A Rheostat of Ceramide and Sphingosine-1-Phosphate as a Determinant of Oxidative Stress-Mediated Kidney Injury
Abstract
:1. Introduction
2. SLs Metabolism
3. The Enzymes That Generate Ceramide and S1P in the Kidney
4. Interaction between ROS, Antioxidants and Ceramide in Oxidant-Induced Kidney Injury
4.1. Increased Ceramide Generation in Oxidant-Induced Kidney Injury
4.2. ROS Activate and Translocate the Enzymes That Generate Ceramide
4.3. Antioxidants Regulate the Enzymes That Generate Ceramide
4.4. Ceramide Stimulates ROS Production and Inhibits Antioxidant Defense System Which Further Activates Ceramide-Producing Enzymes
5. Role of Mitochondrial Function in the Regulation of Ceramide and ROS Formation
5.1. Interaction between Bcl-2 Family Proteins and the Enzymes That Generate Ceramide
5.2. Bcl-2 Family Proteins Regulate Ceramide-Induced ROS Production
6. Role of Mitochondria and Cell Signaling Pathways for Ceramide-Induced Apoptosis in Oxidant-Induced Kidney Injury
6.1. Ceramide-Induced Cell Death in Oxidant-Induced Kidney Injury
6.2. A Role of Mitochondria for the Regulation of ROS/Antioxidants and Ceramide Generation
6.3. Ceramide-Induced Mitochondrial Dysfunction Leading to Apoptosis
6.4. Ceramide-Induced Cell Signaling Pathway for Cell Death
7. Interaction between ROS and S1P in Oxidant-Induced Kidney Injury
7.1. Alteration of S1P Levels in Oxidative Stress-Mediated Kidney Disease
| Oxidative Kidney Diseases | S1P Levels in Kidney and Plasma/Serum | Enzymes for S1P Alteration | Kidney Cell Response | References | |
|---|---|---|---|---|---|
| Toxic nephropathy Cadmium | kidney, plasma | kidney→, plasma↑ | kidney SphK1/2→, S1PR1→ | Necrosis/ Fibrosis | [208] |
| Radiation | podocyte | ↓ | SMPDL3b↓ | Apoptosis | [35] |
| Ischemia/reperfusion | RTC | ↓ | SphK1↓ | Necroptosis | [195] |
| kidney | NA | SphK1↑, SphK2→, S1PR1/3↑ | Apoptosis/necrosis | [196] | |
| EC | NA | SphK1↑, SphK2→ | Angiogenesis | [197] | |
| Unilateral ureteral obstruction | Kidney, plasma | kidney; NA, plasma↑ | RTC; SphK1↑ | Autophagy/fibrosis | [201] |
| Kidney | → | SphK→ | NA | [81] | |
| Kidney | kidney; NA | SphK1↑, S1PR1-3 ↑ | Necrosis/fibrosis | [202] | |
| Kidney, plasma | ↑ | SphK2↑ | Necrosis/fibrosis | [194] | |
| Anti-GBM Ab GN | Spleen | NA | S1PR1/2/5 ↑ | Necrosis | [203] |
| Nephrotic syndrome | Podocyte, serum | serum↑ | SGPL1↓ | FSGS | [83] |
| Podocyte, fibroblast | fibroblast↓ | SGPL1↓ | FSGS | [84] | |
| Chronic GN | Plasma | ↑ | UN | Necrosis | [209] |
| Plasma | S1P↑, HDL-S1P↑ | UN | ESRD | [210] | |
| Lupus nephritis | Plasma | ↓ | SGPL1↑ | NA | [93] |
| Serum, urine | serum↑, urine→ | UN | NA | [211] | |
| Kidney, serum | serum S1P↑, kidney, serum dhS1P↑ | SphK1/2↑ | Necrosis | [92,94] | |
| Serum | serum S1P↑, dhS1P↑ | PBMC SphK2→ | NA | [212] | |
| Diabetic nephropathy | Kidney | ↓ | UN | Apoptosis/MME/inflammation/ fibrosis | [43] |
| Kidney, glomerulus, MC | ↑ | SphK↑ | Proliferation | [46,47] | |
| MC | NA | SphK1↑ | Fibrosis | [198] | |
| Kidney | ↑ | SphK1↑ | Necrosis/proliferation/ inflammation/fibrosis | [199] | |
| Kidney, RTC | ↑ | SphK1↑ | Inflammation/fibrosis | [200] | |
| Kidney, MC | NA | SphK1↑ | Fibrosis | [204] | |
| VSMC | ↑ | SphK1↑, SphK2→ | Apoptosis | [205] | |
| Plasma | ↑ | UN | Necrosis | [96,213] | |
| Plasma | ↓ | UN | Necrosis | [214] | |
7.2. ROS Regulate SphKs/S1P and Vice Versa and the Role of Mitochondria for Regulation of ROS by S1P
8. Roles of Mitochondria and Cell Signaling Pathways for S1P-Induced Cellular Function in Oxidant-Induced Kidney Injury
8.1. A Role of SphK1/2 for S1P-Induced Survival
8.2. Role of Mitochondria for S1P-Induced Cell Survival
8.3. Cell Signaling Pathways for S1P-Induced Cell Survival, Proliferation, Inflammation and Fibrosis
8.3.1. Apoptosis
8.3.2. Cell Proliferation
8.3.3. Inflammation
8.3.4. Renal Fibrosis
9. A Rheostat of Ceramide-S1P in the Regulation of Oxidative Stress-Mediated Kidney Injury
9.1. Balance between Ceramide and S1P Regulates Oxidant-Induced Kidney Injury
9.2. Targeting Enzymes for Ceramide Generation and SphKs That Improve Ceramide-S1P Rheostat, Contributing to Prevention against Oxidant-Induced Kidney Injury
10. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AIF | apoptosis-inducing factor |
| AA | arachidonic acid |
| aCDase | acid ceramidase |
| AGEs | advanced glycation endproducts |
| Ang II | angiotensin II |
| apoM | apolipoprotein M |
| AR | adenosine receptors |
| aSMase | acid sphingomyelinase |
| AdipoR | adiponectin receptor |
| alCDase | alkaline CDase |
| anti-GBM Ab GN | anti-glomerular basement membrane antibody-induced glomerulonephritis |
| AMK | AMP-activated protein kinase |
| BMMC | bone marrow-derived mononuclear cells |
| BPGM | bisphosphoglycerate mutase |
| CAMP | cyclic adenosine monophosphate |
| CCl4 | carbon tetrachloride |
| CDDO | methyl-2-cyano-3,12-dioxooleano-1,9-dien-28-oate |
| Cer | ceramide |
| CerK | Cer kinase |
| CerS | Cer synthase |
| CDase | ceramidase |
| C1P | Cer-1-phosphate |
| C1PP | C1P phosphatase |
| C1PTP | C1P ransport protein |
| CERT | ceramide transport protein |
| CGN | chronic glomerulonephritis |
| CKD | chronic kidney disease |
| CK2α | casein kinase 2α |
| COX-2 | cyclooxygenase-2 |
| cPLA2 | cytosolic phospholipase A2 |
| CREB | CAMP response element-binding protein |
| CTGF | connective tissue growth factor |
| Cyto C | cytochrome C |
| DES | dihydroceramide desaturase |
| dhCer | dihydroceramide |
| dhSph | dihydrosphingosine |
| DM | diabetes mellitus |
| DN | diabetic nephropathy |
| EA1P | ethanolamine 1-phosphate |
| EC | endothelial cell |
| ER | endoplasmic reticulum |
| ERK | extracellular signal-regulated kinase |
| eNOS | endothelial nitric oxide synthase |
| FSGS | focal segmental glomerulosclerosis |
| FPE | foot process effacement |
| GCase | glycosidase |
| GlcCer | glucosylceramide |
| GCSs | glycosylceramide synthases |
| GEC | glomerular EC |
| GSLs | glycosphingolipids |
| GPx | glutathione peroxidase |
| GSSG | glutathione disulfide |
| GSH | glutathione |
| GST | glutathione s-transferase |
| GTPase | guanosine triphosphatase |
| HD | hexadecenal |
| H2O2 | hydrogen peroxide |
| HIF | hypoxia-inducible factor |
| H/R | hypoxia/reoxygenation |
| HO-1 | heme oxygenase-1 |
| HREs | HIF-responsive-elements |
| HSP | heat-shock protein |
| ICAM-1 | intercellular adhesion molecule-1 |
| IGF | insulin growth factor |
| IGF-IIR | IGF-II receptor |
| IGFBP-3 | IGF binding protein-3 |
| iNOS | inducible nitric oxide synthase |
| IL | interleukin |
| I/R | ischemia/reperfusion |
| JNK | Jun N-terminal protein kinase |
| 3-KdhSph | 3-ketodihydrosphingosine |
| 3-KR | 3-KdhSph reductase |
| LIMK-1 | LIM kinase-1 |
| LN | lupus nephritis |
| LOX-1 | oxLDL receptor-1 |
| LPS | lipopolysaccharide |
| LRs | lipid rafts |
| MAPK | mitogen-activated protein kinase |
| MC | mesangial cell |
| MAM | mitochondria-associated membrane |
| MA-nSMase | mitochondria-associated nSMase |
| MCLR | microcystin-LR |
| MCP-1 | monocyte chemotactic protein-1 |
| MEKK | mitogen-activated protein kinase kinase kinase |
| MME | mesangial matrix expansion |
| MMP | mitochondrial membrane potential |
| MMP-9 | matrix metalloproteinase-9 |
| MOMP | mitochondrial outer membrane permeability |
| M6P | mannose 6-phosphate |
| MPTP | mitochondrial transition pore |
| MRCC | mitochondrial respiratory chain complex |
| M6P/IGF-IIR | M6P/insulin-like growth factor II receptor |
| NAC | N-acetylcysteine |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| nCDase | neutral CDase |
| Nfr-2 | nuclear factor-erythroid 2-related factor-2 |
| NO | nitric oxide |
| NOX | NADPH oxidase |
| NF-κB | nuclear factor-κB |
| nSMase | neutral SMase |
| NS | nephrotic syndrome |
| OxLDL | oxidized low-density lipoprotein |
| PI3K | phosphatidylinositol3-kinase |
| PDGF | platelet-derived growth factor |
| PK | protein kinase |
| PLA2 | phospholipase A2 |
| PP2A | protein phosphatase 2A |
| PHB2 | prohibitin 2 |
| ROS | reactive oxygen species |
| RTC | renal tubular cell |
| SAPK | stress-activated protein kinase |
| SGK-1 | serum- and glucocorticoid-inducible protein kinase-1 |
| SLE | systemic lupus erythematosus |
| SLs | sphingolipids |
| SM | sphingomyelin |
| SMPDL3b | sphingomyelin phosphodiesterase acid-like 3b |
| SMS | SM synthase |
| SOD | superoxide dismutase |
| Sph | sphingosine |
| SphKs | sphingosine kinases |
| Spns2 | spinster homologue 2 |
| S1P | sphingosine-1-phosphate |
| S1PL | S1P lyase |
| S1PP | S1P phosphatase |
| S1PRs | S1P receptors |
| SPT | serine palmitoyltransferase |
| STAT | signal transducer and activator of transcription |
| TIMP-1 | tissue inhibitor of matrix metalloproteinase-1 |
| TNF | tumor necrosis factor |
| TRAIL | TNF-related apoptosis inducing ligand |
| UUO | unilateral ureteral obstruction |
| UV | ultraviolet |
| VDAC | voltage-dependent anion channel |
| VEGF | vascular endothelial growth factor |
| VSMC | vascular smooth muscle cell |
References
- Sedeek, M.; Nasrallah, R.; Touyz, R.M.; Hébert, R.L. NADPH oxidases, reactive oxygen species, and the kidney: Friend and foe. J. Am. Soc. Nephrol. 2013, 24, 1512–1518. [Google Scholar] [CrossRef] [PubMed]
- Ratliff, B.B.; Abdulmahdi, W.; Pawar, R.; Wolin, M.S. Oxidant mechanisms in renal injury and disease. Antioxid. Redox Signal. 2016, 25, 119–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Brocklyn, J.R.; Williams, J.B. The control of the balance between ceramide and sphingosine-1-phosphate by sphingosine kinase: Oxidative stress and the seesaw of cell survival and death. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2012, 163, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Ueda, N. Ceramide-induced apoptosis in renal tubular cells: A role of mitochondria and sphingosine-1-phoshate. Int. J. Mol. Sci. 2015, 16, 5076–5124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ueda, N. Sphingolipids in genetic and acquired forms of chronic kidney diseases. Curr. Med. Chem. 2017, 24, 1238–1275. [Google Scholar] [CrossRef]
- Bhat, O.M.; Yuan, X.; Li, G.; Lee, R.M.; Li, P.L. Sphingolipids and redox signaling in renal regulation and chronic kidney diseases. Antioxid. Redox Signal. 2018, 28, 1008–1026. [Google Scholar] [CrossRef]
- Rojas-Rivera, J.; Ortiz, A.; Egido, J. Antioxidants in kidney diseases: The impact of bardoxolone methyl. Int. J. Nephrol. 2012, 2012, 321714. [Google Scholar] [CrossRef] [Green Version]
- Gyurászová, M.; Gurecká, R.; Bábíčková, J.; Tóthová, L. Oxidative stress in the pathophysiology of kidney disease: Implications for noninvasive monitoring and identification of biomarkers. Oxid. Med. Cell. Longev. 2020, 2020, 5478708. [Google Scholar] [CrossRef] [Green Version]
- Hannun, Y.A.; Obeid, L.M. Sphingolipids and their metabolism in physiology and disease. Nat. Rev. Mol. Cell. Biol. 2018, 19, 175–191. [Google Scholar] [CrossRef]
- Lone, M.A.; Hülsmeier, A.J.; Saied, E.M.; Karsai, G.; Arenz, C.; von Eckardstein, A.; Hornemann, T. Subunit composition of the mammalian serine-palmitoyltransferase defines the spectrum of straight and methyl-branched long-chain bases. Proc. Natl. Acad. Sci. USA 2020, 117, 15591–15598. [Google Scholar] [CrossRef]
- Vasiliauskaité-Brooks, I.; Healey, R.D.; Rochaix, P.; Saint-Paul, J.; Sounier, R.; Grison, C.; Waltrich-Augusto, T.; Fortier, M.; Hoh, F.; Saied, E.M.; et al. Structure of a human intramembrane ceramidase explains enzymatic dysfunction found in leukodystrophy. Nat. Commun. 2018, 9, 5437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vasiliauskaité-Brooks, I.; Sounier, R.; Rochaix, P.; Bellot, G.; Fortier, M.; Hoh, F.; De Colibus, L.; Bechara, C.; Saied, E.M.; Arenz, C.; et al. Structural insights into adiponectin receptors suggest ceramidase activity. Nature 2017, 544, 120–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abou Daher, A.; Francis, M.; Azzam, P.; Ahmad, A.; Eid, A.A.; Fornoni, A.; Marples, B.; Zeidan, Y.H. Modulation of radiation-induced damage of human glomerular endothelial cells by SMPDL3B. FASEB J. 2020, 34, 7915–7926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Batheja, A.D.; Uhlinger, D.J.; Carton, J.M.; Ho, G.; D’Andrea, M.R. Characterization of serine palmitoyltransferase in normal human tissues. J. Histochem. Cytochem. 2003, 51, 687–696. [Google Scholar] [CrossRef] [Green Version]
- Mallela, S.K.; Mitrofanova, A.; Merscher, S.; Fornoni, A. Regulation of the amount of ceramide-1-phosphate synthesized in differentiated human podocytes. Biochim. Biophys. Acta Mol. Cell. Biol. Lipids 2019, 1864, 158517. [Google Scholar] [CrossRef]
- Causeret, C.; Geeraert, L.; Van der Hoeven, G.; Mannaerts, G.P.; Van Veldhoven, P.P. Further characterization of rat dihydroceramide desaturase: Tissue distribution, subcellular localization, and substrate specificity. Lipids 2000, 35, 1117–1125. [Google Scholar] [CrossRef]
- Alsanafi, M.; Kelly, S.L.; Jubair, K.; McNaughton, M.; Tate, R.J.; Merrill, A.H., Jr.; Pyne, S.; Pyne, N.J. Native and polyubiquitinated forms of dihydroceramide desaturase are differentially linked to human embryonic kidney cell survival. Mol. Cell. Biol. 2018, 38, e00222-18. [Google Scholar] [CrossRef] [Green Version]
- Sudarkina, O.Y.; Filippenkov, I.B.; Brodsky, H.B.; Limborska, S.A.; Dergunova, L.V. Comparative analysis of sphingomyelin synthase 1 gene expression at the transcriptional and translational levels in human tissues. Mol. Cell. Biochem. 2015, 406, 91–99. [Google Scholar] [CrossRef]
- Sugimoto, M.; Wakabayashi, M.; Shimizu, Y.; Yoshioka, T.; Higashino, K.; Numata, Y.; Okuda, T.; Zhao, S.; Sakai, S.; Igarashi, Y.; et al. Imaging mass spectrometry reveals acyl-chain- and region-specific sphingolipid metabolism in the kidneys of sphingomyelin synthase 2-deficient mice. PLoS ONE 2016, 11, e0152191. [Google Scholar] [CrossRef] [Green Version]
- Favale, N.O.; Santacreu, B.J.; Pescio, L.G.; Marquez, M.G.; Sterin-Speziale, N.B. Sphingomyelin metabolism is involved in the differentiation of MDCK cells induced by environmental hypertonicity. J. Lipid Res. 2015, 56, 786–800. [Google Scholar]
- Zhang, P.; Hua, L.; Hou, H.; Du, X.; He, Z.; Liu, M.; Hu, X.; Yan, N. Sphingomyelin synthase 2 promotes H2O2-induced endothelial dysfunction by activating the Wnt/β-catenin signaling pathway. Int. J. Mol. Med. 2018, 42, 3344–3354. [Google Scholar] [CrossRef] [PubMed]
- Zager, R.A.; Conrad, S.; Lochhead, K.; Sweeney, E.A.; Igarashi, Y.; Burkhart, K.M. Altered sphingomyelinase and ceramide expression in the setting of ischemic and nephrotoxic acute renal failure. Kidney Int. 1998, 53, 573–582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, R.; Patni, H.; Tandon, P.; Luan, L.; Sharma, B.; Salhan, D.; Saleem, M.A.; Mathieson, P.W.; Malhotra, A.; Husain, M.; et al. Nef interaction with actin compromises human podocyte actin cytoskeletal integrity. Exp. Mol. Pathol. 2013, 94, 51–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huwiler, A.; Pfeilschifter, J.; van den Bosch, H. Nitric oxide donors induce stress signaling via ceramide formation in rat renal mesangial cells. J. Biol. Chem. 1999, 274, 7190–7195. [Google Scholar] [CrossRef] [Green Version]
- Franzen, R.; Fabbro, D.; Aschrafi, A.; Pfeilschifter, J.; Huwiler, A. Nitric oxide induces degradation of the neutral ceramidase in rat renal mesangial cells and is counterregulated by protein kinase C. J. Biol. Chem. 2002, 277, 46184–46190. [Google Scholar] [CrossRef] [Green Version]
- Boini, K.M.; Xia, M.; Li, C.; Zhang, C.; Payne, L.P.; Abais, J.M.; Poklis, J.L.; Hylemon, P.B.; Li, P.L. Acid sphingomyelinase gene deficiency ameliorates the hyperhomocysteinemia-induced glomerular injury in mice. Am. J. Pathol. 2011, 179, 2210–2219. [Google Scholar] [CrossRef]
- Chatterjee, M.; Wu, S. Cell line dependent involvement of ceramide in ultraviolet light-induced apoptosis. Mol. Cell. Biochem. 2001, 219, 21–27. [Google Scholar] [CrossRef]
- Ghosh, P.; Chatterjee, S. Effects of gentamicin on sphingomyelinase activity in cultured human renal proximal tubular cells. J. Biol. Chem. 1987, 262, 12550–12556. [Google Scholar] [CrossRef]
- Boini, K.M.; Zhang, C.; Xia, M.; Han, W.Q.; Brimson, C.; Poklis, J.L.; Li, P.L. Visfatin-induced lipid raft redox signaling platforms and dysfunction in glomerular endothelial cells. Biochim. Biophys. Acta 2010, 1801, 1294–1304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Q.; Band, M.R.; Hernandez, A.; Liu, Z.L.; Kummerow, F.A. 27-Hydroxycholesterol inhibits neutral sphingomyelinase in cultured human endothelial cells. Life Sci. 2004, 75, 1567–1577. [Google Scholar] [CrossRef] [PubMed]
- Ichi, I.; Kamikvawa, C.; Nakagawa, T.; Kobayashi, K.; Kataoka, R.; Nagata, E.; Kitamura, Y.; Nakazaki, C.; Matsura, T.; Kojo, S. Neutral sphingomyelinase-induced ceramide accumulation by oxidative stress during carbon tetrachloride intoxication. Toxicology 2009, 261, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Kaszkin, M.; Huwiler, A.; Scholz, K.; van den Bosch, H.; Pfeilschifter, J. Negative regulation of interleukin-1β-activated neutral sphingomyelinase by protein kinase C in rat mesangial cells. FEBS Lett. 1998, 440, 163–166. [Google Scholar] [CrossRef] [Green Version]
- Martín, S.F.; Sawai, H.; Villalba, J.M.; Hannun, Y.A. Redox regulation of neutral sphingomyelinase-1 activity in HEK293 cells through a GSH-dependent mechanism. Arch. Biochem. Biophys. 2007, 459, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Camaré, C.; Augé, N.; Pucelle, M.; Saint-Lebes, B.; Grazide, M.H.; Nègre-Salvayre, A.; Salvayre, R. The neutral sphingomyelinase-2 is involved in angiogenic signaling triggered by oxidized LDL. Free Radic. Biol. Med. 2016, 93, 204–216. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Mitrofanova, A.; Bielawski, J.; Yang, Y.; Marples, B.; Fornoni, A.; Zeidan, Y.H. Sphingomyelinase-like phosphodiesterase 3b mediates radiation-induced damage of renal podocytes. FASEB J. 2017, 31, 771–780. [Google Scholar] [CrossRef] [Green Version]
- Sugiura, M.; Kono, K.; Liu, H.; Shimizugawa, T.; Minekura, H.; Spiegel, S.; Kohama, T. Ceramide kinase, a novel lipid kinase. Molecular cloning and functional characterization. J. Biol. Chem. 2002, 277, 23294–23300. [Google Scholar]
- Pastukhov, O.; Schwalm, S.; Römer, I.; Zangemeister-Wittke, U.; Pfeilschifter, J.; Huwiler, A. Ceramide kinase contributes to proliferation but not to prostaglandin E2 formation in renal mesangial cells and fibroblasts. Cell. Physiol. Biochem. 2014, 34, 119–133. [Google Scholar] [CrossRef] [Green Version]
- Sridevi, P.; Alexander, H.; Laviad, E.L.; Pewzner-Jung, Y.; Hannink, M.; Futerman, A.H.; Alexander, S. Ceramide synthase 1 is regulated by proteasomal mediated turnover. Biochim. Biophys. Acta 2009, 1793, 1218–1227. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Pan, Y.; Bian, Z.; Chen, P.; Zhu, S.; Gu, H.; Guo, L.; Hu, C. Ceramide production mediates aldosterone-induced human umbilical vein endothelial cell (HUVEC) damages. PLoS ONE 2016, 11, e0146944. [Google Scholar] [CrossRef]
- Mullen, T.D.; Hannun, Y.A.; Obeid, L.M. Ceramide synthases at the centre of sphingolipid metabolism and biology. Biochem. J. 2012, 441, 789–802. [Google Scholar] [CrossRef] [Green Version]
- Pescio, L.G.; Santacreu, B.J.; Lopez, V.G.; Paván, C.H.; Romero, D.J.; Favale, N.O.; Sterin-Speziale, N.B. Changes in ceramide metabolism are essential in Madin-Darby canine kidney cell differentiation. J. Lipid Res. 2017, 58, 1428–1438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lahiri, S.; Futerman, A.H. LASS5 is a bona fide dihydroceramide synthase that selectively utilizes palmitoyl-CoA as acyl donor. J. Biol. Chem. 2005, 280, 33735–33738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, S.R.; Lim, J.H.; Kim, M.Y.; Kim, E.N.; Kim, Y.; Choi, B.S.; Kim, Y.S.; Kim, H.W.; Lim, K.M.; Kim, M.J.; et al. Adiponectin receptor agonist AdipoRon decreased ceramide, and lipotoxicity, and ameliorated diabetic nephropathy. Metabolism 2018, 85, 348–360. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Kidd, J.; Kaspar, C.; Dempsey, S.; Bhat, O.M.; Camus, S.; Ritter, J.K.; Gehr, T.W.B.; Gulbins, E.; Li, P.L. Podocytopathy and nephrotic syndrome in mice with podocyte-specific deletion of the asah1 gene: Role of ceramide accumulation in glomeruli. Am. J. Pathol. 2020, 190, 1211–1223. [Google Scholar] [CrossRef]
- Mitsutake, S.; Tani, M.; Okino, N.; Mori, K.; Ichinose, S.; Omori, A.; Iida, H.; Nakamura, T.; Ito, M. Purification, characterization, molecular cloning, and subcellular distribution of neutral ceramidase of rat kidney. J. Biol. Chem. 2001, 276, 26249–26259. [Google Scholar] [CrossRef] [Green Version]
- Geoffroy, K.; Troncy, L.; Wiernsperger, N.; Lagarde, M.; El Bawab, S. Glomerular proliferation during early stages of diabetic nephropathy is associated with local increase of sphingosine-1-phosphate levels. FEBS Lett. 2005, 579, 1249–1254. [Google Scholar] [CrossRef] [Green Version]
- Geoffroy, K.; Wiernsperger, N.; Lagarde, M.; El Bawab, S. Bimodal effect of advanced glycation end products on mesangial cell proliferation is mediated by neutral ceramidase regulation and endogenous sphingolipids. J. Biol. Chem. 2004, 279, 34343–34352. [Google Scholar] [CrossRef] [Green Version]
- Coroneos, E.; Martinez, M.; McKenna, S.; Kester, M. Differential regulation of sphingomyelinase and ceramidase activities by growth factors and cytokines. Implications for cellular proliferation and differentiation. J. Biol. Chem. 1995, 270, 23305–23309. [Google Scholar] [CrossRef] [Green Version]
- Facchinetti, M.M.; Leocata Nieto, F.; Márquez, M.G.; Sterin-Speziale, N. Stratification of sphingosine kinase-1 expression and activity in rat kidney. Cells Tissues Organs 2008, 188, 384–392. [Google Scholar] [CrossRef]
- Igarashi, N.; Okada, T.; Hayashi, S.; Fujita, T.; Jahangeer, S.; Nakamura, S. Sphingosine kinase 2 is a nuclear protein and inhibits DNA synthesis. J. Biol. Chem. 2003, 278, 46832–46839. [Google Scholar] [CrossRef] [Green Version]
- Awad, A.S.; Rouse, M.D.; Khutsishvili, K.; Huang, L.; Bolton, W.K.; Lynch, K.R.; Okusa, M.D. Chronic sphingosine 1-phosphate 1 receptor activation attenuates early-stage diabetic nephropathy independent of lymphocytes. Kidney Int. 2011, 79, 1090–1098. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, S.; Xin, C.; Pfeilschifter, J.; Huwiler, A. A novel mode of action of the putative sphingosine kinase inhibitor 2-(p-hydroxyanilino)-4-(p-chlorophenyl) thiazole (SKI II): Induction of lysosomal sphingosine kinase 1 degradation. Cell. Physiol. Biochem. 2010, 26, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Beyer, S.; Schwalm, S.; Pfeilschifter, J.; Huwiler, A. Renal mesangial cells isolated from sphingosine kinase 2 transgenic mice show reduced proliferation and are more sensitive to stress-induced apoptosis. Cell. Physiol. Biochem. 2018, 47, 2522–2533. [Google Scholar] [CrossRef]
- Zhu, X.; Shi, D.; Cao, K.; Ru, D.; Ren, J.; Rao, Z.; Chen, Y.; You, Q.; Dai, C.; Liu, L.; et al. Sphingosine kinase 2 cooperating with Fyn promotes kidney fibroblast activation and fibrosis via STAT3 and AKT. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 3824–3836. [Google Scholar] [CrossRef] [PubMed]
- Billich, A.; Bornancin, F.; Dévay, P.; Mechtcheriakova, D.; Urtz, N.; Baumruker, T. Phosphorylation of the immunomodulatory drug FTY720 by sphingosine kinases. J. Biol. Chem. 2003, 278, 47408–47415. [Google Scholar] [CrossRef] [Green Version]
- Bionda, C.; Portoukalian, J.; Schmitt, D.; Rodriguez-Lafrasse, C.; Ardail, D. Subcellular compartmentalization of ceramide metabolism: MAM (mitochondria-associated membrane) and/or mitochondria? Biochem. J. 2004, 382, 527–533. [Google Scholar] [CrossRef] [Green Version]
- Novgorodov, S.A.; Wu, B.X.; Gudz, T.I.; Bielawski, J.; Ovchinnikova, T.V.; Hannun, Y.A.; Obeid, L.M. Novel pathway of ceramide production in mitochondria: Thioesterase and neutral ceramidase produce ceramide from sphingosine and acyl-CoA. J. Biol. Chem. 2011, 286, 25352–25362. [Google Scholar] [CrossRef] [Green Version]
- Rajagopalan, V.; Canals, D.; Luberto, C.; Snider, J.; Voelkel-Johnson, C.; Obeid, L.M.; Hannun, Y.A. Critical determinants of mitochondria-associated neutral sphingomyelinase (MA-nSMase) for mitochondrial localization. Biochim. Biophys. Acta 2015, 1850, 628–639. [Google Scholar] [CrossRef] [Green Version]
- Wei, J.; Yerokun, T.; Leipelt, M.; Haynes, C.A.; Radhakrishna, H.; Momin, A.; Kelly, S.; Park, H.; Wang, E.; Carton, J.M.; et al. Serine palmitoyltransferase subunit 1 is present in the endoplasmic reticulum, nucleus and focal adhesions, and functions in cell morphology. Biochim. Biophys. Acta 2009, 1791, 746–756. [Google Scholar] [CrossRef] [Green Version]
- Jain, A.; Beutel, O.; Ebell, K.; Korneev, S.; Holthuis, J.C. Diverting CERT-mediated ceramide transport to mitochondria triggers Bax-dependent apoptosis. J. Cell. Sci. 2017, 130, 360–371. [Google Scholar] [CrossRef] [Green Version]
- Olivera, A.; Kohama, T.; Tu, Z.; Milstien, S.; Spiegel, S. Purification and characterization of rat kidney sphingosine kinase. J. Biol. Chem. 1998, 273, 12576–12583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, W.K.; Torchalski, B.; Thévenod, F. Cadmium-induced ceramide formation triggers calpain-dependent apoptosis in cultured kidney proximal tubule cells. Am. J. Physiol. Cell Physiol. 2007, 293, C839–C847. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, J.Q.; Liu, C.M.; Yang, W. Protective effect of rutin against carbon tetrachloride-induced oxidative stress, inflammation and apoptosis in mouse kidney associated with the ceramide, MAPKs, p53 and calpain activities. Chem. Biol. Interact. 2018, 286, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.L.; Chiou, S.H.; Tai, W.C.; Joseph, N.A.; Chow, K.C. Trivalent chromium induces autophagy by activating sphingomyelin phosphodiesterase 2 and increasing cellular ceramide levels in renal HK2 cells. Mol. Carcinog. 2017, 56, 2424–2433. [Google Scholar] [CrossRef]
- Siskind, L.J.; Mullen, T.D.; Romero Rosales, K.; Clarke, C.J.; Hernandez-Corbacho, M.J.; Edinger, A.L.; Obeid, L.M. The BCL-2 protein BAK is required for long-chain ceramide generation during apoptosis. J. Biol. Chem. 2010, 28, 11818–11826. [Google Scholar] [CrossRef] [Green Version]
- Dupre, T.V.; Doll, M.A.; Shah, P.P.; Sharp, C.N.; Siow, D.; Megyesi, J.; Shayman, J.; Bielwska, A.; Bielawski, J.; Beverly, L.J.; et al. Inhibiting glucosylceramide synthase exacerbates cisplatin-induced acute kidney injury. J. Lipid Res. 2017, 58, 1439–1452. [Google Scholar] [CrossRef] [Green Version]
- Dahdouh, F.; Raane, M.; Thévenod, F.; Lee, W.K. Nickel-induced cell death and survival pathways in cultured renal proximal tubule cells: Roles of reactive oxygen species, ceramide and ABCB1. Arch. Toxicol. 2014, 88, 881–892. [Google Scholar] [CrossRef]
- Bonnaud, S.; Niaudet, C.; Pottier, G.; Gaugler, M.H.; Millour, J.; Barbet, J.; Sabatier, L.; Paris, F. Sphingosine-1-phosphate protects proliferating endothelial cells from ceramide-induced apoptosis but not from DNA damage-induced mitotic death. Cancer Res. 2007, 67, 1803–1811. [Google Scholar] [CrossRef] [Green Version]
- Niaudet, C.; Bonnaud, S.; Guillonneau, M.; Gouard, S.; Gaugler, M.H.; Dutoit, S.; Ripoche, N.; Dubois, N.; Trichet, V.; Corre, I.; et al. Plasma membrane reorganization links acid sphingomyelinase/ceramide to p38 MAPK pathways in endothelial cells apoptosis. Cell. Signal. 2017, 33, 10–21. [Google Scholar] [CrossRef]
- Wortel, R.C.; Mizrachi, A.; Li, H.; Markovsky, E.; Enyedi, B.; Jacobi, J.; Brodsky, O.; Cao, J.; Lippert, A.R.; Incrocci, L.; et al. Sildenafil protects endothelial cells from radiation-induced oxidative stress. J. Sex Med. 2019, 16, 1721–1733. [Google Scholar] [CrossRef] [Green Version]
- Itoh, Y.; Yano, T.; Sendo, T.; Sueyasu, M.; Hirano, K.; Kanaide, H.; Oishi, R. Involvement of de novo ceramide synthesis in radiocontrast-induced renal tubular cell injury. Kidney Int. 2006, 69, 288–297. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cao, L.C.; Honeyman, T.; Jonassen, J.; Scheid, C. Oxalate-induced ceramide accumulation in Madin-Darby canine kidney and LLC-PK1 cells. Kidney Int. 2000, 57, 2403–2411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miller, C.; Kennington, L.; Cooney, R.; Kohjimoto, Y.; Cao, L.C.; Honeyman, T.; Pullman, J.; Jonassen, J.; Scheid, C. Oxalate toxicity in renal epithelial cells: Characteristics of apoptosis and necrosis. Toxicol. Appl. Pharmacol. 2000, 162, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Yi, F.; Zhang, A.Y.; Li, N.; Muh, R.W.; Fillet, M.; Renert, A.F.; Li, P.L. Inhibition of ceramide-redox signaling pathway blocks glomerular injury in hyperhomocysteinemic rats. Kidney Int. 2006, 70, 88–96. [Google Scholar] [CrossRef] [Green Version]
- Boini, K.M.; Xia, M.; Abais, J.M.; Xu, M.; Li, C.X.; Li, P.L. Acid sphingomyelinase gene knockout ameliorates hyperhomocysteinemic glomerular injury in mice lacking cystathionine-β-synthase. PLoS ONE 2012, 7, e45020. [Google Scholar] [CrossRef]
- Ueda, N.; Kaushal, G.P.; Hong, X.; Shah, S.V. Role of enhanced ceramide generation in DNA damage and cell death in chemical hypoxic injury to LLC-PK1 cells. Kidney Int. 1998, 54, 399–406. [Google Scholar] [CrossRef] [Green Version]
- Ueda, N.; Camargo, S.M.; Hong, X.; Basnakian, A.G.; Walker, P.D.; Shah, S.V. Role of ceramide synthase in oxidant injury to renal tubular epithelial cells. J. Am. Soc. Nephrol. 2001, 12, 2384–2391. [Google Scholar] [CrossRef]
- Basnakian, A.G.; Ueda, N.; Hong, X.; Galitovsky, V.E.; Yin, X.; Shah, S.V. Ceramide synthase is essential for endonuclease-mediated death of renal tubular epithelial cells induced by hypoxia-reoxygenation. Am. J. Physiol. Renal Physiol. 2005, 288, F308–F314. [Google Scholar] [CrossRef]
- Therade-Matharan, S.; Laemmel, E.; Carpentier, S.; Obata, Y.; Levade, T.; Duranteau, J.; Vicaut, E. Reactive oxygen species production by mitochondria in endothelial cells exposed to reoxygenation after hypoxia and glucose depletion is mediated by ceramide. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 289, R1756–R1762. [Google Scholar] [CrossRef] [Green Version]
- Malik, R.K.; Thornhill, B.A.; Chang, A.Y.; Kiley, S.C.; Chevalier, R.L. Renal apoptosis parallels ceramide content after prolonged ureteral obstruction in the neonatal rat. Am. J. Physiol. Renal Physiol. 2001, 281, F56–F61. [Google Scholar] [CrossRef]
- Verdoorn, K.S.; Lindoso, R.S.; Lowe, J.; Lara, L.S.; Vieyra, A.; Einicker-Lamas, M. Bone marrow mononuclear cells shift bioactive lipid pattern in injured kidney towards tissue repair in rats with unilateral ureteral obstruction. Nephrol. Dial. Transplant. 2010, 25, 3867–3874. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eckes, T.; Trautmann, S.; Djudjaj, S.; Beyer, S.; Patyna, S.; Schwalm, S.; Gauer, S.; Thomas, D.; Schaefer, L.; Boor, P.; et al. Consistent alteration of chain length-specific ceramides in human and mouse fibrotic kidneys. Biochim. Biophys. Acta Mol. Cell. Biol. Lipids 2021, 1866, 15882. [Google Scholar] [CrossRef] [PubMed]
- Prasad, R.; Hadjidemetriou, I.; Maharaj, A.; Meimaridou, E.; Buonocore, F.; Saleem, M.; Hurcombe, J.; Bierzynska, A.; Barbagelata, E.; Bergadá, I.; et al. Sphingosine-1-phosphate lyase mutations cause primary adrenal insufficiency and steroid-resistant nephrotic syndrome. J. Clin. Investig. 2017, 127, 942–953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lovric, S.; Goncalves, S.; Gee, H.Y.; Oskouian, B.; Srinivas, H.; Choi, W.I.; Shril, S.; Ashraf, S.; Tan, W.; Rao, J.; et al. Mutations in sphingosine-1-phosphate lyase cause nephrosis with ichthyosis and adrenal insufficiency. J. Clin. Investig. 2017, 127, 912–928. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, G.; Han, F.; Yang, Y.; Xie, Y.; Jiang, H.; Mao, Y.; Wang, H.; Wang, M.; Chen, R.; Yang, J.; et al. Evaluation of sphingolipid metabolism in renal cortex of rats with streptozotocin-induced diabetes and the effects of rapamycin. Nephrol. Dial. Transplant. 2011, 26, 1493–1502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woo, C.Y.; Baek, J.Y.; Kim, A.R.; Hong, C.H.; Yoon, J.E.; Kim, H.S.; Yoo, H.J.; Park, T.S.; Kc, R.; Lee, K.U.; et al. Inhibition of ceramide accumulation in podocytes by myriocin prevents diabetic nephropathy. Diabetes Metab. J. 2020, 44, 581–591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, M.; Huang, S.; Duan, W.; Liu, Q.; Lei, M. Inhibition of acid sphingomyelinase activity ameliorates endothelial dysfunction in db/db mice. Biosci. Rep. 2019, 39, BSR20182144. [Google Scholar] [CrossRef] [Green Version]
- Luo, Y.; Lei, M. α-Mangostin protects against high-glucose induced apoptosis of human umbilical vein endothelial cells. Biosci. Rep. 2017, 37, BSR20170779. [Google Scholar] [CrossRef] [Green Version]
- Chun, L.; Junlin, Z.; Aimin, W.; Niansheng, L.; Benmei, C.; Minxiang, L. Inhibition of ceramide synthesis reverses endothelial dysfunction and atherosclerosis in streptozotocin-induced diabetic rats. Diabetes Res. Clin. Pract. 2011, 93, 77–85. [Google Scholar] [CrossRef]
- Mitsnefes, M.; Scherer, P.E.; Friedman, L.A.; Gordillo, R.; Furth, S.; Warady, B.A.; CKiD Study Group. Ceramides and cardiac function in children with chronic kidney disease. Pediatr. Nephrol. 2014, 29, 415–422. [Google Scholar] [CrossRef] [Green Version]
- Mantovani, A.; Lunardi, G.; Bonapace, S.; Dugo, C.; Altomari, A.; Molon, G.; Conti, A.; Bovo, C.; Laaksonen, R.; Byrne, C.D.; et al. Association between increased plasma ceramides and chronic kidney disease in patients with and without ischemic heart disease. Diabetes Metab. 2021, 47, 101152. [Google Scholar] [CrossRef] [PubMed]
- Patyna, S.; Büttner, S.; Eckes, T.; Obermüller, N.; Bartel, C.; Braner, A.; Trautmann, S.; Thomas, D.; Geiger, H.; Pfeilschifter, J.; et al. Blood ceramides as novel markers for renal impairment in systemic lupus erythematosus. Prostaglandins Other Lipid Mediat. 2019, 144, 106348. [Google Scholar] [CrossRef] [PubMed]
- Checa, A.; Idborg, H.; Zandian, A.; Sar, D.G.; Surowiec, I.; Trygg, J.; Svenungsson, E.; Jakobsson, P.J.; Nilsson, P.; Gunnarsson, I.; et al. Dysregulations in circulating sphingolipids associate with disease activity indices in female patients with systemic lupus erythematosus: A cross-sectional study. Lupus 2017, 26, 1023–1033. [Google Scholar] [CrossRef] [PubMed]
- Snider, A.J.; Ruiz, P.; Obeid, L.M.; Oates, J.C. Inhibition of sphingosine kinase-2 in a murine model of lupus nephritis. PLoS ONE 2013, 8, e53521. [Google Scholar]
- Oates, J.C.; Mashmoushi, A.K.; Shaftman, S.R.; Gilkeson, G.S. NADPH oxidase and nitric oxide synthase-dependent superoxide production is increased in proliferative lupus nephritis. Lupus 2013, 22, 1361–1370. [Google Scholar] [CrossRef] [Green Version]
- Fox, T.E.; Bewley, M.C.; Unrath, K.A.; Pedersen, M.M.; Anderson, R.E.; Jung, D.Y.; Jefferson, L.S.; Kim, J.K.; Bronson, S.K.; Flanagan, J.M.; et al. Circulating sphingolipid biomarkers in models of type 1 diabetes. J. Lipid Res. 2011, 52, 509–517. [Google Scholar] [CrossRef] [Green Version]
- Kim, M.; Jeong, H.; Lee, B.; Cho, Y.; Yoon, W.K.; Cho, A.; Kwon, G.; Nam, K.T.; Ha, H.; Lim, K.M. Enrichment of short-chain ceramides and free fatty acids in the skin epidermis, liver, and kidneys of db/db mice, a type 2 diabetes mellitus model. Biomol. Ther. 2019, 27, 457–465. [Google Scholar] [CrossRef] [Green Version]
- Sas, K.M.; Nair, V.; Byun, J.; Kayampilly, P.; Zhang, H.; Saha, J.; Brosius, F.C., 3rd; Kretzler, M.; Pennathur, S. Targeted lipidomic and transcriptomic analysis identifies dysregulated renal ceramide metabolism in a mouse model of diabetic kidney disease. J. Proteom. Bioinform. 2015, (Suppl. 14), 002. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.J.; Ghosh, S.; Kovalik, J.P.; Ching, J.; Choi, H.W.; Tavintharan, S.; Ong, C.N.; Sum, C.F.; Summers, S.A.; Tai, E.S.; et al. Profiling of plasma metabolites suggests altered mitochondrial fuel usage and remodeling of sphingolipid metabolism in individuals with type 2 diabetes and kidney disease. Kidney Int. Rep. 2016, 2, 470–480. [Google Scholar] [CrossRef] [Green Version]
- Ichi, I.; Nakahara, K.; Fujii, K.; Iida, C.; Miyashita, Y.; Kojo, S. Increase of ceramide in the liver and plasma after carbon tetrachloride intoxication in the rat. J. Nutr. Sci. Vitaminol. 2007, 53, 53–56. [Google Scholar] [CrossRef] [Green Version]
- Klein, R.L.; Hammad, S.M.; Baker, N.L.; Hunt, K.J.; Al Gadban, M.M.; Cleary, P.A.; Virella, G.; Lopes-Virella, M.F. DCCT/EDIC Research Group. Decreased plasma levels of select very long chain ceramide species are associated with the development of nephropathy in type 1 diabetes. Metabolism 2014, 63, 1287–1295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magagnotti, C.; Zerbini, G.; Fermo, I.; Carletti, R.M.; Bonfanti, R.; Vallone, F.; Andolfo, A. Identification of nephropathy predictors in urine from children with a recent diagnosis of type 1 diabetes. J. Proteomics 2019, 193, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Morita, Y.; Kurano, M.; Sakai, E.; Nishikawa, T.; Nishikawa, M.; Sawabe, M.; Aoki, J.; Yatomi, Y. Analysis of urinary sphingolipids using liquid chromatography-tandem mass spectrometry in diabetic nephropathy. J. Diabetes Investig. 2020, 11, 441–449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pautz, A.; Franzen, R.; Dorsch, S.; Böddinghaus, B.; Briner, V.A.; Pfeilschifter, J.; Huwiler, A. Cross-talk between nitric oxide and superoxide determines ceramide formation and apoptosis in glomerular cells. Kidney Int. 2002, 61, 790–796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Filosto, S.; Ashfaq, M.; Chung, S.; Fry, W.; Goldkorn, T. Neutral sphingomyelinase 2 activity and protein stability are modulated by phosphorylation of five conserved serines. J. Biol. Chem. 2012, 287, 514–522. [Google Scholar] [CrossRef] [Green Version]
- Zhang, A.Y.; Yi, F.; Jin, S.; Xia, M.; Chen, Q.Z.; Gulbins, E.; Li, P.L. Acid sphingomyelinase and its redox amplification in formation of lipid raft redox signaling platforms in endothelial cells. Antioxid. Redox Signal. 2007, 9, 817–828. [Google Scholar] [CrossRef]
- Bao, J.X.; Chang, H.; Lv, Y.G.; Yu, J.W.; Bai, Y.G.; Liu, H.; Cai, Y.; Wang, L.; Ma, J.; Chang, Y.M. Lysosome-membrane fusion mediated superoxide production in hyperglycaemia-induced endothelial dysfunction. PLoS ONE 2012, 7, e30387. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Han, W.Q.; Boini, K.M.; Xia, M.; Zhang, Y.; Li, P.L. TRAIL death receptor 4 signaling via lysosome fusion and membrane raft clustering in coronary arterial endothelial cells: Evidence from ASM knockout mice. J. Mol. Med. 2013, 91, 25–36. [Google Scholar] [CrossRef] [Green Version]
- Min, J.; Mesika, A.; Sivaguru, M.; Van Veldhoven, P.P.; Alexander, H.; Futerman, A.H.; Alexander, S. (Dihydro)ceramide synthase 1 regulated sensitivity to cisplatin is associated with the activation of p38 mitogen-activated protein kinase and is abrogated by sphingosine kinase 1. Mol. Cancer Res. 2007, 5, 801–812. [Google Scholar] [CrossRef] [Green Version]
- Zager, R.A.; Conrad, D.S.; Burkhart, K. Ceramide accumulation during oxidant renal tubular injury: Mechanisms and potential consequences. J. Am. Soc. Nephrol. 1998, 9, 1670–1680. [Google Scholar] [CrossRef]
- Sawada, M.; Nakashima, S.; Kiyono, T.; Nakagawa, M.; Yamada, J.; Yamakawa, H.; Banno, Y.; Shinoda, J.; Nishimura, Y.; Nozawa, Y.; et al. p53 regulates ceramide formation by neutral sphingomyelinase through reactive oxygen species in human glioma cells. Oncogene 2001, 20, 1368–1378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Owumi, S.E.; Olayiwola, Y.O.; Alao, G.E.; Gbadegesin, M.A.; Odunola, O.A. Cadmium and nickel co-exposure exacerbates genotoxicity and not oxido-inflammatory stress in liver and kidney of rats: Protective role of omega-3 fatty acid. Environ. Toxicol. 2019, 35, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Böhler, T.; Waiser, J.; Hepburn, H.; Gaedeke, J.; Lehmann, C.; Hambach, P.; Budde, K.; Neumayer, H.H. TNF-α and IL-1α induce apoptosis in subconfluent rat mesangial cells. Evidence for the involvement of hydrogen peroxide and lipid peroxidation as second messengers. Cytokine 2000, 12, 986–991. [Google Scholar] [CrossRef]
- Pastore, D.; Della-Morte, D.; Coppola, A.; Capuani, B.; Lombardo, M.F.; Pacifici, F.; Ferrelli, F.; Arriga, R.; Mammi, C.; Federici, M.; et al. SGK-1 protects kidney cells against apoptosis induced by ceramide and TNF-α. Cell. Death Dis. 2015, 6, e1890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shabahang, S.; Liu, Y.H.; Huwiler, A.; Pfeilschifter, J. Identification of the LIM kinase-1 as a ceramide-regulated gene in renal mesangial cells. Biochem. Biophys. Res. Commun. 2002, 298, 408–413. [Google Scholar] [CrossRef]
- Kim, M.H.; Ahn, H.K.; Lee, E.J.; Kim, S.J.; Kim, Y.R.; Park, J.W.; Park, W.J. Hepatic inflammatory cytokine production can be regulated by modulating sphingomyelinase and ceramide synthase 6. Int. J. Mol. Med. 2017, 39, 453–462. [Google Scholar] [CrossRef]
- Yoo, J.M.; Lee, Y.S.; Choi, H.K.; Lee, Y.M.; Hong, J.T.; Yun, Y.P.; Oh, S.; Yoo, H.S. Protection of LLC-PK1 cells against hydrogen peroxide-induced cell death by modulation of ceramide level. Arch. Pharm. Res. 2005, 28, 311–318. [Google Scholar] [CrossRef]
- Rutkute, R.; Asmis, R.H.; Nikolova-Karakashian, M.N. Regulation of neutral sphingomyelinase02 by GSH: A new insight into the role of oxidative stress in aging-associated inflammation. J. Lipid Res. 2007, 48, 2443–2452. [Google Scholar] [CrossRef] [Green Version]
- Smith, A.R.; Visioli, F.; Frei, B.; Hagen, T.M. Lipoic acid significantly restores, in rats, the age-related decline in vasomotion. Br. J. Pharmacol. 2008, 153, 1615–1622. [Google Scholar] [CrossRef]
- Thayyullathil, F.; Chathoth, S.; Hago, A.; Patel, M.; Szulc, Z.M.; Hannun, Y.; Galadari, S. Purification and characterization of a second type of neutral ceramidase from rat brain: A second more hydrophobic form of rat brain ceramidase. Biochim. Biophys. Acta 2011, 1811, 242–252. [Google Scholar] [CrossRef]
- Đurašević, S.; Stojković, M.; Bogdanović, L.; Pavlović, S.; Borković-Mitić, S.; Grigorov, I.; Bogojević, D.; Jasnić, N.; Tosti, T.; Đurović, S.; et al. The effects of meldonium on the renal acute ischemia/reperfusion injury in rats. Int. J. Mol. Sci. 2019, 20, 5747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watt, M.J.; Barnett, A.C.; Bruce, C.R.; Schenk, S.; Horowitz, J.F.; Hoy, A.J. Regulation of plasma ceramide levels with fatty acid oversupply: Evidence that the liver detects and secretes de novo synthesised ceramide. Diabetologia 2012, 55, 2741–2746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsunaga, T.; Kotamraju, S.; Kalivendi, S.V.; Dhanasekaran, A.; Joseph, J.; Kalyanaraman, B. Ceramide-induced intracellular oxidant formation, iron signaling, and apoptosis in endothelial cells: Protective role of endogenous nitric oxide. J. Biol. Chem. 2004, 279, 28614–28624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mogami, K.; Kishi, H.; Kobayashi, S. Sphingomyelinase causes endothelium-dependent vasorelaxation through endothelial nitric oxide production without cytosolic Ca2+ elevation. FEBS Lett. 2005, 579, 393–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corda, S.; Laplace, C.; Vicaut, E.; Duranteau, J. Rapid reactive oxygen species production by mitochondria in endothelial cells exposed to tumor necrosis factor-α is mediated by ceramide. Am. J. Respir. Cell. Mol. Biol. 2001, 24, 762–768. [Google Scholar] [CrossRef]
- Kitatani, K.; Akiba, S.; Sato, T. Ceramide-induced enhancement of secretory phospholipase A2 expression via generation of reactive oxygen species in tumor necrosis factor-α-stimulated mesangial cells. Cell. Signal. 2004, 16, 967–974. [Google Scholar] [CrossRef]
- Park, I.N.; Cho, I.J.; Kim, S.G. Ceramide, an apoptotic rheostat, inhibits CCAAT/enhancer binding protein-beta and NF-E2-related factor-2 activation: The role in glutathione S-transferase A2 gene repression. Drug. Metab. Dispos. 2004, 32, 893–897. [Google Scholar]
- Gómez-Sierra, T.; Molina-Jijón, E.; Tapia, E.; Hernández-Pando, R.; García-Niño, W.R.; Maldonado, P.D.; Reyes, J.L.; Barrera-Oviedo, D.; Torres, I.; Pedraza-Chaverri, J. S-allylcysteine prevents cisplatin-induced nephrotoxicity and oxidative stress. J. Pharm. Pharmacol. 2014, 66, 1271–1281. [Google Scholar] [CrossRef]
- Nezu, M.; Souma, T.; Yu, L.; Suzuki, T.; Saigusa, D.; Ito, S.; Suzuki, N.; Yamamoto, M. Transcription factor Nrf2 hyperactivation in early-phase renal ischemia-reperfusion injury prevents tubular damage progression. Kidney Int. 2017, 91, 387–401l. [Google Scholar] [CrossRef]
- Popgeorgiev, N.; Jabbour, L.; Gillet, G. Subcellular localization and dynamics of the Bcl-2 family of proteins. Front. Cell. Dev. Biol. 2018, 6, 13. [Google Scholar] [CrossRef] [Green Version]
- Pohl, S.Ö.; Agostino, M.; Dharmarajan, A.; Pervaiz, S. Cross talk between cellular redox state and the antiapoptotic protein Bcl-2. Antioxid. Redox Signal. 2018, 29, 1215–1236. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.F.; Shyu, H.W.; Chang, Y.C.; Tseng, W.C.; Huang, Y.L.; Lin, K.H.; Chou, M.C.; Liu, H.L.; Chen, C.Y. Nickel (II)-induced cytotoxicity and apoptosis in human proximal tubule cells through a ROS- and mitochondria-mediated pathway. Toxicol. Appl. Pharmacol. 2012, 259, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Kaushal, G.P.; Liu, L.; Kaushal, V.; Hong, X.; Melnyk, O.; Seth, R.; Safirstein, R.; Shah, S.V. Regulation of caspase-3 and -9 activation in oxidant stress to RTE by forkhead transcription factors, Bcl-2 proteins, and MAP kinases. Am. J. Physiol. Renal Physiol. 2004, 287, F1258–F1268. [Google Scholar] [CrossRef] [PubMed]
- Beverly, L.J.; Howell, L.A.; Hernandez-Corbacho, M.; Casson, L.; Chipuk, J.E.; Siskind, L.J. BAK activation is necessary and sufficient to drive ceramide synthase-dependent ceramide accumulation following inhibition of BCL2-like proteins. Biochem. J. 2013, 452, 111–119. [Google Scholar] [CrossRef] [Green Version]
- Zhang, T.; Barclay, L.; Walensky, L.D.; Saghatelian, A. Regulation of mitochondrial ceramide distribution by members of the BCL-2 family. J. Lipid Res. 2015, 56, 1501–1510. [Google Scholar] [CrossRef] [Green Version]
- Mullen, T.D.; Jenkins, R.W.; Clarke, C.J.; Bietawski, J.; Hannun, Y.A.; Obeid, L.M. Ceramide synthase-dependent ceramide generation and programmed cell death: Involvement of salvage pathway in regulating postmitochondrial events. J. Biol. Chem. 2011, 286, 15929–15942. [Google Scholar] [CrossRef] [Green Version]
- Kashkar, H.; Wiegmann, K.; Yazdanpanah, B.; Haubert, D.; Krönke, M. Acid sphingomyelinase is indispensable for UV light-induced Bax conformational change at the mitochondrial membrane. J. Biol. Chem. 2005, 280, 20804–20813. [Google Scholar] [CrossRef] [Green Version]
- Sawada, M.; Nakashima, S.; Banno, Y.; Yamakawa, H.; Takenaka, K.; Shinoda, J.; Nishimura, Y.; Sakai, N.; Nozawa, Y. Influence of Bax or Bcl-2 overexpression on the ceramide-dependent apoptotic pathway in glioma cells. Oncogene 2000, 19, 3508–3520. [Google Scholar] [CrossRef] [Green Version]
- Chipuk, J.E.; McStay, G.P.; Bharti, A.; Kuwana, T.; Clarke, C.J.; Siskind, L.J.; Obeid, L.M.; Green, D.R. Sphingolipid metabolism cooperates with BAK and BAX to promote the mitochondrial pathway of apoptosis. Cell 2012, 148, 988–1000. [Google Scholar] [CrossRef] [Green Version]
- Hockenbery, D.M.; Oltvai, Z.N.; Yin, X.M.; Milliman, C.L.; Korsmeyer, S.J. Bcl-2 functions in an antioxidant pathway to prevent apoptosis. Cell 1993, 75, 241–251. [Google Scholar] [CrossRef]
- El-Assaad, W.; Kozhaya, I.; Araysi, S.; Panjarian, S.; Bitar, F.F.; Baz, E.; El-Sabban, M.E. Ceramide and glutathione define two independently regulated pathways of cell death initiated by p53 in Molt-4 leukaemia cells. Biochem. J. 2003, 376, 725–732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faucher, K.; Rabinovitch-Chable, H.; Cook-Moreau, J.; Barrière, G.; Sturtz, F.; Rigaud, M. Overexpression of human GPX1 modifies Bax to Bcl-2 apoptotic ratio in human endothelial cells. Mol. Cell. Biochem. 2005, 277, 81–87. [Google Scholar] [CrossRef]
- Li, L.; Du, G.P.; Wang, D.J.; Zhou, J.; Jiang, G.; Jiang, H. Overexpression of heme oxygenase-1 in mesenchymal stem cells augments their protection on retinal cells in vitro and attenuates retinal ischemia/reperfusion injury in vivo against oxidative stress. Stem. Cells Int. 2017, 2017, 4985323. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhang, W.; Xiao, E. SOD2 overexpression in bone marrow-derived mesenchymal stem cells ameliorates hepatic ischemia/reperfusion injury. Mol. Med. Rep. 2021, 24, 671. [Google Scholar] [CrossRef] [PubMed]
- Zigdon, H.; Kogot-Levin, A.; Park, J.W.; Goldschmidt, R.; Kelly, S.; Merrill, A.H., Jr.; Scherz, A.; Pewzner-Jung, Y.; Saada, A.; Futerman, A.H. Ablation of ceramide synthase 2 causes chronic oxidative stress due to disruption of the mitochondrial respiratory chain. J. Biol. Chem. 2013, 288, 4947–4956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moser, M.D.; Matsuzaki, S.; Humphries, K.M. Inhibition of succinate-linked respiration and complex II activity by hydrogen peroxide. Arch. Biochem. Biophys. 2009, 488, 69–75. [Google Scholar] [CrossRef]
- Sundaram, K.; Mather, A.R.; Marimuthu, S.; Shah, P.P.; Snider, A.J.; Obeid, L.M.; Hannun, Y.A.; Beverly, L.J.; Siskind, L.J. Loss of neutral ceramidase protects cells from nutrient- and energy -deprivation-induced cell death. Biochem. J. 2016, 473, 743–755. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Wu, Y.; Luo, K.; Liu, Y.; Zhou, M.; Yan, S.; Shi, H.; Cai, Y. The protective effects of selenium on cadmium-induced oxidative stress and apoptosis via mitochondria pathway in mice kidney. Food Chem. Toxicol. 2013, 58, 61–67. [Google Scholar] [CrossRef]
- Jiang, X.; Liao, X.H.; Huang, L.L.; Sun, H.; Liu, Q.; Zhang, L. Overexpression of augmenter of liver regeneration (ALR) mitigates the effect of H 2O2-induced endoplasmic reticulum stress in renal tubule epithelial cells. Apoptosis 2019, 24, 278–289. [Google Scholar] [CrossRef]
- Correa, F.; Pavón, N.; Buelna-Chontal, M.; Chiquete-Félix, N.; Hernández-Esquivel, L.; Chávez, E. Calcium induces mitochondrial oxidative stress because of its binding to adenine nucleotide translocase. Cell. Biochem. Biophys. 2018, 76, 445–450. [Google Scholar] [CrossRef]
- Ichimiya, M.; Chang, S.H.; Liu, H.; Berezesky, I.K.; Trump, B.F.; Amstad, P.A. Effect of Bcl-2 on oxidant-induced cell death and intracellular Ca2+ mobilization. Am. J. Physiol. 1998, 275, C832–C839. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhang, L.; Zhao, J.; Guo, X.; Luo, Y.; Hu, W.; Zhao, T. Tumor necrosis factor receptor-associated protein 1 protects against mitochondrial injury by preventing high glucose-induced mPTP opening in diabetes. Oxid. Med. Cell. Longev. 2020, 2020, 6431517. [Google Scholar] [CrossRef] [PubMed]
- García-Ruiz, C.; Colell, A.; Marí, M.; Morales, A.; Fernández-Checa, J.C. Direct effect of ceramide on the mitochondrial electron transport chain leads to generation of reactive oxygen species. Role of mitochondrial glutathione. J. Biol. Chem. 1997, 272, 11369–11377. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Wu, C.G.; Fang, C.Q.; Gao, J.; Liu, Y.Z.; Chen, Y.; Chen, Y.N.; Xu, Z.G. The protective effect of α-Lipoic acid on mitochondria in the kidney of diabetic rats. Int. J. Clin. Exp. Med. 2013, 6, 90–97. [Google Scholar]
- Wang, L.; Lin, S.Q.; He, Y.L.; Liu, G.; Wang, Z.Y. Protective effects of quercetin on cadmium-induced cytotoxicity in primary cultures of rat proximal tubular cells. Biomed. Environ. Sci. 2013, 26, 258–267. [Google Scholar] [PubMed]
- Huang, S.; Tang, Y.; Liu, T.; Zhang, N.; Yang, X.; Yang, D.; Hong, G. A novel antioxidant protects against contrast medium-induced acute kidney injury in rats. Front. Pharmacol. 2020, 11, 599577. [Google Scholar] [CrossRef]
- Ahmed, L.A.; Shehata, N.I.; Abdelkader, N.A.; Khattab, M.M. Tempol, a superoxide dismutase mimetic agent, ameliorates cisplatin-induced nephrotoxicity through alleviation of mitochondrial dysfunction in mice. PLoS ONE 2014, 9, e108889. [Google Scholar] [CrossRef] [Green Version]
- Scorrano, L.; Oakes, S.A.; Opferman, J.T.; Cheng, E.H.; Sorcinelli, M.D.; Pozzan, T.; Korsmeyer, S.J. BAX and BAK regulation of endoplasmic reticulum Ca2+: A control point for apoptosis. Science 2003, 300, 135–139. [Google Scholar] [CrossRef]
- Perera, M.N.; Lin, S.H.; Peterson, Y.K.; BielawskaRoy, S.S.; Madesh, M.; Davies, E.; Antonsson, B.; Danial, N.; Hajnóczky, G. Bad targets the permeability transition pore independent of Bax or Bak to switch between Ca2+-dependent cell survival and death. Mol. Cell. 2009, 33, 377–388. [Google Scholar]
- Verweij, M.; Sluiter, W.; van den Engel, S.; Jansen, E.; Ijzermans, J.N.M.; de Bruin, R.W.F. Altered mitochondrial functioning induced by preoperative fasting may underlie protection against renal ischemia/reperfusion injury. J. Cell. Biochem. 2013, 114, 230–237. [Google Scholar] [CrossRef]
- Iwayama, H.; Ueda, N. Role of mitochondrial Bax, caspases, and MAPKs for ceramide-induced apoptosis in renal proximal tubular cells. Mol. Cell. Biochem. 2013, 379, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Pastorino, J.G.; Tafani, M.; Rothman, R.J.; Marcinkeviciute, A.; Hoek, J.B.; Farber, J.L. Functional consequences of the sustained or transient activation by Bax of the mitochondrial permeability transition pore. J. Biol. Chem. 1999, 274, 31734–31739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ganesan, V.; Perera, M.N.; Colombini, D.; Datskovskiy, D.; Chadha, K.; Colombini, M. Ceramide and activated Bax act synergistically to permeabilize the mitochondrial outer membrane. Apoptosis 2010, 15, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Pinton, P.; Ferrari, D.; Rapizzi, E.; Di Virgilio, F.; Pozzan, T.; Rizzuto, R. The Ca2+ concentration of the endoplasmic reticulum is a key determinant of ceramide-induced apoptosis: Significance for the molecular mechanism of Bcl-2 action. EMBO J. 2001, 20, 2690–2701. [Google Scholar] [CrossRef]
- Colombini, M. Ceramide channels and mitochondrial outer membrane permeability. J. Bioenerg. Biomembr. 2017, 49, 57–64. [Google Scholar] [CrossRef]
- Perera, M.N.; Lin, S.H.; Peterson, Y.K.; Bielawska, A.; Szulc, Z.M.; Bittman, R.; Colombini, M. Bax and Bcl-xL exert their regulation on different sites of the ceramide channel. Biochem. J. 2012, 445, 81–91. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Abundis, E.; Correa, F.; Pavón, N.; Zazueta, C. Bax distribution into mitochondrial detergent-resistant microdomains is related to ceramide and cholesterol content in postischemic hearts. FEBS J. 2009, 276, 5579–5588. [Google Scholar] [CrossRef]
- Dadsena, S.; Bockelmann, S.; Mina, J.G.M.; Hassan, D.G.; Korneev, S.; Razzera, G.; Jahn, H.; Niekamp, P.; Müller, D.; Schneider, M.; et al. Ceramides bind VDAC2 to trigger mitochondrial apoptosis. Nat. Commun. 2019, 10, 1832. [Google Scholar] [CrossRef]
- Scharstuhl, A.; Mutsaers, H.A.; Pennings, S.W.; Russel, F.G.; Wagener, F.A. Involvement of VDAC, Bax and ceramides in the efflux of AIF from mitochondria during curcumin-induced apoptosis. PLoS ONE 2009, 4, e6688. [Google Scholar] [CrossRef]
- Kong, J.N.; Zhu, Z.; Itokazu, Y.; Wang, G.; Dinkins, M.B.; Zhong, L.; Lin, H.P.; Elsherbini, A.; Leanhart, S.; Jiang, X.; et al. Novel function of ceramide for regulation of mitochondrial ATP release in astrocytes. J. Lipid Res. 2018, 59, 488–506. [Google Scholar] [CrossRef] [Green Version]
- Gupta, K.; Kshirsagar, S.; Li, W.; Gui, L.; Ramakrishnan, S.; Gupta, P.; Law, P.Y.; Hebbel, R. P.VEGF prevents apoptosis of human microvascular endothelial cells via opposing effects on MAPK/ERK and SAPK/JNK signaling. Exp. Cell. Res. 1999, 247, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Pfeilschifter, J.; Huwiler, A. Identification of ceramide targets in interleukin-1 and tumor necrosis factor-alpha signaling in mesangial cells. Kidney Int. Suppl. 1998, 67, S34–S639. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loidl, A.; Claus, R.; Ingolic, E.; Deigner, H.P.; Hermetter, A. Role of ceramide in activation of stress-associated MAP kinases by minimally modified LDL in vascular smooth muscle cells. Biochim. Biophys. Acta 2004, 1690, 150–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huwiler, A.; Xin, C.; Brust, A.K.; Briner, V.A.; Pfeilschifter, J. Differential binding of ceramide to MEKK1 in glomerular endothelial and mesangial cells. Biochim. Biophys. Acta 2004, 1636, 159–168. [Google Scholar] [CrossRef]
- Bourbon, N.A.; Yun, J.; Kester, M. Ceramide directly activates protein kinase C zeta to regulate a stress-activated protein kinase signaling complex. J. Biol. Chem. 2000, 275, 35617–35623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dagher, P.C. Apoptosis in ischemic renal injury: Roles of GTP depletion and p53. Kidney Int. 2004, 66, 506–509. [Google Scholar] [CrossRef] [Green Version]
- Fekry, B.; Jeffries, K.A.; Esmaeilniakooshkghazi, A.; Szulc, Z.M.; Knagge, K.J.; Kirchner, D.R.; Horita, D.A.; Krupenko, S.A.; Krupenko, N.I. C 16-ceramide is a natural regulatory ligand of p53 in cellular stress response. Nat. Commun. 2018, 9, 4149. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Higuchi, M. Role of ceramide in tumour necrosis factor-mediated apoptosis and nuclear factor-kB activation. Biochem. Soc. Trans. 1997, 25, 1166–1171. [Google Scholar] [CrossRef] [Green Version]
- Ansari, M.A.; Raish, M.; Ahmad, A.; Alkharfy, K.M.; Ahmad, S.F.; Attia, S.M.; Alsaad, A.M.S.; Bakheet, S.A. Sinapic acid ameliorate cadmium-induced nephrotoxicity: In vivo possible involvement of oxidative stress, apoptosis, and inflammation via NF-κB downregulation. Environ. Toxicol. Pharmacol. 2017, 51, 100–107. [Google Scholar] [CrossRef]
- Majumdar, S.; Aggarwal, B.B. Adenosine suppresses activation of nuclear factor-kB selectively induced by tumor necrosis factor in different cell types. Oncogene 2003, 22, 1206–1218. [Google Scholar] [CrossRef]
- Markó, L.; Vigolo, E.; Hinze, C.; Park, J.K.; Roël, G.; Balogh, A.; Choi, M.; Wübken, A.; Cording, J.; Blasig, I.E.; et al. Tubular epithelial NF-κB activity regulates ischemic AKI. J. Am. Soc. Nephrol. 2016, 27, 2658–2669. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cao, L.C.; Honeyman, T.W.; Cooney, R.; Kennington, L.; Scheid, C.R.; Jonassen, J.A. Mitochondrial dysfunction is a primary event in renal cell oxalate toxicity. Kidney Int. 2004, 66, 1890–1900. [Google Scholar] [CrossRef] [Green Version]
- Harris, R.C.; Zhang, M.Z. Cyclooxygenase metabolites in the kidney. Compr. Physiol. 2011, 1, 1729–1758. [Google Scholar] [PubMed]
- Huwiler, A.; Johansen, B.; Skarstad, A.; Pfeilschifter, J. Ceramide binds to the CaLB domain of cytosolic phospholipase A2 and facilitates its membrane docking and arachidonic acid release. FASEB J. 2001, 15, 7–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, H.; Murayama, T. Role of sphingolipids in arachidonic acid metabolism. J. Pharmacol. Sci. 2014, 124, 307–312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zager, R.A.; Burkhart, K.M.; Johnson, A. Sphingomyelinase and membrane sphingomyelin content: Determinants of proximal tubule cell susceptibility to injury. J. Am. Soc. Nephrol. 2000, 11, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.L.; Kang, B.; Yang, L.J.; Williamson, J.R. Tumor necrosis factor-alpha and ceramide induce cell death through different mechanisms in rat mesangial cells. Am. J. Physiol. 1999, 276, F390–F397. [Google Scholar] [PubMed]
- Tsao, C.C.; Nica, A.F.; Kurinna, S.M.; Jiffar, T.; Mumby, M.; Ruvolo, P.P. Mitochondrial protein phosphatase 2A regulates cell death induced by simulated ischemia in kidney NRK-52E cells. Cell. Cycle 2007, 6, 2377–2385. [Google Scholar] [CrossRef] [Green Version]
- Li, T.; Ying, L.; Wang, H.; Li, N.; Fu, W.; Guo, Z.; Xu, L. Microcystin-LR induces ceramide to regulate PP2A and destabilize cytoskeleton in HEK293 cells. Toxicol. Sci. 2012, 128, 147–157. [Google Scholar] [CrossRef] [Green Version]
- Lang, F.; Böhmer, C.; Palmada, M.; Seebohm, G.; Strutz-Seebohm, N.; Vallon, V. (Patho)physiological significance of the serum- and glucocorticoid-inducible kinase isoforms. Physiol. Rev. 2006, 86, 1151–1178. [Google Scholar] [CrossRef]
- Basnet, R.; Gong, G.Q.; Li, C.; Wang, M.W. Serum and glucocorticoid inducible protein kinases (SGKs): A potential target for cancer intervention. Acta Pharm. Sin. B 2018, 8, 767–771. [Google Scholar] [CrossRef] [PubMed]
- Rusai, K.; Wagner, B.; Roos, M.; Schmaderer, C.; Strobl, M.; Boini, K.M.; Grenz, A.; Kuhl, D.; Heemann, U.; Lang, F.; et al. The serum and glucocorticoid-regulated kinase 1 in hypoxic renal injury. Cell. Physiol. Biochem. 2009, 24, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Ferrelli, F.; Pastore, D.; Capuani, B.; Lombardo, M.F.; Blot-Chabaud, M.; Coppola, A.; Basello, K.; Galli, A.; Donadel, G.; Romano, M.; et al. Serum glucocorticoid inducible kinase (SGK)-1 protects endothelial cells against oxidative stress and apoptosis induced by hyperglycaemia. Acta Diabetol. 2015, 52, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Schwalm, S.; Beyer, S.; Frey, H.; Haceni, R.; Grammatikos, G.; Thomas, D.; Geisslinger, G.; Schaefer, L.; Huwiler, A.; Pfeilschifter, J. Sphingosine kinase-2 deficiency ameliorates kidney fibrosis by up-regulating Smad7 in a mouse model of unilateral ureteral obstruction. Am. J. Pathol. 2017, 187, 2413–2429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, X.; Li, D.; Chen, X.; Han, C.; Xu, L.; Huang, T.; Dong, Z.; Zhang, M. Extracellular vesicles from human-induced pluripotent stem cell-derived mesenchymal stromal cells (hiPSC-MSCs) protect against renal ischemia/reperfusion injury via delivering specificity protein (SP1) and transcriptional activating of sphingosine kinase 1 and inhibiting necroptosis. Cell. Death Dis. 2017, 8, 3200. [Google Scholar]
- Jo, S.K.; Bajwa, A.; Ye, H.; Vergis, A.L.; Awad, A.S.; Kharel, Y.; Lynch, K.R.; Okusa, M.D. Divergent roles of sphingosine kinases in kidney ischemia-reperfusion injury. Kidney Int. 2009, 75, 167–175. [Google Scholar] [CrossRef] [Green Version]
- Schwalm, S.; Döll, F.; Römer, I.; Bubnova, S.; Pfeilschifter, J.; Huwiler, A. Sphingosine kinase-1 is a hypoxia-regulated gene that stimulates migration of human endothelial cells. Biochem. Biophys. Res. Commun. 2008, 368, 1020–1025. [Google Scholar] [CrossRef]
- Lan, T.; Liu, W.; Xie, X.; Huang, J.; Peng, J.; Huang, J.; Shen, X.; Liu, P.; Yang, H.; Huang, H. Berberine suppresses high glucose-induced TGF-β1 and fibronectin synthesis in mesangial cells through inhibition of sphingosine kinase 1/AP-1 pathway. Eur. J. Pharmacol. 2012, 697, 165–172. [Google Scholar] [CrossRef]
- Chen, C.; Huang, K.; Hao, J.; Huang, J.; Yang, Z.; Xiong, F.; Liu, P.; Huang, H. Polydatin attenuates AGEs-induced upregulation of fibronectin and ICAM-1 in rat glomerular mesangial cells and db/db diabetic mice kidneys by inhibiting the activation of the SphK1-S1P signaling pathway. Mol. Cell. Endocrinol. 2016, 427, 45–56. [Google Scholar] [CrossRef]
- Yaghobian, D.; Don, A.S.; Yaghobian, S.; Chen, X.; Pollock, C.A.; Saad, S. Increased sphingosine 1-phosphate mediates inflammation and fibrosis in tubular injury in diabetic nephropathy. Clin. Exp. Pharmacol. Physiol. 2016, 43, 56–66. [Google Scholar] [CrossRef]
- Du, C.; Ren, Y.; Yao, F.; Duan, J.; Zhao, H.; Du, Y.; Xiao, X.; Duan, H.; Shi, Y. Sphingosine kinase 1 protects renal tubular epithelial cells from renal fibrosis via induction of autophagy. Int. J. Biochem. Cell. Biol. 2017, 90, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, W.; Ji, X.Y.; Ritter, J.K.; Li, N. Knockout of sphingosine kinase 1 attenuates renal fibrosis in unilateral ureteral obstruction model. Am. J. Nephrol. 2019, 50, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Sui, M.; Zhou, J.; Xie, R.; Liu, X.; Mu, S.; Jia, X.; Ma, J.; Wu, H. The sphingosine-1-phosphate receptor agonist FTY720 prevents the development of anti-glomerular basement membrane glomerulonephritis. Mol. Biol. Rep. 2012, 39, 389–397. [Google Scholar] [CrossRef]
- Huang, J.; Li, J.; Chen, Z.; Li, J.; Chen, Q.; Gong, W.; Liu, P.; Huang, H. Sphingosine kinase 1 mediates diabetic renal fibrosis via NF-κB signaling pathway: Involvement of CK2α. Oncotarget 2017, 8, 88988–89004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- You, B.; Ren, A.; Yan, G.; Sun, J. Activation of sphingosine kinase-1 mediates inhibition of vascular smooth muscle cell apoptosis by hyperglycemia. Diabetes 2007, 56, 1445–1453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, S.; Babelova, A.; Moreth, K.; Xin, C.; Eberhardt, W.; Doller, A.; Pavenstädt, H.; Schaefer, L.; Pfeilschifter, J.; Huwiler, A. Transforming growth factor-β2 upregulates sphingosine kinase-1 activity, which in turn attenuates the fibrotic response to TGF-β2 by impeding CTGF expression. Kidney Int. 2009, 76, 857–867. [Google Scholar] [CrossRef] [Green Version]
- Imeri, F.; Tanturovska, B.S.; Schwalm, S.; Saha, S.; Zeng-Brouwers, J.; Pavenstädt, H.; Pfeilschifter, J.; Schaefer, L.; Huwiler, A. Loss of sphingosine kinase 2 enhances Wilm’s tumor suppressor gene 1 and nephrin expression in podocytes and protects from streptozotocin-induced podocytopathy and albuminuria in mice. Matrix Biol. 2021, 98, 32–48. [Google Scholar] [CrossRef]
- Tsutsumi, T.; Ishihara, A.; Yamamoto, A.; Asaji, H.; Yamakawa, S.; Tokumura, A. The potential protective role of lysophospholipid mediators in nephrotoxicity induced by chronically exposed cadmium. Food Chem. Toxicol. 2014, 65, 52–62. [Google Scholar] [CrossRef]
- Benito, S.; Sánchez-Ortega, A.; Unceta, N.; Andrade, F.; Aldámiz-Echevarria, L.; Goicolea, M.A.; Barrio, R.J. Untargeted metabolomics for plasma biomarker discovery for early chronic kidney disease diagnosis in pediatric patients using LC-QTOF-MS. Analyst 2018, 143, 4448–4458. [Google Scholar] [CrossRef]
- Brinck, J.W.; Thomas, A.; Brulhart-Meynet, M.C.; Lauer, E.; Frej, C.; Dahlbäck, B.; Stenvinkel, P.; James, R.W.; Frias, M.A. High-density lipoprotein from end-stage renal disease patients exhibits superior cardioprotection and increase in sphingosine-1-phosphate. Eur. J. Clin. Investig. 2018, 48, e12866. [Google Scholar] [CrossRef] [Green Version]
- Watson, L.; Tullus, K.; Marks, S.D.; Holt, R.C.; Pilkington, C.; Beresford, M.W. Increased serum concentration of sphingosine-1-phosphate in juvenile-onset systemic lupus erythematosus. J. Clin. Immunol. 2012, 32, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, S.; Vineetha, N.S.; James, S.; Aparna, J.S.; Lankadasari, M.B.; Allegood, J.C.; Li, Q.Z.; Spiegel, S.; Harikumar, K.B. Examination of the role of sphingosine kinase 2 in a murine model of systemic lupus erythematosus. FASEB J. 2019, 33, 7061–7071. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Kanazawa, I.; Sugimoto, T. Visceral fat accumulation is associated with increased plasma sphingosine-1-phosphate levels in type 2 diabetes mellitus. Diabetes Res. Clin. Pract. 2018, 143, 146–150. [Google Scholar] [CrossRef] [PubMed]
- Bekpinar, S.; Yenidunya, G.; Gurdol, F.; Unlucerci, Y.; Aycan-Ustyol, E.; Dinccag, N. The effect of nephropathy on plasma sphingosine 1-phosphate concentrations in patients with type 2 diabetes. Clin. Biochem. 2015, 48, 1264–1267. [Google Scholar] [CrossRef]
- Kurano, M.; Tsukamoto, K.; Shimizu, T.; Kassai, H.; Nakao, K.; Aiba, A.; Hara, M.; Yatomi, Y. Protection against insulin resistance by apolipoprotein m/sphingosine-1-phosphate. Diabetes 2020, 69, 867–881. [Google Scholar] [CrossRef]
- Sutter, I.; Park, R.; Othman, A.; Rohrer, L.; Hornemann, T.; Stoffel, M.; Devuyst, O.; von Eckardstein, A. Apolipoprotein M modulates erythrocyte efflux and tubular reabsorption of sphingosine-1-phosphate. J. Lipid Res. 2014, 55, 1730–1737. [Google Scholar] [CrossRef] [Green Version]
- Cinq-Frais, C.; Coatrieux, C.; Grazide, M.H.; Hannun, Y.A.; Nègre-Salvayre, A.; Salvayre, R.; Augé, N. A signaling cascade mediated by ceramide, src and PDGFRβ coordinates the activation of the redox-sensitive neutral sphingomyelinase-2 and sphingosine kinase-1. Biochim. Biophys. Acta 2013, 1831, 1344–1356. [Google Scholar] [CrossRef]
- Chen, H.C.; Guh, J.Y.; Shin, S.J.; Tomino, Y.; Lai, Y.H. Effects of pravastatin on superoxide and fibronectin production of mesangial cells induced by low-density lipoprotein. Kidney Blood Press. Res. 2002, 25, 2–6. [Google Scholar] [CrossRef]
- El-Shewy, H.M.; Sohn, M.; Wilson, P.; Lee, M.H.; Hammad, S.M.; Luttrell, L.M.; Jaffa, A.A. Low-density lipoprotein induced expression of connective tissue growth factor via transactivation of sphingosine 1-phosphate receptors in mesangial cells. Mol. Endocrinol. 2012, 26, 833–845. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.B.; Kim, J.J.; Kim, T.W.; Kim, B.S.; Lee, M.S.; Yoo, Y.D. Serum deprivation- induced reactive oxygen species production is mediated by Romo1. Apoptosis 2010, 15, 204–218. [Google Scholar] [CrossRef]
- Truman, J.P.; Ruiz, C.F.; Trayssac, M.; Mao, C.; Hannun, Y.A.; Obeid, L.M. Sphingosine kinase 1 downregulation is required for adaptation to serine deprivation. FASEB J. 2021, 35, e21284. [Google Scholar] [CrossRef] [PubMed]
- Okada, T.; Ding, G.; Sonoda, H.; Kajimoto, T.; Haga, Y.; Khosrowbeygi, A.; Gao, S.; Miwa, N.; Jahangeer, S.; Nakamura, S. Involvement of N-terminal-extended form of sphingosine kinase 2 in serum-dependent regulation of cell proliferation and apoptosis. J. Biol. Chem. 2005, 280, 36318–36325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shao, J.J.; Peng, Y.; Wang, L.M.; Wang, J.K.; Chen, X. Activation of sphK1 by K6PC-5 inhibits oxygen-glucose deprivation/reoxygenation-induced myocardial cell death. DNA Cell. Biol. 2015, 34, 669–676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, S.; Yang, J.; Xiang, H.; Chen, W.; Zhong, H.; Yang, G.; Fang, T.; Deng, H.; Yuan, H.; Chen, A.F.; et al. Role of sphingosine-1-phosphate receptor 1 and sphingosine-1-phosphate receptor 2 in hyperglycemia-induced endothelial cell dysfunction. Int. J. Mol. Med. 2015, 35, 1103–1108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, F.C.; Yuan, C.X.; Tong, J.Y.; Zhang, G.H.; Zhou, F.P.; Yang, F. Protective effect of sphingosine-1-phosphate for chronic intermittent hypoxia-induced endothelial cell injury. Biochem. Biophys. Res. Commun. 2018, 498, 1016–1021l. [Google Scholar] [CrossRef]
- Bajwa, A.; Rosin, D.L.; Chroscicki, P.; Lee, S.; Dondeti, K.; Ye, H.; Kinsey, G.R.; Stevens, B.K.; Jobin, K.; Kenwood, B.M.; et al. Sphingosine 1-phosphate receptor-1 enhances mitochondrial function and reduces cisplatin-induced tubule injury. J. Am. Soc. Nephrol. 2015, 26, 908–925. [Google Scholar] [CrossRef]
- Strub, G.M.; Paillard, M.; Liang, J.; Gomez, L.; Allegood, J.C.; Hait, N.C.; Maceyka, M.; Price, M.M.; Chen, Q.; Simpson, D.C.; et al. Sphingosine-1-phosphate produced by sphingosine kinase 2 in mitochondria interacts with prohibitin 2 to regulate complex IV assembly and respiration. FASEB J. 2011, 25, 600–612. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Toman, R.E.; Goparaju, S.K.; Maceyka, M.; Nava, V.E.; Sankala, H.; Payne, S.G.; Bektas, M.; Ishii, I.; Chun, J.; et al. Sphingosine kinase type 2 is a putative BH3-only protein that induces apoptosis. J. Biol. Chem. 2003, 278, 40330–40336. [Google Scholar] [CrossRef] [Green Version]
- Maceyka, M.; Sankala, H.; Hait, N.C.; Le Stunff, H.; Liu, H.; Toman, R.; Collier, C.; Zhang, M.; Satin, L.S.; Merrill, A.H., Jr.; et al. SphK1 and SphK2, sphingosine kinase isoenzymes with opposing functions in sphingolipid metabolism. J. Biol. Chem. 2005, 280, 37118–37129. [Google Scholar] [CrossRef] [Green Version]
- Park, S.W.; Kim, M.; Kim, M.; D’Agati, V.D.; Lee, H.T. Sphingosine kinase 1 protects against renal ischemia-reperfusion injury in mice by sphingosine-1-phosphate1 receptor activation. Kidney Int. 2011, 80, 1315–1327. [Google Scholar] [CrossRef] [Green Version]
- Park, S.W.; Kim, M.; Brown, K.M.; D’Agati, V.D.; Lee, H.T. Inhibition of sphingosine 1-phosphate receptor 2 protects against renal ischemia-reperfusion injury. J. Am. Soc. Nephrol. 2012, 23, 266–280. [Google Scholar] [CrossRef] [PubMed]
- Gamble, J.R.; Sun, W.Y.; Li, X.; Hahn, C.N.; Pitson, S.M.; Vadas, M.A.; Bonder, C.S. Sphingosine kinase-1 associates with integrin αVβ3 to mediate endothelial cell survival. Am. J. Pathol. 2009, 175, 2217–2225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Limaye, V.; Li, X.; Hahn, C.; Xia, P.; Berndt, M.C.; Vadas, M.A.; Gamble, J.R. Sphingosine kinase-1 enhances endothelial cell survival through a PECAM-1-dependent activation of PI-3K/Akt and regulation of Bcl-2 family members. Blood 2005, 105, 3169–3177. [Google Scholar] [CrossRef] [Green Version]
- Song, Z.; Wang, W.; Li, N.; Yan, S.; Rong, K.; Lan, T.; Xia, P. Sphingosine kinase 2 promotes lipotoxicity in pancreatic β-cells and the progression of diabetes. FASEB J. 2019, 33, 3636–3646. [Google Scholar] [CrossRef]
- Kumar, A.; Byun, H.S.; Bittman, R.; Saba, J.D. The sphingolipid degradation product trans-2-hexadecenal induces cytoskeletal reorganization and apoptosis in a JNK-dependent manner. Cell. Signal. 2011, 23, 1144–1152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agrawal, M.; Kumar, V.; Singh, A.K.; Kashyap, M.P.; Khanna, V.K.; Siddiqui, M.A.; Pant, A.B. trans-Resveratrol protects ischemic PC12 cells by inhibiting the hypoxia associated transcription factors and increasing the levels of antioxidant defense enzymes. ACS Chem. Neurosci. 2013, 4, 285–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.W.; Kim, M.; Kim, J.Y.; Brown, K.M.; Haase, V.H.; D’Agati, V.D.; Lee, H.T. Proximal tubule sphingosine kinase-1 has a critical role in A1 adenosine receptor-mediated renal protection from ischemia. Kidney Int. 2012, 82, 878–891. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.T.; Park, S.W.; Kim, M.; Ham, A.; Anderson, L.J.; Brown, K.M.; D’Agati, V.D.; Cox, G.N. Interleukin-11 protects against renal ischemia and reperfusion injury. Am. J. Physiol. Renal Physiol. 2012, 303, F1216–F1224. [Google Scholar] [CrossRef] [Green Version]
- Ham, A.; Kim, M.; Kim, J.Y.; Brown, K.M.; Fruttiger, M.; D’Agati, V.D.; Lee, H.T. Selective deletion of the endothelial sphingosine-1-phosphate 1 receptor exacerbates kidney ischemia-reperfusion injury. Kidney Int. 2014, 85, 807–823. [Google Scholar] [CrossRef] [Green Version]
- Pitson, S.M.; Moretti, P.A.; Zebol, J.R.; Lynn, H.E.; Xia, P.; Vadas, M.A.; Wattenberg, B.W. Activation of sphingosine kinase 1 by ERK1/2-mediated phosphorylation. EMBO J. 2003, 22, 5491–5500. [Google Scholar] [CrossRef] [Green Version]
- Bajwa, A.; Jo, S.K.; Ye, H.; Huang, L.; Dondeti, K.R.; Rosin, D.L.; Haase, V.H.; Macdonald, T.L.; Lynch, K.R.; Okusa, M.D. Activation of sphingosine-1-phosphate 1 receptor in the proximal tubule protects against ischemia-reperfusion injury. J. Am. Soc. Nephrol. 2010, 21, 955–965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bakar, A.M.; Park, S.W.; Kim, M.; Lee, H.T. Isoflurane protects against human endothelial cell apoptosis by inducing sphingosine kinase-1 via ERK MAPK. Int. J. Mol.Sci. 2012, 13, 977–993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Min, J.; Van Veldhoven, P.P.; Zhang, L.; Hanigan, M.H.; Alexander, H.; Alexander, S. Sphingosine-1-phosphate lyase regulates sensitivity of human cells to select chemotherapy drugs in a p38-dependent manner. Mol. Cancer Res. 2005, 3, 287–296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, S.; Chintapalli, J.; Sodagum, L.; Baskin, S.; Malhotra, A.; Reiss, K.; Meggs, L.G. Activated IGF-1R inhibits hyperglycemia-induced DNA damage and promotes DNA repair by homologous recombination. Am. J. Physiol. Renal Physiol. 2005, 289, F1144–F1152. [Google Scholar] [CrossRef]
- Kang, B.P.S.; Urbonas, A.; Baddoo, A.; Baskin, S.; Malhotra, A.; Meggs, L.G. IGF-1 inhibits the mitochondrial apoptosis program in mesangial cells exposed to high glucose. Am. J. Physiol. Renal Physiol. 2003, 285, F1013–F1024. [Google Scholar] [CrossRef] [Green Version]
- Granata, R.; Trovato, L.; Garbarino, G.; Taliano, M.; Ponti, R.; Sala, G.; Ghidoni, R.; Ghigo, E. Dual effects of IGFBP-3 on endothelial cell apoptosis and survival: Involvement of the sphingolipid signaling pathways. FASEB J. 2004, 18, 1456–1458. [Google Scholar] [CrossRef]
- Granata, R.; Trovato, L.; Lupia, E.; Sala, G.; Settanni, F.; Camussi, G.; Ghidoni, R.; Ghigo, E. Insulin-like growth factor binding protein-3 induces angiogenesis through IGF-I- and SphK1-dependent mechanisms. J. Thromb. Haemost. 2007, 5, 835–845. [Google Scholar] [CrossRef]
- Sun, B.; Lu, C.; Zhou, G.P.; Xing, C.Y. Suppression of Par-4 protects human renal proximal tubule cells from apoptosis induced by oxidative stress. Nephron Exp. Nephrol. 2011, 117, e53–e61. [Google Scholar] [CrossRef]
- Moriue, T.; Igarashi, J.; Yoneda, K.; Nakai, K.; Kosaka, H.; Kubota, Y. Sphingosine 1-phosphate attenuates H2O2-induced apoptosis in endothelial cells. Biochem. Biophys. Res. Commun. 2008, 368, 852–857. [Google Scholar] [CrossRef]
- Barr, R.K.; Lynn, H.E.; Moretti, P.A.B.; Khew-Goodall, Y.; Pitson, S.M. Deactivation of sphingosine kinase 1 by protein phosphatase 2A. J. Biol. Chem. 2008, 283, 34994–35002. [Google Scholar] [CrossRef] [Green Version]
- Ishibashi, Y.; Matsui, T.; Maeda, S.; Higashimoto, Y.; Yamagishi, S. Advanced glycation end products evoke endothelial cell damage by stimulating soluble dipeptidyl peptidase-4 production and its interaction with mannose 6-phosphate/insulin-like growth factor II receptor. Cardiovasc. Diabetol. 2013, 12, 125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Shewy, H.M.; Abdel-Samie, S.A.; Al Qalam, A.M.; Lee, M.H.; Kitatani, K.; Anelli, V.; Jaffa, A.A.; Obeid, L.M.; Luttrell, L.M. Phospholipase C and protein kinase C-β 2 mediate insulin-like growth factor II-dependent sphingosine kinase 1 activation. Mol. Endocrinol. 2011, 25, 2144–2156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deng, Y.; Lan, T.; Huang, J.; Huang, H. Sphingosine kinase-1/sphingosine 1-phosphate pathway in diabetic nephropathy. Chin. Med. J. 2014, 127, 3004–3010. [Google Scholar] [PubMed]
- Katsuma, S.; Hada, Y.; Ueda, T.; Shiojima, S.; Hirasawa, A.; Tanoue, A.; Takagaki, K.; Ohgi, T.; Yano, J.; Tsujimoto, G. Signalling mechanisms in sphingosine 1-phosphate-promoted mesangial cell proliferation. Genes Cells. 2002, 7, 1217–1230. [Google Scholar] [CrossRef]
- Zahner, G.; Wolf, G.; Schroeder, S.; Stahl, R.A. Inhibition of platelet-derived growth factor-induced mesangial cell proliferation by cyclooxygenase-2 overexpression is abolished through reactive oxygen species. FEBS Lett. 2006, 580, 2523–2528. [Google Scholar] [CrossRef] [Green Version]
- Schwalm, S.; Timcheva, T.M.; Filipenko, I.; Ebadi, M.; Hofmann, L.P.; Zangemeister-Wittke, U.; Pfeilschifter, J.; Huwiler, A. Sphingosine kinase 2 deficiency increases proliferation and migration of renal mouse mesangial cells and fibroblasts. J. Biol. Chem. 2015, 396, 813–825. [Google Scholar] [CrossRef] [Green Version]
- Lien, Y.H.; Yong, K.C.; Cho, C.; Igarashi, S.; Lai, L.W. S1P(1)-selective agonist, SEW2871, ameliorates ischemic acute renal failure. Kidney Int. 2006, 69, 1601–1608. [Google Scholar] [CrossRef] [Green Version]
- Blanchard, O.; Stepanovska, B.; Starck, M.; Erhardt, M.; Römer, I.; Meyer Zu Heringdorf, D.; Pfeilschifter, J.; Zangemeister-Wittke, U.; Huwiler, A. Downregulation of the S1P transporter spinster homology protein 2 (Spns2) exerts an anti-fibrotic and anti-inflammatory effect in human renal proximal tubular epithelial cells. Int. J. Mol. Sci. 2018, 19, 1498. [Google Scholar] [CrossRef] [Green Version]
- Bajwa, A.; Huang, L.; Kurmaeva, E.; Ye, H.; Dondeti, K.R.; Chroscicki, P.; Foley, L.S.; Balogun, Z.A.; Alexander, K.J.; Park, H.; et al. Sphingosine kinase 2 deficiency attenuates kidney fibrosis via IFN-γ. J. Am. Soc. Nephrol. 2017, 28, 1145–1161. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, M.; Thangada, S.; Dasgupta, O.; Khanna, K.M.; Yamase, H.T.; Kashgarian, M.; Hla, T.; Shapiro, L.H.; Ferrer, F.A. Cell-intrinsic sphingosine kinase 2 promotes macrophage polarization and renal inflammation in response to unilateral ureteral obstruction. PLoS ONE 2018, 13, e0194053. [Google Scholar] [CrossRef]
- Shiohira, S.; Yoshida, T.; Sugiura, H.; Nishida, M.; Nitta, K.; Tsuchiya, K. Sphingosine-1-phosphate acts as a key molecule in the direct mediation of renal fibrosis. Physiol. Rep. 2013, 1, e00172. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Fan, M.; Jin, Y.; Wang, H.; Ding, L.; Fan, J.; Gu, S.; Xu, W. Sphingosine kinase 1 inhibition decreases the epithelial-mesenchymal transition and ameliorates renal fibrosis via modulating NF-κB signaling. Am. J. Transl. Res. 2019, 11, 5879–5887. [Google Scholar] [PubMed]
- Richter, K.; Konzack, A.; Pihlajaniemi, T.; Heljasvaara, R.; Kietzmann, T. Redox-fibrosis: Impact of TGFβ1 on ROS generators, mediators and functional consequences. Redox Biol. 2015, 6, 344–352. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Liu, W.; Lan, T.; Xie, X.; Peng, J.; Huang, J.; Wang, S.; Shen, X.; Liu, P.; Huang, H. Berberine reduces fibronectin expression by suppressing the S1P-S1P2 receptor pathway in experimental diabetic nephropathy models. PLoS ONE 2012, 7, e43874. [Google Scholar] [CrossRef]
- Lahiri, S.; Park, H.; Laviad, E.L.; Lu, X.; Bittman, R.; Futerman, A.H. Ceramide synthesis is modulated by the sphingosine analog FTY720 via a mixture of uncompetitive and noncompetitive inhibition in an Acyl-CoA chain length-dependent manner. J. Biol. Chem. 2009, 284, 16090–16098. [Google Scholar] [CrossRef] [Green Version]
- Mandala, S.M.; Thornton, R.; Galve-Roperh, I.; Poulton, S.; Peterson, C.; Olivera, A.; Bergstrom, J.; Kurtz, M.B.; Spiegel, S. Molecular cloning and characterization of a lipid phosphohydrolase that degrades sphingosine-1- phosphate and induces cell death. Proc. Natl. Acad. Sci. USA 2000, 97, 7859–7864. [Google Scholar] [CrossRef] [Green Version]
- Santacreu, B.J.; Pescio, L.G.; Romero, D.J.; Corradi, G.R.; Sterin-Speziale, N.; Favale, N.O. Sphingosine kinase and sphingosine-1-phosphate regulate epithelial cell architecture by the modulation of de novo sphingolipid synthesis. PLoS ONE 2019, 14, e0213917. [Google Scholar] [CrossRef]
- Nieto, F.L.; Pescio, L.G.; Favale, N.O.; Adamo, A.M.; Sterin-Speziale, N.B. Sphingolipid metabolism is a crucial determinant of cellular fate in nonstimulated proliferating Madin-Darby canine kidney (MDCK) cells. J. Biol. Chem. 2008, 283, 25682–25691. [Google Scholar] [CrossRef] [Green Version]
- Kohama, T.; Olivera, A.; Edsall, L.; Nagiec, M.M.; Dickson, R.; Spiegel, S. Molecular cloning and functional characterization of murine sphingosine kinase. J. Biol. Chem. 1998, 273, 23722–23728. [Google Scholar] [CrossRef] [Green Version]
- Gault, C.R.; Eblen, S.T.; Neumann, C.A.; Hannun, Y.A.; Obeid, L.M. Oncogenic K-Ras regulates bioactive sphingolipids in a sphingosine kinase 1-dependent manner. J. Biol. Chem. 2012, 287, 31794–31803. [Google Scholar] [CrossRef] [Green Version]
- Tominaga, T.; Sharma, I.; Fujita, Y.; Doi, T.; Wallner, A.K.; Kanwar, Y.S. Myo-inositol oxygenase accentuates renal tubular injury initiated by endoplasmic reticulum stress. Am. J. Physiol. Renal Physiol. 2019, 316, F301–F315. [Google Scholar] [CrossRef] [PubMed]
- Aslan, M.; Elpek, Ö.; Akkaya, B.; Balaban, H.T.; Afşar, E. Organ function, sphingolipid levels and inflammation in tunicamycin induced endoplasmic reticulum stress in male rats. Hum. Exp. Toxicol. 2021, 40, 259–273. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Chen, C.; Peng, Z.; Brown, B.C.; Reisz, J.A.; Xu, P.; Zhou, Z.; Song, A.; Zhang, Y.; Bogdanov, M.V.; et al. Erythrocyte metabolic reprogramming by sphingosine 1-phosphate in chronic kidney disease and therapies. Circ. Res. 2020, 127, 360–375. [Google Scholar] [CrossRef] [PubMed]
- Jessup, C.F.; Bonder, C.S.; Pitson, S.M.; Coates, P.T.H. The sphingolipid rheostat: A potential target for improving pancreatic islet survival and function. Endocr. Metab. Immune Disord. Drug Targets 2011, 11, 262–272. [Google Scholar] [CrossRef]
- Syed, I.; Szulc, Z.M.; Ogretmen, B.; Kowluru, A. L- threo-C6-pyridinium-ceramide bromide, a novel cationic ceramide, induces NADPH oxidase activation, mitochondrial dysfunction and loss in cell viability in INS 832/13 β-cells. Cell. Physiol. Biochem. 2012, 30, 1051–1058. [Google Scholar] [CrossRef]
- Qi, Y.; Chen, J.; Lay, A.; Don, A.; Vadas, M.; Xia, P. Loss of sphingosine kinase 1 predisposes to the onset of diabetes via promoting pancreatic β-cell death in diet-induced obese mice. FASEB J. 2013, 27, 4294–4304. [Google Scholar] [CrossRef]
- Bruce, C.R.; Risis, S.; Babb, J.R.; Yang, C.; Kowalski, G.M.; Selathurai, A.; Lee-Young, R.S.; Weir, J.M.; Yoshioka, K.; Takuwa, Y.; et al. Overexpression of sphingosine kinase 1 prevents ceramide accumulation and ameliorates muscle insulin resistance in high-fat diet-fed mice. Diabetes 2012, 61, 3148–3155. [Google Scholar] [CrossRef] [Green Version]
- Haass, N.K.; Nassif, N.; McGowan, E.M. Switching the sphingolipid rheostat in the treatment of diabetes and cancer comorbidity from a problem to an advantage. BioMed Res. Int. 2015, 2015, 165105. [Google Scholar] [CrossRef] [Green Version]
- Nègre-Salvayre, A.; Augé, N.; Camaré, C.; Bacchetti, T.; Ferretti, G.; Salvayre, R. Dual signaling evoked by oxidized LDLs in vascular cells. Free Radic. Biol. Med. 2017, 106, 118–133. [Google Scholar] [CrossRef]
- Ishimaru, K.; Yoshioka, K.; Kano, K.; Kurano, M.; Saigusa, D.; Aoki, J.; Yatomi, Y.; Takuwa, N.; Okamoto, Y.; Proia, R.L.; et al. Sphingosine kinase-2 prevents macrophage cholesterol accumulation and atherosclerosis by stimulating autophagic lipid degradation. Sci. Rep. 2019, 9, 18329. [Google Scholar] [CrossRef]
- Schulz, M.E.; Katunaric, B.; Hockenberry, J.C.; Gutterman, D.D.; Freed, J.K. Manipulation of the sphingolipid rheostat influences the mediator of flow-induced dilation in the human microvasculature. J. Am. Heart Assoc. 2019, 8, e013153. [Google Scholar] [CrossRef] [PubMed]
- Fenger, M.; Linneberg, A.; Jørgensen, T.; Madsbad, S.; Søbye, K.; Eugen-Olsen, J.; Jeppesen, J. Genetics of the ceramide/sphingosine-1-phosphate rheostat in blood pressure regulation and hypertension. BMC Genet. 2011, 12, 44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skácel, J.; Slusher, B.S.; Tsukamoto, T. Small molecule inhibitors targeting biosynthesis of ceramide, the central hub of the sphingolipid network. J. Med. Chem. 2021, 64, 279–297. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Park, C.W. Mechanisms of adiponectin action: Implication of adiponectin receptor agonism in diabetic kidney disease. Int. J. Mol. Sci. 2019, 20, 1782. [Google Scholar] [CrossRef] [Green Version]
- Kim, M.; Kim, M.; Kim, N.; D’Agati, V.D.; Emala Sr, C.W.; Lee, H.T. Isoflurane mediates protection from renal ischemia-reperfusion injury via sphingosine kinase and sphingosine-1-phosphate-dependent pathways. Am. J. Physiol. Renal Physiol. 2007, 293, F1827–F1835. [Google Scholar] [CrossRef] [Green Version]
- Schwalm, S.; Beyer, S.; Hafizi, R.; Trautmann, S.; Geisslinger, G.; Adams, D.R.; Pyne, S.; Pyne, N.; Schaefer, L.; Huwiler, A.; et al. Validation of highly selective sphingosine kinase 2 inhibitors SLM6031434 and HWG-35D as effective anti-fibrotic treatment options in a mouse model of tubulointerstitial fibrosis. Cell. Signal. 2021, 79, 109881. [Google Scholar] [CrossRef]
- Awad, A.S.; Ye, H.; Huang, L.; Li, L.; Foss, F.W., Jr.; Macdonald, T.L.; Lynch, K.R.; Okusa, M.D. Selective sphingosine 1-phosphate 1 receptor activation reduces ischemia-reperfusion injury in mouse kidney. Am. J. Physiol. Renal Physiol. 2006, 290, F1516–F1524. [Google Scholar] [CrossRef] [Green Version]
- Sugahara, K.; Maeda, Y.; Shimano, K.; Murase, M.; Mochiduki, S.; Takemoto, K.; Kakimoto, T.; Utsumi, H.; Oshita, K.; Kataoka, H. Amiselimod (MT-1303), a novel sphingosine 1-phosphate receptor-1 modulator, potently inhibits the progression of lupus nephritis in two murine SLE models. J. Immunol. Res. 2019, 2019, 5821589. [Google Scholar] [CrossRef] [Green Version]
- Meadows, K.R.T.; Steinberg, M.W.; Clemons, B.; Stokes, M.E.; Opiteck, G.J.; Peach, R.; Scott, F.L. Ozanimod (RPC1063), a selective S1PR1 and S1PR5 modulator, reduces chronic inflammation and alleviates kidney pathology in murine systemic lupus erythematosus. PLoS ONE 2018, 13, e0193236. [Google Scholar]
- Wenderfer, S.E.; Stepkowski, S.M.; Braun, M.C. Increased survival and reduced renal injury in MRL/lpr mice treated with a novel sphingosine-1-phosphate receptor agonist. Kidney Int. 2008, 74, 1319–1326. [Google Scholar] [CrossRef] [Green Version]
- Kurano, M.; Tsuneyama, K.; Morimoto, Y.; Nishikawa, M.; Yatomi, Y. Apolipoprotein M suppresses the phenotypes of IgA nephropathy in hyper-IgA mice. FASEB J. 2019, 33, 5181–5195. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Lan, T.; Xie, X.; Huang, K.; Peng, J.; Huang, J.; Shen, X.; Liu, P.; Huang, H. S1P2 receptor mediates sphingosine-1-phosphate-induced fibronectin expression via MAPK signaling pathway in mesangial cells under high glucose condition. Exp. Cell. Res. 2012, 318, 936–943. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Xiang, H.; Chen, R.; Yang, J.; Yang, X.; Zhou, J.; Liu, H.; Zhao, S.; Xiao, J.; Chen, P.; et al. S1PR2 antagonist ameliorate high glucose-induced fission and dysfunction of mitochondria in HRGECs via regulating ROCK1. BMC Nephrol. 2019, 20, 135. [Google Scholar] [CrossRef] [PubMed]
- Gholamnezhadjafari, R.; Falak, R.; Tajik, N.; Aflatoonian, R.; Keshtkar, A.A.; Rezaei, A. Effect of FTY720 (fingolimod) on graft survival in renal transplant recipients: A systematic review protocol. BMJ. Open 2016, 6, e010114. [Google Scholar] [CrossRef]
- Ettenger, R.; Schmouder, R.; Kovarik, J.M.; Bastien, M.C.; Hoyer, P.F. Pharmacokinetics, pharmacodynamics, safety, and tolerability of single-dose fingolimod (FTY720) in adolescents with stable renal transplants. Pediatr. Transplant. 2011, 15, 406–413. [Google Scholar] [CrossRef]
- Zywno, H.; Bzdega, W.; Kolakowski, A.; Kurzyna, P.; Harasim-Symbor, E.; Sztolsztener, K.; Chabowski, A.; Konstantynowicz-Nowicka, K. The influence of coumestrol on sphingolipid signaling pathway and insulin resistance development in primary rat hepatocytes. Biomolecule 2021, 11, 268. [Google Scholar] [CrossRef]
- Hodun, K.; Sztolsztener, K.; Chabowski, A. Antioxidants supplementation reduces ceramide synthesis improving the cardiac insulin transduction pathway in a rodent model of obesity. Nutrients 2021, 13, 3413. [Google Scholar] [CrossRef]
- Li, G.; Zhang, Q.; Hong, J.; Ritter, J.K.; Li, P.L. Inhibition of pannexin-1 channel activity by adiponectin in podocytes: Role of acid ceramidase activation. Biochim. Biophys. Acta Mol. Cell. Biol. Lipids 2018, 863, 1246–1256. [Google Scholar] [CrossRef]
- Förster, A.; Emmler, T.; Schwalm, S.; Ebadi, M.; Heringdorf, D.M.; Nieuwenhuis, B.; Kleuser, B.; Huwiler, A.; Pfeilschifter, J. Glucocorticoids protect renal mesangial cells from apoptosis by increasing cellular sphingosine-1-phosphate. Kidney Int. 2010, 77, 870–879. [Google Scholar] [CrossRef] [Green Version]



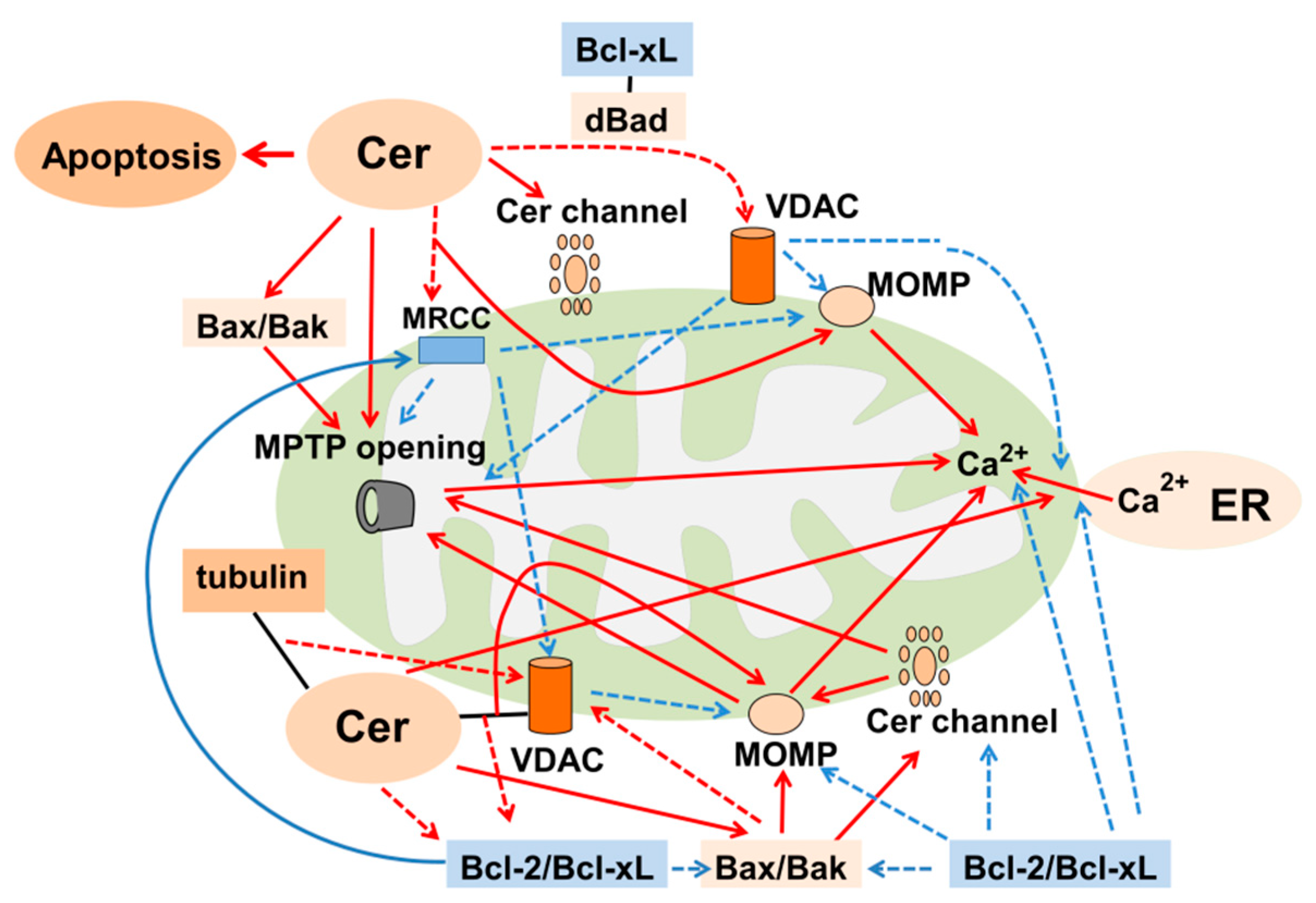

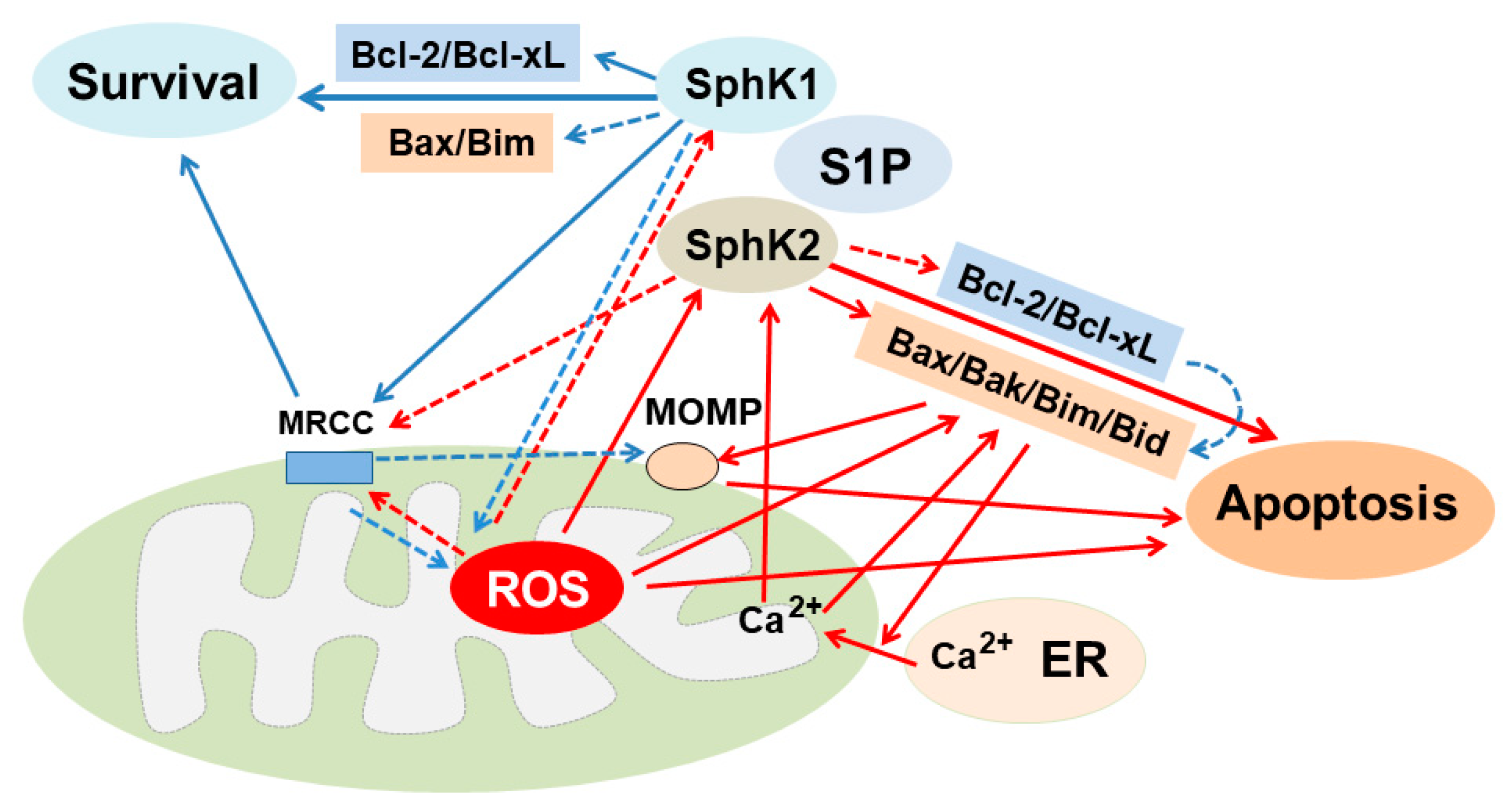


| Enzymes | Localization | References |
|---|---|---|
| SPT | kidney, podocyte, RTC, GEC | [14,15] |
| DES | kidney, RTC | [16,17] |
| SMS1/2 | kidney, RTC, EC | [18,19,20,21] |
| aSMase | kidney, podocyte, glomerulus, MC, RTC, GEC, EC | [22,23,24,25,26,27,28,29,30,31] |
| nSMase | kidney, MC, RTC, EC | [22,24,25,28,30,31,33,34] |
| SMPDL3b | kidney, glomeruli, podocyte, GEC | [13,15,35] |
| CerK | kidney, podocyte, MC, RTC, GEC | [13,15,36,37] |
| CerS1 | RTC, EC | [38,39,40] |
| CerS2 | kidney, RTC | [40,41] |
| CerS4 | kidney, RTC | [40,41] |
| CerS5 | kidney, RTC | [40,42] |
| CerS6 | kidney, RTC | [40,41] |
| aCDase | kidney, MC, podocyte, GEC | [24,35,43,44] |
| nCDase | kidney, glomerulus, podocyte, MC, RTC | [24,25,35,45,46,47] |
| alkaline CDase | MC, podocyte | [15,35,48] |
| SphK1 | kidney, podocyte, MC, RTC, EC | [17,34,49,51,52,55] |
| SphK2 | kidney, podocyte, MC, RTC, EC | [17,50,51,53,54,55] |
| Oxidative Kidney Diseases | Cer Levels in Kidney, Plasma/Serum and Urine | Enzymes for Cer Production | Kidney Cell Response | References | |
|---|---|---|---|---|---|
| Toxic nephropathy | RTC | ↑ | CerS↑ | Apoptosis | [62] |
| Cadmium | |||||
| Carbon tetrachloride | Kidney, plasma | ↑ | nSMase↑, aSMase↓ | Apoptosis/necrosis | [31,63] |
| Chromium | RTC | ↑ | SMPD2↑ | Autophagy | [64] |
| Cisplatin | Kidney, BMK | ↑ | SPT↑, CerS↑, aSMae↑, GCS↑ | Apoptosis/necrosis | [65,66] |
| Nickel | RTC | ↑ | CerS↑, GCS↑ | Apoptosis | [67] |
| UV-irradiation | RTC, BMK | ↑ | SPT↑, aSMase↑, CerS↑ | Apoptosis | [27,65] |
| Radiation | Podocyte | ↑ | SMPDL3b↓, nCDase↓, aCDase→, alkaline CDase→ | Apoptosis | [35] |
| EC | ↑ | aSMase↑ | Apoptosis | [68,69,70] | |
| GEC | ↑ | SMPDL3b↑, CerK→ | Apoptosis | [13] | |
| Radiocontrasts | RTC | ↑ | CerS↑ | Apoptosis | [71] |
| Oxalate nephrolithiasis | RTC | ↑ | SMase↑ | Apoptosis/necrosis | [72,73] |
| Hypermocysteinemia | Kidney, glomerulus, Podocyte | ↑ | aSMase↑, SPT↑ | MME/necrosis/sclerosis/ fibrosis | [26,74,75] |
| Myohemoglobinuria | Kidney, RTC | ↑ | aSMase↓ nSMase↓, CerS↑ | Necrosis | [22] |
| Ischemia/reperfusion | Kidney, RTC | ↑ | aSMase↓, nSMase↓, CerS↑ | Apoptosis/necrosis | [22,76,77,78] |
| EC | ↑ | SMase↑ | Necrosis | [79] | |
| Unilateral ureteral obstruction | Kidney | NA | CerS↑ | Apoptosis | [71] |
| Kidney | ↑ | UN | Apoptosis | [80,81] | |
| Kidney | LCCer↓ | UN | Fibrosis | [82] | |
| Anti-GBM Ab GN | Kidney | ↑ | aSMase↑, nSMase↑ | Necrosis | [22] |
| Nephrotic syndrome | Glomerulus, podocyte | ↑ | aSMase↑, aCDase↓ | FPE | [44] |
| Podocyte, fibroblast | ↑ | SGPL1↓, CerS2↑ | FPE, FSGS | [83] | |
| Podocyte, serum | ↑ | SGPL1↓ | FPE, FSGS | [84] | |
| Chronic GN | Plasma/serum | ↑ | UN | Necrosis | [90,91] |
| Lupus nephritis | Plasma/serum | ↑ | UN | Necrosis | [92] |
| Plasma | ↑ | CerS5↑ | Necrosis | [93] | |
| Plasma | → | UN | Necrosis | [94] | |
| Diabetic nephropathy | Kidney | ↑ | aCDase↓ | Apoptosis | [43] |
| MC | → | nCDase↑,↓ | Proliferation | [47] | |
| Kidney, RTC | ↑ | SPT↑, aSMase→ | Apoptosis | [85] | |
| Podocyte | ↑ | SPT↑ | Apoptosis | [86] | |
| Kidney | ↑ | UN | NA | [97] | |
| Kidney | ↓ | CerS5↓, nSMase↓, alkaline CDase↑ | MME | [98] | |
| EC | ↑ | aSMase↑, CerS↑ | Apoptosis/ necrosis | [87,88,89] | |
| Plasma | ↑ | UN | NA | [96] | |
| Plasma | ↑ | UN | MME/necrosis | [98,99] | |
| Plasma | VLCer↓ | UN | Necrosis | [101] | |
| Urine | ↑ | UN | Necrosis | [102,103] | |
| Targeted Enzyme | Effector (Function) | Model/Oxidant Stimuli | Tissue/Cell Type/Plasma | Effects | References |
|---|---|---|---|---|---|
| SPT | Myriosin (inhibitor) | Cisplatin nephropathy | kidney | Protects against apoptosis/necrosis | [66] |
| hHcys | kidney | Protects against proteinuria and necrosis | [74] | ||
| DN | kidney/podocyte | Prevents ROS production, albuminuria and apoptosis | [86] | ||
| Coumesterol (inhibitor) | DM | hepatocyte | Improves insulin resistance | [296] | |
| NAC * (inhibitor) | obesity/DM | plasma/cardiocyte | Improves insulin resistance | [297] | |
| CerS | Fumonisin B1 (inhibitor) | Cadmium nephropathy | RTC | Prevents apoptosis | [62] |
| Radiocontrast nephropathy | RTC | Prevents apoptosis | [71] | ||
| H/R | RTC | Prevents apoptosis/necrosis | [76,78] | ||
| CerS4,5 | Coumesterol (inhibitor) | DM | hepatocyte | Improves insulin resistance | [296] |
| CerS5 | NAC * (inhibitor) | Obesity/DM | plasma/cardiocyte | Improves insulin resistance | [297] |
| aSMase | Amitriptyline (inhibitor) | Cisplatin nephropathy | kidney | Prevents apoptosis/necrosis | [66] |
| hHcys | podocyte | Protects against ROS production and necrosis | [75] | ||
| Desipramin (inhibitor) | UV-irradiation nephropathy | RTC | Prevents apoptosis | [27] | |
| alSMase, nSMase | NAC* (inhibitor) | Obesity/DM | plasma/cardiocyte | Improves insulin resistance | [297] |
| aCDase | AdipoRon * (activator) | DN | kidney, podocyte, GEC | Increases S1P and protects against apoptosis | [43,284,298] |
| nCDase | AdipoRon * (activator) | H2O2 | EC | Improves EC function | [281] |
| Dexamethasone ** (activator) | Staurosporine, TNF-α | MC | Prevents apoptosis | [299] | |
| SphK1 | SKI-II (inhibitor) | DN | RTC | Prevents inflammation and fibrosis | [200] |
| Berberine (inhibitor) | DN | MC | Prevents fibrosis | [198] | |
| kidney | Prevents injury | [264] | |||
| Dimethylsphingosine (inhibitor) | DN | MC | Prevents fibrosis | [198] | |
| Dimethylsphingosine (inhibitor) | LDL/DN | MC | Prevents fibrosis | [219] | |
| Isoflurane (activator) | I/R | kidney | Prevents apoptosis | [285] | |
| Coumesterol * (activator) | DM | hepatocyte | Improves insulin resistance | [296] | |
| Dexamethasone (activator) | Staurosporine, TNF-α | MC | Prevents apoptosis | [299] | |
| SphK2 | SLM6031434 (inhibitor) | UUO | renal fibroblast | Prevents fibrosis | [286] |
| SLM6031434 (inhibitor) | DN | podocyte | Prevents podocyte function and fibrosis | [207] | |
| HWG-35D (inhibitor) | UUO | renal fibroblast | Prevents fibrosis | [286] | |
| SLP 120701 (inhibitor) | kidney | Prevents inflammation/fibrosis | [260] | ||
| ABC294640 (inhibitor) | LN | glomeruli | Prevents injury | [94] | |
| Coumesterol * (activator) | DM | hepatocyte | Improves insulin resistance | [296] | |
| S1PR1 | FTY720 (activator) | Cisplatin nephropathy | kidney | Prevents injury | [226] |
| FTY720 (activator) | I/R | kidney | Prevents injury | [287] | |
| SEW2871 (activator) | |||||
| Amiselimod (MT-1303) (activator) | LN | kidney | Prevents injury | [288] | |
| Ozanimid (RPC-1063) (activator) | [289] | ||||
| RP-101075 (activator) | [289] | ||||
| KRP-203 (activator) | Prevents proteinuria and apoptosis | [290] | |||
| S1PR2 | VPC23019 (inhibitor) | IgA GN | MC, RTC | Prevents cell proliferation and fibrosis | [291] |
| JTE-013 (inhibitor) | DN | MC | Prevents fibrosis | [292] | |
| GEC | Prevents ROS production and apoptosis | [293] | |||
| Beberine (inhibitor) | kidney, MC | Prevents fibrosis | [264] | ||
| S1PR1/3 | VPC23019 (inhibitor) | LDL/DN | MC | Prevents fibrosis | [219] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ueda, N. A Rheostat of Ceramide and Sphingosine-1-Phosphate as a Determinant of Oxidative Stress-Mediated Kidney Injury. Int. J. Mol. Sci. 2022, 23, 4010. https://doi.org/10.3390/ijms23074010
Ueda N. A Rheostat of Ceramide and Sphingosine-1-Phosphate as a Determinant of Oxidative Stress-Mediated Kidney Injury. International Journal of Molecular Sciences. 2022; 23(7):4010. https://doi.org/10.3390/ijms23074010
Chicago/Turabian StyleUeda, Norishi. 2022. "A Rheostat of Ceramide and Sphingosine-1-Phosphate as a Determinant of Oxidative Stress-Mediated Kidney Injury" International Journal of Molecular Sciences 23, no. 7: 4010. https://doi.org/10.3390/ijms23074010





