Abstract
Obesity has a noteworthy role in breast tumor initiation and progression. Among the mechanisms proposed, the most validated is the development of chronic low-grade inflammation, supported by immune cell infiltration along with dysfunction in adipose tissue biology, characterized by an imbalance in adipocytokines secretion and alteration of their receptors within the tumor microenvironment. Many of these receptors belong to the seven-transmembrane receptor family, which are involved in physiological features, such as immune responses and metabolism, as well as in the development and progression of several malignancies, including breast cancer. These receptors are classified as canonical (G protein-coupled receptors, GPCRs) and atypical receptors, which fail to interact and activate G proteins. Among the atypical receptors, adiponectin receptors (AdipoRs) mediate the effect of adiponectin, the most abundant adipocytes-derived hormone, on breast cancer cell proliferation, whose serum levels are reduced in obesity. The adiponectin/AdipoRs axis is becoming increasingly important regarding its role in breast tumorigenesis and as a therapeutic target for breast cancer treatment. The objectives of this review are as follows: to point out the structural and functional differences between GPCRs and AdipoRs, and to focus on the effect of AdipoRs activation in the development and progression of obesity-dependent breast cancer.
1. Introduction
Obesity is a multifactorial metabolic disorder characterized by an abnormal accumulation of adipose tissue and is associated with an increased incidence of several diseases and malignancies [1]. The World Health Organization (WHO) and the National Institute of Health (NIH) have provided accurate guidelines to clinically define an obese subject, according to the body mass index (BMI). Although BMI is the mainstream indicator for diagnosing obesity, it is not sufficient to explain the complexity of obesity status and its related disorders [2]. Particularly, it has been evidenced that body composition and adipose tissue depots distribution also play a relevant role in contributing to metabolic and hormonal abnormalities, which are linked to an increased risk of developing several cancer types, including breast cancer [2]. In this context, it has been observed that the association between breast cancer risk and metabolic dysfunction is independent of BMI. Indeed, individuals with metabolically unhealthy normal weight or overweight/obesity may have an increased risk and incidence of breast cancer compared to metabolically healthy normal-weight individuals [3].
Obesity and breast cancer are both public health issues, and strong evidence correlates obesity with worse disease-free and overall survival [4].
Currently, breast cancer is the most commonly diagnosed cancer and represents the leading cause of cancer-related mortality among women worldwide. According to the latest cancer data accessible from the GLOBOCAN 2020 database, globally breast cancer accounts for 24.5% of all new cancer cases and 15.5% of all cancer-related deaths, with an estimated number of incident cases of 531.086 and 141.765 deaths in Europe [5]. Tumor initiation and development are closely dependent on the cross-talk between breast epithelial cells and the surrounding tumor microenvironment (TME), mainly composed of adipocytes, peripheral immune and inflammatory cells, cancer-associated fibroblasts and microvessels, embedded with various cytokines and chemokines [6].
Among the cellular components of TME, adipocytes are the main actors in regard to the link between obesity and breast cancer risk. Indeed, adipose tissue has been identified as an active organ engaged in the regulation of inflammation, metabolism, and tissue remodeling through the production of many bioactive polypeptides known as adipokines [7].
Obesity is associated with chronic low-grade inflammation, which is related to multiple changes in adipose tissue biology, including dysfunction in adipokines secretion such as adiponectin [1].
A large body of evidence suggests that the infiltration of proinflammatory macrophages into the adipose tissue, resulting in the secretion of inflammatory mediators, contributes to the maintenance of chronic low-grade inflammation, an essential condition to create a suitable TME for breast cancer development, progression, and metastasis [6,8].
These secretory factors act by binding to specific receptors, whose regulation is affected by autocrine, paracrine and endocrine mechanisms. Most of these receptors are cell surface receptors belonging to the seven-transmembrane receptor family. This receptor group consists of two distinct subfamilies, canonical G protein-coupled receptors (GPCRs) and atypical receptors, characterized by a number of peculiar structural and functional properties [9,10]. Although they share a seven-transmembrane architecture, each receptor subfamily presents a unique conformation and the ability to initiate signaling through the engagement of distinct transduction molecules.
There are two main hallmarks for a protein to be classified as a GPCR: the presence of seven α-helices that span the plasma membrane, and the ability of the receptor to interact with a G-protein [9,10].
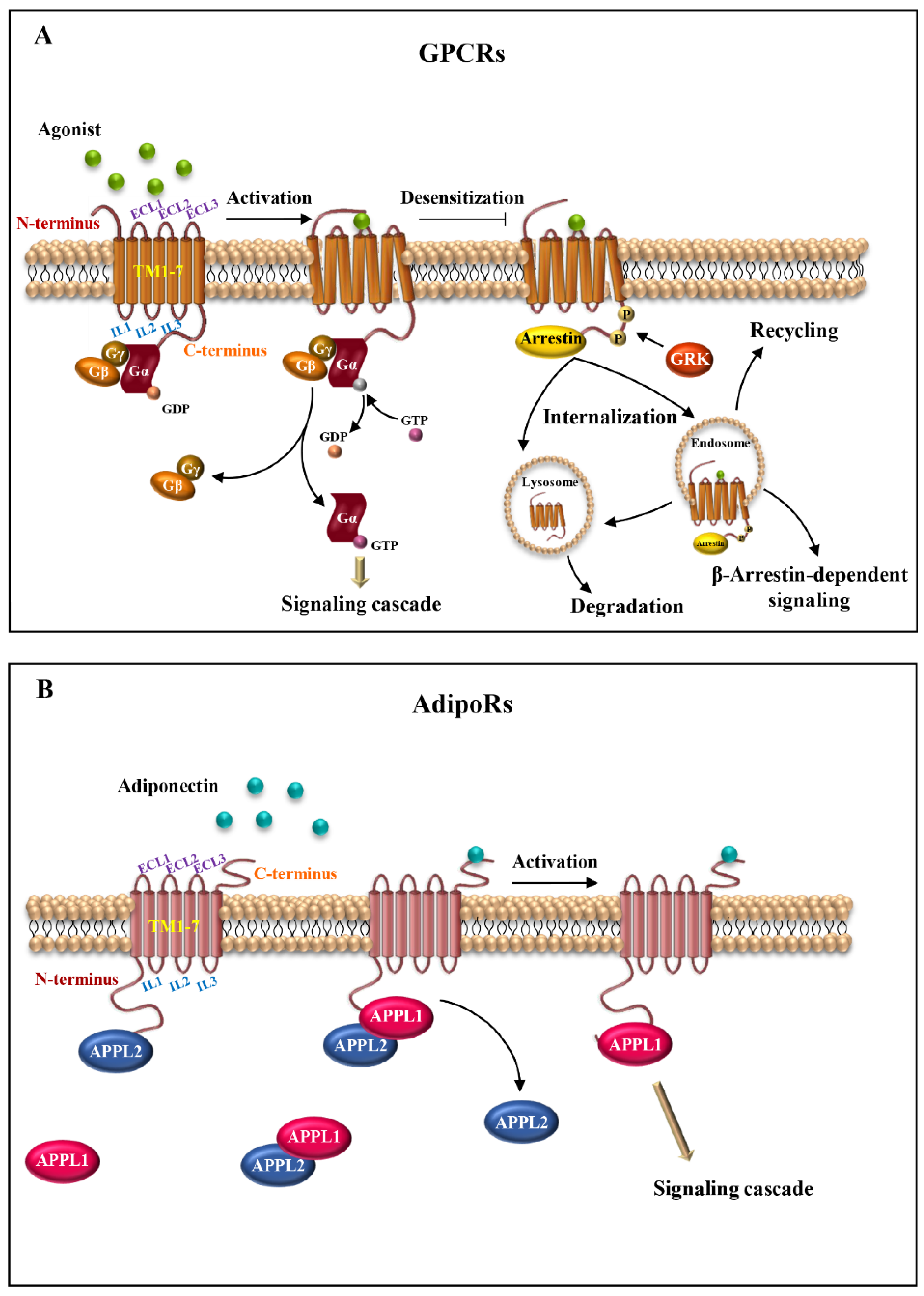
Structurally, these receptors consist of three parts: the extracellular, the transmembrane (TM) and the intracellular region. The extracellular region contains the N-terminus and three extracellular loops (ECL1-ECL3); the transmembrane region is composed of seven transmembrane domains (alpha-helices, TM1-TM7) that span the plasma membrane in a counter-clockwise manner, forming a recognition and connection unit; the intracellular region presents three intracellular loops (ICL1-ICL3), a short intracellular amphipathic alpha-helix (H8) and the C-terminus [9,10]. The extracellular region regulates the interaction with the ligands; the TM regions bind the ligands and transduce the signal to the intracellular side, causing a conformational change, which allows the interaction with cytosolic signaling proteins (Figure 1A) [11].
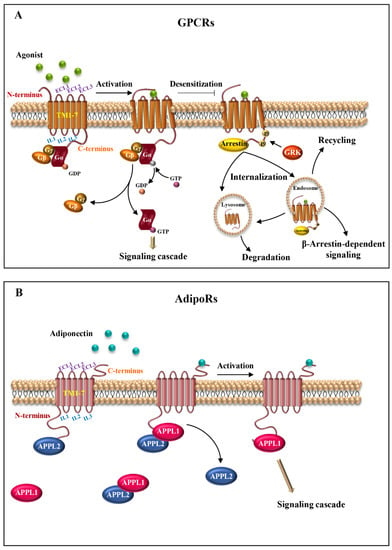
Figure 1.
Seven-transmembrane receptor structure and activation. (A) Schematic representation of the basic structure of GPCRs. The receptors comprise seven transmembrane helices (TM1-7) connected by intracellular (IL1-3) and extracellular (ECL1-3) loops, with extracellular N-terminus and intracellular C-terminus. The binding of the agonist to the receptor induces conformational changes and activation of heterotrimeric G proteins (αβγ). Next, after the exchange between GDP and GTP, α-subunit dissociates from βγ dimer, initiating signal cascades. (B) Illustrative scheme of AdipoRs structure organization. AdipoRs are atypical seven-transmembrane proteins, consisting of seven integral membrane domains (TM1-7) and extracellular and intracellular loops (ECL1-3 and IL1-3, respectively), with an internal N-terminus and an external C-terminus. AdipoRs signaling is mediated through APPL isoforms, APPL1 and APPL2. In the absence of adiponectin, APPL2 binds to the N-terminal domain of AdipoRs or forms an APPL1/APPL2 heterodimer, preventing receptors activation. Conversely, in the presence of adiponectin, APPL2 dissociates from APPL1 and AdipoRs. Adiponectin/AdipoRs interaction promotes APPL1 association and subsequent activation of different signaling pathways.
These receptors transduce a wide array of signals and regulate many cellular physiological and pathological processes, representing attractive drug targets.
GPCRs detect multiple extracellular signals and activate intracellular responses through the interaction with heterotrimeric G-proteins (α, β and γ subunits), initiating a cascade of G protein-mediated signaling pathways, which are also involved in the desensitization and internalization of the receptor (Figure 1A) [9].
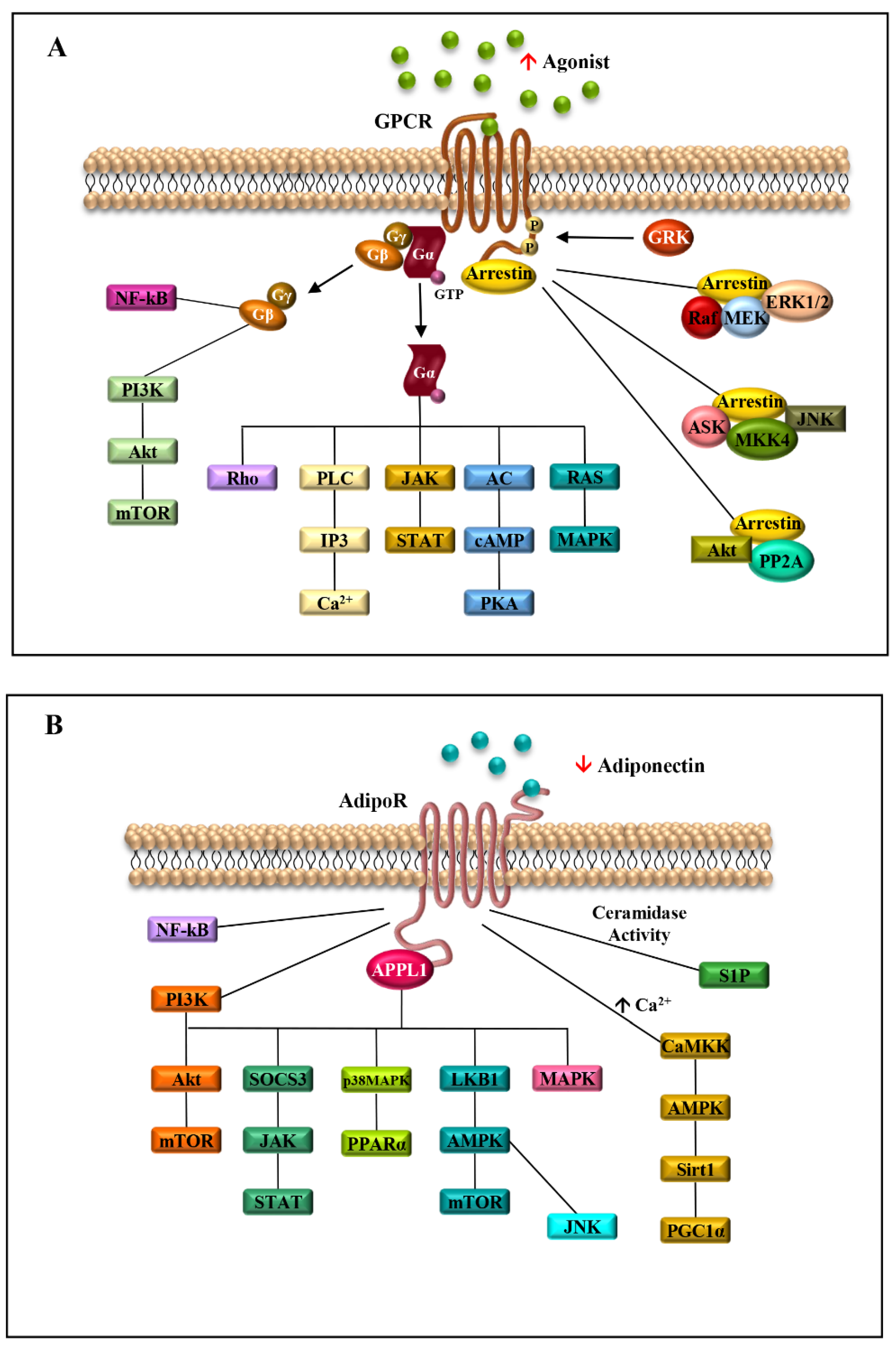
Due to the great heterogeneity and their involvement in a plethora of biological processes, GPCRs take part in the activation of a variety of intracellular signalings, such as ERK1/2, JNK, p38 mitogen-activated protein kinases (MAPKs) and PI3K/Akt (Figure 2) [12].
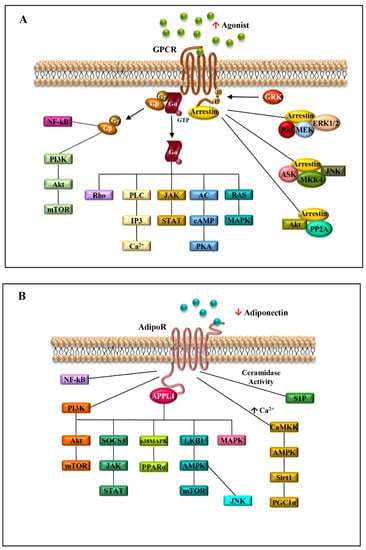
Figure 2.
Seven-transmembrane receptors signaling pathways activated in obesity-related breast cancer. Schematic representation of the signaling cascades activated by the binding of agonists to GPCRs (A) or adiponectin to AdipoRs (B) in obesity conditions. ↑ agonist indicates the high level of GPCR agonists in obesity; ↓ adiponectin shows the reduced level of adiponectin, typical of obese status.
The seven-transmembrane receptors are key regulator elements in a wide range of physiological processes, including hormonal signal transduction, cell survival, proliferation and differentiation [13].
In the last few years, many studies have demonstrated evidence that different cell types (e.g., immune, normal mesenchymal, stromal and tumor cells) express atypical chemokine receptors (ACKRs) that are seven-transmembrane cell surface proteins. These receptors regulate cell migration in inflammatory, immune and pathological contexts. ACKRs, in contrast to canonical GPCRs, fail to induce the full spectrum of signaling activated by G proteins. This is due to a structural peculiarity in ACKRs represented by the modified DRYLAIV motif within the second intracellular loop. These amino acid substitutions make ACKRs unable to bind to the G protein α subunit and to activate the related downstream signaling and cellular responses [14,15].
A singular case of seven-transmembrane domain receptors, not coupled with G proteins, is represented by the adiponectin receptors, AdipoR1 and AdipoR2 (AdipoRs) [7,16].
Adiponectin is one of the adipokines produced by adipose tissue that has recently attracted more attention for its crucial role in obesity-related disorders [1,17].
In adipocytes, adiponectin is produced as a monomeric protein, which undergoes posttranslational modification into different multimers before secretion into the bloodstream [18]. AdipoRs are ubiquitously expressed with a predominant level of one of the two receptor subtypes depending on the specific tissue. AdipoR1 is highly expressed in adipose tissue, suggesting that adiponectin may exert its biological effects in an autocrine/paracrine manner [7,19]. Paradoxically, in the adipose tissue of obese individuals, AdipoR1 expression is reduced, whereas weight loss elicits its increase [20].
AdipoRs regulate a plethora of biological effects, including insulin-sensitizing, anti-inflammatory, anti-angiogenic and vasodilatory properties [21]. Adiponectin also plays an important role in energy homeostasis by enhancing fatty acid oxidation in skeletal muscle and inhibiting glucose production in the liver [22].
AdipoRs topology is opposite to that of the classical GPCRs, with an internal N-terminus and an external C-terminus, enabling them to interact with G proteins [19]. Indeed, the AdipoRs bind to the adaptor protein containing a pleckstrin homology domain, phosphotyrosine binding domain and leucine zipper motif (APPL). APPL exhibits two isoforms, APPL1 and APPL2, which positively and negatively regulate adiponectin signaling, respectively [23]. In the absence of adiponectin stimulation, APPL2 binds to the N-terminus domain of AdipoRs or undergoes heterodimerization with APPL1, preventing the APPL1/AdipoRs from binding. On the other hand, upon adiponectin binding to AdipoRs, APPL1 dissociates from APPL2, leading to the activation of downstream signal transduction (Figure 1B) [19,24,25,26].
Multiple epidemiological evidence correlates low adiponectin level, characterizing obesity status, with an enhanced risk of cancer development, such as kidney, esophagus, prostate, thyroid, pancreatic, colorectal, breast cancer and many others [27].
In cancer cells, the activation of AdipoRs impacts cell proliferation, survival, apoptosis, migration and invasion, supporting tumor growth and progression [1,27].
Although AdipoRs and GPCRs enroll different intracellular protein effectors, their activation may trigger signaling pathways that mediate similar biological effects in tumor cells.
The objective of this review is to summarize the prevailing knowledge on atypical seven-transmembrane receptors, focusing on the role of AdipoRs in sustaining obesity-related breast cancer growth and progression.
2. Adiponectin Receptors and Adiponectin
Many reports validate the involvement of seven-transmembrane receptors in the pathogenesis of several human diseases, such as diabetes, obesity, cardiovascular dysfunctions and malignancies [1,28,29]. Emerging evidence underlines the important role of seven-transmembrane receptors and their signaling in tumor growth and development [2,25,30,31]. In fact, seven-transmembrane receptors control many features of tumor biology, including proliferation, invasion, survival, and metastasis. These receptors are not only expressed by cancer cells but also by different cell types in the tumor microenvironment, such as stromal, vascular, and immune cells [6,32,33].
As seven-transmembrane receptors play an important role in a wide variety of physiological and pathological processes, they are commonly targeted for medicinal therapeutics [25,28,34,35].
Adiponectin receptors, AdipoR1 and AdipoR2, cloned for the first time in 2003, are located at chromosome 1p36.13-q41 and 1 E4, and chromosome 12p13.31 and 6 F1, respectively. The analysis of cDNA encoded in these proteins showed that human and mouse AdipoR1 share 96.8% identity, whereas human and mouse AdipoR2 share 95.2% [36,37].
Similarly to other GPCRs, both AdipoR1 and AdipoR2 are integral membrane proteins, containing seven-transmembrane domains, an N-terminus and a C-terminus. However, their configuration is opposite to GPCRs, with an internal N-terminus and an external C-terminus. This makes AdipoRs unable to interact with G proteins [38,39] (Figure 1B). These peculiarities make adiponectin receptors structurally and functionally distinct from GPCRs [16,39].
Thus, even though AdipoRs and GPCRs families share the same seven-transmembrane domain structure, their sequence homology is weak [38].
In contrast to most of the GPCRs, knowledge of the conformational status of AdipoRs with respect to transmembrane signaling remains to be better defined [9,40].
Thus, structural details about AdipoRs could provide important information on the activation of specific signaling pathways, as well as for the development of specific agonists. For instance, Tanabe and coworkers demonstrated by crystallographic studies that AdipoR1 assumes a dual conformation, consisting of the closed and open forms, related to the signaling mechanisms [40].
AdipoRs mediate the pleiotropic actions of adiponectin, the most abundant fat-derived hormone, whose reduction plays a pivotal role in obesity-linked diseases, including insulin resistance/type 2 diabetes, atherosclerosis and malignancies [21,37].
The gene encoding adiponectin is expressed mainly in adipocytes and is regulated by different transcriptional factors, such as C/EBPs [41], sterol regulatory element binding to protein 1c (SREBP1c) [42] and PPARγ [43]. This adipokine consists of a C-terminal globular domain and an N-terminal collagen-like domain, which makes it structurally similar to complement 1q, belonging to a protein family that forms characteristic multimers [22]. Once synthesized, adiponectin is secreted into the bloodstream in different isoforms: a low-molecular-weight (LMW) trimer, a middle-molecular-weight (MMW) hexamer and a high-molecular-weight (HMW) 12- to 18-mer [37,44,45]. Moreover, a globular form, derived from full-length adiponectin by proteolytic cleavage, has been detected in plasma [37].
The interaction between ligand and receptor takes place through the globular domain of adiponectin and the extracellular portion of the receptors [46]. AdipoRs are unique and ubiquitously expressed, even though they differ from each other, both for the predominance of tissue localization and for their affinity to adiponectin isoforms [7]. In particular, AdipoR1 shows a higher affinity for globular adiponectin and is predominant in the skeletal muscle [7]. In contrast, AdipoR2 exhibits a higher affinity for full-length adiponectin and is most abundant in the liver [19,27]. AdipoRs contain a zinc-binding catalytic site coordinated by three histamine residues located near the inner surface of the plasma membrane [38]. The binding of adiponectin to the zinc-binding motif of AdipoRs triggers the activation of downstream signaling pathways in several target tissues, including skeletal muscle, liver, heart, kidney, and pancreas [47].
The signal transduction is mainly regulated by the adaptor protein APPL1, which interacts with the intracellular region of AdipoRs [48].
The engagement of AdipoRs by APPL1 increases the phosphorylation of adenosine monophosphate protein kinase (AMPK) and p38 mitogen-activated protein kinase (p38 MAPK), and enhances peroxisome proliferator-activated receptor alpha (PPARα) ligand activity, thereby inducing increased fatty acid oxidation and glucose consumption [36]. In particular, AMPK activation also depends on the different mechanisms, such as LKB1 cytosolic localization, driven by APPL1, or phospholipase C/Ca2+/Ca2+/calmodulin-dependent protein kinase kinase-dependent pathway, triggering an increased release of Ca2+ from the intracellular stores [1]. In addition, AdipoRs possess intrinsic ceramidase activity, which leads to a decrease in intracellular ceramide, a sphingolipid involved in insulin resistance, atherosclerosis, inflammation, and cell death (Figure 2B) [47]. The adiponectin/AdipoRs complex also stimulates fatty acid oxidation in skeletal muscle, inhibits glucose production in the liver, prompting an improvement in energy homeostasis, and mediates anti-inflammatory and anti-apoptotic actions in different cell types [46].
Obesity not only leads to a decrease in plasma adiponectin levels but also to an altered AdipoRs expression, modulating adiponectin sensitivity and causing insulin resistance with consequent hyperinsulinemia [49]. Moreover, a decreased expression of AdipoRs in the skeletal muscle of type 2 diabetic patients has also been found [50]. Therefore, further studies are needed for a deeper understanding of the role of the adiponectin/AdipoRs complex in obesity-associated diseases and in the identification of molecular targets to be used for therapeutic purposes.
Signaling Pathways Activated by Seven-Transmembrane Receptors in Breast Cancer
Despite AdipoRs and GPCRs specifically exploiting different transduction proteins to initiate signaling cascades, they activate downstream signaling involved in different proliferative and pro-inflammatory cytokine actions.
Chronic inflammatory status, supported by cytokines, is an important aspect of the pathogenesis of obesity-related breast cancer [51]. It is largely known that obese individuals are affected by more aggressive breast tumors, with an increased risk of recurrence and higher mortality [52]. Indeed, in an obese adipose tissue, the imbalance of cytokine production creates a chronic inflammatory microenvironment that promotes breast tumor cell motility, invasion, and epithelial-mesenchymal transition, enhancing the tumor cells’ metastatic potential [7,52,53,54]. Moreover, in this scenario, different cytokines may act through autocrine and paracrine mechanisms to modulate the activation of specific receptors, influencing breast tumor behavior.
In recent decades, growing evidence has highlighted the critical role of adiponectin/AdipoRs signaling as one of the main prediction markers of breast tumor clinical outcomes in obese patients. Indeed, in obesity-related breast cancer, the activation of AdipoRs affects cell proliferation, survival, apoptosis, migration and invasion, endosomal trafficking, metabolism and chromatin remodeling [7,48,55,56,57,58].
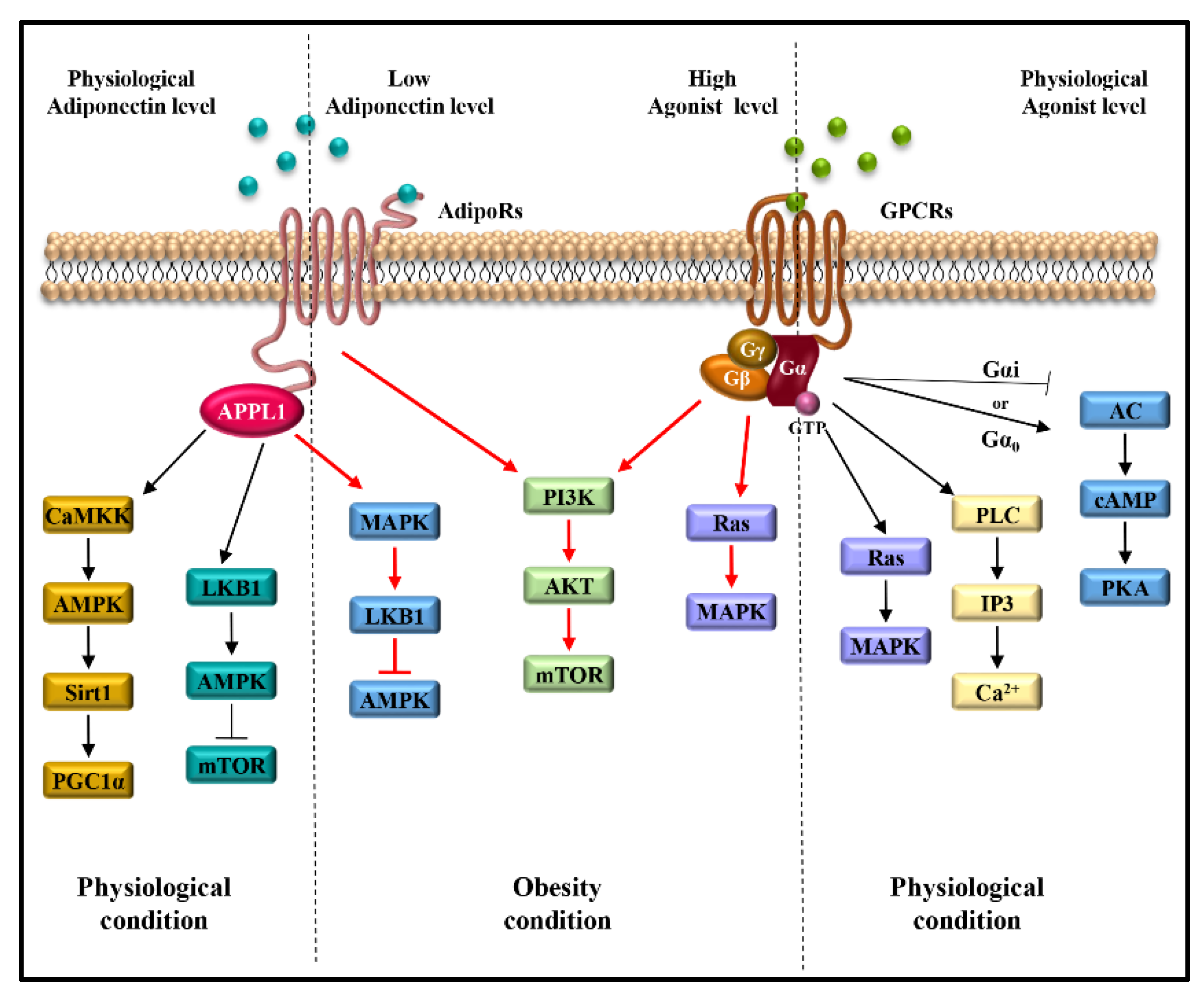
The main transduction pathways activated by adiponectin/AdipoRs/APPL1 axis include JAK/STAT, LKB1/AMPK/mTOR, AMPK/Sirt1/PGC1α, NF-kB, JNK and MAPK, sustaining breast tumor development and progression through the induction of cell proliferation and survival, cell cycle progression and anti-apoptotic response [18,59] (Figure 2B). Moreover, the stimulation of AdipoR1 and AdipoR2 induced by adiponectin leads to the activation of AMPK and PPARs, respectively, involved in the regulation of inflammatory responses, and lipid and glucose metabolism [60]. In the context of TME, the stimulation of AdipoR1 may potentiate the effect of pro-inflammatory cytokines. For example, in the TME, the high levels of CXCL12, which binds its seven-transmembrane receptor, CXCR4, and the low levels of adiponectin may concomitantly contribute to mediating cancer cell growth and progression [61,62,63,64]. Indeed, the adiponectin/AdipoR1 complex and the CXCL12/CXCR4 axis trigger the activation of several transduction signalings—Ras/MAPK, PI3K/AKT/mTOR, JNK/p38MAPK, Jak/STAT, NF-kB, PLC and MAPK—involved in cell survival, cell proliferation and migration [65,66,67,68,69]. Moreover, the inhibition of adenylate cyclase and consequent cAMP formation, induced by the CXCL12/CXCR4 complex, favors cancer cell proliferation [70,71]. Thus, the activation of all these mentioned pathways may create a suitable TME more prone to sustain breast cancer growth and progression (Figure 3).
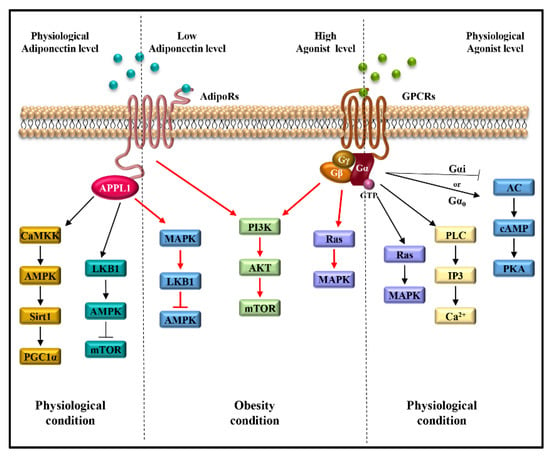
Figure 3.
Signaling pathways activated by atypical (AdipoRs) and classical (GPCRs) seven-transmembrane receptors in obesity-related breast cancer. Red lines indicate the shared signaling activated by low concentrations of adiponectin and by high levels of GPCRs agonists, which characterize obesity status, and favor breast cancer growth and progression.
3. Breast Cancer and Obesity
Breast cancer consists of a heterogeneous group of malignant tumors that are different in morphology, gene expression patterns, tumor behavior, clinical management, and prognosis [72].
The traditional, histological, pathological staging and tumor differentiation classifications provide diagnostic and prognostic information that is still important but not sufficient to satisfy the need for personalized therapies [72].
Indeed, tumors that at first glance are similar in morphology and stage actually may have a variable prognosis and therapeutic responses.
Thus, due to breast cancer’s biological and clinical complexity, a more accurate tumor classification is critical for individualized patient care [72].
According to the current molecular classification, breast cancers are grouped as follows: luminal A: ER+ and/or PR+, HER2- and low Ki-67 (<14%); luminal B: ER+ and/or PR+, HER2+ or HER2- and high Ki-67 (>14%); HER2+: ER-, PR- and HER2+; and basal-like (BLBC): ER-, PR-, HER2- (triple negative), plus CK 5/6+ and/or EGFR+ [72]. More recently, PD-L1 has been recognized as a predictive marker for the potential response to immunotherapy in the treatment of positive advanced/metastatic triple-negative breast cancer [73].
In addition, to better understand the heterogeneous nature of breast cancer, close attention is being paid to protective and negative factors, which may affect the development and progression of the disease.
Obesity/overweight is the main lifestyle-related risk factor associated with increased comorbidities and premature deaths [2,74].
A meta-analysis of 82 follow-up studies showed a 35% higher risk of breast cancer mortality in obese women than in their normal-weight counterparts, ascribing obesity to a relevant role as a risk factor for breast tumorigenesis [75].
The mechanisms connecting obesity with cellular pathways contributing to breast tumorigenesis include the hypertrophic and hyperplastic status of adipocytes, which entail an altered adipokine, cytokine and hormone production and secretion, as well as a modulation of their own receptor expression [4]. Moreover, the dysfunctional adipocytes engulf vascular stroma, sustaining local hypoxia that leads to the recruitment of inflammatory cells.
Many reports have addressed the critical role of these factors in sustaining low-grade chronic inflammation and a favorable tumor microenvironment in the development and progression of several obesity-related neoplasias [2,24,70]. In addition, adipose tissue, which acts as a source of estrogens, may contribute to obesity-associated hormone-responsive cancer growth. Moreover, the reduction in adiponectin levels in obese subjects has drawn particular attention for its paracrine and autocrine influence on the mammary epithelium.
Growing evidence correlates low adiponectin levels with a more aggressive breast cancer phenotype, characterized by a higher histological grade, large tumor size, lymph node metastasis and increased mortality [4,22,76]. In this scenario, the action of adiponectin on the tumor microenvironment occurs in the context of altered metabolic homeostasis due to adipocytes dysfunction and the activation of epigenetic pathways [70].
The correlation between tumor growth/progression and low adiponectin levels mainly depends on the disruption of adiponectin signaling mediated through AMPK, which normally inhibits proliferative pathways in non-tumoral cells [11].
Interestingly, in obesity, low levels of adiponectin are associated with altered levels of AdipoRs both in breast cancer cells and tumor microenvironment cell components.
Thus, these findings, due to the close interaction between mammary epithelial cells and adipose tissue, address how any imbalance in the hormonal milieu affects not only the initiation and progression of breast cancer but could also affect its response to therapies [76].
3.1. Adiponectin Receptors in Obesity-Related Breast Cancer
Many studies demonstrated that adiponectin and its receptors are expressed both in normal and neoplastic breast epithelial cells. However, conflicting data showed no differences between AdipoR1 and AdipoR2 expression between normal breast epithelial cells and breast cancer cells [77]. Thus, further studies are needed to clarify this issue.
Interestingly, adiponectin receptors are known to be differently expressed in breast cancer cell lines independent of ERα status, with a higher expression of AdipoR1 in MCF-7, T47D and MDA-MB-231 cells, and a higher level of AdipoR2 in MDA-MB-361 cells. Although AdipoRs expression was markedly increased in breast tumor tissues compared with control tissues [78], it has been shown that the high expression of AdipoR2 in breast cancer tissue is significantly and positively associated with vascular and lymph vascular invasion [27].
Reports have revealed that AdipoRs in breast cancer cell lines are also modulated by adiponectin [19,24,27], the levels of which were significantly downregulated in cancer patients compared to control subjects. Particularly, the low adiponectin serum levels upregulate AdipoR1 expression in breast cancer tissue as a kind of feedback loop [79].
Interestingly, the downregulation of AdipoR1 expression was observed in the epithelial cells of pre-invasive ductal carcinoma in situ (DCIS) compared to cells of adjacent invasive breast cancer [79,80]. These findings suggested that the loss of AdipoR1 favors the progression of the pre-invasive lesion [79]. On the other hand, it has been shown that decreased adiponectin serum levels and increased AdipoR1 and AdipoR2 expression occur in response to the development of diet-induced obesity in mice [81]. However, it is worth mentioning that another event increasing the incidence of breast cancer may come from ADIPOQ and/or ADIPOR1 single nucleotide polymorphisms [82].
Convincing evidence has demonstrated that low adiponectin concentrations affect breast cancer cell growth both in vitro and in vivo. Specifically, low adiponectin levels are induced by the formation of a multiprotein complex, where AdipoR1 cross-talks with ERα, c-Src and IGF-1R, converging with MAPK activation and increasing cell proliferation [58]. These results suggest a pivotal role of ERα in the adiponectin response. Indeed, in ERα-positive breast cancer cells, it has been demonstrated that among the mechanisms through which low adiponectin levels stimulate cell proliferation, there is the upregulation of Cyclin D1 and the inhibition of LKB1/AMPK/mTOR signaling [57,83]. On the contrary, in ERα-negative breast cancer cells, the activation of anti-proliferative and pro-apoptotic signaling pathways has been observed [57,83,84,85,86,87]. Although adiponectin has been conventionally considered an anticancer agent, all these data ascribe an ERα signaling amplifier role to this adipokine, favoring breast tumor growth and progression.
3.2. Adiponectin Receptors in Breast Tumor Microenvironment
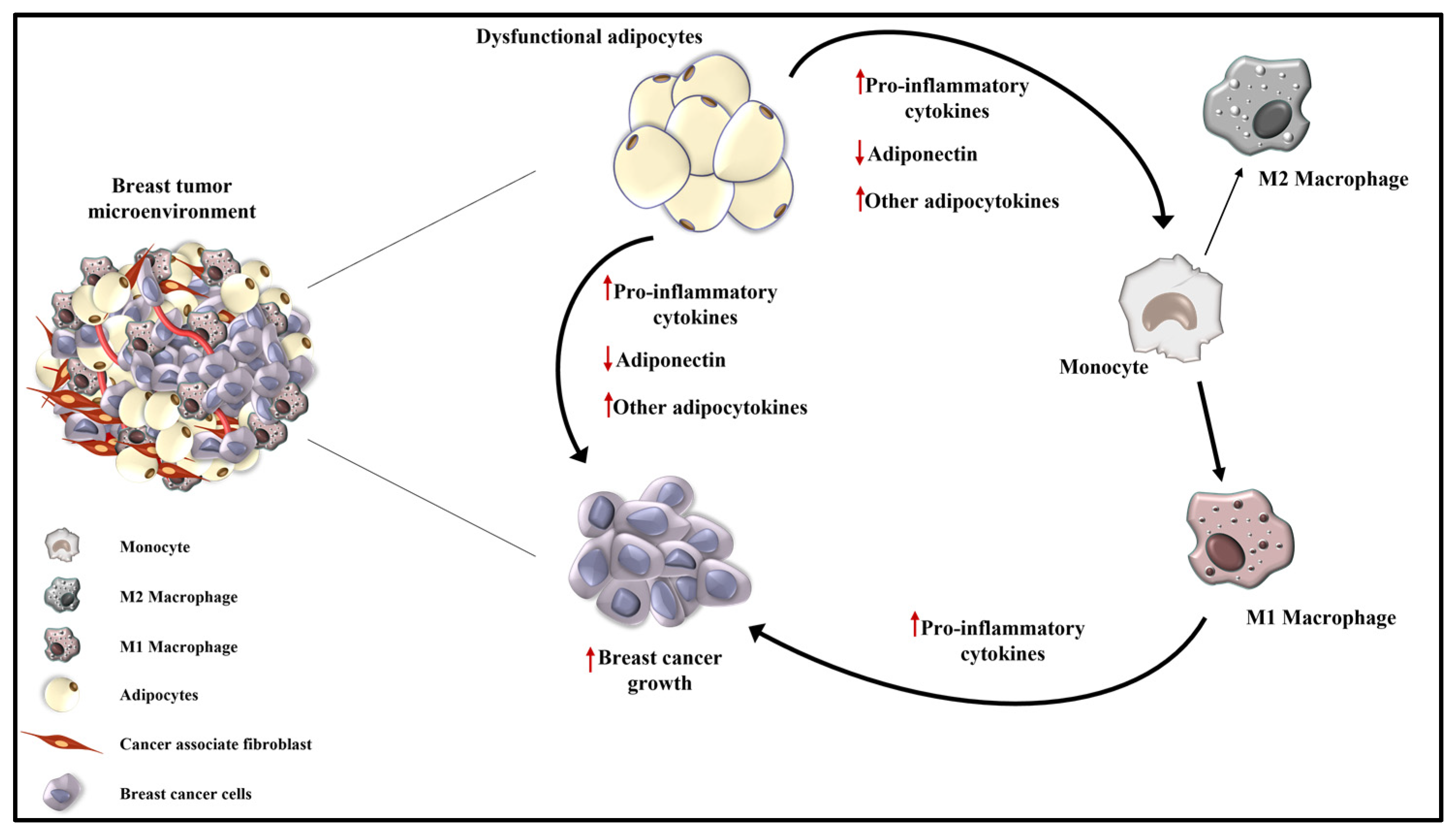
It is widely recognized that tumor development and progression depend on epithelial tissue and the surrounding microenvironment. TME is a highly complex and heterogeneous intricate network, mainly composed of tumor cells, immune and inflammatory cells, cancer-associated fibroblasts and adipocytes, embedded with different factors, such as cytokines, chemokines, growth factors, soluble receptors and exosomes (Figure 4) [6].
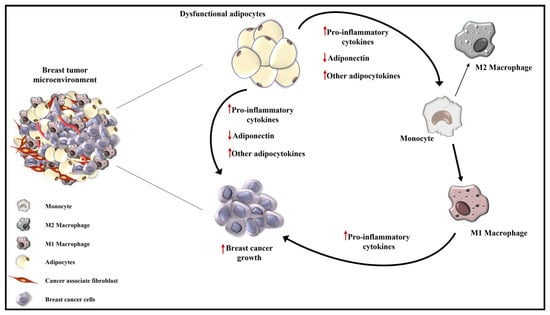
Figure 4.
Schematic representation of tumor microenvironment in obesity-related breast cancer. Obesity is characterized by a low-grade inflammatory status sustained by an altered adipocytokines secretion by dysfunctional adipocytes and by an increased infiltration of M1 polarized macrophages that release pro-inflammatory cytokines. All this creates a suitable TME more prone to sustain breast cancer growth and progression. ↑ indicates the increased production of pro-inflammatory cytokines and adipocytokines; ↓ represents the reduced level of adiponectin.
Recent evidence has highlighted the existence of a dynamic interaction between breast cancer and the cellular components of TME that drives remodeling of extracellular matrix, neovascularization and epithelial cell transition to stem cells [88]. Thus, by reshaping the TME through the identification of specific molecular targets may represent a cancer treatment strategy.
Adipocytes are considered to be essential components of breast cancer TME [6,89]. In vitro, in vivo and clinical data demonstrate that breast tumor cells significantly affect the surrounding adipocytes, whose derived factors change during tumor progression [6].
Growing evidence has demonstrated that breast cancer progression may be promoted by the interplay of invasive cancer cells with peritumoral adipocytes, referred to as cancer-associated adipocytes (CAAs) [90]. Indeed, breast cancer cells induce the differentiation of normal adipocytes into CAAs, which, compared to mature adipocytes, have smaller cell sizes, irregular shapes, and smaller lipid droplets [91,92]. Furthermore, CAAs are characterized by decreased differentiation markers of mature adipocytes and a more aggressive secretome depending on the overexpression of inflammatory cytokines and proteases, aberrant secretion of adipokines (such as leptin and adiponectin), and promotion of metabolic reprogramming of breast cancer cells [6,90,93].
The bidirectional interaction between breast cancer cells and CAAs triggers the shaping of the TME, leading to the acquisition of an invasive phenotype characterized by proliferation, angiogenesis, tumor dissemination, invasion and metastasis by a less physical barrier.
Notably, it has been demonstrated that there is a significant reduction in adiponectin levels in adipose tissue of breast cancer patients compared to normal breast adipose tissue. On the contrary, the mRNA expression levels of AdipoRs are significantly increased in CAAs [6].
In obesity, a reduction in the adiponectin level is related to DNA methylation of its promoter region in adipocytes. Moreover, low adiponectin expression, in turn, induces an increase in AdipoR2 gene methylation, as reported in humans and obese mice models, suppressing adiponectin binding and action. On the other hand, it has been reported that obesity does not affect AdipoR1 methylation [6,94].
Cicekdal MB and coworkers demonstrated a high methylation level of AdipoR1 in mammary tumor virus-transforming growth factor-α female mice that underwent a chronic energy restriction compared to ad libitum mice [95].
Overall, similarly to mammary adipocytes in obese patients, the reduction in adiponectin secretion by CAAs has been recognized as a crucial event in stimulating breast cancer progression [96].
Moreover, CAAs, once involved in a complex inflammatory process, contribute to the development of drug resistance, which is a crucial driver of tumor progression.
The establishment of chronic low-grade inflammation depends on the increased recruitment of macrophages, distinct in two subpopulations: the pro-inflammatory M1-polarized macrophages and the anti-inflammatory M2 type [97].
More evidence suggests that in lean subjects, the M2 macrophage type is prevalent, while in obese individuals, the M1 type predominates, addressing an M2 to M1 phenotypic switch in adipose tissue in obesity [98,99]. The prevalence of M1 macrophages in the breast cancer microenvironment is also sustained by the recruitment of additional circulating macrophages characterized by a high M1 gene expression that forms inflammatory loci called crown-like structures [1].
Recent data indicate that, through its receptors, different adiponectin isoforms induce isoform-specific responses in human monocytes. Particularly, LMW adiponectin reduces IL-6 release and stimulates IL-10 secretion in differentiated macrophages, favoring anti-inflammatory effects, while HMW increases the secretion of IL-6 in human monocytes, mediating pro-inflammatory responses [100].
Nevertheless, the regulation of AdipoRs expression in monocytes and macrophages deserves to be further explored.
Many studies have suggested the involvement of different hormones and macrophage polarization processes in the modulation of AdipoRs levels in monocytes and macrophages.
Particularly, reports reveal that AdipoRs expression is increased by nuclear hormone receptor ligands, such as LXR and PPAR [101].
Moreover, it has been observed that AdipoR1 is more expressed than AdipoR2 in monocytes, while its expression decreases in M1 macrophages, indicating an important role of macrophage differentiation and polarization in the regulation of the AdipoRs expression.
In M1 polarized macrophages, adiponectin restores AdipoR1 and AdipoR2 levels compared to those in nonpolarized macrophages, thus mediating an inflammatory response. On the contrary, M2 polarized macrophages exhibited higher AdipoRs expression than non-polarized macrophages, regardless of adiponectin stimulation [102].
These data provide important evidence of macrophage polarization in AdipoRs expression and adiponectin-mediated inflammatory response in the breast TME, which profoundly affects cancer progression.
4. Conclusions and Future Directions
As breast cancer is the most common cancer and the leading cause of mortality in women worldwide, there is a compelling need to design new therapeutic strategies for the personalized treatment of this malignancy. Tumor initiation and development are affected by the cross-talk between epithelial cells and the surrounding TME components [6]. The chronic low-grade inflammation that characterizes TME in obesity is sustained by immune cell infiltration and altered expression and secretion profiles of inflammatory mediators and their receptors in dysfunctional adipocytes [33]. In this scenario, the decreased adiponectin secreted levels and the dysregulated expression of AdipoRs in breast tumor cells and TME cell components are gaining increasing attention to understand the molecular mechanisms that drive the growth and progression of breast cancer, as well as to identify new therapeutic targets [79,81,101,102]. Particularly, the remodeling of TME is becoming a promising candidate for the development of innovative breast cancer treatment strategies. Indeed, several therapeutic approaches targeted to increase the assembly and secretion of adiponectin have been investigated [1,25]. However, due to adiponectin’s multimeric structure, short half-life and high serum concentrations, it is difficult to obtain benefits from adiponectin replacement therapy.
Thus, the design of AdipoRs agonists aimed at potentiating adiponectin action, making it able to antagonize pro-inflammatory signaling in the TME, represents an attractive therapeutic strategy.
Therefore, the current druggable options focus on the development of orally active small-molecule agonists for AdipoRs. Among these, the ADP355 molecule is the pioneer of the first class of AdipoR1 agonists that reproduces globular adiponectin anti-proliferative activity through the regulation of different signals, such as AMPK, Akt, STAT3 and ERK1/2 [103,104]. Three other peptides designed to mimic adiponectin actions are BHD1028, BHD43 and BHD44, which show a higher stability, solubility and affinity for AdipoR1 compared to ADP355 [105]. Currently, AdipoRon, an oral non-peptidic AdipoRs agonist, is extensively studied due to its adiponectin replacement therapy potentiality. AdipoRon’s main functions are through the activation of AMPK and PPARγ pathways in obesity-related disorders [106]. Despite these molecules being promising candidates in preclinical studies, the AdipoRs agonists listed in this study are currently unavailable for human clinical trials.
In conclusion, the design of innovative AdipoRs agonists and/or the refinement of those that are already available could extend the therapeutic strategies for the treatment of obesity-related breast cancer.
Author Contributions
L.M., G.D.N., A.P. and M.F. prepared the manuscript, collected the data, wrote, edited, and prepared the figures. L.M., S.A., M.L.P. and F.G. prepared, revised, and provided critical consideration for the manuscript, design, and editing. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the University of Calabria and MIUR Excellence Department Project funds (L.232/2016), awarded to the Department of Pharmacy, Health and Nutritional Sciences, University of Calabria, Italy. Associazione Italiana per la Ricerca sul Cancro (AIRC), Grant/Award Number: IG-18602. PON Salute ARS01_00568 SI.F.I.PA.CRO.DE.
Acknowledgments
Giuseppina Daniela Naimo was supported by the Italian Minister of University and Research (MIUR, D.M. 1062/2021) -PON “Ricerca e Innovazione” 2014–2020, Azione IV.4 “Dottorati e contratti di ricerca su tematiche dell’innovazione”.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Andò, S.; Naimo, G.D.; Gelsomino, L.; Catalano, S.; Mauro, L. Novel insights into adiponectin action in breast cancer: Evidence of its mechanistic effects mediated by ERα expression. Obes. Rev. 2020, 21, e13004. [Google Scholar]
- Barone, I.; Caruso, A.; Gelsomino, L.; Giordano, C.; Bonofiglio, D.; Catalano, S.; Andò, S. Obesity and endocrine therapy resistance in breast cancer: Mechanistic insights and perspectives. Obes. Rev. 2022, 23, e13358. [Google Scholar]
- Karra, P.; Winn, M.; Pauleck, S.; Bulsiewicz-Jacobsen, A.; Peterson, L.; Coletta, A.; Doherty, J.; Ulrich, C.M.; Summers, S.A.; Gunter, M. Metabolic dysfunction and obesity-related cancer: Beyond obesity and metabolic syndrome. Obesity 2022, 30, 1323–1334. [Google Scholar]
- Lohmann, A.E.; Soldera, S.V.; Pimentel, I.; Ribnikar, D.; Ennis, M.; Amir, E.; Goodwin, P.J. Association of obesity with breast cancer outcome in relation to cancer subtypes: A meta-analysis. JNCI J. Natl. Cancer Inst. 2021, 113, 1465–1475. [Google Scholar]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.; Torre, L.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA-A Cancer J. Clin. 2020, 70, 313. [Google Scholar]
- Yao, H.; He, S. Multi-faceted role of cancer-associated adipocytes in the tumor microenvironment. Mol. Med. Rep. 2021, 24, 886. [Google Scholar]
- Mauro, L.; Naimo, G.D.; Ricchio, E.; Panno, M.L.; Andò, S. Cross-talk between adiponectin and IGF-IR in breast cancer. Front. Oncol. 2015, 5, 157. [Google Scholar]
- Ouchi, N.; Parker, J.L.; Lugus, J.J.; Walsh, K. Adipokines in inflammation and metabolic disease. Nat. Rev. Immunol. 2011, 11, 85–97. [Google Scholar]
- Rosenbaum, D.M.; Rasmussen, S.G.; Kobilka, B.K. The structure and function of G-protein-coupled receptors. Nature 2009, 459, 356–363. [Google Scholar]
- Tuteja, N. Signaling through G protein coupled receptors. Plant Signal. Behav. 2009, 4, 942–947. [Google Scholar]
- Zhao, J.; Deng, Y.; Jiang, Z.; Qing, H. G protein-coupled receptors (GPCRs) in Alzheimer’s disease: A focus on BACE1 related GPCRs. Front. Aging Neurosci. 2016, 8, 58. [Google Scholar]
- McCudden, C.; Hains, M.; Kimple, R.; Siderovski, D.; Willard, F. G-protein signaling: Back to the future. Cell Mol. Life Sci. 2005, 62, 551–577. [Google Scholar]
- Zhou, X.E.; Melcher, K.; Xu, H.E. Understanding the GPCR biased signaling through G protein and arrestin complex structures. Curr. Opin. Struct. Biol. 2017, 45, 150–159. [Google Scholar]
- Lokeshwar, B.L.; Kallifatidis, G.; Hoy, J.J. Atypical chemokine receptors in tumor cell growth and metastasis. In Advances in Cancer Research; Elsevier: Amsterdam, The Netherlands, 2020; Volume 145, pp. 1–27. [Google Scholar]
- Bonecchi, R.; Graham, G.J. Atypical chemokine receptors and their roles in the resolution of the inflammatory response. Front. Immunol. 2016, 7, 224. [Google Scholar]
- Vasiliauskaité-Brooks, I.; Healey, R.D.; Granier, S. 7TM proteins are not necessarily GPCRs. Mol. Cell. Endocrinol. 2019, 491, 110397. [Google Scholar]
- Avgerinos, K.I.; Spyrou, N.; Mantzoros, C.S.; Dalamaga, M. Obesity and cancer risk: Emerging biological mechanisms and perspectives. Metabolism 2019, 92, 121–135. [Google Scholar]
- Panno, M.L.; Naimo, G.D.; Spina, E.; Andò, S.; Mauro, L. Different molecular signaling sustaining adiponectin action in breast cancer. Curr. Opin. Pharmacol. 2016, 31, 1–7. [Google Scholar]
- Kadowaki, T.; Yamauchi, T. Adiponectin and adiponectin receptors. Endocr. Rev. 2005, 26, 439–451. [Google Scholar]
- Rasmussen, M.S.; Lihn, A.S.; Pedersen, S.B.; Bruun, J.M.; Rasmussen, M.; Richelsen, B. Adiponectin receptors in human adipose tissue: Effects of obesity, weight loss, and fat depots. Obesity 2006, 14, 28–35. [Google Scholar]
- Esmaili, S.; Hemmati, M.; Karamian, M. Physiological role of adiponectin in different tissues: A review. Arch. Physiol. Biochem. 2020, 126, 67–73. [Google Scholar]
- Gelsomino, L.; Naimo, G.D.; Catalano, S.; Mauro, L.; Andò, S. The emerging role of adiponectin in female malignancies. Int. J. Mol. Sci. 2019, 20, 2127. [Google Scholar]
- Wang, C.; Xin, X.; Xiang, R.; Ramos, F.J.; Liu, M.; Lee, H.J.; Chen, H.; Mao, X.; Kikani, C.K.; Liu, F. Yin-Yang regulation of adiponectin signaling by APPL isoforms in muscle cells. J. Biol. Chem. 2009, 284, 31608–31615. [Google Scholar]
- Dalamaga, M.; Diakopoulos, K.N.; Mantzoros, C.S. The role of adiponectin in cancer: A review of current evidence. Endocr. Rev. 2012, 33, 547–594. [Google Scholar]
- Naimo, G.D.; Gelsomino, L.; Catalano, S.; Mauro, L.; Andò, S. Interfering role of ERα on adiponectin action in breast cancer. Front. Endocrinol. 2020, 11, 66. [Google Scholar]
- Barbe, A.; Bongrani, A.; Mellouk, N.; Estienne, A.; Kurowska, P.; Grandhaye, J.; Elfassy, Y.; Levy, R.; Rak, A.; Froment, P. Mechanisms of adiponectin action in fertility: An overview from gametogenesis to gestation in humans and animal models in normal and pathological conditions. Int. J. Mol. Sci. 2019, 20, 1526. [Google Scholar]
- Parida, S.; Siddharth, S.; Sharma, D. Adiponectin, obesity, and cancer: Clash of the bigwigs in health and disease. Int. J. Mol. Sci. 2019, 20, 2519. [Google Scholar]
- Hauser, A.S.; Attwood, M.M.; Rask-Andersen, M.; Schiöth, H.B.; Gloriam, D.E. Trends in GPCR drug discovery: New agents, targets and indications. Nat. Rev. Drug Discov. 2017, 16, 829–842. [Google Scholar]
- Schöneberg, T.; Schulz, A.; Biebermann, H.; Hermsdorf, T.; Römpler, H.; Sangkuhl, K. Mutant G-protein-coupled receptors as a cause of human diseases. Pharmacol. Ther. 2004, 104, 173–206. [Google Scholar]
- Talia, M.; De Francesco, E.M.; Rigiracciolo, D.C.; Muoio, M.G.; Muglia, L.; Belfiore, A.; Maggiolini, M.; Sims, A.H.; Lappano, R. The G Protein-Coupled Estrogen Receptor (GPER) Expression Correlates with Pro-Metastatic Pathways in ER-Negative Breast Cancer: A Bioinformatics Analysis. Cells 2020, 9, 622. [Google Scholar]
- Treeck, O.; Schüler-Toprak, S.; Ortmann, O. Estrogen Actions in Triple-Negative Breast Cancer. Cells 2020, 9, 2358. [Google Scholar]
- Insel, P.A.; Sriram, K.; Wiley, S.Z.; Wilderman, A.; Katakia, T.; McCann, T.; Yokouchi, H.; Zhang, L.; Corriden, R.; Liu, D. GPCRomics: GPCR expression in cancer cells and tumors identifies new, potential biomarkers and therapeutic targets. Front. Pharmacol. 2018, 9, 431. [Google Scholar]
- Artemniak-Wojtowicz, D.; Kucharska, A.; Pyrżak, B. Obesity and chronic inflammation crosslinking. Cent. Eur. J. Immunol. 2020, 45, 461–468. [Google Scholar]
- Cosín-Roger, J.; Ortiz-Masia, D.; Barrachina, M.D.; Calatayud, S. Metabolite Sensing GPCRs: Promising Therapeutic Targets for Cancer Treatment? Cells 2020, 9, 2345. [Google Scholar]
- Luo, J.; Yu, F.-X. GPCR-hippo signaling in cancer. Cells 2019, 8, 426. [Google Scholar]
- Yamauchi, T.; Kamon, J.; Ito, Y.; Tsuchida, A.; Yokomizo, T.; Kita, S.; Sugiyama, T.; Miyagishi, M.; Hara, K.; Tsunoda, M. Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature 2003, 423, 762–769. [Google Scholar]
- Yamauchi, T.; Iwabu, M.; Okada-Iwabu, M.; Kadowaki, T. Adiponectin receptors: A review of their structure, function and how they work. Best Pract. Res. Clin. Endocrinol. Metab. 2014, 28, 15–23. [Google Scholar]
- Tanabe, H.; Fujii, Y.; Okada-Iwabu, M.; Iwabu, M.; Nakamura, Y.; Hosaka, T.; Motoyama, K.; Ikeda, M.; Wakiyama, M.; Terada, T. Crystal structures of the human adiponectin receptors. Nature 2015, 520, 312–316. [Google Scholar]
- Iwabu, M.; Okada-Iwabu, M.; Yamauchi, T.; Kadowaki, T. Adiponectin/adiponectin receptor in disease and aging. NPJ Aging Mech. Dis. 2015, 1, 15013. [Google Scholar]
- Tanabe, H.; Fujii, Y.; Okada-Iwabu, M.; Iwabu, M.; Kano, K.; Kawana, H.; Hato, M.; Nakamura, Y.; Terada, T.; Kimura-Someya, T. Human adiponectin receptor AdipoR1 assumes closed and open structures. Commun. Biol. 2020, 3, 446. [Google Scholar]
- Shehzad, A.; Iqbal, W.; Shehzad, O.; Lee, Y.S. Adiponectin: Regulation of its production and its role in human diseases. Hormones 2012, 11, 8–20. [Google Scholar]
- Seo, J.B.; Moon, H.M.; Noh, M.J.; Lee, Y.S.; Jeong, H.W.; Yoo, E.J.; Kim, W.S.; Park, J.; Youn, B.-S.; Kim, J.W. Adipocyte determination-and differentiation-dependent factor 1/sterol regulatory element-binding protein 1c regulates mouse adiponectin expression. J. Biol. Chem. 2004, 279, 22108–22117. [Google Scholar]
- Maeda, N.; Takahashi, M.; Funahashi, T.; Kihara, S.; Nishizawa, H.; Kishida, K.; Nagaretani, H.; Matsuda, M.; Komuro, R.; Ouchi, N. PPARγ ligands increase expression and plasma concentrations of adiponectin, an adipose-derived protein. Diabetes 2001, 50, 2094–2099. [Google Scholar]
- Pajvani, U.B.; Du, X.; Combs, T.P.; Berg, A.H.; Rajala, M.W.; Schulthess, T.; Engel, J.; Brownlee, M.; Scherer, P.E. Structure-function studies of the adipocyte-secreted hormone Acrp30/adiponectin implications for metabolic regulation and bioactivity. J. Biol. Chem. 2003, 278, 9073–9085. [Google Scholar]
- Waki, H.; Yamauchi, T.; Kamon, J.; Ito, Y.; Uchida, S.; Kita, S.; Hara, K.; Hada, Y.; Vasseur, F.; Froguel, P. Impaired multimerization of human adiponectin mutants associated with diabetes molecular structure and multimer formation of adiponectin. J. Biol. Chem. 2003, 278, 40352–40363. [Google Scholar]
- Fang, H.; Judd, R.L. Adiponectin regulation and function. Compr. Physiol. 2011, 8, 1031–1063. [Google Scholar]
- Holland, W.L.; Miller, R.A.; Wang, Z.V.; Sun, K.; Barth, B.M.; Bui, H.H.; Davis, K.E.; Bikman, B.T.; Halberg, N.; Rutkowski, J.M. Receptor-mediated activation of ceramidase activity initiates the pleiotropic actions of adiponectin. Nat. Med. 2011, 17, 55–63. [Google Scholar]
- Deepa, S.S.; Dong, L.Q. APPL1: Role in adiponectin signaling and beyond. Am. J. Physiol. -Endocrinol. Metab. 2009, 296, E22–E36. [Google Scholar]
- Tsuchida, A.; Yamauchi, T.; Ito, Y.; Hada, Y.; Maki, T.; Takekawa, S.; Kamon, J.; Kobayashi, M.; Suzuki, R.; Hara, K. Insulin/Foxo1 pathway regulates expression levels of adiponectin receptors and adiponectin sensitivity. J. Biol. Chem. 2004, 279, 30817–30822. [Google Scholar]
- Iwabu, M.; Okada-Iwabu, M.; Yamauchi, T.; Kadowaki, T. Adiponectin/AdipoR research and its implications for lifestyle-related diseases. Front. Cardiovasc. Med. 2019, 6, 116. [Google Scholar]
- Landskron, G.; De la Fuente, M.; Thuwajit, P.; Thuwajit, C.; Hermoso, M.A. Chronic inflammation and cytokines in the tumor microenvironment. J. Immunol. Res. 2014, 2014, 149185. [Google Scholar]
- Zielińska, K.; Kwasniak, K.; Tabarkiewicz, J.; Karczmarek-Borowska, B. The role of pro-inflammatory cytokines in the pathogenesis and progression of neoplasms. Adv. Hyg. Exp. Med./Postep. Hig. I Med. Dosw. 2018, 72, 896–905. [Google Scholar]
- Amin, M.N.; Siddiqui, S.A.; Ibrahim, M.; Hakim, M.L.; Ahammed, M.S.; Kabir, A.; Sultana, F. Inflammatory cytokines in the pathogenesis of cardiovascular disease and cancer. SAGE Open Med. 2020, 8, 2050312120965752. [Google Scholar]
- Chu, D.-T.; Nguyen Thi Phuong, T.; Tien, N.L.B.; Tran, D.-K.; Nguyen, T.-T.; Thanh, V.V.; Luu Quang, T.; Minh, L.B.; Pham, V.H.; Ngoc, V.T.N. The effects of adipocytes on the regulation of breast cancer in the tumor microenvironment: An update. Cells 2019, 8, 857. [Google Scholar]
- Jia, Z.; Liu, Y.; Cui, S. Adiponectin induces breast cancer cell migration and growth factor expression. Cell Biochem. Biophys. 2014, 70, 1239–1245. [Google Scholar]
- Jeong, Y.-J.; Bong, J.-G.; Park, S.-H.; Choi, J.-H.; Oh, H.-K. Expression of leptin, leptin receptor, adiponectin, and adiponectin receptor in ductal carcinoma in situ and invasive breast cancer. J. Breast Cancer 2011, 14, 96–103. [Google Scholar]
- Mauro, L.; Naimo, G.D.; Gelsomino, L.; Malivindi, R.; Bruno, L.; Pellegrino, M.; Tarallo, R.; Memoli, D.; Weisz, A.; Panno, M.L. Uncoupling effects of estrogen receptor α on LKB1/AMPK interaction upon adiponectin exposure in breast cancer. FASEB J. 2018, 32, 4343–4355. [Google Scholar]
- Mauro, L.; Pellegrino, M.; De Amicis, F.; Ricchio, E.; Giordano, F.; Rizza, P.; Catalano, S.; Bonofiglio, D.; Sisci, D.; Panno, M.L. Evidences that estrogen receptor α interferes with adiponectin effects on breast cancer cell growth. Cell Cycle 2014, 13, 553–564. [Google Scholar]
- Kelesidis, I.; Kelesidis, T.; Mantzoros, C. Adiponectin and cancer: A systematic review. Br. J. Cancer 2006, 94, 1221–1225. [Google Scholar]
- Pham, D.-V.; Nguyen, T.-K.; Park, P.-H. Adipokines at the crossroads of obesity and mesenchymal stem cell therapy. Exp. Mol. Med. 2023, 55, 313–324. [Google Scholar]
- Adams, V.; Heiker, J.T.; Höllriegel, R.; Beck, E.B.; Woitek, F.J.; Erbs, S.; Blüher, M.; Stumvoll, M.; Beck-Sickinger, A.G.; Schuler, G. Adiponectin promotes the migration of circulating angiogenic cells through p38-mediated induction of the CXCR4 receptor. Int. J. Cardiol. 2013, 167, 2039–2046. [Google Scholar]
- Shibata, R.; Skurk, C.; Ouchi, N.; Galasso, G.; Kondo, K.; Ohashi, T.; Shimano, M.; Kihara, S.; Murohara, T.; Walsh, K. Adiponectin promotes endothelial progenitor cell number and function. FEBS Lett. 2008, 582, 1607–1612. [Google Scholar]
- Nakamura, N.; Naruse, K.; Matsuki, T.; Hamada, Y.; Nakashima, E.; Kamiya, H.; Matsubara, T.; Enomoto, A.; Takahashi, M.; Oiso, Y. Adiponectin promotes migration activities of endothelial progenitor cells via Cdc42/Rac1. FEBS Lett. 2009, 583, 2457–2463. [Google Scholar]
- Chen, L.; Wu, F.; Xia, W.-H.; Zhang, Y.-Y.; Xu, S.-Y.; Cheng, F.; Liu, X.; Zhang, X.-Y.; Wang, S.-M.; Tao, J. CXCR4 gene transfer contributes to in vivo reendothelialization capacity of endothelial progenitor cells. Cardiovasc. Res. 2010, 88, 462–470. [Google Scholar]
- Yin, X.; Liu, Z.; Zhu, P.; Wang, Y.; Ren, Q.; Chen, H.; Xu, J. CXCL12/CXCR4 promotes proliferation, migration, and invasion of adamantinomatous craniopharyngiomas via PI3K/AKT signal pathway. J. Cell. Biochem. 2019, 120, 9724–9736. [Google Scholar]
- Tian, Y.; Yin, H.; Deng, X.; Tang, B.; Ren, X.; Jiang, T. CXCL12 induces migration of oligodendrocyte precursor cells through the CXCR4-activated MEK/ERK and PI3K/AKT pathways. Mol. Med. Rep. 2018, 18, 4374–4380. [Google Scholar]
- Jiménez, C.; Portela, R.A.; Mellado, M.; Rodríguez-Frade, J.M.; Collard, J.; Serrano, A.; Martínez-a, C.; Avila, J.; Carrera, A.C. Role of the PI3K regulatory subunit in the control of actin organization and cell migration. J. Cell Biol. 2000, 151, 249–262. [Google Scholar]
- Teixido, J.; Martinez-Moreno, M.; Diaz-Martinez, M.; Sevilla-Movilla, S. The good and bad faces of the CXCR4 chemokine receptor. Int. J. Biochem. Cell Biol. 2018, 95, 121–131. [Google Scholar]
- Döring, Y.; Pawig, L.; Weber, C.; Noels, H. The CXCL12/CXCR4 chemokine ligand/receptor axis in cardiovascular disease. Front. Physiol. 2014, 5, 212. [Google Scholar]
- Mellado, M.; Rodríguez-Frade, J.M.; Mañes, S.; Martínez-A, C. Chemokine signaling and functional responses: The role of receptor dimerization and TK pathway activation. Annu. Rev. Immunol. 2001, 19, 397–421. [Google Scholar]
- Domanska, U.M.; Kruizinga, R.C.; Nagengast, W.B.; Timmer-Bosscha, H.; Huls, G.; de Vries, E.G.; Walenkamp, A.M. A review on CXCR4/CXCL12 axis in oncology: No place to hide. Eur. J. Cancer 2013, 49, 219–230. [Google Scholar]
- Zhang, X. Molecular Classification of Breast Cancer: Relevance and Challenges. Arch. Pathol. Lab. Med. 2023, 147, 46–51. [Google Scholar]
- Muller, K.; Jorns, J.M.; Tozbikian, G. Journal of Pathology and Translational Medicine. J. Pathol. Transl. Med. 2022, 56, 170–171. [Google Scholar]
- Ryan, D.; Barquera, S.; Barata Cavalcanti, O.; Ralston, J. The global pandemic of overweight and obesity: Addressing a twenty-First century multifactorial disease. In Handbook of Global Health; Springer: Berlin/Heidelberg, Germany, 2021; pp. 739–773. [Google Scholar]
- Chan, D.; Vieira, A.; Aune, D.; Bandera, E.; Greenwood, D.; McTiernan, A.; Navarro Rosenblatt, D.; Thune, I.; Vieira, R.; Norat, T. Body mass index and survival in women with breast cancer—Systematic literature review and meta-analysis of 82 follow-up studies. Ann. Oncol. 2014, 25, 1901–1914. [Google Scholar]
- Surmacz, E. Leptin and adiponectin: Emerging therapeutic targets in breast cancer. J. Mammary Gland Biol. Neoplasia 2013, 18, 321–332. [Google Scholar]
- Takahata, C.; Miyoshi, Y.; Irahara, N.; Taguchi, T.; Tamaki, Y.; Noguchi, S. Demonstration of adiponectin receptors 1 and 2 mRNA expression in human breast cancer cells. Cancer Lett. 2007, 250, 229–236. [Google Scholar]
- Körner, A.; Pazaitou-Panayiotou, K.; Kelesidis, T.; Kelesidis, I.; Williams, C.J.; Kaprara, A.; Bullen, J.; Neuwirth, A.; Tseleni, S.; Mitsiades, N. Total and high-molecular-weight adiponectin in breast cancer: In vitro and in vivo studies. J. Clin. Endocrinol. Metab. 2007, 92, 1041–1048. [Google Scholar]
- Pfeiler, G.; Hudelist, G.; Wülfing, P.; Mattsson, B.; Königsberg, R.; Kubista, E.; Singer, C.F. Impact of AdipoR1 expression on breast cancer development. Gynecol. Oncol. 2010, 117, 134–138. [Google Scholar]
- Jardé, T.; Caldefie-Chézet, F.; Goncalves-Mendes, N.; Mishellany, F.; Buechler, C.; Penault-Llorca, F.; Vasson, M.-P. Involvement of adiponectin and leptin in breast cancer: Clinical and in vitro studies. Endocr. -Relat. Cancer 2009, 16, 1197–1210. [Google Scholar]
- Bullen, J.W., Jr.; Bluher, S.; Kelesidis, T.; Mantzoros, C.S. Regulation of adiponectin and its receptors in response to development of diet-induced obesity in mice. Am. J. Physiol. Endocrinol. Metab. 2007, 292, E1079–E1086. [Google Scholar]
- Kaklamani, V.G.; Sadim, M.; Hsi, A.; Offit, K.; Oddoux, C.; Ostrer, H.; Ahsan, H.; Pasche, B.; Mantzoros, C. Variants of the adiponectin and adiponectin receptor 1 genes and breast cancer risk. Cancer Res. 2008, 68, 3178–3184. [Google Scholar]
- Mauro, L.; Pellegrino, M.; Giordano, F.; Ricchio, E.; Rizza, P.; De Amicis, F.; Catalano, S.; Bonofiglio, D.; Panno, M.L.; Andò, S. Estrogen receptor-α drives adiponectin effects on cyclin D1 expression in breast cancer cells. FASEB J. 2015, 29, 2150–2160. [Google Scholar]
- Taliaferro-Smith, L.; Nagalingam, A.; Knight, B.B.; Oberlick, E.; Saxena, N.K.; Sharma, D. Integral role of PTP1B in adiponectin-mediated inhibition of oncogenic actions of leptin in breast carcinogenesis. Neoplasia 2013, 15, 23-IN11. [Google Scholar]
- Wang, Y.; Lam, J.B.; Lam, K.S.; Liu, J.; Lam, M.C.; Hoo, R.L.; Wu, D.; Cooper, G.J.; Xu, A. Adiponectin modulates the glycogen synthase kinase-3β/β-catenin signaling pathway and attenuates mammary tumorigenesis of MDA-MB-231 cells in nude mice. Cancer Res. 2006, 66, 11462–11470. [Google Scholar]
- Nakayama, S.; Miyoshi, Y.; Ishihara, H.; Noguchi, S. Growth-inhibitory effect of adiponectin via adiponectin receptor 1 on human breast cancer cells through inhibition of S-phase entry without inducing apoptosis. Breast Cancer Res. Treat. 2008, 112, 405–410. [Google Scholar]
- Dos Santos, E.; Benaitreau, D.; Dieudonne, M.-N.; Leneveu, M.-C.; Serazin, V.; Giudicelli, Y.; Pecquery, R. Adiponectin mediates an antiproliferative response in human MDA-MB 231 breast cancer cells. Oncol. Rep. 2008, 20, 971–977. [Google Scholar]
- Baghban, R.; Roshangar, L.; Jahanban-Esfahlan, R.; Seidi, K.; Ebrahimi-Kalan, A.; Jaymand, M.; Kolahian, S.; Javaheri, T.; Zare, P. Tumor microenvironment complexity and therapeutic implications at a glance. Cell Commun. Signal. 2020, 18, 59. [Google Scholar]
- Lazar, I.; Clement, E.; Attane, C.; Muller, C.; Nieto, L. A new role for extracellular vesicles: How small vesicles can feed tumors’ big appetite. J. Lipid Res. 2018, 59, 1793–1804. [Google Scholar]
- Wu, Q.; Li, B.; Li, Z.; Li, J.; Sun, S.; Sun, S. Cancer-associated adipocytes: Key players in breast cancer progression. J. Hematol. Oncol. 2019, 12, 95. [Google Scholar]
- Dirat, B.; Bochet, L.; Dabek, M.; Daviaud, D.; Dauvillier, S.; Majed, B.; Wang, Y.Y.; Meulle, A.; Salles, B.; Le Gonidec, S. Cancer-associated adipocytes exhibit an activated phenotype and contribute to breast cancer invasion. Cancer Res. 2011, 71, 2455–2465. [Google Scholar]
- Suárez–Nájera, L.E.; Chanona–Pérez, J.J.; Valdivia–Flores, A.; Marrero–Rodríguez, D.; Salcedo–Vargas, M.; García–Ruiz, D.I.; Castro–Reyes, M.A. Morphometric study of adipocytes on breast cancer by means of photonic microscopy and image analysis. Microsc. Res. Tech. 2018, 81, 240–249. [Google Scholar]
- Crake, R.L.I.; Phillips, E.; Kleffmann, T.; Currie, M.J. Co-culture with human breast adipocytes differentially regulates protein abundance in breast Cancer cells. Cancer Genom. Proteom. 2019, 16, 319–332. [Google Scholar]
- Engin, A. Adiponectin-resistance in obesity. Obes. Lipotoxic. 2017, 960, 415–441. [Google Scholar]
- Cicekdal, M.B.; Kazan, B.; Tuna, B.; Ozorhan, U.; Ekici, I.; Zhu, F.; Suakar, O.; Kuskucu, A.; Bayrak, O.; Arcaro, K. Effects of two types of energy restriction on methylation levels of adiponectin receptor 1 and leptin receptor overlapping transcript in a mouse mammary tumour virus-transforming growth factor-α breast cancer mouse model. Br. J. Nutr. 2021, 125, 1–9. [Google Scholar]
- Zhao, C.; Wu, M.; Zeng, N.; Xiong, M.; Hu, W.; Lv, W.; Yi, Y.; Zhang, Q.; Wu, Y. Cancer-associated adipocytes: Emerging supporters in breast cancer. J. Exp. Clin. Cancer Res. 2020, 39, 156. [Google Scholar]
- Zatterale, F.; Longo, M.; Naderi, J.; Raciti, G.A.; Desiderio, A.; Miele, C.; Beguinot, F. Chronic adipose tissue inflammation linking obesity to insulin resistance and type 2 diabetes. Front. Physiol. 2020, 10, 1607. [Google Scholar] [CrossRef]
- Lumeng, C.N.; Bodzin, J.L.; Saltiel, A.R. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Investig. 2007, 117, 175–184. [Google Scholar]
- Kwiecień, I.; Polubiec-Kownacka, M.; Dziedzic, D.; Wołosz, D.; Rzepecki, P.; Domagała-Kulawik, J. CD163 and CCR7 as markers for macrophage polarisation in lung cancer microenvironment. Cent. Eur. J. Immunol. 2019, 44, 395–402. [Google Scholar]
- Neumeier, M.; Weigert, J.; Schäffler, A.; Wehrwein, G.; Müller-Ladner, U.; Schölmerich, J.; Wrede, C.; Buechler, C. Different effects of adiponectin isoforms in human monocytic cells. J. Leukoc. Biol. 2006, 79, 803–808. [Google Scholar]
- Chinetti, G.; Zawadski, C.; Fruchart, J.-C.; Staels, B. Expression of adiponectin receptors in human macrophages and regulation by agonists of the nuclear receptors PPARα, PPARγ, and LXR. Biochem. Biophys. Res. Commun. 2004, 314, 151–158. [Google Scholar]
- Van Stijn, C.M.; Kim, J.; Lusis, A.J.; Barish, G.D.; Tangirala, R.K. Macrophage polarization phenotype regulates adiponectin receptor expression and adiponectin anti-inflammatory response. FASEB J. 2015, 29, 636. [Google Scholar]
- Otvos Jr, L.; Kovalszky, I.; Olah, J.; Coroniti, R.; Knappe, D.; Nollmann, F.I.; Hoffmann, R.; Wade, J.D.; Lovas, S.; Surmacz, E. Optimization of adiponectin-derived peptides for inhibition of cancer cell growth and signaling. Pept. Sci. 2015, 104, 156–166. [Google Scholar]
- Otvos, L.; Haspinger, E.; La Russa, F.; Maspero, F.; Graziano, P.; Kovalszky, I.; Lovas, S.; Nama, K.; Hoffmann, R.; Knappe, D. Design and development of a peptide-based adiponectin receptor agonist for cancer treatment. BMC Biotechnol. 2011, 11, 90. [Google Scholar]
- Kim, S.; Lee, Y.; Kim, J.W.; Son, Y.-J.; Ma, M.J.; Um, J.-H.; Kim, N.D.; Min, S.H.; Kim, D.I.; Kim, B.B. Discovery of a novel potent peptide agonist to adiponectin receptor 1. PLoS ONE 2018, 13, e0199256. [Google Scholar]
- Okada-Iwabu, M.; Yamauchi, T.; Iwabu, M.; Honma, T.; Hamagami, K.-i.; Matsuda, K.; Yamaguchi, M.; Tanabe, H.; Kimura-Someya, T.; Shirouzu, M. A small-molecule AdipoR agonist for type 2 diabetes and short life in obesity. Nature 2013, 503, 493–499. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).