New Poly(N-isopropylacrylamide-butylacrylate) Copolymer Biointerfaces and Their Characteristic Influence on Cell Behavior In Vitro
Abstract
:1. Introduction
2. Results and Discussions
2.1. p(NIPAM-BA) Interfaces’ Morphology Obtained by MAPLE
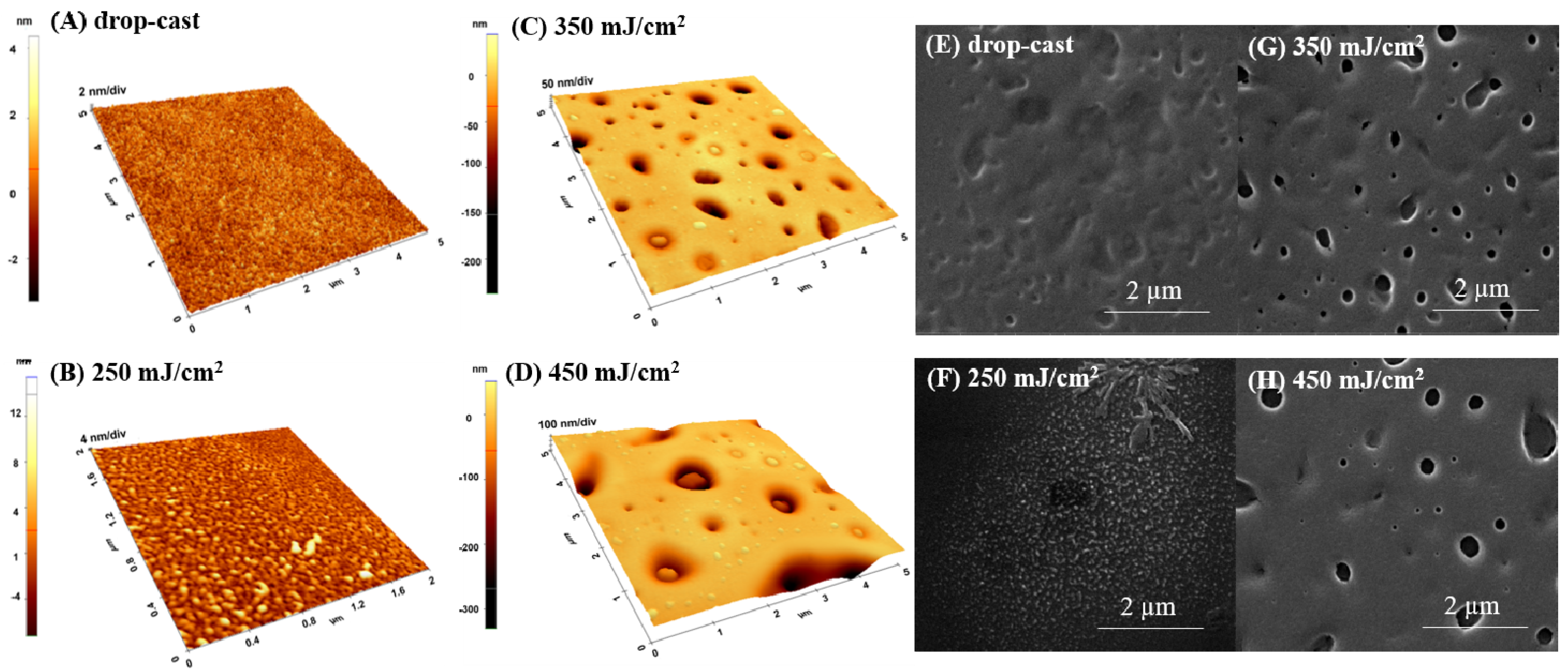
2.1.1. Water-Solvent
2.1.2. DMSO-Solvent
2.1.3. Chloroform-Solvent
2.2. p(NIPAM-BA) Interfaces’ Morphology Obtained by Spin Coating (SC)
2.3. FTIR Investigations of the p(NIPAM-BA) Copolymer Coatings
2.4. Surface Composition Measurements by X-Ray Photoelectron Spectroscopy (XPS) Analysis
2.5. Contact Angles Measurements and Swelling Behavior
2.6. Biological In Vitro Investigations
2.6.1. Cell Viability
2.6.2. Adhesion and Distribution of Cells onto the p(NIPAM-BA) Layers
3. Materials and Methods
3.1. Materials and Reagents
3.2. Surface Designing/Tailoring/Deposition
3.2.1. MAPLE Method
3.2.2. Spin Coating Method
3.3. p(NIPAM-BA) Morphologic Analysis
3.3.1. Scanning Electron Microscopy (SEM)
3.3.2. Atomic Force Microscopy (AFM)
3.4. Wettability Characterization by Contact Angle (CA)
3.5. Chemical Profile: Analysis of p(NIPAM-BA)
3.5.1. Fourier Transform Infrared Spectroscopy (FTIR) Analysis
3.5.2. X-ray Photoelectron Spectroscopy (XPS) Analysis
3.6. Biological Investigations
3.6.1. Cells
3.6.2. Cell Viability Assay
3.6.3. Cell Adhesion by Scanning Electron Microscopy (SEM)
3.6.4. Statistical Analysis
3.7. Samples/Batch Production
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, Q.; Ma, L.; Gao, C. Biomaterials for in situ tissue regeneration: Development and perspectives. J. Mater. Chem. B 2015, 3, 8921–8938. [Google Scholar] [CrossRef] [PubMed]
- Adel, I.M.; ElMeligy, M.F.; Elkasabgy, N.A. Conventional and Recent Trends of Scaffolds Fabrication: A Superior Mode for Tissue Engineering. Pharmaceutics 2022, 14, 306. [Google Scholar] [CrossRef] [PubMed]
- Shimojo, A.A.M.; Rodrigues, I.C.P.; Perez, A.G.M.; Souto, E.M.B.; Gabriel, L.P.; Webster, T. Scaffolds for Tissue Engineering: A State of-the-Art Review Concerning Types, Properties, Materials, Processing, and Characterization BT. In Racing for the Surface: Antimicrobial and Interface Tissue Engineering; Li, B., Moriarty, T.F., Webster, T., Xing, M., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 647–676. [Google Scholar]
- Abdelaal, O.A.M.; Darwish, S.M.H. Review of rapid prototyping techniques for tissue engineering scaffolds fabrication. In Characterization and Development of Biosystems and Biomaterials; Springer: Berlin/Heidelberg, Germany, 2021; pp. 33–54. [Google Scholar]
- Chang, H.-I. Cell Responses to surface and architecture of tissue engineering scaffolds. In Regenerative Medicine and Tissue Engineering-Cells and Biomaterials; Eberli, Y.W.E.-D., Ed.; IntechOpen: Rijeka, Croatia, 2011; p. 27. [Google Scholar]
- Schmidt, S.; Zeiser, M.; Hellweg, T.; Duschl, C.; Fery, A.; Moehwald, H. Adhesion and Mechanical Properties of PNIPAM Microgel Films and Their Potential Use as Switchable Cell Culture Substrates. Adv. Funct. Mater. 2010, 20, 3235–3243. [Google Scholar] [CrossRef]
- Gil, E.S.; Hudson, S.M. Stimuli-reponsive polymers and their bioconjugates. Prog. Polym. Sci. 2004, 29, 1173–1222. [Google Scholar] [CrossRef]
- Lanzalaco, S.; Armelin, E. Poly(N-isopropylacrylamide) and Copolymers: A Review on Recent Progresses in Biomedical Applications. Gels 2017, 3, 36. [Google Scholar] [CrossRef]
- Roy, D.; Brooks, W.L.; Sumerlin, B.S. New directions in thermoresponsive polymers. Chem. Soc. Rev. 2013, 42, 7214–7243. [Google Scholar] [CrossRef]
- Buratti, E.; Sanzari, I.; Dinelli, F.; Prodromakis, T.; Bertoldo, M. Formation and Stability of Smooth Thin Films with Soft Microgels Made of Poly(N-Isopropylacrylamide) and Poly(Acrylic Acid). Polymers 2020, 12, 2638. [Google Scholar] [CrossRef]
- Fan, X.; Gu, S.; Wu, L.; Yang, L. Preparation and characterization of thermoresponsive poly(N-isopropylacrylamide) copolymers with enhanced hydrophilicity. Polymers 2020, 20, 561–570. [Google Scholar] [CrossRef]
- Becerra, N.; Restrepo, L.M.; López, B.L. Synthesis and characterization of a biocompatible copolymer to be used as cell culture support. Macromol. Symp. 2007, 258, 30–37. [Google Scholar] [CrossRef]
- Becerra, N.Y.; López, B.L.; Restrepo, L.M. Thermosensitive behavior in cell culture media and cytocompatibility of a novel copolymer: Poly(N-isopropylacrylamide-co-butylacrylate). J. Mater. Sci. Mater. Med. 2013, 24, 1043–1052. [Google Scholar] [CrossRef]
- Calandrelli, L.; Annunziata, M.; Ragione, F.D.; Laurienzo, P.; Malinconico, M.; Oliva, A. Development and performance analysis of PCL/silica nanocomposites for bone regeneration. J. Mater. Sci. Mater. Med. 2010, 21, 2923–2936. [Google Scholar] [CrossRef] [PubMed]
- Piqué, A.; McGill, R.A.; Chrisey, D.B.; Leonhardt, D.; Mslna, E.; Spargo, B.J.; Callahan, J.H.; Vachet, R.W.; Chung, R.; Bucaro, M.A. Growth of organic thin films by the matrix assisted pulsed laser evaporation (MAPLE) technique. Thin Solid Films 1999, 355, 536–541. [Google Scholar] [CrossRef]
- Caricato, A.P.; Luches, A.; Leggieri, G.; Martino, M.; Rella, R. Matrix-assisted pulsed laser deposition of polymer and nanoparticle films. Vacuum 2012, 86, 661–666. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, Y.; Yu, S.; Zhang, Z.; He, C.; Chen, X. pH- and Amylase-Responsive Carboxymethyl Starch/Poly(2-isobutyl-acrylic acid) Hybrid Microgels as Effective Enteric Carriers for Oral Insulin Delivery. Biomacromolecules 2018, 19, 2123–2136. [Google Scholar] [CrossRef]
- Ahiabu, A.; Serpe, J.M. Rapidly Responding pH- and Temperature-Responsive Poly(N-Isopropylacrylamide)-Based Microgels and Assemblies. ACS Omega 2017, 2, 1769–1777. [Google Scholar] [CrossRef] [Green Version]
- Yin, X.; Hoffman, S.A.; Stayton, S.P. Poly(N-isopropylacrylamide-co-propylacrylic acid) Copolymers That Respond Sharply to Temperature and pH. Biomacromolecules 2006, 7, 1381–1385. [Google Scholar] [CrossRef]
- Garbern, J.C.; Hoffman, A.S.; Stayton, P.S. Injectable pH- and Temperature-Responsive Poly(N-isopropylacrylamide-co-propylacrylic acid) Copolymers for Delivery of Angiogenic Growth Factors. Biomacromolecules 2010, 11, 1833–1839. [Google Scholar] [CrossRef] [Green Version]
- Sigma Aldrich. Poly(N-isopropylacrylamide-co-butylacrylate). Available online: https://www.sigmaaldrich.com/RO/en/product/aldrich/762857 (accessed on 10 January 2022).
- Caricato, A.P.; Arima, V.; Cesaria, M.; Martino, M.; Tunno, T.; Rinaldi, R.; Zacheo, A. Solvent-related effects in MAPLE mechanism. Appl. Phys. B 2013, 113, 463–471. [Google Scholar] [CrossRef]
- Chen, L.; Yan, C.; Zheng, Z. Functional polymer surfaces for controlling cell behaviors. Mater. Today 2018, 21, 38–59. [Google Scholar] [CrossRef]
- Leveugle, E.; Zhigilei, L.V. Molecular dynamics simulation study of the ejection and transport of polymer molecules in matrix-assisted pulsed laser evaporation. J. Appl. Phys. 2007, 102, 074914. [Google Scholar] [CrossRef] [Green Version]
- Huang, G.; Chen, Y.; Zhang, J. Nanocomposited coatings produced by laser-assisted process to prevent silicone hydogels from protein fouling and bacterial contamination. Appl. Surf. Sci. 2016, 360, 383–388. [Google Scholar] [CrossRef]
- Icriverzi, M.; Rusen, L.; Sima, L.E.; Moldovan, A.; Brajnicov, S.; Bonciu, A.; Mihailescu, N.; Dinescu, M.; Cimpean, A.; Roseanu, A.; et al. In vitro behavior of human mesenchymal stem cells on poly(N-isopropylacrylamide) based biointerfaces obtained by matrix assisted pulsed laser evaporation. Appl. Surf. Sci. 2018, 440, 712–724. [Google Scholar] [CrossRef]
- Loh, Q.L.; Choong, C. Three-dimensional scaffolds for tissue engineering applications: Role of porosity and pore size. Tissue Eng. Part B Rev. 2013, 19, 485–502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rana, M.M.; De la Hoz Siegler, H. Tuning the Properties of PNIPAm-Based Hydrogel Scaffolds for Cartilage Tissue Engineering. Polymers 2021, 13, 3154. [Google Scholar] [CrossRef] [PubMed]
- Leveugle, E.; Sellinger, A.; Fitz-Gerald, J.M.; Zhigilei, L.V. Making Molecular Balloons in Laser-Induced Explosive Boiling of Polymer Solutions. Phys. Rev. Lett. 2007, 98, 216101. [Google Scholar] [CrossRef] [Green Version]
- Zhigilei, L.V.; Volkov, A.N.; Leveugle, E.; Tabetah, M. The effect of the target structure and composition on the ejection and transport of polymer molecules and carbon nanotubes in matrix-assisted pulsed laser evaporation. Appl. Phys. A 2011, 105, 529–546. [Google Scholar] [CrossRef]
- Wu, O.; Wang, R.; Zhou, Y.; Zhou, Y.; Huang, Y.; Ghosh, R.; Chen, X. Poly(N-isopropylacrylamide)-grafted dual stimuli-responsive filter paper for protein separation. Chin. J. Polym. Sci. 2015, 33, 1048–1057. [Google Scholar] [CrossRef]
- Dybal, J.; Trchová, M.; Schmidt, P. The role of water in structural changes of poly(N-isopropylacrylamide) and poly(N-isopropylmethacrylamide) studied by FTIR, Raman spectroscopy and quantum chemical calculations. Vib. Spectrosc. 2009, 51, 44–51. [Google Scholar] [CrossRef]
- Sanzari, I.; Buratti, E.; Huang, R.; Tusan, C.G.; Dinelli, F.; Evans, N.D.; Prodromakis, T.; Bertoldo, M. Poly(N-isopropylacrylamide) based thin microgel films for use in cell culture applications. Sci. Rep. 2020, 10, 6126. [Google Scholar] [CrossRef]
- Felgueiras, H.P.; Antunes, J.C.; Martins, M.C.L.; Barbosa, M.A. Fundamentals of protein and cell interactions in biomaterials. In Peptides and Proteins as Biomaterials for Tissue Regeneration and Repair; Barbosa, M., Martins, M.C., Eds.; Woodhead Publishing: Cambridge, UK, 2018; pp. 1–27. [Google Scholar]
- Ning, L.; Xu, N.; Wang, R.; Liu, Y. Fibrous membranes electrospun from the suspension polymerization product of styrene and butyl acrylate for oil-water separation. RSC Adv. 2015, 5, 57101. [Google Scholar] [CrossRef]
- Zuo, Y.; Zhao, J.; Gao, Y.; Zhang, Y. Controllable synthesis of P(NIPAM-co-MPTMS)/PAA-Au composite materials with tunable LSPR performance. J. Mater. Sci. 2017, 52, 9584–9601. [Google Scholar] [CrossRef]
- Kou, R.; Xu, Z.; Deng, H.; Liu, Z.; Seta, P.; Xu, Y. Surface modification of microporous polypropylene membranes by plasma-induced graft polymerization of α-allyl glucoside. Langmuir 2003, 19, 6869–6875. [Google Scholar] [CrossRef]
- Mizutani, A.; Kikuchi, A.; Yamato, M.; Kanazawa, H.; Okano, T. Preparation of thermo-responsive polymer brush surfaces and their interaction with cells. Biomaterials 2008, 29, 2073–2081. [Google Scholar] [CrossRef]
- Tang, Y.; Lu, J.R.; Lewis, A.L.; Vick, T.A.; Stratford, P.W. Swelling of zwitterionic polymer films characterized by spectroscopic ellipsometry. Macromolecules 2001, 34, 8768–8776. [Google Scholar] [CrossRef]
- Gilcreest, V.P.; Carroll, W.M.; Rochev, Y.A.; Blute, I.; Dawson, K.A.; Gorelov, A.V. Thermoresponsive Poly(N-isopropylacrylamide) Copolymers: Contact Angles and Surface Energies of Polymer Films. Langmuir 2004, 20, 10138–10145. [Google Scholar] [CrossRef] [PubMed]
- Zohuriaan-Mehr, M.J.; Kabiri, K.; Kheirabadi, M. Extraordinary Swelling Behavior of Poly(AMPS) Organogel in Solvent/DMSO Binary Mixed Media. J. Appl. Polym. Sci. 2010, 117, 1127–1136. [Google Scholar] [CrossRef]
- Tavagnacco, L.; Zaccarelli, E.; Chiessi, E. On the Molecular Origin of the Cooperative Coil-to-globule Transition of Poly(N-isopropylacrylamide) in Water. Phys. Chem. Chem. Phys. 2018, 20, 9997–10010. [Google Scholar] [CrossRef] [Green Version]
- Natalia, B.; Henry, A.; Betty, L.; Marina, R.L.; Roberto, R. Probing poly(N-isopropylacrylamide-co-butylacrylate)/cell interactions by atomic force microscopy. J. Biomed. Mater. Res. A 2015, 103, 145–153. [Google Scholar] [CrossRef]
- Tsai, H.-Y.; Vats, K.; Yates, M.Z.; Benoit, D.S.W. Two-Dimensional Micropatterns of Self-Assembled Poly(Nisopropylacrylamide) Microgels for Patterned Adhesion and Temperature-Responsive Detachment of Fibroblasts. Langmuir 2013, 29, 12183–12193. [Google Scholar] [CrossRef] [Green Version]
- Geiger, B.; Spatz, J.P.; Bershadsky, A.D. Environmental sensing through focal adhesions. Nat. Rev. Mol. Cell Biol. 2009, 10, 21–33. [Google Scholar] [CrossRef]
- ISO 10993-5:2009; Biological Evaluation of Medical Devices—Part 5: Tests for In Vitro Cytotoxicity. International Standards Organization: Geneva, Switzerland, 2009.
- Li, Y.; Xiao, Y.; Liu, C. The Horizon of Materiobiology: A Perspective on Materi-al-Guided Cell Behaviors and Tissue Engineering. Chem. Rev. 2017, 117, 4376–4421. [Google Scholar] [CrossRef] [PubMed]
- Capella, V.; Rivero, R.E.; Liaudat, A.C.; Ibarra, L.E.; Roma, D.A.; Alustiza, F.; Manas, F.; Barbero, C.A.; Bosch, P.; Rivarola, C.R.; et al. Cytotoxicity and bioadhesive properties of poly-N-isopropylacrylamide hydrogel. Heliyon 2019, 5, e01474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lian, J.; Xu, H.; Duan, S.; Ding, X.; Hu, Y.; Zhao, N.; Ding, X.; Xu, F.J. Tunable Adhesion of Different Cell Types Modulated by Thermoresponsive Polymer Brush Thickness. Biomacromolecules 2020, 21, 732–742. [Google Scholar] [CrossRef] [PubMed]
- Litowczenko, J.; Gapiński, J.; Markiewicz, R.; Woźniak, A.; Wychowaniec, J.K.; Peplińska, B.; Jurga, S.; Patkowski, A. Synthesis, characterization and in vitro cytotoxicity studies of poly-N-isopropyl acrylamide gel nanoparticles and films. Mater. Sci. Eng. C 2021, 118, 111507. [Google Scholar] [CrossRef] [PubMed]
- Dumitrescu, L.N.; Neacsu, P.; Necula, M.G.; Bonciu, A.; Marascu, V.; Cimpean, A.; Dinca, V.; Dinescu, M. Induced Hydrophilicity and In Vitro Preliminary Osteoblast Response of Polyvinylidene Fluoride (PVDF) Coatings Obtained via MAPLE Deposition and Subsequent Thermal Treatment. Molecules 2020, 25, 582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kosoff, D.; Yu, J.; Suresh, V.; Beebe, D.J.; Lang, J.M. Surface topography and hydrophilicity regulate macrophage phenotype in milled microfluidic systems. Lab Chip 2018, 18, 3011–3017. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.M.; Jiang, Z.; Bastmeyer, M.; Lahann, J. Physical Aspects of Cell Culture Substrates: Topography, Roughness, and Elasticity. Small 2012, 8, 336–355. [Google Scholar] [CrossRef] [Green Version]
- Sima, F.; Davidson, P.; Pauthe, E.; Gallet, O.; Anselme, K.; Mihailescu, I. Thin films of vitronectin transferred by MAPLE. Appl. Phys. A 2011, 105, 611–617. [Google Scholar] [CrossRef]
- Dinca, V.; Mocanu, A.; Isopencu, G.; Busuioc, C.; Brajnicov, S.; Vlad, A.; Icriverzi, M.; Roseanu, A.; Dinescu, M.; Stroescu, M.; et al. Biocompatible pure ZnO nanoparticles-3D bacterial cellulose biointerfaces with antibacterial properties. Arab. J. Chem. 2020, 13, 3521–3533. [Google Scholar] [CrossRef]












| Roughness—RMS (nm) | ||||
|---|---|---|---|---|
| Deposition Methods | p(NIPAM-BA) 2 wt.%+ H2O | p(NIPAM-BA) 2 wt.%+ DMSO | p(NIPAM-BA) 2 wt.%+ Chloroform | |
| Drop-cast | 4.4 | 0.8 | 2.8 | |
| MAPLE | 250 mJ/cm2 | 36.3 | 1.5 | 25.7 |
| 350 mJ/cm2 | 139 | 27.7 | 143 | |
| 450 mJ/cm2 | 346 | 49.4 | 153.2 | |
| Spin Coating | 3.6 | 7 | 0.9 | |
| Solvent Used | Deposition Technique | |||||
|---|---|---|---|---|---|---|
| Drop-Cast | MAPLE Laser Fluence | Spin Coating | Atomic Composition (%) | |||
| 250 mJ/cm2 | 350 mJ/cm2 | 450 mJ/cm2 | ||||
| Water | 73.48 | 54.54 | 69.73 | 72.10 | 73.23 | C1s |
| 12.01 | 9.66 | 13.19 | 13.18 | 13.03 | N1s | |
| 14.51 | 26.79 | 17.08 | 14.71 | 13.74 | O1s | |
| - | 9.01 | - | - | - | Si2p | |
| DMSO | 73.72 | 55.96 | 64.57 | 64.00 | 72.99 | C1s |
| 12.30 | 9.91 | 11.76 | 11.17 | 13.57 | N1s | |
| 13.98 | 27.34 | 20.30 | 20.77 | 13.44 | O1s | |
| - | 6.79 | 3.38 | 4.06 | - | S2p | |
| Chlorofom | 72.71 | 59.83 | 67.83 | 66.16 | 74.15 | C1s |
| 12.82 | 5.62 | 10.70 | 9.23 | 12.78 | N1s | |
| 14.47 | 18.04 | 16.63 | 17.11 | 13.07 | O1s | |
| - | 16.51 | 4.84 | 7.50 | - | Cl2p | |
| p(NIPAM-BA) _Water | ||
|---|---|---|
| Laser Fluence (mJ/cm2) | CA (RT) CA (37 °C) | |
| Drop-Cast | 63.73 ± 0.34 | 70.87 ± 0.72 |
| 250 | 25.85 ± 0.52 | 28.24 ± 0.5 |
| 350 | 28.99 ± 0.13 | 33.4 ± 0.22 |
| 450 | 36.75 ± 0.41 | 45.07 ± 0.37 |
| p(NIPAM-BA) _DMSO | ||
|---|---|---|
| Laser Fluence (mJ/cm2) | CA (RT) CA (37 °C) | |
| Drop-Cast | 75.79 ± 1.24 | 60 ± 0.76 |
| 250 | 56.47 ± 0.84 | 62.31 ± 0.46 |
| 350 | 78.49 ± 0.58 | 75.7 ± 0.66 |
| 450 | 84.14 ± 1.36 | 61.64 ± 0.89 |
| p(NIPAM-BA) _ Chloroform | ||
|---|---|---|
| Laser Fluence (mJ/cm2) | CA (RT) CA (37 °C) | |
| Drop-Cast | 92.58 ± 1.89 | 70.37 ± 0.79 |
| 250 | 91.09 ± 1.65 | 99.44 ± 1.53 |
| 350 | 77.44 ± 0.68 | 74.3 ± 0.85 |
| 450 | 77.89 ± 0.66 | 75.43 ± 0.98 |
| p(NIPAM-BA) | CA (RT) | CA (37 °C) |
|---|---|---|
| Water | 63.73 ± 0.99 | 65.36 ± 0.36 |
| DMSO | 59.2 ± 0.24 | 66.78 ± 0.15 |
| Chloroform | 66.77 ± 1.166 68.77 ± 1.16 | 69.18 ± 0.72 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dumitrescu, N.-L.; Icriverzi, M.; Bonciu, A.; Florian, P.; Moldovan, A.; Roseanu, A.; Rusen, L.; Dinca, V.; Grama, F. New Poly(N-isopropylacrylamide-butylacrylate) Copolymer Biointerfaces and Their Characteristic Influence on Cell Behavior In Vitro. Int. J. Mol. Sci. 2022, 23, 3988. https://doi.org/10.3390/ijms23073988
Dumitrescu N-L, Icriverzi M, Bonciu A, Florian P, Moldovan A, Roseanu A, Rusen L, Dinca V, Grama F. New Poly(N-isopropylacrylamide-butylacrylate) Copolymer Biointerfaces and Their Characteristic Influence on Cell Behavior In Vitro. International Journal of Molecular Sciences. 2022; 23(7):3988. https://doi.org/10.3390/ijms23073988
Chicago/Turabian StyleDumitrescu, Nicoleta-Luminita, Madalina Icriverzi, Anca Bonciu, Paula Florian, Antoniu Moldovan, Anca Roseanu, Laurentiu Rusen, Valentina Dinca, and Florin Grama. 2022. "New Poly(N-isopropylacrylamide-butylacrylate) Copolymer Biointerfaces and Their Characteristic Influence on Cell Behavior In Vitro" International Journal of Molecular Sciences 23, no. 7: 3988. https://doi.org/10.3390/ijms23073988








