Relative Contribution of Blood Pressure and Renal Sympathetic Nerve Activity to Proximal Tubular Sodium Reabsorption via NHE3 Activity
Abstract
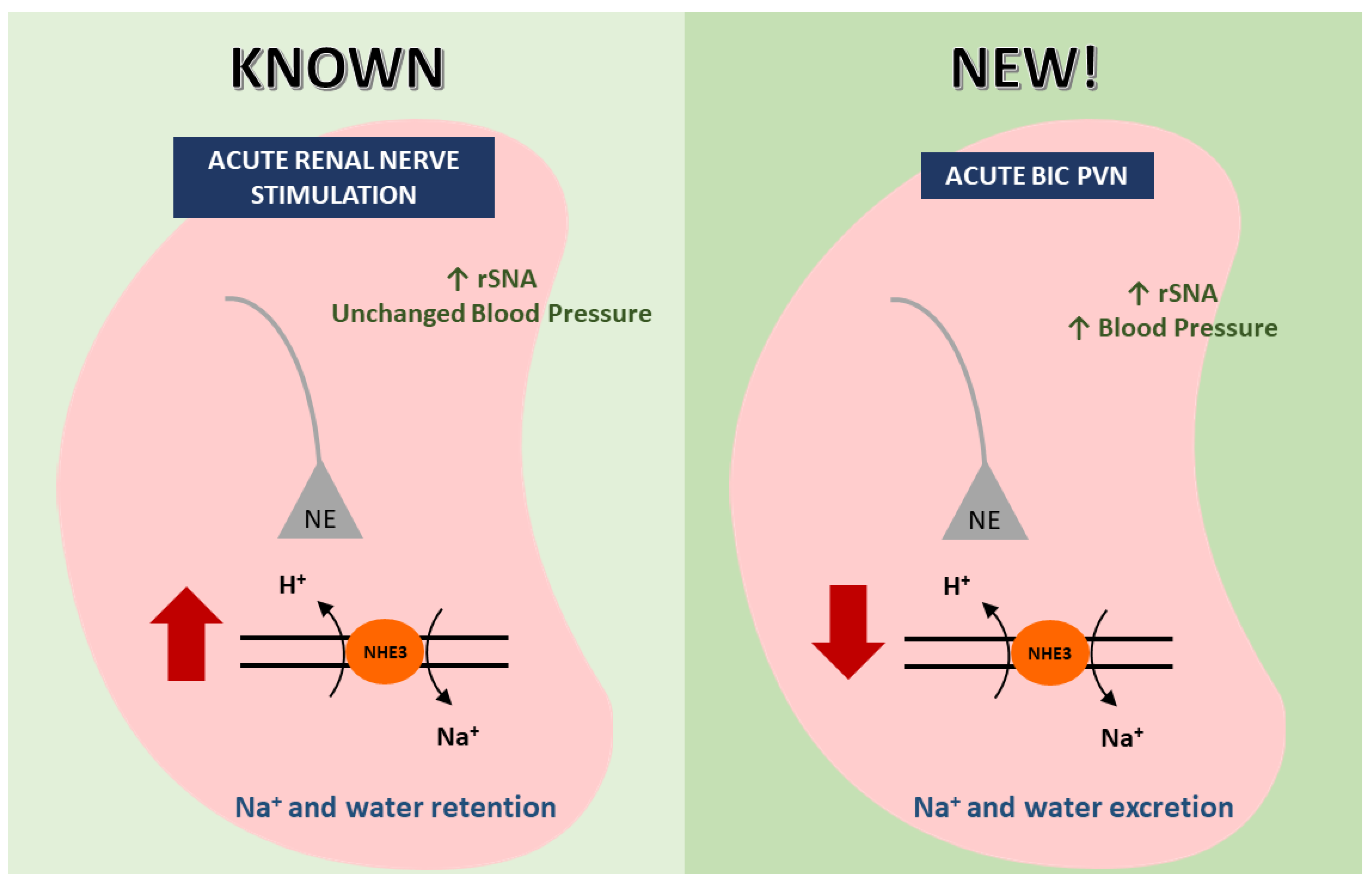
:1. Introduction
2. Results
2.1. First and Second Series of Experiments
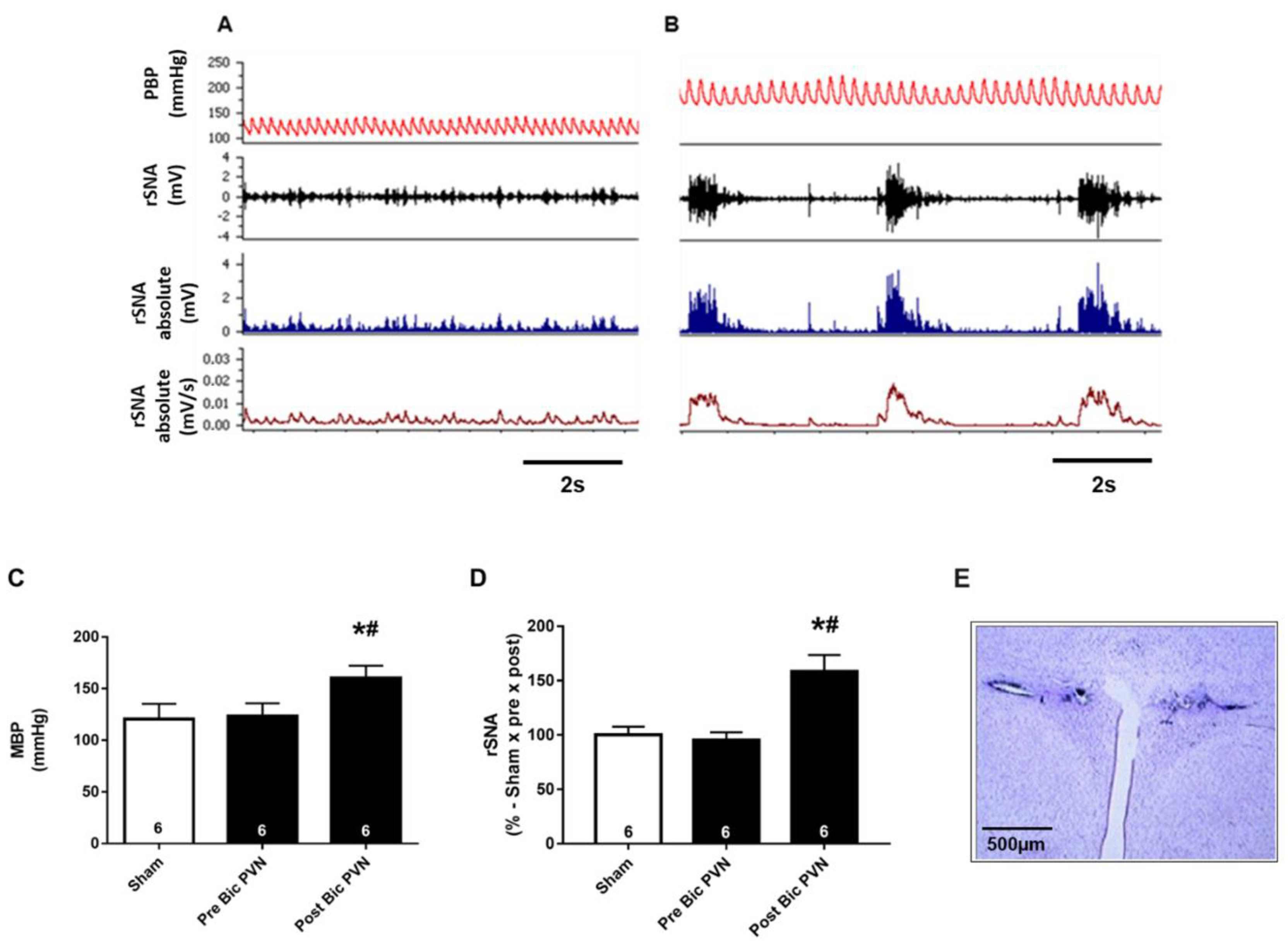
2.1.1. Bic Injection into the PVN Increases BP and rSNA
2.1.2. Bic Injection in the PVN Increases Urinary Flow Rate and Urinary Sodium Rate but Not Creatinine Clearance
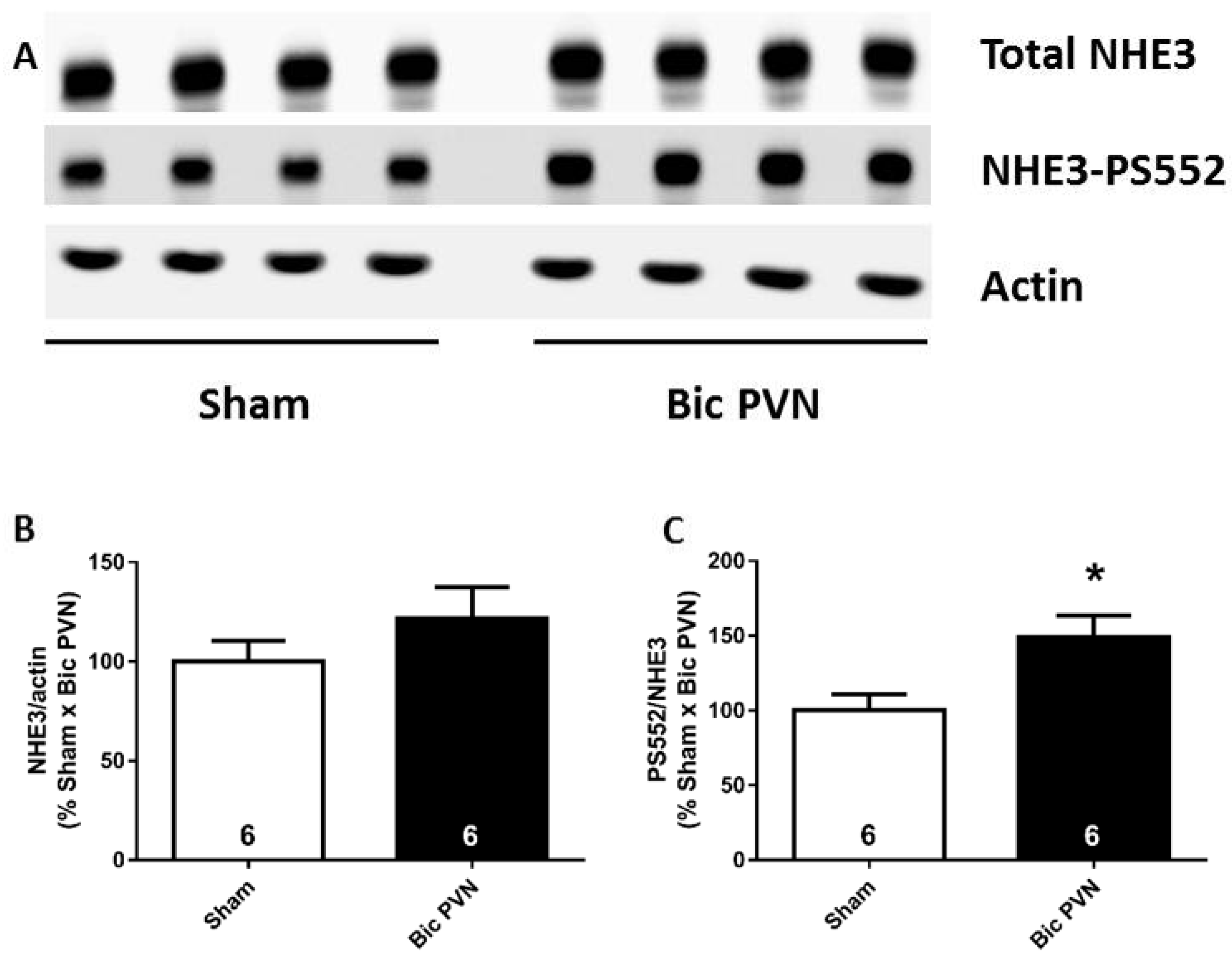
2.1.3. Bic Injection in the PVN Increases NHE3 Phosphorylation Levels at Serine 552
2.2. Third Series of Experiments
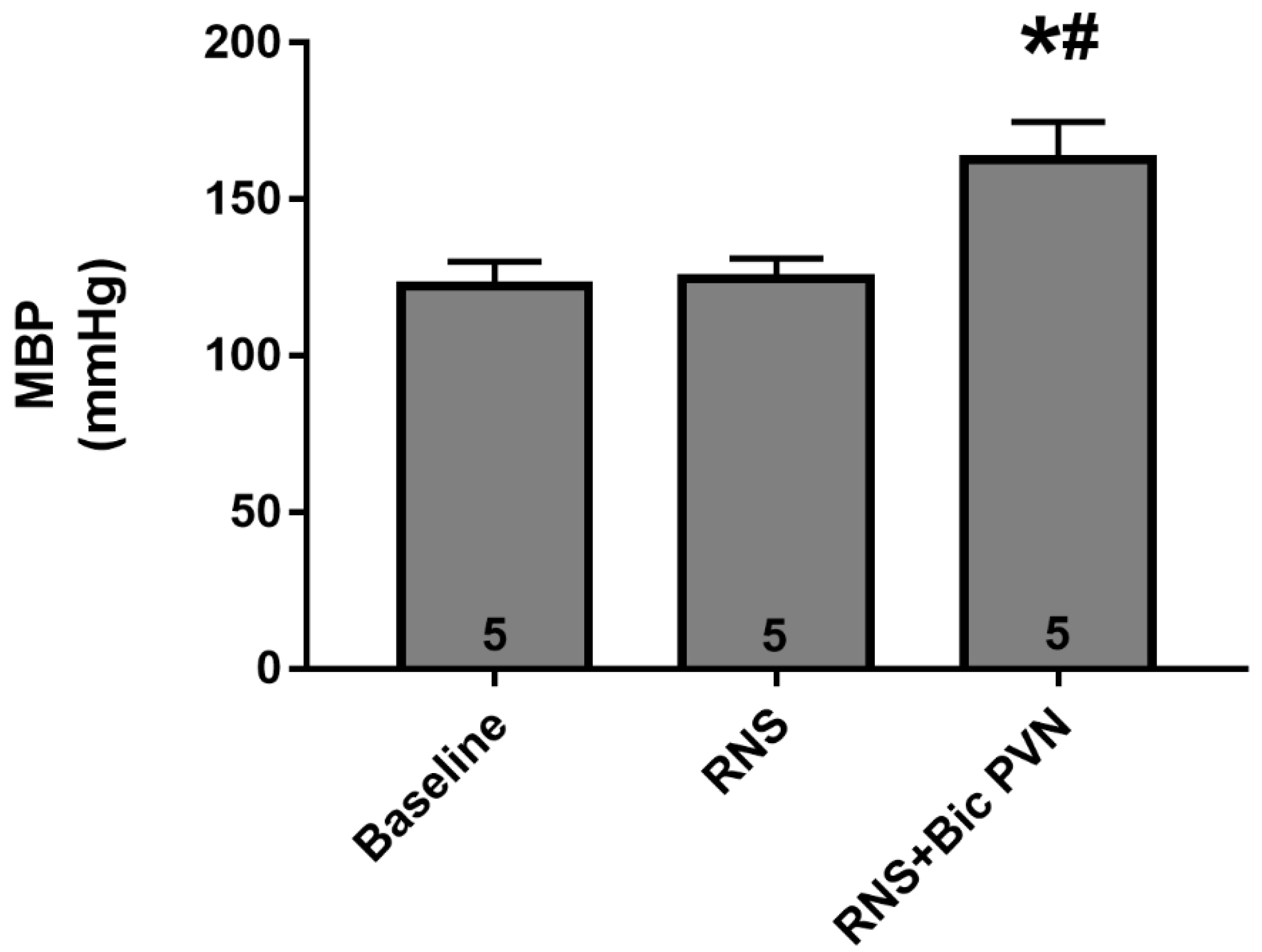
2.2.1. Effect of RNS and RNS + Bic PVN on BP
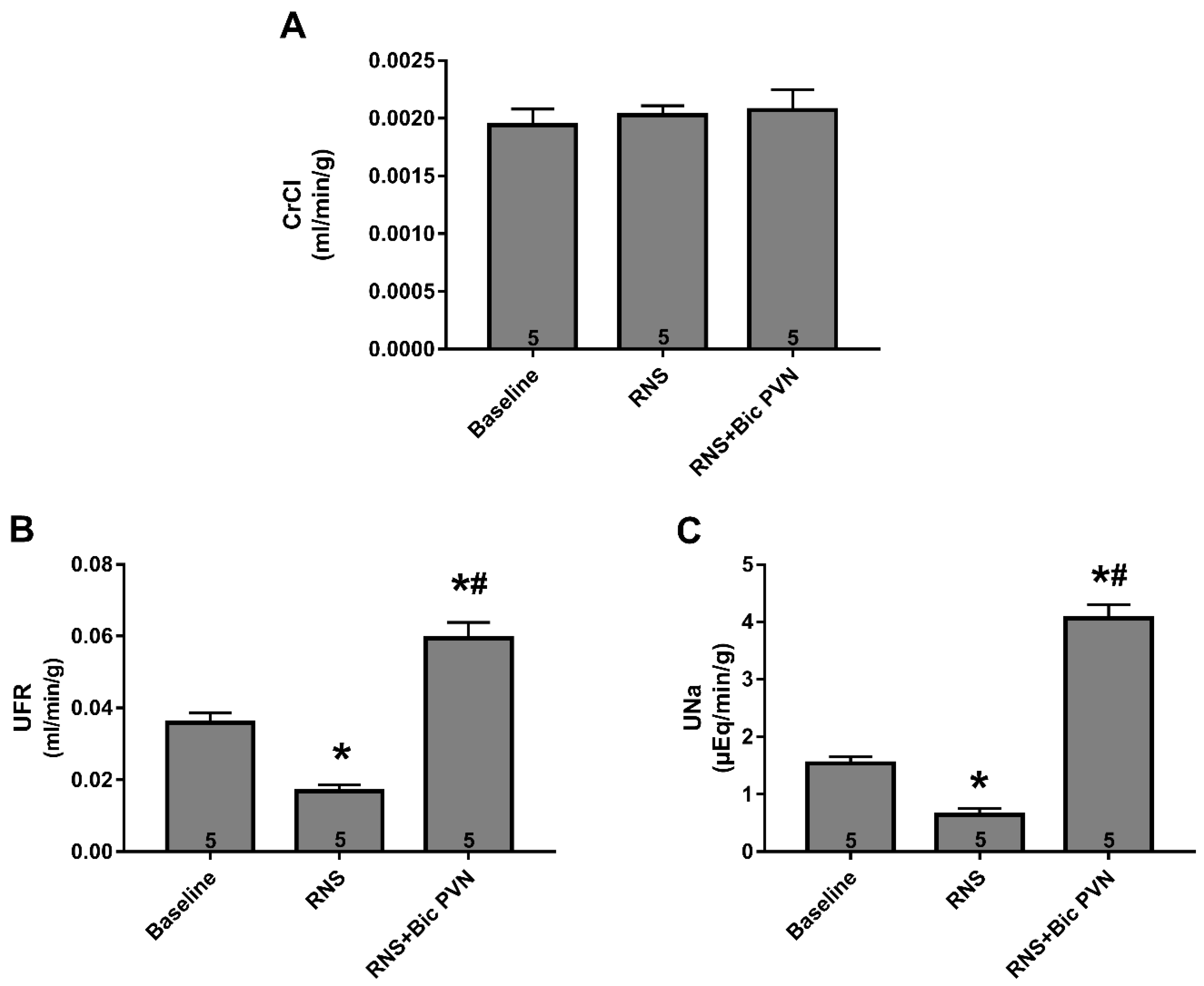
2.2.2. Effect of RNS and RNS + Bic PVN on Renal Function: CrCl, UFR, and UNa
3. Discussion
3.1. Limitations of the Present Study
3.2. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Animals
4.3. Study Design
4.4. rSNA Signal Acquisition
4.5. RNS
4.6. Bic PVN Injection
4.7. Analysis of Baseline rSNA
4.8. Plasma and Urine Analyses
4.9. Renal Cortical Membrane Protein Isolation
4.10. Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis and Immunoblotting
4.11. Statistics Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- DiBona, G.F.; Kopp, U.C. Neural control of renal function. Physiol. Rev. 1997, 77, 75–197. [Google Scholar] [CrossRef] [PubMed]
- Guyenet, P.G. The sympathetic control of blood pressure. Nat. Rev. Neurosci. 2006, 7, 335–346. [Google Scholar] [CrossRef] [PubMed]
- Akine, A.; Montanaro, M.; Allen, A.M. Hypothalamic paraventricular nucleus inhibition decreases renal sympathetic nerve activity in hypertensive and normotensive rats. Auton Neurosci. 2003, 108, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Dampney, R.A.; Michelini, L.C.; Li, D.P.; Pan, H.L. Regulation of sympathetic vasomotor activity by the hypothalamic paraventricular nucleus in normotensive and hypertensive states. Am. J. Physiol. Heart Circ. Physiol. 2018, 315, H1200–H1214. [Google Scholar] [CrossRef] [Green Version]
- Chen, Q.H.; Haywood, J.R.; Toney, G.M. Sympathoexcitation by PVN-injected bicuculline requires activation of excitatory amino acid receptors. Hypertension 2003, 42, 725–731. [Google Scholar] [CrossRef] [Green Version]
- Bell-Reuss, E.; Trevino, D.L.; Gottschalk, C.W. Effect of renal sympathetic nerve stimulation on proximal water and sodium reabsorption. J. Clin. Investig. 1976, 57, 1104–1107. [Google Scholar] [CrossRef] [Green Version]
- Healy, V.; Thompson, C.; Johns, E.J. The adrenergic regulation of proximal tubular Na+/H+ exchanger 3 in the rat. Acta Physiol. 2014, 210, 678–689. [Google Scholar] [CrossRef]
- Pontes, R.B.; Crajoinas, R.O.; Nishi, E.E.; Oliveira-Sales, E.B.; Girardi, A.C.; Campos, R.R.; Bergamaschi, C.T. Renal nerve stimulation leads to the activation of the Na+/H+ exchanger isoform 3 via angiotensin II type I receptor. Am. J. Physiol. Renal. Physiol. 2015, 308, F848–F856. [Google Scholar] [CrossRef] [Green Version]
- Pontes, R.B.; Girardi, A.C.; Nishi, E.E.; Campos, R.R.; Bergamaschi, C.T. Crosstalk between the renal sympathetic nerve and intrarenal angiotensin II modulates proximal tubular sodium reabsorption. Exp. Physiol. 2015, 100, 502–506. [Google Scholar] [CrossRef]
- Aronson, P.S. Ion exchangers mediating NaCl transport in the renal proximal tubule. Cell Biochem. Biophys. 2002, 36, 147–153. [Google Scholar] [CrossRef]
- McDonough, A.A. Mechanisms of proximal tubule sodium transport regulation that link extracellular fluid volume and blood pressure. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R851–R861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.C.; Zhu, D.; Chen, X.; Zheng, X.; Zhao, C.; Zhang, J.; Soleimani, M.; Rubera, I.; Tauc, M.; Zhou, X.; et al. Proximal Tubule-Specific Deletion of the NHE3 (Na+/H+ Exchanger 3) in the Kidney Attenuates Ang II (Angiotensin II)-Induced Hypertension in Mice. Hypertension 2019, 74, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.T.; Lee, D.H.; Delpire, E.; McDonough, A.A. Differential regulation of Na+ transporters along nephron during ANG II-dependent hypertension: Distal stimulation counteracted by proximal inhibition. Am. J. Physiol. Renal. Physiol. 2013, 305, F510–F519. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nishi, E.E.; Almeida, V.R.; Amaral, F.G.; Simon, K.A.; Futuro-Neto, H.A.; Pontes, R.B.; Cespedes, J.G.; Campos, R.R.; Bergamaschi, C.T. Melatonin attenuates renal sympathetic overactivity and reactive oxygen species in the brain in neurogenic hypertension. Hypertens. Res. 2019, 42, 1683–1691. [Google Scholar] [CrossRef] [PubMed]
- Oliveira-Sales, E.B.; Nishi, E.E.; Carillo, B.A.; Boim, M.A.; Dolnikoff, M.S.; Bergamaschi, C.T.; Campos, R.R. Oxidative stress in the sympathetic premotor neurons contributes to sympathetic activation in renovascular hypertension. Am. J. Hypertens. 2009, 22, 484–492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oliveira-Sales, E.B.; Toward, M.A.; Campos, R.R.; Paton, J.F. Revealing the role of the autonomic nervous system in the development and maintenance of Goldblatt hypertension in rats. Auton. Neurosci. 2014, 183, 23–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zambrano, L.I.; Pontes, R.B.; Garcia, M.L.; Nishi, E.E.; Nogueira, F.N.; Higa, E.M.S.; Cespedes, J.G.; Bergamaschi, C.T.; Campos, R.R. Pattern of sympathetic vasomotor activity in a model of hypertension induced by nitric oxide synthase blockade. Physiol. Rep. 2019, 7, e14183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Esler, M.D.; Krum, H.; Sobotka, P.A.; Schlaich, M.P.; Schmieder, R.E.; Böhm, M.; Symplicity HTN-2 Investigators. Renal sympathetic denervation in patients with treatment-resistant hypertension (The Symplicity HTN-2 Trial): A randomised controlled trial. Lancet 2010, 376, 1903–1909. [Google Scholar]
- Krum, H.; Schlaich, M.; Whitbourn, R.; Sobotka, P.A.; Sadowski, J.; Bartus, K.; Kapelak, B.; Walton, A.; Sievert, H.; Thambar, S.; et al. Catheter-based renal sympathetic denervation for resistant hypertension: A multicentre safety and proof-of-principle cohort study. Lancet 2009, 373, 1275–1281. [Google Scholar] [CrossRef]
- Schlaich, M.P.; Sobotka, P.A.; Krum, H.; Lambert, E.; Esler, M.D. Renal sympathetic-nerve ablation for uncontrolled hypertension. N. Engl. J. Med. 2009, 361, 932–934. [Google Scholar] [CrossRef]
- Kocinsky, H.S.; Girardi, A.C.; Biemesderfer, D.; Nguyen, T.; Mentone, S.; Orlowski, J.; Aronson, P.S. Use of phospho-specific antibodies to determine the phosphorylation of endogenous Na+/H+ exchanger NHE3 at PKA consensus sites. Am. J. Physiol. Renal. Physiol. 2005, 289, F249–F258. [Google Scholar] [CrossRef] [PubMed]
- Crajoinas, R.O.; Lessa, L.M.; Carraro-Lacroix, L.R.; Davel, A.P.; Pacheco, B.P.; Rossoni, L.V.; Malnic, G.; Girardi, A.C. Posttranslational mechanisms associated with reduced NHE3 activity in adult vs. young prehypertensive SHR. Am. J. Physiol. Renal. Physiol. 2010, 299, F872–F881. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crajoinas, R.O.; Pessoa, T.D.; Rodrigues, M.V.; Malnic, G.; Girardi, A.C. Changes in the activity and expression of protein phosphatase-1 accompany the differential regulation of NHE3 before and after the onset of hypertension in spontaneously hypertensive rats. Acta Physiol. 2014, 211, 395–408. [Google Scholar] [CrossRef] [PubMed]
- Veiras, L.C.; Girardi, A.C.C.; Curry, J.; Pei, L.; Ralph, D.L.; Tran, A.; Castelo-Branco, R.C.; Pastor-Soler, N.; Arranz, C.T.; Yu, A.S.L.; et al. Sexual Dimorphic Pattern of Renal Transporters and Electrolyte Homeostasis. J. Am. Soc. Nephrol. 2017, 28, 3504–3517. [Google Scholar] [CrossRef] [Green Version]
- Garcia, M.L.; Pontes, R.B.; Nishi, E.E.; Ibuki, F.K.; Oliveira, V.; Sawaya, A.C.; Carvalho, P.O.; Nogueira, F.N.; Franco, M.D.; Campos, R.R.; et al. The antioxidant effects of green tea reduces blood pressure and sympathoexcitation in an experimental model of hypertension. J. Hypertens. 2017, 35, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Fardin, N.M.; Oyama, L.M.; Campos, R.R. Changes in baroreflex control of renal sympathetic nerve activity in high-fat-fed rats as a predictor of hypertension. Obesity 2012, 20, 1591–1597. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Porter, J.P.; Brody, M.J. A comparison of the hemodynamic effects produced by electrical stimulation of subnuclei of the paraventricular nucleus. Brain Res. 1986, 375, 20–29. [Google Scholar] [CrossRef]
- Dampney, R.A. Functional organization of central pathways regulating the cardiovascular system. Physiol. Rev. 1994, 74, 323–364. [Google Scholar] [CrossRef] [PubMed]
- Borges-Júnior, F.A.; Silva Dos Santos, D.; Benetti, A.; Polidoro, J.Z.; Wisnivesky, A.C.T.; Crajoinas, R.O.; Antônio, E.L.; Jensen, L.; Caramelli, B.; Malnic, G.; et al. Empagliflozin Inhibits Proximal Tubule NHE3 Activity, Preserves GFR, and Restores Euvolemia in Nondiabetic Rats with Induced Heart Failure. J. Am. Soc. Nephrol. 2021, 32, 1616–1629. [Google Scholar] [CrossRef]
- Crajoinas, R.O.; Polidoro, J.Z.; Carneiro de Morais, C.P.; Castelo-Branco, R.C.; Girardi, A.C. Angiotensin II counteracts the effects of cAMP/PKA on NHE3 activity and phosphorylation in proximal tubule cells. Am. J. Physiol. Cell Physiol. 2016, 311, C768–C776. [Google Scholar] [CrossRef] [Green Version]
- Martins, F.L.; Bailey, M.A.; Girardi, A.C.C. Endogenous Activation of Glucagon-Like Peptide-1 Receptor Contributes to Blood Pressure Control: Role of Proximal Tubule Na+/H+ Exchanger Isoform 3, Renal Angiotensin II, and Insulin Sensitivity. Hypertension 2020, 76, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Luchi, W.M.; Crajoinas, R.O.; Martins, F.L.; Castro, P.C.; Venturini, G.; Seguro, A.C.; Girardi, A.C.C. High blood pressure induced by vitamin D deficiency is associated with renal overexpression and hyperphosphorylation of Na+-K+-2Cl- cotransporter type 2. J. Hypertens. 2021, 39, 880–891. [Google Scholar] [CrossRef] [PubMed]
- Bello-Reuss, E.; Pastoriza-Muńoz, E.; Colindres, R.E. Acute unilateral renal denervation in rats with extracellular volume expansion. Am. J. Physiol. 1977, 232, F26–F32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carillo, B.A.; Oliveira-Sales, E.B.; Andersen, M.; Tufik, S.; Hipolide, D.; Santos, A.A.; Tucci, P.J.; Bergamaschi, C.T.; Campos, R.R. Changes in GABAergic inputs in the paraventricular nucleus maintain sympathetic vasomotor tone in chronic heart failure. Auton. Neurosci. 2012, 171, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pontes, R.B.; Nishi, E.E.; Crajoinas, R.O.; Milanez, M.I.O.; Girardi, A.C.C.; Campos, R.R.; Bergamaschi, C.T. Relative Contribution of Blood Pressure and Renal Sympathetic Nerve Activity to Proximal Tubular Sodium Reabsorption via NHE3 Activity. Int. J. Mol. Sci. 2023, 24, 349. https://doi.org/10.3390/ijms24010349
Pontes RB, Nishi EE, Crajoinas RO, Milanez MIO, Girardi ACC, Campos RR, Bergamaschi CT. Relative Contribution of Blood Pressure and Renal Sympathetic Nerve Activity to Proximal Tubular Sodium Reabsorption via NHE3 Activity. International Journal of Molecular Sciences. 2023; 24(1):349. https://doi.org/10.3390/ijms24010349
Chicago/Turabian StylePontes, Roberto B., Erika E. Nishi, Renato O. Crajoinas, Maycon I. O. Milanez, Adriana C. C. Girardi, Ruy R Campos, and Cassia T Bergamaschi. 2023. "Relative Contribution of Blood Pressure and Renal Sympathetic Nerve Activity to Proximal Tubular Sodium Reabsorption via NHE3 Activity" International Journal of Molecular Sciences 24, no. 1: 349. https://doi.org/10.3390/ijms24010349
APA StylePontes, R. B., Nishi, E. E., Crajoinas, R. O., Milanez, M. I. O., Girardi, A. C. C., Campos, R. R., & Bergamaschi, C. T. (2023). Relative Contribution of Blood Pressure and Renal Sympathetic Nerve Activity to Proximal Tubular Sodium Reabsorption via NHE3 Activity. International Journal of Molecular Sciences, 24(1), 349. https://doi.org/10.3390/ijms24010349





