Diabetes Mellitus Alters the Immuno-Expression of Neuronal Nitric Oxide Synthase in the Rat Pancreas
Abstract
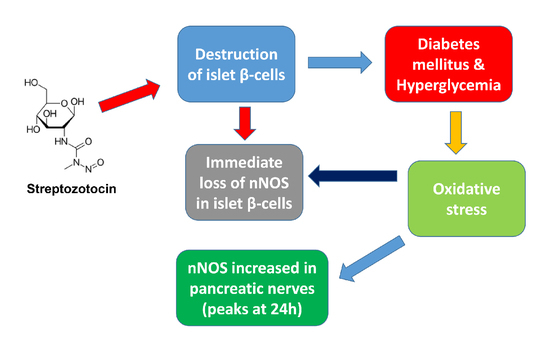
:1. Introduction
2. Results
2.1. Weight and Blood Glucose Levels of Normal and Diabetic Rats
2.2. Immunohistochemistry
2.3. Western Blot
2.4. Immunofluorescence Study of Insulin and nNOS
2.4.1. Hours after the Onset of Diabetes Mellitus
2.4.2. Weeks after the Induction of Diabetes
2.4.3. Months after the Onset of Diabetes
2.5. Immunofluorescence Study of Glucagon and nNOS
2.5.1. Hours after the Onset of Diabetes Mellitus
2.5.2. Weeks after the Induction of Diabetes Mellitus
2.5.3. Months after the Onset of Diabetes Mellitus
2.6. Measurement of Thiobarbituric Acid Reactive Substances (TBARS), Rate of TBARS, Reactive Oxygen Species (ROS) and Glutathione (GSH)
2.7. Reactive Oxygen Species (ROS)
3. Discussion
3.1. nNOS in Pancreatic Beta Cell
3.2. nNOS in Pancreatic Nerve Profiles
3.3. Markers of Oxidative Stress
4. Materials and Methods
4.1. Experimental Animals and Induction of Diabetes Mellitus
4.2. Immunohistochemistry
4.3. Immunofluorescence
4.4. Measurement of Thiobarbituric Acid Reactive Substances (TBARS), Rate of TBARS, Reactive Oxygen Species (ROS), Glutathione (GSH)
4.5. Lipid Peroxidation
4.6. Reactive Oxygen Species (ROS)
4.7. Glutathione (GSH)
4.8. Western Blot Analysis of Neural Nitric Oxide Synthase (nNOS)
4.9. Densitometric Analysis of Intensity of nNOS Immunofluorescence
5. Statistical Analysis
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Diabetes Atlas, 10th ed.; International Diabetes Federation: Brussels, Belgium, 2021.
- Adeghate, E.; Schattner, P.; Dunn, E. An update on the etiology and epidemiology of diabetes mellitus. Ann. N. Y. Acad. Sci. 2006, 1084, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Calejman, C.M.; Doxsey, W.G.; Fazakerley, D.; Guertin, D.A. Integrating adipocyte insulin signaling and metabolism in the multi-omics era. Trends Biochem. Sci. 2022. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, J.S.; Sehrawat, A.; Mishra, J.; Sidhu, I.S.; Navik, U.; Khullar, N.; Kumar, S.; Bhatti, G.K.; Reddy, P.H. Oxidative stress in the pathophysiology of type 2 diabetes and related complications: Current therapeutics strategies and future perspectives. Free Radic Biol. Med. 2022, 184, 114–134. [Google Scholar] [CrossRef] [PubMed]
- Adeghate, E.A.; Kalász, H.; Al Jaberi, S.; Adeghate, J.; Tekes, K. Tackling type 2 diabees-associated cardiovascular and renal comorbidities: A key challenge for drug development. Expert Opin. Investig. Drugs 2021, 30, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Rodeberg, D.A.; Chaet, M.S.; Bass, R.C.; Arkovitz, M.S.; Garcia, V.F. Nitric oxide: An overview. Am. J. Surg. 1995, 170, 292–303. [Google Scholar] [CrossRef]
- Whittle, B.J. Nitric oxide—A mediator of inflammation or mucosal defence. Eur. J. Gastroenterol. Hepatol. 1997, 9, 1026–1032. [Google Scholar] [CrossRef]
- Brown, M.D.; Schoenfisch, M.H. Electrochemical Nitric Oxide Sensors: Principles of Design and Characterization. Chem. Rev. 2019, 119, 11551–11575. [Google Scholar] [CrossRef]
- Porrini, C.; Ramarao, N.; Tran, S.L. Dr. NO and Mr. Toxic—The versatile role of nitric oxide. Biol. Chem. 2020, 401, 547–572. [Google Scholar] [CrossRef]
- Caballano-Infantes, E.; Cahuana, G.M.; Bedoya, F.J.; Salguero-Aranda, C.; Tejedo, J.R. The Role of Nitric Oxide in Stem Cell Biology. Antioxidants 2022, 11, 497. [Google Scholar] [CrossRef]
- Roszer, T. The Biology of Subcellular Nitric Oxide; Springer: Dordrecht, The Netherlands, 2012; ISBN 978-94-007-2818-9. [Google Scholar]
- Culotta, E.; Koshland, D.E., Jr. NO news is good news. Science 1992, 258, 1862–1865. [Google Scholar] [CrossRef]
- Poon, C.H.; Tsui, K.C.; Chau, S.C.; Chong, P.S.; Lui, S.W.Y.; Aquili, L.; Wong, K.H.; Lim, L.W. Functional Roles of Neuronal Nitric; Oxide Synthase in Neurodegenerative Diseases and Mood Disorders. Curr. Alzheimer Res. 2021, 18, 831–840. [Google Scholar] [CrossRef] [PubMed]
- Parillo, F.; Catone, G.; Boiti, C.; Zerani, M. Immunopresence and enzymatic activity of nitric oxide synthases, cyclooxygenases and PGE2-9-ketoreductase and in vitro production of PGF2α, PGE2 and testosterone in the testis of adult and prepubertal alpaca (Lama pacos). Gen. Comp. Endocrinol. 2011, 171, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Saluja, R.; Saini, R.; Mitra, K.; Bajpai, V.K.; Dikshit, M. Ultrastructural immunogold localization of nitric oxide synthase isoforms in rat and human eosinophils. Cell Tissue Res. 2010, 340, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Lajoix, A.D.; Reggio, H.; Chardès, T.; Péraldi-Roux, S.; Tribillac, F.; Roye, M.; Dietz, S.; Broca, C.; Manteghetti, M.; Ribes, G.; et al. A neuronal isoform of nitric oxide synthase expressed in pancreatic beta cells controls insulin secretion. Diabetes 2001, 50, 1311–1323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ember, Z.; Réti, A.; Fehér, E. Enzyme and immunohistochemical localization of nitric oxide synthase in nerves of the porcine pancreas. Neurosci. Lett. 2000, 292, 163–166. [Google Scholar] [CrossRef]
- Arciszewski, M.B. Expression of neuronal nitric oxide synthase in the pancreas of the sheep. Anat. Histol. Embryol. 2007, 36, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Dorff, G.; Meyer, G.; Krone, D.; Pozzilli, P.; Zülke, H. Neuronal NO synthase and its inhibitor PIN are present and influenced by glucose in the human beta cell line CM and in rats INS-1 cells. Biol. Chem. 2002, 383, 1357–1361. [Google Scholar] [CrossRef]
- Ort, T.; Maksimova, E.; Dirkx, R.; Kachinsky, A.M.; Berghs, S.; Froehner, S.C.; Solimena, M. The receptor tyrosine phosphatase-like protein ICA512 binds the PDZ domains of beta2-syntrophin and nNOS in pancreatic beta-cells. Eur. J. Cell Biol. 2000, 79, 621–630. [Google Scholar] [CrossRef]
- Yaribeygi, H.; Sathyapalan, T.; Atkin, S.L.; Sahebkar, A. Molecular Mechanisms Linking Oxidative Stress and Diabetes Mellitus. Oxidative Med. Cell. Longev. 2020, 2020, 8609213. [Google Scholar] [CrossRef] [Green Version]
- Raza, H.; Prabu, S.K.; Robin, M.A.; Avadhani, N.G. Elevated mitochondrial cytochrome P450 2E1 and glutathione S-transferase A4-4 in streptozotocin-induced diabetic rats: Tissue-specific variations and roles in oxidative stress. Diabetes 2004, 53, 185–194. [Google Scholar] [CrossRef] [Green Version]
- Lotfy, M.; Adeghate, J.; Kalasz, H.; Singh, J.; Adeghate, E. Chronic Complications of Diabetes Mellitus: A Mini Review. Curr. Diabetes Rev. 2017, 13, 3–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsunami, T.; Sato, Y.; Sato, T.; Ariga, S.; Shimomura, T.; Yukawa, M. Oxidative stress and gene expression of antioxidant enzymes in the streptozotocin-induced diabetic rats under hyperbaric oxygen exposure. Int. J. Clin. Exp. Pathol. 2009, 3, 177–188. [Google Scholar] [PubMed]
- Fiorentino, T.V.; Prioletta, A.; Zuo, P.; Folli, F. Hyperglycemia-induced oxidative stress and its role in diabetes mellitus related cardiovascular diseases. Curr. Pharm. Des. 2013, 19, 5695–5703. [Google Scholar] [CrossRef] [PubMed]
- VanderJagt, T.J.; Ghattas, R.; VanderJagt, D.J.; Crossey, M.; Glew, R.H. Comparison of the total antioxidant content of 30 widely used medicinal plants of New Mexico. Life Sci. 2002, 70, 1035–1040. [Google Scholar] [CrossRef]
- Seghrouchni, I.; Drai, J.; Bannier, E.; Riviere, J.; Calmard, P.; Garcia, I.; Orgiazzi, J.; Revol, A. Oxidative stress parameters in type I, type II and insulin-treated type 2 diabetes mellitus; insulin treatment efficiency. Clin. Chim. Acta 2002, 321, 89–96. [Google Scholar] [CrossRef]
- Adeghate, E.; D’Souza, C.M.; Saeed, Z.; Al Jaberi, S.; Tariq, S.; Kalász, H.; Tekes, K.; Adeghate, E.A. Nociceptin Increases Antioxidant Expression in the Kidney, Liver and Brain of Diabetic Rats. Biology 2021, 10, 621. [Google Scholar] [CrossRef] [PubMed]
- Pi, J.; Zhang, Q.; Fu, J.; Woods, C.G.; Hou, Y.; Corkey, B.E.; Collins, S.; Andersen, M.E. ROS signaling, oxidative stress and Nrf2 in pancreatic beta-cell function. Toxicol. Appl. Pharmacol. 2010, 244, 77–83. [Google Scholar] [CrossRef] [Green Version]
- Redza-Dutordoir, M.; Averill-Bates, D.A. Activation of apoptosis signalling pathways by reactive oxygen species. Biochim. Biophys. Acta 2016, 63, 2977–2992. [Google Scholar] [CrossRef]
- Novo, E.; Parola, M. Redox mechanisms in hepatic chronic wound healing and fibrogenesis. Fibrogenesis Tissue Repair 2008, 1, 5. [Google Scholar] [CrossRef] [Green Version]
- Coelho, A.M.; Queiroz, I.F.; Lima, W.G.; Talvani, A.; Perucci, L.O.; Oliveira de Souza, M.; Costa, D.C. Temporal analysis of paracetamol-induced hepatotoxicity. Drug Chem. Toxicol. 2022, 1–10. [Google Scholar] [CrossRef]
- Elabadlah, H.; Hameed, R.; D’Souza, C.; Mohsin, S.; Adeghate, E.A. Exogenous Ghrelin Increases Plasma Insulin Level in Diabetic Rats. Biomolecules 2020, 10, 633. [Google Scholar] [CrossRef] [PubMed]
- Adeghate, E.; Ponery, A.S. Ghrelin stimulates insulin secretion from the pancreas of normal and diabetic rats. J. Neuroendocrinol. 2002, 14, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Adeghate, E.; Ponery, A.S. GABA in the endocrine pancreas: Cellular localization and function in normal and diabetic rats. Tissue Cell 2002, 34, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Al-Salam, S.; Rashed, H.; Adeghate, E. Diabetes mellitus is associated with an increased expression of resistin in human pancreatic islet cells. Islets 2011, 3, 246–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adeghate, E.; Fernandez-Cabezudo, M.; Hameed, R.; El-Hasasna, H.; El Wasila, M.; Abbas, T.; Al-Ramadi, B. Orexin-1 receptor co-localizes with pancreatic hormones in islet cells and modulates the outcome of streptozotocin-induced diabetes mellitus. PLoS ONE 2010, 5, e8587. [Google Scholar] [CrossRef] [Green Version]
- Adeghate, E.; Ponery, A.S. Large reduction in the number of galanin-immunoreactive cells in pancreatic islets of diabetic rats. J. Neuroendocrinol. 2001, 13, 706–710. [Google Scholar] [CrossRef]
- Tariq, S.; Rashed, H.; Nurulain, S.M.; Emerald, B.S.; Koturan, S.; Tekes, K.; Adeghate, E. Distribution of nociceptin in pancreatic islet cells of normal and diabetic rats. Pancreas 2015, 44, 602–607. [Google Scholar] [CrossRef]
- Adeghate, E.; Saeed, Z.; D’Souza, C.; Tariq, S.; Kalász, H.; Tekes, K.; Adeghate, E.A. Effect of nociceptin on insulin release in normal and diabetic rat pancreas. Cell Tissue Res. 2018, 374, 517–529. [Google Scholar] [CrossRef]
- Adeghate, E.; Ponery, A. Diabetes mellitus influences the degree of colocalization of calcitonin gene-related peptide with insulin and somatostatin in the rat pancreas. Pancreas 2004, 29, 311–319. [Google Scholar] [CrossRef]
- Nekrep, N.; Wang, J.; Miyatsuka, T.; German, M.S. Signals from the neural crest regulate beta-cell mass in the pancreas. Development 2008, 135, 2151–2160. [Google Scholar] [CrossRef] [Green Version]
- Arntfield, M.E.; van der Kooy, D. β-Cell evolution: How the pancreas borrowed from the brain: The shared toolbox of genes expressed by neural and pancreatic endocrine cells may reflect their evolutionary relationship. Bioessays 2011, 33, 582–587. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Liu, Y.; Zheng, Q.; Hu, Q. Increased proportion of nitric oxide synthase immunoreactive neurons in rat ileal myenteric ganglia after severe acute pancreatitis. BMC Gastroenterol. 2011, 11, 127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adeghate, E.; Al-Ramadi, B.; Saleh, A.M.; Vijayarasathy, C.; Ponery, A.S.; Arafat, K.; Howarth, F.C.; El-Sharkawy, T. Increase in neuronal nitric oxide synthase content of the gastroduodenal tract of diabetic rats. Cell. Mol. Life Sci. CMLS 2003, 60, 1172–1179. [Google Scholar] [CrossRef] [PubMed]
- Idrizaj, E.; Traini, C.; Vannucchi, M.G.; Baccari, M.C. Nitric Oxide: From Gastric Motility to Gastric Dysmotility. Int. J. Mol. Sci. 2021, 22, 9990. [Google Scholar] [CrossRef] [PubMed]
- Lanas, A. Role of nitric oxide in the gastrointestinal tract. Arthritis Res. Ther. 2008, 10 (Suppl. 2), S4. [Google Scholar] [CrossRef] [Green Version]
- El-Huneidi, W.; Anjum, S.; Saleh, M.A.; Bustanji, Y.; Abu-Gharbieh, E.; Taneera, J. Carnosic Acid Protects INS-1 β-Cells against Streptozotocin-Induced Damage by Inhibiting Apoptosis and Improving Insulin Secretion and Glucose Uptake. Molecules 2022, 27, 2102. [Google Scholar] [CrossRef]
- Spinas, G.A. The Dual Role of Nitric Oxide in Islet beta-Cells. News Physiol Sci. 1999, 14, 49–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajapakse, N.W.; Mattson, D.L. Role of L-arginine in nitric oxide production in health and hypertension. Clin. Exp. Pharmacol. Physiol. 2009, 36, 249–255. [Google Scholar] [CrossRef]
- Adeghate, E.; Ponery, A.S.; El-Sharkawy, T.; Parvez, H. L-arginine stimulates insulin secretion from the pancreas of normal and diabetic rats. Amino Acids 2001, 21, 205–209. [Google Scholar] [CrossRef]
- Adeghate, E.; Parvez, S.H. Nitric oxide and neuronal and pancreatic beta cell death. Toxicology 2000, 153, 143–156. [Google Scholar] [CrossRef]
- De Giorgio, R.; Brecha, N.C.; Brunicardi, F.C.; Becker, J.M.; Go, V.L.W.; Sternini, C. Nitric oxide producing neurons in the monkey and human digestive system. J. Comp. Neurol. 1994, 342, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Adeghate, E.; Hameed, R.S.; Ponery, A.S.; Tariq, S.; Sheen, R.S.; Shaffiullah, M.; Donáth, T. Streptozotocin causes pancreatic beta cell failure via early and sustained biochemical and cellular alterations. Exp. Clin. Endocrinol. Diabetes 2010, 118, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Wrzos, H.F.; Cruz, A.; Polavarapu, R.; Shearer, D.; Ouyang, A. Nitric oxide synthase (NOS) expression in the myenteric plexus of streptozotocin-diabetic rats. Dig. Dis. Sci. 1997, 42, 2106–2110. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Nakamura, K.; Itoh, H.; Sima, A.A.; Owyang, C. Impaired expression of nitric oxide synthase in the gastric myenteric plexus of spontaneously diabetic rats. Gastroenterology 1997, 113, 1535–1544. [Google Scholar] [CrossRef] [PubMed]
- Giglio, R.V.; Stoian, A.P.; Haluzik, M.; Pafili, K.; Patti, A.M.; Rizvi, A.A.; Ciaccio, M.; Papanas, N.; Rizzo, M. molecular markers of cardiovascular disease risk in type 2 diabetes mellitus. Biochim. Biophys. Acta Mol. Basis Dis. 2021, 1867, 166148. [Google Scholar] [CrossRef]
- Alhakamy, N.A.; Caruso, G.; Eid, B.G.; Fahmy, U.A.; Ahmed, O.A.A.; Abdel-Naim, A.B.; Alamoudi, A.J.; Alghamdi, S.A.; Al Sadoun, H.; Eldakhakhny, B.M.; et al. Ceftriaxone and Melittin Synergistically Promote Wound Healing in Diabetic Rats. Pharmaceutics 2021, 13, 1622. [Google Scholar] [CrossRef]
- Lotfy, M.; Singh, J.; Rashed, H.; Tariq, S.; Zilahi, E.; Adeghate, E. Mechanism of the beneficial and protective effects of exenatide in diabetic rats. J. Endocrinol. 2014, 220, 291–304. [Google Scholar] [CrossRef] [Green Version]
- Adeghate, E. Effect of subcutaneous pancreatic tissue transplants on streptozotocin-induced diabetes in rats. II. Endocrine and Metabolic functions. Tissue Cell 1999, 31, 73–83. [Google Scholar]
- Howarth, F.C.; Jacobson, M.; Shafiullah, M.; Adeghate, E. Long-term effects of type 2 diabetes mellitus on heart rhythm in the Goto-Kakizaki rat. Exp. Physiol. 2008, 93, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Adeghate, E.; Donath, T. Morphological findings in long-term pancreatic tissue transplants in the anterior eye chamber of rats. Pancreas 1990, 5, 208–305. [Google Scholar] [CrossRef]
- Adeghate, E. Distribution of calcitonin-gene-related peptide, neuropeptide-Y, vasoactive intestinal polypeptide, cholecystokinin-8, substance P and islet peptides in the pancreas of normal and diabetic rats. Neuropeptides 1999, 33, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Adeghate, J.O.; D’Souza, C.; Kántor, O.; Tariq, S.; Souid, A.K.; Adeghate, E. Early (5-Day) Onset of Diabetes Mellitus Causes Degeneration of Photoreceptor Cells, Overexpression of Incretins, and Increased Cellular Bioenergetics in Rat Retina. Cells 2021, 10, 1981. [Google Scholar] [CrossRef] [PubMed]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Buttar, H.S.; Chow, A.Y.; Downie, R.H. Glutathione alterations in rat liver after acute and subacute oral administration of paracetamol. Clin. Exp. Pharmacol. Physiol. 1977, 4, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Rubino, F.M. The Redox Potential of the β-93-Cysteine Thiol Group in Human Hemoglobin Estimated from In Vitro Oxidant Challenge Experiments. Molecules 2021, 26, 2528. [Google Scholar] [CrossRef]
- George, J.; Bashir, G.; Qureshi, M.M.; Mohamed, Y.A.; Azzi, J.; Al-Ramadi, B.K.; Fernandez-Cabezudo, M.J. Cholinergic stimulation prevents the development of autoimmune diabetes: Evidence for the modulation of Th17 effector cells via an IFNγ-dependent mechanism. Front. Immunol. 2016, 7, 419. [Google Scholar] [CrossRef] [Green Version]
- Aryappalli, P.; Al-Qubaisi, S.S.; Attoub, S.; George, J.A.; Arafat, K.; Ramadi, K.B.; Mohamed, Y.A.; Al-Dhaheri, M.M.; Al-Sbiei, A.; Fernandez-Cabezudo, M.J.; et al. The IL-6/STAT3 signaling pathway is an early target of Manuka honey-induced suppression of human breast cancer cells. Front. Oncol. 2017, 7, 167. [Google Scholar] [CrossRef] [Green Version]
- Aryappalli, P.; Shabbiri, K.; Masad, R.J.; Al-Marri, R.H.; Haneefa, S.M.; Mohamed, Y.A.; Arafat, K.; Attoub, S.; Cabral-Marques, O.; Ramadi, K.B.; et al. Inhibition of tyrosine-phosphorylated STAT3 in human breast and lung cancer cells by Manuka honey is mediated by selective antagonism of the IL-6 receptor. Int. J. Mol. Sci. 2019, 20, 4340. [Google Scholar] [CrossRef] [Green Version]














| Normal | Diabetic | Difference | |
|---|---|---|---|
| Weight (g) | 303.33 ± 14.2 | 244.67 ± 33.6 | p < 0.004 |
| Blood glucose (mg/dL) | 55.9 ± 5.5 | 528.8 ± 55.14 | p < 0.0005 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Emerald, B.S.; Mohsin, S.; D’Souza, C.; John, A.; El-Hasasna, H.; Ojha, S.; Raza, H.; al-Ramadi, B.; Adeghate, E. Diabetes Mellitus Alters the Immuno-Expression of Neuronal Nitric Oxide Synthase in the Rat Pancreas. Int. J. Mol. Sci. 2022, 23, 4974. https://doi.org/10.3390/ijms23094974
Emerald BS, Mohsin S, D’Souza C, John A, El-Hasasna H, Ojha S, Raza H, al-Ramadi B, Adeghate E. Diabetes Mellitus Alters the Immuno-Expression of Neuronal Nitric Oxide Synthase in the Rat Pancreas. International Journal of Molecular Sciences. 2022; 23(9):4974. https://doi.org/10.3390/ijms23094974
Chicago/Turabian StyleEmerald, Bright Starling, Sahar Mohsin, Crystal D’Souza, Annie John, Hussain El-Hasasna, Shreesh Ojha, Haider Raza, Basel al-Ramadi, and Ernest Adeghate. 2022. "Diabetes Mellitus Alters the Immuno-Expression of Neuronal Nitric Oxide Synthase in the Rat Pancreas" International Journal of Molecular Sciences 23, no. 9: 4974. https://doi.org/10.3390/ijms23094974