Variations in the Gene Expression Profile in Atherosclerotic Patients with Non-Fatal ACS: A Preliminary Study
Abstract
:1. Introduction
2. Results
2.1. Formation of Platelet–Leukocyte Aggregates (PLAs) and Expression of P-Selectin Measured by Flow Cytometry
2.2. The Concentration of PF-4 and Soluble (s)P-Selectin in Human Plasma
2.3. Pro-Inflammatory Cytokines Measurement in Human Plasma
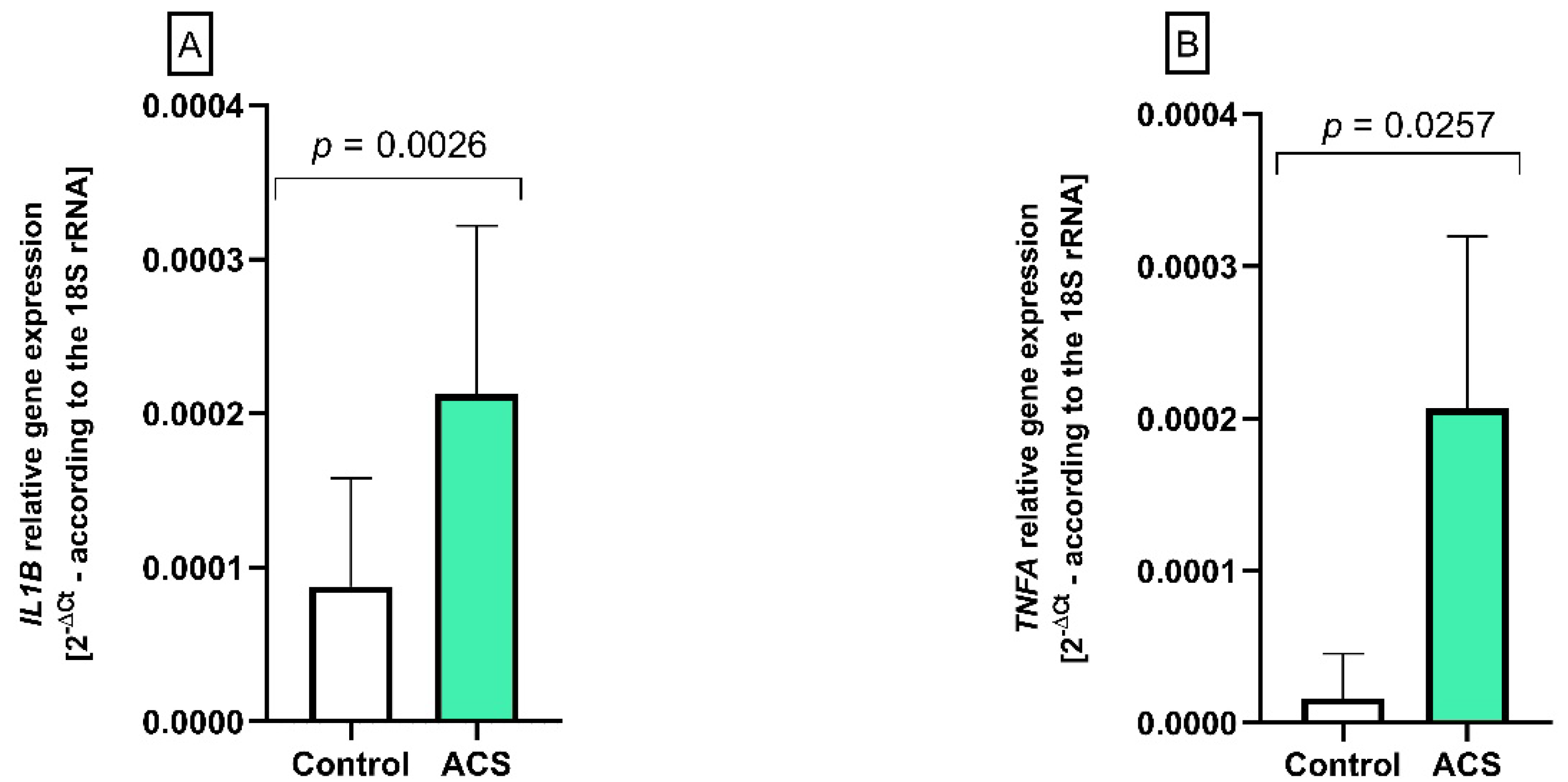
2.4. The Analysis of IL1B and TNFA Gene Expression by Real-Time PCR
2.5. The Expression of 92 Immune Response Associated Genes of Selected Pro-Inflammatory Factors
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Analysis Using Flow Cytometry
4.3. Cytokine Level Analysis
4.4. PF-4 Level Analysis
4.5. Soluble P-Selectin Measurement
4.6. Isolation of Total RNA and Reverse Transcription
4.7. Analysis of Gene Expression
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global burden of cardiovascular diseases and risk factors, 1990–2019: Update from the GBD 2019 study. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef] [PubMed]
- Gotlieb, A.I. Atherosclerosis and acute coronary syndromes. Cardiovasc. Pathol. 2005, 14, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Avis, S.R.; Vernon, S.T.; Hagström, E.; Figtree, G.A. Coronary artery disease in the absence of traditional risk factors: A call for action. Eur. Heart J. 2021, 42, 3822–3824. [Google Scholar] [CrossRef] [PubMed]
- Body, R.; McDowell, G.; Carley, S.; Mackway-Jones, K. Do risk factors for chronic coronary heart disease help diagnose acute myocardial infarction in the emergency department? Resuscitation 2008, 79, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Aukrust, P.; Berge, R.K.; Ueland, T.; Aaser, E.; Damås, J.K.; Wikeby, L.; Brunsvig, A.; Müller, F.; Forfang, K.; Frøland, S.S.; et al. Interaction between chemokines and oxidative stress: Possible pathogenic role in acute coronary syndromes. J. Am. Coll. Cardiol. 2001, 37, 485–491. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Tang, C. Targeting platelet in atherosclerosis plaque formation: Current knowledge and future perspectives. Int. J. Mol. Sci. 2020, 21, 9760. [Google Scholar] [CrossRef]
- Adamski, P.; Buszko, K.; Sikora, J.; Niezgoda, P.; Fabiszak, T.; Ostrowska, M.; Barańska, M.; Karczmarska-Wódzka, A.; Navarese, E.P.; Kubica, J. Determinants of high platelet reactivity in patients with acute coronary syndromes treated with ticagrelor. Sci. Rep. 2019, 9, 3924. [Google Scholar] [CrossRef]
- McNicol, A.; Israels, S.J. Beyond hemostasis: The role of platelets in inflammation, malignancy and infection. Cardiovasc. Hematol. Disord. Drug Targets 2008, 8, 99–117. [Google Scholar] [CrossRef] [Green Version]
- Whiss, P.A.; Andersson, R.G.; Srinivas, U. Kinetics of platelet P-selectin mobilization: Concurrent surface expression and release induced by thrombin or PMA, and inhibition by the NO donor SNAP. Cell Adhes. Commun. 1998, 6, 289–300. [Google Scholar] [CrossRef] [Green Version]
- Bigalke, B.; Stellos, K.; Geisler, T.; Kremmer, E.; Seizer, P.; May, A.E.; Lindemann, S.; Melms, A.; Luft, A.; Gawaz, M. Expression of platelet glycoprotein VI is associated with transient ischemic attack and stroke. Eur. J. Neurol. 2010, 17, 111–117. [Google Scholar] [CrossRef] [Green Version]
- Liverani, E.; Kilpatrick, L.E.; Tsygankov, A.Y.; Kunapuli, S.P. The role of P2Y₁₂ receptor and activated platelets during inflammation. Curr. Drug Targets 2014, 15, 720–728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Z.; Mondal, N.K.; Ding, J.; Koenig, S.C.; Slaughter, M.S.; Griffith, B.P.; Wu, Z.J. Activation and shedding of platelet glycoprotein IIb/IIIa under non-physiological shear stress. Mol. Cell Biochem. 2015, 409, 93–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feghhi, S.; Munday, A.D.; Tooley, W.W.; Rajsekar, S.; Fura, A.M.; Kulman, J.D.; López, J.A.; Sniadecki, N.J. Glycoprotein Ib-IX-V complex transmits cytoskeletal forces that enhance platelet adhesion. Biophys. J. 2016, 111, 601–608. [Google Scholar] [CrossRef] [Green Version]
- Zaslavsky, A.; Baek, K.H.; Lynch, R.C.; Short, S.; Grillo, J.; Folkman, J.; Italiano, J.E.J.; Ryeom, S. Platelet-derived thrombospondin-1 is a critical negative regulator and potential biomarker of angiogenesis. Blood 2010, 115, 4605–4613. [Google Scholar] [CrossRef] [Green Version]
- Bryckaert, M.; Rosa, J.P.; Denis, C.V.; Lenting, P.J. Of von willebrand factor and platelets. Cell Mol. Life Sci. 2015, 72, 307–326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hou, Y.; Carrim, N.; Wang, Y.; Gallant, R.C.; Marshall, A.; Ni, H. Platelets in hemostasis and thrombosis: Novel mechanisms of fibrinogen-independent platelet aggregation and fibronectin-mediated protein wave of hemostasis. J. Biomed. Res. 2015, 29, 437–444. [Google Scholar] [CrossRef] [Green Version]
- Beikmann, B.S.; Tomlinson, I.D.; Rosenthal, S.J.; Andrews, A.M. Serotonin uptake is largely mediated by platelets versus lymphocytes in peripheral blood cells. ACS Chem. Neurosci. 2013, 4, 161–170. [Google Scholar] [CrossRef] [Green Version]
- Chen, R.; Jin, G.; Li, W.; McIntyre, T.M. Epidermal growth factor (EGF) autocrine activation of human platelets promotes EGF receptor-dependent oral squamous cell carcinoma invasion, migration, and epithelial mesenchymal transition. J. Immunol. 2018, 201, 2154–2164. [Google Scholar] [CrossRef]
- Meyer, A.; Wang, W.; Qu, J.; Croft, L.; Degen, J.L.; Coller, B.S.; Ahamed, J. Platelet TGF-β1 contributions to plasma TGF-β1, cardiac fibrosis, and systolic dysfunction in a mouse model of pressure overload. Blood 2012, 119, 1064–1074. [Google Scholar] [CrossRef] [Green Version]
- Ahamed, J.; Laurence, J. Role of platelet-derived transforming growth factor-β1 and reactive oxygen species in radiation-induced organ fibrosis. Antioxid. Redox Signal. 2017, 27, 977–988. [Google Scholar] [CrossRef]
- Gremmel, T.; Michelson, A.; Frelinger, A.L. III. Platelet physiology. Semin. Thromb. Hemost. 2016, 42, 191–204. [Google Scholar] [CrossRef] [Green Version]
- Badimon, L.; Padró, T.; Vilahur, G. Atherosclerosis, platelets and thrombosis in acute ischaemic heart disease. Eur. Heart J. Acute Cardiovasc. Care 2012, 1, 60–74. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Ridker, P.M.; Hansson, G.K.; Leducq. Transatlantic network on atherothrombosis, 2009. Inflammation in atherosclerosis: From pathophysiology to practice. J. Am. Coll. Cardiol. 2009, 54, 2129–2138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abbasi, S.H.; Boroumand, M.A. Expanded network of inflammatory markers of atherogenesis: Where are we now? Open Cardiovasc. Med. J. 2010, 4, 38–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ammirati, E.; Moroni, F.; Norata, G.D.; Magnoni, M.; Camici, P.G. Markers of inflammation associated with plaque progression and instability in patients with carotid atherosclerosis. Mediat. Inflamm. 2015, 2015, 718329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zernecke, A.; Weber, C. Chemokines in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef]
- Mourouzis, K.; Oikonomou, E.; Siasos, G.; Tsalamadris, S.; Vogiatzi, G.; Antonopoulos, A.; Fountoulakis, P.; Goliopoulou, A.; Papaioannou, S.; Tousoulis, D. Pro-inflammatory cytokines in acute coronary syndromes. Curr. Pharm. Des. 2020, 26, 4624–4647. [Google Scholar] [CrossRef]
- Stakos, D.A.; Gatsiou, A.; Stamatelopoulos, K.; Tselepis, A.D.; Stellos, K. Platelet microRNAs: From platelet biology to possible disease biomarkers and therapeutic targets. Platelets 2013, 24, 579–589. [Google Scholar] [CrossRef]
- Muñoz-Carrillo, J.L.; Contreras-Cordero, J.; Gutierrez, O.; Villalobos-Gutiérrez, P.; Ramos-Gracia, L.; Hernández-Reyes, V. Cytokine profiling plays a crucial role in activating immune system to clear infectious pathogens. In Immune Response Activation and Immunomodulation, 1st ed.; Tyagi, R., Singh Bisen, P., Eds.; IntechOpen: London, UK, 2018; pp. 1–30. [Google Scholar]
- Lins Ferreira, V.; Borba, H.; Bonetti, A.; Leonart, L.; Pontarolo, R. Cytokines and interferons: Types and functions. In Autoantibodies and Cytokines; IntechOpen: London, UK, 2018. [Google Scholar]
- Brambilla, M.; Camera, M.; Colnago, D.; Marenzi, G.; De Metrio, M.; Giesen, P.L.; Balduini, A.; Veglia, F.; Gertow, K.; Biglioli, P.; et al. Tissue factor in patients with acute coronary syndromes: Expression in platelets, leukocytes, and platelet-leukocyte aggregates. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 947–953. [Google Scholar] [CrossRef] [Green Version]
- Schrottmaier, W.C.; Mussbacher, M.; Salzmann, M.; Assinger, A. Platelet-leukocyte interplay during vascular disease. Atherosclerosis 2020, 307, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Rauova, L.; Zhai, L.; Kowalska, M.A.; Arepally, G.M.; Cines, D.B.; Poncz, M. Role of platelet surface PF4 antigenic complexes in heparin-induced thrombocytopenia pathogenesis: Diagnostic and therapeutic implications. Blood 2006, 107, 2346–2353. [Google Scholar] [CrossRef] [PubMed]
- Sachais, B.S.; Turrentine, T.; Dawicki McKenna, J.M.; Rux, A.H.; Rader, D.; Kowalska, M.A. Elimination of platelet factor 4 (PF4) from platelets reduces atherosclerosis in C57Bl/6 and apoE-/- mice. Thromb. Haemost. 2007, 98, 1108–1113. [Google Scholar] [PubMed]
- von Hundelshausen, P.; Weber, C. Platelets as immune cells: Bridging inflammation and cardiovascular disease. Circ. Res. 2007, 100, 27–40. [Google Scholar] [CrossRef]
- Kamath, S.; Blann, A.D.; Caine, G.J.; Gurney, D.; Chin, B.S.P.; Lip, G.Y.H. Platelet p-selectin levels in relation to plasma soluble p-selectin and β-thromboglobulin levels in atrial fibrillation. Stroke 2002, 33, 1237–1242. [Google Scholar] [CrossRef] [Green Version]
- Woollard, K.J.; Suhartoyo, A.; Harris, E.E.; Eisenhardt, S.U.; Jackson, S.P.; Peter, K.; Dart, A.M.; Hickey, M.J.; Chin-Dusting, J.P.F. Pathophysiological levels of soluble p-selectin mediate adhesion of leukocytes to the endothelium through mac-1 activation. Circ. Res. 2008, 103, 1128–1138. [Google Scholar] [CrossRef] [Green Version]
- Ridker, P.M.; Buring, J.E.; Rifai, N. Soluble P-selectin and the risk of future cardiovascular events. Circulation 2001, 103, 491–495. [Google Scholar] [CrossRef]
- Finsterbusch, M.; Schrottmaier, W.C.; Kral-Pointner, J.B.; Salzmann, M.; Assinger, A. Measuring and interpreting platelet-leukocyte aggregates. Platelets 2018, 29, 677–685. [Google Scholar] [CrossRef]
- Zhu, L.; Yin, Y.; Zhou, R.; Lin, J.; Li, J.; Ye, J. Changes of monocyte subsets in patients with acute coronary syndrome and correlation with myocardial injury markers. Int. J. Clin. Exp. Pathol. 2015, 8, 7266–7271. [Google Scholar]
- Furman, M.I.; Barnard, M.R.; Krueger, L.A.; Fox, M.L.; Shilale, E.A.; Lessard, D.M.; Marchese, P.; Frelinger, A.L.; Goldberg, R.J.; Michelson, A.D. Circulating monocyte-platelet aggregates are an early marker of acute myocardial infarction. J. Am. Coll. Cardiol. 2001, 38, 1002–1006. [Google Scholar] [CrossRef] [Green Version]
- Gasson, J.C. Molecular physiology of granulocyte-macrophage colony-stimulating factor. Blood 1991, 77, 1131–1145. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Théroux, P. Clopidogrel inhibits platelet-leukocyte interactions and thrombin receptor agonist peptide-induced platelet activation in patients with an acute coronary syndrome. J. Am. Coll. Cardiol. 2004, 43, 1982–1988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dann, R.; Hadi, T.; Montenont, E.; Boytard, L.; Alebrahim, D.; Feinstein, J.; Allen, N.; Simon, R.; Barone, K.; Uryu, K.; et al. Platelet-derived MRP-14 induces monocyte activation in patients with symptomatic peripheral artery disease. J. Am. Coll. Cardiol. 2018, 71, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Živković, L.; Asare, Y.; Bernhagen, J.; Dichgans, M.; Georgakis, M. CCL2/CCR2 inhibition in atherosclerosis: A meta-analysis of preclinical studies. bioRxiv 2021. [Google Scholar]
- Quinones, M.P.; Martinez, H.G.; Jimenez, F.; Estrada, C.A.; Dudley, M.; Willmon, O.; Kulkarni, H.; Reddick, R.L.; Fernandes, G.; Kuziel, W.A.; et al. CC chemokine receptor 5 influences late-stage atherosclerosis. Atherosclerosis 2007, 195, e92–e103. [Google Scholar] [CrossRef]
- de Jager, S.C.; Bongaerts, B.W.; Weber, M.; Kraaijeveld, A.O.; Rousch, M.; Dimmeler, S.; van Dieijen-Visser, M.P.; Cleutjens, K.B.; Nelemans, P.J.; van Berkel, T.J.; et al. Chemokines CCL3/MIP1α, CCL5/RANTES and CCL18/PARC are independent risk predictors of short-term mortality in patients with acute coronary syndromes. PLoS ONE 2012, 7, e45804. [Google Scholar] [CrossRef] [PubMed]
- An, Z.; Li, J.; Yu, J.; Wang, X.; Gao, H.; Zhang, W.; Wei, Z.; Zhang, J.; Zhang, Y.; Zhao, J.; et al. Neutrophil extracellular traps induced by IL-8 aggravate atherosclerosis via activation NF-κB signaling in macrophages. Cell Cycle 2019, 18, 2928–2938. [Google Scholar] [CrossRef]
- Kleemann, R.; Zadelaar, S.; Kooistra, T. Cytokines and atherosclerosis: A comprehensive review of studies in mice. Cardiovasc. Res. 2008, 79, 360–376. [Google Scholar] [CrossRef] [Green Version]
- Boesten, L.S.M.; Zadelaar, A.S.M.; van Nieuwkoop, A.; Gijbels, M.J.J.; de Winther, M.P.J.; Havekes, L.M.; van Vlijmen, B.J.M. Tumor necrosis factor-α promotes atherosclerotic lesion progression in APOE*3-leiden transgenic mice. Cardiovasc. Res. 2005, 66, 179–185. [Google Scholar] [CrossRef] [Green Version]
- Maury, C.P.; Teppo, A.M. Circulating tumour necrosis factor-alpha (cachectin) in myocardial infarction. J. Intern. Med. 1989, 225, 333–336. [Google Scholar] [CrossRef]
- Mazzone, A.; De Servi, S.; Vezzoli, M.; Fossati, G.; Mazzucchelli, I.; Gritti, D.; Ottini, E.; Mussini, A.; Specchia, G. Plasma levels of interleukin 2, 6, 10 and phenotypic characterization of circulating T lymphocytes in ischemic heart disease. Atherosclerosis 1999, 145, 369–374. [Google Scholar] [CrossRef]
- Upadhya, S.; Mooteri, S.; Peckham, N.; Pai, R.G. Atherogenic effect of interleukin-2 and antiatherogenic effect of interleukin-2 antibody in apo-e-deficient mice. Angiology 2004, 55, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Voloshyna, I.; Littlefield, M.J.; Reiss, A.B. Atherosclerosis and interferon-γ: New insights and therapeutic targets. Trends Cardiovasc. Med. 2014, 24, 45–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ranjbaran, H.; Sokol, S.I.; Gallo, A.; Eid, R.E.; Iakimov, A.O.; D’Alessio, A.; Kapoor, J.R.; Akhtar, S.; Howes, C.J.; Aslan, M.; et al. An inflammatory pathway of IFN-gamma production in coronary atherosclerosis. J. Immunol. 2007, 178, 592–604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frangogiannis, N. Transforming growth factor-β in tissue fibrosis. J. Exp. Med. 2020, 217, e20190103. [Google Scholar] [CrossRef]
- Hiebert, P.R.; Boivin, W.A.; Zhao, H.; McManus, B.M.; Granville, D.J. Perforin and granzyme B have separate and distinct roles during atherosclerotic plaque development in apolipoprotein E knockout mice. PLoS ONE 2013, 8, e78939. [Google Scholar] [CrossRef] [Green Version]
- Streicher, J.M.; Wang, Y. The role of COX-2 in heart pathology. Cardiovasc. Hematol. Agents Med. Chem. 2008, 6, 69–79. [Google Scholar] [CrossRef]
- Cipollone, F.; Prontera, C.; Pini, B.; Marini, M.; Fazia, M.; De Cesare, D.; Iezzi, A.; Ucchino, S.; Boccoli, G.; Saba, V.; et al. Overexpression of functionally coupled cyclooxygenase-2 and prostaglandin E synthase in symptomatic atherosclerotic plaques as a basis of prostaglandin E(2)-dependent plaque instability. Circulation 2001, 104, 921–927. [Google Scholar] [CrossRef] [Green Version]
- Afek, A.; Harats, D.; Roth, A.; Keren, G.; George, J. A functional role for inducible costimulator (ICOS) in atherosclerosis. Atherosclerosis 2005, 183, 57–63. [Google Scholar] [CrossRef]
- Becker, R.C.; Tracy, R.P.; Bovill, E.G.; Mann, K.G.; Ault, K. The clinical use of flow cytometry for assessing platelet activation in acute coronary syndromes. TIMI-III thrombosis and anticoagulation group. Coron. Artery Dis. 1994, 5, 339–345. [Google Scholar] [CrossRef]
- Watała, C.; Różalski, M.; Boncler, M.; Kaźmierczak, P. Badania i Publikacje w Naukach Biomedycznych; Alfa-Medica Press: Bielsko-Biała, Poland, 2011; Volume 1. [Google Scholar]



| Cytokine | Control Group | ACS | p-Value | Increment |
|---|---|---|---|---|
| IL-1β (pg/mL) | 21.7 (±12.25) | 45.50 (±15.39) | p = 0.0003 | ↑ 216% |
| TNF-α (pg/mL) | 356.33 (±105.53) | 752.33 (±169.20) | p < 0.0001 | ↑ 211% |
| IL-2 (pg/mL) | 95 (89–121.30) | 126 (111.5–156) | p = 0.011 | ↑ 133% |
| IFN-γ (pg/mL) | 206.50 (204.50–299.80) | 304 (245–340) | p = 0.033 | ↑ 128% |
| TGF-β (pM) | 100 (86.25–98.75) | 97 (94.25–102) | p = 0.678 | N/S |
| IFN-α (pg/mL) | 11.50 (9.25–19.75) | 16 (12.75–24.75) | p = 0.091 | N/S |
| No. | Assay ID | Gene Symbol | Gene Expression (2−ΔCt) ± SD in ACS |
|---|---|---|---|
| 1 | Hs00234140_m1 | CCL2 | 0.5 ± 0.3 [× 10−2] |
| 2 | Hs00174150_m1 | CCR2 | 2.0 ± 1.3 [× 10−2] |
| 3 | Hs00171266_m1 | CSF2 | 1.0 ± 0.6 [× 10−2] |
| 4 | Hs00188051_m1 | GZMB | 42 ± 27 [× 10−2] |
| 5 | Hs00359999_m1 | ICOS | 1.5 ± 1 [× 10−2] |
| No. | Assay ID | Gene Symbol | Gene Expression (2−ΔCt) for Control | Gene Expression (2−ΔCt) for ACS |
|---|---|---|---|---|
| 1 | Hs00174092_m1 | IL1A | N/A | N/A |
| 2 | Hs00174097_m1 | IL1B | 2.304 | 2.021 |
| 3 | Hs00174114_m1 | IL2 | 0.011 | N/A |
| 4 | Hs00174117_m1 | IL3 | N/A | N/A |
| 5 | Hs00174122_m1 | IL4 | N/A | N/A |
| 6 | Hs00174200_m1 | IL5 | N/A | N/A |
| 7 | Hs00174202_m1 | IL7 | N/A | N/A |
| 8 | Hs00174103_m1 | IL8 | 3.148 | 3.398 |
| 9 | Hs00174125_m1 | IL9 | N/A | N/A |
| 10 | Hs00174086_m1 | IL10 | N/A | N/A |
| 11 | Hs00168405_m1 | IL12A | 0.026 | 0.015 |
| 12 | Hs00233688_m1 | IL12B | N/A | N/A |
| 13 | Hs00174379_m1 | IL13 | N/A | N/A |
| 14 | Hs00174106_m1 | IL15 | N/A | N/A |
| 15 | Hs00174383_m1 | IL17A | N/A | N/A |
| 16 | Hs00155517_m1 | IL18 | 0.034 | 0.029 |
| 17 | Hs00171149_m1 | CCL19 | N/A | N/A |
| 18 | Hs00171041_m1 | CXCR3 | N/A | N/A |
| 19 | Hs00171042_m1 | CXCL10 | 0.025 | 0.015 |
| 20 | Hs00171138_m1 | CXCL11 | 0.016 | N/A |
| 21 | Hs00174164_m1 | CSF1 | 0.007 | 0.005 |
| 22 | Hs00357085_g1 | CSF3 | N/A | N/A |
| 23 | Hs00234174_m1 | STAT3 | 0.630 | 0.362 |
| 24 | Hs00174517_m1 | NFKB2 | 0.315 | 0.324 |
| 25 | Hs00233284_m1 | IKBKB | 0.065 | 0.035 |
| 26 | Hs00167894_m1 | CD3E | 1.046 | 1.380 |
| 27 | Hs00181217_m1 | CD4 | 0.239 | 0.271 |
| 28 | Hs00233520_m1 | CD8A | 0.509 | 0.530 |
| 29 | Hs00174333_m1 | CD19 | 0.057 | 0.014 |
| 30 | Hs00166229_m1 | IL2RA | 0.085 | 0.021 |
| 31 | Hs00174796_m1 | CD28 | 0.130 | 0.115 |
| 32 | Hs00233552_m1 | CD38 | 0.074 | 0.031 |
| 33 | Hs00365634_g1 | PTPRC | 3.397 | 2.818 |
| 34 | Hs00154355_m1 | CD68 | 1.370 | 0.653 |
| 35 | Hs00175478_m1 | CD80 | 0.009 | 0.007 |
| 36 | Hs00199349_m1 | CD86 | 0.443 | 0.347 |
| 37 | Hs00175480_m1 | CTLA4 | 0.063 | 0.070 |
| 38 | Hs00163934_m1 | CD40LG | 0.133 | 0.017 |
| 39 | Hs00219575_m1 | HLA-DRA | 7.697 | 9.746 |
| 40 | Hs99999917_m1 | HLA-DRB1 | N/A | N/A |
| 41 | Hs00203436_m1 | TBX21 | 0.057 | 0.049 |
| 42 | Hs00188346_m1 | TNFRSF18 | N/A | N/A |
| 43 | Hs00167248_m1 | NOS2 | N/A | N/A |
| 44 | Hs00169141_m1 | BCL2L1 | 0.431 | 0.239 |
| 45 | Hs00180269_m1 | BAX | 0.410 | 0.244 |
| 46 | Hs00164932_m1 | ICAM1 | 0.111 | 0.135 |
| 47 | Hs00174583_m1 | SELP | 0.218 | 0.110 |
| 48 | Hs00174057_m1 | SELE | N/A | N/A |
| 49 | Hs00157965_m1 | HMOX1 | 0.194 | 0.055 |
| 50 | Hs00189742_m1 | LRP2 | N/A | N/A |
| 51 | Hs00167927_m1 | CYP1A2 | N/A | N/A |
| 52 | Hs00167982_m1 | CYP7A1 | N/A | N/A |
| 53 | Hs00174143_m1 | IFNG | 0.008 | 0.010 |
| 54 | Hs00246266_m1 | GNLY | 4.939 | 7.386 |
| 55 | Hs00163653_m1 | FAS | 0.242 | 0.296 |
| 56 | Hs00181225_m1 | FASLG | N/A | N/A |
| 57 | Hs00171257_m1 | TGFB1 | 2.304 | 2.403 |
| 58 | Hs00232222_m1 | SMAD3 | 0.035 | 0.037 |
| 59 | Hs00178696_m1 | SMAD7 | 0.075 | 0.035 |
| 60 | Hs00365052_m1 | FN1 | N/A | N/A |
| 61 | Hs00163811_m1 | C3 | 0.014 | 0.006 |
| 62 | Hs00174128_m1 | TNF | 0.062 | 0.074 |
| 63 | Hs00236874_m1 | LTA | N/A | N/A |
| 64 | Hs00174179_m1 | ACE | N/A | N/A |
| 65 | Hs00173626_m1 | VEGFA | N/A | N/A |
| 66 | Hs00161707_m1 | SKI | 0.035 | 0.047 |
| 67 | Hs00156373_m1 | CD34 | N/A | N/A |
| 68 | Hs00241341_m1 | AGTR1 | N/A | N/A |
| 69 | Hs00169126_m1 | AGTR2 | N/A | N/A |
| 70 | Hs00174961_m1 | EDN1 | 0.043 | N/A |
| 71 | Hs00171455_m1 | LIF | N/A | N/A |
| 72 | Hs00209771_m1 | LY96 | 0.043 | 0.618 |
| 73 | Hs00236988_g1 | MIF | 6.889 | 5.080 |
| 74 | Hs00190046_m1 | NFATC3 | 1.664 | 1.735 |
| 75 | Hs00190037_m1 | NFATC4 | N/A | N/A |
| 76 | Hs00236998_m1 | PF4 | 18.692 | 14.772 |
| 77 | Hs00374292_m1 | SYK | N/A | N/A |
| Characteristic [Clinical Standards] | Control Group (n = 12) | Study Group (ACS) (n = 12) |
|---|---|---|
| Age | 55 ± 6.61 | 57 ± 11 (p = 0.100; n.s.) |
| Gender (M/F) | M8/F4 | M8/F4 |
| BMI [<30] | 26.40 ± 6.61 | 21.9 ± 4.85 (p = 0.413; n.s.) |
| Triglycerides [<2.5 mmol/L] | 1.18 ± 0.54 | 2.17 ± 0.82 (p = 0.005) |
| Total cholesterol [3–5 mmol/L] | 4.02 ± 1.34 | 4.68 ± 1.12 (p = 0.196; n.s.) |
| HDL [>1 mmol/L] | 1.31 ± 0.53 | 1.17 ± 0.21 (p = 0.700; n.s.) |
| LDL [<2.9 mmol/L] | 2.89 ± 1.09 | 1.54 ± 0.97 (p = 0.413; n.s.) |
| Leukocytes on admission [4–11 × 103/μL] | 7.78 ± 2.52 | 9.63 ± 2.42 (p = 0.027) |
| Erythrocytes on admission [4–6 × 106/μL] | 5.08 ± 0.41 | 4.72 ± 0.66 (p = 0.118) |
| Platelets on admission [150–400 × 103/μL] | 316.17 ± 119.70 | 242.3 ± 87.2 (p = 0.098; n.s.) |
| Creatinine [64–104 μmol/L] | 83.94 ± 15.70 | 84.25 ± 14.01 (p = 0.960; n.s.) |
| Troponin peak (hs-TnT) [<14 ng/L] | 13.34 ± 0.23 | 2778 ± 2361.33 (p = 0.0005) |
| ALT [0–45 UI] | 23.92 ± 12.80 | 30.07 ± 13.18 (p = 0.259) |
| AST [0–35 UI] | 21.30 ± 7.12 | 33.44 ± 15.07 (p = 0.019) |
| Glucose [4.1–5.5 mmol/L] | 5.07 ± 0.60 | 5.40 ± 0.44 (p = 0.131) |
| GFR [ml/min./1.73 m2] | 94 ± 14 | 93 ± 9 (p = 0.735) |
| NSTEMI/STEMI | - | 4/8 |
| Localization of culprit lesion | - | Cx (n = 5); RCA (n = 4); LCA (n = 1); LAD (n = 2) |
| Statins administration | - | Patients before admission to hospital were not treated with statins. All patients received first dose of statin (at least 20 mg dose of Rosuvastatin) during the first 24 h of ACS. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dziedzic, A.; Szelenberger, R.; Kacprzak, M.; Czarny, P.; Synowiec, E.; Saluk-Bijak, J.; Sliwinski, T.; Zielinska, M.; Bijak, M. Variations in the Gene Expression Profile in Atherosclerotic Patients with Non-Fatal ACS: A Preliminary Study. Int. J. Mol. Sci. 2022, 23, 5017. https://doi.org/10.3390/ijms23095017
Dziedzic A, Szelenberger R, Kacprzak M, Czarny P, Synowiec E, Saluk-Bijak J, Sliwinski T, Zielinska M, Bijak M. Variations in the Gene Expression Profile in Atherosclerotic Patients with Non-Fatal ACS: A Preliminary Study. International Journal of Molecular Sciences. 2022; 23(9):5017. https://doi.org/10.3390/ijms23095017
Chicago/Turabian StyleDziedzic, Angela, Rafal Szelenberger, Michal Kacprzak, Piotr Czarny, Ewelina Synowiec, Joanna Saluk-Bijak, Tomasz Sliwinski, Marzenna Zielinska, and Michal Bijak. 2022. "Variations in the Gene Expression Profile in Atherosclerotic Patients with Non-Fatal ACS: A Preliminary Study" International Journal of Molecular Sciences 23, no. 9: 5017. https://doi.org/10.3390/ijms23095017








