardant (tetrabromobisphenol A) and its derivatives to aquatic organisms. Comp. Biochem. Physiol. Part C 2010, 152, 407–412. [Google Scholar]
- Ogunbayo, O.A.; Jensen, K.T.; Michelangeli, F. The interaction of the brominated flame retardant: Tetrabromobisphenol A with phospholipid membranes. Biochim. Biophys. Acta 2007, 1768, 1559–1566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Glass, C.A.; Perrin, R.M.; Pocock, T.M.; Bates, D.O. Transient osmotic absorption of fluid in microvessels exposed to low concentrations of dimethylsulfoxide. Microcirculation 2006, 13, 29–40. [Google Scholar] [CrossRef] [PubMed]
- American Public Health Association. Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association, American Water Works Association, Water Environment Federation: Washington, DC, USA, 2005. [Google Scholar]
- PN-ISO 6060. Water Quality: Determination of Chemical Oxygen Demand (COD). ISO: Geneva, Switzerland, 2006. [Google Scholar]
- PN-EN 1484. Water Analysis—Guidelines for the Determination of Total Organic Carbon (TOC) and Dissolved Organic Carbon (DOC). ISO: Geneva, Switzerland, 1999. [Google Scholar]
- Thamnotoxkit F™. Crustacean Toxicity Screening Test for Freshwater. Standard Operational Procedure; MicroBioTests Inc.: Nazareth, Belgium, 1995; 23p. [Google Scholar]
- Artoxkit F™. Artemia Toxicity Screening Test for Estuarine and Marine Waters. Standard Operational Procedure; MicroBioTests Inc.: Nazareth, Belgium, 1995. [Google Scholar]

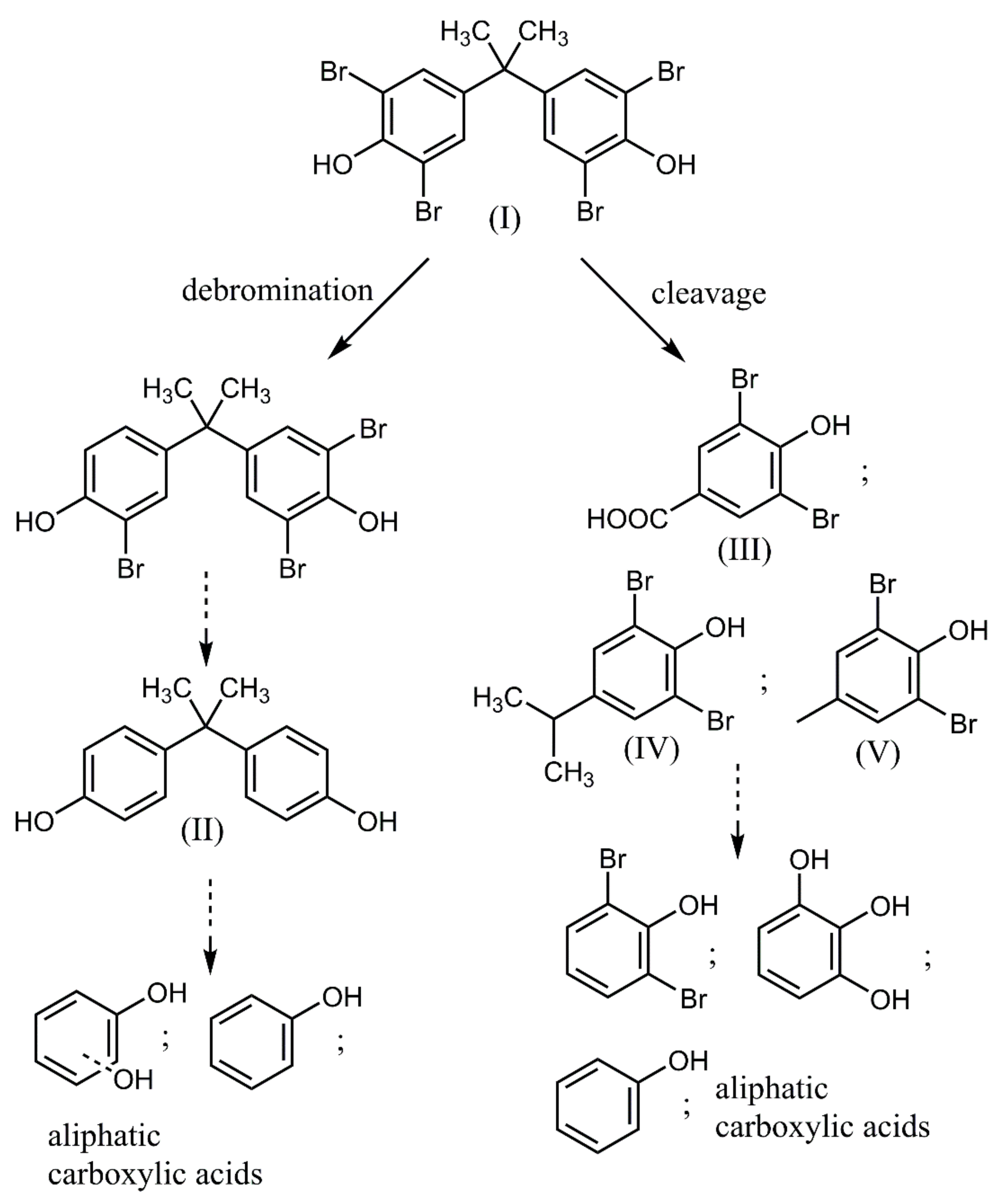
| Reaction System Number | Degradation Process |
|---|---|
| 1 | μ-Fe3O4 photocatalysis |
| 2 | n-Fe3O4 photocatalysis |
| 3 | ozonolysis |
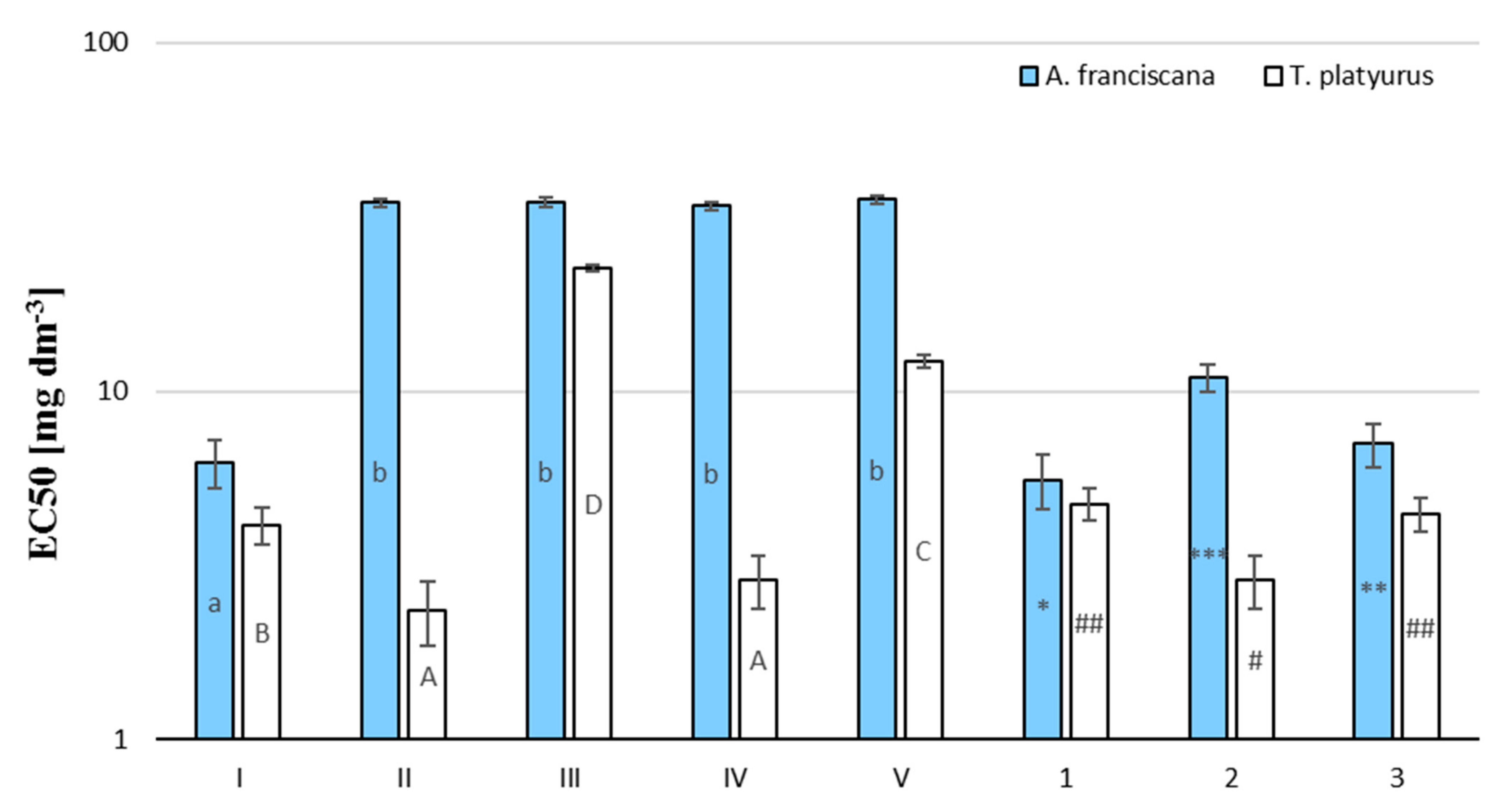
| Compound | EC50 (24 h), Artemia franciscana [mg dm−3] | EC50 (24 h), Thamnocephalus platyurus [mg dm−3] |
|---|---|---|
| TBBPA (I) | 6.243 (4.931–6.772) | 4.141 (3.47–4.61) |
| BPA (II) | 34.74 (28.35–43.26) | 2.35 (2.03–3.04) |
| 2,6-dibromo-4-hydroxybenzoic acid (III) | 34.89 (31.15–41.53) | 22.57 (19.44–28.76) |
| 2,6-dibromo-4-isopropylphenol (IV) | 34.01 (30.31–39.58) | 2.88 (2.64–3.23) |
| 2,6-dibromo-4-methylphenol (V) | 35.56 (28.08–39.43) | 12.15 (11.54–15.79) |
| TBBPA/μ-Fe3O4 (1) | 5.56 (4.61–6.25) | 4.748 (4.31–5.34) |
| TBBPA/n-Fe3O4 (2) | 10.95 (10.40–13.36) | 2.884 (2.24–3.14) |
| TBBPA/O3 (3) | 7.048 (6.14–7.82) | 4.436 (4.01–5.65) |
| Compound/Removal System | Extremely Toxic (<0.1 mg dm−3) | Very Toxic (0.1–1 mg dm−3) | Toxic (1–10 mg dm−3) | Harmful (10–100 mg dm−3) | Non Toxic (>100 mg dm−3) |
|---|---|---|---|---|---|
| TBBPA (I) | + • | ||||
| BPA (II) | • | + | |||
| 2,6-dibromo-4-hydroxybenzoic acid (III) | + • | ||||
| 2,6-dibromo-4-isopropylphenol (IV) | • | + | |||
| 2,6-dibromo-4-methylphenol (V) | + • | ||||
| TBBPA/μ-Fe3O4 (1) | + • | ||||
| TBBPA/n-Fe3O4 (2) | • | + | |||
| TBBPA/O3 (3) | + • |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kisała, J.; Tomaszewska, A.; Barylyak, A.; Bobitski, Y.; Balawejder, M. Photocatalytic Degradation of 4,4′-Isopropylidenebis(2,6-dibromophenol) on Magnetite Catalysts vs. Ozonolysis Method: Process Efficiency and Toxicity Assessment of Disinfection By-Products. Int. J. Mol. Sci. 2022, 23, 3438. https://doi.org/10.3390/ijms23073438
Kisała J, Tomaszewska A, Barylyak A, Bobitski Y, Balawejder M. Photocatalytic Degradation of 4,4′-Isopropylidenebis(2,6-dibromophenol) on Magnetite Catalysts vs. Ozonolysis Method: Process Efficiency and Toxicity Assessment of Disinfection By-Products. International Journal of Molecular Sciences. 2022; 23(7):3438. https://doi.org/10.3390/ijms23073438
Chicago/Turabian StyleKisała, Joanna, Anna Tomaszewska, Adriana Barylyak, Yaroslav Bobitski, and Maciej Balawejder. 2022. "Photocatalytic Degradation of 4,4′-Isopropylidenebis(2,6-dibromophenol) on Magnetite Catalysts vs. Ozonolysis Method: Process Efficiency and Toxicity Assessment of Disinfection By-Products" International Journal of Molecular Sciences 23, no. 7: 3438. https://doi.org/10.3390/ijms23073438







