Macrophage Dysfunction in Autoimmune Rheumatic Diseases and Atherosclerosis
Abstract
:1. Introduction
- -
- The systemic effect of proinflammatory cytokines: interleukin (IL)-1β, IL-6, tumor necrosis factor (TNF)-α, and interferon (IFN)-γ;
- -
- Increased adhesion of activated neutrophils, monocytes, and platelets to the vascular endothelium under the influence of neutrophil chemokine (C-X-C motif) ligand 8 (CXCL8) or IL-8 and monocyte chemokine (C-C motif) ligand 2 (CCL2) or monocyte chemoattractant protein 1 (MCP-1);
- -
- Further activation of platelets by neutrophils/monocytes via proteinase-activated receptors (PARs) 1 and 4 and anticitrullinated protein antibodies (ACPAs);
- -
- Activation of vascular endothelial PAR-1 by adherent neutrophils/macrophages, exacerbating systemic inflammation and endothelial dysfunction;
- -
- Chronic low-grade inflammation contributing to proatherogenic oxidized low-density lipoprotein (ox-LDL) modification;
- -
- Neutrophil effect on activated platelets with the intravascular formation of a neutrophil extracellular trap (NET), which maintains intravascular proinflammatory potential through the expression of tissue factor, endothelium-activating proteases, and histones.
2. Macrophage Polarization
3. Macrophage Dysfunction in Atherosclerosis
4. Macrophage Dysfunction and Autoimmune Rheumatic Diseases
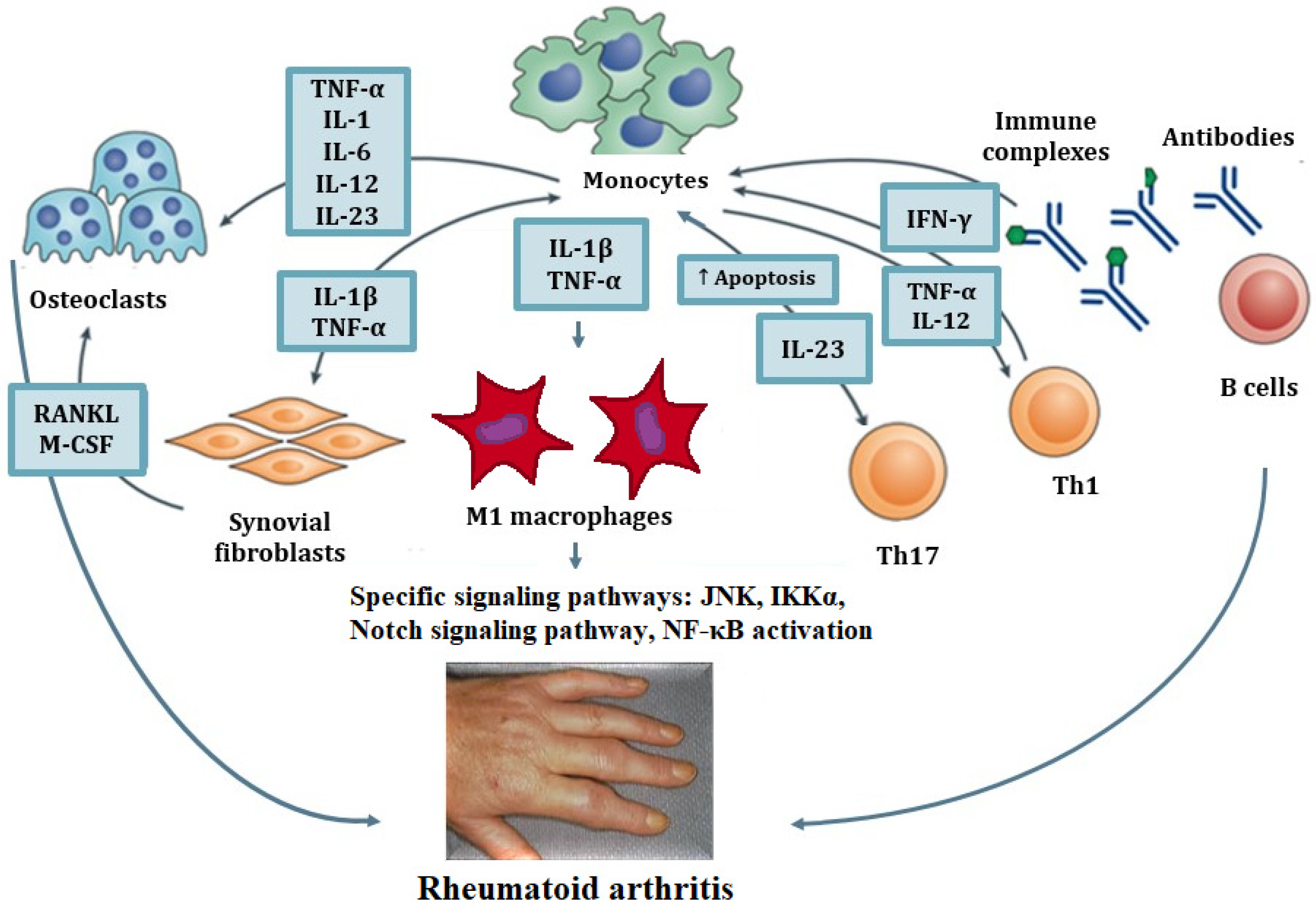
4.1. Macrophage M1/M2 Polarization in Rheumatoid Arthritis
4.2. Macrophage Dysfunction in Systemic Lupus Erythematosus
4.3. Macrophage Dysfunction in Systemic Sclerosis
4.4. Macrophage and Sjögren’s Syndrome
5. Macrophages and Accelerated Atherosclerosis in ARDs
6. Antirheumatic Drugs and Macrophages
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Abbreviations
| ACPAs | anticitrullinated protein antibodies |
| ARDs | autoimmune rheumatic diseases |
| ATP | adenosine triphosphate |
| CAD | coronary artery disease |
| CCL2 | chemokine (C-C motif) ligand 2 |
| CHB | congenital heart block |
| CRP | C-reactive protein |
| CSF-1 | colony-stimulating factor 1 |
| CTGF | connective tissue growth factor |
| CVCs | cardiovascular complications |
| CVDs | cardiovascular diseases |
| CVR | cardiovascular risk |
| CXCL8 | chemokine (C-X-C motif) ligand 8 |
| ESR | erythrocyte sedimentation rate |
| GM-CSF | granulocyte/monocyte colony-stimulating factor |
| GSK3b | glycogen synthase kinase 3b |
| HC | healthy controls |
| HSPCs | hematopoietic stem and progenitor cells |
| IFN-γ | interferon-gamma |
| IKKα | IκB kinase alpha |
| IL | interleukin |
| IRF5 | interferon regulatory factor 5 |
| ITGB1 | integrin beta-1 |
| JNK | C-Jun N-terminal kinase |
| LPS | lipopolysaccharide |
| LXR | liver X receptor |
| M1 | classically activated macrophages |
| M2 | alternatively activated macrophages |
| MCP-1 | monocyte chemoattractant protein 1 |
| M-CSF | macrophage colony-stimulating factor |
| MDM | monocyte-derived macrophages |
| MIP-1α | macrophage inflammatory protein 1 alpha |
| MMP | matrix metalloproteinase |
| MP-IC | microparticles immune complexes |
| NET | neutrophil extracellular trap |
| NF-κB | nuclear factor kappa-B |
| NLRP | NLR family pyrin domain-containing 3 |
| OA | osteoarthritis |
| OC | osteoclast |
| Ox-LDL | oxidized low-density lipoprotein |
| PAR | proteinase-activated receptors |
| RA | rheumatoid arthritis |
| RANKL | receptor activator of nuclear factor kappa-B (NF-κB) ligand |
| RF | rheumatoid Factor |
| Sema3A | semaphorin 3A |
| Siglec-1 | sialic acid-binding Ig-like lectin 1 |
| SIR1 | silent information regulator 1 proteins |
| SIRT | sirtuins |
| SLE | systemic lupus erythematosus |
| sPAP | systolic pulmonary artery pressure |
| SSc | systemic sclerosis |
| STAT6 | signal transducer and activator of transcription |
| TGF-β | transforming growth factor-beta |
| Th | T-helper |
| TLR | toll-like receptor |
| TNF-α | tumoral necrosis factor-alpha |
| VEGF | vascular endothelial growth factor |
References
- Agca, R.; Heslinga, S.C.; van Halm, V.P.; Nurmohamed, M.T. Atherosclerotic cardiovascular disease in patients with chronic inflammatory joint disorders. Heart 2016, 102, 790–795. [Google Scholar] [CrossRef] [PubMed]
- Van den Hoek, J.; Boshuizen, H.C.; Roorda, L.D.; Tijhuis, G.J.; Nurmohamed, M.T.; van den Bos, G.A.M.; Dekker, J. Mortality in patients with rheumatoid arthritis: A 15-year prospective cohort study. Rheumatol. Int. 2017, 37, 487–493. [Google Scholar] [CrossRef] [Green Version]
- Mahtta, D.; Gupta, A.; Ramsey, D.J.; Rifai, M.A.; Mehta, A.; Krittanawong, C.; Lee, M.T.; Nasir, K.; Samad, Z.; Blumenthal, R.S.; et al. Autoimmune Rheumatic Diseases and Premature Atherosclerotic Cardiovascular Disease: An Analysis From the VITAL Registry. Am. J. Med. 2020, 133, 1424–1432. [Google Scholar] [CrossRef] [PubMed]
- Martinez, S.C.; Mohamed, M.; Potts, J.; Abhishek, A.; Roddy, E.; Savage, M.; Bharadwaj, A.; Kwok, C.S.; Bagur, R.; Mamas, M.A. Percutaneous coronary intervention outcomes in patients with rheumatoid arthritis, systemic lupus erythematosus and systemic sclerosis. Rheumatology 2020, 59, 2512–2522. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, M.; Wang, M.; Tai, Y.; Tao, J.; Zhou, W.; Han, Y.; Wei, W. Triggers of Cardiovascular Diseases in Rheumatoid Arthritis. Curr. Probl. Cardiol. 2021, 6, 100853. [Google Scholar]
- Avina-Zubieta, J.A.; Thomas, J.; Sadatsafavi, M.; Lehman, A.J.; Lacaille, D. Risk of incident cardiovascular events in patients with rheumatoid arthritis: A meta-analysis of observational studies. Ann. Rheum. Dis. 2012, 71, 1524–1529. [Google Scholar] [CrossRef]
- Dadoun, S.; Zeboulon-Ktorza, N.; Combescure, C.; Elhai, M.; Rozenberg, S.; Gossec, L.; Fautrel, B. Mortality in rheumatoid arthritis over the last fifty years: Systematic review and meta-analysis. Jt. Bone Spine 2013, 80, 29–33. [Google Scholar] [CrossRef]
- Buleu, F.; Sirbu, E.; Caraba, A.; Dragan, S. Heart Involvement in Inflammatory Rheumatic Diseases: A Systematic Literature Review. Medicina 2019, 55, 249. [Google Scholar] [CrossRef] [Green Version]
- Nasonov, E.L.; Popkova, T.V. Atherosclerosis: Perspectives of anti-inflammatory therapy. Ter. Arkhiv (Ter. Arkh.) 2018, 90, 4–12. (In Russian) [Google Scholar]
- Kurmann, R.D.; Mankad, R. Atherosclerotic vascular disease in the autoimmune rheumatologic woman. Clin. Cardiol. 2018, 41, 258–263. [Google Scholar] [CrossRef]
- Dadonienė, J.; Charukevič, G.; Jasionytė, G.; Staškuvienė, K.; Miltinienė, D. Mortality in Inflammatory Rheumatic Diseases: Lithuanian National Registry Data and Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 12338. [Google Scholar] [CrossRef]
- Hedar, A.M.; Stradner, M.H.; Roessler, A.; Goswami, N. Autoimmune Rheumatic Diseases and Vascular Function: The Concept of Autoimmune Atherosclerosis. J. Clin. Med. 2021, 10, 4427. [Google Scholar] [CrossRef] [PubMed]
- Ambrosino, P.; Lupoli, R.; Di Minno, A.; Tasso, M.; Peluso, R.; Di Minno, M.N. Subclinical atherosclerosis in patients with rheumatoid arthritis. A meta-analysis of literature studies. Thromb. Haemost. 2015, 113, 916–930. [Google Scholar] [CrossRef]
- Sciarra, I.; Vasile, M.; Carboni, A.; Stefanantoni, K.; Iannace, N.; Angelelli, C.; Scarno, A.G.; Valesini, G.; Riccieri, V. Subclinical atherosclerosis in systemic sclerosis: Different risk profiles among patients according to clinical manifestations. Int. J. Rheum. Dis. 2021, 24, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Gerasimova, E.V.; Popkova, T.V.; Gerasimova, D.A.; Glukhova, S.I.; Nasonov, E.L.; Lila, A.M. Application of cardiovascular risk scales to identify carotid atherosclerosis in patients with rheumatoid arthritis. Ter. Arkhiv (Ter. Arkh.). 2021, 93, 561–567. (In Russian) [Google Scholar] [CrossRef]
- Dalbeni, A.; Giollo, A.; Bevilacqua, M.; Cioffi, G.; Tagetti, A.; Cattazzo, F.; Orsolini, G.; Ognibeni, F.; Minuz, P.; Rossini, M.; et al. Traditional cardiovascular risk factors and residual disease activity are associated with atherosclerosis progression in rheumatoid arthritis patients. Hypertens. Res. 2020, 43, 922–928. [Google Scholar] [CrossRef] [PubMed]
- Meyer, P.W.; Anderson, R.; Ker, J.A.; Ally, M.T. Rheumatoid arthritis and risk of cardiovascular disease. Cardiovasc. J. Afr. 2018, 29, 317–321. [Google Scholar] [CrossRef] [Green Version]
- Atzeni, F.; Nucera, V.; Gerratana, E.; Fiorenza, A.; Gianturco, L.; Corda, M.; Sarzi-Puttini, P. Cardiovascular Consequences of Autoimmune Rheumatic Diseases. Curr. Vasc. Pharmacol. 2020, 18, 566–579. [Google Scholar] [CrossRef]
- Kennedy, A.; Fearon, U.; Veale, D.J.; Godson, C. Macrophages in synovial inflammation. Front. Immunol. 2011, 2, 52. [Google Scholar] [CrossRef] [Green Version]
- Yunna, C.; Mengru, H.; Lei, W.; Weidong, C. Macrophage M1/M2 polarization. Eur. J. Pharmacol. 2020, 877, 173090. [Google Scholar] [CrossRef]
- Sica, A.; Mantovani, A. Macrophage plasticity and polarization: In vivo veritas. J. Clin. Investig. 2012, 122, 787–795. [Google Scholar] [CrossRef]
- Stein, M.; Keshav, S.; Harris, N.; Gordon, S. Interleukin 4 potently enhances murine macrophage mannose receptor activity: A marker of alternative immunologic macrophage activation. J. Exp. Med. 1992, 176, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S. Alternative activation of macrophages. Nat. Rev. Immunol. 2003, 3, 23–35. [Google Scholar] [CrossRef]
- Hao, N.B.; Lü, M.H.; Fan, Y.H. Macrophages in tumor microenvironments and the progression of tumor. Clin. Dev. Immunol. 2012, 2012, 948098. [Google Scholar]
- Gratchev, A.; Kzhyshkowska, J.; Köthe, K.; Muller-Molinet, I.; Kannookadan, S.; Utikal, J.; Goerdtet, S. Mphi1 and Mphi2 can be re-polarized by Th2 or Th1 cytokines, respectively, and respond to exogenous danger signals. Immunobiology 2006, 211, 473–486. [Google Scholar] [CrossRef]
- Tatano, Y.; Shimizu, T.; Tomioka, H. Unique macrophages different from M1/M2 macrophages inhibit T cell mitogenesis while upregulating Th17 polarization. Sci. Rep. 2014, 4, 4146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mantovani, A.; Sica, A.; Sozzani, S.; Vecchi, A.; Locati, M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 2004, 25, 677–686. [Google Scholar] [CrossRef]
- Graff, J.W.; Dickson, A.M.; Clay, G.; McCaffrey, A.P.; Wilson, M.E. Identifying functional microRNAs in macrophages with polarized phenotypes. J. Biol. Chem. 2012, 286, 21816–21825. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spiller, K.L.; Anfang, R.R.; Spiller, K.J. The role of macrophage phenotype in vascularization of tissue engineering scaffolds. Biomaterials 2014, 35, 4477–4488. [Google Scholar] [CrossRef] [Green Version]
- Arabpour, M.; Saghazadeh, A.; Rezaei, N. Anti-inflammatory and M2 macrophage polarization-promoting effect of mesenchymal stem cell-derived exosomes. Int. Immunopharmacol. 2021, 97, 107823. [Google Scholar] [CrossRef]
- Barrett, T.J. Macrophages in Atherosclerosis Regression. Arter. Thromb. Vasc. Biol. 2020, 40, 20–33. [Google Scholar] [CrossRef]
- Bories, G.; Leitinger, N. Macrophage metabolism in atherosclerosis. FEBS Lett. 2017, 591, 3042–3060. [Google Scholar] [CrossRef] [Green Version]
- Riksen, N.P.; Stienstra, R. Metabolism of innate immune cells: Impact on atherosclerosis. Curr. Opin. Lipidol. 2018, 29, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Tabas, I.; Bornfeldt, K.E. Macrophage Phenotype and Function in Different Stages of Atherosclerosis. Circ. Res. 2016, 118, 653–667. [Google Scholar] [CrossRef] [Green Version]
- Kim, H. The transcription factor MafB promotes anti-inflammatory M2 polarization and cholesterol efflux in macrophages. Sci. Rep. 2017, 7, 7591. [Google Scholar] [CrossRef]
- Gong, M.; Zhuo, X.; Ma, A. STAT6 Upregulation Promotes M2 Macrophage Polarization to Suppress Atherosclerosis. Med. Sci. Monit. Basic Res. 2017, 23, 240–249. [Google Scholar] [CrossRef]
- Rahman, K.; Vengrenyuk, Y.; Ramsey, S.A.; Vila, N.R.; Girgis, N.; Liu, J.; Gusarova, V.; Gromada, J.; Weinstock, A.; Moore, K.; et al. Inflammatory Ly6Chi monocytes and their conversion to M2 macrophages drive atherosclerosis regression. J. Clin. Investig. 2017, 127, 2904–2915. [Google Scholar] [CrossRef] [PubMed]
- Vats, D.; Mukundan, L.; Odegaard, J.I.; Zhang, L.; Smith, K.L.; Morel, C.R.; Wagner, R.A.; Greaves, D.; Murray, P.J.; Chawlaet, A. Oxidative metabolism and PGC-1beta attenuate macrophage-mediated inflammation. Cell Metab. 2006, 4, 13–24. [Google Scholar] [CrossRef] [Green Version]
- Stöger, J.L.; Gijbels, M.J.; van der Velden, S.; Manca, M.; van der Loos, C.M.; Biessen, E.A.; Daemen, M.J.; Lutgens, E.; de Winther, M.P. Distribution of macrophage polarization markers in human atherosclerosis. Atherosclerosis 2012, 225, 461–468. [Google Scholar] [CrossRef] [Green Version]
- Navegantes, K.C.; de Souza Gomes, R.; Pereira, P.A.T.; Czaikoski, P.G.; Azevedo, C.M.; Monteiro, M.C. Immune modulation of some autoimmune diseases: The critical role o macrophages and neutrophils in the innate and adaptive immunity. J. Transl. Med. 2017, 15, 36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atri, C.; Guerfali, F.Z.; Laouini, D. Role of human macrophage polarization in inflammation during infectious diseases. Int. J. Mol. Sci. 2018, 19, 1801. [Google Scholar] [CrossRef] [Green Version]
- Tardito, S.; Martinelli, G.; Soldano, S.; Paolino, S.; Pacini, G.; Patane, M.; Alessandri, E.; Smith, V.; Cutolo, M. Macrophage M1/M2 polarization and rheumatoid arthritis: A systematic review. Autoimmun. Rev. 2019, 18, 102397. [Google Scholar] [CrossRef] [Green Version]
- Maruotti, N.; Cantatore, F.P.; Crivellato, E.; Vacca, A.; Ribatti, D. Macrophages in rheumatoid arthritis. Histol. Histopathol. 2007, 22, 581–586. [Google Scholar] [PubMed]
- Wendling, D.; Abbas, W.; Godfrin-Valnet, M.; Kumar, A.; Guillot, X.; Khan, K.A.; Vidon, C.; Coquard, L.; Toussirot, E.; Prati, C.; et al. Dysregulated serum IL-23 and SIRT1 activity in peripheral blood mononuclear cells of patients with rheumatoid arthritis. PLoS ONE 2015, 10, e0119981. [Google Scholar] [CrossRef]
- Hannemann, N.; Apparailly, F.; Courties, G. New insights into macrophage heterogeneity in rheumatoid arthritis. Jt. Bone Spine 2021, 88, 105091. [Google Scholar] [CrossRef]
- Lu, M.C.; Lai, N.S.; Yin, W.Y.; Yu, C.H.; Huang, H.-B.; Tung, H.-C.; Huang, K.-Y.; Yu, H.-L. Anti-citrullinated protein antibodies activated ERK1/2 and JNK mitogen-activated protein kinases via binding to surface-expressed citrullinated GRP78 on mononuclear cells. J. Clin. Immunol. 2013, 33, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Fukui, S.; Iwamoto, N.; Takatani, A.; Igawa, T.; Shimizu, T.; Umeda, M.; Nishino, A.; Horai, Y.; Hirai, Y.; Kogaet, T.; et al. M1 and M2 monocytes in rheumatoid arthritis: A contribution of imbalance of M1/M2 monocytes to osteoclastogenesis. Front. Immunol. 2017, 8, 1958–1968. [Google Scholar] [CrossRef] [Green Version]
- Kokkonen, H.; Soderstrom, I.; Rocklov, J.; Hallmans, G.; Lejon, K.; Dahlqvistet, S.R. Up-regulation of cytokines and chemokines predates the onset of rheumatoid arthritis. Arthritis Rheum. 2010, 62, 383–391. [Google Scholar] [CrossRef]
- Kurowska, W.; Kuca-Warnawin, E.H.; Radzikowska, A.; Maśliński, W. The role of anticitrullinated protein antibodies (ACPA) in the pathogenesis of rheumatoid arthritis. Cent. J. Immunol. 2017, 42, 390–398. [Google Scholar] [CrossRef]
- Zhu, W.; Li, X.; Fang, S.; Zhang, X.; Wang, Y.; Zhang, T.; Li, Z.; Xu, Y.; Qu, S.; Liuet, C.; et al. Anti-Citrullinated Protein Antibodies Induce Macrophage Subset Disequilibrium in RA Patients. Inflammation 2015, 38, 2067–2075. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.; Park, S.H.; Chen, J.; Qiao, Y.; Giannopoulou, E.; Berg, K.; Hanidu, A.; Li, J.; Nabozny, G.; Kang, K.; et al. Interferon-γ represses M2 gene expression in human macrophages by disassembling enhancers bound by the transcription factor MAF. Immunity 2017, 47, 235–250. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Zheng, Z.; Lin, P.; Fu, X.; Li, F.; Jiang, J.; Zhu, P. ACPAs promote IL-1beta production in rheumatoid arthritis by activating the NLRP3 inflammasome. Cell. Mol. Immunol. 2020, 17, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Alvarado-Vazquez, P.A.; Bernal, L.; Paige, C.A.; Grosick, R.L.; Vilrriales, C.M.; Ferreira, D.W.; Ulecia-Morón, C.; Romero-Sandoval, E.A. Macrophage-specific nanotechnology-driven CD163 overexpression in human macrophages results in an M2 phenotype under inflammatory conditions. Immunobiology 2017, 222, 900–912. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.A.; Dickinson, A.K.; Taams, L.S. The Interplay Between Monocytes/Macrophages and CD4(+) T Cell Subsets in Rheumatoid Arthritis. Front. Immunol. 2015, 19, 571. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, T.; Takeda, S.; Xu, R.; Ochi, H.; Sunamura, S.; Sato, T.; Shibata, S.; Yoshida, Y.; Gu, Z.; Kimura, A.; et al. Sema3A regulates bone-mass accrual through sensory innervations. Nature 2013, 497, 490–493. [Google Scholar] [CrossRef]
- Teng, Y.; Yin, Z.; Li, J.; Li, K.; Li, X.; Zhang, Y. Adenovirus-mediated delivery of Sema3A alleviates rheumatoid arthritis in a serum-transfer induced mouse model. Oncotarget 2017, 8, 66270–66280. [Google Scholar] [CrossRef] [Green Version]
- Dongsheng, Z.; Zhiguang, F.; Junfeng, J.; Zifan, L.; Li, W. Cyclophilin A Aggravates Collagen-Induced Arthritis via Promoting Classically Activated Macrophages. Inflammation 2017, 40, 1761–1772. [Google Scholar] [CrossRef]
- Li, F.; Yang, Y.; Zhu, X.; Huang, L.; Xu, J. Macrophage Polarization Modulates Development of Systemic Lupus Erythematosus. Cell. Physiol. Biochem. 2015, 37, 1279–1288. [Google Scholar] [CrossRef]
- Funes, S.C.; Rios, M.; Escobar-Vera, J.; Kalergis, A.M. Implications of Macrophage Polarization in Autoimmunity. Immunology 2018, 154, 186–195. [Google Scholar] [CrossRef] [Green Version]
- Labonte, A.C.; Kegerreis, B.; Geraci, N.S.; Bachali, P.; Madamanchi, S.; Robl, R.; Catalina, M.; Lipsky, P.; Grammer, A. Identification of Alterations in Macrophage Activation Associated with Disease Activity in Systemic Lupus Erythematosus. PLoS ONE 2018, 13, e0208132. [Google Scholar] [CrossRef] [Green Version]
- Vilaiyuk, S.; Sirachainan, N.; Wanitkun, S.; Pirojsakul, K.; Vaewpanich, J. Recurrent macrophage activation syndrome as the primary manifestation in systemic lupus erythematosus and the benefit of serial ferritin measurements: A case-based review. Clin. Rheumatol. 2013, 32, 899–904. [Google Scholar] [CrossRef] [PubMed]
- Olmes, G.; Büttner-Herold, M.; Ferrazzi, F.; Distel, L.; Amann, K.; Daniel, C. CD163+ M2c-Like Macrophages Predominate in Renal Biopsies from Patients with Lupus Nephritis. Arthritis Res. Ther. 2016, 18, 90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sung, S.J.; Ge, Y.; Dai, C.; Wang, H.; Fu, S.M.; Sharma, R. Dependence of Glomerulonephritis Induction on Novel Intraglomerular Alternatively Activated Bone Marrow-Derived Macrophages and Mac-1 and PD-L1 in Lupus-Prone NZM2328 Mice. J. Immunol. 2017, 198, 2589–2601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iwata, Y.; Boström, E.; Menke, J.; Rabacal, W.; Morel, L.; Wada, T.; Kelley. Aberrant macrophages mediate defective kidney repair that triggers nephritis in lupus-susceptible mice. J. Immunol. 2012, 188, 4568–4580. [Google Scholar] [CrossRef]
- Schaper, F.; de Leeuw, F.; Horst, G.; Bootsma, H.; Limburg, P.C.; Heeringa, P.; Bijl, M.; Westra, J. High mobility group box 1 skews macrophage polarization and negatively influences phagocytosis of apoptotic cells. Rheumatology 2016, 55, 2260–2270. [Google Scholar] [CrossRef] [Green Version]
- Mohammadi, S.; Saghaeian-Jazi, M.; Sedighi, S.; Memarian, A. Immunomodulation in systemic lupus erythematosus: Induction of M2 population in monocyte-derived macrophages by pioglitazone. Lupus 2017, 26, 1318–1327. [Google Scholar] [CrossRef]
- Burbano, C.; Villar-Vesga, J.; Orejuela, J.; Muñoz, C.; Vanegas, A.; Vásquez, G.; Rojas, M.; Castaño, D. Potential involvement of platelet-derived microparticles and microparticles forming immune complexes during monocyte activation in patients with systemic lupus erythematosus. Front. Immunol. 2018, 9, 322. [Google Scholar] [CrossRef]
- Ahamada, M.M.; Jia, Y.; Wu, X. Macrophage Polarization and Plasticity in Systemic Lupus Erythematosus. Front. Immunol. 2021, 12, 734008. [Google Scholar] [CrossRef]
- Foocharoen, C.; Pussadhamma, B.; Mahakkanukrauh, A.; Suwannaroj, S.; Nanagara, R. Asymptomatic cardiac involvement in Thai systemic sclerosis: Prevalence and clinical correlations with non-cardiac manifestations (pre-liminary report). Rheumatology 2015, 54, 1616–1621. [Google Scholar] [CrossRef] [Green Version]
- Nihtyanova, S.I.; Denton, C.P. Pathogenesis of systemic sclerosis associated interstitial lung disease. J. Scleroderma Relat. Disord. 2020, 5 (Suppl. 2), 6–16. [Google Scholar] [CrossRef]
- Mohamed, M.E.; Gamal, R.M.; Hassan, A.T.; Hassan, A.; Abozaid, H.; Ghandour, A.; Ismail, S.; Yousef, H.; El-Hakeim, E.; Makarem, Y.; et al. Peripheral cells from patients with systemic sclerosis disease co-expressing M1 and M2 monocyte/macrophage surface markers: Relation to the degree of skin involvement. Hum. Immunol. 2021, 82, 634–639. [Google Scholar] [PubMed]
- Higashi-Kuwata, N.; Jinnin, M.; Makino, T.; Fukushima, S.; Inoue, Y.; Muchemwa, F.; Yonemura, Y.; Komohara, Y.; Takeya, M.; Mitsuya, H.; et al. Characterization of monocyte/macrophage subsets in the skin and peripheral blood derived from patients with systemic sclerosis. Arthritis Res. Ther. 2010, 12, R128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahoney, J.M.; Taroni, J.; Martyanov, V.; Wood, T.; Greene, C.; Pioli, P.; Hinchcliff, M.; Mhitfield, M. Systems level analysis of systemic sclerosis shows a network of immune and profibrotic pathways connected with genetic polymorphisms. PLoS Comput. Biol. 2015, 11, e1004005. [Google Scholar] [CrossRef] [Green Version]
- Higashi-Kuwata, N.; Makino, T.; Inoue, Y.; Takeya, M.; Ihn, H. Alternatively activated macrophages (M2 macrophages) in the skin of patient with localized scleroderma. Exp. Dermatol. 2009, 18, 727–729. [Google Scholar] [PubMed]
- Meng, X.M.; Nikolic-Paterson, D.J.; Lan, H.Y. TGF-beta: The master regulator of fibrosis. Nat. Rev. Nephrol. 2016, 12, 325–338. [Google Scholar] [PubMed]
- Mathai, S.K.; Gulati, M.; Peng, X.; Russell, T.R.; Shaw, A.C.; Rubinowitz, A.N.; Murray, L.A.; Siner, J.M.; Antin-Ozerkis, D.E.; Montgomery, R.R.; et al. Circulating monocytes from systemic sclerosis patients with interstitial lung disease show an enhanced profibrotic phenotype. Lab. Investig. 2010, 90, 812–823. [Google Scholar] [CrossRef]
- Ototake, Y.; Yamaguchi, Y.; Asami, M.; Komitsu, N.; Akita, A.; Watanabe, T.; Kanaoka, M.; Kurotaki, D.; Tamura, T.; Aihara, M. Downregulated IRF8 in Monocytes and Macrophages of Patients with Systemic Sclerosis May Aggravate the Fibrotic Phenotype. J. Investig. Dermatol. 2021, 141, 1954–1963. [Google Scholar] [CrossRef]
- Trombetta, A.C.; Soldano, S.; Contini, P.; Tomatis, V.; Ruaro, B.; Paolino, S.; Brizzolara, R.; Montagna, P.; Sulli, A.; Pizzorni, C.; et al. A circulating cell population showing both M1 and M2 monocyte/macrophage surface markers characterizes systemic sclerosis patients with lung involvement. Respir. Res. 2018, 19, 186. [Google Scholar] [CrossRef]
- Lescoat, A.; Lecureur, V.; Varga, J. Contribution of monocytes and macrophages to the pathogenesis of systemic sclerosis: Recent insights and therapeutic implications. Curr. Opin. Rheumatol. 2021, 33, 463–470. [Google Scholar] [CrossRef]
- Aota, K.; Yamanoi, T.; Kani, K.; Nakashiro, K.I.; Ishimaru, N.; Azuma, M. Inverse correlation between the number of CXCR3(+) macrophages and the severity of inflammatory lesions in Sjogren’s syndrome salivary glands: A pilot study. J. Oral Pathol. Med. 2018, 47, 710–718. [Google Scholar] [CrossRef]
- Rose, T.; Grützkau, A.; Hirseland, H.; Huscher, D.; Dähnrich, C.; Dzionek, A.; Ozimkowski, T.; Schlumberger, W.; Enghard, P.; Radbruch, A.; et al. IFNα and its response proteins, IP-10 and SIGLEC-1, are biomarkers of disease activity in systemic lupus erythematosus. Ann. Rheum. Dis. 2013, 72, 1639–1645. [Google Scholar] [CrossRef] [PubMed]
- Lisney, A.R.; Szelinski, F.; Reiter, K.; Burmester, G.R.; Rose, T.; Dorner, T. High maternal expression of SIGLEC1 on monocytes as a surrogate marker of a type I interferon signature is a risk factor for the development of autoimmune congenital heart block. Ann. Rheum. Dis. 2017, 76, 1476–1480. [Google Scholar] [CrossRef] [PubMed]
- Clancy, R.M.; Halushka, M.; Rasmussen, S.E.; Lhakhang, T.; Chang, M.; Buyon, J.P. Siglec-1 Macrophages and the Contribution of IFN to the Development of Autoimmune Congenital Heart Block. J. Immunol. 2019, 202, 48–55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kiss, M.; Czimmerer, Z.; Nagy, L. The role of lipid-activated nuclear receptors in shaping macrophage and dendritic cell function: From physiology to pathology. J. Allergy Clin. Immunol. 2013, 132, 264–286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dragoljevic, D.; Kraakman, M.J.; Nagareddy, P.R.; Ngo, D.; Shihata, W.; Kammoun, H.L.; Whillas, A.; Lee, M.K.S.; Al-Sharea, A.; Pernes, G.; et al. Defective cholesterol metabolism in haematopoietic stem cells promotes monocyte-driven atherosclerosis in rheumatoid arthritis. Eur. Heart J. 2018, 39, 2158–2167. [Google Scholar] [CrossRef] [Green Version]
- Murphy, A.J.; Dragoljevic, D.; Tall, A.R. Cholesterol Efflux Pathways Regulate Myelopoiesis: A Potential Link to Altered Macrophage Function in Atherosclerosis. Front. Immunol. 2014, 5, 490. [Google Scholar] [CrossRef] [Green Version]
- Westerterp, M.; Murphy, A.J.; Wang, M.; Pagler, T.A.; Vengrenyuk, Y.; Kappus, M.S.; Gorman, D.J.; Nagareddy, P.R.; Zhu, X.; Abramowicz, S.; et al. Deficiency of ATP-binding cassette transporters A1 and G1 in macrophages increases inflammation and accelerates atherosclerosis in mice. Circ. Res. 2013, 112, 1456–1465. [Google Scholar] [CrossRef]
- Duewell, P.; Kono, H.; Rayner, K.J.; Sirois, C.M.; Vladimer, G.; Bauernfeind, F.G.; Abela, G.S.; Franchi, L.; Nuñez, G.; Schnurr, M.; et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 2010, 464, 1357–1361. [Google Scholar] [CrossRef] [Green Version]
- Lancaster, G.I.; Langley, K.G.; Berglund, N.A.; Kammoun, H.L.; Reibe, S.; Estevez, E.; Weir, J.; Mellett, N.A.; Pernes, G.; Conway, J.R.W.; et al. Evidence that TLR4 is not a receptor for saturated fatty acids but mediates lipid-induced inflammation by reprogramming macrophage metabolism. Cell Metab. 2018, 27, 1096–1110. [Google Scholar] [CrossRef] [Green Version]
- Thomas, D.G.; Doran, A.C.; Fotakis, P.; Westerterp, M.; Antonson, P.; Jiang, H.; Jiang, X.C.; Gustafsson, J.Å.; Tabas, I.; Tall, A.R. LXR suppresses inflammatory gene expression and neutrophil migration through cis-repression and cholesterol efflux. Cell Rep. 2018, 25, 3774–3785. [Google Scholar] [CrossRef] [Green Version]
- Jeon, J.Y.; Nam, J.Y.; Kim, H.A.; Park, Y.B.; Bae, S.C.; Suh, C.H. Liver X receptors alpha gene (NR1H3) promoter polymorphisms are associated with systemic lupus erythematosus in Koreans. Arthritis Res. Ther 2014, 16, R112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noelia, N.; Bensinger, S.J.; Hong, C.; Beceiro, S.; Bradley, M.N.; Zelcer, N.; Deniz, J.; Ramirez, C.; Díaz, M.; Gallardo, G.; et al. Apoptotic cells promote their own clearance and immune tolerance through activation of the nuclear receptor LXR. Immunity 2009, 31, 245–258. [Google Scholar] [CrossRef] [Green Version]
- Zeisbrich, M.; Yanes, R.E.; Zhang, H.; Watanabe, R.; Li, Y.; Brosig, L.; Hong, J.; Wallis, B.B.; Giacomini, J.C.; Assimes, T.L.; et al. Hypermetabolic macrophages in rheumatoid arthritis and coronary artery disease due to glycogen synthase kinase 3b inactivation. Ann. Rheum. Dis. 2018, 77, 1053–1062. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Li, W.; Li, X.; Zhou, H. Inflammation: A Novel Therapeutic Target/Direction in Atherosclerosis. Curr. Pharm. Des. 2017, 23, 1216–1227. [Google Scholar] [CrossRef]
- Ridker, P.M.; MacFadyen, J.G.; Thuren, T.; Everett, B.M.; Libby, P.; Glynn, R.J. CANTOS Trial Group. Effect of interleukin-1β inhibition with canakinumab on incident lung cancer in patients with atherosclerosis: Exploratory results from a randomised, double-blind, placebo-controlled trial. Lancet 2017, 390, 1833–1842. [Google Scholar]
- Ridker, P.M.; Everett, B.M.; Pradhan, A.; MacFadyen, J.G.; Solomon, D.H.; Zaharris, E.; Mam, V.; Hasan, A.; Rosenberg, Y.; Iturriaga, E.; et al. Low-dose methotrexate for the prevention of atherosclerotic events. N. Engl. J. Med. 2019, 380, 752–762. [Google Scholar] [CrossRef]
- Tardif, J.C.; Kouz, S.; Waters, D.D.; Bertrand, O.F.; Diaz, R.; Maggioni, A.P.; Pinto, F.J.; Ibrahim, R.; Gamra, H.; Kiwan, G.S.; et al. Efficacy and safety of low-dose colchicine after myocardial infarction. N. Engl. J. Med. 2019, 381, 2497–2505. [Google Scholar] [CrossRef]
- Ridker, P.M. Anticytokine agents: Targeting interleukin signaling pathways for the treatment of atherothrombosis. Circ. Res. 2019, 124, 437–450. [Google Scholar]
- Dinarello, C.A. An expanding role for interleukin-1 blockade from gout to cancer. Mol. Med. 2014, 20, S43–S58. [Google Scholar] [CrossRef]
- Karasawa, T.; Takahashi, M. Role of NLRP3 Inflammasomes in Atherosclerosis. J. Atheroscler Thromb. 2017, 24, 443–451. [Google Scholar] [CrossRef] [Green Version]
- Fuster, J.J.; MacLauchlan, S.; Zuriaga, M.A.; Polackal, M.N.; Ostriker, A.C.; Chakraborty, R.; Wu, C.-L.; Sano, S.; Muralidharan, S.; RiusEt, C.; et al. Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice. Science 2017, 355, 842–847. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Busque, L.; Patel, J.P.; Figueroa, M.E.; Vasanthakumar, A.; Provost, S.; Hamilou, Z.; Mollica, L.; Li, J.; Viale, A.; Heguy, A.; et al. Recurrent somatic TET2 mutations in normal elderly individuals with clonal hematopoiesis. Nat. Genet. 2012, 44, 1179–1181. [Google Scholar] [CrossRef] [PubMed]
- Reiss, B.; Carsons, S.E.; Anwar, K.; Rao, S.; Edelman, S.D.; Zhang, H.; Fernandez, P.; Cronstein, B.N.; Chan, E.S. Atheroprotective effects of methotrexate on reverse cholesterol transport proteins and foam cell transformation in human THP-1 monocyte/macrophages. Arthritis Rheum. 2008, 58, 3675–3683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olsen, N.J.; Spurlock, C.F., 3rd; Aune, T.M. Methotrexate induces production of IL-1 and IL-6 in the monocytic cell line U937. Arthritis Res. Ther. 2014, 16, R17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallet, M.A.; Wallet, S.M.; Guiulfo, G.; Sleasman, J.W.; Goodenowet, M.M. IFNgamma primes macrophages for inflammatory activation by high molecular weight hyaluronan. Cell Immunol. 2010, 262, 84–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tracey, D.; Klareskog, L.; Sasso, E.H.; Salfeld, J.G.; Tak, P.P. Tumor necrosis factor antagonist mechanisms of action: A comprehensive review. Pharmacol. Ther. 2008, 117, 244–279. [Google Scholar] [CrossRef]
- Degboé, Y.; Rauwel, B.; Baron, M.; Boyer, J.F.; Ruyssen-Witrand, A.; Constantin, A.; Davignon, J.L. Polarization of Rheumatoid Macrophages by TNF Targeting Through an IL-10/STAT3 Mechanism. Front. Immunol. 2019, 10, 3. [Google Scholar] [CrossRef] [Green Version]
- Koelink, P.J.; Bloemendaal, F.M.; Li, B.; Westera, L.; Vogels, E.W.M.; van Roest, M.; Gloudemans, A.K.; van ’t Wout, A.B.; Korf, H.; Vermeire, S.; et al. Anti-TNF therapy in IBD exerts its therapeutic effect through macrophage IL-10 signalling. Gut 2020, 69, 1053–1063. [Google Scholar] [CrossRef] [Green Version]
- Mauri, C.; Gray, D.; Mushtaq, N.; Londei, M. Prevention of arthritis by interleukin 10-producing B cells. J. Exp. Med. 2003, 197, 489–501. [Google Scholar] [CrossRef] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gerasimova, E.V.; Popkova, T.V.; Gerasimova, D.A.; Kirichenko, T.V. Macrophage Dysfunction in Autoimmune Rheumatic Diseases and Atherosclerosis. Int. J. Mol. Sci. 2022, 23, 4513. https://doi.org/10.3390/ijms23094513
Gerasimova EV, Popkova TV, Gerasimova DA, Kirichenko TV. Macrophage Dysfunction in Autoimmune Rheumatic Diseases and Atherosclerosis. International Journal of Molecular Sciences. 2022; 23(9):4513. https://doi.org/10.3390/ijms23094513
Chicago/Turabian StyleGerasimova, Elena V., Tatiana V. Popkova, Daria A. Gerasimova, and Tatiana V. Kirichenko. 2022. "Macrophage Dysfunction in Autoimmune Rheumatic Diseases and Atherosclerosis" International Journal of Molecular Sciences 23, no. 9: 4513. https://doi.org/10.3390/ijms23094513





