Molecular and Cytogenetic Analysis of rDNA Evolution in Crepis Sensu Lato
Abstract
1. Introduction
2. Results
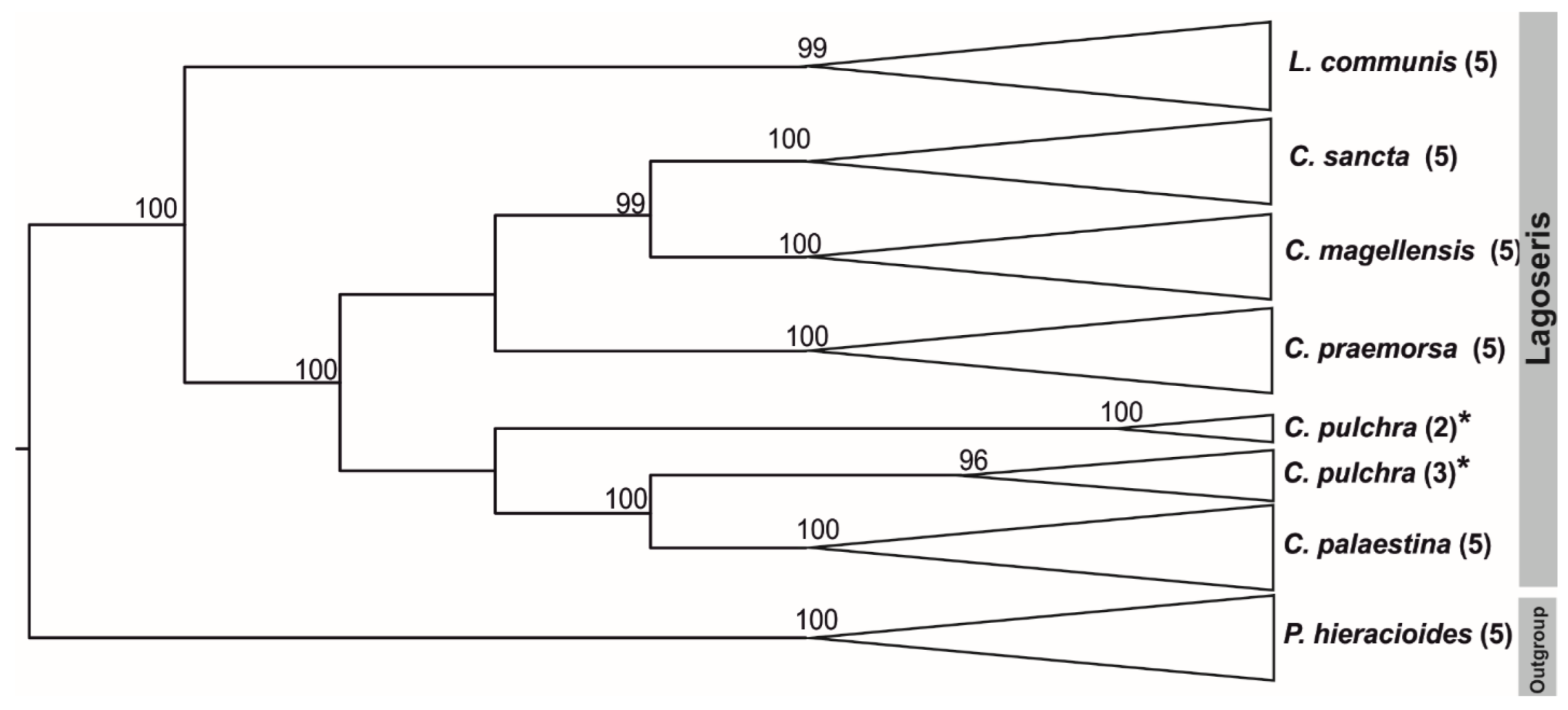
2.1. Phylogenetic Analyses of the 5S rDNA NTS and nrITS
2.2. Chromosome Number Evolution
2.3. Chromosomal Organisation of the rDNA Loci
2.4. Fluorochrome Banding
2.5. Patterns of Ribosomal DNA Loci Evolution
3. Discussion
4. Materials and Methods
4.1. Plant Material and DNA Isolation
4.2. DNA Amplification and Sequencing
4.3. Phylogenetic Analyses, Inferences of the Patterns of the Evolution of the rDNA Loci and Ancestral State Reconstructions
4.4. Chromosome Preparation and Fluorescent In Situ Hybridisation
4.5. Fluorochrome Banding
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, S.-F.; Su, T.; Cheng, G.-Q.; Wang, B.-X.; Li, X.; Deng, C.-L.; Gao, W.-J. Chromosome evolution in connection with repetitive sequences and epigenetics in plants. Genes 2017, 8, 290. [Google Scholar] [CrossRef] [PubMed]
- Schubert, I.; Vu, G.T.H. Genome stability and evolution: Attempting a holistic view. Trends Plant Sci. 2016, 21, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Sancho, R.; Inda, L.A.; Díaz-Pérez, A.; Des Marais, D.L.; Gordon, S.; Vogel, J.; Lusinska, J.; Hasterok, R.; Contreras-Moreira, B.; Catalán, P. Tracking the ancestry of known and ‘Ghost’ homeologous subgenomes in model grass Brachypodium polyploids. Plant J. 2022. [Google Scholar] [CrossRef] [PubMed]
- Mandáková, T.; Guo, X.; Özüdoğru, B.; Mummenhoff, K.; Lysak, M.A. Hybridization-facilitated genome merger and repeated chromosome fusion after 8 million years. Plant J. 2018, 96, 748–760. [Google Scholar] [CrossRef]
- Mota, L.; Torices, R.; Loureiro, J. The evolution of haploid chromosome numbers in the sunflower family. Genome Biol. Evol. 2016, 8, 3516–3528. [Google Scholar] [CrossRef]
- Ferraz, M.E.; Fonsêca, A.; Pedrosa-Harand, A. Multiple and independent rearrangements revealed by comparative cytogenetic mapping in the dysploid Leptostachyus group (Phaseolus L., Leguminosae). Chromosome Res. 2020, 28, 395–405. [Google Scholar] [CrossRef]
- Lusinska, J.; Betekhtin, A.; Lopez-Alvarez, D.; Catalan, P.; Jenkins, G.; Wolny, E.; Hasterok, R. Comparatively barcoded chromosomes of Brachypodium perennials tell the story of their karyotype structure and evolution. Int. J. Mol. Sci. 2019, 20, 5557. [Google Scholar] [CrossRef]
- Lusinska, J.; Majka, J.; Betekhtin, A.; Susek, K.; Wolny, E.; Hasterok, R. Chromosome identification and reconstruction of evolutionary rearrangements in Brachypodium distachyon, B. stacei and B. hybridum. Ann. Bot. 2018, 122, 445–459. [Google Scholar] [CrossRef]
- Liu, X.; Sun, S.; Wu, Y.; Zhou, Y.; Gu, S.; Yu, H.; Yi, C.; Gu, M.; Jang, J.; Liu, B.; et al. Dual-color oligo-FISH can reveal chromosomal variations and evolution in Oryza species. Plant J. 2020, 101, 112–121. [Google Scholar] [CrossRef]
- Lysak, M.A.; Mandáková, T.; Schranz, M.E. Comparative paleogenomics of crucifers: Ancestral genomic blocks revisited. Curr. Opin. Plant Biol. 2016, 30, 108–115. [Google Scholar] [CrossRef]
- Mandaákovaá, T.; Lysak, M.A. Chromosomal phylogeny and karyotype evolution in x=7 crucifer species (Brassicaceae). Plant Cell 2008, 20, 2559–2570. [Google Scholar] [CrossRef] [PubMed]
- Mandáková, T.; Lysak, M.A. Post-polyploid diploidization and diversification through dysploid changes. Curr. Opin. Plant Biol. 2018, 42, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Braz, G.T.; He, L.; Zhao, H.; Zhang, T.; Semrau, K.; Rouillard, J.M.; Torres, G.A.; Jiang, J. Comparative oligo-FISH mapping: An efficient and powerful methodology to reveal karyotypic and chromosomal evolution. Genetics 2018, 208, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J. Fluorescence in Situ hybridization in plants: Recent developments and future applications. Chromosome Res. 2019, 27, 153–165. [Google Scholar] [CrossRef]
- Lee, Y.-I.; Yap, J.W.; Izan, S.; Leitch, I.J.; Fay, M.F.; Lee, Y.-C.; Hidalgo, O.; Dodsworth, S.; Smulders, M.J.M.; Gravendeel, B.; et al. Satellite DNA in Paphiopedilum subgenus Parvisepalum as revealed by high-throughput sequencing and fluorescent in situ hybridization. BMC Genom. 2018, 19, 578. [Google Scholar] [CrossRef]
- Waminal, N.E.; Pellerin, R.J.; Kang, S.-H.; Kim, H.H. Chromosomal mapping of tandem repeats revealed massive chromosomal rearrangements and insights into Senna tora dysploidy. Front. Plant Sci. 2021, 12, 629898. [Google Scholar] [CrossRef]
- Moraes, A.P.; Olmos Simões, A.; Ojeda Alayon, D.I.; de Barros, F.; Forni-Martins, E.R. Detecting mechanisms of karyotype evolution in Heterotaxis (Orchidaceae). PLoS ONE 2016, 11, e0165960. [Google Scholar] [CrossRef]
- Costa, L.; Oliveira, Á.; Carvalho-Sobrinho, J.; Souza, G. Comparative cytomolecular analyses reveal karyotype variability related to biogeographic and species richness patterns in Bombacoideae (Malvaceae). Plant Syst. Evol. 2017, 303, 1131–1144. [Google Scholar] [CrossRef]
- Volkov, R.; Medina, F.; Zentgraf, U.; Hemleben, V. Organization and molecular evolution of rDNA nucleolar dominance and nucleolus structure. In Progress in Botany; Esser, K., Luttge, U., Beyschlag, W., Murata, J., Eds.; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 2004; Volume 65. [Google Scholar]
- Vasconcelos, E.V.; Vasconcelos, S.; Ribeiro, T.; Benko-Iseppon, A.M.; Brasileiro-Vidal, A.C. Karyotype heterogeneity in Philodendron s.l. (Araceae) revealed by chromosome mapping of rDNA loci. PLoS ONE 2018, 13, e0207318. [Google Scholar] [CrossRef]
- Kolano, B.; Siwinska, D.; McCann, J.; Weiss-Schneeweiss, H. The evolution of genome size and rDNA in diploid species of Chenopodium s.l. (Amaranthaceae). Bot. J. Linn. Soc. 2015, 179, 218–235. [Google Scholar] [CrossRef]
- Jang, T.-S.; Emadzade, K.; Parker, J.; Temsch, E.M.; Leitch, A.R.; Speta, F.; Weiss-Schneeweiss, H. Chromosomal diversification and karyotype evolution of diploids in the cytologically diverse genus Prospero (Hyacinthaceae). BMC Evol. Biol. 2013, 13, 136. [Google Scholar] [CrossRef] [PubMed]
- Costa, L.; Jimenez, H.; Carvalho, R.; Carvalho-Sobrinho, J.; Escobar, I.; Souza, G. Divide to conquer: Evolutionary history of Allioideae tribes (Amaryllidaceae) is linked to distinct trends of karyotype evolution. Front. Plant Sci. 2020, 11, 320. [Google Scholar] [CrossRef] [PubMed]
- Feliner, G.N.; Rossello, J.A. Better the devil you know? Guidelines for insightful utilization of nrDNA ITS in species-level evolutionary studies in plants. Mol. Phylogenet. Evol. 2007, 44, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Garcia, S.; Panero, J.L.; Siroky, J.; Kovarik, A. Repeated reunions and splits freature the highly dynamic evolution of 5S and 35S ribosomal RNA genes (rDNA) in the Asteraceae family. BMC Plant Biol. 2010, 10, 176. [Google Scholar] [CrossRef] [PubMed]
- Enke, N.; Gemeinholzer, B. Babcock revisited: New insights into generic delimitation and character evolution in Crepis L. (Compositae: Cichorieae) from ITS and matK sequence data. Taxon 2008, 57, 756–768. [Google Scholar] [CrossRef]
- Senderowicz, M.; Nowak, T.; Rojek-Jelonek, M.; Bisaga, M.; Papp, L.; Weiss-Schneeweiss, H.; Kolano, B. Descending dysploidy and bidirectional changes in genome size accompanied Crepis (Asteraceae) evolution. Genes 2021, 12, 1436. [Google Scholar] [CrossRef]
- Babcock, E.B. The Genus Crepis I. The Taxonomy, Phylogeny, Distribution and Evolution of Crepis. The Genus Crepis II. Systematic Treatment; University of California Press: Berkeley, CA, USA; Los Angeles, CA, USA, 1947. [Google Scholar]
- Jamilena, M.; Ruiz Rejón, C.; Ruiz Rejón, M. Repetitive DNA sequence families in Crepis capillaris. Chromosoma 1993, 102, 272–278. [Google Scholar] [CrossRef]
- Enke, N.; Kunze, R.; Pustahija, F.; Glöckner, G.; Zimmermann, J.; Oberländer, J.; Kamari, G.; Siljak-Yakovlev, S. Genome size shifts: Karyotype evolution in Crepis section Neglectoides (Asteraceae). Plant Biol. 2015, 17, 775–786. [Google Scholar] [CrossRef]
- Maluszynska, J.; Schweizer, D. Ribosomal RNA genes in B chromosomes of Crepis capillaris detected by non-radioactive in situ hybridization. Heredity 1989, 62, 59–65. [Google Scholar] [CrossRef]
- Hizume, M. Chromosomal localization of 5S rRNA genes in Vicia faba and Crepis capillaris. Cytologia 1993, 58, 417–421. [Google Scholar] [CrossRef]
- Schweizer, D. Reverse fluorescent chromosome banding with chromomycin and DAPI. Chromosoma 1976, 58, 307–324. [Google Scholar] [CrossRef] [PubMed]
- Fehrer, J.; Slavíková, R.; Paštová, L.; Josefiová, J.; Mráz, P.; Chrtek, J.; Bertrand, Y.J.K. Molecular evolution and organization of ribosomal DNA in the Hawkweed Tribe Hieraciinae (Cichorieae, Asteraceae). Front. Plant Sci. 2021, 12, 647375. [Google Scholar] [CrossRef] [PubMed]
- Kolano, B.; McCann, J.A.M.I.E.; Oskędra, M.; Chrapek, M.; Rojek, M.; Nobis, A.; Weiss-Schneeweiss, H. Parental origin and genome evolution of several eurasian hexaploid species of chenopodium (Chenopodiaceae). Phytotaxa 2019, 392, 163–185. [Google Scholar] [CrossRef]
- Volkov, R.A.; Panchuk, I.I.; Borisjuk, N.V.; Hosiawa-Baranska, M.; Maluszynska, J.; Hemleben, V. Evolutional dynamics of 45S and 5S ribosomal DNA in ancient allohexaploid Atropa belladonna. BMC Plant Biol. 2017, 17, 21. [Google Scholar] [CrossRef] [PubMed]
- Matyácek, R.; Fulnecek, J.; Lim, K.Y.; Leitch, A.R.; Kovarík, A. Evolution of 5S rDNA unit arrays in the plant genus Nicotiana (Solanaceae). Genome 2002, 45, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Persson, C. Phylogeny of the Neotropical Alibertia group (Rubiaceae), with emphasis on the genus Alibertia, inferred from ITS and 5S ribosomal DNA sequences. Am. J. Bot. 2000, 87, 1018–1028. [Google Scholar] [CrossRef] [PubMed]
- Waminal, N.E.; Ryu, K.B.; Park, B.R.; Kim, H.H. Phylogeny of Cucurbitaceae species in Korea based on 5S rDNA non-transcribed spacer. Genes Genom. 2014, 36, 57–64. [Google Scholar] [CrossRef]
- Grabiele, M.; Aguilera, P.M.; Ducasse, D.A.; Debat, H.J. Molecular characterization of the 5S rDNA non-Transcribed spacer and reconstruction of phylogenetic relationships in Capsicum. Rodriguesia 2021, 72. [Google Scholar] [CrossRef]
- Pornpongrungrueng, P.; Borchsenius, F.; Gustafsson, M.H.G. Relationships within Blumea (Inuleae, Asteraceae) and the Utility of the 5S-NTS in Species-Level Phylogeny Reconstruction. Taxon 2009, 58, 1181–1193. [Google Scholar] [CrossRef]
- Garcia, S.; Wendel, J.F.; Borowska-Zuchowska, N.; Aïnouche, M.; Kuderova, A.; Kovarik, A. The utility of graph clustering of 5S ribosomal DNA homoeologs in plant allopolyploids, homoploid hybrids, and cryptic introgressants. Front. Plant Sci. 2020, 11, 41. [Google Scholar] [CrossRef]
- Simon, L.; Rabanal, F.A.; Dubos, T.; Oliver, C.; Lauber, D.; Poulet, A.; Vogt, A.; Mandlbauer, A.; Le Goff, S.; Sommer, A.; et al. Genetic and epigenetic variation in 5S ribosomal RNA genes reveals genome dynamics in Arabidopsis thaliana. Nucleic Acids Res. 2018, 46, 3019–3033. [Google Scholar] [CrossRef] [PubMed]
- Garcia, S.; Kovařík, A.; Leitch, A.R.; Garnatje, T. Cytogenetic features of rRNA genes across land plants: Analysis of the Plant rDNA database. Plant J. 2017, 89, 1020–1030. [Google Scholar] [CrossRef] [PubMed]
- Matoba, H.; Mizutani, T.; Nagano, K.; Hoshi, Y.; Uchiyama, H. Chromosomal study of lettuce and its allied species (Lactuca spp., Asteraceae) by means of karyotype analysis and fluorescence in situ hybridization. Hereditas 2007, 144, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Roa, F.; Guerra, M. Non-random distribution of 5S rDNA sites and its association with 45S rDNA in plant chromosomes. Cytogenet. Genome Res. 2015, 146, 243–249. [Google Scholar] [CrossRef]
- Clarkson, J.J.; Lim, K.Y.; Kovarik, A.; Chase, M.W.; Knapp, S.; Leitch, A.R. Long-term genome diploidization in allopolyploid Nicotiana section Repandae (Solanaceae). New Phytol. 2005, 168, 241–252. [Google Scholar] [CrossRef]
- Jang, T.-S.; McCann, J.; Parker, J.S.; Takayama, K.; Hong, S.-P.; Schneeweiss, G.M.; Weiss-Schneeweiss, H. rDNA loci evolution in the genus Glechoma (Lamiaceae). PLoS ONE 2016, 11, e0167177. [Google Scholar] [CrossRef]
- Książczyk, T.; Taciak, M.; Zwierzykowski, Z. Variability of ribosomal DNA sites in Festuca pratensis, Lolium perenne, and their intergeneric hybrids, revealed by FISH and GISH. J. Appl. Genet. 2010, 51, 449–460. [Google Scholar] [CrossRef]
- Belyayev, A. Bursts of transposable elements as an evolutionary driving force. J. Evol. Biol. 2014, 27, 2573–2584. [Google Scholar] [CrossRef]
- Kalendar, R.; Raskina, O.; Belyayev, A.; Schulman, A.H. Long tandem arrays of Cassandra retroelements and their role in genome dynamics in plants. Int. J. Mol. Sci. 2020, 21, 2931. [Google Scholar] [CrossRef]
- Raskina, O.; Barber, J.C.; Nevo, E.; Belyayev, A. Repetitive DNA and chromosomal rearrangements: Speciation-related events in plant genomes. Cytogenet. Genome Res. 2008, 120, 351–357. [Google Scholar] [CrossRef]
- Książczyk, T.; Zwierzykowska, E.; Molik, K.; Taciak, M.; Krajewski, P.; Zwierzykowski, Z. Genome-dependent chromosome dynamics in three successive generations of the allotetraploid Festuca pratensis × Lolium perenne hybrid. Protoplasma 2015, 252, 985–996. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Thomas, H.M.; Harper, J.A.; Morgan, W.G. Gross chromosome rearrangements are occurring in an accession of the grass Lolium rigidum. Chromosome Res. 2001, 9, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Lan, H.; Chen, C.-L.; Miao, Y.; Yu, C.-X.; Guo, W.-W.; Xu, Q.; Deng, X.-X. Fragile Sites of ‘Valencia’ Sweet Orange (Citrus sinensis) Chromosomes Are Related with Active 45s rDNA. PLoS ONE 2016, 11, e0151512. [Google Scholar] [CrossRef] [PubMed]
- Rocha, L.C.; Ferreira, M.T.M.; Cunha, I.M.F.; Mittelmann, A.; Techio, V.H. 45S rDNA sites in meiosis of Lolium multiflorum Lam.: Variability, non-homologous associations and lack of fragility. Protoplasma 2019, 256, 227–235. [Google Scholar] [CrossRef]
- Novák, P.; Guignard, M.S.; Neumann, P.; Kelly, L.J.; Mlinarec, J.; Koblížková, A.; Dodsworth, S.; Kovařík, A.; Pellicer, J.; Wang, W.; et al. Repeat-sequence turnover shifts fundamentally in species with large genomes. Nat. Plants 2020, 6, 1325–1329. [Google Scholar] [CrossRef]
- Lan, T.; Albert, V.A. Dynamic distribution patterns of ribosomal DNA and chromosomal evolution in Paphiopedilum, a lady’s slipper orchid. BMC Plant Biol. 2011, 11, 126. [Google Scholar] [CrossRef]
- Siroký, J.; Lysák, M.A.; Dolezel, J.; Kejnovský, E.; Vyskot, B. Heterogeneity of rDNA distribution and genome size in Silene spp. Chromosome Res. 2001, 9, 387–393. [Google Scholar] [CrossRef]
- Silva, J.C.; Soares, F.A.F.; Sattler, M.C.; Clarindo, W.R. Repetitive sequences and structural chromosome alterations promote intraspecific variations in Zea mays L. karyotype. Sci. Rep. 2020, 10, 8866. [Google Scholar] [CrossRef]
- Zozomová-Lihová, J.; Mandáková, T.; Kovaříková, A.; Mühlhausen, A.; Mummenhoff, K.; Lysak, M.A.; Kovařík, A. When fathers are instant losers: Homogenization of rDNA loci in recently formed Cardamine × schulzii trigenomic allopolyploid. New Phytol. 2014, 203, 1096–1108. [Google Scholar] [CrossRef]
- Chiarini, F.E.; Santiñaque, F.F.; Urdampilleta, J.D.; Las Peñas, M.L. Genome size and karyotype diversity in Solanum sect. Acanthophora (Solanaceae). Plant Syst. Evol. 2014, 300, 113–125. [Google Scholar] [CrossRef]
- Hasterok, R.; Wolny, E.; Hosiawa, M.; Kowalczyk, M.; Kulak-Ksiazczyk, S.; Ksiazczyk, T.; Heneen, W.K.; Maluszynska, J. Comparative analysis of rDNA distribution in chromosomes of various species of Brassicaceae. Ann. Bot. 2006, 97, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Bernardes, E.C.S.; Benko-Iseppon, A.M.; Vasconcelos, S.; Carvalho, R.; Brasileiro-Vidal, A.C. Intra- and interspecific chromosome polymorphisms in cultivated Cichorium L. species (Asteraceae). Genet. Mol. Biol. 2013, 36, 357–363. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Huang, J.; Ma, L.; Yang, F.; Fei, S.-z.; Li, L. 45S rDNA regions are chromosome fragile sites expressed as gaps in vitro on metaphase chromosomes of root-tip meristematic cells in Lolium spp. PLoS ONE 2008, 3, e2167. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Rieseberg, L.H. Frequency, origins, and evolutionary role of chromosomal inversions in plants. Front. Plant Sci. 2020, 11, 296. [Google Scholar] [CrossRef]
- Zhang, S.-J.; Liu, L.; Yang, R.; Wang, X. Genome size evolution mediated by Gypsy retrotransposons in Brassicaceae. Genom. Proteom. Bioinform. 2020, 18, 321–332. [Google Scholar] [CrossRef]
- Liu, Z.; Liu, Y.; Liu, F.; Zhang, S.; Wang, X.; Lu, Q.; Wang, K.; Zhang, B.; Peng, R. Genome-wide survey and comparative analysis of long terminal repeat (LTR) retrotransposon families in four Gossypium species. Sci. Rep. 2018, 8, 9399. [Google Scholar] [CrossRef]
- Raskina, O.; Belyayev, A.; Nevo, E. Activity of the En/Spm-like transposons in meiosis as a base for chromosome repatterning in a small, isolated, peripheral population of Aegilops speltoides Tausch. Chromosome Res. 2004, 12, 153–161. [Google Scholar] [CrossRef]
- Symonová, R.; Majtánová, Z.; Sember, A.; Staaks, G.B.; Bohlen, J.; Freyhof, J.; Rábová, M.; Ráb, P. Genome differentiation in a species pair of coregonine fishes: An extremely rapid speciation driven by stress-activated retrotransposons mediating extensive ribosomal DNA multiplications. BMC Evol. Biol. 2013, 13, 42. [Google Scholar] [CrossRef]
- Xiong, Z.; Gaeta, R.T.; Edger, P.P.; Cao, Y.; Zhao, K.; Zhang, S.; Pires, J.C. Chromosome inheritance and meiotic stability in allopolyploid Brassica napus. G3 Genes Genomes Genet. 2020, 11, jkaa011. [Google Scholar] [CrossRef]
- Emadzade, K.; Jang, T.-S.; Macas, J.; Kovařík, A.; Novák, P.; Parker, J.; Weiss-Schneeweiss, H. Differential amplification of satellite PaB6 in chromosomally hypervariable Prospero autumnale complex (Hyacinthaceae). Ann. Bot. 2014, 114, 1597–1608. [Google Scholar] [CrossRef]
- Venora, G.; Blangiforti, S.; Frediani, M.; Maggini, F.; Gelati, M.T.; Castiglione, M.R.; Cremonini, R. Nuclear DNA contents, rDNAs, chromatin organization, and karyotype evolution in Vicia sect, faba. Protoplasma 2000, 213, 118–125. [Google Scholar] [CrossRef]
- Kolano, B.; McCann, J.; Orzechowska, M.; Siwinska, D.; Temsch, E.; Weiss-Schneeweiss, H. Molecular and cytogenetic evidence for an allotetraploid origin of Chenopodium quinoa and C. berlandieri (Amaranthaceae). Mol. Phylogenet. Evol. 2016, 100, 109–123. [Google Scholar] [CrossRef] [PubMed]
- Löytynoja, A.; Goldman, N. WebPRANK: A phylogeny-aware multiple sequence aligner with interactive alignment browser. BMC Bioinform. 2010, 11, 579. [Google Scholar] [CrossRef] [PubMed]
- Collingridge, P.W.; Kelly, S. MergeAlign: Improving multiple sequence alignment performance by dynamic reconstruction of consensus multiple sequence alignments. BMC Bioinform. 2012, 13, 117. [Google Scholar] [CrossRef]
- Minh, B.Q.; Schmidt, H.A.; Chernomor, O.; Schrempf, D.; Woodhams, M.D.; von Haeseler, A.; Lanfear, R. IQ-TREE 2: New models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef]
- Maddison, W.P.; Maddison, D.R. Mesquite: A Modular System for Evolutionary Analysis. Version 3.7. Available online: http://mesquiteproject.org (accessed on 1 March 2021).
- Glick, L.; Mayrose, I. ChromEvol: Assessing the pattern of chromosome number evolution and the inference of polyploidy along a phylogeny. Mol. Biol. Evol. 2014, 31, 1914–1922. [Google Scholar] [CrossRef]
- Dydak, M.; Kolano, B.; Nowak, T.; Siwinska, D.; Maluszynska, J. Cytogenetic studies of three European species of Centaurea L. (Asteraceae). Hereditas 2009, 146, 152–161. [Google Scholar] [CrossRef]
- Unfried, I.; Gruendler, P. Nucleotide sequence of the 5.8S and 25S rRNA genes and of the internal transcribed spacers from Arabidopsis thaliana. Nucleic Acids Res. 1990, 18, 4011. [Google Scholar] [CrossRef]
- Gerlach, W.L.; Dyer, T.A. Sequence organization of the repeating units in the nucleus of wheat which contain 5S rRNA genes. Nucleic Acids Res. 1980, 8, 4851–4865. [Google Scholar] [CrossRef]
- Kolano, B.; Saracka, K.; Broda-Cnota, A.; Maluszynska, J. Localization of ribosomal DNA and CMA3/DAPI heterochromatin in cultivated and wild Amaranthus species. Sci. Hortic. 2013, 164, 249–255. [Google Scholar] [CrossRef]







| Species | Chromosome Number | Number and Localisation of 35S  and 5S and 5S  rDNA Loci rDNA Lociand CMA3+  Bands Bands | |||
|---|---|---|---|---|---|
| 5S rDNA | 35S rDNA | CMA3+ | |||
| Crepis s.s. | |||||
| Crepis aculeata | 2n = 2x = 8 | 1 | 2 |  | 1 (35S rDNA) * |
| C. albida | 2n = 2x = 10 | 1 | 1 |  | 1 (35S rDNA) |
| C. alpestris | 2n = 2x = 8 | 1 | 1 |  | 1 (35S rDNA) |
| C. alpina | 2n = 2x = 10 | 1 | 1 |  | 1 (35S rDNA) |
| C. aspera | 2n = 2x = 8 | 2 | 1 |  | - |
| C. aurea | 2n = 2x = 10 | 1 | 1 |  | 2 (1 with 35S & 1 with 5S rDNA) |
| C. capillaris | 2n = 2x = 6 | 1 | 1 |  | 1 (35S rDNA) |
| C. conyzifolia (1) | 2n = 2x = 8 | 3 | 4 |  | 1 (35S rDNA) |
| C. conyzifolia (2) | 2n = 2x = 8 | 3 | 4 |  | 1 (35S rDNA) |
| C. conyzifolia subsp. dshimilensis (3) | 2n = 2x = 8 | 2 | 4 |  | 1 (35S rDNA) |
| C. chondrilloides | 2n = 2x = 8 | 1 | 3 |  | - |
| C. dioscoridis | 2n = 2x = 8 | 2 | 2 |  | 3 (2 with 35S & 1 with 5S rDNA) |
| C. foetida (1) | 2n = 2x = 10 | 1 | 1 |  | 1 (35S rDNA) |
| C. foetida subsp. rhoaedifolia (2) | 2n = 2x = 10 | 3 | 1 |  | 1 (35S rDNA) |
| C. jacquinii | 2n = 2x = 12 | 1 | 1 |  | - |
| C. kotschyana | 2n = 2x = 8 | 1 | 1 |  | 1 (35S rDNA) |
| C. lacera | 2n = 2x = 8 | 2 | 1 |  | 2 (1 with 35S & 1 with 5S rDNA) |
| C. leontodontoides | 2n = 2x = 10 | 1 | 1 |  | 1 (35S rDNA) |
| C. lyrata | 2n = 2x = 12 | 1 | 1 |  | 1 (35S rDNA) |
| C. mollis | 2n = 2x = 12 | 2 | 1 |  | 1 (35S rDNA) |
| C. nicaeensis | 2n = 2x = 8 | 1 | 2 |  | - |
| C. nigrescens | 2n = 2x = 8 | 1 | 1 |  | - |
| C. oporinoides | 2n = 2x = 8 | 1 | 2 |  | 1 (35S rDNA) |
| C. paludosa | 2n = 2x = 12 | 1 | 1 |  | 1 (35S rDNA) |
| C. pannonica (1) | 2n = 2x = 8 | 2 | 3 |  | 2 (35S rDNA) |
| C. pannonica (2) | 2n = 2x = 8 | 3 | 4 |  | - |
| C. polymorpha | 2n = 2x = 8 | 2 | 1 |  | - |
| C. pygmeae | 2n = 2x = 12 | 3 | 1 |  | 1 (35S rDNA) |
| C. pyrenaica | 2n = 2x = 8 | 1 | 1 |  | 1 (35S rDNA) |
| C. rubra | 2n = 2x = 10 | 1 | 2 |  | 4 (2 with 35S rDNA) |
| C. setosa 1 | 2n = 2x = 8 | 1 | 1 + (1) |  | 1 (35S rDNA) |
| C. setosa 2 | 2n = 2x = 8 | 1 | 1 or 2 |  | 1 (35S rDNA) |
| C. sibirica | 2n = 2x = 10 | 1 | 1 |  | - |
| C. succisifolia | 2n = 2x = 12 | 2 | 1 |  | 1 (35S rDNA) |
| C. syriaca | 2n = 2x = 10 | 1 | 1 |  | 1 (35S rDNA) |
| C. taraxacifolia | 2n = 2x = 8 | 2 | 1 |  | 1 (35S rDNA) |
| C. tectorum | 2n = 2x = 8 | 1 | 1 |  | 1 (35S rDNA) |
| C. vesicaria (3) | 2n = 2x = 8 | 2 | 1 |  | 1 (35S rDNA) |
| C. vesicaria (1) | 2n = 4x = 16 | 2 | 1 |  | 2 (35S rDNA) |
| C. vesicaria (2) | 2n = 4x = 16 | 2 | 1 |  | 2 (35S rDNA) |
| C. zacintha | 2n = 2x = 6 | 1 | 1 |  | 1 (35S rDNA) |
| Lagoseris | |||||
| C. magellensis | 2n = 2x = 10 | 1 | 2 |  | 1 (35S rDNA) |
| C. palaestina | 2n = 2x = 8 | 2 | 3 |  | 2 (35S rDNA) |
| C. praemorsa | 2n = 2x = 8 | 2 | 1 |  | - |
| C. pulchra | 2n = 2x = 8 | 2 | 2 |  | 2 (35S rDNA) |
| C. sancta | 2n = 2x = 10 | 1 | 2 |  | 2 (35S rDNA) |
| Lapsana communis | 2n = 2x = 14 | 3 | 1 |  | 1 (35S rDNA) |
| Species | Collection Number | Voucher Number | 5S rDNA NTS |
|---|---|---|---|
| GenBank Number | |||
| Crepis s.s. | |||
| Crepis aculeata Boiss. | BGT 38 | KTU154623 | MZ226690–MZ226694 |
| C. albida Vill. | UGA 233 | - | MZ226725–MZ226729 |
| C. alpestris (Jacq.) Tausch | BGUG N49 | KTU157712 | MZ226695–MZ226699 |
| C. alpina L. | USDA PI 274367 | KTU154609 | MZ226705–MZ226709 |
| C. aspera L. | LBG 006722 | KTU157716 | MZ226715–MZ226719 |
| C. aurea (L.) Cass. | USDA PI 312843 | KTU157719 | MZ226720–MZ226724 |
| C. capillaris Wallr. | BGGU 335 | KTU154610 | MZ226735–MZ226739 |
| C. conyzifolia (Gouan) A.Kern. (1) | GBA 462 | KTU157720 | MZ226740–MZ226744 |
| C. conyzifolia (Gouan) A.Kern. (2) | UGA 236 | - | MZ226745–MZ226749 |
| C. conyzifolia subsp. dshimilensis (K.Koch) Lamond (3) | GBBG | - | MZ226839–MZ226843 |
| C. chondrilloides Jacq. | Triest, Italy 45°36′46.54″ N 13°51′36.96″ E | - | MZ226952–MZ226956 |
| C. dioscoridis L. | IPK CRE2 | KTU154619 | MZ226750–MZ226754 |
| C. foetida L. | USDA PI 296071 | KTU154612 | MZ226755–MZ226759 |
| C. foetida subsp. rhoaedifolia (M.Bieb.) Celak. | HBBH 1734 | KTU154614 | MZ226864–MZ226868 |
| C. jacquinii Tausch | Sarnia Skała Tatra Mts, Poland 49°15′52.77″ N 19°56′30.36″ E | KTU159736 | MZ226760–MZ226764 |
| C. kotschyana Boiss. | USDA PI 310392 | KTU164608 | MZ226765–MZ226769 |
| C. lacera Ten. | BGMN | KTU159735 | MZ226770–MZ226774 |
| C. leontodontoides All. | BGDG 658 | KTU154631 | MZ226775–MZ226779 |
| C. lyrata (L.) Froel. | SSBG | - | MZ226780–MZ226785 |
| C. mollis Asch. | Sławków, Poland 50°17′45.90″ N 19°16′59.06″ E | KTU154630 | MZ226792–MZ226796 |
| C. nicaeensis Balb. | BGEU | KTU157730 | MZ226797–MZ226801 |
| C. nigrescens Pohle | HUM | - | MZ226802–MZ226806 |
| C. oporinoides Boiss. ex Froel. | ABGL 1516 | KTU154622 | MZ226710–MZ226714 |
| C. paludosa Moench | Sławków, Poland 50°18′07.51″ N 19°21′19.10″ E | KTU154625 | MZ226807–MZ226811 |
| C. pannonica (Jacq.) K.Koch (1) | BGBD 256-01-00-14 | KTU154627 | MZ226817–MZ226826 |
| C. pannonica (Jacq.) K.Koch (2) | BGEU | KTU157729 | MZ226827–MZ226833 |
| C. polymorpha Pourr | JBN 149 | KTU157725 | MZ226834–MZ226838 |
| C. pygmeae L. | UGA 239 | KTU157722 | MZ226854–MZ226858 |
| C. pyrenaica (L.) Greuter | BGBD 1010 | KTU154621 | MZ226859–MZ226863 |
| C. rubra L. | BGK 364 | KTU154607 | MZ226869–MZ226873 |
| C. setosa Haller f. 1 | HBUR 1275 | KTU154620 | MZ226879–MZ226883 |
| C. sibirica L. | BGBD 738 | KTU157721 | MZ226884–MZ226889 |
| C. succisifolia Tausch | Rędziny, Poland 50°49′08.66″ N 15°55′55.27″ E | KTU154656 | MZ226890–MZ226894 |
| C. syriaca (Bornm.) Babc. & Navashin | KEW 0129064 | KTU154615 | MZ226895–MZ226899 |
| C. taraxacifolia Thuill. | BGGU 347 | KTU157723 | MZ226900–MZ226904 |
| C. tectorum L. | Ustroń, Poland 49°43′14.68″ N 18°49′29.11″ E | KTU157717 | MZ226905–MZ226909 |
| C. veiscaria L. 1 (4x) | BGBD 1014 | KTU154616 | MZ226910–MZ226920 |
| C. vesicaria L. 2 (4x) | BGBD 918 | KTU157726 | MZ226921–MZ226931 |
| C. vesicaria L. 3 (2x) | OBUP | KTU157724 | MZ226932–MZ226936 |
| C. zacintha (L.) Loisel. | BGT 92 | KTU154606 | MZ226937–MZ226941 |
| Lagoseris | |||
| C. magellensis F. Conti & Uzunov | BGMN | KTU157727 | MZ226787–MZ226791 |
| C. palaestina Bornm. | BGGU 335 | KTU154611 | MZ226812–MZ226816 |
| C. pulchra L. | BGGU 341 | KTU154648 | MZ226849–MZ226853 |
| C. preamorsa (L.) Tausch | BGBD 662 | KTU154628 | MZ226844–MZ226848 |
| C. sancta (L.) Bornm. | BGUK 104 | KTU154613 | MZ226874–MZ226878 |
| Lapsana communis L. | KEW 0018568 | KTU154617 | MZ226942–MZ226946 |
| Outgroup | |||
| Picris hieracioides L. | Jaworzno Poland 50°13′31.43″ N 19°16′28.63″ E | KTU157710 | MZ226947–MZ226951 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Senderowicz, M.; Nowak, T.; Weiss-Schneeweiss, H.; Papp, L.; Kolano, B. Molecular and Cytogenetic Analysis of rDNA Evolution in Crepis Sensu Lato. Int. J. Mol. Sci. 2022, 23, 3643. https://doi.org/10.3390/ijms23073643
Senderowicz M, Nowak T, Weiss-Schneeweiss H, Papp L, Kolano B. Molecular and Cytogenetic Analysis of rDNA Evolution in Crepis Sensu Lato. International Journal of Molecular Sciences. 2022; 23(7):3643. https://doi.org/10.3390/ijms23073643
Chicago/Turabian StyleSenderowicz, Magdalena, Teresa Nowak, Hanna Weiss-Schneeweiss, Laszlo Papp, and Bozena Kolano. 2022. "Molecular and Cytogenetic Analysis of rDNA Evolution in Crepis Sensu Lato" International Journal of Molecular Sciences 23, no. 7: 3643. https://doi.org/10.3390/ijms23073643
APA StyleSenderowicz, M., Nowak, T., Weiss-Schneeweiss, H., Papp, L., & Kolano, B. (2022). Molecular and Cytogenetic Analysis of rDNA Evolution in Crepis Sensu Lato. International Journal of Molecular Sciences, 23(7), 3643. https://doi.org/10.3390/ijms23073643






